Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates
Abstract
1. Introduction
2. Methods
2.1. Study Patients and Data Collection
- Patient history and symptoms: disease duration from first non-Raynaud’s phenomenon, smoking exposure (past or current), New York Heart Association (NYHA) functional class for dyspnoea, history of chest pain, palpitations, and syncope;
- Full rheumatological assessments: fulfillment of the EULAR/ACR 2013 classification criteria [26], specifying when this was obtained through a very early diagnosis of systemic sclerosis (VEDOSS) features only [27]; modified Rodnan skin score (mRSS) and type of skin involvement (limited or diffuse) according to LeRoy classification [28]; nailfold video capillaroscopy (NVC) pattern [29]; presence of interstitial lung disease (ILD) on high resolution computed tomography (HRCT) and its functional assessment with pulmonary function tests, including forced vital capacity (FVC) and diffusion lung capacity of carbon oxide (DLco); history of digital ulcers (DUs) or current DUs; gastro-intestinal involvement; history of renal crisis; ongoing vasodilating; and/or ongoing and past immunosuppressive medications;
- Cardiological workup: evaluation of traditional cardiovascular risk factors; standard 12-lead ECG; 24 h ECG Holter monitoring; standard transthoracic echocardiography; morpho functional and tissue characterization by CMR. The following CMR parameters were included: left and right chamber volumes; left and right ventricular function; presence and pattern of late gadolinium enhancement (LGE); qualitatively evaluated T2 short T inversion recovery (STIR) images for increased signal at myocardial and pericardial level; and presence of pericardial effusion;
- Laboratory parameters: anti-nuclear antibodies (ANA); anti-topoisomerase I (ATA) antibodies; anti-centromere antibodies (ACA); anti-RNA polymerase III antibodies and other rare SSc-related antibodies by standard methods according to local laboratory; type B pro-natriuretic peptide (NT-proBNP); high-sensitive troponin I or T; and creatinine clearance.
2.2. Serum AHA and AIDA Testing by Indirect Immunofluorescence (IF)
2.3. Prognostic Evaluation
2.4. Statistical Analysis
3. Results
3.1. Baseline Features of the Study Population
3.2. AHA and AIDA: Frequency and Associations with Baseline Clinical and Diagnostic Features
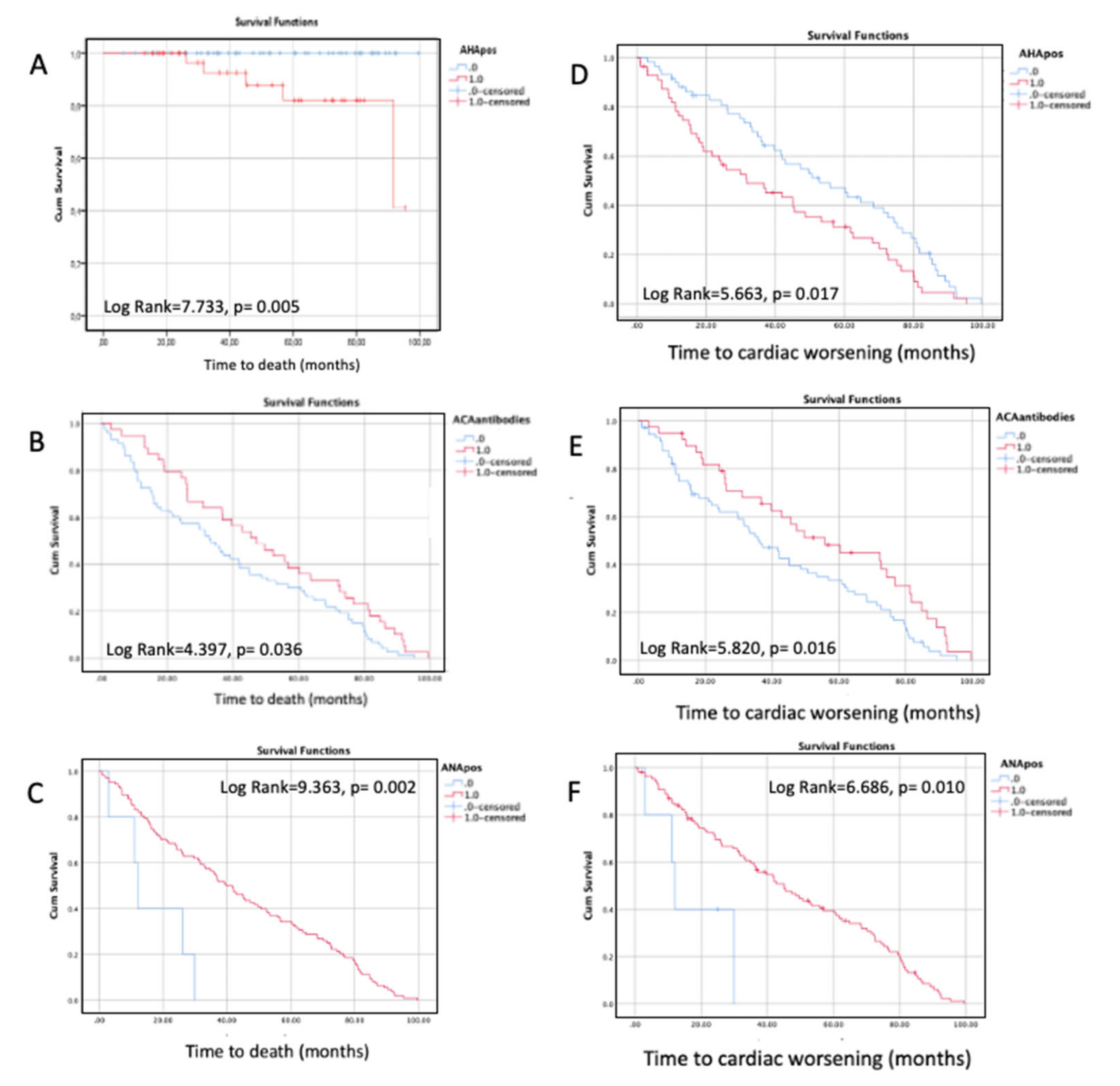
3.3. Follow-Up and Prognostic Evaluation
4. Discussion
4.1. Frequency and Significance of AHA and AIDA in SSc
4.2. AHA and AIDA: Significance of Associations with Baseline Clinical and Diagnostic Features
4.3. AHA, AIDA, and Prognostic Outcomes
4.4. Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACA | anti-centromere antibodies |
| AHA | anti-heart antibody |
| AIDA | anti-intercalated disk antibodies |
| ANA | anti-nuclear antibodies |
| ATA | anti-topoisomerase I antibodies |
| CAD | coronary artery disease |
| CI | confidence interval |
| CMR | cardiac magnetic resonance |
| DLco | diffusion capacity of the lung for carbone oxyde |
| DUs | digital ulcers |
| ECG | electrocardiogram |
| Echo | echocardiography |
| FVC | forced vital capacity |
| HF | heart failure |
| HInv | heart involvement |
| HR | hazard ratio |
| HRCT | high-resolution computed toography |
| ICD | implantable cardiac defibrillator |
| ID | immunofluorescence |
| IHD | ischemich heart disease |
| ILD | interstitial lung disease |
| IS | immunosuppression |
| LGE | late gadolinium enhancement |
| LVEF | left ventricle ejection fraction |
| mRSS | modified Rodnan skin score |
| NBD | normal blood donors |
| NICD | non-inflammatory cardiac disease |
| nSVT | non-sustained ventricular tachycardia |
| NT-proBNP | type B pro-natriuretic peptide |
| NVC | nailfold videocapillaroscopy |
| NYHA | New York heart association |
| OR | odds ration |
| PAPs | pulmonary artery pressure in systole |
| PH | pulmonary hypertension |
| RNA | ribonucleic acid |
| RVEF | right ventricle ejection fraction |
| SD | standard deviation |
| SIDs | systemic immune-mediated diseases |
| SSc | systemic sclerosis |
| STIR | short T inversion recovery |
| TAPSE | tricuspid Annulus Plane Systolic Excursion |
| VEDOSS | very early diagnosis of systemic sclerosis |
References
- Bournia, V.-K.; Tountas, C.; Protogerou, A.D.; Panopoulos, S.; Mavrogeni, S.; Sfikakis, P.P. Update on assessment and management of primary cardiac involvement in systemic sclerosis. J. Scleroderma Relat. Disord. 2018, 3, 53–65. [Google Scholar] [CrossRef]
- Moore, D.F.; Steen, V.D. Overall mortality. J. Scleroderma Relat. Disord. 2020, 6, 3–10. [Google Scholar] [CrossRef]
- Bruni, C.; Ross, L. Cardiac involvement in systemic sclerosis: Getting to the heart of the matter. Best Pract. Res. Clin. Rheumatol. 2021, 35, 101668. [Google Scholar] [CrossRef]
- Bruni, C.; Buch, M.H.; Furst, D.E.; De Luca, G.; Djokovic, A.; Dumitru, R.B.; Giollo, A.; Polovina, M.; Steelandt, A.; Bratis, K.; et al. Primary systemic sclerosis heart involvement: A systematic literature review and preliminary data-driven, consensus-based WSF/HFA definition. J. Scleroderma Relat. Disord. 2021. [Google Scholar] [CrossRef]
- Mavrogeni, S.; Gargani, L.; Pepe, A.; Monti, L.; Markousis-Mavrogenis, G.; De Santis, M.; De Marchi, D.; Koutsogeorgopoulou, L.; Karabela, G.; Stavropoulos, E.; et al. Cardiac magnetic resonance predicts ventricular arrhythmias in scleroderma: The Scleroderma Arrhythmia Clinical Utility Study (SAnCtUS). Rheumatology 2020, 59, 1938–1948. [Google Scholar]
- Follansbee, W.P.; Zerbe, T.R.; Medsger, T.A. Cardiac and skeletal muscle disease in systemic sclerosis (scleroderma): A high risk association. Am. Heart J. 1993, 125, 194–203. [Google Scholar] [CrossRef]
- Venalis, P.; Kumánovics, G.; Schulze-Koops, H.; Distler, A.; Dees, C.; Zerr, P.; Palumbo-Zerr, K.; Czirják, L.; Mackevic, Z.; Lundberg, I.E.; et al. Cardiomyopathy in murine models of systemic sclerosis. Arthritis Rheumatol. 2015, 67, 508–516. [Google Scholar] [CrossRef]
- Varga, J.; Trojanowska, M.; Kuwana, M. Pathogenesis of systemic sclerosis: Recent insights of molecular and cellular mechanisms and therapeutic opportunities. J. Scleroderma Relat. Disord. 2017, 2, 137–152. [Google Scholar] [CrossRef]
- Mueller, K.A.; Mueller, I.I.; Eppler, D.; Zuern, C.S.; Seizer, P.; Kramer, U.; Koetter, I.; Roecken, M.; Kandolf, R.; Gawaz, M.; et al. Clinical and histopathological features of patients with systemic sclerosis undergoing endomyocardial biopsy. PLoS ONE 2015, 10, e0126707. [Google Scholar]
- Pieroni, M.; De Santis, M.; Zizzo, G.; Bosello, S.; Smaldone, C.; Campioni, M.; De Luca, G.; Laria, A.; Meduri, A.; Bellocci, F.; et al. Recognizing and treating myocarditis in recent-onset systemic sclerosis heart disease: Potential utility of immunosuppressive therapy in cardiac damage progression. Semin. Arthritis Rheum. 2014, 43, 526–533. [Google Scholar] [CrossRef]
- De Luca, G.; Campochiaro, C.; De Santis, M.; Sartorelli, S.; Peretto, G.; Sala, S.; Canestrari, G.; De Lorenzis, E.; Basso, C.; Rizzo, S.; et al. Systemic sclerosis myocarditis has unique clinical, histological and prognostic features: A comparative histological analysis. Rheumatology 2020, 59, 2523–2533. [Google Scholar] [CrossRef]
- Caforio, A.L.; Pankuweit, S.; Arbustini, E.; Basso, C.; Gimeno-Blanes, J.; Felix, S.B.; Fu, M.; Heliö, T.; Heymans, S.; Jahns, R.; et al. European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013, 34, 2636–2648. [Google Scholar]
- Caforio, A.; Adler, Y.; Agostini, C.; Allanore, Y.; Anastasakis, A.; Arad, M.; Böhm, M.; Charron, P.; Elliott Pm Eriksson, U.; Felix Sb Garcia-Pavia, P.; et al. Diagnosis and management of myocardial involvement in systemic immune-mediated diseases: A position statement of the ESC Working Group on Myocardial and Pericardial Disease. Eur. Heart J. 2017, 38, 2649–2662. [Google Scholar] [CrossRef]
- Peretto, G.; Sala, S.; De Luca, G.; Campochiaro, C.; Sartorelli, S.; Cappelletti, A.M.; Rizzo, S.; Palmisano, A.; Esposito, A.; Margonato, A.; et al. Impact of systemic immune-mediated diseases on clinical features and prognosis of patients with biopsy-proved myocarditis. Int. J. Cardiol. 2019, 280, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Bosello, S.; De Luca, G.; Berardi, G.; Canestrari, G.; de Waure, C.; Gabrielli, F.A.; Di Mario, C.; Forni, F.; Gremese, E.; Ferraccioli, G. Cardiac troponin T and NT-proBNP as diagnostic and prognostic biomarkers of primary cardiac involvement and disease severity in systemic sclerosis: A prospective study. Eur. J. Intern. Med. 2019, 60, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Allanore, Y.; Meune, C.; Vonk, M.C.; Airo, P.; Hachulla, E.; Caramaschi, P.; Riemekasten, G.; Cozzi, F.; Beretta, L.; Derk, C.T.; et al. Prevalence and factors associated with left ventricular dysfunction in the EULAR Scleroderma Trial and Research group (EUSTAR) database of patients with systemic sclerosis. Ann. Rheum. Dis. 2010, 69, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Tzelepis, G.E.; Kelekis, N.L.; Plastiras, S.C.; Mitseas, P.; Economopoulos, N.; Kampolis, C.; Gialafos, E.J.; Moyssakis, I.; Moutsopoulos, H.M. Pattern and distribution of myocardial fibrosis in systemic scleroderma, a delayed enhanced MRI study. Arthritis Rheum. 2007, 56, 3827–3836. [Google Scholar] [CrossRef]
- Hachulla, A.L.; Launay, D.; Gaxotte, V.; de Groote, P.; Lamblin, N.; Devos, P.; Hatron, P.Y.; Beregi, J.P.; Hachulla, E. Cardiac magnetic resonance imaging in systemic sclerosis: A cross-sectional observational study of 52 patients. Ann. Rheum. Dis. 2009, 68, 1878–1884. [Google Scholar] [CrossRef]
- Gargani, L.; Todiere, G.; Guiducci, S.; Bruni, C.; Pingitore, A.; De Marchi, D.; Bellando Randone, S.; Aquaro, G.D.; Bazzichi, L.; Mosca, M.; et al. Early Detection of Cardiac Involvement in Systemic Sclerosis: The Added Value of Magnetic Resonance Imaging. JACC Cardiovasc. Imaging. 2019, 12, 927–928. [Google Scholar] [CrossRef]
- Pingitore, A.; Guiducci, S.; Conforti, M.L.; De Marchi, D.; Gargani, L.; Moggi-Pignone, A.; Randone, S.B.; Lombardi, M.; Picano, E.; Matucci-Cerinic, M. Early detection of myocardial and pulmonary oedema with MRI in an asymptomatic systemic sclerosis patient: Successful recovery with pulse steroid. Rheumatology 2013, 52, 1920–1921. [Google Scholar] [CrossRef][Green Version]
- Caforio, A.L.; Bonifacio, E.; Stewart, J.T.; Neglia, D.; Parodi, O.; Bottazzo, G.F.; Mckenna, W.J. Novel organ-specific circulating cardiac autoantibodies in dilated cardiomyopathy. J. Am. Coll Cardiol. 1990, 15, 1527–1534. [Google Scholar] [CrossRef]
- Caforio, A.L.; Mahon, N.G.; Baig, M.K.; Tona, F.; Murphy, R.T.; Elliott, P.M.; McKenna, W.J. Prospective Familial Assessment in Dilated Cardiomyopathy. Circulation 2007, 115, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Caforio, A.L.; Calabrese, F.; Angelini, A.; Tona, F.; Vinci, A.; Bottaro, S.; Ramondo, A.; Carturan, E.; Iliceto, S.; Thiene, G.; et al. A prospective study of biopsy-proven myocarditis: Prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur. Heart J. 2007, 28, 1326–1333. [Google Scholar] [CrossRef]
- Peretto, G.; Sala, S.; De Luca, G.; Marcolongo, R.; Campochiaro, C.; Sartorelli, S.; Tresoldi, M.; Foppoli, L.; Palmisano, A.; Esposito, A.; et al. Immunosuppressive Therapy and Risk Stratification of Patients With Myocarditis Presenting With Ventricular Arrhythmias. JACC Clin. Electrophysiol. 2020, 6, 1221–1234. [Google Scholar] [CrossRef]
- Caforio, A.L.P.; Baritussio, A.; Marcolongo, R.; Cheng, C.Y.; Pontara, E.; Bison, E.; Cattini, M.G.; Gallo, N.; Plebani, M.; Iliceto, S.; et al. Serum Anti-Heart and Anti-Intercalated Disk Autoantibodies: Novel Autoimmune Markers in Cardiac Sarcoidosis. J. Clin. Med. 2021, 10, 2476. [Google Scholar] [CrossRef] [PubMed]
- Minier, T.; Guiducci, S.; Bellando-Randone, S.; Bruni, C.; Lepri, G.; Czirják, L.; Distler, O.; Walker, U.A.; Fransen, J.; Allanore, Y.; et al. Preliminary analysis of the very early diagnosis of systemic sclerosis (VEDOSS) EUSTAR multicentre study: Evidence for puffy fingers as a pivotal sign for suspicion of systemic sclerosis. Ann. Rheum. Dis. 2014, 73, 2087–2093. [Google Scholar] [CrossRef]
- Van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013, 65, 2737–2747. [Google Scholar] [CrossRef] [PubMed]
- LeRoy, E.C.; Medsger, T.A., Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 2001, 28, 1573–1576. [Google Scholar] [PubMed]
- Ruaro, B.; Nallino, M.G.; Casabella, A.; Salton, F.; Confalonieri, P.; De Tanti, A.; Bruni, C. Monitoring the microcirculation in the diagnosis and follow-up of systemic sclerosis patients: Focus on pulmonary and peripheral vascular manifestations. Microcirculation 2020, 27, e12647. [Google Scholar] [CrossRef]
- Caforio, A.L.P.; Re, F.; Avella, A.; Marcolongo, R.; Baratta, P.; Seguso, M.; Gallo, N.; Plebani, M.; Izquierdo-Bajo, A.; Cheng, C.Y.; et al. Evidence from family studies for autoimmunity in arrhythmogenic right ventricular cardiomyopathy: Associations of circulating anti-heart and anti-intercalated disk autoantibodies with disease severity and family history. Circulation 2020, 141, 1238–1248. [Google Scholar] [CrossRef]
- Galiè, N.; Humbert, M.; Vachiery, J.L.; Gibbs, S.; Lang, I.; Torbicki, A.; Simonneau, G.; Peacock, A.; Vonk Noordegraaf, A.; Beghetti, M.; et al. 2015 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: The joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Heart J. 2016, 37, 67–119. [Google Scholar]
- Blagova, O.; Varionchik, N.; Zaidenov, V.; Savina, P.; Sarkisova, N. Anti-heart antibodies levels and their correlation with clinical symptoms and outcomes in patients with confirmed or suspected diagnosis COVID-19. Eur. J. Immunol. 2021, 51, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Caforio, A.L.P.; Angelini, A.; Blank, M.; Shani, A.; Kivity, S.; Goddard, G.; Doria, A.; Bottaro, S.; Marcolongo, R.; Thiene, G.; et al. Passive Transfer of Affinity-Purified Anti-Heart Autoantibodies (AHA) from Sera of Patients with Myocarditis Induces Experimental Myocarditis in Mice. Int. J. Cardiol. 2015, 179, 166–177. [Google Scholar] [CrossRef] [PubMed]
- Caforio, A.L.P.; Grazzini, M.; Mann, J.M.; Keeling, P.J.; Bottazzo, G.F.; McKenna, W.J.; Schiaffino, S. Identification of alpha- and beta-cardiac myosin heavy chain isoforms as major autoantigens in dilated cardiomyopathy. Circulation 1992, 85, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Ciaffi, J.; van Leeuwen, N.M.; Huizinga, T.W.J.; de Vries-Bouwstra, J.K. Smoking and systemic sclerosis: Influence on microangiopathy and expression of anti-topoisomerase I antibodies in a monocentric cohort. Clin. Exp. Rheumatol. 2020, 38 (Suppl. S125), 25–28. [Google Scholar]
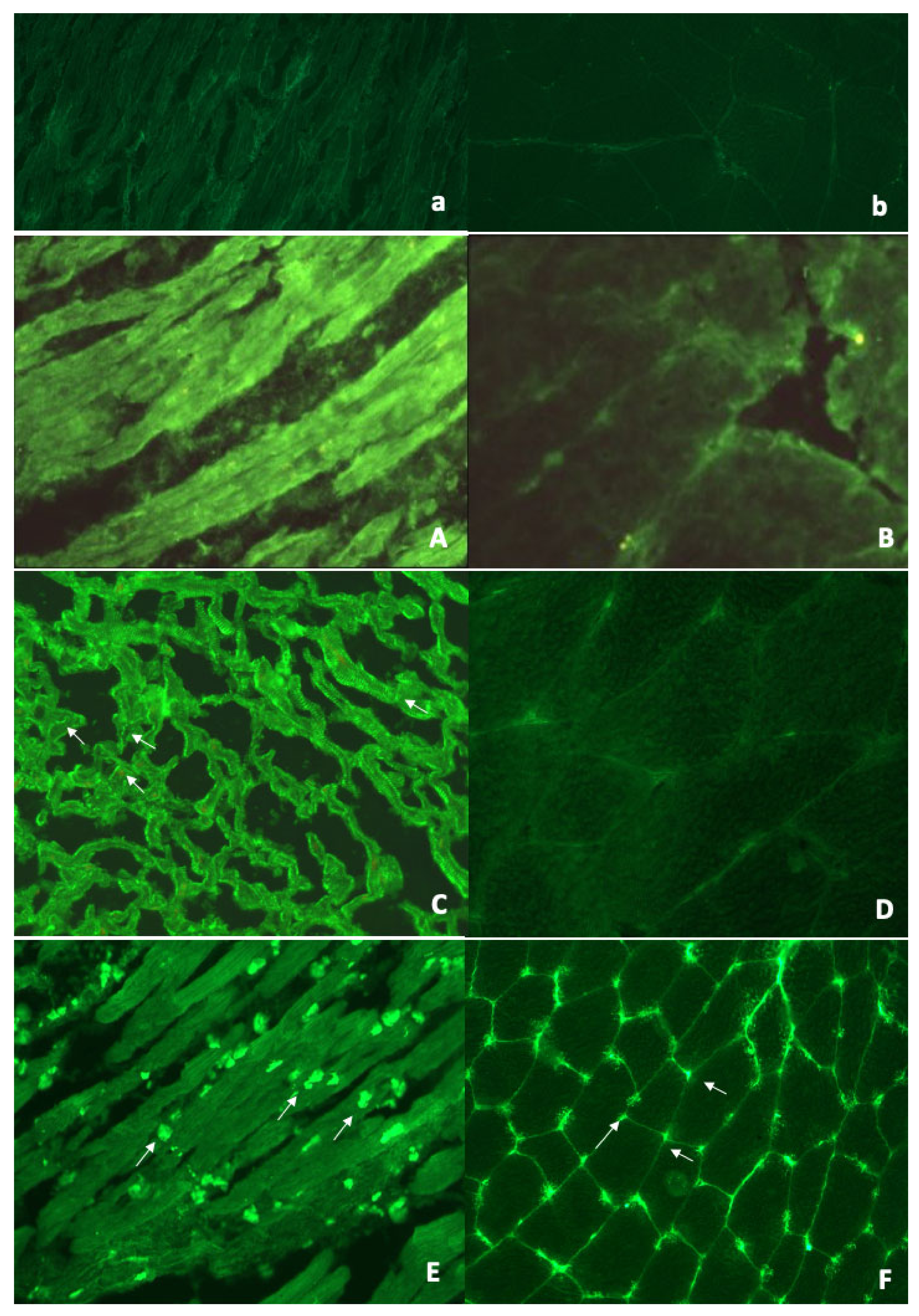
| n = 116 | |
|---|---|
| Female gender, n (%) | 97 (83.6) |
| Age, years | 53 ± 13 |
| Median disease duration (range), years | 7 (0–41) |
| Previous smoker, n (%) | 55 (47) |
| SSc types (limited; diffuse; sine scleroderma) n (%) | 77 (66); 36 (31); 3 (3) |
| mRSS (data available, n = 105), median, (range) | 2 (0–35) |
| VEDOSS status (yes) n (%) | 28 (24) |
| Digital ulcers (at any time, previous or current) (n = 115) | 42 (36) |
| NVC pattern (n = 113): Early, n (%); Active, n (%); Late, n (%) | 31 (27); 48 (42); 23 (21) |
| ILD on HRCT (yes), (n = 105), n (%) | 55 (52) |
| Syncope (yes), n (%) | 2 (2) |
| Palpitation (yes), n (%) | 19 (16) |
| Chest pain (yes), n (%) | 5 (4) |
| Advanced NYHA class (III and IV) at diagnosis, n (%) | 8 (6) |
| Patients with supraventricular ectopic beats on ECG Holter monitoring (n = 78), n (%) | 53 (67.9) |
| Patients with supraventricular tachycardia on ECG Holter monitoring (n = 77), n (%) | 12 (15.5) |
| Patients with ventricular ectopic beats on ECG Holter monitoring (n = 77), n (%) | 51 (66) |
| Patients with ventricular couplets on 24 h ECG Holter monitoring (n = 78), n (%) | 15 (19%) |
| Ventricular ectopic beats total number on 24-h ECG Holter monitoring (n = 67), median (range) | 4 (0–32,024) |
| LVEF Echo, (n = 113), mean ± SD | 63 ± 6 |
| TAPSE, mm (n = 84), mean ± SD | 22 ± 3 |
| PAPs on Echo, mmHg, (n = 68), mean ± SD | 26 ± 6 |
| LVEF% at CMR, (n = 116), mean ± SD | 65 ± 7 |
| RVEF% at CMR, (n = 112), mean ± SD | 65 ± 7 |
| Presence of LGE by CMR, (n = 114), n (%) | 47 (40.5) |
| Presence of myocardial oedema by T2 STIR, (n = 115), n (%) | 15 (13) |
| Oedema distribution on CMR, (n = 13) concordant with LGE site, another site from LGE, n (%) | 7 (54); 6 (46) |
| Presence of pericardial oedema by T2 STIR, (n = 115), n (%) | 4 (3.5) |
| Pericardial effusion by CMR, (n = 115), n (%) | 22 (19) |
| NT-proBNP, (n = 87), median (range) | 110 (10–2374) |
| Creatinine clearance, (n = 76), mean ± SD | 94 ± 29 |
| High sensitivity troponin I, (n = 55), median (range) | 0.26 (0–490) |
| AHA positivity, n (%) | 57 (49) |
| AIDA positivity, n (%) | 65 (56) |
| ATA positivity, (n = 114) n (%) | 41 (36) |
| ACA positivity, (n = 113) n (%) | 39 (34.5) |
| ANA positivity, (n = 114) n (%) | 109 (95.6) |
| Currently on IS (yes), n (%) | 36 (31) |
| Current IS type, (n = 36), n (%): methotrexate; azathioprine; cyclophosphamide; mycophenolate mofetil; biologic agents | 8 (22);2 (5.5); 2 (5.5);16 (44%); 8 (22) |
| Past IS (yes), n (%) | 13 (11) |
| Past IS type, (n = 13), n (%): methotrexate; azathioprine; cyclophosphamide; mycophenolate mofetil; biologic agents | 4 (31); 1 (7.6); 5 (38); 1 (7.6%); 2 (15) |
| Past steroid therapy, n (%): | 15 (12.9) |
| AHA Positive n = 57 | AHA Negative n = 59 | p-Value | |
|---|---|---|---|
| Age, mean ± SD, years | 51.8 ± 13 | 54 ± 13 | 0.814 |
| Female gender, n (%) | 51 (89.5) | 46 (78) | 0.097 |
| Smoking exposure, n (%) | 34 (59.6) | 21(35.6) | 0.009 |
| SSc types (limited; diffuse; sine scleroderma) n | 37/18/2 | 40/18/1 | 0.93 |
| VEDOSS status (yes) n (%) | 11 (19.3) | 17 (28.8) | 0.230 |
| Digital ulcers (at any time, previous or current) (n = 115) | 25 (43.9) | 17 (29.3) | 0.102 |
| NVC pattern (n = 113): Early; Active, Late; | 13/14/26 | 18/9/22 | 0.2208 |
| ILD on HRCT (yes), (n = 105), n (%) | 33 (62.3) | 22 (42.3) | 0.041 |
| Syncope (yes), n (%) | 1 (1.8) | 1 (1.7) | >0.999 |
| Palpitation (yes), n (%) | 10 (17.5) | 9 (15.3) | 0.733 |
| Chest pain (yes), n (%) | 5 (8.8) | 0 (0) | 0.026 |
| NYHA class (I-II, III, and IV) at diagnosis (n = 115) | 51/5/1 | 56/2/0 | 0.285 |
| Presence of LGE by CMR, (n = 114), n (%) | 25 (44.6) | 22 (37.9) | 0.597 |
| Presence of myocardial oedema by T2 STIR, (n = 115), n (%) | 9 (16.4) | 6 (10.2) | 0.320 |
| Presence of pericardial oedema by T2 STIR, (n = 115), n (%) | 4 (7.1) | 0 (0) | 0.053 |
| Pericardial effusion by CMR, (n = 115), n (%) | 13 (23.2) | 9 (15.3) | 0.345 |
| Patients with ventricular ectopic beats on ECG Holter monitoring (n = 77), n (%) | 23 (63.9) | 28 (68.3) | 0.680 |
| High sensitivity troponin I, (n = 55), median (range) | 63 (10–121) | 25 (1–59) | 0.006 |
| ATA positivity, (n = 114) n (%) | 25 (44.6) | 16 (27.6) | 0.057 |
| AIDA positivity, n (%) | 48 (84) | 17 (28.8) | <0.001 |
| ACA positivity, (n = 113) n (%) | 15 (27.3) | 24 (41.4) | 0.118 |
| ANA positivity, (n = 114) n (%) | 52 (92.9) | 57 (98.3) | 0.204 |
| Currently on IS (yes), n (%) | 24 (42.1) | 12 (20.3) | 0.011 |
| Currently on Prostanoids (n = 115) (yes), n (%) | 19 (33.9) | 11 (18.6) | 0.062 |
| AIDA Positive n = 65 | AIDA Negative n = 51 | p-Value | |
|---|---|---|---|
| Age, years | 52.6 ± 12 | 53.2 ± 14 | 0.812 |
| Female gender, n (%) | 58 (89.2) | 39 (76.5) | 0.067 |
| Smoking exposure, n (%) | 38 (58.5) | 17(33.3) | 0.004 |
| SSc types (limited; diffuse; sine scleroderma) n | 45/19/1 | 32/17/2 | 0.619 |
| VEDOSS status (yes) n (%) | 14 (21.5) | 14 (27.5) | 0.460 |
| Digital ulcers (at any time, previous or current) (n = 115) | 28 (43.8) | 14 (27.5) | 0.071 |
| NVC pattern (n = 113): Early; Active, Late. | 17/15/29 | 14/8/19 | 0.056 |
| ILD on HRCT (yes), (n = 105), n (%) | 34 (57.6) | 21 (45.7) | 0.226 |
| Syncope (yes), n (%) | 1 (1.5) | 1 (2) | >0.999 |
| Palpitation (yes), n (%) | 7 (10.8) | 12 (23.5) | 0.065 |
| Chest pain (yes), n (%) | 4 (6.2) | 1 (2) | 0.388 |
| NYHA class (I-II, III, and IV) at diagnosis (n = 115) | 58/6/0 | 49/1/1 | 0.142 |
| Presence of LGE on CMR, (n = 114), n (%) | 25 (53.2) | 22 (46,8) | 0.566 |
| Presence of myocardial oedema by CMR, (n = 115), n (%) | 10 (15.6) | 5 (10.0) | 0.371 |
| Presence of pericardial oedema by CMR, (n = 115), n (%) | 4 (6.2) | 0 (0) | 0.135 |
| Pericardial effusion by CMR, (n = 115), n (%) | 14 (21.5) | 8 (16.0) | 0.485 |
| Patients with ventricular ectopic beats on ECG Holter monitoring (n = 77), n (%) | 22 (52.4) | 29 (82.3) | 0.007 |
| ATA positivity, (n = 114) n (%) | 27 (42.9) | 14 (27.5) | 0.080 |
| ACA positivity, (n = 113) n (%) | 19 (30.6) | 20 (39.2) | 0.345 |
| ANA positivity, (n = 114) n (%) | 62 (96.9) | 47 (94.0) | 0.657 |
| Currently on IS (yes), n (%) | 22 (33.8) | 14 (27.5) | 0.469 |
| Currently on Prostanoids (n = 115) (yes), n (%) | 24 (37.5) | 6 (11.8) | 0.002 |
| AHA Positive n = 56 | AHA Negative n = 59 | p-Value | |
|---|---|---|---|
| Any cardiac event, n (%) | 7 (11.9%) | 8 (14.6%) | 0.785 |
| Pulmonary arterial hypertension, n (%) | 0 (0) | 1 (1.7) | >0.999 |
| Right heart failure, n (%) | 1 (1.8) | 0 (0) | 0.482 |
| Left heart failure, n (%) | 2 (3.6) | 0 (0) | 0.234 |
| Non sustained ventricular tachycardia, n (%) | 2 (3.6) | 1 (1.7) | 0.616 |
| Implantable cardioverter defibrillator for sustained ventricular tachycardia, n (%) | 1 (1.8) | 2 (3.4) | >0.999 |
| Coronary artery disease, n (%) | 1 (1.8) | 2 (3.4) | >0.999 |
| Any other arrhythmia (yes), n (%) | 6 (10.7) | 2 (3.4) | 0.123 |
| Death, n (%) | 5 (8.9) | 0 (0) | 0.021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caforio, A.L.P.; De Luca, G.; Baritussio, A.; Seguso, M.; Gallo, N.; Bison, E.; Cattini, M.G.; Pontara, E.; Gargani, L.; Pepe, A.; et al. Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates. Diagnostics 2021, 11, 2165. https://doi.org/10.3390/diagnostics11112165
Caforio ALP, De Luca G, Baritussio A, Seguso M, Gallo N, Bison E, Cattini MG, Pontara E, Gargani L, Pepe A, et al. Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates. Diagnostics. 2021; 11(11):2165. https://doi.org/10.3390/diagnostics11112165
Chicago/Turabian StyleCaforio, Alida Linda Patrizia, Giacomo De Luca, Anna Baritussio, Mara Seguso, Nicoletta Gallo, Elisa Bison, Maria Grazia Cattini, Elena Pontara, Luna Gargani, Alessia Pepe, and et al. 2021. "Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates" Diagnostics 11, no. 11: 2165. https://doi.org/10.3390/diagnostics11112165
APA StyleCaforio, A. L. P., De Luca, G., Baritussio, A., Seguso, M., Gallo, N., Bison, E., Cattini, M. G., Pontara, E., Gargani, L., Pepe, A., Campochiaro, C., Plebani, M., Iliceto, S., Peretto, G., Esposito, A., Tofani, L., Moggi-Pignone, A., Dagna, L., Marcolongo, R., ... Bruni, C. (2021). Serum Organ-Specific Anti-Heart and Anti-Intercalated Disk Autoantibodies as New Autoimmune Markers of Cardiac Involvement in Systemic Sclerosis: Frequency, Clinical and Prognostic Correlates. Diagnostics, 11(11), 2165. https://doi.org/10.3390/diagnostics11112165












