Optimization of Point-Shear Wave Elastography by Skin-to-Liver Distance to Assess Liver Fibrosis in Patients Undergoing Bariatric Surgery
Abstract
:1. Introduction
2. Material and Methods
2.1. Liver Elastography
2.2. Ultrasound Evaluation of Steatosis
2.3. Liver Biopsy
2.4. Statistical Analysis
3. Results
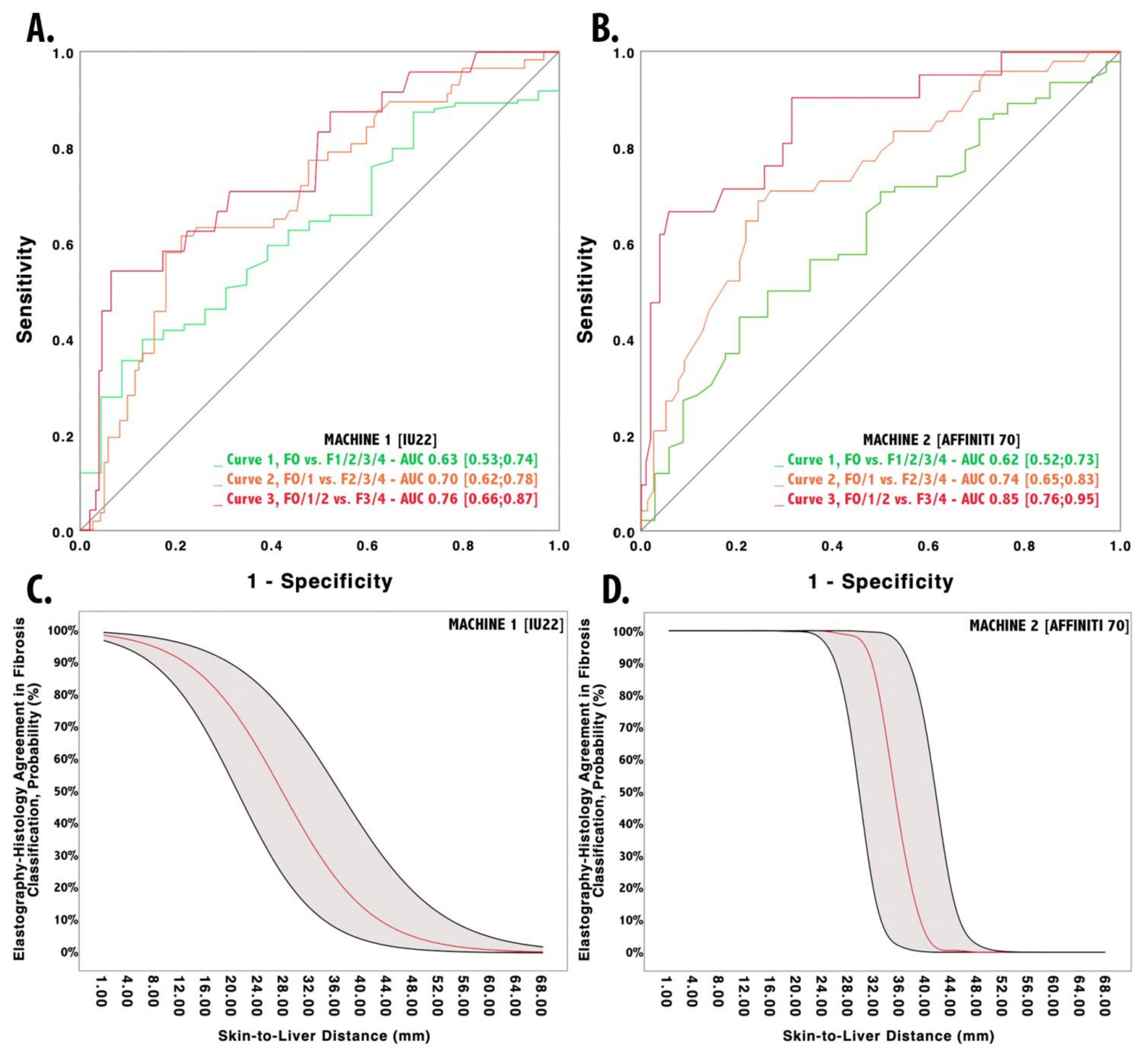
3.1. The Impact of SLD on Philips IU22 Measurements
3.2. The Impact of SLD on Philips Affiniti 70 Measurements
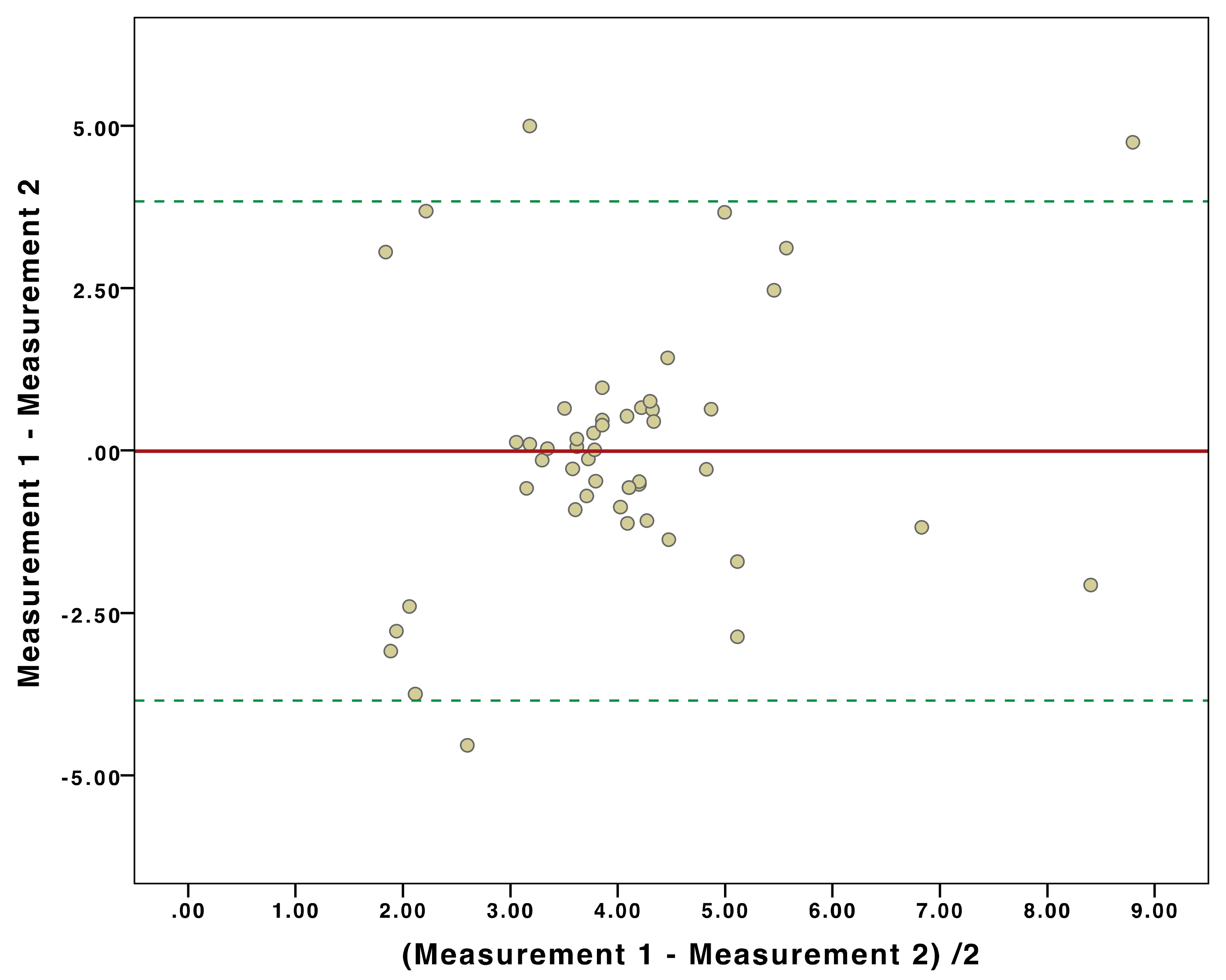
3.3. Inter-Machine Liver Stiffness Measurement Agreement
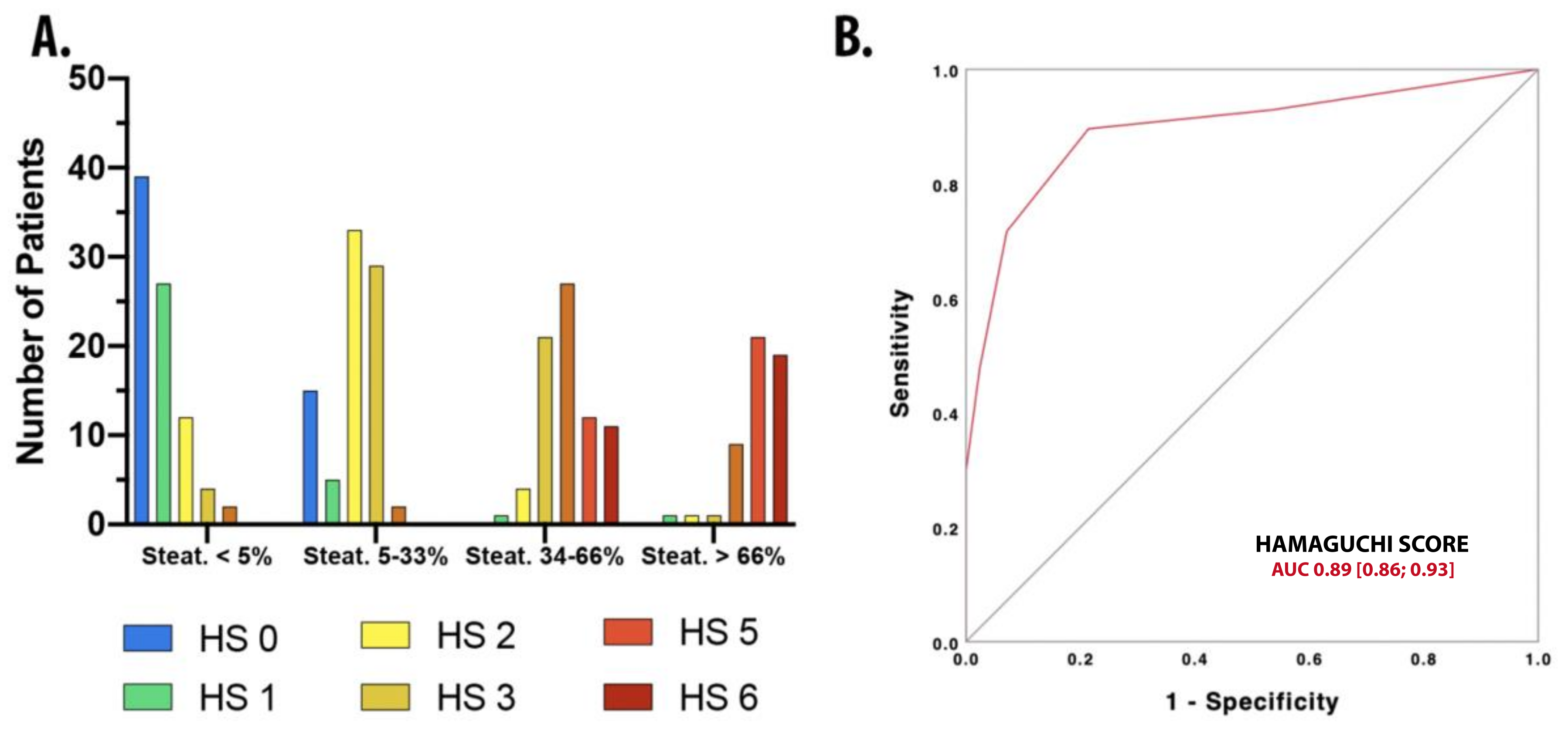
3.4. Application of Hamaguchi Score for Steatosis
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| NAFLD | non-alcoholic fatty liver disease |
| NAFL | non-alcoholic fatty liver |
| NASH | non-alcoholic steatohepatitis |
| LD | linear dichroism |
| TE | transient elastography |
| pSWE | point Shear Wave Elastography |
| AWT | abdominal wall thickness |
| LSM | liver stiffness measurement |
| ElastPQ | Elastography Point Quantification |
| HS | Hamaguchi Score |
| BMI | body mass index |
| EHA | elastography–histology agreement |
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| GGT | gamma-glutamyl-transferase |
| HDL | high density lipoprotein |
| LDL | low density lipoprotein |
| IQR | interquartile range |
References
- WHO. Prevalence Obesity and Overweight. In World Health Organisation Media Centre Fact Sheet No 311 2014. Available online: http://www.who.Int/growthref/who.bmi.for.age/enhtml (accessed on 1 March 2020).
- Stigler, F.L.; Lustig, R.H.; Ma, J.I. Mechanisms, pathophysiology, and management of obesity. N. Engl. J. Med. 2017, 376, 1490–1491. [Google Scholar] [CrossRef] [Green Version]
- Malaguarnera, M.; Di Rosa, M.; Nicoletti, F.; Malaguarnera, L. Molecular mechanisms involved in NAFLD progression. J. Mol.Med. 2009, 87, 679–695. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Chen, L.; Leung, L.; Yen, T.S.B.; Lee, C.; Chan, J.Y. Liver-specific inactivation of the Nrf1 gene in adult mouse leads to nonalcoholic steatohepatitis and hepatic neoplasia. Proc. Natl. Acad. Sci. USA 2005, 102, 4120–4125. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giuffrè, M.; Campigotto, M.; Campisciano, G.; Comar, M.; Crocè, L.S. A Story of Liver and Gut Microbes: How Does the Intestinal Flora Affect Liver Disease? Am. J. Physiol. Gastrointest. Liver Physiol. 2020. [CrossRef]
- Machado, M.; Marques-Vidal, P.; Cortez-Pinto, H. Hepatic histology in obese patients undergoing bariatric surgery. J. Hepatol. 2006, 45, 600–606. [Google Scholar] [CrossRef]
- Cassinotto, C.; Boursier, J.; de Lédinghen, V.; Lebigot, J.; Lapuyade, B.; Cales, P.; Hiriart, J.; Michalak, S.; le Bail, B.; Cartier, V.; et al. Liver stiffness in nonalcoholic fatty liver disease: A comparison of supersonic shear imaging, FibroScan, and ARFI with liver biopsy. Hepatology 2016, 63, 1817–1827. [Google Scholar] [CrossRef] [PubMed]
- Foschi, F.G.; Domenicali, M.; Giacomoni, P.; Dall’Aglio, A.C.; Conti, F.; Borghi, A.; Bevilacqua, V.; Napoli, L.; Mirici, F.; Cucchetti, A. Is there an association between commonly employed biomarkers of liver fibrosis and liver stiffness in the general population? Annals Hepatol. 2020, 19, 380–387. [Google Scholar] [CrossRef]
- De Ledinghen, V.; Vergniol, J.; Foucher, J.; El-Hajbi, F.; Merrouche, W.; Rigalleau, V. Feasibility of liver transient elastography with FibroScan® using a new probe for obese patients. Liver Int. 2010, 30, 1043–1048. [Google Scholar] [CrossRef]
- Myers, R.P.; Pomier-Layrargues, G.; Kirsch, R.; Pollett, A.; Duarte-Rojo, A.; Wong, D.; Beaton, M.; Levstik, M.; Crotty, P.; Elkashab, M. Feasibility and diagnostic performance of the FibroScan XL probe for liver stiffness measurement in overweight and obese patients. Hepatology 2012, 55, 199–208. [Google Scholar] [CrossRef]
- Jiang, W.; Huang, S.; Teng, H.; Wang, P.; Wu, M.; Zhou, X.; Ran, H. Diagnostic accuracy of point shear wave elastography and transient elastography for staging hepatic fibrosis in patients with non-alcoholic fatty liver disease: A meta-analysis. BMJ Open 2018, 8, e021787. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Fu, J.; Hong, R.; Liu, L.; Li, F. Acoustic radiation force impulse elastography for the non-invasive evaluation of hepatic fibrosis in non-alcoholic fatty liver disease patients: A systematic review & meta-analysis. PLoS ONE 2015, 10, e0127782. [Google Scholar] [CrossRef] [Green Version]
- Cho, Y.S.; Lim, S.; Kim, Y.; Kim, T.Y.; Jeong, W.K.; Sohn, J.H. Abdominal Wall Thickness Affects Liver Stiffness Measurements by 2-D Shear Wave Elastography in Patients with Chronic Liver Disease. Ultrasound Med. Biol. 2019, 45, 2697–2703. [Google Scholar] [CrossRef]
- Mulabecirovic, A.; Mjelle, A.B.; Gilja, O.H.; Vesterhus, M.; Havre, R.F. Repeatability of shear wave elastography in liver fibrosis Phantoms—evaluation of five different systems. PLoS ONE 2018, 13, e0189671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karlas, T.; Dietrich, A.; Peter, V.; Wittekind, C.; Lichtinghagen, R.; Garnov, N.; Linder, N.; Schaudinn, A.; Busse, H.; Prettin, C.; et al. Evaluation of transient elastography, Acoustic Radiation Force Impulse Imaging (ARFI), and Enhanced Liver Function (ELF) score for detection of fibrosis in morbidly obese patients. PLoS ONE 2015, 10, e0141649. [Google Scholar] [CrossRef] [PubMed]
- Hamaguchi, M.; Kojima, T.; Itoh, Y.; Yuichi, H.; Kota, F.; Tomoaki, N.; Takahiro, K.; Noriyuki, T.; Junichi, O.; Kazunori, I.; et al. The Severity of Ultrasonographic Findings in Nonalcoholic Fatty Liver Disease Reflects the Metabolic Syndrome and Visceral Fat Accumulation. Am. J. Gastroenterol. 2007. [CrossRef]
- Grundy, S.M.; Barondess, J.A.; Bellegie, N.J.; Fromm, H.; Greenway, F.; Halsted, C.H.; Huth, E.J.; Kumanyika, S.K.; Reisin, E.; Robinson, M.K.; et al. Gastrointestinal surgery for severe obesity. Ann. Intern. Med. 1991, 956–961. [Google Scholar] [CrossRef]
- Giuffrè, M.; Macor, D.; Masutti, F.; Abazia, C.; Tinè, F.; Bedogni, G.; Tiribelli, C.; Crocè, L.S. Spleen Stiffness Probability Index (SSPI): A simple and accurate method to detect esophageal varices in patients with compensated liver cirrhosis. Ann. Hepatol. 2020, 19, 53–61. [Google Scholar] [CrossRef]
- Giuffrè, M.; Fouraki, S.; Campigotto, M.; Colombo, A.; Visintin, A.; Buonocore, M.R.; Aversano, A.; Budel, M.; Tinè, F.; Abazia, C.; et al. Alanine aminotransferase and spleno-portal dynamics affect spleen stiffness measured by point shear-wave elastography in patients with chronic hepatitis C in the absence of significant liver fibrosis. J. Ultrasound 2020. [CrossRef]
- Giuffrè, M.; Bedogni, G.; Abazia, C.; Masutti, F.; Tiribelli, C.; Crocè, L.S. Spleen stiffness can be employed to assess the efficacy of spontaneous portosystemic shunts in relieving portal hypertension. Ann. Hepatol. 2020, 7–9. [Google Scholar] [CrossRef]
- Giuffrè, M.; Macor, D.; Masutti, F.; Abazia, C.; Tinè, F.; Patti, R.; Buonocore, M.R.; Colombo, A.; Visintin, A.; Campigotto, M.; et al. Evaluation of spleen stiffness in healthy volunteers using point shear wave elastography. Ann. Hepatol. 2019, 18, 736–741. [Google Scholar] [CrossRef]
- Sporea, I.; Bota, S.; Grădinaru-Taşcău, O.; Sirli, R.; Popescu, A. Comparative study between two point shear wave elastographic techniques: Acoustic Radiation Force Impulse (ARFI) elastography and ElastPQ. Med. Ultrason. 2014, 16, 309–314. [Google Scholar] [CrossRef]
- Yoo, H.; Lee, J.M.; Yoon, J.H.; Lee, D.H.; Chang, W.; Han, J.K. Prospective comparison of liver stiffness measurements between two point shear wave elastography methods: Virtual touch quantification and elastography point quantification. Korean J. Radiol. 2016, 17, 750–757. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. A Coefficient of Agreement for Nominal Scales. Educ. Psychol. Meas. 1960, 20, 37–46. [Google Scholar] [CrossRef]
- Hanley, J.A.; McNeil, B.J. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology 1983, 148, 839–843. [Google Scholar] [CrossRef] [Green Version]
- Fagerland, M.W.; Hosmer, D.W. A goodness-of-fit test for the proportional odds regression model. Stat. Med. 2013, 32, 2235–2249. [Google Scholar] [CrossRef]
- Sambataro, G.; Giuffrè, M.; Sambataro, D.; Palermo, A.; Vignigni, G.; Cesareo, R.; Crimi, N.; Torrisi, S.E.; Vancheri, C.; Malatino, L.; et al. The Model for Early COvid-19 Recognition (MECOR) Score: A Proof-of-Concept for a Simple and Low-Cost Tool to Recognize a Possible Viral Etiology in Community-Acquired Pneumonia Patients during COVID-19 Outbreak. Diagnostics 2020, 10, 619. [Google Scholar] [CrossRef]
- Liao, J.J.Z. Sample size calculation for an agreement study. Pharm. Stat. 2010, 9, 125–132. [Google Scholar] [CrossRef]
- Cicchetti, D.V. Guidelines, Criteria, and Rules of Thumb for Evaluating Normed and Standardized Assessment Instruments in Psychology. Psychol. Assess. 1994, 6, 284–290. [Google Scholar] [CrossRef]
- Altman, D.G.; Bland, J.M. Measurement in Medicine: The Analysis of Method Comparison Studies. Statistician 1983, 32, 307–317. [Google Scholar] [CrossRef]
- Cadranel, J.F.; Rufat, P.; Degos, F. Practices of liver biopsy in France: Results of a prospective nationwide survey. Hepatology 2000, 32, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, B.; Leary, W.P.; Naran, A.D.; Maharaj, R.J.; Cooppan, R.M.; Pirie, D.; Pudifin, J.D. Sampling Variability and Its Influence on the Diagnostic Yield of Percutaneous Needle Biopsy of the Liver. Lancet 1986, 327, 523–525. [Google Scholar] [CrossRef]
- Bedossa, P. Intraobserver and Interobserver Variations in Liver Biopsy Interpretation in Patients with Chronic Hepatitis, C. Hepatology 1994, 20, 15–20. [Google Scholar] [CrossRef]
- Masuzaki, R.; Tateishi, R.; Yoshida, H.; Goto, E.; Sato, T.; Ohki, T.; Imamura, J.; Goto, T.; Kanai, F.; Kato, N.; et al. Prospective risk assessment for hepatocellular carcinoma development in patients with chronic hepatitis C by transient elastography. Hepatology 2009, 49, 1954–1961. [Google Scholar] [CrossRef]
- Sigrist, R.M.S.; Liau, J.; el Kaffas, A.; Chammas, M.C.; Willmann, J.K. Ultrasound elastography: Review of techniques and clinical applications. Theranostics 2017, 7, 1303–1329. [Google Scholar] [CrossRef]
- Cho, H.J.; Seo, Y.S.; Lee, K.G.; Hyun, J.J.; An, H.; Keum, B.; Kim, J.H.; Yim, H.J.; Jeen, Y.T.; Lee, H.S.; et al. Serum aminotransferase levels instead of etiology affects the accuracy of transient elastography in chronic viral hepatitis patients. J. Gastroenterol. Hepatol. 2011, 26, 492–500. [Google Scholar] [CrossRef]
- Giuffrè, M.; Fouraki, S.; Comar, M.; Masutti, F.; Crocè, L.S. The importance of transaminases flare in liver elastography: Characterization of the probability of liver fibrosis overestimation by hepatitis C virus-induced cytolysis. Microorganisms 2020, 8, 348. [Google Scholar] [CrossRef] [Green Version]
- Haberkorn, U.; Layer, G.; Rudat, V.; Zuna, I.; Lorenz, A.; van Kaick, G. Ultrasound image properties influenced by abdominal wall thickness and composition. J. Clin. Ultrasound 1993, 21, 423–429. [Google Scholar] [CrossRef]
- Ferraioli, G.; Monteiro, L.B.S. Ultrasound-based techniques for the diagnosis of liver steatosis. World J. Gastroenterol. 2019, 25, 6053–6062. [Google Scholar] [CrossRef]
- Larson, S.P.; Bowers, S.P.; Palekar, N.A.; Ward, J.A.; Pulcini, J.P.; Harrison, S.A. Histopathologic Variability Between the Right and Left Lobes of the Liver in Morbidly Obese Patients Undergoing Roux-en-Y Bypass. Clin. Gastroenterol. Hepatol. 2007, 5, 1329–1332. [Google Scholar] [CrossRef]
- Sasaki, A.; Nitta, H.; Otsuka, K.; Umemura, A.; Baba, S.; Obuchi, T.; Wakabayashi, G. Bariatric surgery and non-alcoholic fatty liver disease: Current and potential future treatments. Front. Endocrinol. 2014, 5, 164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristic | Value |
|---|---|
| Age, years | 53 (41; 57) |
| Gender, female, n (%) | 174 (62.2 %) |
| BMI, kg/m2 | 44 (40; 50) |
| Waist Circumference, cm | 135 (121; 146) |
| SLD, mm | 32 (26; 40) |
| Glycated Hemoglobin, % | 6.3 (± 0.8) |
| Fasting Plasma Glucose, mg/dL | 110 (99; 131) |
| Fasting Plasma Insulin, mIU/L | 20 (13; 36) |
| HOMA-IR Index | 5.10 (3.85; 8.53) |
| HOMA-IR Index >2.73, n (%) | 160 (54.7%) |
| Previous Diagnosis of Type 2 Diabetes Mellitus, n (%) | 49 (16.4%) |
| HDL Cholesterol, mg/dL | 38 (36;43) |
| LDL Cholesterol, mg/dL | 171 (151; 199) |
| Triglycerides, mg/dL | 143 (110; 210) |
| AST, U/L | 26 (21; 32) |
| ALT, U/L | 29 (23; 39) |
| GGT, U/L | 33 (21; 49) |
| ALP, U/L | 75 (64; 102) |
| Total Bilirubin, mg/dL | 0.5 (0.34; 0.68) |
| Albumin, g/dL | 3.9 (3.7; 4.4) |
| Liver Fibrosis (Kleiner), n (%) Score 0 (F0) Score 1 (F1) Score 2 (F2) Score 3 (F3) Score 4 (F4) | 55 (18.4%) 142 (47.5%) 59 (19.7%) 19 (6.4%) 24 (8%) |
| Liver Steatosis Grade, n (%) Score 0 Score 1 Score 2 Score 3 | 74 (24.8%) 106 (35.4%) 71 (23.8%) 48 (16%) |
| Fibrosis Staging According to Philips IU 22 (n = 183) | ||||
| Values | Kleiner F0, n = 24 | Kleiner F1, n = 101 | Kleiner F2, n = 33 | Kleiner F3–4, n = 25 |
| Liver Stiffness (kPa), Median (IQR) | 4.1 (3.6;5.3) | 4.34 (3.6;5.5) | 6.6 (4;6.7) | 8.1 (4.9;9.4) |
| Correctly Classified by Arbitrary Cut-Off | 15/24 (62.5%) | 25/101 (24.75%) | 18/33 (54.5%) | 14/25 (56%) |
| Fibrosis Staging According to Philips Affiniti 70 (n = 125) | ||||
| Values | Kleiner F0 n = 45 | Kleiner F1 n = 54 | Kleiner F2 n = 26 | Kleiner F3–4 n = 20 |
| Liver Stiffness (kPa), Median (IQR) | 4.5 (3.9;6.20) | 4.6 (4.1;6.10) | 6.5 (4.3;7.2) | 8.6 (6.3;9.3) |
| Correctly Classified by Arbitrary Cut-Off | 29/45 (64.4%) | 17/54 (31.5%) | 14/26 (53.8%) | 16/20 (80%) |
| Logistic Regression Probability Models | ||
|---|---|---|
| Impact of Abdominal Wall Stiffness on Elastography–Histology Agreement | ||
| Variables | [M1] Philips IU 22 | [M2] Philips Affiniti 70 |
| Abdominal Wall Thickness | −0.144 [−0.16;−0.13] | −0.591 [−0.617;−0.564] |
| Intercept | 3.99 [3.35;3.63] | 20.86 [18.3;23.4] |
| Nagelkerke Pseudo-R2 | 0.266 | 0.894 |
| AIC | 207.7 | 38.5 |
| BIC | 214.1 | 44.2 |
| AUROC | 0.79 [0.72;0.86] | 0.90 [0.85;0.93] |
| Hosmer–Lemeshow p-value | 0.09 | 0.15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giuffrè, M.; Giuricin, M.; Bonazza, D.; Rosso, N.; Giraudi, P.J.; Masutti, F.; Palmucci, S.; Basile, A.; Zanconati, F.; de Manzini, N.; et al. Optimization of Point-Shear Wave Elastography by Skin-to-Liver Distance to Assess Liver Fibrosis in Patients Undergoing Bariatric Surgery. Diagnostics 2020, 10, 795. https://doi.org/10.3390/diagnostics10100795
Giuffrè M, Giuricin M, Bonazza D, Rosso N, Giraudi PJ, Masutti F, Palmucci S, Basile A, Zanconati F, de Manzini N, et al. Optimization of Point-Shear Wave Elastography by Skin-to-Liver Distance to Assess Liver Fibrosis in Patients Undergoing Bariatric Surgery. Diagnostics. 2020; 10(10):795. https://doi.org/10.3390/diagnostics10100795
Chicago/Turabian StyleGiuffrè, Mauro, Michela Giuricin, Deborah Bonazza, Natalia Rosso, Pablo José Giraudi, Flora Masutti, Stefano Palmucci, Antonio Basile, Fabrizio Zanconati, Nicolò de Manzini, and et al. 2020. "Optimization of Point-Shear Wave Elastography by Skin-to-Liver Distance to Assess Liver Fibrosis in Patients Undergoing Bariatric Surgery" Diagnostics 10, no. 10: 795. https://doi.org/10.3390/diagnostics10100795
APA StyleGiuffrè, M., Giuricin, M., Bonazza, D., Rosso, N., Giraudi, P. J., Masutti, F., Palmucci, S., Basile, A., Zanconati, F., de Manzini, N., Tiribelli, C., Palmisano, S., & Crocè, L. S., for the ElastPQ-XL-Group. (2020). Optimization of Point-Shear Wave Elastography by Skin-to-Liver Distance to Assess Liver Fibrosis in Patients Undergoing Bariatric Surgery. Diagnostics, 10(10), 795. https://doi.org/10.3390/diagnostics10100795







