Point-of-Care Ultrasound Use in Cardiac Arrest Patients
Abstract


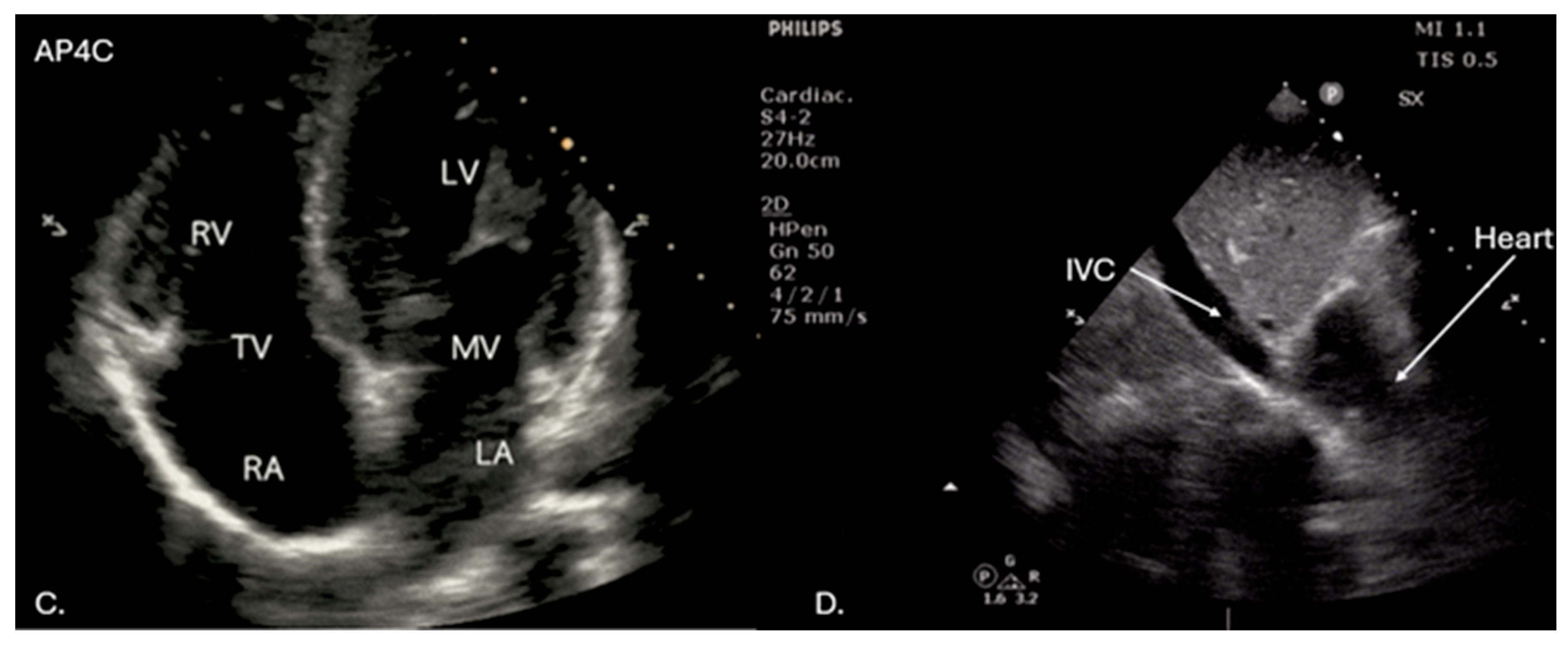

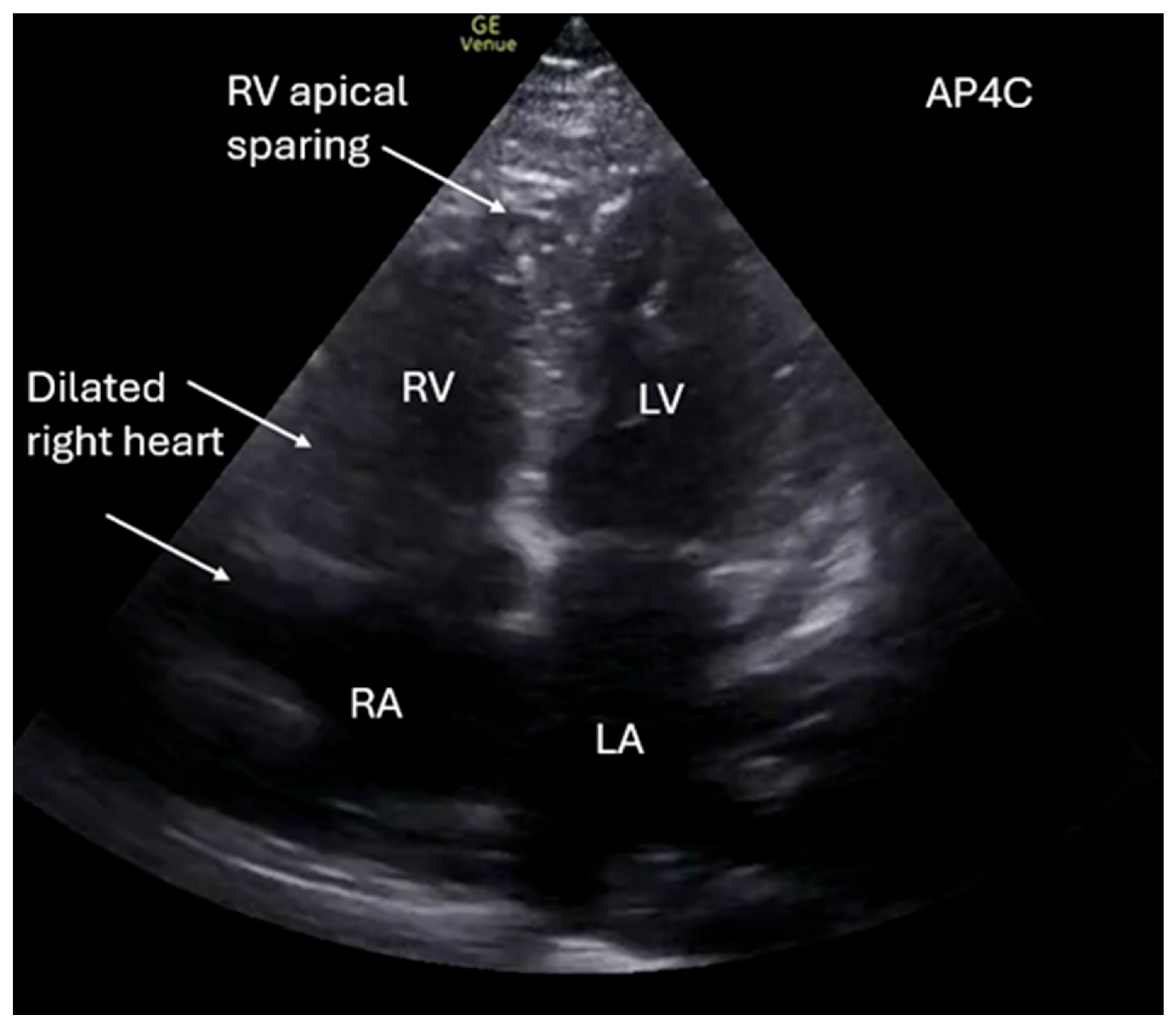

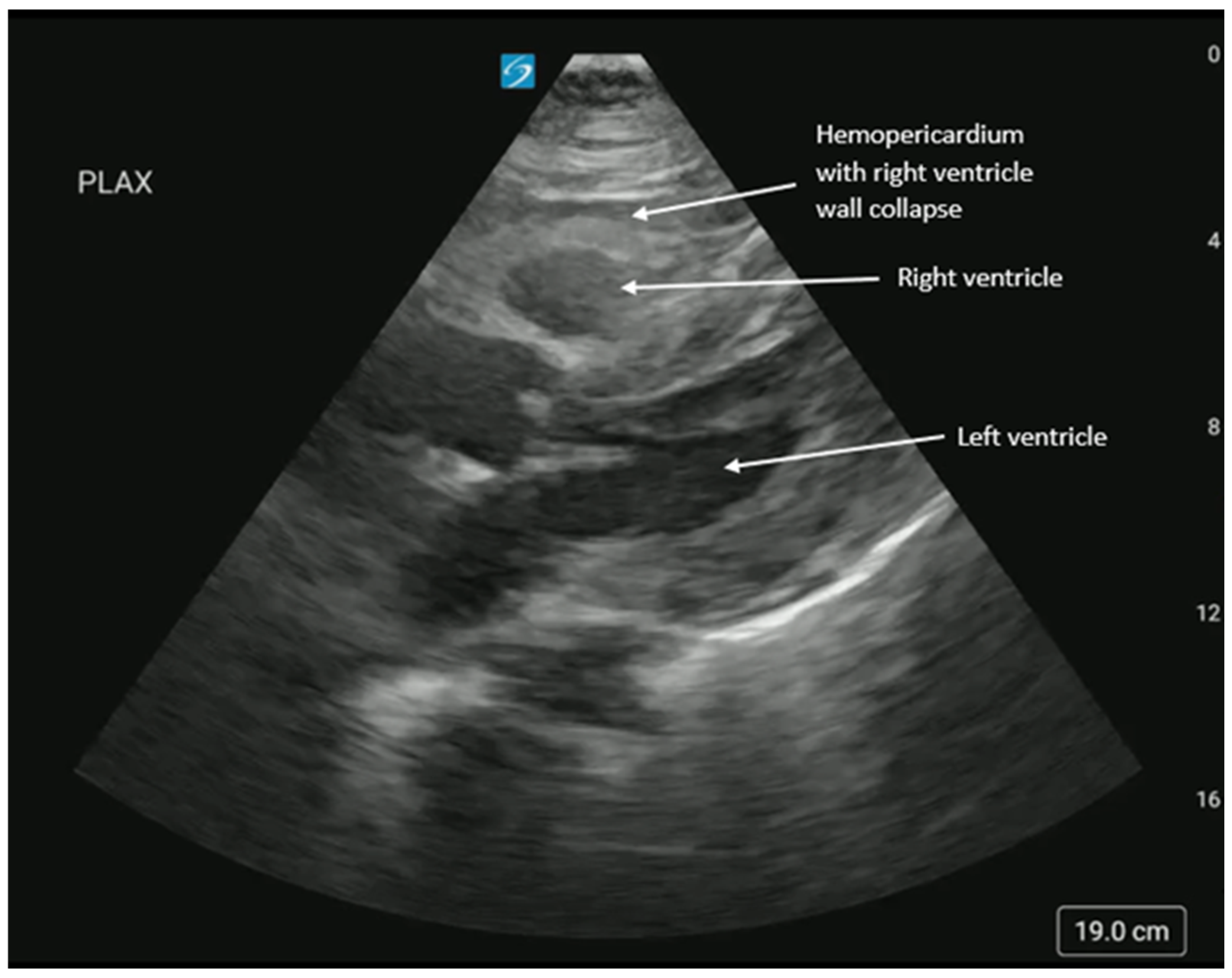

Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| POCUS | Point-of-care ultrasound |
| VF | Ventricular fibrillation |
| ED | Emergency department |
| PEA | Pulseless electrical activity |
| RV | Right ventricle |
| LV | Left ventricle |
| PE | Pulmonary embolism |
| ROSC | Return of spontaneous circulation |
| CPR | Cardiopulmonary resuscitation |
References
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Anderson, C.A.; Arora, P.; Avery, C.L.; Baker-Smith, C.M.; Beaton, A.Z.; Boehme, A.K.; Buxton, A.E.; et al. Heart disease and stroke statistics—2023 update: A report from the American Heart Association. Circulation 2023, 147, e93–e621. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, R.; Weekes, A.; Adhikari, S.; Noble, V.E.; Nomura, J.T.; Theodoro, D.; Woo, M.; Atkinson, P.; Blehar, D.; Brown, S.M.; et al. Emergency department point-of-care ultrasound in out-of-hospital and in-ED cardiac arrest. Resuscitation 2016, 109, 33–39. [Google Scholar] [CrossRef]
- Salen, P.; Melniker, L.; Chooljian, C.; Rose, J.S.; Alteveer, J.; Reed, J.; Heller, M. Does the presence or absence of sonographically identified cardiac activity predict resuscitation outcomes of cardiac arrest patients? Am. J. Emerg. Med. 2005, 23, 459–462. [Google Scholar] [CrossRef]
- Gottlieb, M.; Sundaram, T.; Olszynski, P.; Atkinson, P. Just the facts: Point-of-care ultrasound in cardiac arrest. Can. J. Emerg. Med. 2022, 24, 579–581. [Google Scholar] [CrossRef]
- Atkinson, P.; Bowra, J.; Milne, J.; Lewis, D.; Lambert, M.; Jarman, B.; Noble, V.E.; Lamprecht, H.; Harris, T.; Connolly, J.; et al. International Federation for Emergency Medicine Consensus Statement: Sonography in hypotension and cardiac arrest (SHoC): An international consensus on the use of point of care ultrasound for undifferentiated hypotension and during cardiac arrest. Can. J. Emerg. Med. 2017, 19, 459–470. [Google Scholar] [CrossRef]
- Clattenburg, E.J.; Wroe, P.C.; Gardner, K.; Schultz, C.; Gelber, J.; Singh, A.; Nagdev, A. Implementation of the Cardiac Arrest Sonographic Assessment (CASA) protocol for patients with cardiac arrest is associated with shorter CPR pulse checks. Resuscitation 2018, 131, 69–73. [Google Scholar] [CrossRef]
- Gardner, K.F.; Clattenburg, E.J.; Wroe, P.; Singh, A.; Mantuani, D.; Nagdev, A. The Cardiac Arrest Sonographic Assessment (CASA) exam—A standardized approach to the use of ultrasound in PEA. Am. J. Emerg. Med. 2018, 36, 729–731. [Google Scholar] [CrossRef]
- Blaivas, M.; Fox, J.C. Outcome in Cardiac Arrest Patients Found to Have Cardiac Standstill on the Bedside Emergency Department Echocardiogram. Acad. Emerg. Med. 2001, 8, 616–621. [Google Scholar] [CrossRef]
- Tayal, V.S.; Kline, J.A. Emergency echocardiography to detect pericardial effusion in patients in PEA and near-PEA states. Resuscitation 2003, 59, 315–318. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, R.; Weekes, A.; Adhikari, S.; Nobled, V.; Nomurae, J.T.; Woog, M.; Atkinsonh, P.; Blehara, D.; Brown, S.; Caffery, T.; et al. A retrospective study of pulseless electrical activity, bedside ultrasound identifies interventions during resuscitation associated with improved survival to hospital admission. A REASON Study. Resuscitation 2017, 120, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Gaspari, R.; Lindsay, R.; She, T.; Acuna, J.; Balk, A.; Bartnik, J.; Baxter, J.; Clare, D.; Caplan, R.J.; DeAngelis, J.; et al. Incidence and clinical relevance of echocardiography visualization of occult ventricular fibrillation: A Multicenter Prospective Study of Patients Presenting to the Emergency Department After Out-of-Hospital Cardiac Arrest. Ann. Emerg. Med. 2025, 86, 328–336. [Google Scholar] [PubMed]
- Nguyen, C.; Parfianowicz, D.; Bennett, C. Point of care ultrasound and shock: The value in bedside diagnosis and hemodynamic assessment in undifferentiated shock patients. J. Transl. Crit. Care Med. 2024, 6, e24-00010. [Google Scholar] [CrossRef]
- Seif, D.; Perera, P.; Mailhot, T.; Riley, D.; Mandavia, D. Bedside ultrasound in resuscitation and the rapid ultrasound in shock protocol. Crit. Care Res. Pract. 2012, 2012, 503254. [Google Scholar] [CrossRef]
- Magon, F.; Longhitano, Y.; Savioli, G.; Piccioni, A.; Tesauro, M.; Del Duca, F.; Napoletano, G.; Volonnino, G.; Maiese, A.; La Russa, R.; et al. Point-of-Care Ultrasound (POCUS) in Adult Cardiac Arrest: Clinical Review. Diagnostics 2024, 14, 434. [Google Scholar] [CrossRef]
- Popat, A.; Harikrishnan, S.; Seby, N.; Sen, U.; Patel, S.K.; Mittal, L.; Patel, M.; Vundi, C.; Patel, Y.; Babita Kumar, A.; et al. Utilization of Point-of-Care Ultrasound as an Imaging Modality in the Emergency Department: A Systematic Review and Meta-Analysis. Cureus 2024, 16, e52371. [Google Scholar] [CrossRef] [PubMed]
- American College of Emergency Physicians Policy Statement. Ultrasound Guidelines: Emergency, Point-of-care, Clinical Ultrasound Guidelines in Medicine. Ann. Emerg. Med. 2023, 82, e115–e155. [Google Scholar] [CrossRef]
- Alerhand, S.; Adrian, R.J. What echocardiographic findings differentiate acute pulmonary embolism and chronic pulmonary hypertension? Am. J. Emerg. Med. 2023, 72, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Nathan, A.S.; Raman, S.; Yang, N.; Painter, I.; Khatana, S.A.M.; Dayoub, E.J.; Herrmann, H.C.; Yeh, R.W.; Groeneveld, P.W.; Doll, J.A.; et al. Association Between 90-Minute Door-to-Balloon Time, Selective Exclusion of Myocardial Infarction Cases, and Access Site Choice: Insights from the Cardiac Care Outcomes Assessment Program (COAP) in Washington State. Circ. Card. Interv. 2020, 13, e009179. [Google Scholar] [CrossRef]
- Nallamothu, B.K.; Normand, S.L.; Wang, Y.; Hofer, T.P.; Brush, J.E.; Messenger, J.C.; Bradley, E.H.; Rumsfeld, J.S.; Krumholz, H.M. Relation between door-to-balloon times and mortality after primary percutaneous coronary intervention over time: A retrospective study. Lancet 2015, 385, 1114–1122. [Google Scholar] [CrossRef]
- Lippmann, M.; Wiley, M.; Freeman, J.; Abicht, T.; Nath, J. Percutaneous Closure of a Traumatic Right Ventricular Pseudoaneurysm: A Novel Interventional Approach. CASE 2019, 3, 60–62. [Google Scholar] [CrossRef]
- Bayat, F.; Tehrani, R.B.; Khani, M.; Akbari, T.; Langroudi, T.F.; Taheri, M.; Alirezaei, A.; Bagheri, A.; Ebrahimi, P. Successful management of a delayed-presented and complicated right ventricular pseudoaneurysm: A case report and literature review. J. Cardiothorac. Surg. 2025, 20, 329. [Google Scholar] [CrossRef] [PubMed]
- Gaddipati, V.C.; Martin, A.I.; Valenzuela, M.O.; Mahmud, A.; Patel, A.A. An Incidentally Detected Right Ventricular Pseudoaneurysm. Case Rep. Cardiol. 2017, 2017, 4352474. [Google Scholar] [CrossRef] [PubMed]
- Alkhouli, M.; Waits, B.; Chaturvedi, A.; Ling, F.S. Percutaneous closure of right ventricular pseudoaneurysm. JACC Cardiovasc. Interv. 2015, 8, e147–e148. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Theophanous, A.G.; Angeles, N.; Elizondo, D.; Bronshteyn, Y.S.; Theophanous, R.G. Point-of-Care Ultrasound Use in Cardiac Arrest Patients. Diagnostics 2026, 16, 1514. https://doi.org/10.3390/diagnostics16101514
Theophanous AG, Angeles N, Elizondo D, Bronshteyn YS, Theophanous RG. Point-of-Care Ultrasound Use in Cardiac Arrest Patients. Diagnostics. 2026; 16(10):1514. https://doi.org/10.3390/diagnostics16101514
Chicago/Turabian StyleTheophanous, Andrew G., Nina Angeles, Denise Elizondo, Yuriy S. Bronshteyn, and Rebecca G. Theophanous. 2026. "Point-of-Care Ultrasound Use in Cardiac Arrest Patients" Diagnostics 16, no. 10: 1514. https://doi.org/10.3390/diagnostics16101514
APA StyleTheophanous, A. G., Angeles, N., Elizondo, D., Bronshteyn, Y. S., & Theophanous, R. G. (2026). Point-of-Care Ultrasound Use in Cardiac Arrest Patients. Diagnostics, 16(10), 1514. https://doi.org/10.3390/diagnostics16101514





