18F-FDG-PET Imaging Patterns in Autoimmune Encephalitis: Impact of Image Analysis on the Results
Abstract
1. Introduction
2. Materials and Methods
2.1. Database Analysis
2.2. FDG-PET Image Analysis
3. Results
3.1. Clinical Findings
3.2. Brain 18F-FDG-PET/CT Findings
3.3. Final Diagnosis after Applying the Proposed Clinical Approach
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Brierley, J.B.; Corsellis, J.A.N.; Hierons, R.; Nevin, S. Subacute encephalitis of later adult life. Mainly affecting the limbic areas. Brain 1960, 83, 357–368. [Google Scholar] [CrossRef]
- Anderson, N.; Barber, P.A. Limbic encephalitis—A review. J. Clin. Neurosci. 2008, 15, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Titulaer, M.J.; Balu, R.; Benseler, S.; Bien, C.G.; Cellucci, T.; Cortese, I.; Dale, R.C.; Gelfand, J.M.; Geschwind, M.; et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016, 15, 391–404. [Google Scholar] [CrossRef]
- Provenzale, J.M.; Barboriak, D.P.; E Coleman, R. Limbic encephalitis: Comparison of FDG PET and MR imaging findings. Am. J. Roentgenol. 1998, 170, 1659–1660. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.Y.; Newberg, A.; Liebeskind, D.S.; Kung, J.; Alavi, A. FDG-PET findings in patients with suspected encephalitis. Clin. Nucl. Med. 2004, 29, 620–625. [Google Scholar] [CrossRef]
- Ances, B.M.; Vitaliani, R.; Taylor, R.A.; Liebeskind, D.S.; Voloschin, A.; Houghton, D.J.; Galetta, S.L.; Dichter, M.; Alavi, A.; Rosenfeld, M.; et al. Treatment-responsive limbic encephalitis identified by neuropil antibodies: MRI and PET correlates. Brain 2005, 128, 1764–1777. [Google Scholar] [CrossRef]
- Fisher, R.E.; Patel, N.R.; Lai, E.C.; Schulz, P.E. Two Different 18F-FDG Brain PET Metabolic Patterns in Autoimmune Limbic Encephalitis. Clin. Nucl. Med. 2012, 37. [Google Scholar] [CrossRef]
- Masangkay, N.; Basu, S.; Moghbel, M.; Kwee, T.; Alavi, A. Brain 18F-FDG-PET characteristics in patients with paraneoplastic neurological syndrome and its correlation with clinical and MRI findings. Nucl. Med. Commun. 2014, 35, 1038–1046. [Google Scholar] [CrossRef]
- Cistaro, A.; Caobelli, F.; Quartuccio, N.; Fania, P.; Pagani, M. Uncommon 18F-FDG-PET/CT findings in patients affected by limbic encephalitis: Hyper-hypometabolic pattern with double antibody positivity and migrating foci of hypermetabolism. Clin. Imaging 2015, 39, 329–333. [Google Scholar] [CrossRef]
- Bien, C.G.; Vincent, A.; Barnett, M.H.; Becker, A.J.; Blümcke, I.; Graus, F.; Jellinger, K.A.; Reuss, D.E.; Ribalta, T.; Schlegel, J.; et al. Immunopathology of autoantibody-associated encephalitides: Clues for pathogenesis. Brain 2012, 135, 1622–1638. [Google Scholar] [CrossRef]
- Hughes, E.G.; Peng, X.; Gleichman, A.J.; Lai, M.; Zhou, L.; Tsou, R.; Parsons, T.D.; Lynch, D.R.; Dalmau, J.; Balice-Gordon, R.J. Cellular and synaptic mechanisms of anti-NMDA receptor encephalitis. J. Neurosci. 2010, 30, 5866–5875. [Google Scholar] [CrossRef] [PubMed]
- Wegner, F.; Wilke, F.; Raab, P.; Ben Tayeb, S.; Boeck, A.-L.; Haense, C.; Trebst, C.; Voss, E.; Schrader, C.; Logemann, F.; et al. Anti-leucine rich glioma inactivated 1 protein and anti-N-methyl-D-aspartate receptor encephalitis show distinct patterns of brain glucose metabolism in 18F-fluoro-2-deoxy-d-glucose positron emission tomography. BMC Neurol. 2014, 14, 136. [Google Scholar] [CrossRef] [PubMed]
- Leypoldt, F.; Buchert, R.; Kleiter, I.; Marienhagen, J.; Gelderblom, M.; Magnus, T.; Dalmau, J.; Gerloff, C.; Lewerenz, J. Fluorodeoxyglucose positron emission tomography in anti-N-methyl-D-aspartate receptor encephalitis: Distinct pattern of disease. J. Neurol. Neurosurg. Psychiatry 2012, 83, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Chanson, J.-B.; Diaconu, M.; Honnorat, J.; Martin, T.; De Seze, J.; Namer, I.; Hirsch, E. PET follow-up in a case of anti-NMDAR encephalitis: Arguments for cingulate limbic encephalitis. Epileptic Disord. 2012, 14, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Greiner, H.; Leach, J.; Lee, K.H.; Krueger, D.A. Anti-NMDA receptor encephalitis presenting with imaging findings and clinical features mimicking Rasmussen syndrome. Seizure 2011, 20, 266–270. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guenther, A.; Brodoehl, S.; Witte, O.W.; Freesmeyer, M.; Dalmau, J.; Redecker, C. Atypical posthypoxic MRI changes in hypermetabolic regions in anti-NMDA-receptor encephalitis. Neurology 2012, 79, 720–721. [Google Scholar] [CrossRef]
- Maeder-Ingvar, M.; Rey, V.; Vincent, A.; Rossetti, A.O.; Prior, J.O.; Irani, S.R. FDG-PET hyperactivity in basal ganglia correlating with clinical course in anti–NDMA-R antibodies encephalitis. J. Neurol. Neurosurg. Psychiatry 2010, 82, 235–236. [Google Scholar] [CrossRef]
- Tzoulis, C.; Vedeler, C.A.; Haugen, M.; Storstein, A.; Tran, G.T.; Gjerde, I.O.; Biermann, M.; Schwarzlmüller, T.; Bindoff, L.A. Progressive striatal necrosis associated with anti-NMDA receptor antibodies. BMC Neurol. 2013, 13, 55. [Google Scholar] [CrossRef][Green Version]
- Novy, J.; Allenbach, G.; Bien, C.G.; Guedj, E.; Prior, J.O.; Rossetti, A.O. FDG-PET hyperactivity pattern in anti-NMDAr encephalitis. J. Neuroimmunol. 2016, 297, 156–158. [Google Scholar] [CrossRef][Green Version]
- Prieto, E.; Martí-Climent, J.; Morán, V.; Rodríguez, L.S.; Barbés, B.; Arbizu, J.; Richter, J.A. Brain PET imaging optimization with time of flight and point spread function modelling. Phys. Med. 2015, 31, 948–955. [Google Scholar] [CrossRef]
- Herholz, K. Guidance for reading FDG PET scans in dementia patients. Q. J. Nucl. Med. Mol. Imaging 2014, 58, 332–343. [Google Scholar] [PubMed]
- SPM—Statistical Parametric Mapping. Available online: https://www.fil.ion.ucl.ac.uk/spm/ (accessed on 29 May 2020).
- Minoshima, S.; A Frey, K.; A Koeppe, R.; Foster, N.L.; Kuhl, D.E. A diagnostic approach in Alzheimer’s disease using three-dimensional stereotactic surface projections of fluorine-18-FDG PET. J. Nucl. Med. 1995, 36, 1238–1248. [Google Scholar] [PubMed]
- Malby, R.; Haworth, S.; Wright, S.; Atkinson, J.; Gooch, S. White paper whitewash. Nurs. Stand. 1989, 3, 42. [Google Scholar] [CrossRef] [PubMed]
- Prieto, E.; Domínguez-Prado, I.; Riverol, M.; Ortega-Cubero, S.; Ribelles, M.J.; Luquin, R.; De Castro, P.; Arbizu, J. Metabolic patterns in prion diseases: An FDG PET voxel-based analysis. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Solnes, L.; Jones, K.M.; Rowe, S.P.; Pattanayak, P.; Nalluri, A.; Venkatesan, A.; Probasco, J.C.; Javadi, M.S. Diagnostic Value of 18 F-FDG PET/CT Versus MRI in the Setting of Antibody-Specific Autoimmune Encephalitis. J. Nucl. Med. 2017, 58, 1307–1313. [Google Scholar] [CrossRef]
- Probasco, J.C.; Solnes, L.; Nalluri, A.; Cohen, J.; Jones, K.M.; Zan, E.; Javadi, M.S.; Venkatesan, A. Abnormal brain metabolism on FDG-PET/CT is a common early finding in autoimmune encephalitis. Neurol. Neuroimmunol. Neuroinflamm. 2017, 4, e352. [Google Scholar] [CrossRef] [PubMed]
- Morbelli, S.; Djekidel, M.; Hesse, S.; Pagani, M.; Barthel, H.; Neuroimaging Committee of the European Association of Nuclear Medicine (EANM); Brain Imaging Council of the Society of Nuclear Medicine and Molecular Imaging (SNMMI). Role of (18)F-FDG-PET imaging in the diagnosis of autoimmune encephalitis. Lancet Neurol. 2016, 15, 1009–1010. [Google Scholar] [CrossRef]
- Titulaer, M.J.; McCracken, L.; Gabilondo, I.; Armangue, T.; Glaser, C.; Iizuka, T.; Honig, L.S.; Benseler, S.M.; Kawachi, I.; Martinez-Hernandez, E.; et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: An observational cohort study. Lancet Neurol. 2013, 12, 157–165. [Google Scholar] [CrossRef]
- Newey, C.R.; Sarwal, A.; Hantus, S. [18F]-Fluoro-Deoxy-Glucose Positron Emission Tomography Scan Should Be Obtained Early in Cases of Autoimmune Encephalitis. Autoimmune Dis. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Mattozzi, S.; Sabater, L.; Escudero, D.; Ariño, H.; Armangue, T.; Simabukuro, M.; Iizuka, T.; Hara, M.; Saiz, A.; Sotgiu, S.; et al. Hashimoto encephalopathy in the 21st century. Neurology 2019, 94, e217–e224. [Google Scholar] [CrossRef]
- Lagström, R.M.B.; Østerbye, N.N.; Henriksen, O.M.; Høgh, P. Hashimoto’s encephalopathy: Follow-up data from neuropsychology, lumbar puncture, and FDG-PET. Clin. Case Rep. 2019, 7, 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Morbelli, S.; Arbizu, J.; Booij, J.; Chen, M.K.; Chetelat, G.; Cross, D.J. The need of standardization and of large clinical studies in an emerging indication of [(18)F]FDG PET: The autoimmune encephalitis. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 353–357. [Google Scholar] [CrossRef] [PubMed]
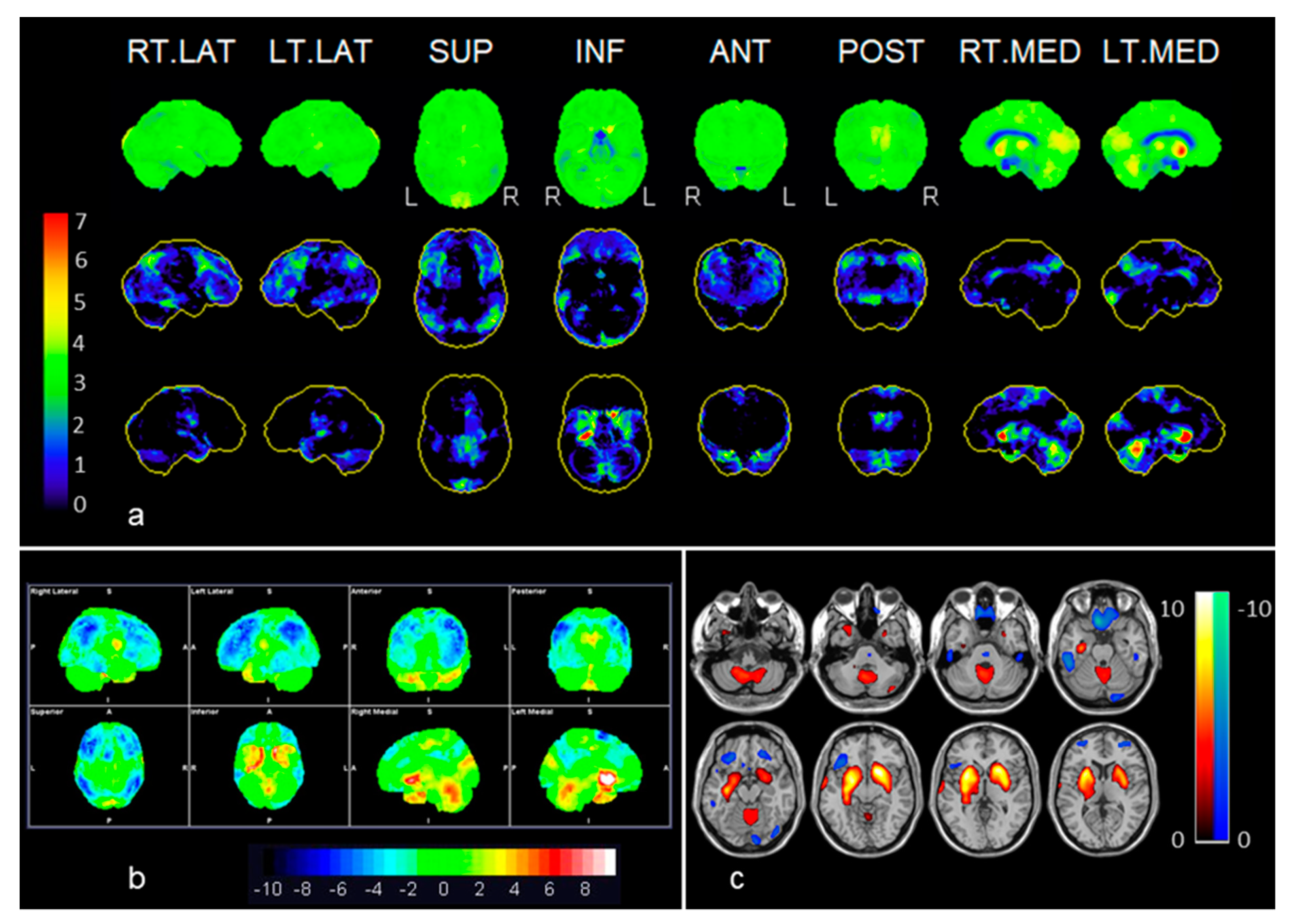

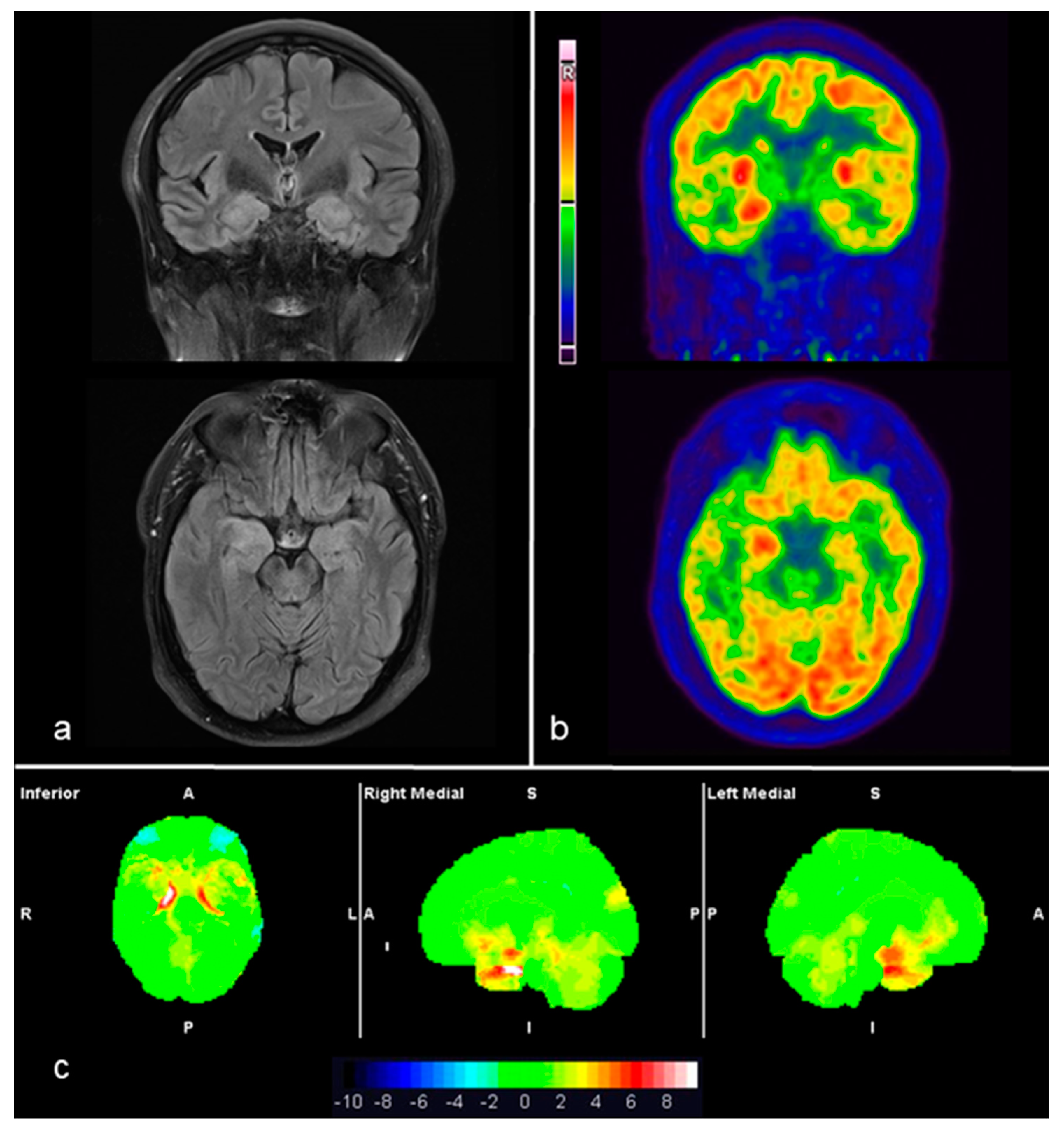
| Case | Age | Gender | Antibodies Type | Cognitive Impairment | Behavioral Disorder | Seizures | Treatment | Improvement after Treatment | EEG | CSF * | PET Result | MRI Result | Additional |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 17 | F | NMDAR | Yes | Yes | Yes | Ig + MP | Yes | + | + | abnormal | normal | - |
| 2 | 74 | M | LGI-1 | Yes | No | Yes | Ig + MP + rituximab | Yes | + | + | abnormal | abnormal | hyponatremia |
| 3 | 65 | M | CASPR2 | Yes | No | Yes | Ig + MP + rituximab | Yes | + | + | abnormal | abnormal | - |
| 4 | 77 | F | LGI-1 | Yes | Yes | Yes | Ig + MP | Yes | + | − | abnormal | abnormal | hyponatremia |
| 5 | 70 | F | No | Yes | No | No | Ig + MP | Yes | − | − | abnormal | normal *** | - |
| 6 | 78 | M | CASPR2 | Yes | Yes | Yes ** | Ig + MP | Yes | − | − | abnormal | abnormal | - |
| Case | Antibodies Type | Visual Assessment | SPM | Syngo.via Database Comparison | Neurostat 3D-SSP | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| p < 0.001 | p < 0.005 | ||||||||||
| Hypo | Hyper | Hypo | Hyper | Hypo | Hyper | Hypo | Hyper | Hypo | Hyper | ||
| 1 | NMDAR | L Frontal, L&R Temporal, Occipital, Motor cortex | L lateral Temporal | R MTL and lateral Temporal, L Frontal. R Motor cortex | R. Insula; L lateral Temporal, L&R Parietal; PC | Similar locations but more extended | Similar locations but more extended | L&R Frontal, R temporal, occipital, L&R Motor cortex | L temporal, medial Frontal, Insula, PC, L&R Parietal, Cerebellum | L&R Frontal, R temporal, occipital, L&R Motor cortex | L Temporal, medial Frontal, Insula, PC, L&R Parietal, Cerebellum |
| 2 | LGI-1 | Frontal, Parietal, Temporal, Thalamus, Occipital. | L&R BC | Orbitofrontal, L Temporal | L&R BG, Cerebellar vermis; L&R MTL | Similar locations and R Parietal | Similar locations but more extended | L&R Lateral Frontal, L Temporal, L&R Parietal, R PC | L&R BG, Cerebellar vermis, L&R MTL | L&R Lateral Frontal, lateral Temporal, Parietal, PC | L&R BG, Cerebellar vermis, L&R MTL |
| 3 | CASPR2 | - | R. MTL R. BG | - | R MTL, R BG | L&R Frontal, R. Temporal | L&R MTL; R BG; Occipital | Frontal, R Temporo-Parietal | L&R MTL, R BG, Occipital | R. Frontal | L&R MTL |
| 4 | LGI-1 | L Frontal, L&R parietal | L&R MTL, Cerebellar vermis, R BG | L&R Frontal, L&R lateral Temporal, R Parietal, L PC | L MTL | Similar but more extended, L&R Parietal, L&R PC | L&R MTL | L&R Frontal, L&R Parietal, L&R PC | L&R MTL Cerebellar Vermis, L&R BG, L&R Motor cortex | L&R Frontal, R Parietal, L&R PC | L&R MTL, Cerebellar vermis, Motor cortex, L&R |
| 5 | Negative | L Frontal, L lateral Temporal | PreCuneus, Occipital | L&R Frontal, L&R Temporal | - | L&R Frontal, R Insula, L&R Temporal | R Parietal | L&R Frontal, L&R Parietal, L Temporal | Parieto-Occipital, Precuneus, | L&R Frontal | Parieto-Occipital |
| 6 | CASPR2 | - | L.MTL. | L&R Fronto-temporal | - | Similar locations but more extended, Parietal | - | L&R Fronto-temporal | L MTL. | L&R Fronto-temporal | L MTL, Parieto-Occipital |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno-Ajona, D.; Prieto, E.; Grisanti, F.; Esparragosa, I.; Sánchez Orduz, L.; Gállego Pérez-Larraya, J.; Arbizu, J.; Riverol, M. 18F-FDG-PET Imaging Patterns in Autoimmune Encephalitis: Impact of Image Analysis on the Results. Diagnostics 2020, 10, 356. https://doi.org/10.3390/diagnostics10060356
Moreno-Ajona D, Prieto E, Grisanti F, Esparragosa I, Sánchez Orduz L, Gállego Pérez-Larraya J, Arbizu J, Riverol M. 18F-FDG-PET Imaging Patterns in Autoimmune Encephalitis: Impact of Image Analysis on the Results. Diagnostics. 2020; 10(6):356. https://doi.org/10.3390/diagnostics10060356
Chicago/Turabian StyleMoreno-Ajona, David, Elena Prieto, Fabiana Grisanti, Inés Esparragosa, Lizeth Sánchez Orduz, Jaime Gállego Pérez-Larraya, Javier Arbizu, and Mario Riverol. 2020. "18F-FDG-PET Imaging Patterns in Autoimmune Encephalitis: Impact of Image Analysis on the Results" Diagnostics 10, no. 6: 356. https://doi.org/10.3390/diagnostics10060356
APA StyleMoreno-Ajona, D., Prieto, E., Grisanti, F., Esparragosa, I., Sánchez Orduz, L., Gállego Pérez-Larraya, J., Arbizu, J., & Riverol, M. (2020). 18F-FDG-PET Imaging Patterns in Autoimmune Encephalitis: Impact of Image Analysis on the Results. Diagnostics, 10(6), 356. https://doi.org/10.3390/diagnostics10060356







