COVID-19 and Radiological Progression of Multiple Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Study Cohort
2.3. Variables and Outcomes
2.4. MRI Scoring
2.5. Analysis and Statistics
3. Results
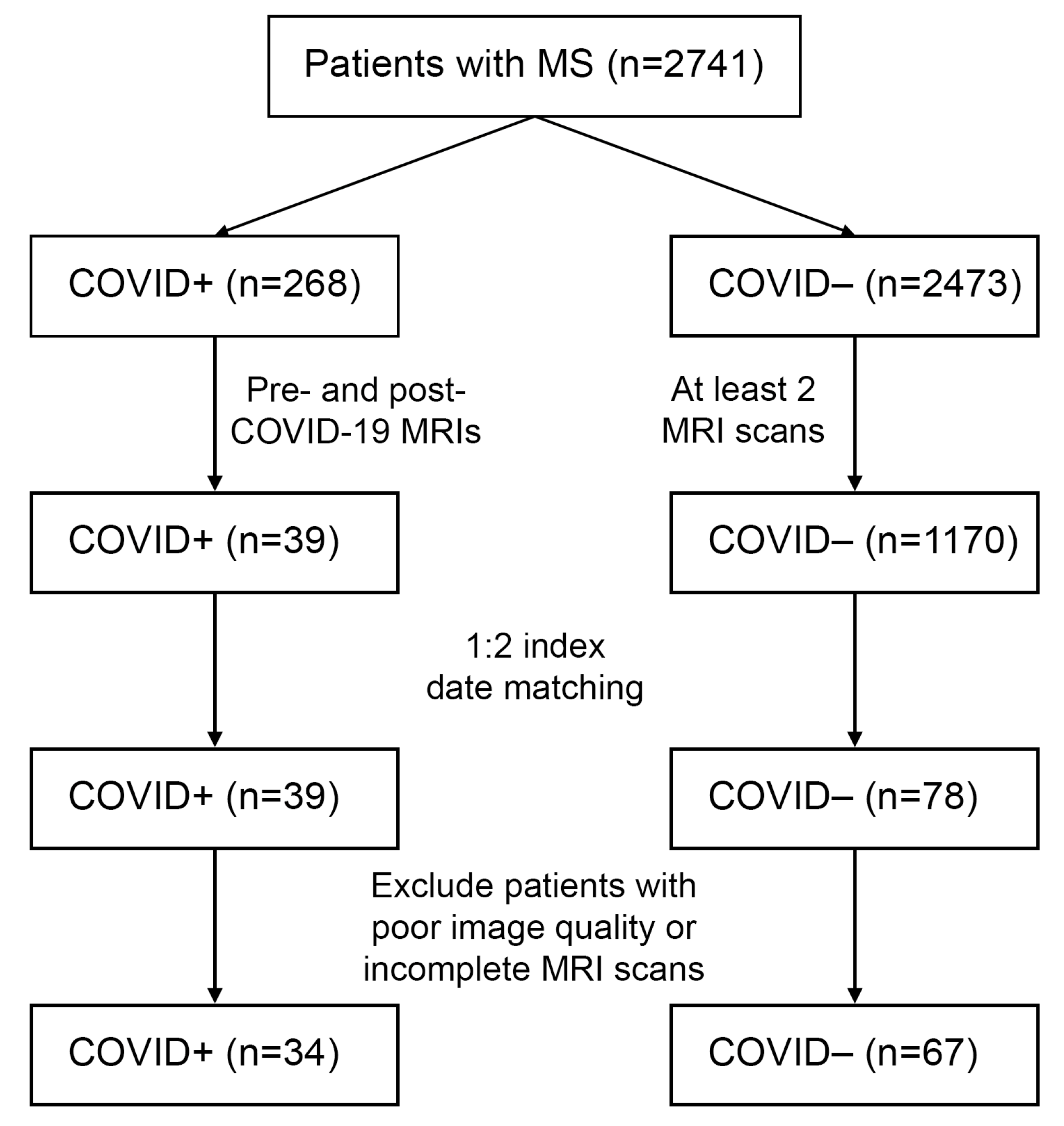
3.1. Cohort Selection
3.2. Baseline Characteristics
3.3. Radiological Outcomes
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviation
| MS | Multiple sclerosis |
References
- Filippi, M.; Bar-Or, A.; Piehl, F.; Preziosa, P.; Solari, A.; Vukusic, S.; Rocca, M.A. Multiple sclerosis. Nat. Rev. Dis. Primers 2018, 4, 43. [Google Scholar] [CrossRef]
- Correale, J.; Fiol, M.; Gilmore, W. The risk of relapses in multiple sclerosis during systemic infections. Neurology 2006, 67, 652–659. [Google Scholar] [CrossRef]
- Simpson-Yap, S.; Pirmani, A.; Kalincik, T.; De Brouwer, E.; Geys, L.; Parciak, T.; Helme, A.; Rijke, N.; Hillert, J.A.; Moreau, Y.; et al. Updated Results of the COVID-19 in MS Global Data Sharing Initiative: Anti-CD20 and Other Risk Factors Associated with COVID-19 Severity. Neurol. Neuroimmunol. Neuroinflamm. 2022, 9, e200021. [Google Scholar] [CrossRef]
- Colais, P.; Cascini, S.; Balducci, M.; Agabiti, N.; Davoli, M.; Fusco, D.; Calandrini, E.; Bargagli, A.M. Impact of the COVID-19 pandemic on access to healthcare services amongst patients with multiple sclerosis in the Lazio region, Italy. Eur. J. Neurol. 2021, 28, 3403–3410. [Google Scholar] [CrossRef]
- Salter, A.; Fox, R.J.; Newsome, S.D.; Halper, J.; Li, D.K.B.; Kanellis, P.; Costello, K.; Bebo, B.; Rammohan, K.; Cutter, G.R.; et al. Outcomes and Risk Factors Associated with SARS-CoV-2 Infection in a North American Registry of Patients with Multiple Sclerosis. JAMA Neurol. 2021, 78, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Portaccio, E.; Fonderico, M.; Hemmer, B.; Derfuss, T.; Stankoff, B.; Selmaj, K.; Tintore, M.; Amato, M.P. Impact of COVID-19 on multiple sclerosis care and management: Results from the European Committee for Treatment and Research in Multiple Sclerosis survey. Mult. Scler. 2022, 28, 132–138. [Google Scholar] [CrossRef]
- Pilotto, A.; Masciocchi, S.; Volonghi, I.; De Giuli, V.; Caprioli, F.; Mariotto, S.; Ferrari, S.; Bozzetti, S.; Imarisio, A.; Risi, B.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Encephalitis Is a Cytokine Release Syndrome: Evidences from Cerebrospinal Fluid Analyses. Clin. Infect. Dis. 2021, 73, e3019–e3026. [Google Scholar] [CrossRef]
- Savelieff, M.G.; Feldman, E.L.; Stino, A.M. Neurological sequela and disruption of neuron-glia homeostasis in SARS-CoV-2 infection. Neurobiol. Dis. 2022, 168, 105715. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhao, H.; Fang, D.; Davis, C.T.; Shi, D.S.; Lei, K.; Rich, B.E.; Winter, J.M.; Guo, L.; Sorensen, L.K.; et al. Neuroinflammatory disease disrupts the blood-CNS barrier via crosstalk between proinflammatory and endothelial-to-mesenchymal-transition signaling. Neuron 2022, 110, 3106–3120.e7. [Google Scholar] [CrossRef]
- Hauptmann, J.; Johann, L.; Marini, F.; Kitic, M.; Colombo, E.; Mufazalov, I.A.; Krueger, M.; Karram, K.; Moos, S.; Wanke, F.; et al. Interleukin-1 promotes autoimmune neuroinflammation by suppressing endothelial heme oxygenase-1 at the blood-brain barrier. Acta Neuropathol. 2020, 140, 549–567. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.H.; Kim, C.Y.; Lee, E.; Lee, C.; Lee, K.S.; Lee, J.; Park, H.; Choi, B.; Hwang, I.; Kim, J.; et al. Microglial NLRP3-gasdermin D activation impairs blood-brain barrier integrity through interleukin-1beta-independent neutrophil chemotaxis upon peripheral inflammation in mice. Nat. Commun. 2025, 16, 699. [Google Scholar] [CrossRef] [PubMed]
- Lake, C.M.; Breen, J.J. Sequence similarity between SARS-CoV-2 nucleocapsid and multiple sclerosis-associated proteins provides insight into viral neuropathogenesis following infection. Sci. Rep. 2023, 13, 389. [Google Scholar] [CrossRef]
- MacDougall, M.; El-Hajj Sleiman, J.; Beauchemin, P.; Rangachari, M. SARS-CoV-2 and Multiple Sclerosis: Potential for Disease Exacerbation. Front. Immunol. 2022, 13, 871276. [Google Scholar] [CrossRef] [PubMed]
- Weissert, R. Nervous system-related tropism of SARS-CoV-2 and autoimmunity in COVID-19 infection. Eur. J. Immunol. 2024, 54, e2250230. [Google Scholar] [CrossRef]
- Albornoz, E.A.; Amarilla, A.A.; Modhiran, N.; Parker, S.; Li, X.X.; Wijesundara, D.K.; Aguado, J.; Zamora, A.P.; McMillan, C.L.D.; Liang, B.; et al. SARS-CoV-2 drives NLRP3 inflammasome activation in human microglia through spike protein. Mol. Psychiatry 2023, 28, 2878–2893. [Google Scholar] [CrossRef] [PubMed]
- Etemadifar, M.; Abhari, A.P.; Nouri, H.; Salari, M.; Maleki, S.; Amin, A.; Sedaghat, N. Does COVID-19 increase the long-term relapsing-remitting multiple sclerosis clinical activity? A cohort study. BMC Neurol. 2022, 22, 64. [Google Scholar] [CrossRef]
- Montini, F.; Nozzolillo, A.; Tedone, N.; Mistri, D.; Rancoita, P.M.; Zanetta, C.; Mandelli, A.; Furlan, R.; Moiola, L.; Martinelli, V.; et al. COVID-19 has no impact on disease activity, progression and cognitive performance in people with multiple sclerosis: A 2-year study. J. Neurol. Neurosurg. Psychiatry 2024, 95, 342–347. [Google Scholar] [CrossRef]
- Salter, A.; Lancia, S.; Cutter, G.R.; Fox, R.J.; Marrie, R.A. Effects of COVID-19 Infection on Symptom Severity and Disability in Multiple Sclerosis. Neurology 2025, 104, e210149. [Google Scholar] [CrossRef]
- Rahmani, M.; Moghadasi, A.N.; Shahi, S.; Eskandarieh, S.; Azizi, H.; Hasanzadeh, A.; Ahmadzade, A.; Dehnavi, A.Z.; Farahani, R.H.; Aminianfar, M.; et al. COVID-19 and its implications on the clinico-radiological course of multiple sclerosis: A case-control study. Med. Clin. 2023, 160, 187–192. [Google Scholar] [CrossRef]
- Hadidchi, R.; Wang, S.H.; Rezko, D.; Henry, S.; Coyle, P.K.; Duong, T.Q. SARS-CoV-2 infection increases long-term multiple sclerosis disease activity and all-cause mortality in an underserved inner-city population. Mult. Scler. Relat. Disord. 2024, 86, 105613. [Google Scholar] [CrossRef]
- Rebsamen, M.; Friedli, C.; Radojewski, P.; Diem, L.; Chan, A.; Wiest, R.; Salmen, A.; Rummel, C.; Hoepner, R. Multiple sclerosis as a model to investigate SARS-CoV-2 effect on brain atrophy. CNS Neurosci. Ther. 2023, 29, 538–543. [Google Scholar] [CrossRef]
- Pakan, R.; Hadidchi, R.; Al-Ani, Y.; Piskun, H.; Duong, K.S.; Henry, S.; Wang, S.; Maurer, C.W.; Duong, T.Q. Long-Term Outcomes of Patients with Pre-Existing Essential Tremor After SARS-CoV-2 Infection. Diagnostics 2024, 14, 2774. [Google Scholar] [CrossRef]
- Lu, J.Q.; Lu, J.Y.; Wang, W.; Liu, Y.; Buczek, A.; Fleysher, R.; Hoogenboom, W.S.; Zhu, W.; Hou, W.; Rodriguez, C.J.; et al. Clinical predictors of acute cardiac injury and normalization of troponin after hospital discharge from COVID-19. eBioMedicine 2022, 76, 103821. [Google Scholar] [CrossRef]
- Lu, J.Y.; Boparai, M.S.; Shi, C.; Henninger, E.M.; Rangareddy, M.; Veeraraghavan, S.; Mirhaji, P.; Fisher, M.C.; Duong, T.Q. Long-term outcomes of COVID-19 survivors with hospital AKI: Association with time to recovery from AKI. Nephrol. Dial. Transplant. 2023, 38, 2160–2169. [Google Scholar] [CrossRef]
- Lu, J.Y.; Buczek, A.; Fleysher, R.; Hoogenboom, W.S.; Hou, W.; Rodriguez, C.J.; Fisher, M.C.; Duong, T.Q. Outcomes of Hospitalized Patients with COVID-19 with Acute Kidney Injury and Acute Cardiac Injury. Front. Cardiovasc. Med. 2021, 8, 798897. [Google Scholar] [CrossRef]
- Lu, J.Y.; Buczek, A.; Fleysher, R.; Musheyev, B.; Henninger, E.M.; Jabbery, K.; Rangareddy, M.; Kanawade, D.; Nelapat, C.; Soby, S.; et al. Characteristics of COVID-19 patients with multiorgan injury across the pandemic in a large academic health system in the Bronx, New York. Heliyon 2023, 9, e15277. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.Y.; Wilson, J.; Hou, W.; Fleysher, R.; Herold, B.C.; Herold, K.C.; Duong, T.Q. Incidence of new-onset in-hospital and persistent diabetes in COVID-19 patients: Comparison with influenza. eBioMedicine 2023, 90, 104487. [Google Scholar] [CrossRef]
- Zhang, V.; Fisher, M.; Hou, W.; Zhang, L.; Duong, T.Q. Incidence of New-Onset Hypertension Post-COVID-19: Comparison with Influenza. Hypertension 2023, 80, 2135–2148. [Google Scholar] [CrossRef]
- Eligulashvili, A.; Darrell, M.; Gordon, M.; Jerome, W.; Fiori, K.P.; Congdon, S.; Duong, T.Q. Patients with unmet social needs are at higher risks of developing severe long COVID-19 symptoms and neuropsychiatric sequela. Sci. Rep. 2024, 14, 7743. [Google Scholar] [CrossRef] [PubMed]
- Eligulashvili, A.; Darrell, M.; Miller, C.; Lee, J.; Congdon, S.; Lee, J.S.; Hsu, K.; Yee, J.; Hou, W.; Islam, M.; et al. COVID-19 Patients in the COVID-19 Recovery and Engagement (CORE) Clinics in the Bronx. Diagnostics 2022, 13, 119. [Google Scholar] [CrossRef] [PubMed]
- Eligulashvili, A.; Gordon, M.; Lee, J.S.; Lee, J.; Mehrotra-Varma, S.; Mehrotra-Varma, J.; Hsu, K.; Hilliard, I.; Lee, K.; Li, A.; et al. Long-term outcomes of hospitalized patients with SARS-CoV-2/COVID-19 with and without neurological involvement: 3-year follow-up assessment. PLoS Med. 2024, 21, e1004263. [Google Scholar] [CrossRef]
- Hadidchi, R.; Al-Ani, Y.; Choi, S.; Renteria, S.; Duong, K.S.; Henry, S.; Wang, S.H.; Duong, T.Q. Long-term outcomes of patients with a pre-existing neurological condition after SARS-CoV-2 infection. J. Neurol. Sci. 2025, 473, 123477. [Google Scholar] [CrossRef]
- Hadidchi, R.; Al-Ani, Y.; Piskun, H.; Pakan, R.; Duong, K.; Jamil, H.; Wang, S.; Henry, S.; Maurer, C.; Duong, T. Long-term outcomes of patients with Parkinson’s disease 3.5 years post SARS-CoV-2 infection in an inner-city population in the Bronx. Res. Sq. 2024. [Google Scholar] [CrossRef]
- Hadidchi, R.; Al-Ani, Y.; Piskun, H.; Pakan, R.; Duong, K.S.; Jamil, H.; Wang, S.H.; Henry, S.; Maurer, C.W.; Duong, T.Q. Impact of COVID-19 on long-term outcomes in Parkinson’s disease. Eur. J. Neurol. 2025, 32, e70013. [Google Scholar] [CrossRef]
- Hadidchi, R.; Pakan, R.; Alamuri, T.; Cercizi, N.; Al-Ani, Y.; Wang, S.H.; Henry, S.; Duong, T.Q. Long COVID-19 outcomes of patients with pre-existing dementia. J. Alzheimers Dis. 2024, 103, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Hadidchi, R.; Vichare, A.; Lu, J.Y.; Hou, W.; Henry, S.; Akalin, E.; Duong, T.Q. SARS-CoV-2 Infection Is Associated with an Accelerated eGFR Decline in Kidney Transplant Recipients up to Four Years Post Infection. Diagnostics 2025, 15, 1091. [Google Scholar] [CrossRef] [PubMed]
- Filippi, M.; Preziosa, P.; Barkhof, F.; Chard, D.T.; De Stefano, N.; Fox, R.J.; Gasperini, C.; Kappos, L.; Montalban, X.; Moraal, B.; et al. Diagnosis of Progressive Multiple Sclerosis From the Imaging Perspective: A Review. JAMA Neurol. 2021, 78, 351–364. [Google Scholar] [CrossRef]
- Devogelaere, J.; Van Remoortel, A.; Van Laethem, D.; Faille, L.D.; D’Hooghe, M.B.; Van Schependom, J.; Stastna, D.; Uher, T.; Nagels, G.; D’Haeseleer, M. Impact of brain volume on coronavirus disease of 2019 severity and subsequent cognitive decline in patients with multiple sclerosis. J. Neurol. Sci. 2025, 475, 123552. [Google Scholar] [CrossRef]
- Bsteh, G.; Assar, H.; Gradl, C.; Heschl, B.; Hiller, M.S.; Krajnc, N.; Di Pauli, F.; Hegen, H.; Traxler, G.; Leutmezer, F.; et al. Long-term outcome after COVID-19 infection in multiple sclerosis: A nation-wide multicenter matched-control study. Eur. J. Neurol. 2022, 29, 3050–3060. [Google Scholar] [CrossRef] [PubMed]
- Amezcua, L.; Rivera, V.M.; Vazquez, T.C.; Baezconde-Garbanati, L.; Langer-Gould, A. Health Disparities, Inequities, and Social Determinants of Health in Multiple Sclerosis and Related Disorders in the US: A Review. JAMA Neurol. 2021, 78, 1515–1524. [Google Scholar] [CrossRef]
- Tranfa, M.; Tortora, M.; Pontillo, G.; Iuzzolino, V.; Riccio, E.; Caccavallo, S.; Di Risi, T.; Monti, S.; Lanzillo, R.; Brescia Morra, V.; et al. The central vein sign helps in differentiating multiple sclerosis from its mimickers: Lessons from Fabry disease. Eur. Radiol. 2022, 32, 3846–3854. [Google Scholar] [CrossRef]
- Somma, F.; Negro, A.; D’Agostino, V.; Piscitelli, V.; Pace, G.; Tortora, M.; Tortora, F.; Gatta, G.; Caranci, F. COVID-19 and low back pain: Previous infections lengthen recovery time after intradiscal ozone therapy in patients with herniated lumbar disc. Radiol. Med. 2022, 127, 673–680. [Google Scholar] [CrossRef]
- Aloisi, F.; Giovannoni, G.; Salvetti, M. Epstein-Barr virus as a cause of multiple sclerosis: Opportunities for prevention and therapy. Lancet Neurol. 2023, 22, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Barzegar, M.; Vaheb, S.; Mirmosayyeb, O.; Afshari-Safavi, A.; Nehzat, N.; Shaygannejad, V. Can coronavirus disease 2019 (COVID-19) trigger exacerbation of multiple sclerosis? A retrospective study. Mult. Scler. Relat. Disord. 2021, 52, 102947. [Google Scholar] [CrossRef]
- Garjani, A.; Middleton, R.M.; Hunter, R.; Tuite-Dalton, K.A.; Coles, A.; Dobson, R.; Duddy, M.; Hughes, S.; Pearson, O.R.; Rog, D.; et al. COVID-19 is associated with new symptoms of multiple sclerosis that are prevented by disease modifying therapies. Mult. Scler. Relat. Disord. 2021, 52, 102939. [Google Scholar] [CrossRef] [PubMed]
- Lotan, I.; Nishiyama, S.; Manzano, G.S.; Lydston, M.; Levy, M. COVID-19 and the risk of CNS demyelinating diseases: A systematic review. Front. Neurol. 2022, 13, 970383. [Google Scholar] [CrossRef]
- Tavazzi, E.; Pichiecchio, A.; Colombo, E.; Rigoni, E.; Asteggiano, C.; Vegezzi, E.; Masi, F.; Greco, G.; Bastianello, S.; Bergamaschi, R. The Potential Role of SARS-CoV-2 Infection and Vaccines in Multiple Sclerosis Onset and Reactivation: A Case Series and Literature Review. Viruses 2023, 15, 1569. [Google Scholar] [CrossRef]
- Jagst, M.; Pottkamper, L.; Gomer, A.; Pitarokoili, K.; Steinmann, E. Neuroinvasion and neurotropism of severe acute respiratory syndrome coronavirus 2 infection. Curr. Opin. Microbiol. 2024, 79, 102474. [Google Scholar] [CrossRef] [PubMed]
- Andrews, H.S.; Herman, J.D.; Gandhi, R.T. Treatments for COVID-19. Annu. Rev. Med. 2024, 75, 145–157. [Google Scholar] [CrossRef]


| Characteristics | COVID+ (n = 34) | COVID− (n = 67) |
|---|---|---|
| Age at index date (years), mean ± SD | 47.50 ± 14.66 | 47.52 ± 12.77 |
| Years between earliest and latest MRI, mean ± SD | 2.20 ± 0.96 | 2.28 ± 1.11 |
| Years between index date and latest MRI, mean ± SD | 1.78 ± 0.98 | 1.67 ± 1.02 |
| Years between MS diagnosis and index date, mean ± SD | 3.34 ± 1.87 | 4.35 ± 1.80 ** |
| Female, n (%) | 27 (79.41%) | 57 (85.07%) |
| Race and Ethnicity, n (%) | ||
| Non-Hispanic White | 7 (20.59%) | 5 (7.46%) |
| Black | 15 (44.12%) | 31 (46.27%) |
| Asian | 0 (0.00%) | 2 (2.99%) |
| Hispanic | 13 (38.24%) | 28 (41.79%) |
| Other race | 12 (35.29%) | 29 (43.28%) |
| Vaccinated for SARS-CoV-2 at index date, n (%) | 21 (61.76%) | 44 (65.67%) |
| Hospitalized due to COVID-19, n (%) | 16 (47.06%) | − |
| Comorbidities, n (%) | ||
| Hypertension | 13 (38.24%) | 32 (47.76%) |
| Type-2 diabetes | 8 (23.53%) | 14 (20.90%) |
| COPD | 0 (0.00%) | 1 (1.49%) |
| Cardiovascular disease | 5 (14.71%) | 11 (16.42%) |
| Chronic kidney disease | 4 (11.76%) | 9 (13.43%) |
| Liver disease | 7 (20.59%) | 8 (11.94%) |
| Tobacco use | 17 (50.00%) | 41 (61.19%) |
| Obesity | 20 (58.82%) | 45 (67.16%) |
| Cerebral infarction | 4 (11.76%) | 5 (7.46%) |
| Intracranial hemorrhage | 0 (0.00%) | 0 (0.00%) |
| Traumatic brain injury | 0 (0.00%) | 1 (1.49%) |
| Intracranial cancer | 0 (0.00%) | 0 (0.00%) |
| Outcome | COVID+ (n = 34) | COVID− (n = 67) | p-Value | OR [95% CI] | aOR [95% CI] |
|---|---|---|---|---|---|
| Enlargement of existing lesions | 1 (2.94%) | 4 (5.97%) | 0.86 | 0.46 [0.05, 4.31] | 0.38 [0.04, 4.00] |
| New lesions | 3 (8.82%) | 6 (8.96%) | 1.00 | 0.95 [0.22, 4.07] | 0.79 [0.17, 3.73] |
| Enhancing lesions | 2 (5.88%) | 1 (1.49%) | 0.54 | 4.00 [0.35, 45.79] | − |
| Brain volume loss | 12 (35.29%) | 32 (47.76%) | 0.33 | 0.60 [0.25, 1.41] | 0.43 [0.14, 1.29] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nguyen, H.Q.; Hadidchi, R.; Eligulashvili, A.; Nandi, S.; Vichare, A.; Patel, B.; Sanchez, J.; Bisulca, J.; Henry, S.; Lee, J.S.; et al. COVID-19 and Radiological Progression of Multiple Sclerosis. Diagnostics 2026, 16, 1513. https://doi.org/10.3390/diagnostics16101513
Nguyen HQ, Hadidchi R, Eligulashvili A, Nandi S, Vichare A, Patel B, Sanchez J, Bisulca J, Henry S, Lee JS, et al. COVID-19 and Radiological Progression of Multiple Sclerosis. Diagnostics. 2026; 16(10):1513. https://doi.org/10.3390/diagnostics16101513
Chicago/Turabian StyleNguyen, Hien Quang, Roham Hadidchi, Anna Eligulashvili, Shounak Nandi, Aditi Vichare, Bhakti Patel, Jimmy Sanchez, Joseph Bisulca, Sonya Henry, Jimmy S. Lee, and et al. 2026. "COVID-19 and Radiological Progression of Multiple Sclerosis" Diagnostics 16, no. 10: 1513. https://doi.org/10.3390/diagnostics16101513
APA StyleNguyen, H. Q., Hadidchi, R., Eligulashvili, A., Nandi, S., Vichare, A., Patel, B., Sanchez, J., Bisulca, J., Henry, S., Lee, J. S., & Duong, T. Q. (2026). COVID-19 and Radiological Progression of Multiple Sclerosis. Diagnostics, 16(10), 1513. https://doi.org/10.3390/diagnostics16101513







