Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature
Abstract
1. Introduction
2. Materials and Methods
3. Cases Retrieved
4. Epidemiology
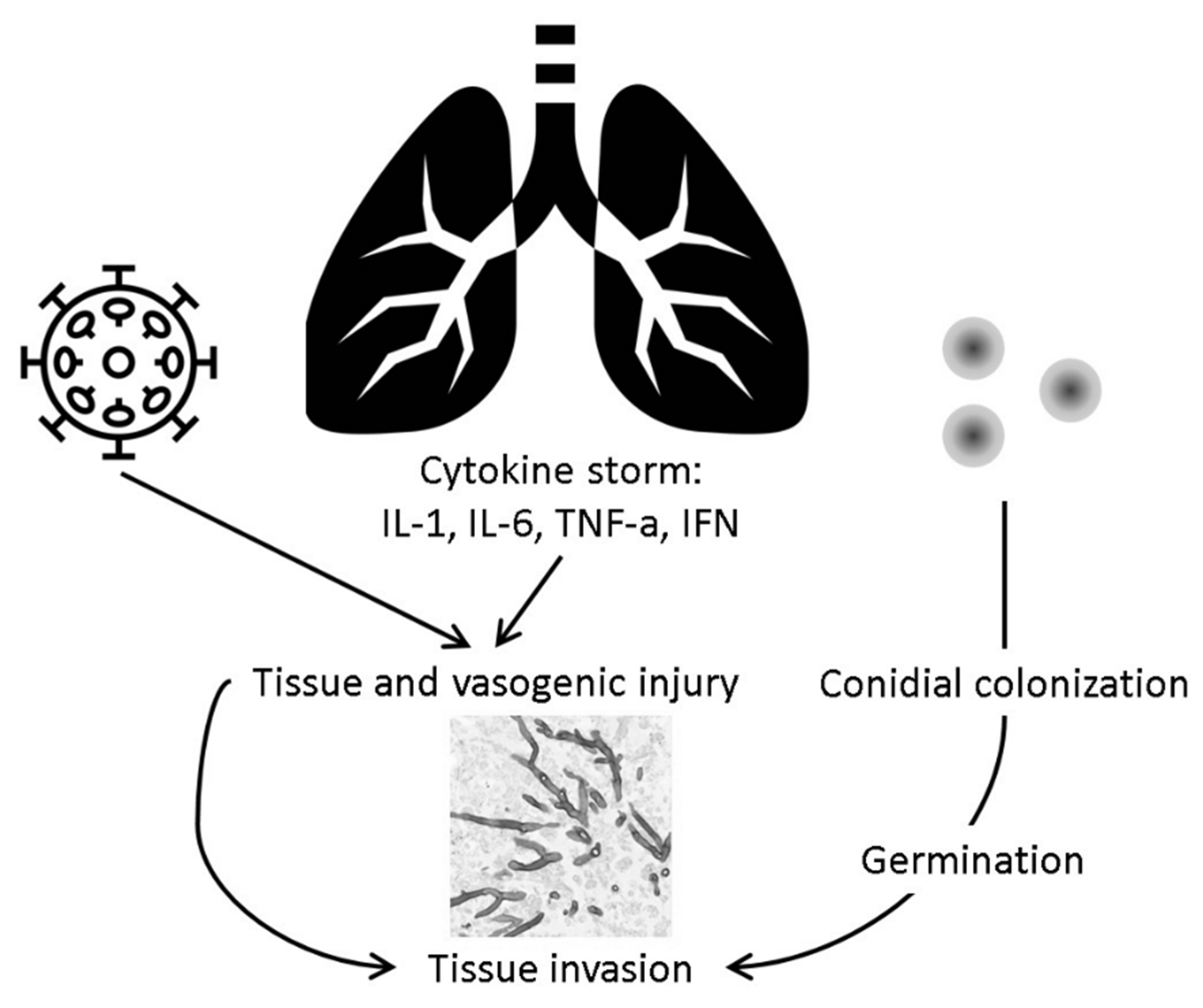
5. Pathogenesis
6. Clinical Presentation
7. Risk Factors
8. Diagnosis
9. Microbiology
10. Treatment
11. Outcomes
12. Discussion
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| N | First Author | Specimen | RdRp Gene Ct | E Gene Ct | S Gene Ct | GADPH Gene Ct | ORF1 Gene Ct |
|---|---|---|---|---|---|---|---|
| 1 * | Lescure [58] | NP, BAL | Negative | 30.3, 27.4 | Not done | 25.7, 24.7 | Not done |
| 2 | Ghelfelstein-Ferreira [37] | NP | Not done | 23.3 | 22.8 | Not done | Not done |
| 3 | Kohler [56] | BAL | Not done | 13.29 | 12.61 | Not done | Not done |
| 4 | Kohler [56] | BAL | Not done | 34.29 | Not done | Not done | 31.47 |
| 5 | Kohler [56] | BAL | Not done | 29.74 | Not done | Not done | 27.86 |
| 6 | Kohler [56] | TA | Not done | 21.47 | Not done | Not done | 20.12 |
| 7 | Meijer [34] | TA | Not done | 30.59 | Not done | Not done | Not done |
| 8 | Sharma [36] | NP | 35.09 | 22.04 | Not done | Not done | Not done |
References
- WHO Statistics. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200726-covid-19-sitrep-188.pdf?sfvrsn=f177c3fa_2 (accessed on 26 July 2020).
- Lansbury, L.; Lim, B.; Baskaran, V.; Lim, W.S. Co-Infections in People with COVID-19: A Systematic Review and Meta-Analysis. SSRN Electron. J. 2020, 81, 266–275. [Google Scholar] [CrossRef]
- De Pauw, B.; Walsh, T.J.; Donnelly, J.P.; Stevens, D.A.; Edwards, J.E.; Calandra, T.; Pappas, P.G.; Maertens, J.; Lortholary, O.; Kauffman, C.A.; et al. Revised Definitions of Invasive Fungal Disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin. Infect. Dis. 2008, 46, 1813–1821. [Google Scholar] [CrossRef]
- Blot, S.; Taccone, F.S.; Abeele, A.-M.V.D.; Bulpa, P.; Meersseman, W.; Brusselaers, N.; Dimopoulos, G.; Paiva, J.A.; Misset, B.; Rello, J.; et al. A Clinical Algorithm to Diagnose Invasive Pulmonary Aspergillosis in Critically Ill Patients. Am. J. Respir. Crit. Care Med. 2012, 186, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 10223. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Q.; Zhang, P.; Sheng, J.; Zhou, J.; Qu, T. Clinical characteristics of invasive pulmonary aspergillosis in patients with COVID-19 in Zhejiang, China: A retrospective case series. Crit. Care 2020, 24, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Flikweert, A.W.; Grootenboers, M.J.; Yick, D.C.; Du Mée, A.W.; Van Der Meer, N.J.; Rettig, T.C.; Kant, M.K. Late histopathologic characteristics of critically ill COVID-19 patients: Different phenotypes without evidence of invasive aspergillosis, a case series. J. Crit. Care 2020, 59, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Bartoletti, M.; Pascale, R.; Cricca, M.; Rinaldi, M.; Maccaro, A.; Bussini, L.; Fornaro, G.; Tonetti, T.; Pizzilli, G.; Francalanci, E.; et al. Epidemiology of Invasive Pulmonary Aspergillosis Among Intubated Patients With COVID-19: A Prospective Study. Clin. Infect. Dis. 2020, ciaa1065. [Google Scholar] [CrossRef] [PubMed]
- Rutsaert, L.; Steinfort, N.; Van Hunsel, T.; Bomans, P.; Naesens, R.; Mertes, H.; Dits, H.; Van Regenmortel, N. COVID-19-associated invasive pulmonary aspergillosis. Ann. Intensiv. Care 2020, 10, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Lamoth, F.; Glampedakis, E.; Boillat-Blanco, N.; Oddo, M.; Pagani, J.-L. Incidence of invasive pulmonary aspergillosis among critically ill COVID-19 patients. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Lionakis, M.S.; Kontoyiannis, D.P. Glucocorticoids and invasive fungal infections. Lancet 2003, 362, 1828–1838. [Google Scholar] [CrossRef]
- Kitmiridou, D.; Aung, S.; Farmakiotis, D. Disseminated Mucormycosis with Positive Aspergillus Galactomannan. Case Rep. Infect. Dis. 2018, 2018, 4294013. [Google Scholar] [CrossRef] [PubMed]
- Meersseman, W.; Lagrou, K.; Maertens, J.; Van Wijngaerden, E. Invasive Aspergillosis in the Intensive Care Unit. Clin. Infect. Dis. 2007, 45, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Schauwvlieghe, A.F.A.D.; Rijnders, B.; Philips, N.; Verwijs, R.; Vanderbeke, L.; Van Tienen, C.; Lagrou, K.; Verweij, P.E.; Van De Veerdonk, F.L.; Gommers, D.; et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: A retrospective cohort study. Lancet Respir. Med. 2018, 6, 782–792. [Google Scholar] [CrossRef]
- Van De Veerdonk, F.; Kolwijck, E.; Lestrade, P.P.A.; Hodiamont, C.J.; Rijnders, B.; Van Paassen, J.; Haas, P.-J.A.; Dos Santos, C.O.; Kampinga, G.A.; Bergmans, D.C.J.J.; et al. Influenza-associated Aspergillosis in Critically Ill Patients. Am. J. Respir. Crit. Care Med. 2017, 196, 524–527. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.-P. The pathobiology of Aspergillus fumigatus. Trends Microbiol. 2001, 9, 382–389. [Google Scholar] [CrossRef]
- Latgé, J.-P.; Chamilos, G. Aspergillus fumigatus and Aspergillosis in 2019. Clin. Microbiol. Rev. 2019, 33, e00140-18. [Google Scholar] [CrossRef] [PubMed]
- Latgé, J.-P. Aspergillus fumigatus and Aspergillosis. Clin. Microbiol. Rev. 1999, 12, 310–350. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group; Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; et al. Dexamethasone in Hospitalized Patients with Covid-19—Preliminary Report. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Gaynor, R.B. Therapeutic potential of inhibition of the NF-kappaB pathway in the treatment of inflammation and cancer. J. Clin. Investig. 2001, 107, 135–142. [Google Scholar] [CrossRef]
- Balloy, V.; Huerre, M.; LatgéJ, -P.; Chignard, M. Differences in Patterns of Infection and Inflammation for Corticosteroid Treatment and Chemotherapy in Experimental Invasive Pulmonary Aspergillosis. Infect. Immun. 2005, 73, 494–503. [Google Scholar] [CrossRef]
- Roilides, E.; Sein, T.; Roden, M.; Schaufele, R.L.; Walsh, T.J. Elevated Serum Concentrations of Interleukin-10 in Nonneutropenic Patients with Invasive Aspergillosis. J. Infect. Dis. 2001, 183, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.E.; Kontoyiannis, D.P. Invasive aspergillosis in glucocorticoid-treated patients. Med. Mycol. 2009, 47, S271–S281. [Google Scholar] [CrossRef] [PubMed]
- Vanderbeke, L.; Spriet, I.; Breynaert, C.; Rijnders, B.J.; Verweij, P.E.; Wauters, J. Invasive pulmonary aspergillosis complicating severe influenza. Curr. Opin. Infect. Dis. 2018, 31, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee of the WHO Consultation on Clinical Aspects of Pandemic (H1N1) 2009 Influenza; Bautista, E.; Chotpitayasunondh, T.; Gao, Z.; Harper, S.A.; Shaw, M.; Uyeki, T.M.; Zaki, S.R.; Hayden, F.G.; Hui, D.S.; et al. Clinical Aspects of Pandemic 2009 Influenza A (H1N1) Virus Infection. N. Engl. J. Med. 2010, 362, 1708–1719. [Google Scholar] [CrossRef] [PubMed]
- Ajmal, S.; Mahmood, M.; Abu Saleh, O.; Larson, J.; Sohail, M. Invasive fungal infections associated with prior respiratory viral infections in immunocompromised hosts. Infection 2018, 46, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Crum-Cianflone, N. Invasive Aspergillosis Associated With Severe Influenza Infections. Open Forum Infect. Dis. 2016, 3, ofw171. [Google Scholar] [CrossRef] [PubMed]
- Schaller, T.; Hirschbühl, K.; Burkhardt, K.; Braun, G.; Trepel, M.; Märkl, B.; Claus, R. Postmortem Examination of Patients With COVID-19. JAMA 2020, 323, 2518. [Google Scholar] [CrossRef]
- Menter, T.; Haslbauer, J.D.; Nienhold, R.; Savic, S.; Hopfer, H.; Deigendesch, N.; Frank, S.; Turek, D.; Willi, N.; Pargger, H.; et al. Post-mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology 2020, 77, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Gandhi, R.T.; Lynch, J.B.; Del Rio, C. Mild or Moderate Covid-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Berlin, D.A.; Gulick, R.M.; Martinez, F.J. Severe Covid-19. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Sun, W.; Li, M.; Dong, L. A complex COVID-19 case with rheumatoid arthritis treated with tocilizumab. Clin. Rheumatol. 2020, 39, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Meijer, E.F.J.; Dofferhoff, A.S.; Hoiting, O.; Buil, J.B.; Meis, J.F. Azole-Resistant COVID-19-Associated Pulmonary Aspergillosis in an Immunocompetent Host: A Case Report. J. Fungi 2020, 6, 79. [Google Scholar] [CrossRef] [PubMed]
- Prattes, J.; Valentin, T.; Hoenigl, M.; Talakic, E.; Reisinger, A.C.; Eller, P. Invasive pulmonary aspergillosis complicating COVID-19 in the ICU—A case report. Med. Mycol. Case Rep. 2020. [Google Scholar] [CrossRef]
- Sharma, A.; Hofmeyr, A.; Bansal, A.; Thakkar, D.; Lam, L.; Harrington, Z.; Bhonagiri, D. COVID-19 associated pulmonary aspergillosis (CAPA): An Australian case report. Med. Mycol. Case Rep. 2020. [Google Scholar] [CrossRef]
- Ghelfenstein-Ferreira, T.; Saade, A.; Alanio, A.; Bretagne, S.; De Castro, R.J.A.; Hamane, S.; Azoulay, E.; Bredin, S.; Dellière, S. Recovery of a triazole-resistant Aspergillus fumigatus in respiratory specimen of COVID-19 patient in ICU—A case report. Med. Mycol. Case Rep. 2020. [Google Scholar] [CrossRef]
- Fernandez, N.B.; Caceres, D.H.; Beer, K.D.; Irrazabal, C.; Delgado, G.; Farias, L.; Chiller, T.M.; Verweij, P.E.; Stecher, D. Ventilator-associated pneumonia involving Aspergillus flavus in a patient with coronavirus disease 2019 (COVID-19) from Argentina. Med. Mycol. Case Rep. 2020. [Google Scholar] [CrossRef]
- Van Biesen, S.; Kwa, D.; Bosman, R.J.; Juffermans, N.P. Detection of Invasive Pulmonary Aspergillosis in COVID-19 with Non-directed Bronchoalveolar Lavage. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef]
- Gangneux, J.-P.; Bougnoux, M.-E.; Dannaoui, E.; Cornet, M.; Zahar, J.; Ralph, Z.J. Invasive fungal diseases during COVID-19: We should be prepared. J. Mycol. Med. 2020, 30, 100971. [Google Scholar] [CrossRef]
- Alanio, A.; Dellière, S.; Fodil, S.; Bretagne, S.; Mégarbane, B. Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Lancet Respir. Med. 2020, 8, e48–e49. [Google Scholar] [CrossRef]
- White, L.; Dhillon, R.; Cordey, A.; Hughes, H.; Faggian, F.; Soni, S.; Pandey, M.; Whitaker, H.; May, A.; Morgan, M.; et al. A National Strategy to Diagnose COVID-19 Associated Invasive Fungal Disease in the ICU. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Mohamed, A.; Rogers, T.R.; Talento, A.F. COVID-19 Associated Invasive Pulmonary Aspergillosis: Diagnostic and Therapeutic Challenges. J. Fungi 2020, 6, 115. [Google Scholar] [CrossRef] [PubMed]
- Blaize, M.; Mayaux, J.; Nabet, C.; Lampros, A.; Marcelin, A.-G.; Thellier, M.; Piarroux, R.; Demoule, A.; Fekkar, A. Fatal Invasive Aspergillosis and Coronavirus Disease in an Immunocompetent Patient. Emerg. Infect. Dis. 2020, 26, 1636–1637. [Google Scholar] [CrossRef] [PubMed]
- Nasir, N.; Farooqi, J.Q.; Mahmood, S.F.; Jabeen, K. COVID-19-associated pulmonary aspergillosis (CAPA) in patients admitted with severe COVID-19 pneumonia: An observational study from Pakistan. Mycoses 2020, 63, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Bruno, G.; Fabrizio, C.; Buccoliero, G.B. COVID-19-associated pulmonary aspergillosis: Adding insult to injury. Lancet Microbe 2020, 1, e106. [Google Scholar] [CrossRef]
- Brown, L.-A.K.; Ellis, J.; Gorton, R.; De, S.; Stone, N. Surveillance for COVID-19-associated pulmonary aspergillosis. Lancet Microbe 2020, 1, e152. [Google Scholar] [CrossRef]
- Farmakiotis, D.; Le, A.; Weiss, Z.F.; Ismail, N.; Kubiak, D.W.; Koo, S. False positive bronchoalveolar lavage galactomannan: Effect of host and cut-off value. Mycoses 2018, 62, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Affolter, K.; Tamm, M.; Jahn, K.; Halter, J.; Passweg, J.R.; Hirsch, H.H.; Stolz, D. Galactomannan in the Bronchoalveolar Lavage for Diagnosing Invasive Fungal Disease. Am. J. Respir. Crit. Care Med. 2014, 190, 309–317. [Google Scholar] [CrossRef]
- Hsu, A.; Matera, R.; Vieira, K.; Reagan, J.L.; Farmakiotis, D. Antifungal prophylaxis during 7 + 3 induction chemotherapy for acute myeloid leukemia is associated with improved survival, in a setting with low incidence of invasive mold infections. Support. Care Cancer 2020, 1–6. [Google Scholar] [CrossRef]
- Tsikala-Vafea, M.; Cao, W.; Olszewski, A.J.; Donahue, J.E.; Farmakiotis, D. Fatal Mucormycosis and Aspergillosis in an Atypical Host: What Do We Know about Mixed Invasive Mold Infections? Case Rep. Infect. Dis. 2020, 2020, 8812528. [Google Scholar] [CrossRef]
- Magira, E.E.; Jiang, Y.; Economides, M.; Tarrand, J.; Kontoyiannis, D.P. Mixed mold pulmonary infections in haematological cancer patients in a tertiary care cancer centre. Mycoses 2018, 61, 861–867. [Google Scholar] [CrossRef]
- Kuderer, N.M.; Choueiri, T.K.; Shah, D.P.; Shyr, Y.; Rubinstein, S.M.; Rivera, D.R.; Shete, S.; Hsu, C.-Y.; Desai, A.; Lopes, G.D.L.; et al. Clinical impact of COVID-19 on patients with cancer (CCC19): A cohort study. Lancet 2020, 395, 1907–1918. [Google Scholar] [CrossRef]
- Acharige, M.J.T.; Koshy, S.; Ismail, N.; Aloum, O.; Jazaerly, M.; Astudillo, C.L.; Koo, S. Breath-based diagnosis of fungal infections. J. Breath Res. 2018, 12, 027108. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.; Thomas, H.R.; Daniels, S.D.; Lynch, R.C.; Fortier, S.M.; Shea, M.M.; Rearden, P.; Comolli, J.C.; Baden, L.R.; Marty, F.M. A Breath Fungal Secondary Metabolite Signature to Diagnose Invasive Aspergillosis. Clin. Infect. Dis. 2014, 59, 1733–1740. [Google Scholar] [CrossRef] [PubMed]
- Koehler, P.; Cornely, O.A.; Böttiger, B.W.; Dusse, F.; Eichenauer, D.A.; Fuchs, F.; Hallek, M.; Jung, N.; Klein, F.; Persigehl, T.; et al. COVID-19 associated pulmonary aspergillosis. Mycoses 2020, 63, 528–534. [Google Scholar] [CrossRef]
- Santana, M.F.; Pivoto, G.; Alexandre, M.A.A.; Baía-Da-Silva, D.C.; Borba, M.G.D.S.; Val, F.A.; Brito-Sousa, J.D.; Melo, G.C.; Monteiro, W.M.; Souza, J.V.B.; et al. Confirmed Invasive Pulmonary Aspergillosis and COVID-19: The value of postmortem findings to support antemortem management. Rev. Soc. Bras. Med. Trop. 2020, 53. [Google Scholar] [CrossRef]
- Lescure, F.-X.; Bouadma, L.; Nguyen, D.; Parisey, M.; Wicky, P.-H.; Behillil, S.; Gaymard, A.; Bouscambert-Duchamp, M.; Donati, F.; Le Hingrat, Q.; et al. Clinical and virological data of the first cases of COVID-19 in Europe: A case series. Lancet Infect. Dis. 2020, 20, 697–706. [Google Scholar] [CrossRef]
- Lahmer, T.; Rasch, S.; Spinner, C.; Geisler, F.; Schmid, R.; Huber, W. Invasive pulmonary aspergillosis in severe coronavirus disease 2019 pneumonia. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Antinori, S.; Rech, R.; Galimberti, L.; Castelli, A.; Angeli, E.; Fossali, T.; Bernasconi, D.; Covizzi, A.; Bonazzetti, C.; Torre, A.; et al. Invasive pulmonary aspergillosis complicating SARS-CoV-2 pneumonia: A diagnostic challenge. Travel Med. Infect. Dis. 2020, 2020, 101752. [Google Scholar] [CrossRef]
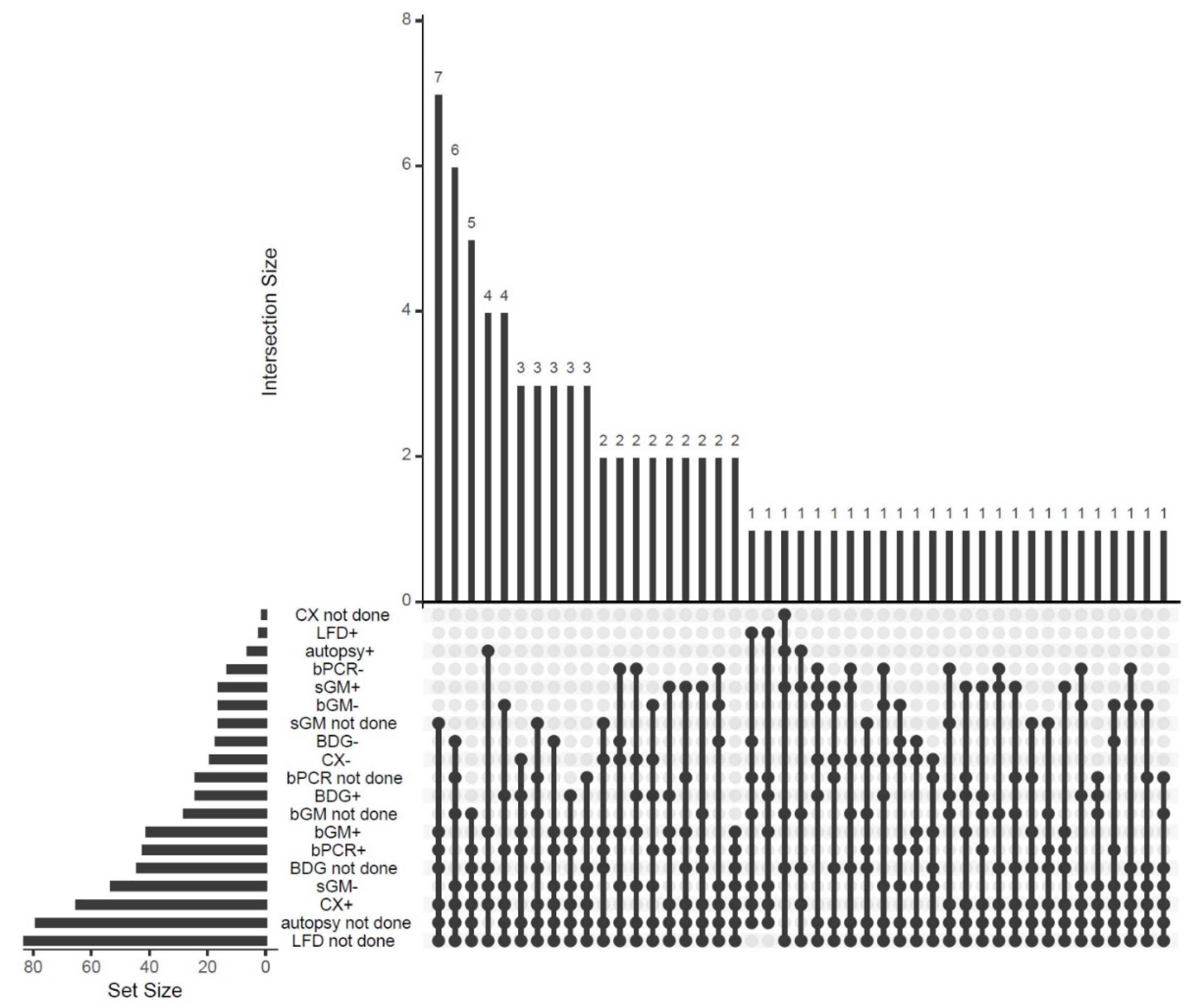
| Parameter | Total | Survived | Died | p-Value |
|---|---|---|---|---|
| N | 85 | 39 | 46 | |
| Age (years, mean ± S.D.) | 67.0 ± 11.5 | 63.2 ± 12.4 | 70.6 ± 9.5 | 0.0098 |
| Missing/unknown | 20 (24) | 8 (21) | 12 (26) | |
| Sex | ||||
| Female | 16 (19) | 11 (28) | 5 (11) | 0.083 |
| Male | 49 (58) | 20 (51) | 29 (63) | |
| Missing/unknown | 20 (24) | 8 (21) | 12 (26) | |
| Comorbidities | ||||
| Asthma or chronic obstructive pulmonary disease (COPD) | 17 (20) | 4 (10) | 13 (28) | 0.056 |
| Diabetes | 15 (18) | 4 (10) | 11 (24) | 0.15 |
| Hypertension | 20 (24) | 9 (23) | 11 (24) | >0.99 |
| Corticosteroids | 39 (46) | 18 (46) | 21 (46) | >0.99 |
| Missing/unknown | 13 (15) | 6 (15) | 7 (15) | |
| Aspergillus species * | ||||
| A. fumigatus | 56 (66) | 26 (67) | 30 (65) | 0.53 |
| A. flavus | 8 (9) | 3 (8) | 5 (11) | |
| A. niger | 1 (1) | 0 (0) | 1 (2) | |
| A. terreus | 1 (1) | 0 (0) | 1 (2) | |
| A. penicilloides | 1 (1) | 0 (0) | 1 (2) | |
| A. tubingensis | 1 (1) | 1 (2) | 0 (0) | |
| Unknown/not speciated | 18 (21) | 9 (23) | 9 (20) | |
| Resistance | 3 (4) | 1 (3) | 2 (4) | >0.99 |
| Missing/unknown | 69 (81) | 34 (87) | 35 (76) | |
| Time to diagnosis (days, median, interquartile range [IQR]) | 10, 5–15 | 12.5, 2.5–19.5 | 9, 6–13 | 0.86 |
| Missing/unknown | 43 (51) | 20 (51) | 23 (50) | |
| CAPA classification | ||||
| Proven | 6 (7) | 3 (8) | 3 (7) | 0.78 |
| Probable or putative | 67 (79) | 31 (79) | 36 (78) | |
| Possible | 12 (14) | 5 (13) | 7 (15) | |
| Treatment ** | ||||
| None | 15 (18) | 5 (13) | 10 (22) | 0.28 |
| Mold-active triazole (MAT: vori-, posa-, isavuconazole) | 61 (72) | 31 (79) | 30 (65) | 0.61 |
| Voriconazole | 55 (65) | 29 (74) | 26 (57) | 0.11 |
| Isavuconazole | 6 (7) | 2 (5) | 4 (9) | 0.68 |
| Echinocandin (caspo-, mica-, anidulafungin) | 14 (16) | 7 (18) | 7 (15) | 0.78 |
| Amphotericin-B | 21 (25) | 11 (28) | 10 (22) | 0.62 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Apostolopoulou, A.; Esquer Garrigos, Z.; Vijayvargiya, P.; Lerner, A.H.; Farmakiotis, D. Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature. Diagnostics 2020, 10, 807. https://doi.org/10.3390/diagnostics10100807
Apostolopoulou A, Esquer Garrigos Z, Vijayvargiya P, Lerner AH, Farmakiotis D. Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature. Diagnostics. 2020; 10(10):807. https://doi.org/10.3390/diagnostics10100807
Chicago/Turabian StyleApostolopoulou, Anna, Zerelda Esquer Garrigos, Prakhar Vijayvargiya, Alexis Hope Lerner, and Dimitrios Farmakiotis. 2020. "Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature" Diagnostics 10, no. 10: 807. https://doi.org/10.3390/diagnostics10100807
APA StyleApostolopoulou, A., Esquer Garrigos, Z., Vijayvargiya, P., Lerner, A. H., & Farmakiotis, D. (2020). Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature. Diagnostics, 10(10), 807. https://doi.org/10.3390/diagnostics10100807





