Assessment of Glutathione Peroxidase-1 (GPX1) Gene Expression as a Specific Diagnostic and Prognostic Biomarker in Malignant Pleural Mesothelioma
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Specimens
2.2. RNA Isolation
2.3. Synthesis of Complementary DNA (cDNA)
2.4. GPX1 Gene Expression Analysis by RT-qPCR
2.5. Statistical Analysis
3. Results
3.1. GPX1-mRNA Levels in MPM and Their ANPT
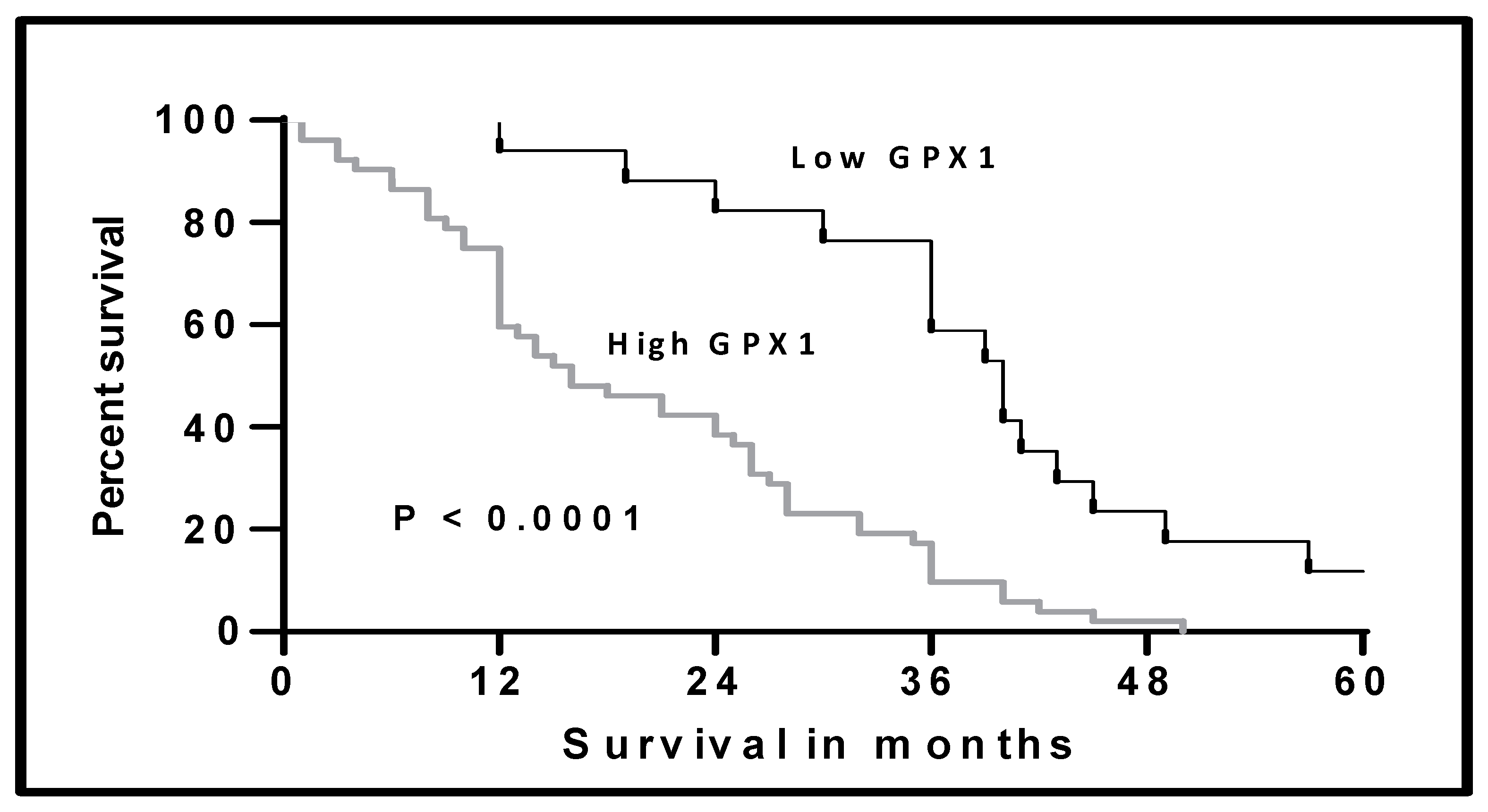
3.2. Clinical Significance of Elevated GPX1-mRNA Levels in MPM
3.3. The Role of Elevated GPX1- mRNA in the Diagnosis of MPM
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vogelzang, N.J.; Rusthoven, J.J.; Symanowski, J.; Denham, C.; Kaukel, E.; Ruffie, P.; Gatzemeier, U.; Boyer, M.; Emri, S.; Manegold, C.; et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J. Clin. Oncol. 2003, 21, 2636–2644. [Google Scholar] [CrossRef] [PubMed]
- Borasio, P.; Berruti, A.; Billé, A.; Lausi, P.O.; Levra, M.G.; Giardino, R.; Ardissone, F. Malignant pleural mesothelioma: Clinicopathologic and survival characteristics in a consecutive series of 394 patients. Eur. J. Cardio-Thoracic Surg. 2008, 33, 307–313. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Koksal, D.; Ozmen, O.; Demirag, F.; Bayiz, H.; Aydogdu, K.; Berkoglu, M.; Koyuncu, A. Prognostic factors in malignant pleural mesothelioma: A retrospective study of 60 Turkish patients. J. Cancer Res. Ther. 2015, 11, 216–222. [Google Scholar] [CrossRef]
- Di Leva, G.; Garofalo, M.; Croce, C.M. MicroRNAs in cancer. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 287–314. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Maiorino, M. Glutathione peroxidases. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2013, 1830, 3289–3303. [Google Scholar] [CrossRef]
- Lubos, E.; Loscalzo, J.; Handy, D.E. Glutathione peroxidase-1 in health and disease: From molecular mechanisms to therapeutic opportunities. Antioxid. Redox Signal. 2011, 15, 1957–1997. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohe, R.; Kipp, A. Glutathione peroxidases in different stages of carcinogenesis. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2009, 1790, 1555–1568. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Wang, H.; Zhuo, P.; Schwartz, J.L.; Diamond, A.M. Selenium and GPx-1 overexpression protect mammalian cells against UV-induced DNA damage. Biol. Trace Elem. Res. 2007, 115, 227–241. [Google Scholar] [CrossRef]
- Liu, J.; Hinkhouse, M.M.; Sun, W.; Weydert, C.J.; Ritchie, J.M.; Oberley, L.W.; Cullen, J.J. Redox regulation of pancreatic cancer cell growth: Role of glutathione peroxidase in the suppression of the malignant phenotype. Hum. Gene Ther. 2004, 15, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Diwadkar-Navsariwala, V.; Koh, T.; Fayad, R.; Fantuzzi, G.; Diamond, A.M. Selenoprotein deficiency enhances radiation-induced micronuclei formation. Mol. Nutr. Food Res. 2008, 52, 1300–1304. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, E.; Capone, F.; Accardo, M.; Sorice, A.; Costantini, M.; Colonna, G.; Castello, G. GPX4 and GPX7 over-expression in human hepatocellular carcinoma tissues. Eur. J. Histochem. 2015, 59, 2540. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.R.; Roh, J.-L.; Lee, S.M.; Park, Y.; Cho, K.-J.; Choi, S.-H.; Nam, S.Y.; Kim, S.Y. Overexpression of glutathione peroxidase 1 predicts poor prognosis in oral squamous cell carcinoma. J. Cancer Res. Clin. Oncol. 2017, 143, 2257–2265. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.B.; Edge, S.B.; Greene, F.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. The eighth edition AJCC cancer staging manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef] [PubMed]
- West, H.J.; Jin, J.O. Performance status in patients with cancer. JAMA Oncol. 2015, 1, 998. [Google Scholar] [CrossRef]
- Nagaraj, N.S.; Washington, M.K.; Merchant, B.N. Combined blockade of Src kinase and epidermal growth factor receptor with gemcitabine overcomes STAT3-mediated resistance of inhibition of pancreatic tumor growth. Clin. Cancer Res. 2011, 17, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Al-Karmalawy, A.A.; Elshal, M.F. Concanavalin—A shows synergistic cytotoxicity with tamoxifen via inducing apoptosis in estrogen receptor-positive breast cancer: In vitro and molecular docking studies. Pharm Sci. 2021. [Google Scholar] [CrossRef]
- Ghanem, A.A.; Emara, H.A.; Muawia, S.; El Maksoud, A.I.A.; Al-Karmalawy, A.A.; Elshal, M.F. Tanshinone IIA synergistically enhances the antitumor activity of doxorubicin by interfering with the PI3K/AKT/mTOR pathway and inhibition of topoisomerase II: In vitro and molecular docking studies. New J. Chem. 2020, 44, 17374–17381. [Google Scholar] [CrossRef]
- Eliaa, S.G.; Al-Karmalawy, A.A.; Saleh, R.M.; Elshal, M.F. Empagliflozin and Doxorubicin Synergistically Inhibit the Survival of Triple-Negative Breast Cancer Cells via Interfering with the mTOR Pathway and Inhibition of Calmodulin: In Vitro and Molecular Docking Studies. ACS Pharmacol. Transl. Sci. 2020, 3, 1330–1338. [Google Scholar] [CrossRef]
- Tafani, M.; Sansone, L.; Limana, F.; Arcangeli, T.; De Santis, E.; Polese, M.; Fini, M.; Russo, M.A. The interplay of reactive oxygen species, hypoxia, inflammation, and sirtuins in cancer initiation and progression. Oxidative Med. Cell. Longev. 2016, 2016, 3907147. [Google Scholar] [CrossRef] [PubMed]
- Filaire, E.; Dupuis, C.; Galvaing, G.; Aubreton, S.; Laurent, H.; Richard, R.; Filaire, M. Lung cancer: What are the links with oxidative stress, physical activity and nutrition. Lung Cancer 2013, 82, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Da Motta, L.L.; Müller, C.B.; De Bastiani, M.A.; Behr, G.A.; Franca, F.S.; Da Rocha, R.F.; Minotto, J.B.; Meurer, R.T.; Fernandes, M.; Roehe, A.; et al. Imbalance in redox status is associated with tumor aggressiveness and poor outcome in lung adenocarcinoma patients. J. Cancer Res. Clin. Oncol. 2014, 140, 461–470. [Google Scholar] [CrossRef]
- Al-Karmalawy, A.A.; Khattab, M. Molecular modelling of mebendazole polymorphs as a potential colchicine binding site inhibitor. New J. Chem. 2020, 44, 13990–13996. [Google Scholar] [CrossRef]
- Khattab, M.; Al-Karmalawy, A.A. Revisiting Activity of Some Nocodazole Analogues as a Potential Anticancer Drugs Using Molecular Docking and DFT Calculations. Front. Chem. 2021, 9, 92. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Xu, J.; Liang, C.; Liu, J.; Hua, J.; Zhang, Y.; Ni, Q.; Shi, S.; Yu, X. GPx1 is involved in the induction of protective autophagy in pancreatic cancer cells in response to glucose deprivation. Cell Death Dis. 2018, 9, 1187. [Google Scholar] [CrossRef]
- Dequanter, D.; Dok, R.; Koolen, L.; Poorten, V.V.; Nuyts, S. Prognostic significance of glutathione peroxidase levels (GPx1) in head and neck cancers. Front. Oncol. 2017, 7, 84. [Google Scholar] [CrossRef] [PubMed]
- Han, J.J.; Xie, D.R.; Wang, L.L.; Liu, Y.Q.; Wu, G.F.; Sun, Q.; Chen, Y.X.; Wei, Y.; Huang, Z.Q.; Li, H.G. Significance of glutathione peroxidase 1 and caudal-related homeodomain transcription factor in human gastric adenocarcinoma. Gastroenterol. Res. Pract. 2013, 2013, 380193. [Google Scholar] [CrossRef] [PubMed]
- Zmorzyński, S.; Świderska-Kołacz, G.; Koczkodaj, D.; Filip, A. Significance of polymorphisms and expression of enzyme-encoding genes related to glutathione in hematopoietic cancers and solid tumors. BioMed Res. Int. 2015, 2015, 853573. [Google Scholar] [CrossRef]
- Metere, A.; Frezzotti, F.; Graves, C.E.; Vergine, M.; De Luca, A.; Pietraforte, D.; Giacomelli, L. A possible role for selenoprotein glutathione peroxidase (GPx1) and thioredoxin reductases (TrxR1) in thyroid cancer: Our experience in thyroid surgery. Cancer Cell Int. 2018, 18, 7. [Google Scholar] [CrossRef]
- Cheng, Y.; Xu, T.; Li, S.; Ruan, H. GPX1, a biomarker for the diagnosis and prognosis of kidney cancer, promotes the progression of kidney cancer. Aging 2019, 11, 12165. [Google Scholar] [CrossRef]
- Min, S.Y.; Kim, H.S.; Jung, E.J.; Jee, C.D.; Kim, W.H. Prognostic significance of glutathione peroxidase 1 (GPX1) down-regulation and correlation with aberrant promoter methylation in human gastric cancer. Anticancer Res. 2012, 32, 3169–3175. [Google Scholar] [PubMed]
- Ekoue, D.N.; Ansong, E.; Hong, L.K.; Nonn, L.; Macias, V.; Deaton, R.; Rupnow, R.; Gann, P.H.; Kajdacsy-Balla, A.; Diamond, A.M. GPX1 localizes to the nucleus in prostate epithelium and its levels are not associated with prostate cancer recurrence. Antioxidants 2018, 7, 167. [Google Scholar] [CrossRef] [PubMed]
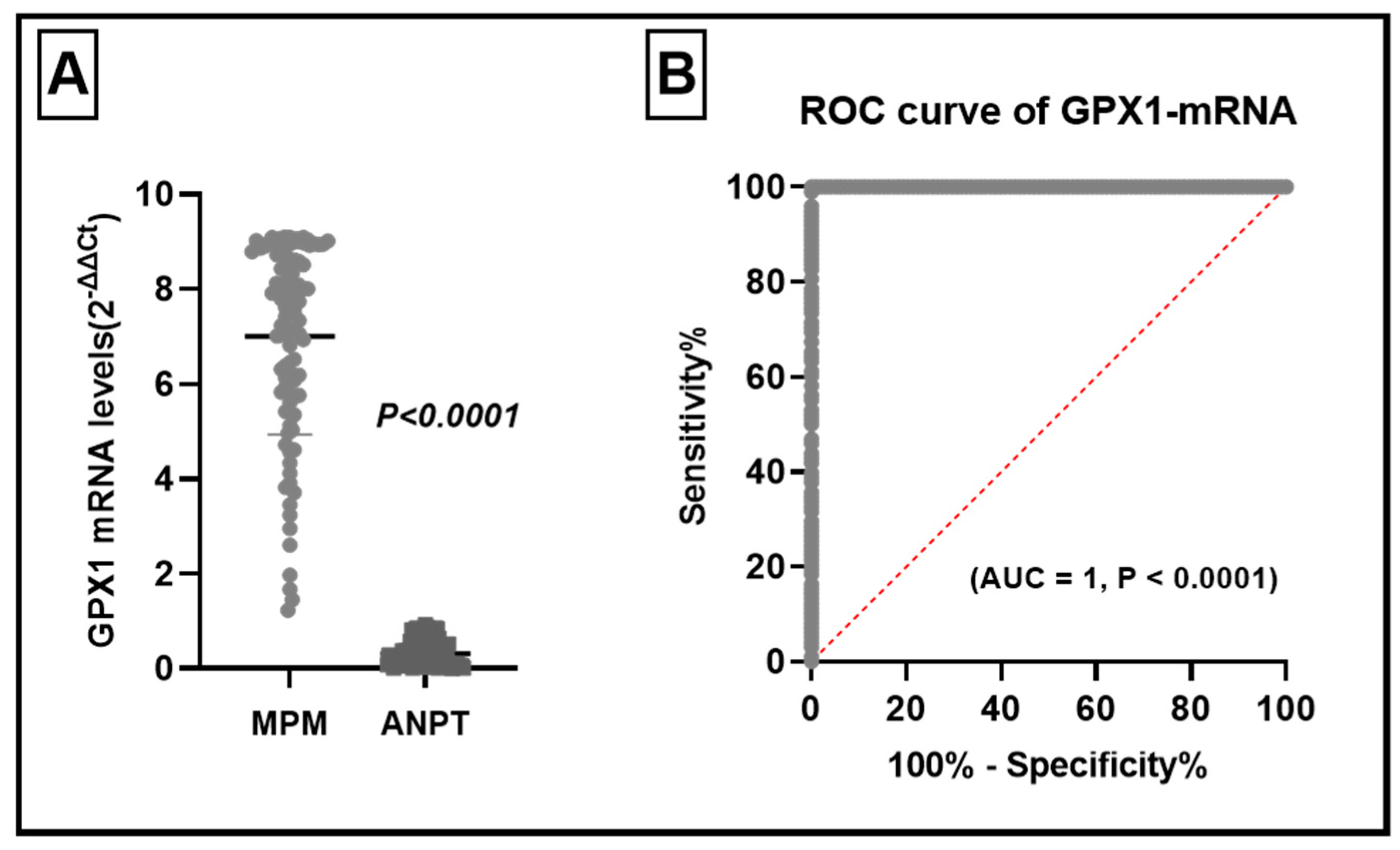

| GPX1 mRNA Level | ||||
|---|---|---|---|---|
| No | Mean | SD | p | |
| Age | ||||
| ≤55 years >55 years | 58 40 | 5.769 8.789 | 1.836 0.33 | <0.0001 * |
| Sex | ||||
| Males Females | 44 54 | 5.157 8.504 | 1.693 0.571 | <0.0001 * |
| Pathology | ||||
| Epithelioid Non-Epithelioid | 66 32 | 6.065 8.932 | 1.899 0.1485 | <0.0001 * |
| Histopathological grade | ||||
| G1–G2 G3–G4 | 35 63 | 4.67 8.297 | 1.56 0.737 | <0.0001 * |
| Tumor Resection | ||||
| Complete Partial Unresectable | 37 49 12 | 4.79 8.17 9.06 | 1.60 0.68 0.03 | <0.0001 ** |
| T | ||||
| T1 T2 T3 | 72 26 | 6.282 8.993 | 1.956 0.075 | <0.0001 * |
| N | ||||
| N1 N2 | 72 26 | 6.282 8.993 | 1.956 0.075 | <0.0001 * |
| Stage | ||||
| I II | 39 59 | 8.81 5.81 | 0.308 1.84 | <0.0001 * |
| Hb% | ||||
| <14 gm >14 gm | 56 42 | 5.69 8.75 | 1.819 0.366 | <0.0001 * |
| Platelet Count | ||||
| Normal High | 46 52 | 5.253 8.548 | 1.717 0.536 | <0.0001 * |
| WBCs | ||||
| Normal High | 57 41 | 5.729 8.77 | 1.827 0.3483 | <0.0001 * |
| LDH | ||||
| Normal High | 72 26 | 6.28 8.99 | 1.96 0.075 | <0.0001 * |
| Performance status | ||||
| 0–1 2 | 72 26 | 6.28 8.99 | 1.96 0.075 | <0.0001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gharib, A.F.; Alaa Eldeen, M.; Khalifa, A.S.; Elsawy, W.H.; Eed, E.M.; Askary, A.E.; Eid, R.A.; Soltan, M.A.; Raafat, N. Assessment of Glutathione Peroxidase-1 (GPX1) Gene Expression as a Specific Diagnostic and Prognostic Biomarker in Malignant Pleural Mesothelioma. Diagnostics 2021, 11, 2285. https://doi.org/10.3390/diagnostics11122285
Gharib AF, Alaa Eldeen M, Khalifa AS, Elsawy WH, Eed EM, Askary AE, Eid RA, Soltan MA, Raafat N. Assessment of Glutathione Peroxidase-1 (GPX1) Gene Expression as a Specific Diagnostic and Prognostic Biomarker in Malignant Pleural Mesothelioma. Diagnostics. 2021; 11(12):2285. https://doi.org/10.3390/diagnostics11122285
Chicago/Turabian StyleGharib, Amal F., Muhammad Alaa Eldeen, Amany Salah Khalifa, Wael H. Elsawy, Emad Mohamed Eed, Ahmad El Askary, Refaat A. Eid, Mohamed A. Soltan, and Nermin Raafat. 2021. "Assessment of Glutathione Peroxidase-1 (GPX1) Gene Expression as a Specific Diagnostic and Prognostic Biomarker in Malignant Pleural Mesothelioma" Diagnostics 11, no. 12: 2285. https://doi.org/10.3390/diagnostics11122285
APA StyleGharib, A. F., Alaa Eldeen, M., Khalifa, A. S., Elsawy, W. H., Eed, E. M., Askary, A. E., Eid, R. A., Soltan, M. A., & Raafat, N. (2021). Assessment of Glutathione Peroxidase-1 (GPX1) Gene Expression as a Specific Diagnostic and Prognostic Biomarker in Malignant Pleural Mesothelioma. Diagnostics, 11(12), 2285. https://doi.org/10.3390/diagnostics11122285






