Hospital Coordination and Protocols Using Serum and Peripheral Blood Cells from Patients and Healthy Donors in a Longitudinal Study of Guillain–Barré Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. GBS Patients and Controls
2.2. Whole Blood Processing for PBMCs and Serum
2.3. Transcriptomics and Real Time RT-PCR of PBMCs
2.4. Proteomic Analysis of Human Serum
2.5. Biochemical Analysis in Human Serum
2.6. Phenotyping in Red Blood Cells
2.7. Statistical Analysis
3. Results
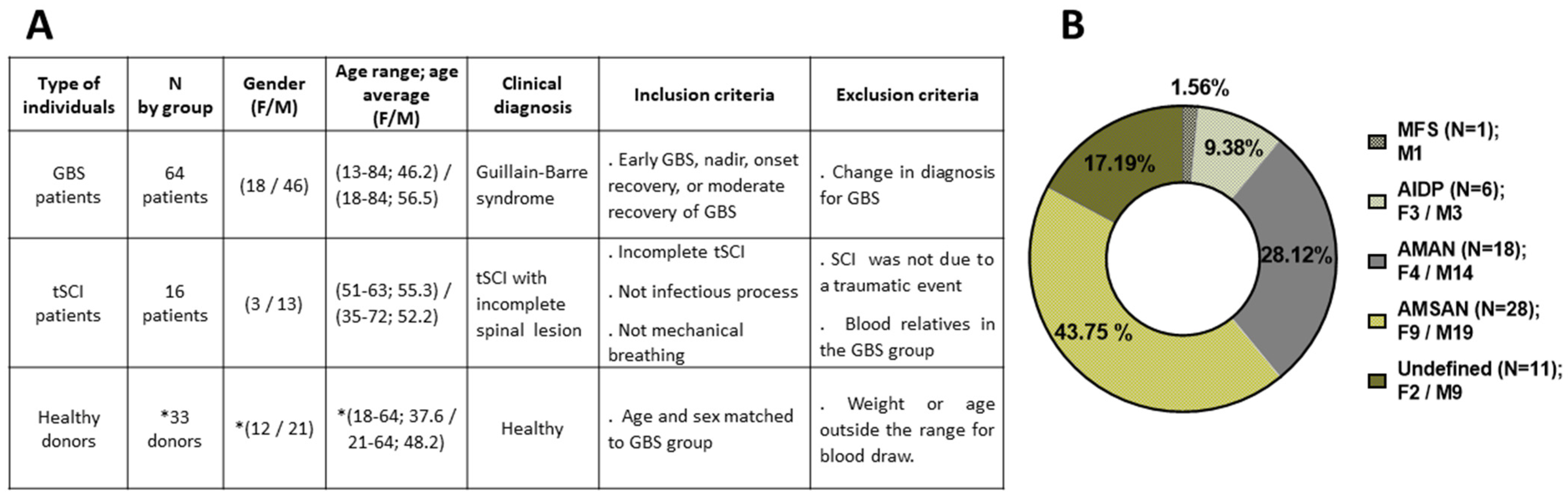
3.1. Recruitment of Participants in BioGBS Study and Identification of GBS Subtypes
3.2. Times Points During GBS Disease and Recovery for Collection of Serum and PBMCs
3.3. Quantitative Real-Time PCR in PBMCs, Serum Proteomic Analysis, and Serum Antibodies Searching from Samples in GBS Biobank Collection
3.4. Serum Biochemical Parameters in GBS Patients During Recovery
3.5. Phenotyping of Red Blood Cells in Donors of GBS Biobank Collection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, L.; Zhao, C.; Bao, Y.; Liu, Y.; Liang, Y.; Wei, J.; Liu, G.; Wang, J.; Zhan, S.; Wang, S.; et al. Variation in Worldwide Incidence of Guillain-Barré Syndrome: A Population-Based Study in Urban China and Existing Global Evidence. Front. Immunol. 2024, 15, 1415986. [Google Scholar] [CrossRef]
- Tikhomirova, A.; McNabb, E.R.; Petterlin, L.; Bellamy, G.L.; Lin, K.H.; Santoso, C.A.; Daye, E.S.; Alhaddad, F.M.; Lee, K.P.; Roujeinikova, A. Campylobacter Jejuni Virulence Factors: Update on Emerging Issues and Trends. J. Biomed. Sci. 2024, 31, 45. [Google Scholar] [CrossRef]
- Vidal, J.E.; Guedes, B.F.; Gomes, H.R.; Mendonça, R.H. Guillain-Barré Syndrome Spectrum as Manifestation of HIV-Related Immune Reconstitution Inflammatory Syndrome: Case Report and Literature Review. Braz. J. Infect. Dis. 2022, 26, 102368. [Google Scholar] [CrossRef] [PubMed]
- Boccia, F.; Florio, L.L.; Durante-Mangoni, E.; Zampino, R. Guillain-Barré Syndrome as Clinical Presentation of a Recently Acquired Hepatitis C. J. Neurovirol. 2023, 29, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.F.d.O.; Nogueira, V.B.; Maury, W.; Wilson, M.E.; Júnior, M.E.T.D.; Teixeira, D.G.; Bezerra Jeronimo, S.M. Altered Cellular Pathways in the Blood of Patients With Guillain-Barre Syndrome. J. Peripher. Nerv. Syst. 2025, 30, e70012. [Google Scholar] [CrossRef]
- Gong, S.; Deng, B.; Yu, H.; Zhang, X.; Yang, W.; Chen, X. Clinical and Immunological Features in Patients with Neuroimmune Complications of COVID-19 during Omicron Wave in China: A Case Series. Front. Immunol. 2024, 15, 1499082. [Google Scholar] [CrossRef]
- Balakrishnan, U.L.; Desai, N.K.; Yen, K.G. Third Nerve Palsy as First Presenting Symptom of Guillain-Barre Syndrome Spectrum Clinical Variant. Am. J. Ophthalmol. Case Rep. 2025, 38, 102291. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, C.; Weng, W.; Wei, Y. Predictors of the Short-Term Outcomes of Guillain–Barré Syndrome: Exploring Electrodiagnostic and Clinical Features. Brain Behav. 2025, 15, e70257. [Google Scholar] [CrossRef]
- Deb, J.; Negi, G.; Mohan, A.K.; Sharawat, I.K.; Banerjee, P.; Chauhan, D.; Kaur, D.; Jain, A. Potential Advantage of Therapeutic Plasma Exchange over Intravenous Immunoglobulin in Children with Axonal Variant of Guillain-Barré Syndrome: A Report of Six Paediatric Cases. Transfus. Clin. Biol. 2025, 32, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Stino, A.M.; Reynolds, E.L.; Watanabe, M.; Callaghan, B.C. Intravenous Immunoglobulin and Plasma Exchange Prescribing Patterns for Guillain-Barre Syndrome in the United States—2001 to 2018. Muscle Nerve 2024, 70, 1192–1199. [Google Scholar] [CrossRef]
- Ullah, W.; Ali, H.; Muhammad, Y.; Ullah, N.; Khalil, I.; Ahmad, S.; Ullah, O.; Ali, A.; Ali, M.Y.; Manan, A.; et al. Evaluating the Efficacy and Cost-Effectiveness of Plasmapheresis and Intravenous Immunoglobulin in Acute Guillain-Barre Syndrome Management in Emergency Departments. Cureus 2024, 16, e7259. [Google Scholar] [CrossRef]
- Announcements. Ann. Neurol. 1978, 3, 565–566. [CrossRef]
- Willison, H.J.; Jacobs, B.C.; van Doorn, P.A. Guillain-Barré Syndrome. Lancet 2016, 388, 717–727. [Google Scholar] [CrossRef]
- Leonhard, S.E.; Mandarakas, M.R.; Gondim, F.A.A.; Bateman, K.; Ferreira, M.L.B.; Cornblath, D.R.; van Doorn, P.A.; Dourado, M.E.; Hughes, R.A.C.; Islam, B.; et al. Diagnosis and Management of Guillain–Barré Syndrome in Ten Steps. Nat. Rev. Neurol. 2019, 15, 671–683. [Google Scholar] [CrossRef]
- Asbury, A.K.; Cornblath, D.R. Assessment of Current Diagnostic Criteria for Guillain-Barré Syndrome. Ann. Neurol. 1990, 27, S21–S24. [Google Scholar] [CrossRef]
- van Doorn, P.A.; Van den Bergh, P.Y.K.; Hadden, R.D.M.; Avau, B.; Vankrunkelsven, P.; Attarian, S.; Blomkwist-Markens, P.H.; Cornblath, D.R.; Goedee, H.S.; Harbo, T.; et al. European Academy of Neurology/Peripheral Nerve Society Guideline on Diagnosis and Treatment of Guillain–Barré Syndrome. Eur. J. Neurol. 2023, 30, 3646–3674. [Google Scholar] [CrossRef] [PubMed]
- Doncel-Pérez, E.; Mateos-Hernández, L.; Pareja, E.; García-Forcada, Á.; Villar, M.; Tobes, R.; Romero Ganuza, F.; Vila del Sol, V.; Ramos, R.; Fernández de Mera, I.G.; et al. Expression of Early Growth Response Gene-2 and Regulated Cytokines Correlates with Recovery from Guillain–Barré Syndrome. J. Immunol. 2016, 196, 1102–1107. [Google Scholar] [CrossRef] [PubMed]
- Mateos-Hernández, L.; Villar, M.; Doncel-Pérez, E.; Trevisan-Herraz, M.; García-Forcada, Á.; Ganuza, F.R.; Vázquez, J.; de la Fuente, J. Quantitative Proteomics Reveals Piccolo as a Candidate Serological Correlate of Recovery from Guillain-Barré Syndrome. Oncotarget 2016, 7, 74582. [Google Scholar] [CrossRef] [PubMed]
- Kleiveland, C.R. Peripheral Blood Mononuclear Cells. In The Impact of Food Bioactives on Health; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 978-3-319-15791-7. [Google Scholar]
- Navarro, P.; Trevisan-Herraz, M.; Bonzon-Kulichenko, E.; Núñez, E.; Martínez-Acedo, P.; Pérez-Hernández, D.; Jorge, I.; Mesa, R.; Calvo, E.; Carrascal, M.; et al. General Statistical Framework for Quantitative Proteomics by Stable Isotope Labeling. J. Proteome Res. 2014, 13, 1234–1247. [Google Scholar] [CrossRef]
- García-Marqués, F.; Trevisan-Herraz, M.; Martínez-Martínez, S.; Camafeita, E.; Jorge, I.; Lopez, J.A.; Méndez-Barbero, N.; Méndez-Ferrer, S.; del Pozo, M.A.; Ibáñez, B.; et al. A Novel Systems-Biology Algorithm for the Analysis of Coordinated Protein Responses Using Quantitative Proteomics. Mol. Cell. Proteom. 2016, 15, 1740–1760. [Google Scholar] [CrossRef]
- Doncel-Pérez, E.; Contreras, M.; Gómez Hernando, C.; Vargas Baquero, E.; Blanco García, J.; Rodríguez Gómez, J.; Velayos Galán, A.; Cabezas-Cruz, A.; Gortázar, C.; de la Fuennte, J. What Is the Impact of the Antibody Response to Glycan Alpha-Gal in Guillain-Barré Syndrome Associated with SARS-CoV-2 Infection? Merit Res. J. Med. Med. Sci. 2020, 8, 730–737. [Google Scholar] [CrossRef]
- Urra, J.M.; Ferreras-Colino, E.; Contreras, M.; Cabrera, C.M.; Fernández de Mera, I.G.; Villar, M.; Cabezas-Cruz, A.; Gortázar, C.; de la Fuente, J. The Antibody Response to the Glycan A-Gal Correlates with COVID-19 Disease Symptoms. J. Med. Virol. 2021, 93, 2065–2075. [Google Scholar] [CrossRef]
- Pacheco, I.; Fernández de Mera, I.G.; Feo Brito, F.; Gómez Torrijos, E.; Villar, M.; Contreras, M.; Lima-Barbero, J.F.; Doncel-Pérez, E.; Cabezas-Cruz, A.; Gortázar, C.; et al. Characterization of the Anti-α-Gal Antibody Profile in Association with Guillain-Barré Syndrome, Implications for Tick-Related Allergic Reactions. Ticks Tick. Borne. Dis. 2021, 12, 101651. [Google Scholar] [CrossRef]
- Bellanti, R.; Rinaldi, S. Guillain-Barré Syndrome: A Comprehensive Review. Eur. J. Neurol. 2024, 31, e16365. [Google Scholar] [CrossRef]
- Kim, S.-H.; Weiß, C.; Hoffmann, U.; Borggrefe, M.; Akin, I.; Behnes, M. Advantages and Limitations of Current Biomarker Research: From Experimental Research to Clinical Application. Curr. Pharm. Biotechnol. 2017, 18, 445–455. [Google Scholar] [CrossRef]
- Cope, H.; Willis, C.R.G.; MacKay, M.J.; Rutter, L.A.; Toh, L.S.; Williams, P.M.; Herranz, R.; Borg, J.; Bezdan, D.; Giacomello, S.; et al. Routine Omics Collection Is a Golden Opportunity for European Human Research in Space and Analog Environments. Patterns 2022, 3, 100550. [Google Scholar] [CrossRef]
- Shi, Q.; Huang, J.; Wan, J.; Zhong, Z.; Sun, Y.; Zhou, Y.; Li, J.; Tan, X.; Yu, B.; Lu, Y.; et al. Physical Frailty, Genetic Predisposition, and Incident Heart Failure. JACC Asia 2024, 4, 547–556. [Google Scholar] [CrossRef]
- Oeztuerk, M.; Henes, A.; Schroeter, C.B.; Nelke, C.; Quint, P.; Theissen, L.; Meuth, S.G.; Ruck, T. Current Biomarker Strategies in Autoimmune Neuromuscular Diseases. Cells 2023, 12, 2456. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.; Joshi, M. Occurrence of Neuropathic Pain and Its Characteristics in Patients with Traumatic Spinal Cord Injury. J. Spinal Cord Med. 2024, 47, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Fredø, H.L.; Rizvi, S.A.M.; Rezai, M.; Rønning, P.; Lied, B.; Helseth, E. Complications and Long-Term Outcomes after Open Surgery for Traumatic Subaxial Cervical Spine Fractures: A Consecutive Series of 303 Patients. BMC Surg. 2016, 16, 56. [Google Scholar] [CrossRef] [PubMed]
- Cicek, M.S.; Olson, J.E. Mini-Review of Laboratory Operations in Biobanking: Building Biobanking Resources for Translational Research. Front. Public Health 2020, 8, 362. [Google Scholar] [CrossRef]
- Keddie, S.; Smyth, D.; Keh, R.Y.S.; Chou, M.K.L.; Grant, D.; Surana, S.; Heslegrave, A.; Zetterberg, H.; Wieske, L.; Michael, M.; et al. Peripherin Is a Biomarker of Axonal Damage in Peripheral Nervous System Disease. Brain 2023, 146, 4562–4573. [Google Scholar] [CrossRef]
- van Doorn, P.A. Diagnosis, Treatment and Prognosis of Guillain-Barré Syndrome (GBS). Presse Med. 2013, 42, e193–e201. [Google Scholar] [CrossRef]
- Doets, A.Y.; Lingsma, H.F.; Walgaard, C.; Islam, B.; Papri, N.; Davidson, A.; Yamagishi, Y.; Kusunoki, S.; Dimachkie, M.M.; Waheed, W.; et al. Predicting Outcome in Guillain-Barré Syndrome. Neurology 2022, 98, e518–e532. [Google Scholar] [CrossRef]
- Jacobs, B.C.; van den Berg, B.; Verboon, C.; Chavada, G.; Cornblath, D.R.; Gorson, K.C.; Harbo, T.; Hartung, H.; Hughes, R.A.C.; Kusunoki, S.; et al. International Guillain-Barré Syndrome Outcome Study: Protocol of a Prospective Observational Cohort Study on Clinical and Biological Predictors of Disease Course and Outcome in Guillain-Barré Syndrome. J. Peripher. Nerv. Syst. 2017, 22, 68–76. [Google Scholar] [CrossRef]
- Taylor, H.; Lewins, M.; Foody, M.G.B.; Gray, O.; Bešević, J.; Conroy, M.C.; Collins, R.; Lacey, B.; Allen, N.; Burkitt-Gray, L. UK Biobank—A Unique Resource for Discovery and Translation Research on Genetics and Neurologic Disease. Neurol. Genet. 2025, 11, e200226. [Google Scholar] [CrossRef]
- Deng, Y.-T.; You, J.; He, Y.; Zhang, Y.; Li, H.-Y.; Wu, X.-R.; Cheng, J.-Y.; Guo, Y.; Long, Z.-W.; Chen, Y.-L.; et al. Atlas of the Plasma Proteome in Health and Disease in 53,026 Adults. Cell 2025, 188, 253–271.e7. [Google Scholar] [CrossRef]
- Stavropoulos, F.; Sargiannidou, I.; Potamiti, L.; Kagiava, A.; Panayiotidis, M.I.; Bae, J.H.; Yeom, S.C.; Lee, J.Y.; Kleopa, K.A. Aberrant Mitochondrial Dynamics and Exacerbated Response to Neuroinflammation in a Novel Mouse Model of CMT2A. Int. J. Mol. Sci. 2021, 22, 11569. [Google Scholar] [CrossRef] [PubMed]
- van Doorn, P.A.; Ruts, L.; Jacobs, B.C. Clinical Features, Pathogenesis, and Treatment of Guillain-Barré Syndrome. Lancet Neurol. 2008, 7, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Disanto, G.; Barro, C.; Benkert, P.; Naegelin, Y.; Schädelin, S.; Giardiello, A.; Zecca, C.; Blennow, K.; Zetterberg, H.; Leppert, D.; et al. Serum Neurofilament Light: A Biomarker of Neuronal Damage in Multiple Sclerosis. Ann. Neurol. 2017, 81, 857–870. [Google Scholar] [CrossRef] [PubMed]
- Ladenson, J.H.; Tsai, L.-M.B.; Michael, J.M.; Kessler, G.; Joist, J.H. Serum versus Heparinized Plasma for Eighteen Common Chemistry Tests: Is Serum the Appropriate Specimen? Am. J. Clin. Pathol. 1974, 62, 545–552. [Google Scholar] [CrossRef]
- Kieseier, B.C.; Mathey, E.K.; Sommer, C.; Hartung, H.-P. Immune-Mediated Neuropathies. Nat. Rev. Dis. Prim. 2018, 4, 31. [Google Scholar] [CrossRef]
- Neurofilament Light Chain: A Prognostic Biomarker in Amyotrophic Lateral Sclerosis. Neurology 2015, 85, 921. [CrossRef] [PubMed]
- Lippi, G.; Guidi, G.C.; Mattiuzzi, C.; Plebani, M. Preanalytical Variability: The Dark Side of the Moon in Laboratory Testing. Clin. Chem. Lab. Med. 2006, 44, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.J.; Vitzthum, F. Effects of Preanalytical Variables on Peptide and Protein Measurements in Human Serum and Plasma: Implications for Clinical Proteomics. Expert Rev. Proteom. 2006, 3, 409–426. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, B.C.; Rothbarth, P.H.; van der Meché, F.G.A.; Herbrink, P.; Schmitz, P.I.M.; de Klerk, M.A.; van Doorn, P.A. The Spectrum of Antecedent Infections in Guillain-Barré Syndrome. Neurology 1998, 51, 1110–1115. [Google Scholar] [CrossRef] [PubMed]




| Biochemical Parameter | Detection Method |
|---|---|
| Creatinine | Jaffé reaction |
| Uric Acid | Uricase-Peroxidase; enzymatic colorimetric |
| Sodium | Indirect potentiometry |
| Potassium | Indirect potentiometry |
| Chloride | Indirect potentiometry |
| Calcium | Photometric NM-BAPTA/EDTA determination |
| Phosphate | Photometric molybdate |
| Magnesium | Xylidyl blue colorimetric method (end-point determination) |
| Albumin | Immunoturbidimetry |
| AST/GOT (Aspartate Aminotransferase) | Enzymatic UV method (NADH-linked) |
| ALT/GPT (Alanine Aminotransferase) | Enzymatic UV Method (NADH-linked) |
| GGT (Gamma Glutamyl Transferase) | Enzymatic Colorimetric Method with γ-Glutamyl-3-carboxy-4-nitroanilide substrate |
| Total Bilirubin | Colorimetric Diazo method |
| C-reactive Protein | Latex particles-enhanced immunoturbidimetry |
| Lipase | Colorimetric enzymatic method by chromogenic substrate (1,2-O-dilauryl-rac-glycero-3-glutaric acid-(6′-methylresorufin) ester) |
| Cholinesterase | Colorimetric assay using butyrylthiocholine and hexacyanoferrate |
| Creatine Kinase | UV kinetic enzymatic method (NADPH-linked) |
| Interleukin-6 | Electrochimioluminiscense immunoessay |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz, R.; Blanco-García, J.; Rodríguez-Gómez, J.; Vargas-Baquero, E.; Fernández-Alarcón, C.; Terán-Tinedo, J.R.; Romero-Ramírez, L.; Mey, J.; de la Fuente, J.; Villar, M.; et al. Hospital Coordination and Protocols Using Serum and Peripheral Blood Cells from Patients and Healthy Donors in a Longitudinal Study of Guillain–Barré Syndrome. Diagnostics 2025, 15, 1900. https://doi.org/10.3390/diagnostics15151900
Díaz R, Blanco-García J, Rodríguez-Gómez J, Vargas-Baquero E, Fernández-Alarcón C, Terán-Tinedo JR, Romero-Ramírez L, Mey J, de la Fuente J, Villar M, et al. Hospital Coordination and Protocols Using Serum and Peripheral Blood Cells from Patients and Healthy Donors in a Longitudinal Study of Guillain–Barré Syndrome. Diagnostics. 2025; 15(15):1900. https://doi.org/10.3390/diagnostics15151900
Chicago/Turabian StyleDíaz, Raquel, Javier Blanco-García, Javier Rodríguez-Gómez, Eduardo Vargas-Baquero, Carmen Fernández-Alarcón, José Rafael Terán-Tinedo, Lorenzo Romero-Ramírez, Jörg Mey, José de la Fuente, Margarita Villar, and et al. 2025. "Hospital Coordination and Protocols Using Serum and Peripheral Blood Cells from Patients and Healthy Donors in a Longitudinal Study of Guillain–Barré Syndrome" Diagnostics 15, no. 15: 1900. https://doi.org/10.3390/diagnostics15151900
APA StyleDíaz, R., Blanco-García, J., Rodríguez-Gómez, J., Vargas-Baquero, E., Fernández-Alarcón, C., Terán-Tinedo, J. R., Romero-Ramírez, L., Mey, J., de la Fuente, J., Villar, M., Beneitez, A., Muñoz-Turrillas, M. d. C., Zurdo-López, M., Sagredo del Río, M., Lorenzo-Lozano, M. d. C., Marsal-Alonso, C., Morales-Casado, M. I., Parra-Serrano, J., & Doncel-Pérez, E. (2025). Hospital Coordination and Protocols Using Serum and Peripheral Blood Cells from Patients and Healthy Donors in a Longitudinal Study of Guillain–Barré Syndrome. Diagnostics, 15(15), 1900. https://doi.org/10.3390/diagnostics15151900








