MNCD: A New Tool for Classifying Parkinson’s Disease in Daily Clinical Practice
Abstract
:1. Introduction
2. Material and Methods
3. Results
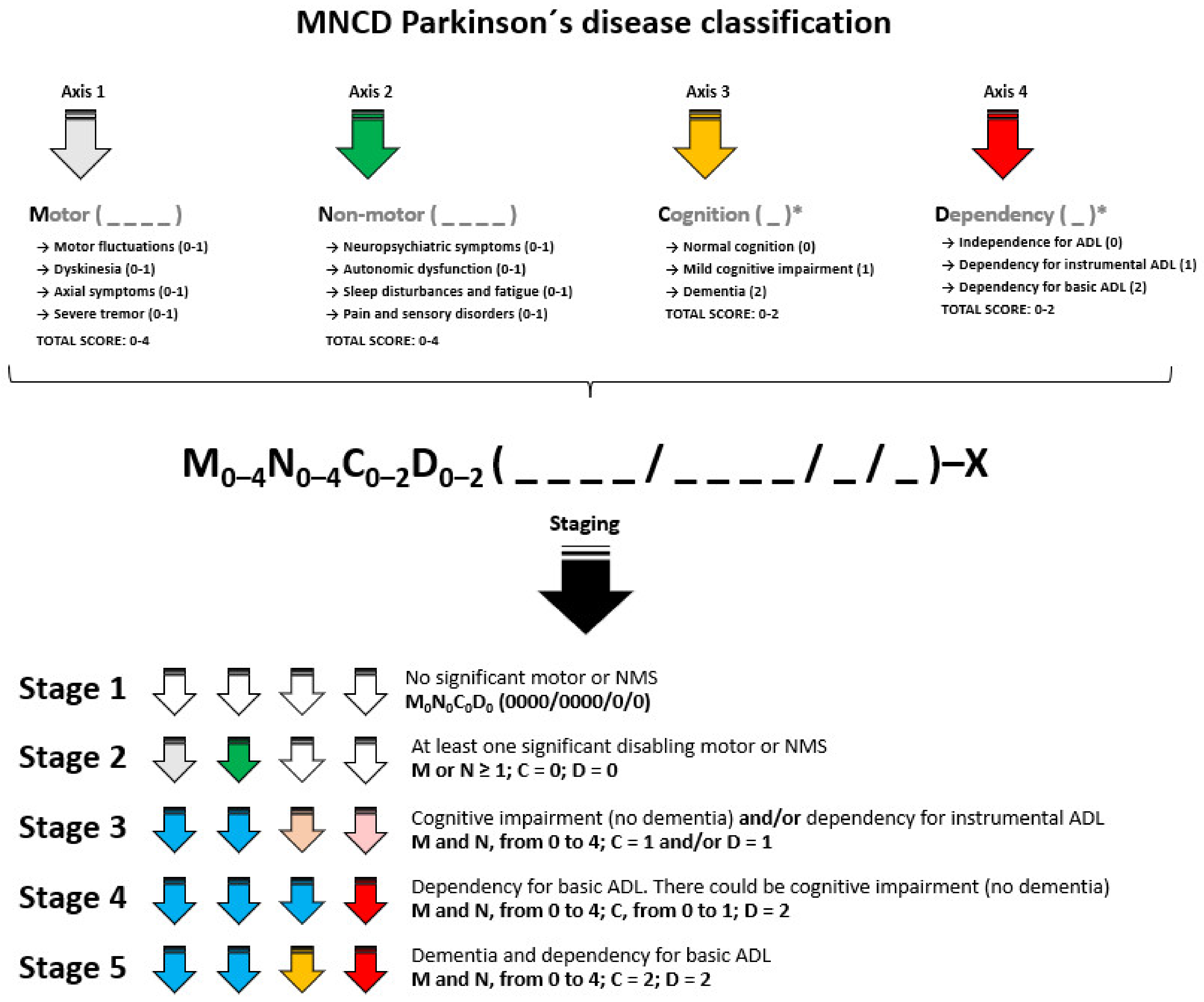
3.1. First Axis
3.2. Second Axis
3.3. Third Axis
3.4. Fourth Axis
3.5. How to Show the MNCD Classification and Interpret It
3.6. Stages of Disease According to the MNCD Classification
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Okun, M.S. Diagnosis and Treatment of Parkinson Disease: A Review. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef]
- Goetz, C.G.; Poewe, W.; Rascol, O.; Sampaio, C.; Stebbins, G.T.; Counsell, C.; Giladi, N.; Holloway, R.G.; Moore, C.G.; Wenning, G.K.; et al. Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. Movement Disorder Society Task Force report on the Hoehn and Yahr staging scale: Status and recommendations. Mov. Disord. 2004, 19, 1020–1028. [Google Scholar] [CrossRef]
- García, D.S.; de Deus Fonticoba, T.; Castro, E.S.; Borrue, C.; Mata, M.; Solano Vila, B.; Cots Foraster, A.; Alvarez Sauco, M.; Rodriguez Perez, A.; Vela, L.; et al. Non-motor symptoms burden, mood, and gait problems are the most significant factors con-tributing to a poor quality of life in non-demented Parkinson’s disease patients: Results from the COPPADIS Study Cohort. Parkinsonism Relat. Disord. 2019, 66, 151–157. [Google Scholar] [CrossRef]
- Ray Chaudhuri, K.; Rojo, J.M.; Schapira, A.H.V.; Brooks, D.J.; Stocchi, F.; Odin, P.; Antonini, A.; Brown, R.J.; Martinez-Martin, P. A proposal for a comprehensive grading of Parkinson’s disease severity combining motor and non-motor assessments: Meeting an unmet need. PLoS ONE 2013, 8, e57221. [Google Scholar]
- García, D.S.; De Deus Fonticoba, T.; Paz González, J.M.; Cores Bartolome, C.; Valdes Aymerich, L.; Munoz Enriquez, J.G.; Suarez, E.; Jesus, S.; Aguilar, M.; Pastor, P.; et al. Staging Parkinson’s Disease Combining Motor and Nonmotor Symptoms Correlates with Disability and Quality of Life. Parkinsons Dis. 2021, 2021, 8871549. [Google Scholar] [PubMed]
- Burciu, R.G.; Ofori, E.; Archer, D.B.; Wu, S.S.; Pasternak, O.; McFarland, N.; Okun, M.S.; Vaillancourt, D.E. Progression marker of Parkinson’s disease: A 4-year multi-site imaging study. Brain 2017, 140, 2183–2192. [Google Scholar] [CrossRef]
- Pfeiffer, R.F. Non-motor symptoms in Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 22, S119–S122. [Google Scholar] [CrossRef]
- Luquin, M.-R.; Kulisevsky, J.; Martinez-Martin, P.; Mir, P.; Tolosa, E.S. Consensus on the Definition of Advanced Parkinson’s Disease: A Neurologists-Based Delphi Study (CEPA Study). Park. Dis. 2017, 2017, 4047392. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Martin, P.; Kulisevsky, J.; Mir, P.; Tolosa, E.; García-Delgado, P.; Luquin, M.R. Validation of a simple screening tool for early diagnosis of advanced Parkinson’s disease in daily practice: The CDEPA questionnaire. NPJ Parkinsons Dis. 2018, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Elbeddini, A.; To, A.; Tayefehchamani, Y.; Wen, C. Potential impact and challenges associated with Parkinson’s disease patient care amidst the COVID-19 global pandemic. J. Clin. Mov. Disord. 2020, 7, 7. [Google Scholar] [CrossRef]
- Aldred, J.; Anca-Herschkovitsch, M.; Antonini, A.; Bajenaru, O.; Bergmann, L.; Bourgeois, P.; Cubo, E.; Davis, T.; Iansek, R.; Kovacs, N. Application of the ‘5-2-1’ screening cri-teria in advanced Parkinson’s disease: Interim analysis of DUOGLOBE. Neurodegener Dis. Manag. 2020, 10, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Santos-García, D.; de Deus Fonticoba, T.; Suárez Castro, E.; Aneiros Díaz, A.; McAfee, D. 5-2-1 Criteria: A Simple Screening Tool for Identifying Advanced PD Patients Who Need an Op-timization of Parkinson’s Treatment. Parkinsons Dis. 2020, 2020, 7537924. [Google Scholar] [CrossRef]
- Antonini, A.; Odin, P.; Schmidt, P.; Cubillos, F.; Standaert, D.G.; Henriksen, T.; Jimenez-Shahed, J.; Alobaidi, A.; Jalundhwala, Y.J.; Bao, Y. Validation and clinical value of the MANAGE-PD tool: A clinician-reported tool to identify Parkinson’s disease patients inadequately controlled on oral medications. Parkinsonism Relat. Disord. 2021, 92, 59–66. [Google Scholar] [CrossRef]
- International Union Against Cancer (UICC). TNM Classfication of Malignant Tumors, 5th ed.; Sobin, L.H., Wittekind, C., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1997. [Google Scholar]
- Brott, T.; Adams, H.P.; Olinger, C.P.; Marler, J.R.; Barsan, W.G.; Biller, J.; Spilker, J.; Holleran, R.; Eberle, R.; Hertzberg, V. Measurements of acute cerebral infarction: A clinical examination scale. Stroke 1989, 20, 864–870. [Google Scholar] [CrossRef]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef]
- Olanow, C.W.; Stern, M.B.; Sethi, K. The scientific and clinical basis for the treatment of Parkinson disease (2009). Neurology 2009, 72, S1–S136. [Google Scholar] [CrossRef] [PubMed]
- Poewe, W. Non-motor symptoms in Parkinson’s disease. Eur. J. Neurol. 2008, 15, S14–S20. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, R.F. Autonomic Dysfunction in Parkinson’s Disease. Neurotherapeutics 2020, 17, 1464–1479. [Google Scholar] [CrossRef] [PubMed]
- Arnulf, I.; Konofal, E.; Merino-Andreu, M.; Houeto, J.L.; Mesnage, V.; Welter, M.L.; Lacomblez, L.; Golmard, J.L.; Derenne, J.P.; Agid, Y. Parkinson’s disease and sleepiness: An integral part of PD. Neurology 2002, 58, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Ford, B. Pain in Parkinson’s disease. Mov. Disord. 2010, 25, S98–S103. [Google Scholar] [CrossRef]
- Barton, B.; Grabli, D.; Bernard, B.; Czernecki, V.; Goldman, J.G.; Stebbins, G.; Dubois, B.; Goetz, C.G. Clinical validation of Movement Disorder Socie-ty-recommended diagnostic criteria for Parkinson’s disease with dementia. Mov. Disord. 2012, 27, 248–253. [Google Scholar] [CrossRef]
- Meireles, J.; Massano, J. Cognitive impairment and dementia in Parkinson’s disease: Clinical features, diagnosis, and management. Front. Neurol. 2012, 3, 88. [Google Scholar] [CrossRef]
- Katz, S.; Ford, A.; Moskowitz, R.W.; Jackson, B.A.; Jaffe, M.W. Studies of illness in the aged. The index of ADL: A standardized measure of biological and psycholosocial function. J. Am. Med. Assoc. 1963, 185, 914–919. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. The functional assessment in rehabilitation of elderly people: Self maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Stocchi, F.; Antonini, A.; Barone, P.; Tinazzi, M.; Zappia, M.; Onofrj, M.; Ruggieri, S.; Morgante, L.; Bonuccelli, U.; Lopiano, L.; et al. Early DEtection of wEaring off in Parkinson disease: The DEEP study. Park. Relat. Disord. 2013, 20, 204–211. [Google Scholar] [CrossRef]
- Santos-García, D.; de Deus Fonticoba, T.; Castro, S.; Diaz, A.; McAfee, D.; Catalan, M.J.; Alonso-Frech, F.; Villanueva, C.; Jesus, S.; Mir, P.; et al. Non-motor symptom burden is strongly correlated to motor complications in patients with Parkinson’s disease. Eur. J. Neurol. 2020, 27, 1210–1223. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S.; Oakes, D.; Shoulson, I.; Kieburtz, K.; Rudolph, A.; Lang, A.; Warren Olanow, C.; Tanner, C.; Marek, K.; Parkinson Study Group. Levodopa and the progression of Parkinson’s disease. N. Engl. J. Med. 2004, 351, 2498–2508. [Google Scholar]
- Turcano, P.; Mielke, M.M.; Bower, J.H.; Parisi, J.E.; Cutsforth-Gregory, J.K.; Ahlskog, J.E.; Savica, R. Levodopa-induced dyskinesia in Parkinson disease: A population-based cohort study. Neurology 2018, 91, e2238-43. [Google Scholar] [CrossRef]
- Fox, S.H.; Katzenschlager, R.; Lim, S.-Y.; Barton, B.; de Bie, R.M.A.; Seppi, K.; Coelho, M.; Sampaio, C. International Parkinson and movement disorder society evidence-based medicine review: Update on treatments for the motor symptoms of Parkinson’s disease. Mov. Disord. 2018, 33, 1248–1266. [Google Scholar] [CrossRef] [PubMed]
- Won, J.H.; Byun, S.J.; Oh, B.-M.; Park, S.J.; Gil Seo, H. Risk and mortality of aspiration pneumonia in Parkinson’s disease: A nationwide database study. Sci. Rep. 2021, 11, 6957. [Google Scholar] [CrossRef] [PubMed]
- García, D.S.; de Deus Fonticoba, T.; Cores Bartolomé, C.; Rios, L.N.; Garcia Roca, L.; Martinez Miro, C.; Canfield, H.; Maestre, S.J.; Aguilar, M.; Pastor, P.; et al. Predictors of Loss of Functional Independence in Parkinson’s Disease: Results from the COPPADIS Cohort at 2-Year Follow-Up and Comparison with a Control Group. Diagnostics 2021, 11, 1801. [Google Scholar] [CrossRef]
- Lau, B.; Meier, N.; Serra, G.; Czernecki, V.; Schuepbach, M.; Navarro, S.; Cornu, P.; Grabli, D.; Agid, Y.; Vidailhet, M.; et al. Axial symptoms predict mortality in patients with Parkinson disease and subthalamic stimulation. Neurology 2019, 92, e2559–e2570. [Google Scholar] [CrossRef]
- Simuni, T.; Caspell-Garcia, C.; Coffey, C.; Lasch, S.; Tanner, C.; Marek, K. PPMI Investigators. How stable are Parkinson’s disease subtypes in de novo patients: Analysis of the PPMI cohort? Parkinsonism Relat. Disord. 2016, 28, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Hacker, M.L.; Delong, M.R.; Turchan, M.; Heusinkveld, L.E.; Ostrem, J.L.; Molinari, A.L.; Currie, A.D.; Konrad, P.E.; Davis, T.L.; Phibbs, F.T.; et al. Effects of deep brain stimulation on rest tremor progression in early stage Parkinson disease. Neurology 2018, 91, e463–e471. [Google Scholar] [CrossRef]
- Martínez-Fernández, R.; Máñez-Miró, J.U.; Rodríguez-Rojas, R.; del Alamo, M.; Shah, B.B.; Hernandez-Fernandez, F.; Pineda-Pardo, J.A.; Monje, M.H.G.; Fernandez-Rodriguez, B.; Sperling, S.A.; et al. Randomized Trial of Fo-cused Ultrasound Subthalamotomy for Parkinson’s Disease. N. Engl. J. Med. 2020, 383, 2501–2513. [Google Scholar] [CrossRef]
- Aarsland, D.; Larsen, J.P.; Karlsen, K.; Lim, N.G.; Tandberg, E. Mental symptoms in Park-inson’s disease are important contributors to caregiver distress. Int. J. Geriatr. Psychiatry 1999, 14, 866–874. [Google Scholar] [CrossRef]
- Aarsland, D.; Larsen, J.P.; Tandberg, E.; Laake, K. Predictors of nursing home placement in Parkinson’s disease: A population-based, prospective study. J. Am. Geriatr. Soc. 2000, 48, 938–942. [Google Scholar] [CrossRef]
- García, D.S.; de Deus Fonticoba, T.; Cores, C.; Canfield, H.; Paz Gonzalez, J.M.; Miro, C.M.; Ayemerich, L.V.; Suarez, E.; Jesus, S.; Aguilar, M.; et al. COPPADIS Study Group. Predictors of Clinically Significant Quality of Life Impairment in Parkinson’s Disease. NPJ Parkinsons Dis. 2021, 11, 626. [Google Scholar]
- Galtier, I.; Nieto, A.; Lorenzo, J.N.; Barroso, J. Subjective cognitive decline and progression to dementia in Parkinson’s disease: A long-term follow-up study. J. Neurol. 2019, 266, 745–754. [Google Scholar] [CrossRef]
- Seppi, K.; Ray Chaudhuri, K.; Coelho, M.; Fox, S.H.; Katzenschlager, R.; Lloret, S.P.; Weintraub, D.; Sampaio, C. Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov. Disord. 2019, 34, 180–198. [Google Scholar] [CrossRef]
- Covinsky, K.E.; Palmer, R.M.; Fortinsky, R.H.; Counsell, S.R.; Stewart, A.L.; Kresevic, D.; Burant, C.J.; Landefeld, C.S. Loss of Independence in Activities of Daily Living in Older Adults Hospitalized with Medical Illnesses: Increased Vulnerability with Age. J. Am. Geriatr. Soc. 2003, 51, 451–458. [Google Scholar] [CrossRef]
- Macleod, A.D.; Dalen, I.; Tysnes, O.B.; Larsen, J.P.; Counsell, C.E. Development and validation of prognostic survival models in newly diagnosed Parkinson’s disease. Mov. Disord. 2018, 33, 108–116. [Google Scholar] [CrossRef]
- Fanhn, S.; Elton, R.L. Unified Parkinson’s Disease Rating Scale. In Recent Developments in Parkinson’s Disease; Fahn, S., Marsden, C.D., Calne, D.B., Goldstein, M., Eds.; Macmillan Health Care Information: Florham Park, NJ, USA, 1987; Volume 2, pp. 153–164. [Google Scholar]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef]
- Rovini, E.; Maremmani, C.; Cavallo, F. How Wearable Sensors Can Support Parkinson’s Disease Diagnosis and Treatment: A Systematic Review. Front Neurosci. 2017, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.; Sanchez-Ferro, A.; Maetzler, W. How Mobile Health Technology and Electronic Health Records Will Change Care of Patients with Parkinson’s Disease. J. Park. Dis. 2018, 8, S41–S45. [Google Scholar] [CrossRef]
- Sauerbier, A.; Jenner, P.; Todorova, A.; Chaudhuri, K.R. Non motor subtypes and Parkinson’s disease. Parkinsonism Relat. Disord. 2016, 22, S41–S46. [Google Scholar] [CrossRef]
- Brendel, M.; Su, C.; Hou, Y.; Henchcliffe, C.; Wang, F. Comprehensive subtyping of Parkinson’s disease patients with similarity fusion: A case study with BioFIND data. NPJ Park. Dis. 2021, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Emamzadeh, F.N.; Surguchov, A. Parkinson’s Disease: Biomarkers, Treatment, and Risk Factors. Front. Neurosci. 2018, 12, 612. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Castrillo, J.C.; Martínez-Martín, P.; Burgos, A.; Arroyo, G.; Luquin, M.R.; Arbelo, J.M. Prevalence of advanced Parkinson’s Disease in patients treated in the hospitals of the Spanish public health care system: PARA-DISE Study (interim analysis). Mov. Disord. 2019, 34, S752. [Google Scholar]
- Pirtošek, Z.; Bajenaru, O.; Kovács, N.; Milanov, I.; Relja, M.; Skorvanek, M. Update on the Man-agement of Parkinson’s Disease for General Neurologists. Parkinsons Dis. 2020, 2020, 9131474. [Google Scholar] [PubMed]

|
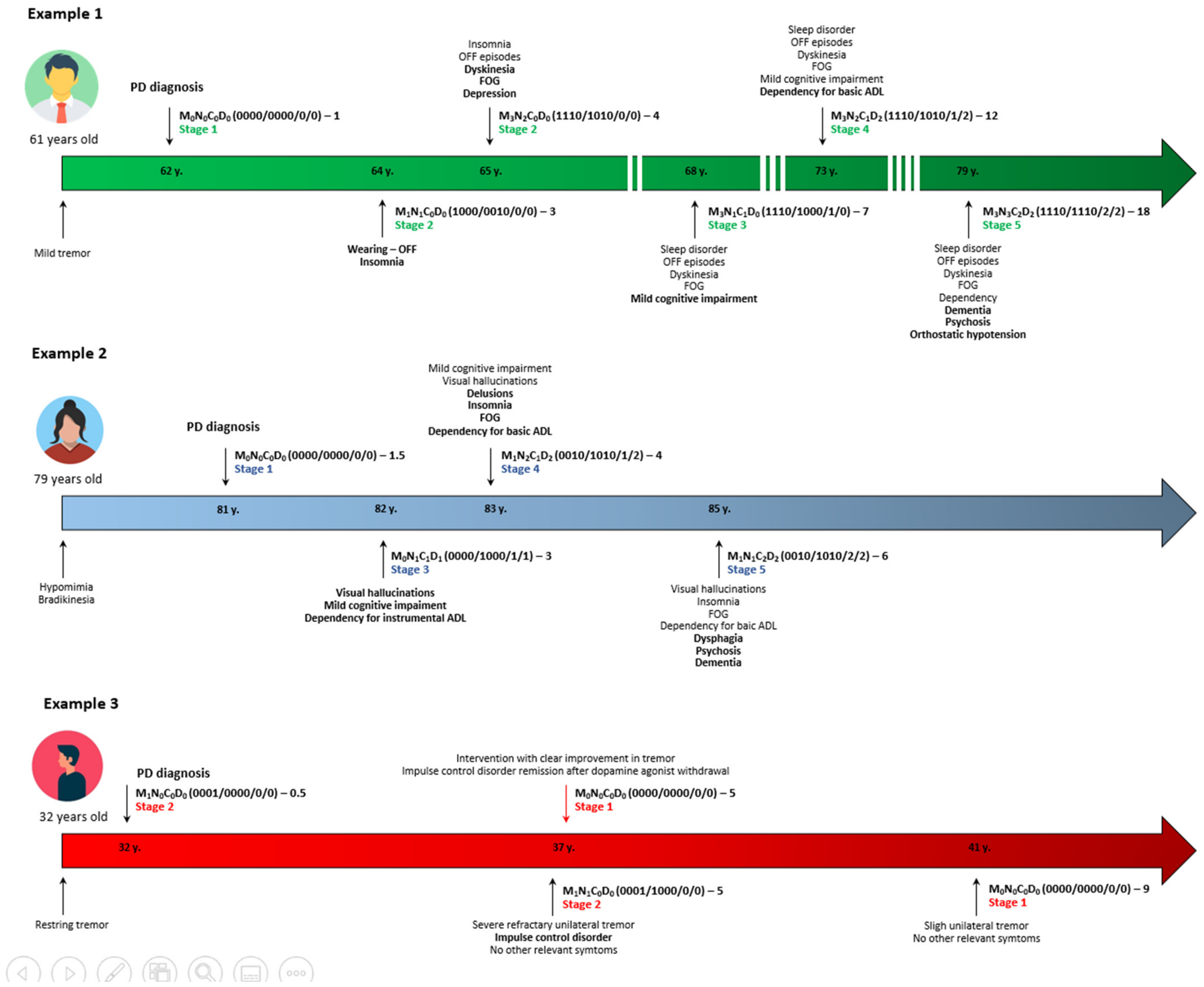
| Example 1 • A 49 year old man, 6 months from symptoms onset with stage 1 of H&Y and Unified Parkinson’s Disease Rating Scale—part III score of 7 points, without cognitive impairment and any significant disabling motor or NMS and independent for ADL. → M0N0C0D0 (0000/0000/0/0)–5. → Stage 1. Example 2 • A 50 year old woman, 2 years from symptoms onset with significant disabling resting tremor on the right part of the body but without axial symptoms, motor fluctuations, dyskinesia or any relevant NMS, with normal cognition and independent for ADL. → M1N0C0D0 (0001/0000/0/0)–2. → Stage 2. Example 3 • A 70 year old woman, 6 years from symptoms onset with motor fluctuations, dyskinesia, FOG, depression, visual hallucinations, mild cognitive impairment and dependency for instrumental ADL. → M3N1C1D1 (1110/1000/1/1)–6. → Stage 3. Example 4 • A 79 year old man, 4 years from symptoms onset with PIGD (postural instability gait difficulty) motor phenotype with FOG and falls but without motor complications, and severe RBD, pain and depression, without cognitive impairment but with loss of independence for basic ADL. → M1N3C0D2 (0010/1011/0/2)–4. → Stage 4. Example 5 • A 69 year old woman, 20 years from symptoms onset with motor fluctuations without dyskinesia, FOG, falls, dysphagia, visual hallucinations with psychosis, orthostatic hypotension, severe insomnia, dementia, and dependency for ADL. → M2N3C2D2 (1010/1110/2/2)–20. → Stage 5. |
| Stage 1 • The patient meets criteria of Parkinson’s disease but neither significant disabling motor or non-motor symptoms and there is no cognitive impairment. The patient is functionally independent for activities of daily living. • MNCD classification is M0N0C0D0 (0000/0000/0/0). Stage 2 • The patient has at least one significant disabling motor or non-motor symptom. There is no cognitive impairment. The patient is functionally independent for instrumental and basic activities of daily living. • MNCD classification: M or N ≥ 1; C = 0; D = 0. Stage 3 • There is cognitive impairment (no dementia) and/or dependency for instrumental ADL. • MNCD classification: M and N, from 0 to 4; At least, C or D = 1. Stage 4 • The patient is functionally dependent for basic activities of daily living. There could be mild cognitive impairment but not dementia. • MNCD classification: M and N, from 0 to 4; C, from 0 to 1; D = 2. Stage 5 • The patient is functionally dependent for basic ADL and there is dementia. • MNCD classification: M and N, from 0 to 4; C = 2; D = 2. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos García, D.; Álvarez Sauco, M.; Calopa, M.; Carrillo, F.; Escamilla Sevilla, F.; Freire, E.; García Ramos, R.; Kulisevsky, J.; Gómez Esteban, J.C.; Legarda, I.; et al. MNCD: A New Tool for Classifying Parkinson’s Disease in Daily Clinical Practice. Diagnostics 2022, 12, 55. https://doi.org/10.3390/diagnostics12010055
Santos García D, Álvarez Sauco M, Calopa M, Carrillo F, Escamilla Sevilla F, Freire E, García Ramos R, Kulisevsky J, Gómez Esteban JC, Legarda I, et al. MNCD: A New Tool for Classifying Parkinson’s Disease in Daily Clinical Practice. Diagnostics. 2022; 12(1):55. https://doi.org/10.3390/diagnostics12010055
Chicago/Turabian StyleSantos García, Diego, María Álvarez Sauco, Matilde Calopa, Fátima Carrillo, Francisco Escamilla Sevilla, Eric Freire, Rocío García Ramos, Jaime Kulisevsky, Juan Carlos Gómez Esteban, Inés Legarda, and et al. 2022. "MNCD: A New Tool for Classifying Parkinson’s Disease in Daily Clinical Practice" Diagnostics 12, no. 1: 55. https://doi.org/10.3390/diagnostics12010055
APA StyleSantos García, D., Álvarez Sauco, M., Calopa, M., Carrillo, F., Escamilla Sevilla, F., Freire, E., García Ramos, R., Kulisevsky, J., Gómez Esteban, J. C., Legarda, I., Luquín, M. R. I., Castrillo, J. C. M., Martínez-Martin, P., Martínez-Torres, I., Mir, P., & Ignacio, Á. S. (2022). MNCD: A New Tool for Classifying Parkinson’s Disease in Daily Clinical Practice. Diagnostics, 12(1), 55. https://doi.org/10.3390/diagnostics12010055







