Dry Eye in Systemic Sclerosis Patients: Novel Methods to Monitor Disease Activity
Abstract
1. Introduction
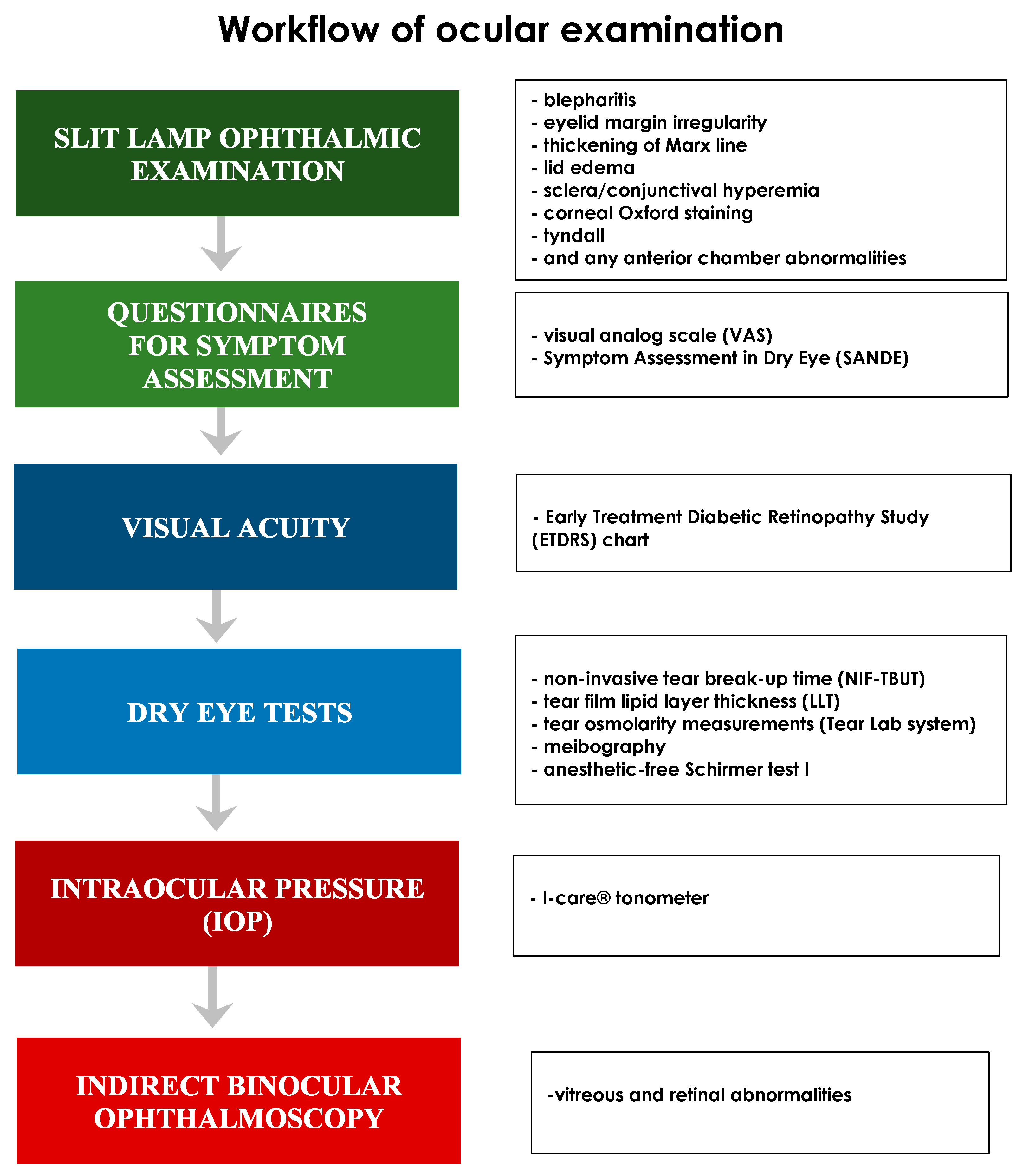
2. Materials and Methods
Statistical Analysis
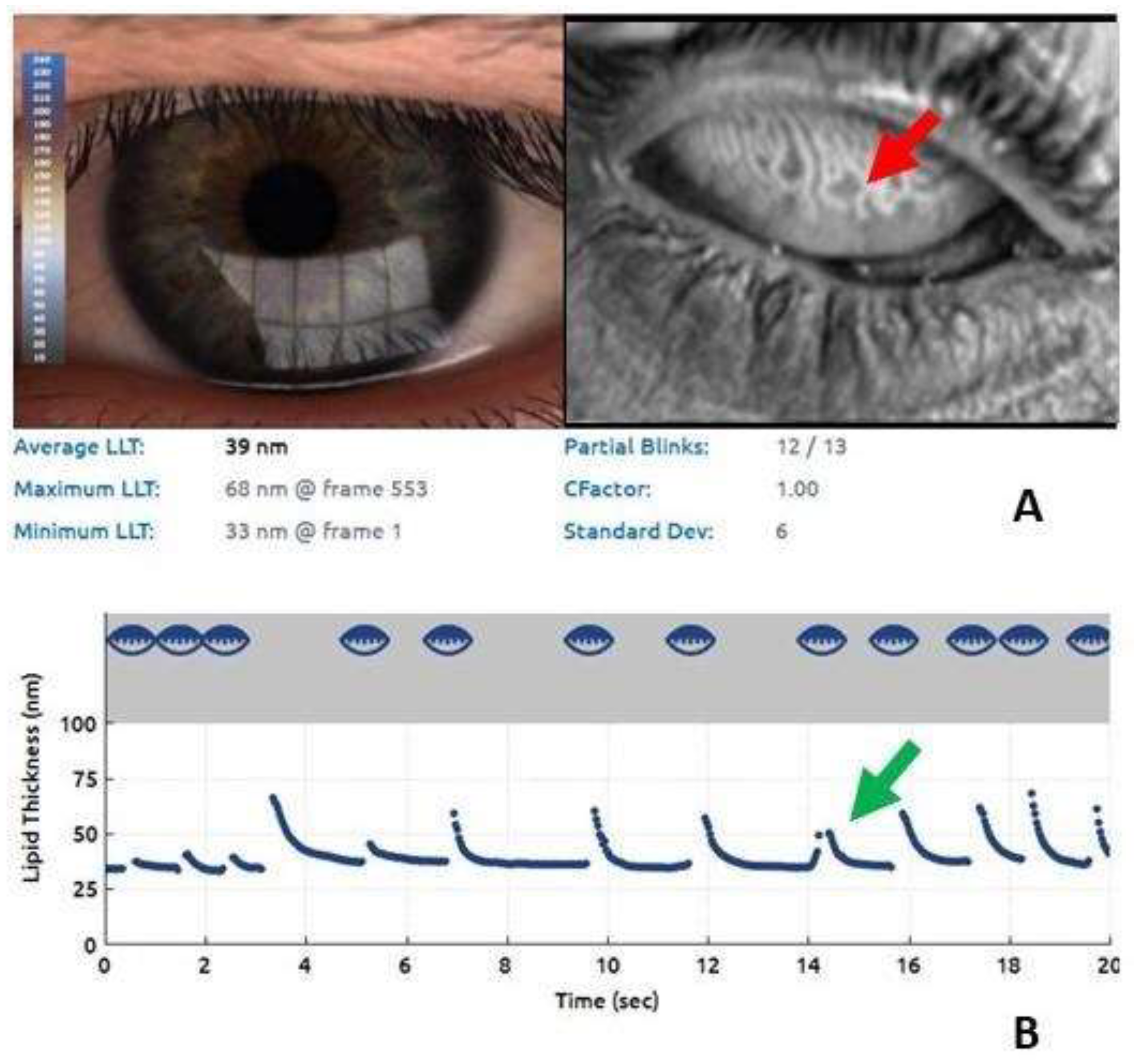
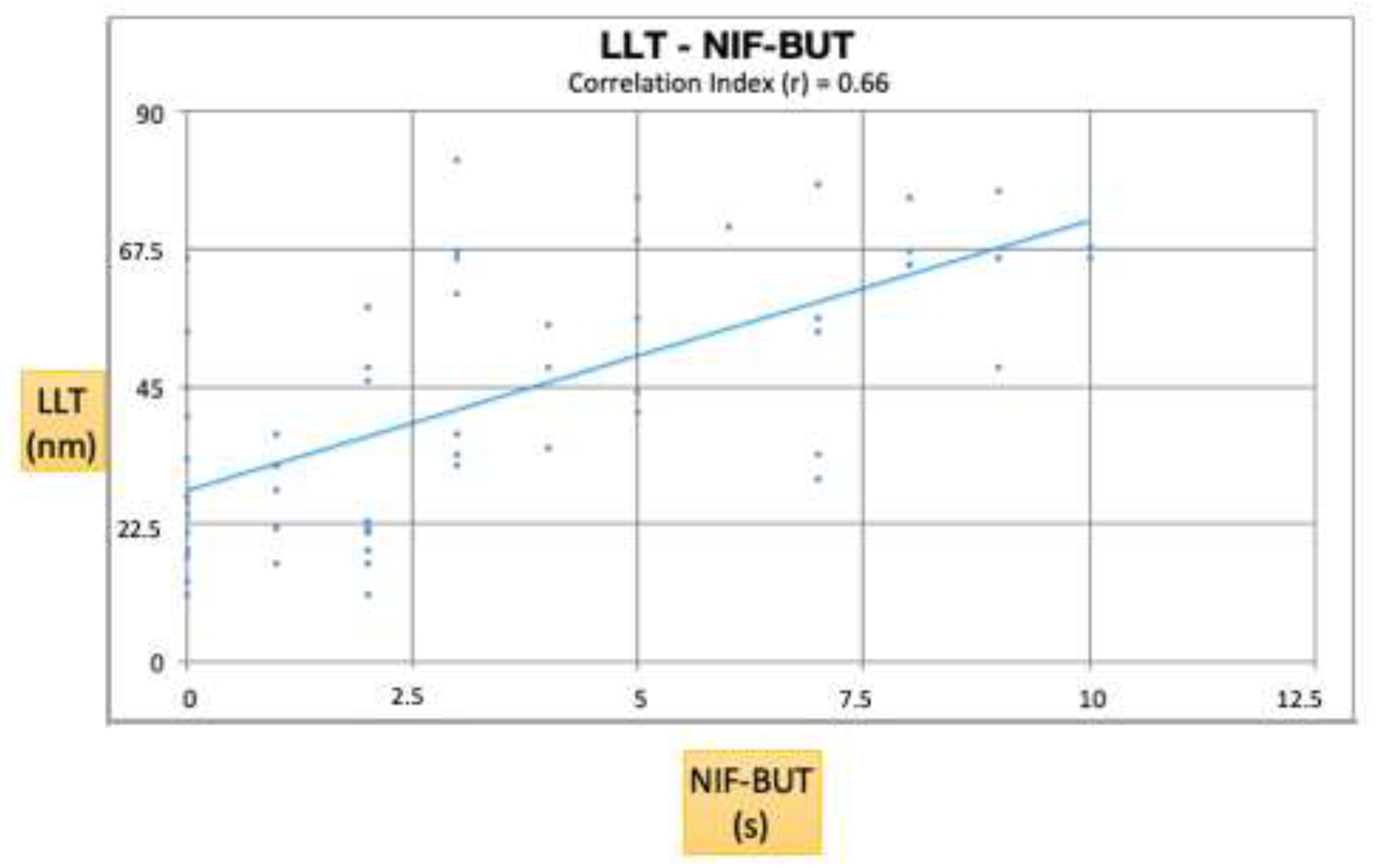
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Asano, Y. Systemic sclerosis. J. Dermatol 2018, 45, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Pattanaik, D.; Brown, M.; Postlethwaite, B.C.; Postlethwaite, A.E. Pathogenesis of Systemic Sclerosis. Front. Immunol. 2015, 6, 272. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.; Pauling, J.D.; Armstrong-James, L.; Denton, C.P.; Galdas, P.; Flurey, C. Gender-related Differences in Systemic Sclerosis. Autoimmun Rev. 2020, 19, 102494. [Google Scholar] [CrossRef] [PubMed]
- Kreps, E.O.; Carton, C.; Cutolo, M.; Cutolo, C.A.; Vanhaecke, A.; Leroy, B.P.; Smith, V. Ocular involvement in systemic sclerosis: A systematic literature review, it’s not all scleroderma that meets the eye. Semin Arthritis Rheum. 2019, 49, 119–125. [Google Scholar] [CrossRef]
- Jimenez, S.A. Role of endothelial to mesenchymal transition in the pathogenesis of the vascular alterations in systemic sclerosis. ISRN Rheumatol. 2013, 835948. [Google Scholar] [CrossRef]
- Wollheim, F.A. Classification of systemic sclerosis. Visions and reality. Rheumatology 2005, 44, 1212–1216. [Google Scholar] [CrossRef]
- Koenig, M.; Joyal, F.; Fritzler, M.J.; Roussin, A.; Abrahamowicz, M.; Boire, G.; Goulet, J.R.; Rich, E.; Grodzicky, T.; Raymond, Y.; et al. Autoantibodies and microvascular damage are independent predictive factors for the progression of Raynaud’s phenomenon to systemic sclerosis: A twenty-year prospective study of 586 patients, with validation of proposed criteria for early systemic sclerosis. Arthritis Rheum. 2008, 58, 3902–3912. [Google Scholar] [CrossRef]
- Waszczykowska, A.; Goś, R.; Waszczykowska, E.; Dziankowska-Bartkowiak, B.; Jurowski, P. Prevalence of ocular manifestations in systemic sclerosis patients. Arch. Med. Sci. 2013, 9, 1107–1113. [Google Scholar] [CrossRef]
- Szucs, G.; Szekanecz, Z.; Aszalos, Z.; Gesztelyi, R.; Zsuga, J.; Szodoray, P.; Kemeny-Beke, A. A Wide Spectrum of Ocular Manifestations Signify Patients with Systemic Sclerosis. Ocul. Immunol. Inflamm. 2019, 1–9. [Google Scholar] [CrossRef]
- International Dry Eye WorkShop Study Group. The epidemiology of dry eye disease: Report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). Ocul. Surf. 2007, 5, 93–107. [Google Scholar]
- Asbell, P.A.; Stapleton, F.J.; Wickström, K.; Akpek, E.K.; Aragona, P.; Dana, R.; Lemp, M.A.; Nichols, K.K. The International Workshop on Meibomian Gland Dysfunction: Report of the clinical trials subcommittee. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2065–2085. [Google Scholar] [CrossRef]
- American Academy of Ophthalmology Cornea/External Disease Panel. Preferred Practice Pattern Guidelines. Dry Eye Syndrome; American Academy of Ophthalmology: San Francisco, CA, USA, 2013. [Google Scholar]
- Yagci, A.; Gurdal, C. The role and treatment of inflammation in dry eye disease. Int. Ophthalmol. 2014, 34, 1291–1301. [Google Scholar] [CrossRef] [PubMed]
- Bose, T.; Diedrichs-Möhring, M.; Wildner, G. Dry eye disease and uveitis: A closer look at immune mechanisms in animal models of two ocular autoimmune diseases. Autoimmun Rev. 2016, 15, 1181–1192. [Google Scholar] [CrossRef]
- Bron, A.J.; Abelson, M.B.; Ousler, G.; Pearce, E.; Tomlinson, A.; Yokoi, N.; Smith, J.A.; Begley, C.; Caffery, B.; Nichols, K.; et al. Methodologies to diagnose and monitor dry eye disease: Report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop (2007). Ocul. Surf. 2007, 5, 108–152. [Google Scholar]
- Choudhary, M.M.; Hajj-Ali, R.A.; Lowder, C.Y. Gender and Ocular Manifestations of Connective Tissue Diseases and Systemic Vasculitides. J. Ophthalmol. 2014, 2014, 403042. [Google Scholar] [CrossRef]
- LeRoy, E.C.; Medsger, T.A., Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 2001, 28, 1573–1576. [Google Scholar]
- Mazi, A.T. Preliminary criteria for the classification of systemic sclerosis (scleroderma). Subcommittee for scleroderma criteria of the American Rheumatism Association Diagnostic and Therapeutic Criteria Committee. Arthritis Rheum. 1980, 23, 581–590. [Google Scholar]
- Rentka, A.; Nagy, A.; Harsfalvi, J.; Szucs, G.; Szekanecz, Z.; Gesztelyi, R.; Szodoray, P.; Kemeny-Beke, A. Association between objective signs and subjective symptoms of dry eye disease in patients with systemic sclerosis. Rheumatol. Int. 2017, 37, 1835–1845. [Google Scholar] [CrossRef] [PubMed]
- Tashbayev, B.; Utheim, T.P.; Utheim, Ø.A.; Ræder, S.; Jensen, J.L.; Yazdani, M.; Lagali, N.; Vitelli, V.; Dartt, D.A.; Chen, X. Utility of Tear Osmolarity Measurement in Diagnosis of Dry Eye Disease. Sci. Rep. 2020, 10, 5542. [Google Scholar] [CrossRef] [PubMed]
- Lemp, M.A.; Foulks, G.N. The definition and classification of dry eye disease: Report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007). Ocul. Surf. 2007, 5, 75–92. [Google Scholar]
- Klimek, L.; Bergmann, K.C.; Biedermann, T.; Bousquet, J.; Hellings, P.; Jung, K.; Merk, H.; Olze, H.; Schlenter, W.; Stock, P.; et al. Visual analogue scales (VAS): Measuring instruments for the documentation of symptoms and therapy monitoring in cases of allergic rhinitis in everyday health care. Allergo J. Int. 2017, 26, 16–24. [Google Scholar] [CrossRef]
- Amparo, F.; Schaumberg, D.A.; Dana, R. Comparison of Two Questionnaires for Dry Eye Symptom Assessment: The Ocular Surface Disease Index and the Symptom Assessment in Dry Eye. Ophthalmology 2015, 122, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.D.; Whitmer, D.; Nichols, K.K.; Tomlinson, A.; Foulks, G.N.; Geerling, G.; Pepose, J.S.; Kosheleff, V.; Porreco, A.; Lemp, M.A. An objective approach to dry eye disease severity. Investig. Ophthalmol. Vis. Sci. 2010, 51, 6125–6130. [Google Scholar] [CrossRef] [PubMed]
- Periman, L.M.; Perez, V.L.; Saban, D.R.; Lin, M.C.; Neri, P. The Immunological Basis of Dry Eye Disease and Current Topical Treatment Options. J. Ocul. Pharmacol. Ther. 2020, 36, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Versura, P.; Profazio, V.; Campos, E.C. Performance of Tear Osmolarity Compared to Previous Diagnostic Tests for Dry Eye Diseases. Curr. Eye Res. 2010, 35, 553–564. [Google Scholar] [CrossRef]
- Goto, E.; Matsumoto, Y.; Kamoi, M.; Endo, K.; Ishida, R.; Dogru, M.; Kaido, M.; Kojima, T.; Tsubota, K. Tear evaporation rates in Sjögren syndrome and non-Sjögren dry eye patients. Am. J. Ophthalmol. 2007, 144, 81–85. [Google Scholar] [CrossRef]
- Wei, Y.; Asbell, P.A. The core mechanism of dry eye disease is inflammation. Eye Contact Lens 2014, 40, 248–256. [Google Scholar] [CrossRef]
- Geerling, G.; Baudouin, C.; Aragona, P.; Rolando, M.; Boboridis, K.G.; Benítez-Del-Castillo, J.M.; Akova, Y.A.; Merayo-Lloves, J.; Labetoulle, M.; Steinhoff, M.; et al. Emerging strategies for the diagnosis and treatment of meibomian gland dysfunction: Proceedings of the OCEAN group meeting. Ocul. Surf. 2017, 15, 179–192. [Google Scholar] [CrossRef]
- Liu, S.; Dong, H.; Huang, X.H.; Tang, S.H. Analysis of factors leading to lid wiper epitheliopathy. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 1593–1601. [Google Scholar] [CrossRef]
- Hassan, A.; Balal, S.; Ahmad, S. Meibomian gland dysfunction, dropout and distress: Emerging therapies. Eye 2020. [Google Scholar] [CrossRef]
- Li, Y.; Lu, J.; Zhou, Q.; Wang, C.; Zeng, Q.; Chen, T.; Liu, C.; Kang, Y.; Li, S. Analysis of clinical and regional distribution characteristics of Obstructive meibomian gland dysfunction in China: A Multicenter Study. Curr. Eye Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Villarreal-Gonzalez, A.J.; Jocelyn Rivera-Alvarado, I.; Rodriguez-Gutierrez, L.A.; Rodriguez-Garcia, A. Analysis of ocular surface damage and visual impact in patients with primary and secondary Sjögren syndrome. Rheumatol. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; de Paiva, C.S.; Chauhan, S.K.; Bonini, S.; Gabison, E.E.; Jain, S.; Knop, E.; Markoulli, M.; Ogawa, Y.; Perez, V.; et al. TFOS DEWS II Pathophysiology Report. Ocul. Surf. 2017, 15, 438–510. [Google Scholar] [CrossRef]
- Pflugfelder, S.C.; de Paiva, C.S. The pathophysiology of dry eye disease: What we know and future directions for research. Ophthalmology 2017, 124, S4–S13. [Google Scholar] [CrossRef]
- Gagliano, C.; Caruso, S.; Napolitano, G.; Malaguarnera, G.; Cicinelli, M.V.; Amato, R.; Reibaldi, M.; Incarbone, G.; Bucolo, C.; Drago, F.; et al. Low levels of 17-β-oestradiol, oestrone and testosterone correlate with severe evaporative dysfunctional tear syndrome in postmenopausal women: A case-control study. Br. J. Ophthalmol. 2014, 98, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Barboza, M.N.; Barboza, G.N.; de Melo, G.M.; Sato, E.; Dantas, M.C.; Dantas, P.E.; Felberg, S. Correlation between signals and symptoms of dry eye in Sjögren’s syndrome patients. Arq. Bras. Oftalmol. 2008, 71, 547–552. [Google Scholar] [CrossRef]
- Wangkaew, S.; Kasitanon, N.; Sivasomboon, C.; Wichainun, R.; Sukitawut, W.; Louthrenoo, W. Sicca symptoms in Thai patients with rheumatoid arthritis, systemic lupus erythematosus and scleroderma: A comparison with age-matched controls and correlation with disease variables. Asian Pac. J. Allergy Immunol. 2006, 24, 213–221. [Google Scholar]

| Demographic Characteristics | |
| Age (y) (mean ± SD) | 57 ± 14.48 |
| Sex (F/M) | 55/5 |
| Race (%) | Caucasian (100) |
| Weight (kg) (mean ± SD) | 56 ± 6.63 |
| Height (cm) (mean ± SD) | 162.71 ± 5.71 |
| BMI (mean ± SD) | 21.14 ± 2.65 |
| Clinical Characteristics | |
| Modified Rodnan skin score (mRSS) (minimum to maximum average) (mean ± SD) | 2–16 [8.86 ± 3.65] |
| SSc type (n) (%) • Early • Limited • Diffuse | 13 [21.66%] 32 [53.33%] 26 [43.33%] |
| Laboratory Findings | |
| Antinuclear antibodies (ANAs) | 78.33% |
| Anti-centromere antibodies (ACAs) | 55% |
| Anti-Scl-70 antibodies (SCL70) | 50% |
| Ophthalmological Findings | ||
|---|---|---|
| n (%) | ±SD | |
| Meibomian glands disease | 52 (86.66%) | |
| Blepharitis | 39 (65%) | |
| Eyelid margin irregularity | 47 (78.33) | |
| Marx line | 48 (80%) | |
| Lid edema | 5 (8.33%) | |
| Scleral/conjunctival hyperemia | 15 (25%) | |
| Cornea (Oxford staining) | 39 (65%) | |
| Tyndall | 4 (6.66%) | |
| Iris | 5 (8.33%) | |
| Lens | 15 (25%) | |
| Vitreous | 35 (58.33%) | |
| Retina | 7 (11.66) | |
| Best corrected visual acuity (BCVA) | 15 (25%) | |
| IOP (mmHg) | 13.68 | 2.45 |
| VAS | 27 | 9.3 |
| SANDE (frequency) (mean ± SD) | 50.93 | 14.26 |
| SANDE (severity) (mean ± SD) | 48.1 | 6.99 |
| Schirmer test I (mm/5 m) (mean ± SD) | 11.04 | 5.3 |
| NIF-TBUT (sec) (mean ± SD) | 3.4 | 3.1 |
| Tear osmolarity (Tear Lab) (mean ± SD) | 328.51 | 23.8 |
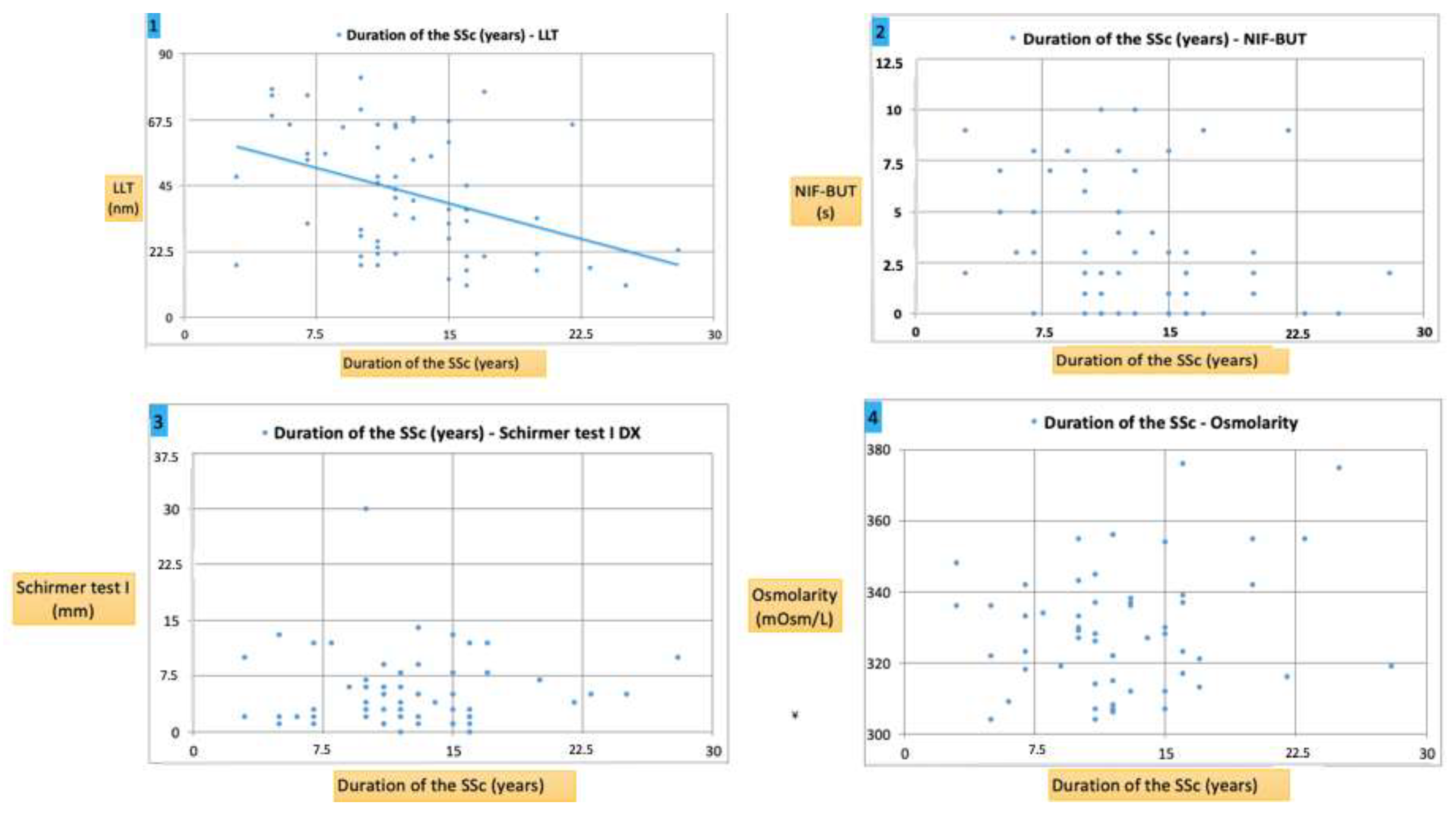
| LLT (nm) (mean ± SD) | 42.95 | 20.91 |
| Group 1 * Mean ± SD | Group 2 ** Mean ± SD | p = Value | ||
|---|---|---|---|---|
| Osmolarity (mOsm/L) | 338.8 ± 14.74 | 333.63 ± 22.65 | 0.94 | NS |
| LLT (nm) | 36.6 ± 21.32 | 44 ± 23.14 | 0.45 | NS |
| NIF-BUT (s) | 2.5 ± 2.71 | 4.09 ± 3.53 | 0.26 | NS |
| Schirmer test I (mm/5 m) | 4.9 ± 4.65 | 3.54 S ± 2.69 | 0.41 | NS |
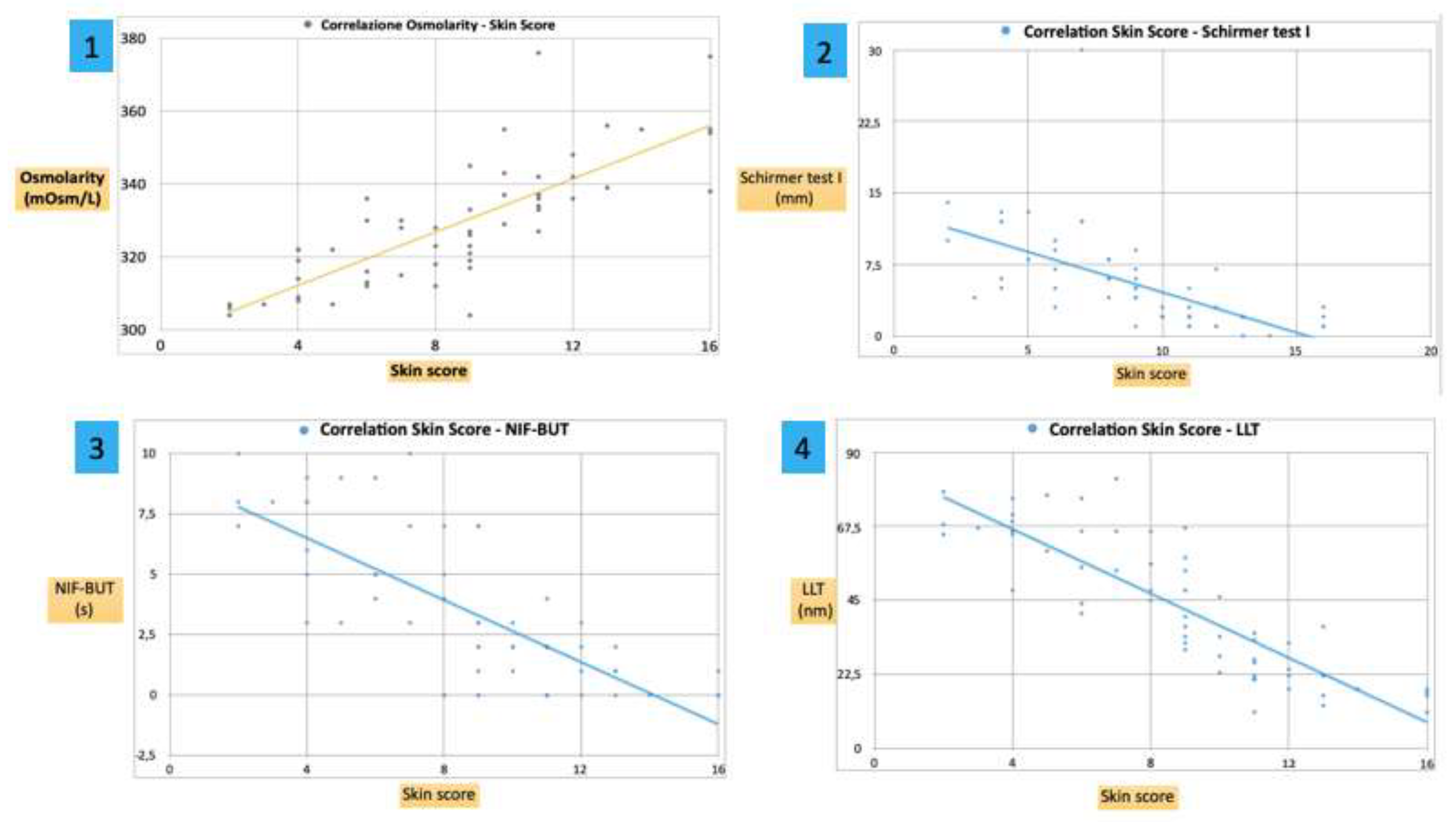
| Linear Correlation Coefficient (Skin Score) | ||||
|---|---|---|---|---|
| r | Significance | p | Correlation | |
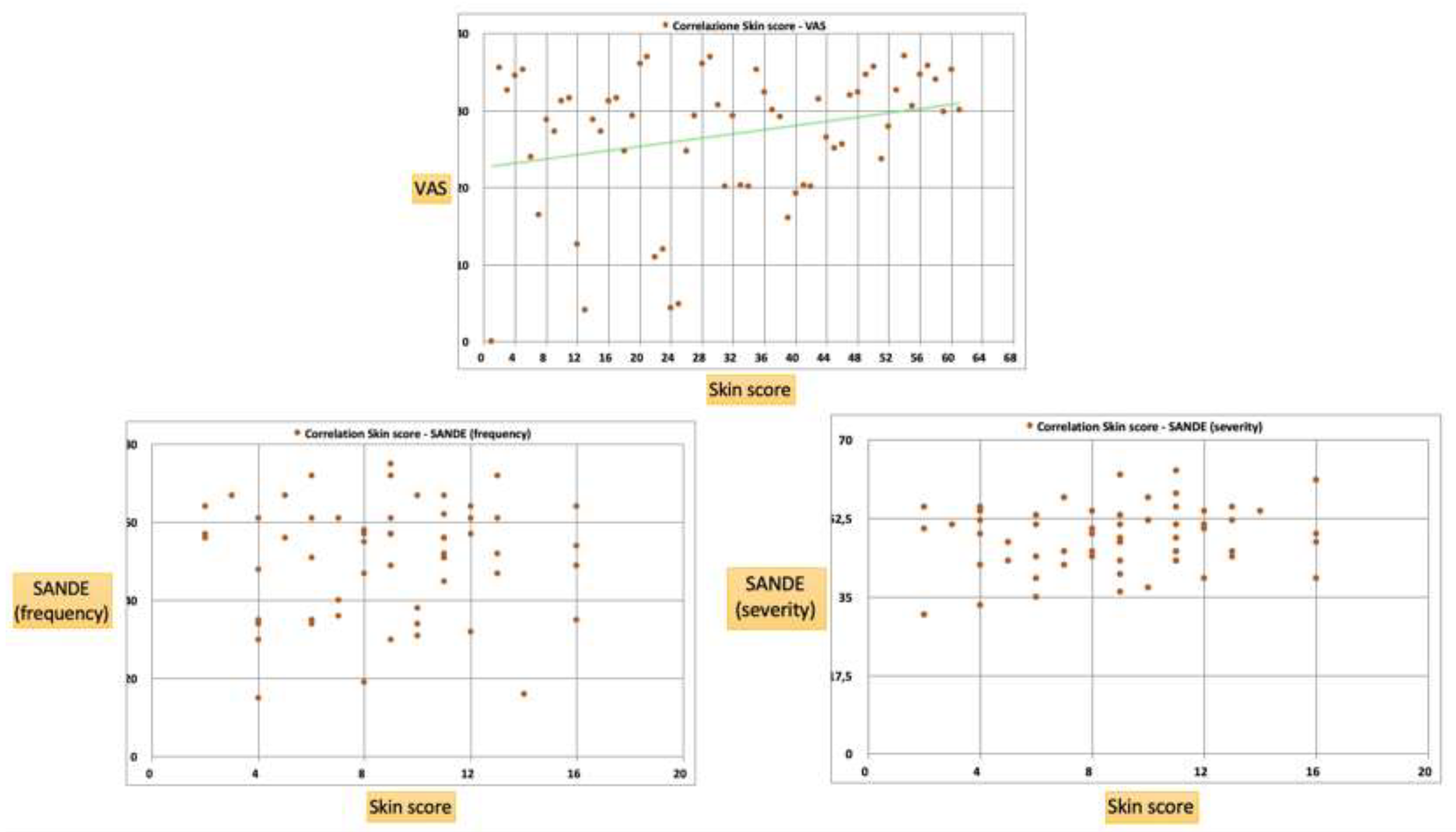
| VAS | 0.38 | S | <0.05 | Positive |
| SANDE (frequency) | 0.03 | NS | >0.05 | No correlation |
| SANDE (severity) | 0.16 | NS | >0.05 | No correlation |
| Schirmer test I (mm) | 0.6 | S | <0.01 | Negative |
| Osmolarity | 0.79 | S | <0.001 | Positive |
| NIF-BUT (sec) | 0.76 | S | <0.01 | Negative |
| LLT (nm) | 0.85 | S | <0.001 | Negative |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gagliano, C.; Visalli, E.; Toro, M.D.; Amato, R.; Panta, G.; Scollo, D.; Scandura, G.; Ficili, S.; Amato, G.; Benenati, A.; et al. Dry Eye in Systemic Sclerosis Patients: Novel Methods to Monitor Disease Activity. Diagnostics 2020, 10, 404. https://doi.org/10.3390/diagnostics10060404
Gagliano C, Visalli E, Toro MD, Amato R, Panta G, Scollo D, Scandura G, Ficili S, Amato G, Benenati A, et al. Dry Eye in Systemic Sclerosis Patients: Novel Methods to Monitor Disease Activity. Diagnostics. 2020; 10(6):404. https://doi.org/10.3390/diagnostics10060404
Chicago/Turabian StyleGagliano, Caterina, Elisa Visalli, Mario Damiano Toro, Roberta Amato, Giovanni Panta, Davide Scollo, Giovanni Scandura, Salvatore Ficili, Giorgio Amato, Alessia Benenati, and et al. 2020. "Dry Eye in Systemic Sclerosis Patients: Novel Methods to Monitor Disease Activity" Diagnostics 10, no. 6: 404. https://doi.org/10.3390/diagnostics10060404
APA StyleGagliano, C., Visalli, E., Toro, M. D., Amato, R., Panta, G., Scollo, D., Scandura, G., Ficili, S., Amato, G., Benenati, A., Foti, R., Malaguarnera, G., Gagliano, G., Falsaperla, R., Avitabile, T., & Foti, R. (2020). Dry Eye in Systemic Sclerosis Patients: Novel Methods to Monitor Disease Activity. Diagnostics, 10(6), 404. https://doi.org/10.3390/diagnostics10060404








