Highly Specific and Rapid Detection of Hepatitis C Virus Using RT-LAMP-Coupled CRISPR–Cas12 Assay
Abstract
:1. Introduction
2. Materials and Methods
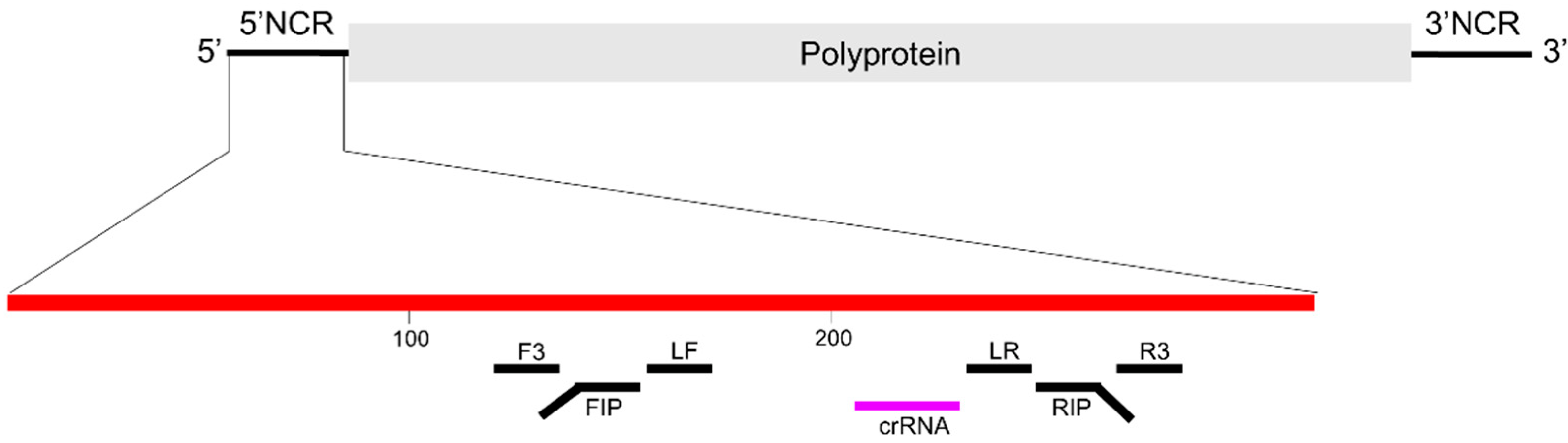
2.1. Design of CRISPR RNA (crRNA)
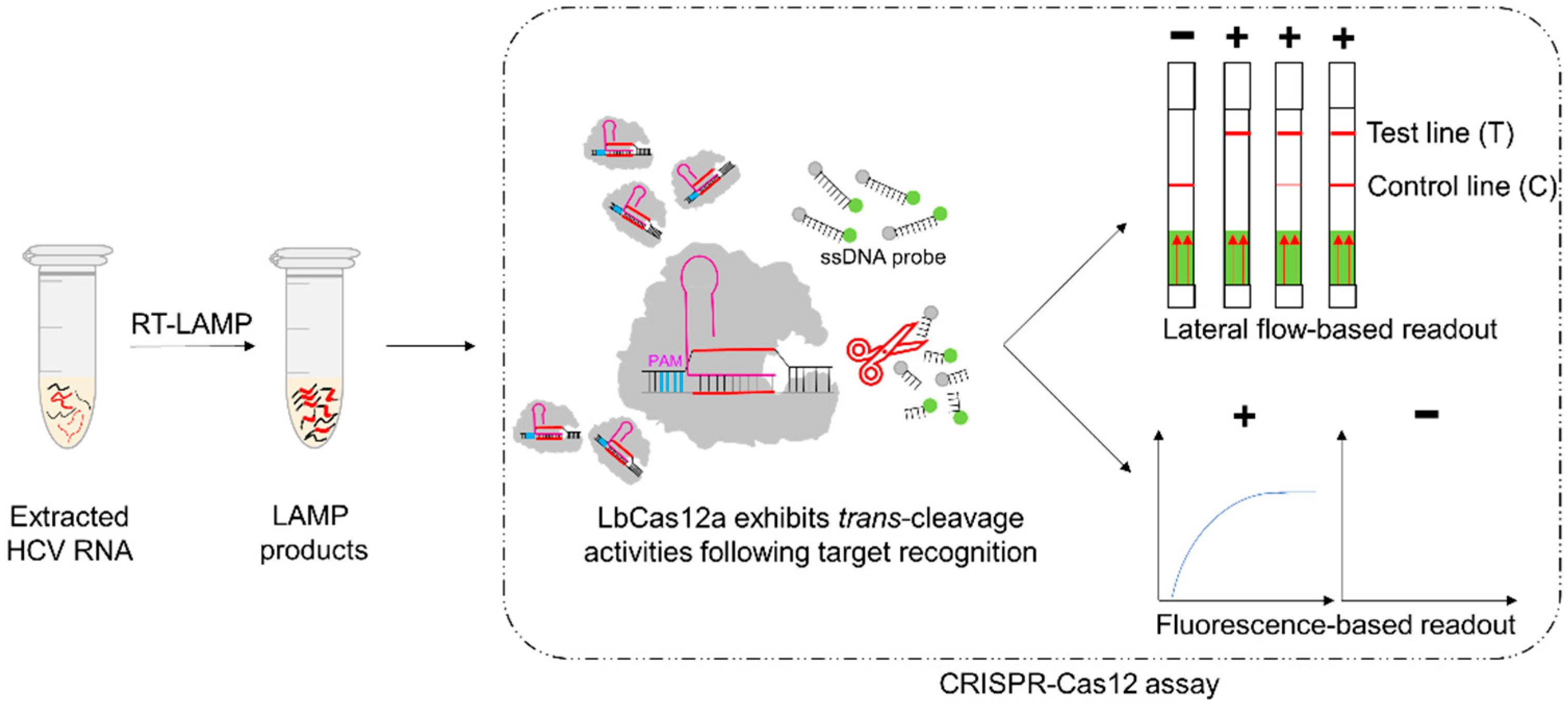
2.2. Development of the RT-LAMP-Coupled CRISPR–Cas12 Assays
2.2.1. RT-LAMP Reaction
2.2.2. CRISPR–Cas12 Assay with Lateral Flow-Based Readout
2.2.3. CRISPR–Cas12 Assay with Fluorescence-Based Readout
2.3. Evaluation with Clinical Samples
2.4. Ethical and Biosafety Statements
2.5. Statistical Analysis
3. Results
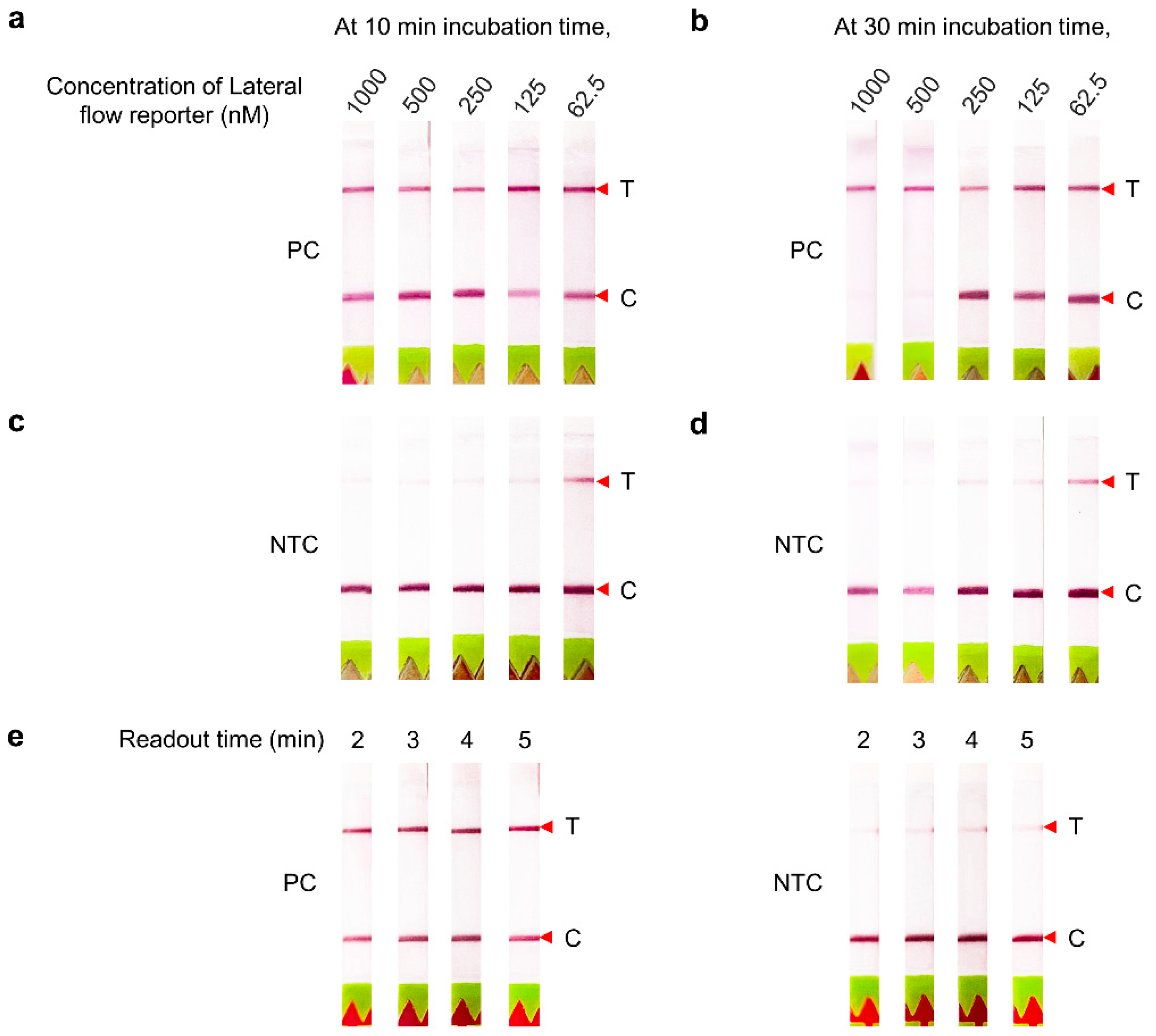
3.1. Optimization of the CRISPR–Cas12 Assay Conditions with Lateral Flow-Based Readout for HCV RNA Detection
3.2. Clinical Evaluation of RT-LAMP-Coupled CRISPR–Cas12 Assay for HCV RNA Detection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Hepatitis C. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-c (accessed on 12 April 2022).
- Rabaan, A.A.; Al-Ahmed, S.H.; Bazzi, A.M.; Alfouzan, W.A.; Alsuliman, S.A.; Aldrazi, F.A.; Haque, S. Overview of hepatitis C infection, molecular biology, and new treatment. J. Infect. Public Health 2020, 13, 773–783. [Google Scholar] [CrossRef] [PubMed]
- Holmes, J.A.; Rutledge, S.M.; Chung, R.T. Direct-acting antiviral treatment for hepatitis C. Lancet 2019, 393, 1392–1394. [Google Scholar] [CrossRef]
- Omata, M.; Kanda, T.; Wei, L.; Yu, M.-L.; Chuang, W.-L.; Ibrahim, A.; Lesmana, C.R.A.; Sollano, J.; Kumar, M.; Jindal, A.; et al. APASL consensus statements and recommendations for hepatitis C prevention, epidemiology, and laboratory testing. Hepatol. Int. 2016, 10, 681–701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, N.; Liu, J.; Sun, D. Detection of HCV genotypes 1b and 2a by a reverse transcription loop-mediated isothermal amplification assay. J. Med. Virol. 2017, 89, 1048–1054. [Google Scholar] [CrossRef]
- Hongjaisee, S.; Doungjinda, N.; Khamduang, W.; Carraway, T.S.; Wipasa, J.; Debes, J.D.; Supparatpinyo, K. Rapid visual detection of hepatitis C virus using a reverse transcription loop-mediated isothermal amplification assay. Int. J. Infect. Dis. 2021, 102, 440–445. [Google Scholar] [CrossRef]
- Soroka, M.; Wasowicz, B.; Rymaszewska, A. Loop-Mediated Isothermal Amplification (LAMP): The Better Sibling of PCR? Cells 2021, 10, 1931. [Google Scholar] [CrossRef]
- Yan, C.; Cui, J.; Huang, L.; Du, B.; Chen, L.; Xue, G.; Li, S.; Zhang, W.; Zhao, L.; Sun, Y.; et al. Rapid and visual detection of 2019 novel coronavirus (SARS-CoV-2) by a reverse transcription loop-mediated isothermal amplification assay. Clin. Microbiol Infect. 2020, 26, 773–779. [Google Scholar] [CrossRef]
- Nyan, D.C.; Swinson, K.L. A method for rapid detection and genotype identification of hepatitis C virus 1-6 by one-step reverse transcription loop-mediated isothermal amplification. Int. J. Infect. Dis. 2016, 43, 30–36. [Google Scholar] [CrossRef] [Green Version]
- Aoki, M.N.; De Oliveira Coelho, B.; Góes, L.G.B.; Minoprio, P.; Durigon, E.L.; Morello, L.G.; Marchini, F.K.; Riediger, I.N.; Do Carmo Debur, M.; Nakaya, H.I.; et al. Colorimetric RT-LAMP SARS-CoV-2 diagnostic sensitivity relies on color interpretation and viral load. Sci. Rep. 2021, 11, 9026. [Google Scholar] [CrossRef]
- Pickar-Oliver, A.; Gersbach, C.A. The next generation of CRISPR–Cas technologies and applications. Nat. Rev. Mol. Cell Biol. 2019, 20, 490–507. [Google Scholar] [CrossRef]
- Nidhi, S.; Anand, U.; Oleksak, P.; Tripathi, P.; Lal, J.A.; Thomas, G.; Kuca, K.; Tripathi, V. Novel CRISPR-Cas Systems: An Updated Review of the Current Achievements, Applications, and Future Research Perspectives. Int. J. Mol. Sci. 2021, 22, 3327. [Google Scholar] [CrossRef] [PubMed]
- Westra, E.R.; Dowling, A.J.; Broniewski, J.M.; Houte, S.V. Evolution and Ecology of CRISPR. Annu. Rev. Ecol. Evol. Syst. 2016, 47, 307–331. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.S.; Ma, E.; Harrington, L.B.; Da Costa, M.; Tian, X.; Palefsky, J.M.; Doudna, J.A. CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science 2018, 360, 436–439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.Y.; Cheng, Q.X.; Liu, J.K.; Nie, X.Q.; Zhao, G.P.; Wang, J. CRISPR-Cas12a has both cis- and trans-cleavage activities on single-stranded DNA. Cell Res. 2018, 28, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Broughton, J.P.; Deng, X.; Yu, G.; Fasching, C.L.; Servellita, V.; Singh, J.; Miao, X.; Streithorst, J.A.; Granados, A.; Sotomayor-Gonzalez, A.; et al. CRISPR-Cas12-based detection of SARS-CoV-2. Nat. Biotechnol. 2020, 38, 870–874. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Zhao, W.; Zhao, W.; Si, Y.; Chen, N.; Chen, X.; Zhang, X.; Fan, L.; Sui, G. Universally Stable and Precise CRISPR-LAMP Detection Platform for Precise Multiple Respiratory Tract Virus Diagnosis Including Mutant SARS-CoV-2 Spike N501Y. Anal. Chem. 2021, 93, 16184–16193. [Google Scholar] [CrossRef]
- Kuiken, C.; Yusim, K.; Boykin, L.; Richardson, R. The Los Alamos hepatitis C sequence database. Bioinformatics 2005, 21, 379–384. [Google Scholar] [CrossRef]
- Bai, J.; Lin, H.; Li, H.; Zhou, Y.; Liu, J.; Zhong, G.; Wu, L.; Jiang, W.; Du, H.; Yang, J.; et al. Cas12a-Based On-Site and Rapid Nucleic Acid Detection of African Swine Fever. Front. Microbiol. 2019, 10, 2830. [Google Scholar] [CrossRef] [Green Version]
- Nagai, K.; Horita, N.; Yamamoto, M.; Tsukahara, T.; Nagakura, H.; Tashiro, K.; Shibata, Y.; Watanabe, H.; Nakashima, K.; Ushio, R.; et al. Diagnostic test accuracy of loop-mediated isothermal amplification assay for Mycobacterium tuberculosis: Systematic review and meta-analysis. Sci. Rep. 2016, 6, 39090. [Google Scholar] [CrossRef]
- Swarts, D.C.; Van der Oost, J.; Jinek, M. Structural basis for guide RNA processing and seed-dependent DNA targeting by CRISPR-Cas12a. Mol. Cell 2017, 66, 221–233. [Google Scholar] [CrossRef] [Green Version]
- Ding, R.; Long, J.; Yuan, M.; Zheng, X.; Shen, Y.; Jin, Y.; Yang, H.; Li, H.; Chen, S.; Duan, G. CRISPR/Cas12-Based Ultra-Sensitive and Specific Point-of-Care Detection of HBV. Int. J. Mol. Sci. 2021, 22, 4842. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Zhang, J.; Yang, Z.; Mou, Q.; Ma, Y.; Xiong, Y.; Lu, Y. Functional DNA Regulated CRISPR-Cas12a Sensors for Point-of-Care Diagnostics of Non-Nucleic-Acid Targets. J. Am. Chem. Soc. 2020, 142, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Samacoits, A.; Nimsamer, P.; Mayuramart, O.; Chantaravisoot, N.; Sitthi-Amorn, P.; Nakhakes, C.; Luangkamchorn, L.; Tongcham, P.; Zahm, U.; Suphanpayak, S.; et al. Machine Learning-Driven and Smartphone-Based Fluorescence Detection for CRISPR Diagnostic of SARS-CoV-2. ACS Omega 2021, 6, 2727–2733. [Google Scholar] [CrossRef] [PubMed]
- Bonini, A.; Poma, N.; Vivaldi, F.; Kirchhain, A.; Salvo, P.; Bottai, D.; Tavanti, A.; Di Francesco, F. Advances in biosensing: The CRISPR/Cas system as a new powerful tool for the detection of nucleic acids. J. Pharm. Biomed. Anal. 2021, 192, 113645. [Google Scholar] [CrossRef]
- Qi, H.; Hu, Z.; Yang, Z.; Zhang, J.; Wu, J.J.; Cheng, C.; Wang, C.; Zheng, L. Capacitive Aptasensor Coupled with Microfluidic Enrichment for Real-Time Detection of Trace SARS-CoV-2 Nucleocapsid Protein. Anal. Chem. 2022, 94, 2812–2819. [Google Scholar] [CrossRef]
- Zhang, Z.; Lou, Y.; Guo, C.; Jia, Q.; Song, Y.; Tian, J.-Y.; Zhang, S.; Wang, M.; He, L.; Du, M. Metal–organic frameworks (MOFs) based chemosensors/biosensors for analysis of food contaminants. Trends Food Sci. Technol. 2021, 118, 569–588. [Google Scholar] [CrossRef]
- Petruzziello, A.; Marigliano, S.; Loquercio, G.; Cozzolino, A.; Cacciapuoti, C. Global epidemiology of hepatitis C virus infection: An up-date of the distribution and circulation of hepatitis C virus genotypes. World J. Gastroenterol. 2016, 22, 7824–7840. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, Y.; Fohlerova, Z.; Chang, H.; Iliescu, C.; Neuzil, P. LAMP-on-a-chip: Revising microfluidic platforms for loop-mediated DNA amplification. Trends Analyt. Chem. 2019, 113, 44–53. [Google Scholar] [CrossRef]


| Assays | RT-LAMP-Coupled CRISPR–Cas12 Assay | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| (1st Round) | (2nd Round) | ||||||||
| Lateral Flow-Based Readout | Fluorescence-Based Readout | Lateral Flow-Based Readout | Fluorescence-Based Readout | ||||||
| Positive (%) | Negative (%) | Positive (%) | Negative (%) | Positive (%) | Negative (%) | Positive (%) | Negative (%) | ||
| Real-time RT-PCR | Detectable (N = 100 a) | 93 (93) | 7 (7) | 93 (93) | 7 (7) | 96 (96) | 4 (4) | 96 (96) | 4 (4) |
| Undetectable (N = 30 b) | 0 (0) | 30 (100) | 0 (0) | 30 (100) | ND | ND | ND | ND | |
| HCV Viral Load (Log10 IU/mL) | All Samples | Genotype 1 | Genotype 3 | Genotype 4 | Genotype 6 |
|---|---|---|---|---|---|
| n/N (%) | n/N (%) | n/N (%) | n/N (%) | n/N (%) | |
| 7.01–8.00 | 15/15 (100) | 7/7 (100) | 5/5 (100) | NA | 3/3 (100) |
| 6.01–7.00 | 34/35 (97) | 15/16 (94) | 12/12 (100) | NA | 7/7 (100) |
| 5.01–6.00 | 24/24 (100) | 10/10 (100) | 10/10 (100) | 1/1 (100) | 3/3 (100) |
| 4.01–5.00 | 21/23 (91) | 9/10 (90) | 10/11 (91) | NA | 2/2 (100) |
| 3.01–4.00 | 2/3 (67) | 2/2 (100) | 0/1 (0) | NA | NA |
| Total | 96/100 (96) | 43/45 (96) | 37/39 (95) | 1/1 (100) | 15/15 (100) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kham-Kjing, N.; Ngo-Giang-Huong, N.; Tragoolpua, K.; Khamduang, W.; Hongjaisee, S. Highly Specific and Rapid Detection of Hepatitis C Virus Using RT-LAMP-Coupled CRISPR–Cas12 Assay. Diagnostics 2022, 12, 1524. https://doi.org/10.3390/diagnostics12071524
Kham-Kjing N, Ngo-Giang-Huong N, Tragoolpua K, Khamduang W, Hongjaisee S. Highly Specific and Rapid Detection of Hepatitis C Virus Using RT-LAMP-Coupled CRISPR–Cas12 Assay. Diagnostics. 2022; 12(7):1524. https://doi.org/10.3390/diagnostics12071524
Chicago/Turabian StyleKham-Kjing, Nang, Nicole Ngo-Giang-Huong, Khajornsak Tragoolpua, Woottichai Khamduang, and Sayamon Hongjaisee. 2022. "Highly Specific and Rapid Detection of Hepatitis C Virus Using RT-LAMP-Coupled CRISPR–Cas12 Assay" Diagnostics 12, no. 7: 1524. https://doi.org/10.3390/diagnostics12071524
APA StyleKham-Kjing, N., Ngo-Giang-Huong, N., Tragoolpua, K., Khamduang, W., & Hongjaisee, S. (2022). Highly Specific and Rapid Detection of Hepatitis C Virus Using RT-LAMP-Coupled CRISPR–Cas12 Assay. Diagnostics, 12(7), 1524. https://doi.org/10.3390/diagnostics12071524







