1. Introduction
All the recent hardware and software advances incorporated in the technology of Wireless Endoscopic Capsule (WEC) robots constantly demonstrate their great potential for improvement, as research boundaries progress even further, revealing new opportunities for gastroenterologists and patients, for better diagnosis and treatment. The WEC has the potential to become a screening, diagnostic and therapeutic technique for the entire GI tract because of its low discomfort to users [
1,
2,
3,
4]. Thus, the WEC has become the first choice for small bowel endoscopy in children [
5,
6,
7]. Moreover, the use of a robotic WEC (in the future) promises to offer some form of targeted “smart” therapy, e.g., drug delivery, clip release for bleeding control, precise biopsy sampling, etc. Nevertheless, there are some specific computer-aided techniques based on AI that are approved by the European Medicines Agency (EMA) and by the Pharmaceuticals and Medical Devices Agency (PMDA) since 2018 [
8]:
- -
CADe by FUJIfilm, PENTAX,
- -
GI Genious by Medtronic,
- -
EndoBRAIN by Cybernet,
- -
Endo-AID by Olympus (as of this time, pending approval).
To the best of our knowledge, no methods or algorithms seem to have been proposed yet to automatically detect the capsule passing by selected GI organs (their extend, from start to finish), although medical reviewers can surely identify such a position through their optical review and diagnosis later on. This kind of automation can play a significant role in the near future, as technology evolves rapidly towards better, cheaper and more efficient endoscopic capsules. The present work reveals such a case, specifically the detection of WEC’s transition and regional awareness while passing the boundaries of gastrointestinal (GI) organs through the GI tract.
A lot of algorithms are proposed for reducing WEC’s videos review time by the experts, by detecting and diagnosing of hemorrhage and GI tract lesions based on AI [
9,
10,
11,
12]. The topic of localizing the capsule inside GI has gained a lot of interest among researchers. Few robotic research groups have proposed methods for the real time localization of the capsule, possibly by providing wireless endoscopic capsule control through magnetic devices and other RF localization techniques [
13,
14].
To the best of our knowledge, the most relevant algorithms capable of classifying single frame images into GI organs, or detecting the boundaries of the GI organs are presented in references [
15,
16]. In [
15], a CNN algorithm with a temporal filtering detects three GI organs (the stomach, the small bowel, and the colon), but is not able to identify the boundary transitions among these organs. On the other hand, the algorithm of [
16], employing the long-term dependencies of WEC frames, detects both transitions between stomach and small bowel and between small bowel to colon. Moreover, the CAD tool based on CNN and SVM algorithms for narrow band imaging endoscopy is implemented into the FPGA-based prototyping platform [
17].
In this study, the problem to solve is the real-time tracking of the WEC transition through the boundaries of GI’s organs (esophagus to stomach, stomach to small intestine, small intestine to colon), from CNN multi-class classification of endoscopy capsule images. Moreover, the algorithms can be executed from a mobile platform that can be physically attached for the time of recording upon the patient. Another aim is to study how the difference of the kernel size and number effects the CNN’s performance or how adding dropout layers will affect the possibility of overfitting and underfitting of the proposed CNN classifier.
2. Materials and Methods
This prospective, noninterventional study was conducted at Aretaeio Hospital, at the Department of Surgery, School of Medicine, National and Kapodistrian University of Athens, Athens, Greece from December 2019 to June 2020. The School of Electrical and Computer Engineering of the Technical University of Crete, Greece and the Department of Electronic Engineering, Hellenic Mediterranean University, were responsible for CAC algorithm development.
2.1. Data and Resources
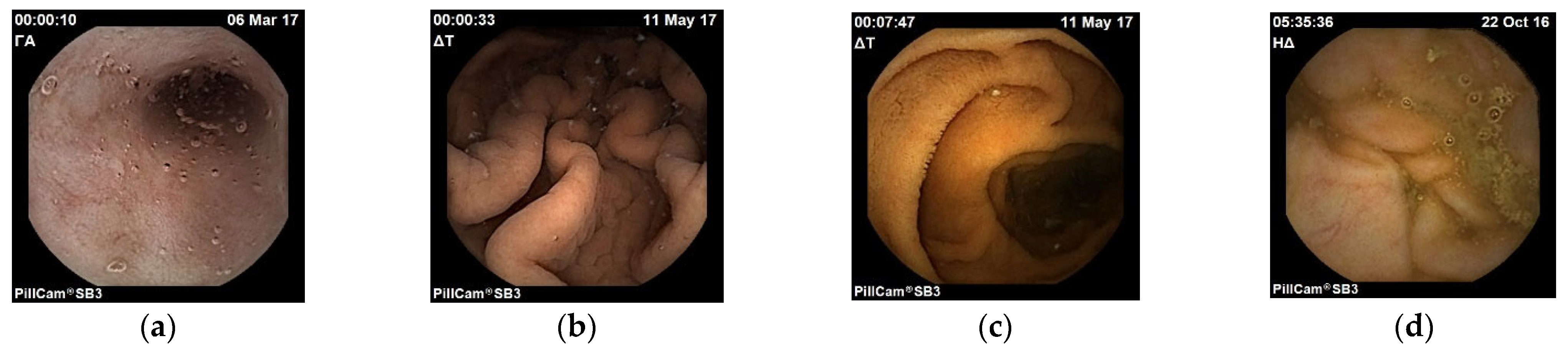
For this research, we have used video data out of a total of 138 PillcamTM SB endoscopic capsule videos collected from random anonymous patients hospitalized in four different Gastroenterology Departments of (a) the Medical School of the National and Kapodistrian University of Athens, (b) the Attikon University Hospital, (c) the Laikon University Hospital and (d) the Medical School of the Aristotle University of Thessaloniki. All these 138 patients have been diagnosed by their attending doctors positive to: (i) bleeding, (ii) angiodysplasias, (iii) haemangiomas, or (iv) lesions which predispose to bleeding in the small intestine. The 138 videos have been evaluated by the third coauthor, who is a very experienced endoscopist, and divided into two sets: the training set with 99 videos which are used by the proposed algorithm for feature extraction and the validation set with the rest of the 39 videos which are used for independent validation as images unseen from the algorithm. From those videos, there was an extraction of total 5520 frames, 1380 frames for each organ (ten frames for each organ boundary of each video) of interest: esophagus, stomach, small intestine, colon. In order to achieve success in regional information extraction of the endoscopy capsule, the problem to be solved is the organ’s entrance (gate) classification.
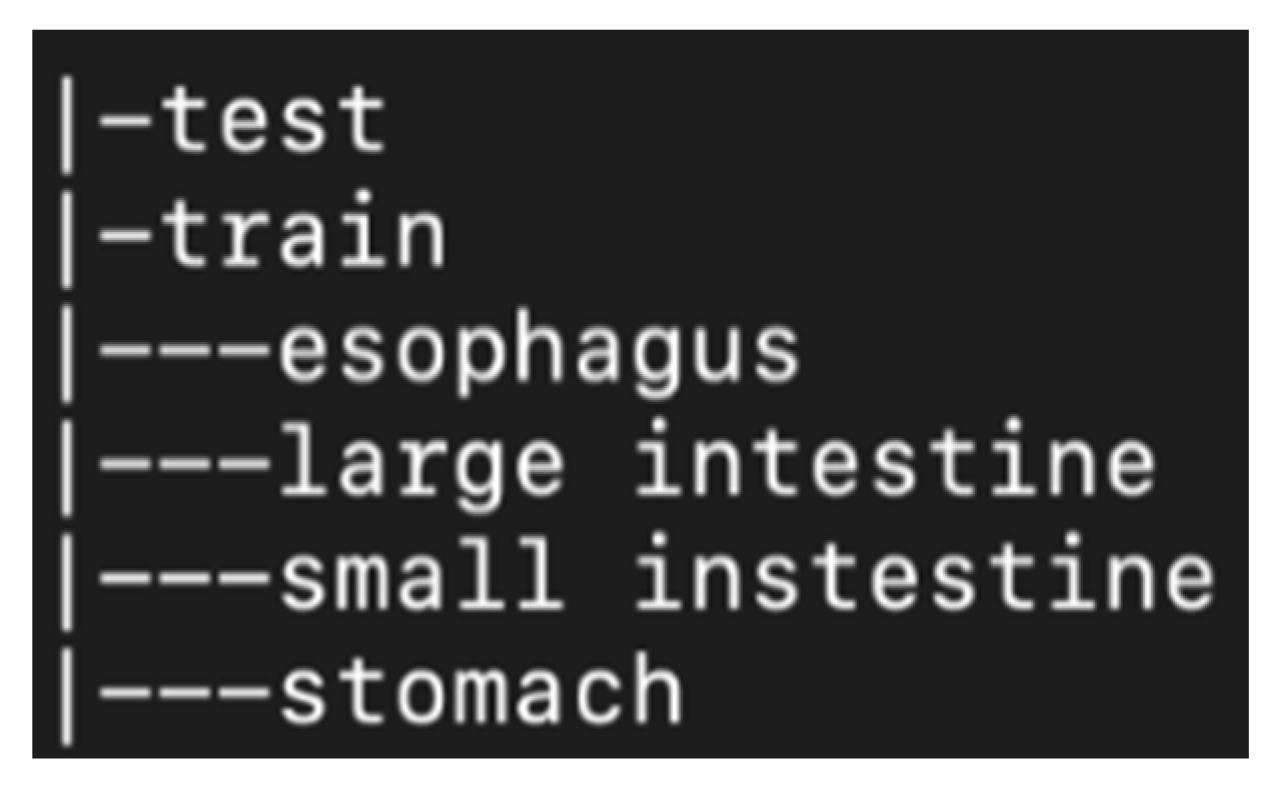
After extracting the images in order to use them for machine learning purposes, images were divided in different folders. First of all, they were distinguished in training data (total 5520 frames, 1380 for each organ) and in testing data (total 496 frames, 124 frames for each organ). Training data are the ones that are fed into the model in the training phase and testing are the ones used in order to verify that the model is producing comprehensive training results. Afterwards, the training data were split in four folders (see
Figure 1). Each folder represents one of the four points of interest. For example, the stomach folder contains only images that are coming from that area. Moreover, for the training process, a CSV file was created in which there is a labelling of the images based on the category they belong to and their name. In
Figure 2, images samples are shown.
All images are in three colour channels (RBG—Red Blue Green), with the typical pixel level [0, 255], although the data are normalized during the experiment. The data of the training folder are separated so that 80% of them are used for the phase of training the model. The rest of the 20% are used as validation data during the training process. Validation data are important so that we have a quicker and better feedback on the model and we do not fall into cases of overfitting or underfitting [
18].
To conclude, before feeding the data into the training phase of the model, the following actions were performed:
create validation data using 20% of the training data, in order to be used as unknown data during the training process;
reduce the size of images from the 576 × 576 original image size to 256 × 256;
define the batch size to be 16;
use autotune data, in order to help our model to be faster, since it will decouple the time when data is produced from the time when data is consumed. It will use a background thread and an internal buffer to prefetch elements from the input dataset ahead of the time they are requested. Autotune function will prompt the tf.data to tune the value dynamically at runtime;
perform data normalization, by transforming all pixel values in the [0, 1] interval.
2.2. Proposed CNNs 4-Class Classifiers
Taking under consideration similar research that classifies images into corresponding categories, the Convolutional Neural Network (CNN) model is well known to be good at image classification. CNN stands for Convolutional Neural Network, where each image goes through a series of convolution and max pooling for features extraction. CNN takes an input image and assigns importance (weights and biases) to various features to help in distinguishing images, for example, animal classification [
19], bleeding and non-bleeding images in endoscopy capsule [
11], ImageNET [
20], concluded that Convolutional Neural Networks were a great starting point.
In general, CNN contains a series of connected layers, containing the convolutional layer, the pooling layer and the dense layer. For this research, three different CNN models were created and evaluated. Additionally, there was a testing of those models including data augmentation. The input of our models at all times are images in three color channels (RGB) with final dimensions [256 × 256]. Then, the pixel values were normalized. This is achieved by subtracting the mean of each pixel and then diving that result by the standard deviation. That will lead to a Gaussian curve centred to zero. In case of images, the possible values are only the positive ones so it will move the data from the value range of [0, 255] to the value range of [0, 1].
The idea around Convolutional Neural Networks is that there are the layers that will help in feature extraction, the convolutional layers in cooperation with the pooling layers, and the layers that will transform their input to a decision, which are the flatten layers. Convolutional layers will perform a linear matrix multiplication by sliding the filter windows through the image pixels.
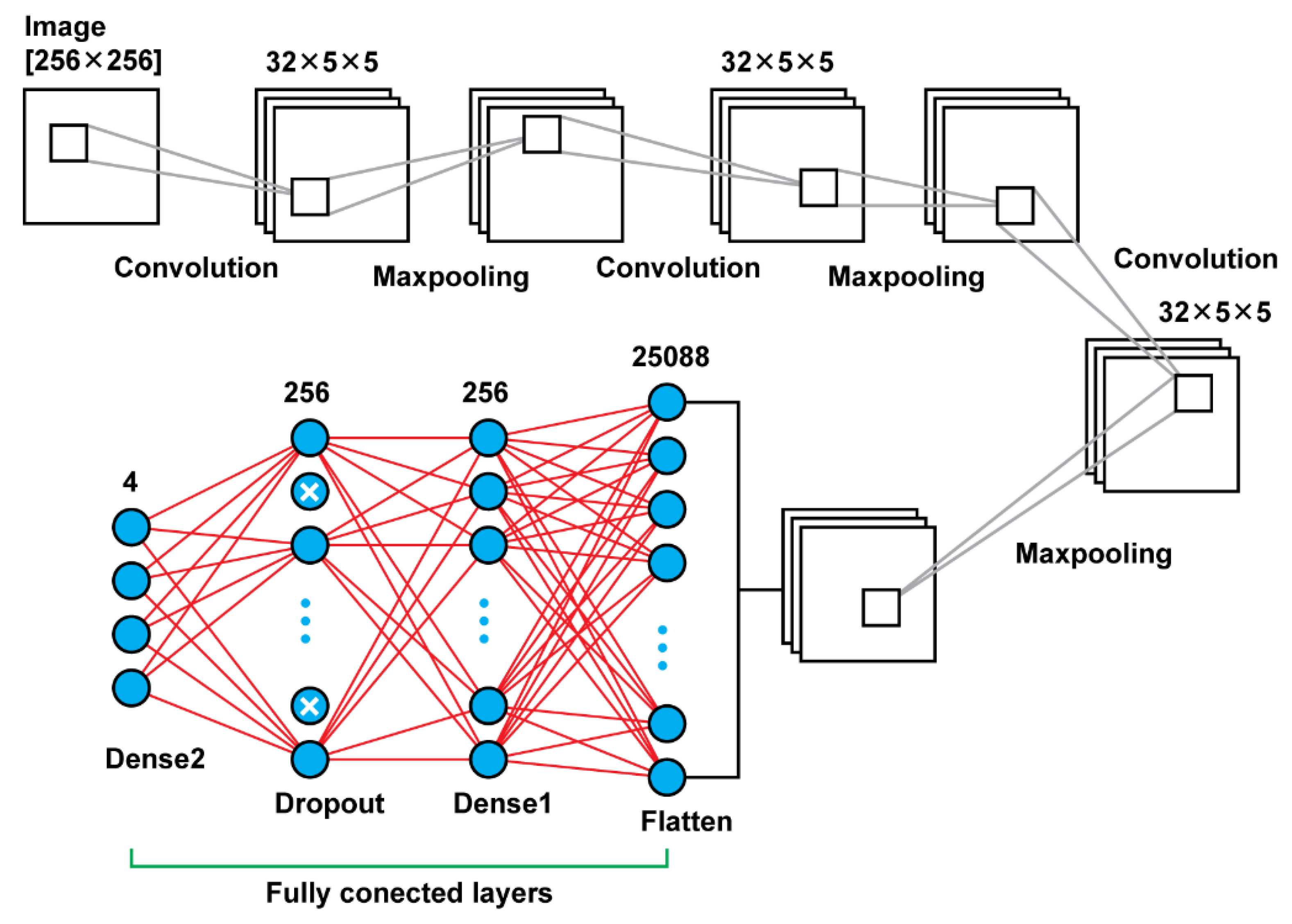
In our research, when it comes to the convolutional layers in Model 1 and 2, the filters are of the size [3 × 3], and in Model 3 are of the size [5 × 5]. From layer to layer in each model, what is changed, also, is the number of filters applied for example, in Model 1 (
Figure 2), firstly, we apply 32 filters, later on 64 and finally 128. Between the Convolutional Layers there is a pooling function, that function helps in order to select the most powerful features, in a window of [2 × 2]; it will parse the outcome of the convolutional layer and select the maximum values. In the next connected layer, the filters will be applied to the strongest feature in order to extract the ones that are more relevant. This will provide also the advantage of downsampling the features and reducing the computational cost of the algorithm and the same time we will care for the most relevant characteristics of the images.
For the code and result production, open source tools were used. All code was written in Python and run in the Google Colab platform in order to have an environment with more resources in memory and GPU than the local system. The libraries that were used for data pre-processing and model creation are Tensorflow and Keras. For image manipulation, the cv2 library was used. From Keras, in order to create the models’ layers, we used the functions: Conv2D, Maxpool2D, Flatten and Dense, Sequential (in order to define that we are referring to a Convolutional Neural Network).
2.2.1. CNN–Model 1
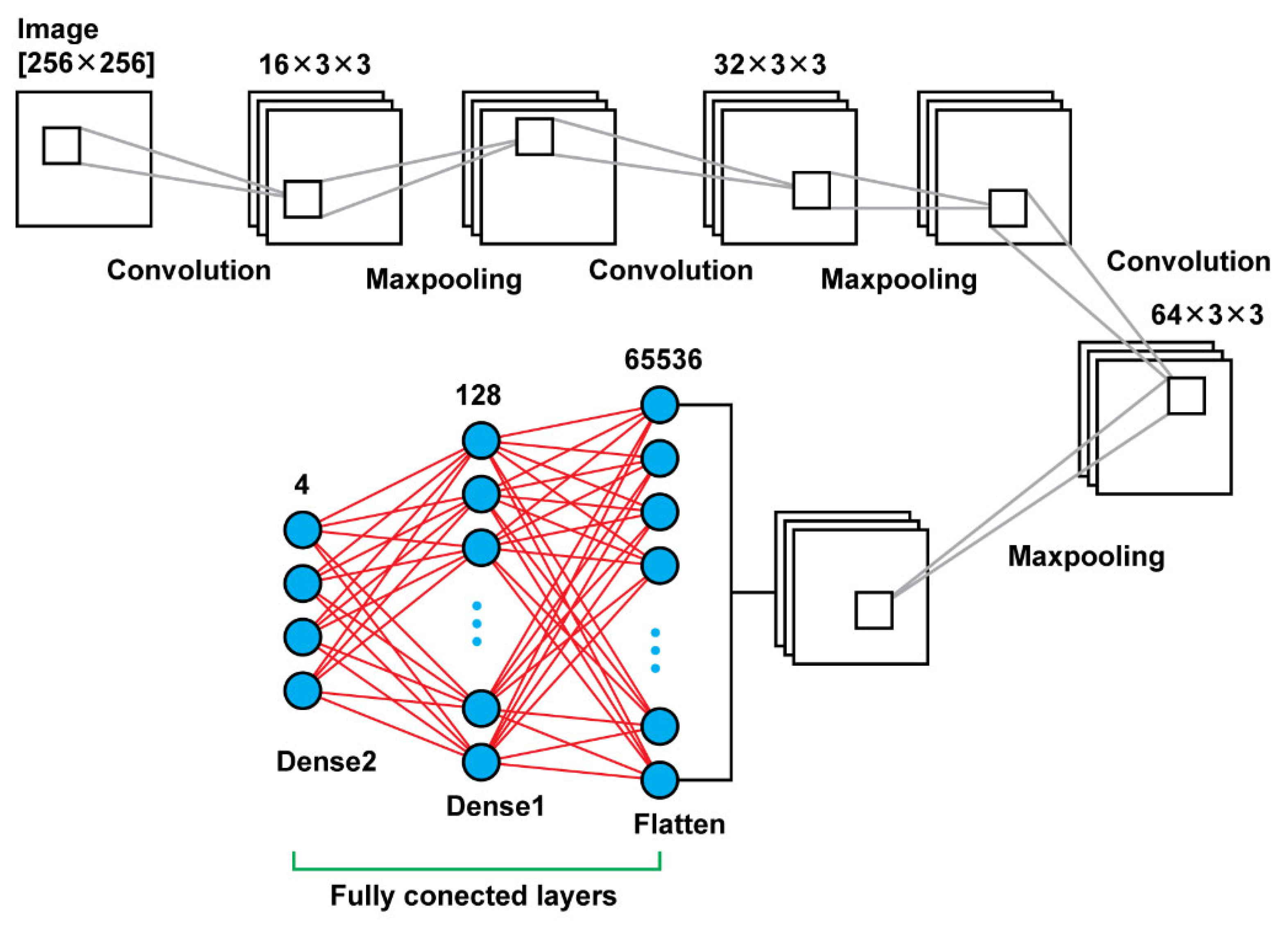
Model 1 consists connected two dimensional convolution layers and two dimensional maxpooling layers as feature extractors, a flatten layer in order to have a vector of features after the last pooling layer that will be an input for the upcoming two dense layers that follow in order to make the final decision of the classification. In total, using that model, there were 8,412,836 trainable parameters. In
Table 1 are shown the details regarding the different layers as well as the sizes of the filters and the trainable parameters. In
Figure 3, there is a graphical representation of Model 1.
2.2.2. CNN–Model 2
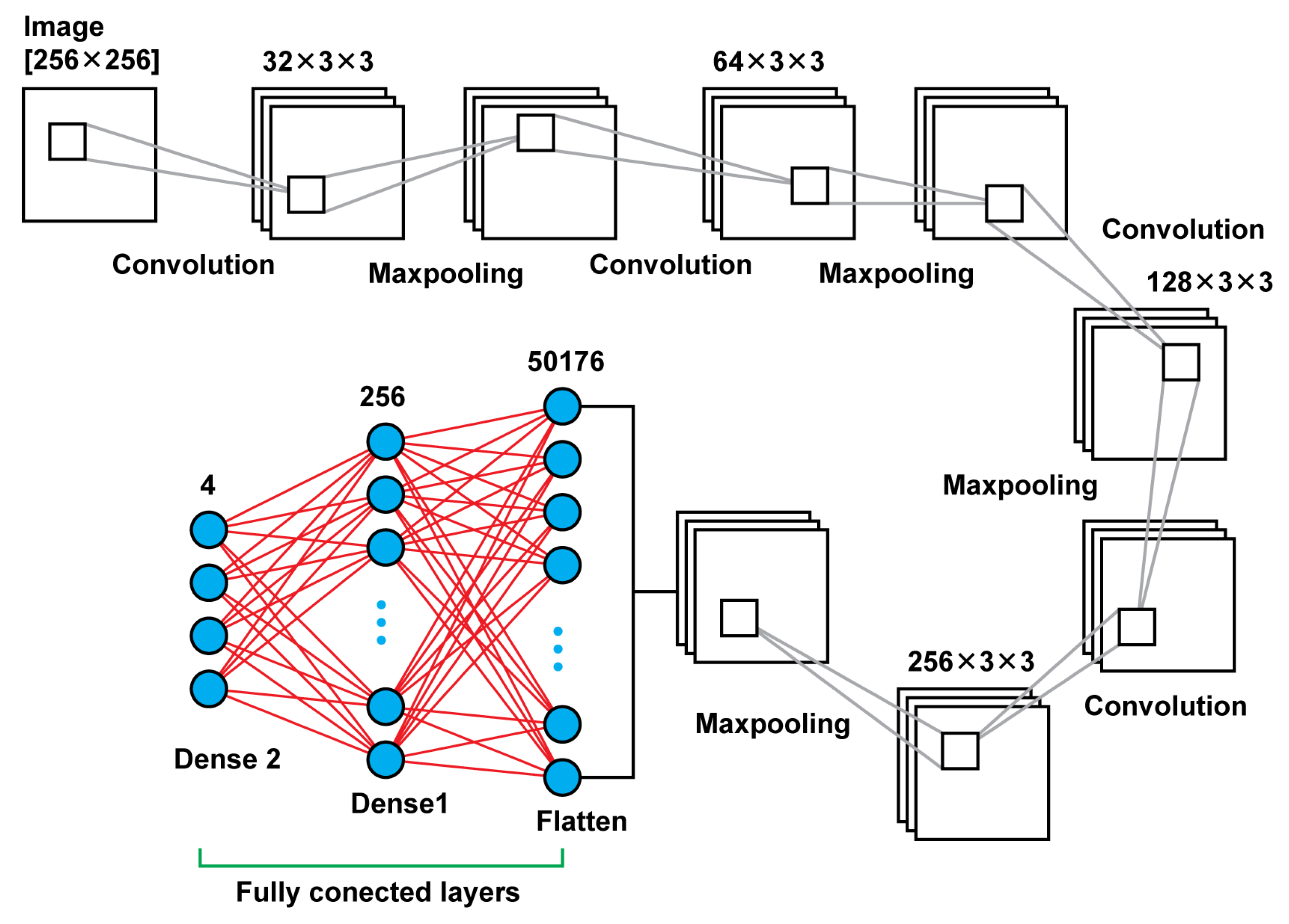
Model 2 is a deeper neural network using, again, connected two dimensional convolution layers and two dimensional maxpooling layers as feature extractors, a flatten layer in order to create a vector of features after the last pooling layer that will be an input for the upcoming two dense layers. In total, that model leads to 13,235,756 trainable parameters. In
Table 2, there are details regarding output size of the different layers as well as the sizes of the filters and the trainable parameters. In
Figure 4, there is a graphical representation of Model 2.
2.2.3. CNN–Model 3
Model 3 is using convolutional layers with filters of the size (5 × 5) instead the filters of size (3 × 3) that were using the previous models. Moreover, in the previous models, there was an increase of the number of filters that were used, as the network was getting deeper. For this model, there were used in all steps filters of the same size and number; in every convolutional layer there were used 32 filters of the size (5 × 5). Another difference of this model is that there was used also a dropout layer between the last two dense layers. Other than those two key differences, the rest remain the same. In total, that model results in 6,477,508 trainable parameters. In
Figure 5, there is a graphical representation of Model 3 and
Table 3 presents the details.
3. Results of CNNs Training
Taking under consideration the relatively small set of data (5520 frames selected from 99 videos) that we are using for training as well as the fact that the data did not undergo any pre-processing before arriving at the training process, our results stand out. There are references on the curves that refer to the loss and accuracy of training and validation data as per epoch of the training process. Those curves are provided as per model.
All models used 30 epochs in order to conclude to a stable value of validation and training accuracy. This amount of epochs worked well enough to allow our training model to conclude with a good training convergence. There was used also instead of individual images, a patch of images of size 16. For the Model 1, training accuracy and validation accuracy start being stable after epoch 15; for Model 2 we see the same behavior after epoch 20 and for the Model 3, those values start being stable after epoch 25. We observed, after a relatively small amount of epochs, the results we were taking were quite sufficient. Model 1 concludes with training accuracy 1 and validation accuracy 0.9677. Model 2 provides training accuracy 1 and validation accuracy 0.9677. Moreover, Model 3 provides training accuracy 1 and validation accuracy 1. All those values refer to the results of the 30th epoch. Among the three models, the one that provides a more stable view, as many times as the experiment of training was reproduced, is Model 2. We observed that increasing the number of filters as the model becomes deeper shows that is providing a more stable result.
4. Methods of Multiclass CNNs Independent Evaluation
The independent evaluation for the prediction quality of our multiclass CNNs, is the trained models test, examining data not used in the training procedure. To evaluate the quality of the performance of any multiclass classifier and compare them to each other, it is broken into multiple binary classification models. The Mattheus correlation coefficient (MCC), calculates a high score only if the prediction obtained correct results in all of the confusion matrix categories (true positives, false negatives, true negatives, and false positives), proportionally both to the size of positive elements and the size of negative elements in the dataset [
21].
where
K is the number of classes,
- -
is the number of times class k truly correct,
- -
is the number of times class k was predicted into class,
- -
is the total number of samples that was correctly predicted,
- -
is the overall number of samples, and
- -
includes the elements wrongly classified by the model and covers multiplicative entities that are weaker than the product c × s.
To evaluate each multi class classifier, the macro average
F1 score indicates how the classifier performs for every class [
22]. The macro average involves computing one versus all confusion matrices for each class, where each individual
ith class is the positive class and all the other classes are the negative class. Since our experiment refers to a 4-class classification, for each class we calculate the values of True Positive (
TP), True Negative (
TN), False Positive (
FP), False Negative (
FN). The macro average
F1 score is defined by the harmonic mean of the macro average of recall (or sensitivity) and macro average precision as:
where the macro average calculates a simple average of the binary classification metric values of all classes,
where precision of the
ith class,
Pi, stands for the fraction of positive class predictions that were actually positive. The value of sensitivity or recall of the
ith class,
Ri, stands for the fraction of all positive samples that were correctly predicted as positive by the classifier.
Accuracy is providing the overall accuracy of a model for a specific class, meaning the fraction of the total class objects that were correctly classified by the classifier. The accuracy of predicting the frames that refer to a class of a model is calculated by Equation (5):
Furthermore,
Error Rate, which indicates the quality of the classifiers towards the wrong predictions, is calculated by Equation (6):
and
Specificity, which presents the fraction of all negative samples that are correctly predicted as negative by the classifier, is calculated by Equation (7):
In the present testing, we evaluate the statistical significance of predictions between the three CNN models by calculating the test result for p. If the p-value is greater than 0.05 then no effect is observed, meaning that our test hypothesis is false and the outcomes are statistically independent. Our null hypothesis is that all the models are equivalent, so “Ho = there is no difference between the models”.
The basic format for reporting a chi-square test result is:
x
2 (degrees of freedom,
N = sample size) = chi-square statistic value,
p =
p value, while our
p-values are calculated by the Pearson’s chi-squared test, using Equation (8):
where
r is the number of rows corresponding to the number of models,
c is the number of columns corresponding to the number of classes of the contingency table,
df is the degrees of freedom calculated by
df = (
r − 1) × (
c − 1) for test of independence,
Ei,j is the “theoretical frequency” for a cell, given the hypothesis of independence, and
Oi,j is the observations of type
j ignoring the row attribute (fraction of column totals).
5. Results of CNN Models Testing
After using the formulas above for each class versus all classes and for each model, the results of the metrics when it comes to each model are presented in
Table 4,
Table 5,
Table 6 and
Table 7. For Model 1, the report is in
Table 4 and its performance analysis in
Table 7. We can observe that the model is working well for all organs, with slightly bigger error rate when it comes to the stomach. After the examination of the error that we saw in the predictions, many times this error occurs because the presence of pituitary or of saliva is misunderstood as the pituitary of the colon which is causing confusion in the model.
Next, for Model 2, the report in
Table 5 and its performance analysis in
Table 7 show that this model is the most accurate among the three, when it comes to predictions; since the error rate is improved for stomach frames prediction and it also provides the best metrics for esophagus frames prediction.
Lastly, the Model 3 report and performance metrics appear in
Table 6 and
Table 7. As we can observe, Model 3 provides the biggest error rate in stomach frames prediction among the rest of the models. Although, the predictions for esophagus and small intestine frames have better metrics using that model.
Our results of independent validation and cross validation, demonstrate that the best of our three distinct models is that of Model 2.
For the 4 × 4 contingency table of each of our CNN1, CNN2 and CNN3 models, our chi-square test results separately in a p-value ≦ 0.00001, showing that the four-class classification of each model is statistically independent.
Moreover, for the 3 × 4 contingency table comparing our three CNN models, the chi-square test results in a p-value ≦ 0.00001. It is showing that the outputs of the three four-class models are statistically independent.
6. Conclusions
By using our research as a tracking and four classes classification algorithm, physicians can successfully detect the gates of the four GI organs (and, furthermore, to recognize the digestive organ) that the capsule is passing by, in real time. The algorithms can be executed from a mobile platform that can be physically attached for the time of recording upon the patient. This can be used as a passive compass while navigating through the digestive tract, to reveal better metrics concerning each capsule design potential (i.e., to overcome palindrome movements or to obtain better insight on how to cope through them), as well as an activation control tool for a number of actions. The whole procedure will provide better accuracy per individual patient (in terms of charting topological idiosyncrasies), without any added cost (same hardware, the processing power needed should be provided by an external unit linked to the capsule). With the future advancement of technology, when sufficient electrical power will be available to ensure the independence of the capsule unit, it will become possible to embed in the capsule a microprocessor to execute the algorithm. For now, the algorithm could be executed upon an external mobile unit, as the video frames are transferred wirelessly in real time.
Author Contributions
Conceptualization, E.S.S., M.E.Z., A.A.P. (Andreas A. Polydorou), G.S.S. and I.O.V.; methodology, E.S.S., M.E.Z., A.A.P. (Andreas A. Polydorou), G.S.S. and I.O.V.; software, S.A.A., A.A.P. (Alexios A. Polydorou) and N.M.A.; validation, S.A.A., A.A.P. (Alexios A. Polydorou) and A.A.P. (Andreas A. Polydorou); investigation, G.S.S. and E.S.S.; resources, A.A.P. (Andreas A. Polydorou); data curation, S.A.A., N.M.A. and A.A.P. (Alexios A. Polydorou); writing—original draft preparation, N.M.A., E.S.S. and S.A.A.; writing—review and editing, E.S.S. supervision, E.S.S., A.A.P. (Andreas A. Polydorou), I.O.V. and M.E.Z.; project administration, E.S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study due to the explicit usage of fully anonymized data.
Informed Consent Statement
Patient consent was waived due to the explicit usage of fully anonymized data.
Data Availability Statement
The data used in this study are available on request from the corresponding authors.
Acknowledgments
The authors are grateful to the Greek Aretaeio University Hospital, Attikon University Hospital, and Laikon General Hospital, and the Aristotle University of Thessaloniki.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vulpoi, R.-A.; Luca, M.; Ciobanu, A.; Olteanu, A.; Barboi, O.-B.; Drug, V.L. Artificial intelligence in digestive endoscopy—Where are we and where are we going? Diagnostics 2022, 12, 927. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Lim, Y.J. Artificial intelligence in capsule endoscopy: A practical guide to its past and future challenges. Diagnostics 2021, 11, 1722. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Hwang, Y.; Yoon, J.H.; Park, M.G.; Kim, J.; Lim, Y.J.; Chun, H.J. Recent development of computer vision technology to improve capsule endoscopy. Clin. Endosc. 2019, 52, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Zhang, W.; Zhang, W.; Paolo, D. A novel biopsy capsule robot based on high-speed cutting tissue. Cyborg Bionic Syst. 2022, 9783517. [Google Scholar] [CrossRef]
- Fritscher-Ravens, A.; Scherbakov, P.; Bufler, P.; Torroni, F.; Ruuska, T.; Nuutinen, H.; Thomson, M.; Tabbers, M.; Milla, P. The feasibility of wireless capsule endoscopy in detecting small intestinal pathology in children under the age of 8 years: A multicentre European study. Gut 2009, 58, 1467–1472. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, G.; Calabrese, C.; Conti Nibali, R.; De Matteis, A.; Casciani, E.; Martemucci, L.; Pagliaro, G.; Pagano, N. Enteroscopy in children. United Eur. Gastroenterol. J. 2018, 6, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Nuutinen, H.; Kolho, K.L.; Salminen, P.; Rintala, R.; Koskenpato, J.; Koivusalo, A.; Sipponen, T.; Färkkilä, M. Capsule endoscopy in pediatric patients: Technique and results in our first 100 consecutive children. Scand. J. Gastroenterol. 2011, 46, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Taghiakbari, M.; Mori, Y.; Renteln, D. Artificial intelligence-assisted colonoscopy: A review of current state of practice and research. World J. Gastroenterol. 2021, 27, 8103–8122. [Google Scholar] [CrossRef] [PubMed]
- Barbagiannis, C.; Polydorou, A.; Zervakis, M.; Polydorou, A.; Sergaki, E. Detection of angioectasias and haemorrhages incorporated into a multi-class classification tool for the GI tract anomalies by using binary CNNs. J. Biomed. Sci. Eng. 2021, 14, 402–414. [Google Scholar] [CrossRef]
- Polydorou, A.; Sergaki, E.; Polydorou, A.; Barbagiannis, C.; Vardiambasis, I.; Giakos, G.; Zervakis, M. Improving CAD hemorrhage detection in capsule endoscopy. J. Biomed. Sci. Eng. 2021, 14, 103–118. [Google Scholar] [CrossRef]
- Pannu, H.S.; Ahuja, S.; Dang, N.; Soni, S.; Malhi, A.K. Deep learning based image classification for intestinal hemorrhage. Multimed Tools 2020, 79, 21941–21966. [Google Scholar] [CrossRef]
- Yu, J.; Chen, L.; Xiang, Z.Q.; Zou, Y. A hybrid convolutional neural networks with extreme learning machine for WCE image classification. In Proceedings of the 2015 IEEE International Conference on Robotics and Biomimetics (ROBIO), Zhuhai, China, 6–9 December 2015. [Google Scholar]
- Oleksy, P.; Januszkiewicz, L. Wireless capsule endoscope localization with phase detection algorithm and adaptive body model. Sensors 2022, 22, 2200. [Google Scholar] [CrossRef] [PubMed]
- Vedaei, S.S.; Wahid, K.A. A localization method for wireless capsule endoscopy using side wall cameras and IMU sensor. Sci. Rep. 2021, 11, 11204. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Li, L.; Wang, Y.; Yu, J.; Li, Y.; Deng, W.J. Classifying digestive organs in wireless capsule endoscopy images based on deep convolutional neural network. In Proceedings of the 2015 International Conference on Digital Signal Processing (DSP), Singapore, 21–24 July 2015. [Google Scholar]
- Son, G.; Eo, T.; An, J.; Oh, D.J.; Shin, Y.; Rha, H.; Kim, Y.J.; Lim, Y.J.; Hwang, D. Small bowel detection for wireless capsule endoscopy using convolutional neural networks with temporal filtering. Diagnostics 2022, 12, 1858. [Google Scholar] [CrossRef] [PubMed]
- Odagawa, M.; Okamoto, T.; Koide, T.; Yoshida, S.; Mieno, H.; Tamaki, T.; Raytchev, B.; Kaneda, K.; Tanaka, S. Classification method with CNN features and SVM for computer-aided diagnosis system in colorectal magnified NBI endoscopy. In Proceedings of the 2020 IEEE Region 10 Conference (TENCON), Osaka, Japan, 16–19 November 2020; pp. 1095–1100. [Google Scholar]
- Tam, A. Training-validation-test split and cross-validation done right. Mach. Learn. Mastery 2021, 23. [Google Scholar]
- Alharbi, F.; Alharbi, A.; Kamioka, E. Animal species classification using machine learning techniques. In Proceedings of the 2018 International Joint Conference on Metallurgical and Materials Engineering (JCMME 2018), Wellington, New Zealand, 10–12 December 2018. [Google Scholar]
- Deng, J.; Dong, W.; Socher, R.; Li, L.-J.; Li, K.; Fei-Fei, L. ImageNet: A large-scale hierarchical image database. In Proceedings of the 2009 IEEE Conference on Computer Vision and Pattern Recognition, Miami, FL, USA, 20–25 June 2009. [Google Scholar]
- Chicco, D.; Jurman, G. The advantages of the Matthews correlation coefficient (MCC) over F1 score and accuracy in binary classification evaluation. BMC Genom. 2020, 21, 6. [Google Scholar] [CrossRef] [PubMed]
- Grandini, M.; Bagli, E.; Visani, G. Metrics for Multi-Class Classification: An Overview. 2020. Available online: https://arxiv.org/abs/2008.05756 (accessed on 10 January 2022).
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).