Smartphone-Enabled Personalized Diagnostics: Current Status and Future Prospects
Abstract
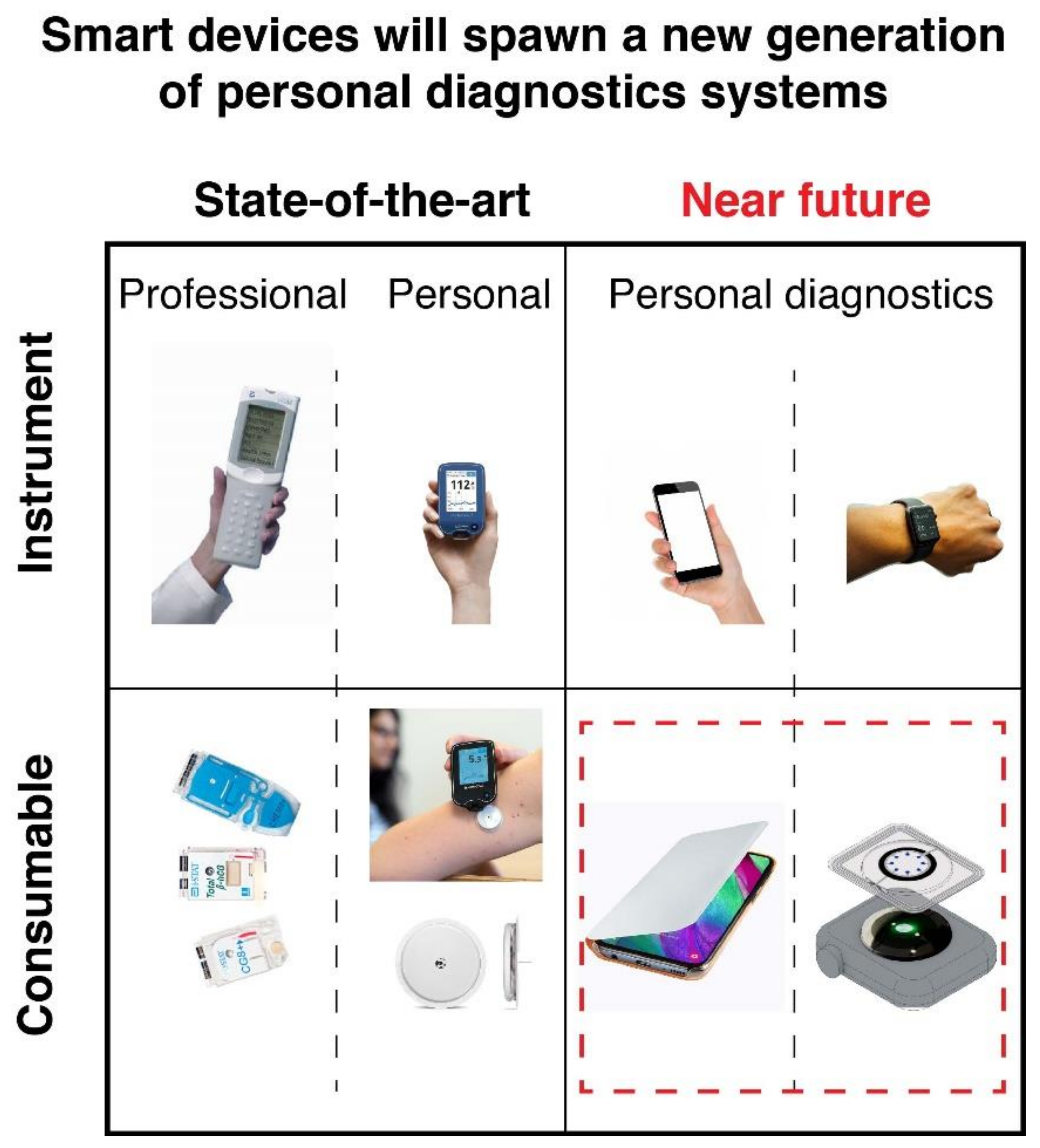
1. Introduction
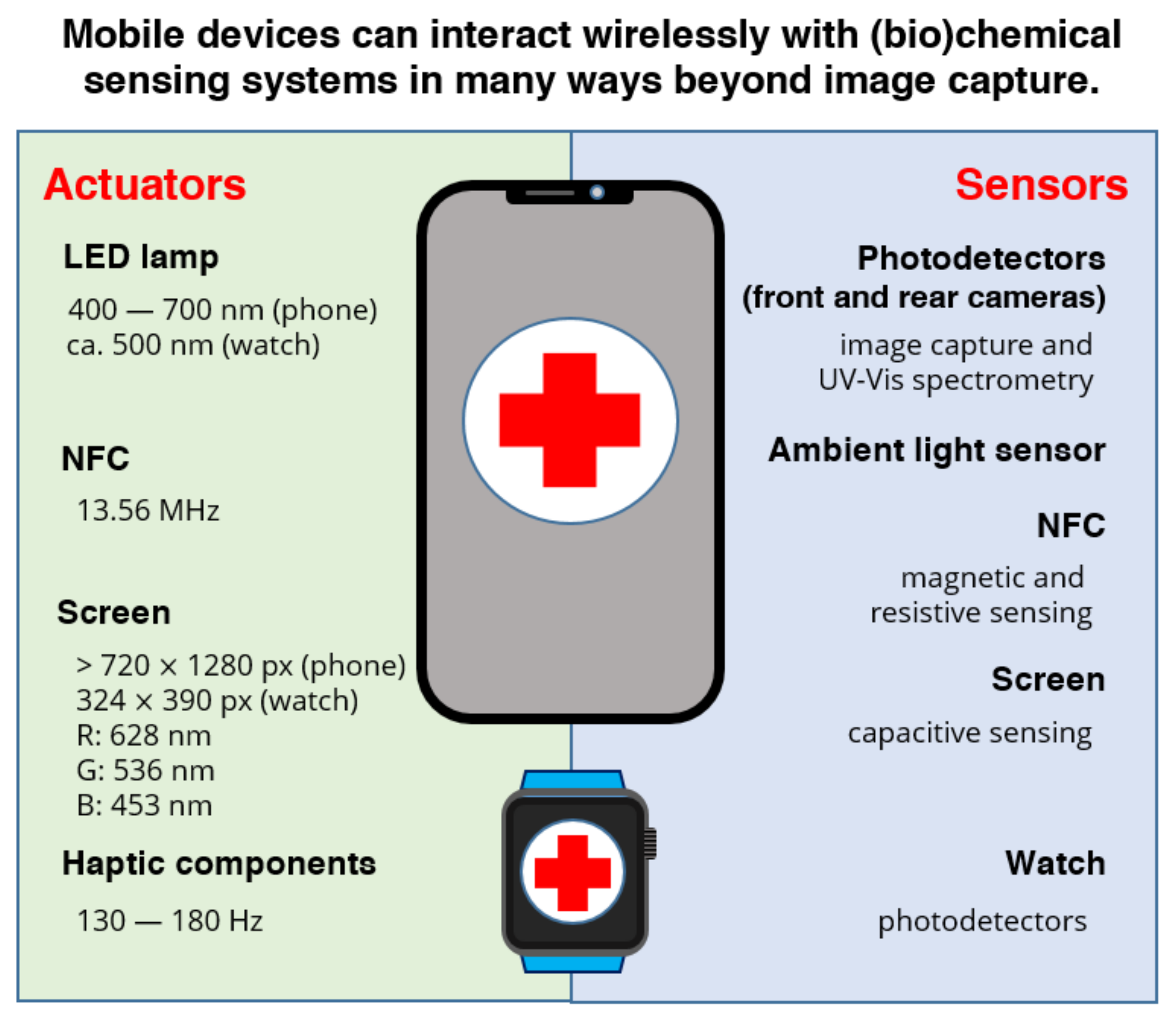
2. Smartphone-Based Diagnostics Today: Optical Detection and Data Transmission
3. Next Steps for Smartphone Full-Capacity Exploitation in Personal Diagnostic Systems
3.1. NFC- and Smartphone-Based Diagnostics
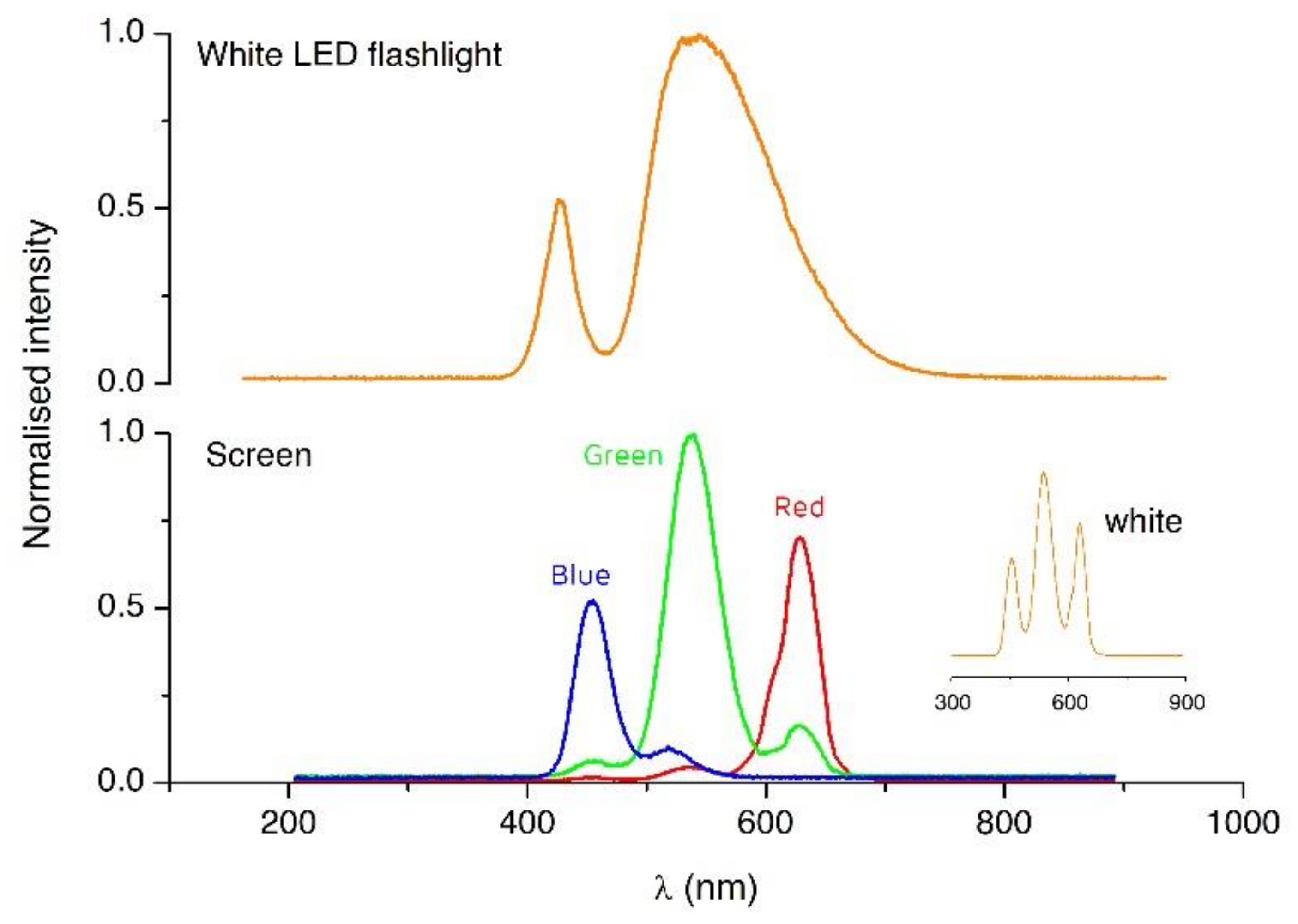
3.2. Light Actuation
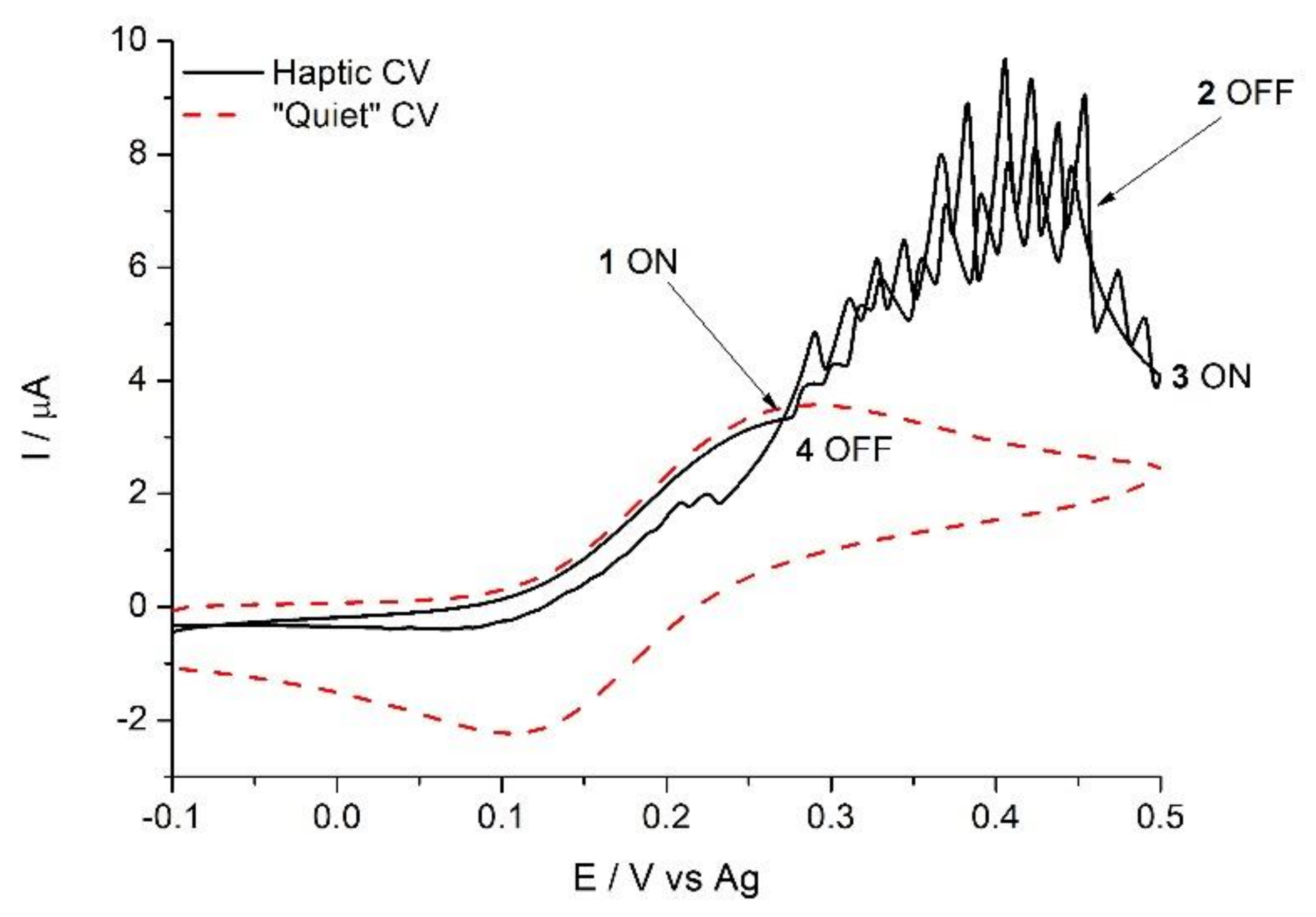
3.3. Haptic Feedback to Enhance Mass Transport
3.4. Magnetic Detection and Actuation
4. Bringing Molecular Diagnostics from the Clinical Lab to the Personal Sphere: Current Methods and Challenges for POC Implementation
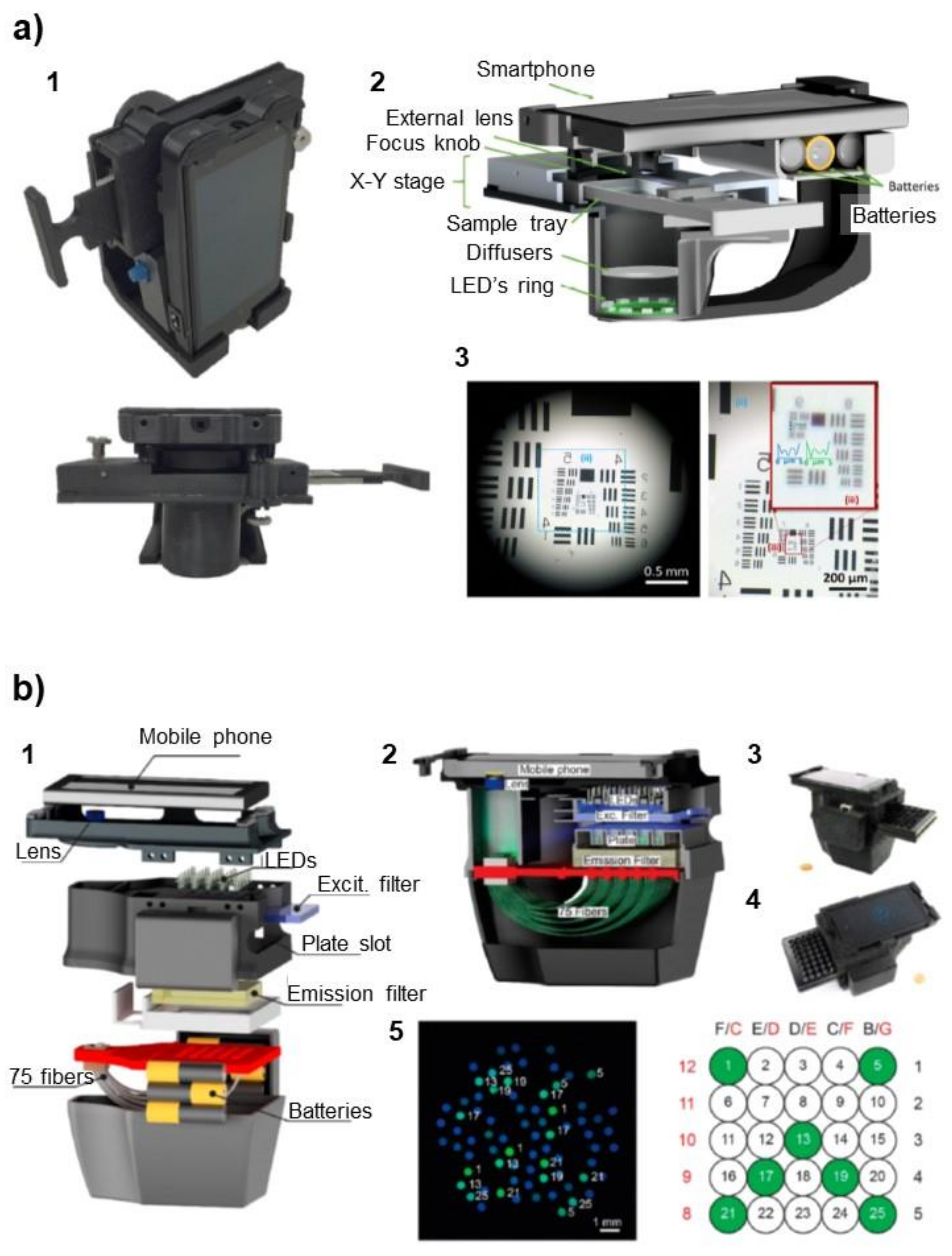
4.1. Microscopy
4.2. Immunoassays
4.3. Nucleic Acid Testing
4.4. Next-Generation Sequencing (NGS)
5. Conclusions and Outlook
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goetz, L.H.; Schork, N.J. Personalized medicine: Motivation, challenges, and progress. Fertil. Steril. 2018, 109, 952–963. [Google Scholar] [CrossRef]
- El Bcheraoui, C.; Weishaar, H.; Pozo-Martin, F.; Hanefeld, J. Assessing COVID-19 through the lens of health systems’ preparedness: Time for a change. Global. Health 2020, 16, 1–5. [Google Scholar] [CrossRef]
- Kosack, C.S.; Page, A.-L.; Klatser, P.R. A guide to aid the selection of diagnostic tests. Bull. World Health Organ. 2017, 95, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Alhabbab, R.Y. Basic Serological Testing; Kalyuzhny, A.E., Ed.; Springer: Cham, Switzerland, 2018; ISBN 978-3-319-77694-1. [Google Scholar]
- Maddocks, S.; Jenkins, R. Understanding PCR: A Practical Bench-Top Guide; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780128026830. [Google Scholar]
- Yetisen, A.K.; Akram, M.S.; Lowe, C.R. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 2013, 13, 2210–2251. [Google Scholar] [CrossRef] [PubMed]
- Morbioli, G.G.; Mazzu-Nascimento, T.; Stockton, A.M.; Carrilho, E. Technical aspects and challenges of colorimetric detection with microfluidic paper-based analytical devices (μPADs)—A review. Anal. Chim. Acta 2017, 970, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Shirshahi, V.; Liu, G. Enhancing the analytical performance of paper lateral flow assays: From chemistry to engineering. TrAC Trends Anal. Chem. 2021, 136, 116200. [Google Scholar] [CrossRef]
- Urusov, A.E.; Zherdev, A.V.; Dzantiev, B.B. Towards lateral flow quantitative assays: Detection approaches. Biosensors 2019, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Shibata, H.; Suzuki, K.; Citterio, D. Toward practical application of paper-based microfluidics for medical diagnostics: State-of-the-art and challenges. Lab Chip 2017, 17, 1206–1249. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-S.; Kim, N.-C.; Ahn, S.-H. Impact of a nurse short message service intervention for patients with diabetes. J. Nurs. Care Qual. 2006, 21, 266–271. [Google Scholar] [CrossRef]
- Blaivas, M.; Lyon, M.; Duggal, S. Ultrasound image transmission via camera phones for overreading. Am. J. Emerg. Med. 2005, 23, 433–438. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Carrilho, E.; Thomas, S.W.; Sindi, H.; Whitesides, G.M. Simple Telemedicine for Developing Regions: Camera Phones and Paper-Based Microfluidic Devices for Real-Time, Off-Site Diagnosis. Anal. Chem. 2008, 80, 3699–3707. [Google Scholar] [CrossRef] [PubMed]
- Quesada-González, D.; Merkoçi, A. Mobile phone-based biosensing: An emerging “diagnostic and communication” technology. Biosens. Bioelectron. 2017, 92, 549–562. [Google Scholar] [CrossRef]
- Rezazadeh, M.; Seidi, S.; Lid, M.; Pedersen-Bjergaard, S.; Yamini, Y. The modern role of smartphones in analytical chemistry. TrAC Trends Anal. Chem. 2019, 118, 548–555. [Google Scholar] [CrossRef]
- Kassal, P.; Steinberg, M.D.; Steinberg, I.M. Wireless chemical sensors and biosensors: A review. Sens. Actuators B Chem. 2018, 266, 228–245. [Google Scholar] [CrossRef]
- Banik, S.; Melanthota, S.K.; Arbaaz; Vaz, J.M.; Kadambalithaya, V.M.; Hussain, I.; Dutta, S.; Mazumder, N. Recent trends in smartphone-based detection for biomedical applications: A review. Anal. Bioanal. Chem. 2021, 413, 2389–2406. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.S.; Jeon, B.G.; Ihm, C.; Park, J.K.; Jung, M.Y. A simple and smart telemedicine device for developing regions: A pocket-sized colorimetric reader. Lab Chip 2011, 11, 120–126. [Google Scholar] [CrossRef]
- Breslauer, D.N.; Maamari, R.N.; Switz, N.A.; Lam, W.A.; Fletcher, D.A. Mobile phone based clinical microscopy for global health applications. PLoS ONE 2009, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Delaney, J.L.; Doeven, E.H.; Harsant, A.J.; Hogan, C.F. Use of a mobile phone for potentiostatic control with low cost paper-based microfluidic sensors. Anal. Chim. Acta 2013, 803, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S. Point of care sensing and biosensing using ambient light sensor of smartphone: Critical review. TrAC Trends Anal. Chem. 2019, 110, 393–400. [Google Scholar] [CrossRef]
- Chen, W.; Yao, Y.; Chen, T.; Shen, W.; Tang, S.; Lee, H.K. Application of smartphone-based spectroscopy to biosample analysis: A review. Biosens. Bioelectron. 2021, 172, 112788. [Google Scholar] [CrossRef]
- Doeven, E.H.; Barbante, G.J.; Harsant, A.J.; Donnelly, P.S.; Connell, T.U.; Hogan, C.F.; Francis, P.S. Mobile phone-based electrochemiluminescence sensing exploiting the ‘USB On-The-Go’ protocol. Sens. Actuators B Chem. 2015, 216, 608–613. [Google Scholar] [CrossRef]
- Moya, A.; Pol, R.; Martínez-Cuadrado, A.; Villa, R.; Gabriel, G.; Baeza, M. Stable Full-Inkjet-Printed Solid-State Ag/AgCl Reference Electrode. Anal. Chem. 2019, 91. [Google Scholar] [CrossRef] [PubMed]
- Nemiroski, A.; Christodouleas, D.C.; Hennek, J.W.; Kumar, A.A.; Maxwell, E.J.; Fernández-Abedul, M.T.; Whitesides, G.M. Universal mobile electrochemical detector designed for use in resource-limited applications. Proc. Natl. Acad. Sci. USA 2014, 111, 11984–11989. [Google Scholar] [CrossRef]
- Guo, J. Smartphone-Powered Electrochemical Biosensing Dongle for Emerging Medical IoTs Application. IEEE Trans. Ind. Inform. 2018, 14, 2592–2597. [Google Scholar] [CrossRef]
- Li, J.; Lillehoj, P.B. Microfluidic Magneto Immunosensor for Rapid, High Sensitivity Measurements of SARS-CoV-2 Nucleocapsid Protein in Serum. ACS Sens. 2021. [Google Scholar] [CrossRef]
- Garoz-Ruiz, J.; Perales-Rondon, J.V.; Heras, A.; Colina, A. Spectroelectrochemical Sensing: Current Trends and Challenges. Electroanalysis 2019, 31, 1254–1278. [Google Scholar] [CrossRef]
- Ainla, A.; Mousavi, M.P.S.; Tsaloglou, M.N.; Redston, J.; Bell, J.G.; Fernández-Abedul, M.T.; Whitesides, G.M. Open-Source Potentiostat for Wireless Electrochemical Detection with Smartphones. Anal. Chem. 2018, 90, 6240–6246. [Google Scholar] [CrossRef] [PubMed]
- Teengam, P.; Siangproh, W.; Tontisirin, S.; Jiraseree-amornkun, A.; Chuaypen, N.; Tangkijvanich, P.; Henry, C.S.; Ngamrojanavanich, N.; Chailapakul, O. NFC-enabling smartphone-based portable amperometric immunosensor for hepatitis B virus detection. Sens. Actuators B Chem. 2021, 326, 128825. [Google Scholar] [CrossRef]
- Chandrasekar, P.; Dutta, A. Recent Developments in Near Field Communication: A Study. Wirel. Pers. Commun. 2021, 116, 2913–2932. [Google Scholar] [CrossRef]
- Kim, H.; Member, S.; Hirayama, H.; Zhang, R.U.I.; Choi, J.; Member, S. Review of near-field wireless power and communication for biomedical applications. IEEE Access 2017, 5, 21264–21285. [Google Scholar] [CrossRef]
- Coskun, V.; Ozdenizci, B.; Ok, K. A survey on near field communication (NFC) technology. Wirel. Pers. Commun. 2013, 71, 2259–2294. [Google Scholar] [CrossRef]
- Victoria, J.; Suarez, A.; Martinez, P.A.; Alcarria, A.; Gerfer, A.; Torres, J. Improving the Efficiency of NFC Systems Through Optimizing the Sintered Ferrite Sheet Thickness Selection. IEEE Trans. Electromagn. Compat. 2020, 62, 1504–1514. [Google Scholar] [CrossRef]
- Zulqarnain, M.; Stanzione, S.; Rathinavel, G.; Smout, S.; Willegems, M.; Myny, K.; Cantatore, E. A flexible ECG patch compatible with NFC RF communication. NPJ Flex. Electron. 2020, 4, 1–8. [Google Scholar] [CrossRef]
- Bansal, R. Near-field magnetic communication. IEEE Antennas Propag. Mag. 2004, 46, 114–115. [Google Scholar] [CrossRef]
- Albrecht, A.; Salmeron, J.F.; Becherer, M.; Lugli, P.; Rivadeneyra, A. Screen-Printed Chipless Wireless Temperature Sensor. IEEE Sens. J. 2019, 19, 12011–12015. [Google Scholar] [CrossRef]
- Larpant, N.; Kalambate, P.K.; Ruzgas, T.; Laiwattanapaisal, W. Paper-Based Competitive Immunochromatography Coupled with an Enzyme-Modified Electrode to Enable the Wireless Monitoring and Electrochemical Sensing of Cotinine in Urine. Sensors 2021, 21, 1659. [Google Scholar] [CrossRef]
- Wagih, M.; Weddell, A.S.; Beeby, S. Rectennas for radio-frequency energy harvesting and wireless power transfer: A review of antenna design [Antenna Applications Corner]. IEEE Antennas Propag. Mag. 2020, 62, 95–107. [Google Scholar] [CrossRef]
- Rajbhandari, G.; Vanaraj, P.W.; Maskey, B.B.; Park, H.; Sapkota, A.; Jung, Y.; Majima, Y.; Cho, G. An Electroactive Binder in the Formulation of IGZO Ink to Print an IGZO-Based Rectifier for Harvesting Direct Current (DC) Power from the Near Field Communication (NFC) Signal of a Smartphone. Adv. Electron. Mater. 2018, 4, 1–7. [Google Scholar] [CrossRef]
- Bhattacharyya, M.; Gruenwald, W.; Jansen, D.; Reindl, L.; Aghassi-Hagmann, J. An Ultra-Low-Power RFID/NFC frontend IC using 0.18 μm CMOS technology for passive tag applications. Sensors 2018, 18, 1452. [Google Scholar] [CrossRef]
- Xie, Z.; Avila, R.; Huang, Y.; Rogers, J.A. Flexible and Stretchable Antennas for Biointegrated Electronics. Adv. Mater. 2020, 32, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Chen, P.; Ma, Z.; Li, S.; Gao, X.; Wu, R.X.; Pan, L.; Shi, Y. Near-field communication sensors. Sensors 2019, 19, 3947. [Google Scholar] [CrossRef] [PubMed]
- Noda, A. Wearable NFC Reader and Sensor Tag for Health Monitoring. In Proceedings of the Biomedical Circuits and Systems Conference, BioCAS 2019, Nara, Japan, 17–19 October 2019; pp. 2–5. [Google Scholar] [CrossRef]
- Applications, B.; Fedtschenko, T.; Utz, A.; Stanitzki, A.; Hennig, A.; Lüdecke, A.; Haas, N.; Kokozinski, R. A New Configurable Wireless Sensor System for Biomedical Applications with ISO 18000-3 Interface in 0.35 µm CMOS. Sensors 2019, 19, 4110. [Google Scholar]
- Rosa, B.M.G.; Anastasova-Ivanova, S.; Yang, G.Z. NFC-Powered Flexible Chest Patch for Fast Assessment of Cardiac, Hemodynamic, and Endocrine Parameters. IEEE Trans. Biomed. Circuits Syst. 2019, 13, 1603–1614. [Google Scholar] [CrossRef]
- Bandodkar, A.J.; Gutruf, P.; Choi, J.; Lee, K.H.; Sekine, Y.; Reeder, J.T.; Jeang, W.J.; Aranyosi, A.J.; Lee, S.P.; Model, J.B.; et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 2019, 5, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Kim, J.; Won, S.M.; Ma, Y.; Kang, D.; Xie, Z.; Lee, K.T.; Chung, H.U.; Banks, A.; Min, S.; et al. Battery-free, wireless sensors for full-body pressure and temperature mapping. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Kim, J.; Salvatore, G.A.; Araki, H.; Chiarelli, A.M.; Xie, Z.; Banks, A.; Sheng, X.; Liu, Y.; Lee, J.W.; Jang, K.-I.; et al. Battery-free, stretchable optoelectronic systems for wireless optical characterization of the skin. Sci. Adv. 2016, 2, e1600418. [Google Scholar] [CrossRef]
- Cheng, C.; Li, X.; Xu, G.; Lu, Y.; Low, S.S.; Liu, G.; Zhu, L.; Li, C.; Liu, Q. Battery-free, wireless, and flexible electrochemical patch for in situ analysis of sweat cortisol via near field communication. Biosens. Bioelectron. 2021, 172. [Google Scholar] [CrossRef] [PubMed]
- Zhdanov, A.; Keefe, J.; Franco-Waite, L.; Konnaiyan, K.R.; Pyayt, A. Mobile phone based ELISA (MELISA). Biosens. Bioelectron. 2018, 103, 138–142. [Google Scholar] [CrossRef]
- Florea, L.; Diamond, D.; Benito-Lopez, F. Photo-Responsive Polymeric Structures Based on Spiropyran. Macromol. Mater. Eng. 2012, 297, 1148–1159. [Google Scholar] [CrossRef]
- Glennon, T.; Saez, J.; Czugala, M.; Florea, L.; McNamara, E.; Fraser, K.J.; Ducree, J.; Diamond, D.; Benito-Lopez, F. Photo-Switchable Microvalve in a Reusable Lab-on-a-Disc. In Proceedings of the 18th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Anchorage, AK, USA, 21–25 June 2015; pp. 109–112. [Google Scholar]
- Delaney, C.; McCluskey, P.; Coleman, S.; Whyte, J.; Kent, N.; Diamond, D. Precision control of flow rate in microfluidic channels using photoresponsive soft polymer actuators. Lab Chip 2017, 17, 2013–2021. [Google Scholar] [CrossRef] [PubMed]
- Saez, J.; Glennon, T.; Czugala, M.; Tudor, A.; Ducreé, J.; Diamond, D.; Florea, L.; Benito-Lopez, F. Reusable ionogel-based photo-actuators in a lab-on-a-disc. Sens. Actuators B Chem. 2018, 257, 963–970. [Google Scholar] [CrossRef]
- Al-Handarish, Y.; Omisore, O.M.; Igbe, T.; Han, S.; Li, H.; Du, W.; Zhang, J.; Wang, L. A Survey of Tactile-Sensing Systems and Their Applications in Biomedical Engineering. Adv. Mater. Sci. Eng. 2020, 2020. [Google Scholar] [CrossRef]
- Compton, R.G.; Eklund, J.C.; Marken, F. Sonoelectrochemical Processes: A Review. Electroanalysis 1997, 9, 509–522. [Google Scholar] [CrossRef]
- Kim, S.E.; Van Tieu, M.; Hwang, S.Y.; Lee, M.H. Magnetic particles: Their applications from sample preparations to biosensing platforms. Micromachines 2020, 11, 302. [Google Scholar] [CrossRef]
- Zhang, Y.; Nguyen, N.-T. Magnetic digital microfluidics—A review. Lab Chip 2017, 17, 994–1008. [Google Scholar] [CrossRef]
- Zhu, H.; Mavandadi, S.; Coskun, A.F.; Yaglidere, O.; Ozcan, A. Optofluidic fluorescent imaging cytometry on a cell phone. Anal. Chem. 2011, 83. [Google Scholar] [CrossRef] [PubMed]
- Doeven, E.H.; Barbante, G.J.; Kerr, E.; Hogan, C.F.; Endler, J.A.; Francis, P.S. Red–Green–Blue Electrogenerated Chemiluminescence Utilizing a Digital Camera as Detector. Anal. Chem. 2014, 86, 2727–2732. [Google Scholar] [CrossRef] [PubMed]
- Delaney, J.L.; Hogan, C.F. Mobile phone based electrochemiluminescence detection in paper-based microfluidic sensors. Methods Mol. Biol. 2015, 1256, 277–289. [Google Scholar]
- Rivenson, Y.; Ceylan Koydemir, H.; Wang, H.; Wei, Z.; Ren, Z.; Günaydın, H.; Zhang, Y.; Göröcs, Z.; Liang, K.; Tseng, D.; et al. Deep Learning Enhanced Mobile-Phone Microscopy. ACS Photonics 2018, 5, 2354–2364. [Google Scholar] [CrossRef]
- Kong, J.E.; Wei, Q.; Tseng, D.; Zhang, J.; Pan, E.; Lewinski, M.; Garner, O.B.; Ozcan, A.; Di Carlo, D. Highly Stable and Sensitive Nucleic Acid Amplification and Cell-Phone-Based Readout. ACS Nano 2017, 11, 2934–2943. [Google Scholar] [CrossRef] [PubMed]
- Crowther, J.R. The ELISA Guidebook; Humana Press: Totowa, NJ, USA, 2001; Volume 149, ISBN 0896037282. [Google Scholar]
- Assays, E.I.; Berg, B.; Cortazar, B.; Tseng, D.; Ozkan, H.; Feng, S.; Wei, Q.; Garner, O.B.; Ozcan, A.; Chan, R.Y.L.; et al. Cellphone-Based Hand-Held Microplate Reader for Point-of-Care Testing of Enzyme-Linked Immunosorbent Assays. ACS Nano 2015, 9, 7857–7866. [Google Scholar] [CrossRef]
- Shin, D.J.; Athamanolap, P.; Chen, L.; Hardick, J.; Lewis, M.; Hsieh, Y.H.; Rothman, R.E.; Gaydos, C.A.; Wang, T.H. Mobile nucleic acid amplification testing (mobiNAAT) for Chlamydia trachomatis screening in hospital emergency department settings. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wang, X.; Hu, J.; Gong, Y.; Wang, L.; Zhou, W.; Li, X.J.; Xu, F. A smartphone-based on-site nucleic acid testing platform at point-of-care settings. Electrophoresis 2019, 40. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Ganguli, A.; Nguyen, J.; Brisbin, R.; Shanmugam, K.; Hirschberg, D.L.; Wheeler, M.B.; Bashir, R.; Nash, D.M.; Cunningham, B.T. Smartphone-based multiplex 30-minute nucleic acid test of live virus from nasal swab extract. Lab Chip 2020, 20, 1621–1627. [Google Scholar] [CrossRef]
- Metzker, M.L. Sequencing technologies the next generation. Nat. Rev. Genet. 2010, 11, 31–46. [Google Scholar] [CrossRef]
- Callahan, N.; Tullman, J.; Kelman, Z.; Marino, J. Strategies for Development of a Next-Generation Protein Sequencing Platform. Trends Biochem. Sci. 2020, 45, 76–89. [Google Scholar] [CrossRef] [PubMed]
- Mrozek, D. A review of Cloud computing technologies for comprehensive microRNA analyses. Comput. Biol. Chem. 2020, 88, 107365. [Google Scholar] [CrossRef]
- Fozouni, P.; Son, S.; Díaz de León Derby, M.; Knott, G.J.; Gray, C.N.; D’Ambrosio, M.V.; Zhao, C.; Switz, N.A.; Kumar, G.R.; Stephens, S.I.; et al. Amplification-free detection of SARS-CoV-2 with CRISPR-Cas13a and mobile phone microscopy. Cell 2021, 184, 323–333. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merazzo, K.J.; Totoricaguena-Gorriño, J.; Fernández-Martín, E.; del Campo, F.J.; Baldrich, E. Smartphone-Enabled Personalized Diagnostics: Current Status and Future Prospects. Diagnostics 2021, 11, 1067. https://doi.org/10.3390/diagnostics11061067
Merazzo KJ, Totoricaguena-Gorriño J, Fernández-Martín E, del Campo FJ, Baldrich E. Smartphone-Enabled Personalized Diagnostics: Current Status and Future Prospects. Diagnostics. 2021; 11(6):1067. https://doi.org/10.3390/diagnostics11061067
Chicago/Turabian StyleMerazzo, Karla Jaimes, Joseba Totoricaguena-Gorriño, Eduardo Fernández-Martín, F. Javier del Campo, and Eva Baldrich. 2021. "Smartphone-Enabled Personalized Diagnostics: Current Status and Future Prospects" Diagnostics 11, no. 6: 1067. https://doi.org/10.3390/diagnostics11061067
APA StyleMerazzo, K. J., Totoricaguena-Gorriño, J., Fernández-Martín, E., del Campo, F. J., & Baldrich, E. (2021). Smartphone-Enabled Personalized Diagnostics: Current Status and Future Prospects. Diagnostics, 11(6), 1067. https://doi.org/10.3390/diagnostics11061067







