Development of an Easy-to-Use Prediction Equation for Body Fat Percentage Based on BMI in Overweight and Obese Lebanese Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Source and Study Population
2.3. Ethics Approval
2.4. Anthropometric Measurements
2.5. Statistical Analysis
2.5.1. Descriptive Statistics
2.5.2. Predictors to Be Included in the Model
2.5.3. Model Derivation
2.5.4. Evaluation of Model Performance and Internal Validity and External Validity
3. Results
3.1. Characteristics of the Study Participants
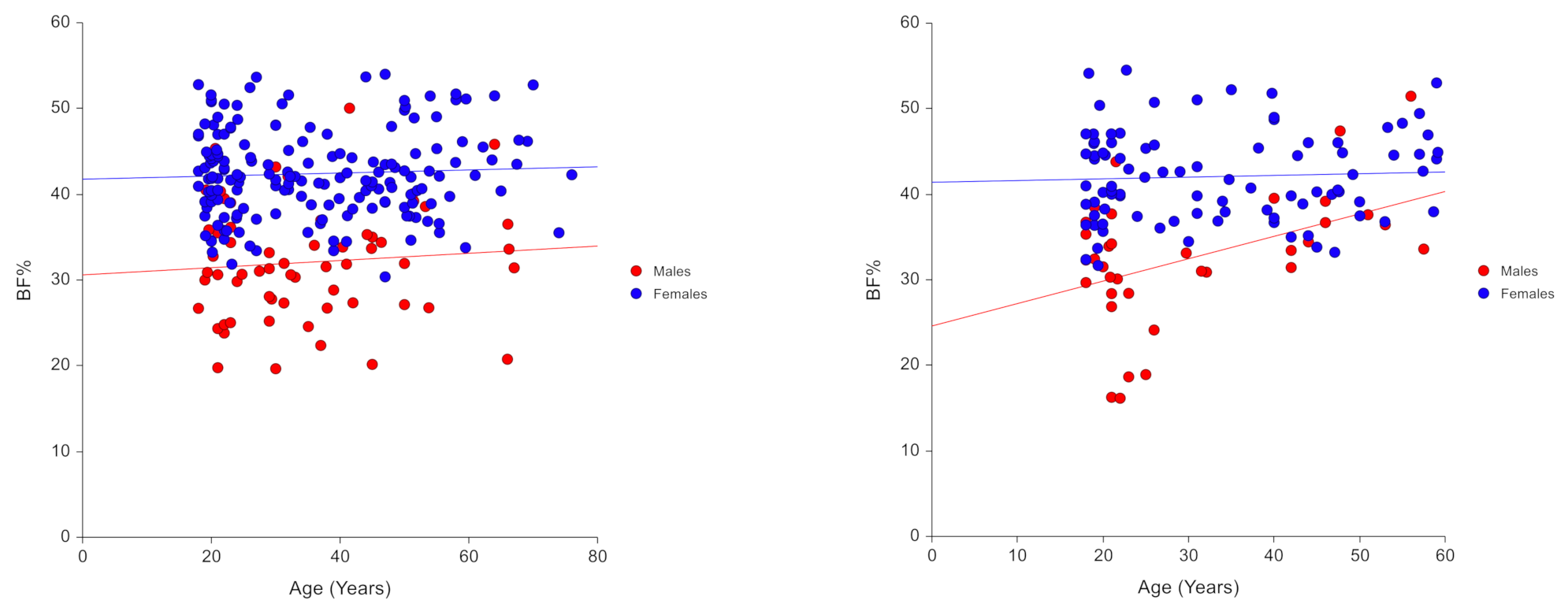
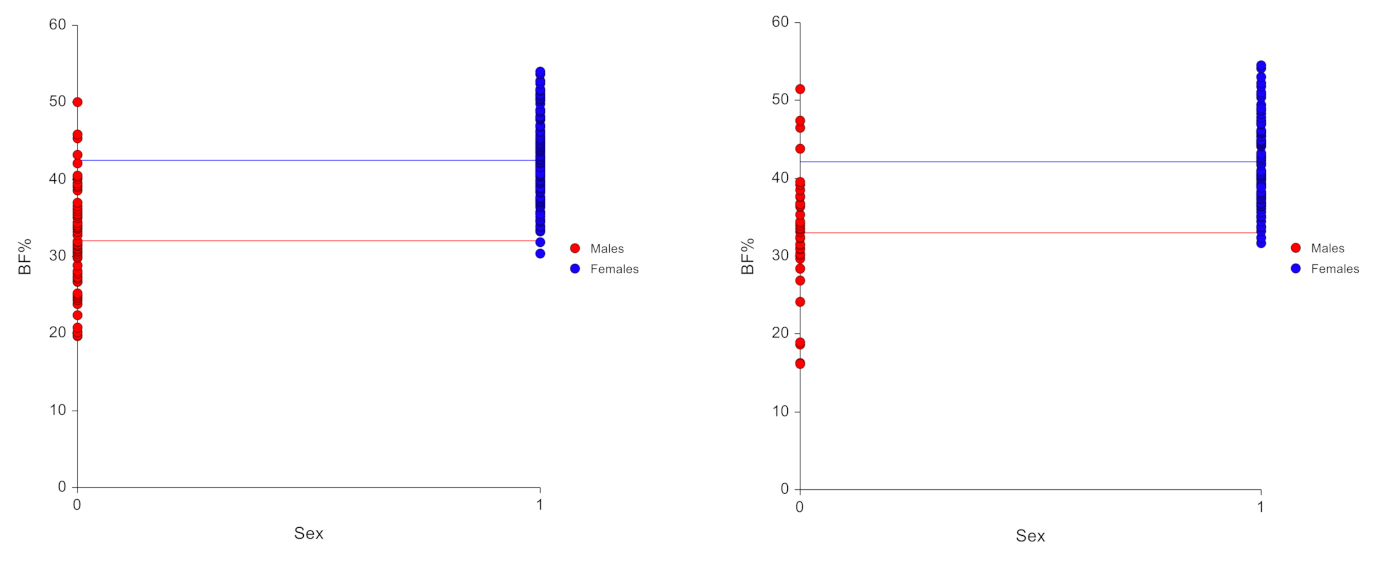
3.2. Model Predictors
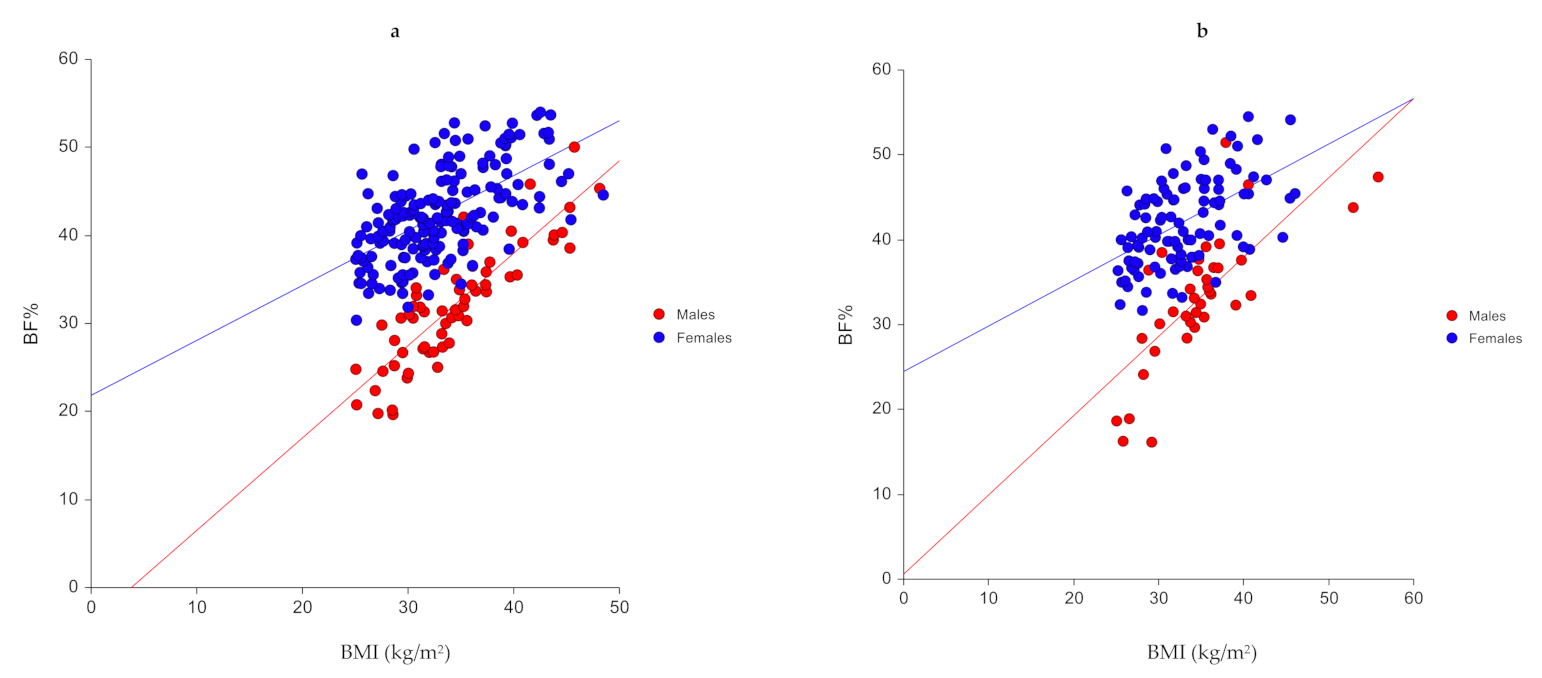
3.3. Derived Model
- BF% females = 0.624 × BMI + 21.835
- BF% males = 1.050 × BMI − 4.001
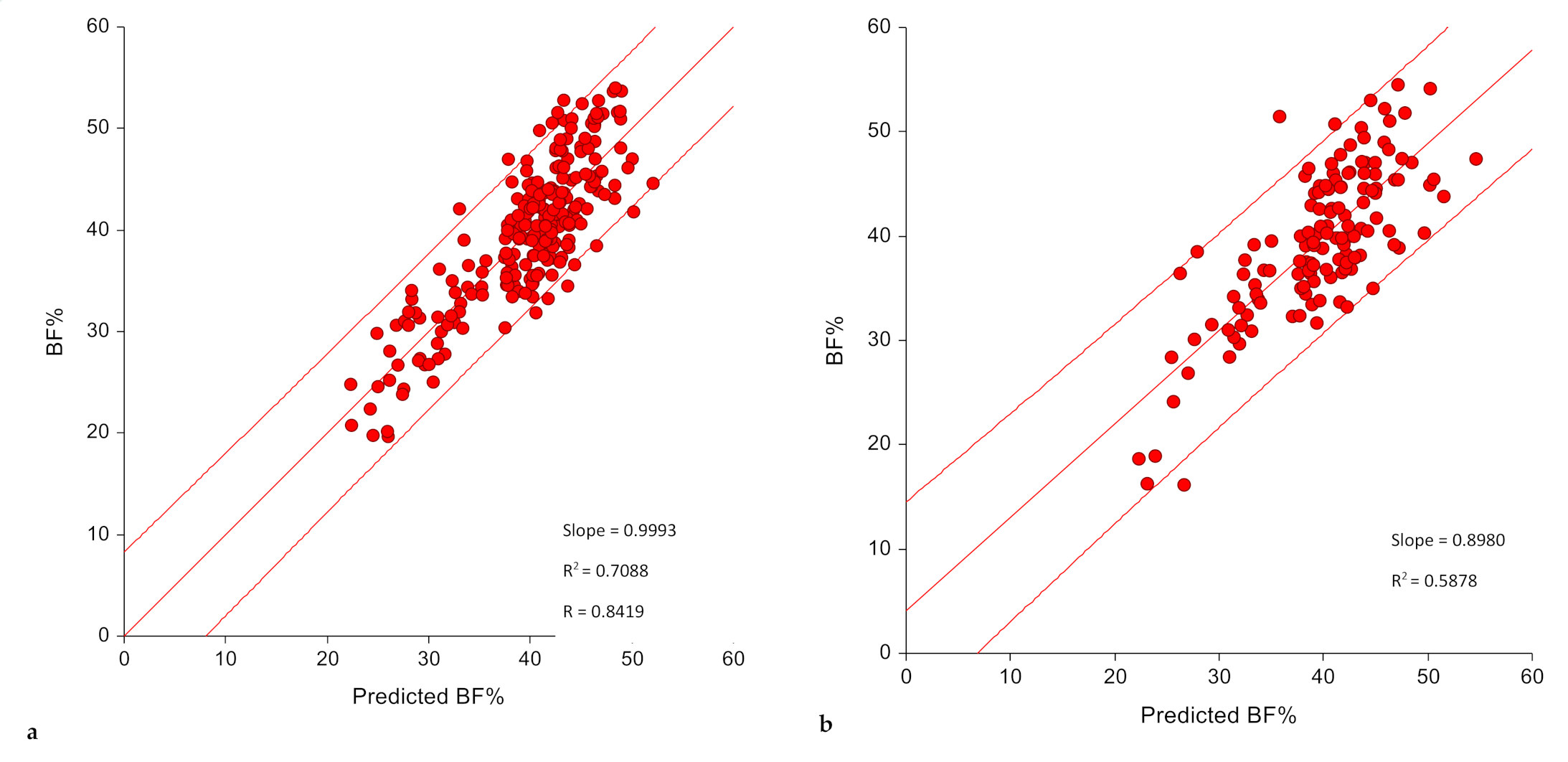
3.4. Model Performamce
4. Discussion
4.1. Findings and Concordance with Previous Studies
4.2. Potential Clinical Implications
4.3. Strengths and Limitations
4.4. New Directions and Areas for Future Research
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organisation (WHO). Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 20 April 2020).
- Maimoun, L.; Mura, T.; Avignon, A.; Mariano-Goulart, D.; Sultan, A. Body Composition in Individuals with Obesity According to Age and Sex: A Cross-Sectional Study. J. Clin. Med. 2020, 9, 1188. [Google Scholar] [CrossRef]
- Pasco, J.A.; Holloway, K.L.; Dobbins, A.G.; Kotowicz, M.A.; Williams, L.J.; Brennan, S.L. Body mass index and measures of body fat for defining obesity and underweight: A cross-sectional, population-based study. BMC Obes. 2014, 1, 9. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Dong, S.Y.; Sun, X.N.; Xie, J.; Cui, Y. Percent body fat is a better predictor of cardiovascular risk factors than body mass index. Braz. J. Med. Biol. Res. 2012, 45, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Padwal, R.; Leslie, W.D.; Lix, L.M.; Majumadar, S.R. Relationship Among Body Fat Percentage, Body Mass Index, and All-Cause Mortality: A Cohort Study. Ann. Intern. Med. 2016, 164, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Thibault, R.; Pichard, C. The evaluation of body composition: A useful tool for clinical practice. Ann. Nutr. Metab. 2012, 60, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Gallagher, D. Assessment methods in human body composition. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Duren, D.L.; Sherwood, R.J.; Czerwinski, S.A.; Lee, M.; Choh, A.C.; Siervogel, R.M.; Cameron Chumlea, W. Body composition methods: Comparisons and interpretation. J. Diabetes. Sci. Technol. 2008, 2, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Lemos, T.; Gallagher, D. Current body composition measurement techniques. Curr. Opin. Endocrinol. Diabetes. Obes. 2017, 24, 310–314. [Google Scholar] [CrossRef]
- Boneva-Asiova, Z.; Boyanov, M.A. Body composition analysis by leg-to-leg bioelectrical impedance and dual-energy X-ray absorptiometry in non-obese and obese individuals. Diabetes. Obes. Metab. 2008, 10, 1012–1018. [Google Scholar] [CrossRef]
- Pateyjohns, I.R.; Brinkworth, G.D.; Buckley, J.D.; Noakes, M.; Clifton, P.M. Comparison of three bioelectrical impedance methods with DXA in overweight and obese men. Obesity (Silver Spring) 2006, 14, 2064–2070. [Google Scholar] [CrossRef]
- Thomson, R.; Brinkworth, G.D.; Buckley, J.D.; Noakes, M.; Clifton, P.M. Good agreement between bioelectrical impedance and dual-energy X-ray absorptiometry for estimating changes in body composition during weight loss in overweight young women. Clin. Nutr. 2007, 26, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Achamrah, N.; Colange, G.; Delay, J.; Rimbert, A.; Folope, V.; Petit, A.; Grigioni, S.; Déchelotte, P.; Coëffier, M. Comparison of body composition assessment by DXA and BIA according to the body mass index: A retrospective study on 3655 measures. PLoS ONE 2018, 137, e0200465. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, A.; Garaci, F.; Cafarelli, F.P.; Guglielmi, G. Body composition in clinical practice. Eur. J. Radiol. 2016, 85, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Womersley, J. A comparison of the skinfold method with extent of ‘overweight’ and various weight-height relationships in the assessment of obesity. Br. J. Nutr. 1977, 38, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.S.; Stanforth, P.R.; Gagnon, J.; Rankinen, T.; Leon, A.S.; Rao, D.C.; Skinner, J.S.; Bouchard, C.; Wilmore, J.H. The effect of sex, age and race on estimating percentage body fat from body mass index: The Heritage Family Study. Int. J. Obes. Relat. Metab. Disord. 2002, 26, 789–796. [Google Scholar] [CrossRef]
- Deurenberg, P.; Weststrate, J.A.; Seidell, J.C. Body mass index as a measure of body fatness: Age- and sex-specific prediction formulas. Br. J. Nutr. 1991, 65, 105–114. [Google Scholar] [CrossRef]
- Gallagher, D.; Heymsfield, S.B.; Heo, M.; Jebb, S.A.; Murgatroyd, P.R.; Sakamoto, Y. Healthy percentage body fat ranges: An approach for developing guidelines based on body mass index. Am. J. Clin. Nutr. 2000, 72, 694–701. [Google Scholar] [CrossRef]
- Gomez-Ambrosi, J.; Silva, C.; Catalan, V.; Rodriguez, A.; Galofre, J.C.; Escalada, J.; Valenti, V.; Rotellar, F.; Romero, S.; Ramirez, B.; et al. Clinical usefulness of a new equation for estimating body fat. Diabetes. Care 2012, 35, 383–388. [Google Scholar] [CrossRef]
- Nickerson, B.S.; McLester, C.N.; McLester, J.R.; Kliszczewicz, B.M. Relative accuracy of anthropometric-based body fat equations in males and females with varying BMI classifications. Clin. Nutr. ESPEN 2020, 35, 136–140. [Google Scholar] [CrossRef]
- Moons, K.G.; Altman, D.G.; Reitsma, J.B.; Ioannidis, J.P.; Macaskill, P.; Steyerberg, E.W.; Vickers, A.J.; Ransohoff, D.F.; Collins, G.S. Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): Explanation and elaboration. Ann. Intern. Med. 2015, 162, W1–W73. [Google Scholar] [CrossRef]
- Expert Panel on Detection Evaluation; Treatment of High Blood Cholesterol in A. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- El Ghoch, M.; Bazzani, P.V.; Calugi, S.; Dalle Grave, R. Weight-Loss Cognitive-Behavioural Treatment and Essential Amino Acid Supplementation in a Patient with Spinal Muscular Atrophy and Obesity. Case. Rep. Med. 2018, 2018, 4058429. [Google Scholar] [CrossRef] [PubMed]
- El Masri, D.; Itani, L.; Kreidieh, D.; Tannir, H.; El Ghoch, M. Relationship between multiple weight cycles and early weight loss in patients with obesity: A longitudinal study. Eat Weight Disord. 2020. [CrossRef]
- Tannir, H.; Itani, L.; El Masri, D.; Kreidieh, D.; El Ghoch, M. Lifetime Weight Cycling and Central Fat Distribution in Females With Obesity: A Brief Report. Diseases 2020, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Verney, J.; Metz, L.; Chaplais, E.; Cardenoux, C.; Pereira, B.; Thivel, D. Bioelectrical impedance is an accurate method to assess body composition in obese but not severely obese adolescents. Nutr. Res. 2016, 36, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Verney, J.; Schwartz, C.; Amiche, S.; Pereira, B.; Thivel, D. Comparisons of a Multi-Frequency Bioelectrical Impedance Analysis to the Dual-Energy X-Ray Absorptiometry Scan in Healthy Young Adults Depending on their Physical Activity Level. J. Hum. Kinet. 2015, 47, 73–80. [Google Scholar] [CrossRef]
- Lohman, T.G. Skinfolds and body density and their relation to body fatness: A review. Hum. Biol. 1981, 53, 181–225. [Google Scholar]
- Garn, S.M.; Leonard, W.R.; Hawthorne, V.M. Three limitations of the body mass index. Am. J. Clin. Nutr. 1986, 44, 996–997. [Google Scholar] [CrossRef]
- Misra, A. Revisions of cutoffs of body mass index to define overweight and obesity are needed for the Asian-ethnic groups. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 1294–1296. [Google Scholar] [CrossRef]
- Kuczmarski, R.J.; Fanelli, M.T.; Koch, G.G. Ultrasonic assessment of body composition in obese adults: Overcoming the limitations of the skinfold caliper. Am. J. Clin. Nutr. 1987, 45, 717–724. [Google Scholar] [CrossRef]
- Damilakis, J.; Adams, J.E.; Guglielmi, G.; Link, T.M. Radiation exposure in X-ray-based imaging techniques used in osteoporosis. Eur. Radiol. 2010, 20, 2707–2714. [Google Scholar] [CrossRef] [PubMed]
- Voogt, M.P.; Opmeer, B.C.; Kastelein, A.W.; Jaspers, M.W.M.; Peute, L.W. Obstacles to Successful Implementation of eHealth Applications into Clinical Practice. Stud. Health Technol. Inform. 2018, 247, 521–525. [Google Scholar] [PubMed]
- Melchionda, N.; Marchesini, G.; Apolone, G.; Cuzzolaro, M.; Mannucci, E.; Grossi, E. The QUOVADIS Study: Features of obese Italian patients seeking treatment at specialist centers. Diab. Nutr. Metab. 2003, 16, 115–124. [Google Scholar]
- Liao, Y.S.; Li, H.C.; Lu, H.K.; Lai, C.L.; Wang, Y.S.; Hsieh, K.C. Comparison of Bioelectrical Impedance Analysis and Dual Energy X-ray Absorptiometry for Total and Segmental Bone Mineral Content with a Three-Compartment Model. Int. J. Environ. Res. Public Health 2020, 17, 2595. [Google Scholar] [CrossRef]
- Kim, H.; Kim, C.H.; Kim, D.W.; Park, M.; Park, H.S.; Min, S.S.; Han, S.H.; Yee, J.Y.; Chung, S.; Kim, C. External cross-validation of bioelectrical impedance analysis for the assessment of body composition in Korean adults. Nutr. Res. Pract. 2011, 5, 246–252. [Google Scholar] [CrossRef]
- Kushner, R.F.; Kunigk, A.; Alspaugh, M.; Andronis, P.T.; Leitch, C.A.; Schoeller, D.A. Validation of bioelectrical-impedance analysis as a measurement of change in body composition in obesity. Am. J. Clin. Nutr. 1990, 52, 219–223. [Google Scholar] [CrossRef]
- Savastano, S.; Belfiore, A.; Di Somma, C.; Mauriello, C.; Rossi, A.; Pizza, G.; De Rosa, A.; Prestieri, G.; Angrisani, L.; Colao, A. Validity of bioelectrical impedance analysis to estimate body composition changes after bariatric surgery in premenopausal morbidly women. Obes. Surg. 2010, 20, 332–339. [Google Scholar] [CrossRef]
- Beeson, W.L.; Batech, M.; Schultz, E.; Salto, L.; Firek, A.; Deleon, M.; Balcazar, H.; Cordero-Macintyre, Z. Comparison of body composition by bioelectrical impedance analysis and dual-energy X-ray absorptiometry in Hispanic diabetics. Int. J. Body Compos. Res. 2010, 8, 45–50. [Google Scholar]
- Macek, P.; Biskup, M.; Terek-Derszniak, M.; Krol, H.; Smok-Kalwat, J.; Gozdz, S.; Zak, M. Optimal cut-off values for anthropometric measures of obesity in screening for cardiometabolic disorders in adults. Sci. Rep. 2020, 10, 11253. [Google Scholar] [CrossRef]
- Kim, C.H.; Park, H.S.; Park, M.; Kim, H.; Kim, C. Optimal cutoffs of percentage body fat for predicting obesity-related cardiovascular disease risk factors in Korean adults. Am. J. Clin. Nutr. 2011, 94, 34–39. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Wang, K.; Wang, W.; Dong, F.; Qian, Y.; Gong, H.; Xu, G.; Li, G.; Pan, L.; et al. Optimal body fat percentage cut-off values for identifying cardiovascular risk factors in Mongolian and Han adults: A population-based cross-sectional study in Inner Mongolia, China. BMJ Open. 2017, 7, e014675. [Google Scholar] [CrossRef] [PubMed]
- Ho-Pham, L.T.; Campbell, L.V.; Nguyen, T.V. More on body fat cutoff points. Mayo Clin. Proc. 2011, 86, 584–585. [Google Scholar] [CrossRef] [PubMed]

| Total n = 375 | Training Sample n = 238 | Validation Sample n = 137 | Significance | |
|---|---|---|---|---|
| Sex | n (%) | χ2 = 0.085; p = 0.770 | ||
| Males | 98 (26.1) | 61 (25.6) | 37 (27.0) | |
| Females | 277 (73.9) | 177 (74.4) | 100 (73.0) | |
| Age (Years) | 35.55 (15.12) | 35.77 (14.72) | 35.18 (15.85) | p = 0.723 |
| BMI (kg/m2) | 33.39 (5.26) | 33.38 (5.16) | 33.39 (5.45) | p = 0.986 |
| χ2 = 0.077; p = 0.781 | ||||
| With overweight | 109 (29.1) | 68 (28.6) | 41 (29.9) | |
| With obesity | 266 (70.9) | 170 (71.4) | 96 (70.1) | |
| BF% measured | 39.80 (7.37) | 39.78 (7.15) | 39.84 (7.77) | p = 0.939 |
| Predictors | Training Sample n = 238 | Validation Sample n = 137 |
|---|---|---|
| Age | 0.069 | 0.242 ** |
| BMI | 0.461 ** | 0.413 ** |
| Sex | 0.635 ** | 0.560 ** |
| Equation | R2 | |||||
|---|---|---|---|---|---|---|
| Training Sample | SEE | TE | Slope | Intercept | p-Value * | |
| Model 1 | 0.718 | |||||
| Model 2 | 0.709 | 3.81 | 3.84 | 1.010 | 0.179 | 0.402 |
| Validation sample ¥ | 3.88 | 3.85 | 0.999 | 0 | 0.981 | |
| Model 1 | 0.561 | |||||
| Model 2 | 0.588 | 4.79 | 4.82 | 0.894 | 4.60 | 0.060 |
| Measured BF% | BF% 1 | Mean Difference (Bias) | 95% CI of Bias (Precision) | Pearson’s Correlation | % Bias | Minimum % Bias | Maximum % Bias | Absolute Percent Error | N (%) Underprediction 2 | N (%) Accurate Prediction 3 | N (%) OverPrediction 4 | % Error | Upper LoA 5 | Lower LoA | Effect Size | p−Value 6 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Training sample | 39.78 ± 7.15 | ||||||||||||||||
| Model 1 | 39.20 ± 5.99 | 0.584 ± 3.81 | 0.098; 1.07 | 0.85 | −0.53 ± 9.88 | −22.92 | 31.27 | 7.91 ± 5.93 | 18.1 | 67.6 | 14.3 | 38.04 | 8.04 | −6.87 | 0.153 | 0.019 | |
| Model 2 | 39.80 ± 6.02 | −0.017 ± 3.86 | −0.51; 0.48 | 0.84 | 1.04 ± 10.10 | −21.53 | 32.25 | 8.16 ± 6.01 | 17.2 | 69.3 | 13.4 | 38.00 | 7.54 | −7.58 | 0.004 | 0.946 | |
| Validation sample | 39.67 ± 7.21 | ||||||||||||||||
| Model 1 | 39.24 ± 6.04 | 0.425 ± 4.82 | −0.39; 1.24 | 0.75 | −0.44 ± 4.82 | −33.22 | 67.16 | 10.11 ± 9.15 | 19.7 | 59.1 | 21.2 | 47.62 | 9.87 | −9.02 | 0.088 | 0.303 | |
| Model 2 | 39.70 ± 6.15 | −0.028 ± 4.67 | −0.82; 0.76 | 0.77 | 1.49 ± 13.14 | −30.46 | 64.95 | 9.85 ± 8.78 | 17.5 | 60.6 | 21.9 | 46.13 | 9.12 | −9.18 | 0.006 | 0.943 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Itani, L.; Tannir, H.; El Masri, D.; Kreidieh, D.; El Ghoch, M. Development of an Easy-to-Use Prediction Equation for Body Fat Percentage Based on BMI in Overweight and Obese Lebanese Adults. Diagnostics 2020, 10, 728. https://doi.org/10.3390/diagnostics10090728
Itani L, Tannir H, El Masri D, Kreidieh D, El Ghoch M. Development of an Easy-to-Use Prediction Equation for Body Fat Percentage Based on BMI in Overweight and Obese Lebanese Adults. Diagnostics. 2020; 10(9):728. https://doi.org/10.3390/diagnostics10090728
Chicago/Turabian StyleItani, Leila, Hana Tannir, Dana El Masri, Dima Kreidieh, and Marwan El Ghoch. 2020. "Development of an Easy-to-Use Prediction Equation for Body Fat Percentage Based on BMI in Overweight and Obese Lebanese Adults" Diagnostics 10, no. 9: 728. https://doi.org/10.3390/diagnostics10090728
APA StyleItani, L., Tannir, H., El Masri, D., Kreidieh, D., & El Ghoch, M. (2020). Development of an Easy-to-Use Prediction Equation for Body Fat Percentage Based on BMI in Overweight and Obese Lebanese Adults. Diagnostics, 10(9), 728. https://doi.org/10.3390/diagnostics10090728







