Prognostic Value of Perineural Invasion on Survival and Recurrence in Oral Squamous Cell Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Statistics
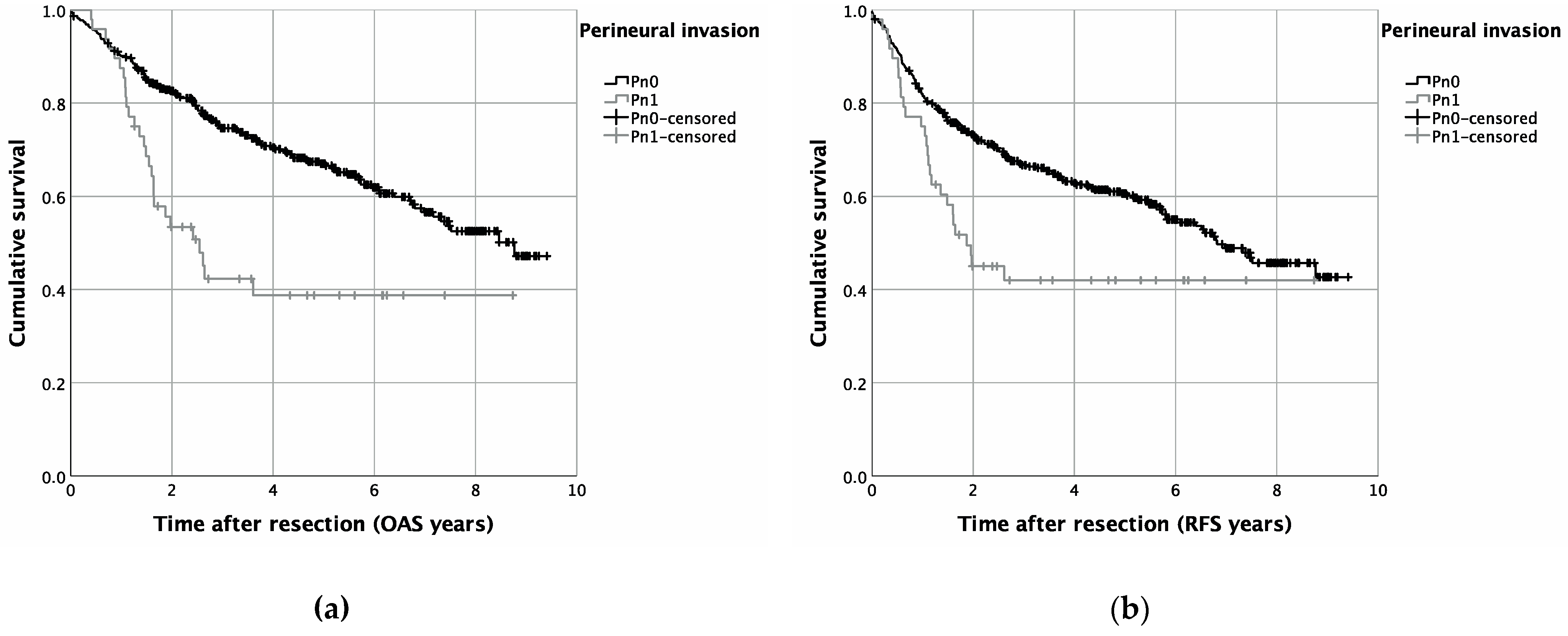
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bonifazi, M.; Malvezzi, M.; Bertuccio, P.; Edefonti, V.; Garavello, W.; Levi, F.; La Vecchia, C.; Negri, E. Age-period-cohort analysis of oral cancer mortality in Europe: The end of an epidemic? Oral Oncol. 2011, 47, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Burtness, B.; Harrington, K.J.; Greil, R.; Soulières, D.; Tahara, M.; de Castro, G., Jr.; Psyrri, A.; Basté, N.; Neupane, P.; Bratland, Å.; et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): A randomised, open-label, phase 3 study. Lancet 2019, 394, 1915–1928. [Google Scholar] [CrossRef]
- Keerthi, R.; Dutta, A.; Agarwal, S.; Kani, V.; Khatua, A. Perineural Invasion of Oral Squamous Cell Carcinoma: A New Hurdle for Surgeons. J. Maxillofac. Oral Surg. 2018, 17, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Chatzistefanou, I.; Lubek, J.; Markou, K.; Ord, R.A. The role of perineural invasion in treatment decisions for oral cancer patients: A review of the literature. J. Cranio-Maxillo-Facial Surg. Off. Publ. Eur. Assoc. Cranio-Maxillo-Facial Surg. 2017, 45, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, K.A.; Hoffman, H.T.; Zimmerman, M.B.; Robinson, R.A. Perineural and vascular invasion in oral cavity squamous carcinoma: Increased incidence on re-review of slides and by using immunohistochemical enhancement. Arch. Pathol. Lab. Med. 2005, 129, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Nair, D.; Mair, M.; Singhvi, H.; Mishra, A.; Nair, S.; Agrawal, J.; Chaturvedi, P. Perineural invasion: Independent prognostic factor in oral cancer that warrants adjuvant treatment. Head Neck 2018, 40, 1780–1787. [Google Scholar] [CrossRef]
- Gaddikeri, S. Perineural Invasion of Skin Cancers in the Head and Neck: An Uncommon Phenomenon Revisited. Otolaryngology 2014, 4, 2. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.W.; Yu, E.H.; Wu, T.H.; Lo, W.L.; Li, W.Y.; Kao, S.Y. Histopathological factors affecting nodal metastasis in tongue cancer: Analysis of 94 patients in Taiwan. Int. J. Oral Maxillofac. Surg. 2008, 37, 912–916. [Google Scholar] [CrossRef] [PubMed]
- Woolgar, J.A.; Scott, J. Prediction of cervical lymph node metastasis in squamous cell carcinoma of the tongue/floor of mouth. Head Neck 1995, 17, 463–472. [Google Scholar] [CrossRef]
- Amit, M.; Na’ara, S.; Gil, Z. Mechanisms of cancer dissemination along nerves. Nat. Rev. Cancer 2016, 16, 399–408. [Google Scholar] [CrossRef]
- Bakst, R.L.; Wong, R.J. Mechanisms of Perineural Invasion. J. Neurol. Surg. Part B Skull Base 2016, 77, 96–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misztal, C.I.; Green, C.; Mei, C.; Bhatia, R.; Velez Torres, J.M.; Kamrava, B.; Moon, S.; Nicolli, E.; Weed, D.; Sargi, Z.; et al. Molecular and Cellular Mechanisms of Perineural Invasion in Oral Squamous Cell Carcinoma: Potential Targets for Therapeutic Intervention. Cancers 2021, 13, 6011. [Google Scholar] [CrossRef]
- Sobin, L.H.; Gospodarowicz, M.K.; Wittekind, C.; International Union against Cancer. TNM Classification of Malignant Tumours, 7th ed.; Sobin, L.H., Gospodarowicz, M.K., Wittekind, C., Eds.; Wiley-Blackwell: Chichester, UK; West Sussex, UK; Hoboken, NJ, USA, 2010. [Google Scholar]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Spanier, G.; Böttcher, J.; Gerken, M.; Fischer, R.; Roth, G.; Lehn, P.; Klingelhöffer, C.; Meier, J.K.; Fraccaroli, A.; Tischer, J.; et al. Prognostic value of perioperative red blood cell transfusion and anemia on survival and recurrence in oral squamous cell carcinoma. Oral Oncol. 2020, 107, 104773. [Google Scholar] [CrossRef] [PubMed]
- Liebig, C.; Ayala, G.; Wilks, J.A.; Berger, D.H.; Albo, D. Perineural invasion in cancer: A review of the literature. Cancer 2009, 115, 3379–3391. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, A.; Panda, S.; Mohanty, N.; Jena, D.; Mishra, N.; Surabhi; Baisakh, M.R. Perinerural, lymphovascular and depths of invasion in extrapolating nodal metastasis in oral cancer. Clin. Oral. Investig. 2020, 24, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Spoerl, S.; Gerken, M.; Fischer, R.; Mamilos, A.; Spoerl, S.; Wolf, S.; Pohl, F.; Klingelhoffer, C.; Ettl, T.; Reichert, T.E.; et al. Lymphatic and vascular invasion in oral squamous cell carcinoma: Implications for recurrence and survival in a population-based cohort study. Oral Oncol. 2020, 111, 105009. [Google Scholar] [CrossRef] [PubMed]
- Varsha, B.K.; Radhika, M.B.; Makarla, S.; Kuriakose, M.A.; Kiran, G.S.; Padmalatha, G.V. Perineural invasion in oral squamous cell carcinoma: Case series and review of literature. J. Oral Maxillofac. Pathol. 2015, 19, 335–341. [Google Scholar] [CrossRef]
- Schmitd, L.B.; Scanlon, C.S.; D’Silva, N.J. Perineural Invasion in Head and Neck Cancer. J. Dent. Res. 2018, 97, 742–750. [Google Scholar] [CrossRef]
- Matsushita, Y.; Yanamoto, S.; Takahashi, H.; Yamada, S.; Naruse, T.; Sakamoto, Y.; Ikeda, H.; Shiraishi, T.; Fujita, S.; Ikeda, T.; et al. A clinicopathological study of perineural invasion and vascular invasion in oral tongue squamous cell carcinoma. Int. J. Oral Maxillofac. Surg. 2015, 44, 543–548. [Google Scholar] [CrossRef] [Green Version]
- Batsakis, J.G. Nerves and neurotropic carcinomas. Ann. Otol. Rhinol. Laryngol. 1985, 94 Pt 1, 426–427. [Google Scholar] [PubMed]
- Chi, A.C.; Katabi, N.; Chen, H.S.; Cheng, Y.L. Interobserver Variation Among Pathologists in Evaluating Perineural Invasion for Oral Squamous Cell Carcinoma. Head Neck Pathol. 2016, 10, 451–464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conte, G.A.; Qari, O.; Fasano, G.A.; Guinto, R.K.; Palo, L.; Parker, G.S.; Rangwala, A.F.; Minassian, H.; Greenberg, P.J.; Dewan, A.A.; et al. S100 Staining Adds to the Prognostic Significance of the Combination of Perineural Invasion and Lymphovascular Invasion in Colorectal Cancer. Appl. Immunohistochem. Mol. Morphol. AIMM 2020, 28, 354–359. [Google Scholar] [CrossRef]
- Shen, W.R.; Wang, Y.P.; Chang, J.Y.; Yu, S.Y.; Chen, H.M.; Chiang, C.P. Perineural invasion and expression of nerve growth factor can predict the progression and prognosis of oral tongue squamous cell carcinoma. J. Oral Pathol. Med. Off. Publ. Int. Assoc. Oral Pathol. Am. Acad. Oral Pathol. 2014, 43, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Laske, R.D.; Scholz, I.; Ikenberg, K.; Meerwein, C.; Vital, D.G.; Studer, G.; Rössle, M.; Huber, G.F. Perineural Invasion in Squamous Cell Carcinoma of the Oral Cavity: Histology, Tumor Stage, and Outcome. Laryngoscope Investig. Otolaryngol. 2016, 1, 13–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tarsitano, A.; Tardio, M.L.; Marchetti, C. Impact of perineural invasion as independent prognostic factor for local and regional failure in oral squamous cell carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Rahima, B.; Shingaki, S.; Nagata, M.; Saito, C. Prognostic significance of perineural invasion in oral and oropharyngeal carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 97, 423–431. [Google Scholar] [CrossRef]
- Blackburn, T.K.; Bakhtawar, S.; Brown, J.S.; Lowe, D.; Vaughan, E.D.; Rogers, S.N. A questionnaire survey of current UK practice for adjuvant radiotherapy following surgery for oral and oropharyngeal squamous cell carcinoma. Oral Oncol. 2007, 43, 143–149. [Google Scholar] [CrossRef]
| Category Group | Perineural Invasion | |||||||
|---|---|---|---|---|---|---|---|---|
| Pn0 | Pn1 | Total | χ2 | |||||
| N | (%) | N | (%) | N | (%) | p | ||
| Gender | Female | 133 | 29.9% | 12 | 25.0% | 145 | 29.4% | 0.480 |
| Male | 312 | 70.1% | 36 | 75.0% | 348 | 70.6% | ||
| Age at diagnosis | <50 | 48 | 10.8% | 5 | 10.4% | 53 | 10.8% | 0.983 |
| 50.0–59.9 | 156 | 35.1% | 17 | 35.4% | 173 | 35.1% | ||
| 60.0–69.9 | 143 | 32.1% | 14 | 29.2% | 157 | 31.8% | ||
| 70.0–79.9 | 77 | 17.3% | 9 | 18.8% | 86 | 17.4% | ||
| 80.0+ | 21 | 4.7% | 3 | 6.3% | 24 | 4.9% | ||
| Age-adjusted CCI | 0 | 36 | 8.1% | 5 | 10.4% | 41 | 8.3% | 0.724 |
| 1 and 2 | 190 | 42.7% | 22 | 45.8% | 212 | 43.0% | ||
| 3+ | 219 | 49.2% | 21 | 43.8% | 240 | 48.7% | ||
| Positive anmnesis smoking | No | 119 | 26.7% | 10 | 20.8% | 129 | 26.2% | 0.376 |
| Yes | 326 | 73.3% | 38 | 79.2% | 364 | 73.8% | ||
| Positive anmnesis alcohol | No | 146 | 32.8% | 13 | 27.1% | 159 | 32.3% | 0.420 |
| Yes | 299 | 67.2% | 35 | 72.9% | 334 | 67.7% | ||
| Anatomical tumor site | Buccal mucosa | 33 | 7.4% | 1 | 2.1% | 34 | 6.9% | 0.332 |
| Upper alveolus and gingiva | 19 | 4.3% | 1 | 2.1% | 20 | 4.1% | ||
| Lower alveolus and gingiva | 71 | 16.0% | 5 | 10.4% | 76 | 15.4% | ||
| Hard palate | 32 | 7.2% | 2 | 4.2% | 34 | 6.9% | ||
| Tongue | 132 | 29.7% | 16 | 33.3% | 148 | 30.0% | ||
| Floor of mouth | 158 | 35.5% | 23 | 47.9% | 181 | 36.7% | ||
| Tumor size | T1 | 183 | 41.1% | 8 | 16.7% | 191 | 38.7% | 0.002 |
| T2 | 141 | 31.7% | 16 | 33.3% | 157 | 31.8% | ||
| T3 | 37 | 8.3% | 9 | 18.8% | 46 | 9.3% | ||
| T4 | 84 | 18.9% | 15 | 31.3% | 99 | 20.1% | ||
| Cervical lymph node metastasis | N0 | 286 | 64.3% | 17 | 35.4% | 303 | 61.5% | <0.001 |
| N1 | 60 | 13.5% | 6 | 12.5% | 66 | 13.4% | ||
| N2/3 | 99 | 22.2% | 25 | 52.1% | 124 | 25.2% | ||
| Extranodal spread | No | 108 | 24.3% | 18 | 37.5% | 126 | 25.6% | <0.001 |
| Yes | 50 | 11.2% | 13 | 27.1% | 63 | 12.8% | ||
| Not applicable | 287 | 64.5% | 17 | 35.4% | 304 | 61.7% | ||
| Grading | G1 | 35 | 7.9% | 1 | 2.1% | 36 | 7.3% | 0.192 |
| G2 | 324 | 72.8% | 34 | 70.8% | 358 | 72.6% | ||
| G3/4 | 86 | 19.3% | 13 | 27.1% | 99 | 20.1% | ||
| Lymphatic invasion | L0 | 399 | 89.7% | 27 | 56.3% | 426 | 86.4% | <0.001 |
| L1 | 46 | 10.3% | 21 | 43.8% | 67 | 13.6% | ||
| Vascular invasion | V0 | 435 | 97.8% | 44 | 91.7% | 479 | 97.2% | 0.016 |
| V1 | 10 | 2.2% | 4 | 8.3% | 14 | 2.8% | ||
| UICC stage | I | 144 | 32.4% | 3 | 6.3% | 147 | 29.8% | <0.001 |
| II | 78 | 17.5% | 8 | 16.7% | 86 | 17.4% | ||
| III | 69 | 15.5% | 4 | 8.3% | 73 | 14.8% | ||
| IV | 154 | 34.6% | 33 | 68.8% | 187 | 37.9% | ||
| Adjuvant therapy | No | 277 | 62.2% | 8 | 16.7% | 285 | 57.8% | <0.001 |
| Radiotherapy | 109 | 24.5% | 24 | 50.0% | 133 | 27.0% | ||
| Radio-chemotherapy | 59 | 13.3% | 16 | 33.3% | 75 | 15.2% | ||
| Life status | Alive | 295 | 66.3% | 21 | 43.8% | 316 | 64.1% | 0.002 |
| Deceased | 150 | 33.7% | 27 | 56.3% | 177 | 35.9% | ||
| Death/recurrence | Alive without recurrence | 263 | 59.1% | 21 | 43.8% | 284 | 57.6% | |
| Death or recurrence | 182 | 40.9% | 27 | 56.3% | 209 | 42.4% | 0.041 | |
| Total | 445 | 100.0% | 48 | 100.0% | 493 | 100.0% | ||
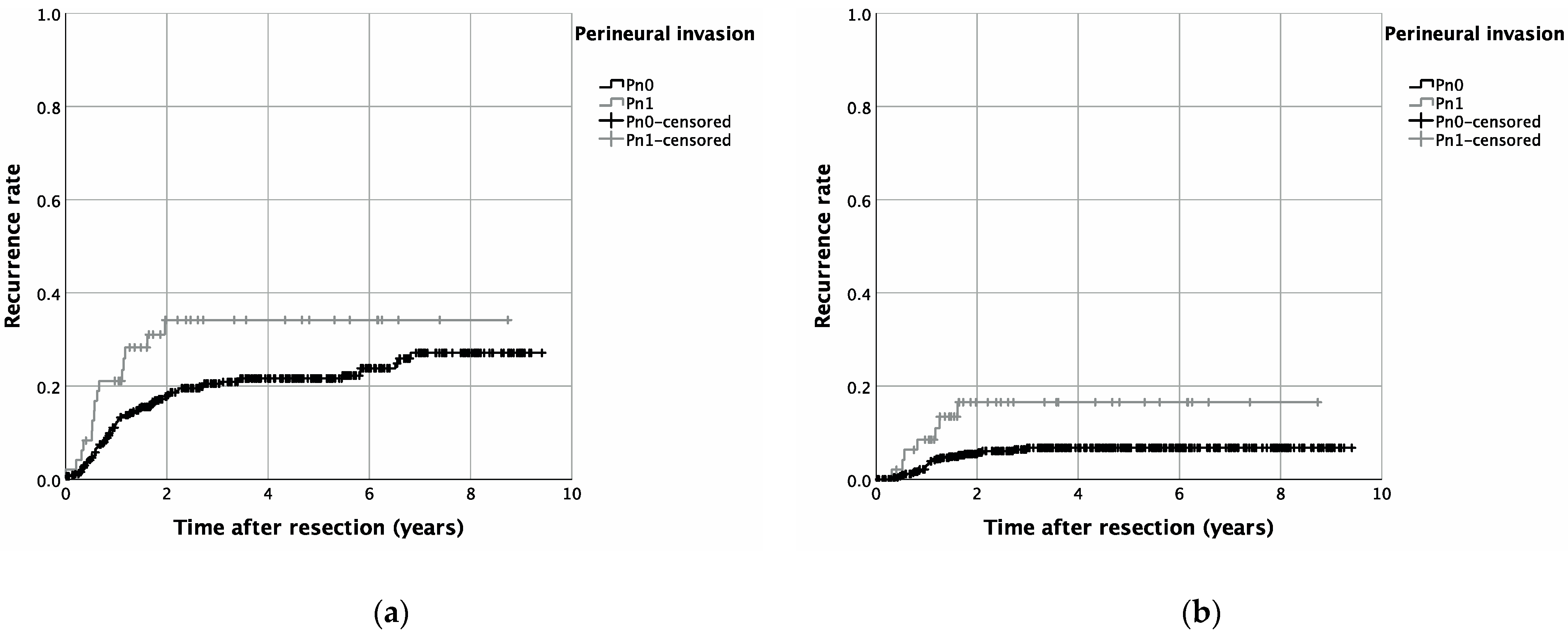
| Category | Group | N (Included Patients) | N (Recurrent Disease) | 5-Year-Recurrence Rate | Log-Rank p |
|---|---|---|---|---|---|
| Cumulative recurrence rate | Pn0 + Pn1 | 493 | 106 | 0.027 | |
| Pn0 | 445 | 91 | 21.6% | ||
| Pn1 | 48 | 15 | 34.2% | ||
| Local recurrence rate | Pn0 + Pn1 | 493 | 67 | 0.134 | |
| Pn0 | 445 | 58 | 13.8% | ||
| Pn1 | 48 | 9 | 21.1% | ||
| Locoregional recurrence rate | Pn0 + Pn1 | 493 | 48 | 0.303 | |
| Pn0 | 445 | 42 | 10.5% | ||
| Pn1 | 48 | 6 | 17.1% | ||
| Distant metastasis recurrence rate | Pn0 + Pn1 | 493 | 33 | 0.011 | |
| Pn0 | 445 | 26 | 6.8% | ||
| Pn1 | 48 | 7 | 16.6% |
| Overall Survival (OAS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Category | Group | Univariable Cox Regression | Multivariable Cox Regression | ||||||
| p | HR | Lower 95%-CI | Upper 95%-CI | p | HR | Lower 95%-CI | Upper 95%-CI | ||
| PNI | Pn0 | 1.000 | 1.000 | ||||||
| Pn1 | <0.001 | 2.312 | 1.530 | 3.493 | 0.009 | 1.820 | 1.164 | 2.847 | |
| Gender | Female | 1.000 | |||||||
| Male | 0.857 | 1.030 | 0.746 | 1.424 | |||||
| Age-adjusted CCI | 0 | <0.001 | 1.000 | <0.001 | 1.000 | ||||
| 1 and 2 | 0.778 | 0.915 | 0.491 | 1.704 | 0.774 | 0.912 | 0.488 | 1.707 | |
| 3+ | 0.043 | 1.854 | 1.020 | 3.369 | 0.040 | 1.877 | 1.028 | 3.430 | |
| Positive anamnesis smoking | No | 1.000 | |||||||
| Yes | 0.522 | 1.118 | 0.795 | 1.572 | |||||
| Positive anamnesis alcohol | No | 1.000 | |||||||
| Yes | 0.684 | 1.068 | 0.777 | 1.469 | |||||
| Anatomical tumor site | Upper alveolus, gingiva and hard palate | 0.23 | 1.000 | ||||||
| Tongue | 0.126 | 0.665 | 0.395 | 1.121 | |||||
| Lower alveolus, floor of mouth, buccal mucosa | 0.499 | 0.851 | 0.534 | 1.358 | |||||
| UICC stage | I/II | 1.000 | 1.000 | ||||||
| III/IV | <0.001 | 3.015 | 2.168 | 4.194 | <0.001 | 2.741 | 1.954 | 3.845 | |
| Grading | G1/2 | 1.000 | 1.000 | ||||||
| G3/4 | 0.068 | 1.395 | 0.976 | 1.993 | 0.124 | 1.328 | 0.925 | 1.906 | |
| Lymphatic invasion | L0 | 1.000 | 1.000 | ||||||
| L1 | 0.001 | 1.906 | 1.297 | 2.800 | 0.595 | 1.122 | 0.735 | 1.713 | |
| Vascular invasion | V0 | 1.000 | |||||||
| V1 | 0.036 | 2.055 | 1.049 | 4.027 | 0.185 | 1.583 | 0.803 | 3.119 | |
| Recurrence-Free Survival (RFS) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Category | Group | Univariable Cox Regression | Multivariable Cox Regression | ||||||
| p | HR | Lower 95%-CI | Upper 95%-CI | p | HR | Lower 95%-CI | Upper 95%-CI | ||
| PNI | Pn0 | 1.000 | 1.000 | ||||||
| Pn1 | 0.005 | 1.794 | 1.195 | 2.692 | 0.097 | 1.461 | 0.934 | 2.284 | |
| Gender | Female | 1.000 | |||||||
| Male | 0.894 | 1.020 | 0.758 | 1.373 | |||||
| Age-adjusted CCI | 0 | <0.001 | 1.000 | <0.001 | 1.000 | ||||
| 1 and 2 | 0.743 | 0.911 | 0.523 | 1.589 | 0.578 | 0.852 | 0.486 | 1.496 | |
| 3+ | 0.070 | 1.642 | 0.960 | 2.808 | 0.108 | 1.561 | 0.907 | 2.688 | |
| Positive anamnesis smoking | No | 1.000 | |||||||
| Yes | 0.684 | 1.066 | 0.782 | 1.454 | |||||
| Positive anamnesis alcohol | No | 1.000 | |||||||
| Yes | 0.638 | 1.072 | 0.801 | 1.436 | |||||
| Anatomical tumor site | Upper alveolus, gingiva and hard palate | 0.028 | 1.000 | 0.290 | |||||
| Tongue | 0.008 | 0.535 | 0.337 | 0.850 | 0.130 | 0.691 | 0.428 | 1.115 | |
| Lower alveolus, floor of mouth, buccal mucosa | 0.099 | 0.709 | 0.471 | 1.067 | 0.108 | 1.561 | 0.907 | 2.688 | |
| UICC stage | I/II | 1.000 | |||||||
| III/IV | <0.001 | 2.331 | 1.745 | 3.113 | <0.001 | 2.074 | 1.534 | 2.805 | |
| Grading | G1/2 | 1.000 | 1.000 | ||||||
| G3/4 | 0.058 | 1.375 | 0.989 | 1.910 | 0.136 | 1.289 | 0.923 | 1.800 | |
| Lymphatic invasion | L0 | 1.000 | 1.000 | ||||||
| L1 | 0.001 | 1.874 | 1.311 | 2.679 | 0.275 | 1.251 | 0.837 | 1.871 | |
| Vascular invasion | V0 | 1.000 | |||||||
| V1 | 0.161 | 1.614 | 0.826 | 3.152 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spoerl, S.; Spoerl, S.; Reil, S.; Gerken, M.; Ludwig, N.; Taxis, J.; Fischer, R.; Ettl, T.; Reichert, T.E.; Spanier, G. Prognostic Value of Perineural Invasion on Survival and Recurrence in Oral Squamous Cell Carcinoma. Diagnostics 2022, 12, 1062. https://doi.org/10.3390/diagnostics12051062
Spoerl S, Spoerl S, Reil S, Gerken M, Ludwig N, Taxis J, Fischer R, Ettl T, Reichert TE, Spanier G. Prognostic Value of Perineural Invasion on Survival and Recurrence in Oral Squamous Cell Carcinoma. Diagnostics. 2022; 12(5):1062. https://doi.org/10.3390/diagnostics12051062
Chicago/Turabian StyleSpoerl, Steffen, Silvia Spoerl, Stephanie Reil, Michael Gerken, Nils Ludwig, Juergen Taxis, René Fischer, Tobias Ettl, Torsten E. Reichert, and Gerrit Spanier. 2022. "Prognostic Value of Perineural Invasion on Survival and Recurrence in Oral Squamous Cell Carcinoma" Diagnostics 12, no. 5: 1062. https://doi.org/10.3390/diagnostics12051062
APA StyleSpoerl, S., Spoerl, S., Reil, S., Gerken, M., Ludwig, N., Taxis, J., Fischer, R., Ettl, T., Reichert, T. E., & Spanier, G. (2022). Prognostic Value of Perineural Invasion on Survival and Recurrence in Oral Squamous Cell Carcinoma. Diagnostics, 12(5), 1062. https://doi.org/10.3390/diagnostics12051062









