Anaplastic Thyroid Carcinoma Histologically Mimicking a Plasmacytoma
Abstract
1. Introduction
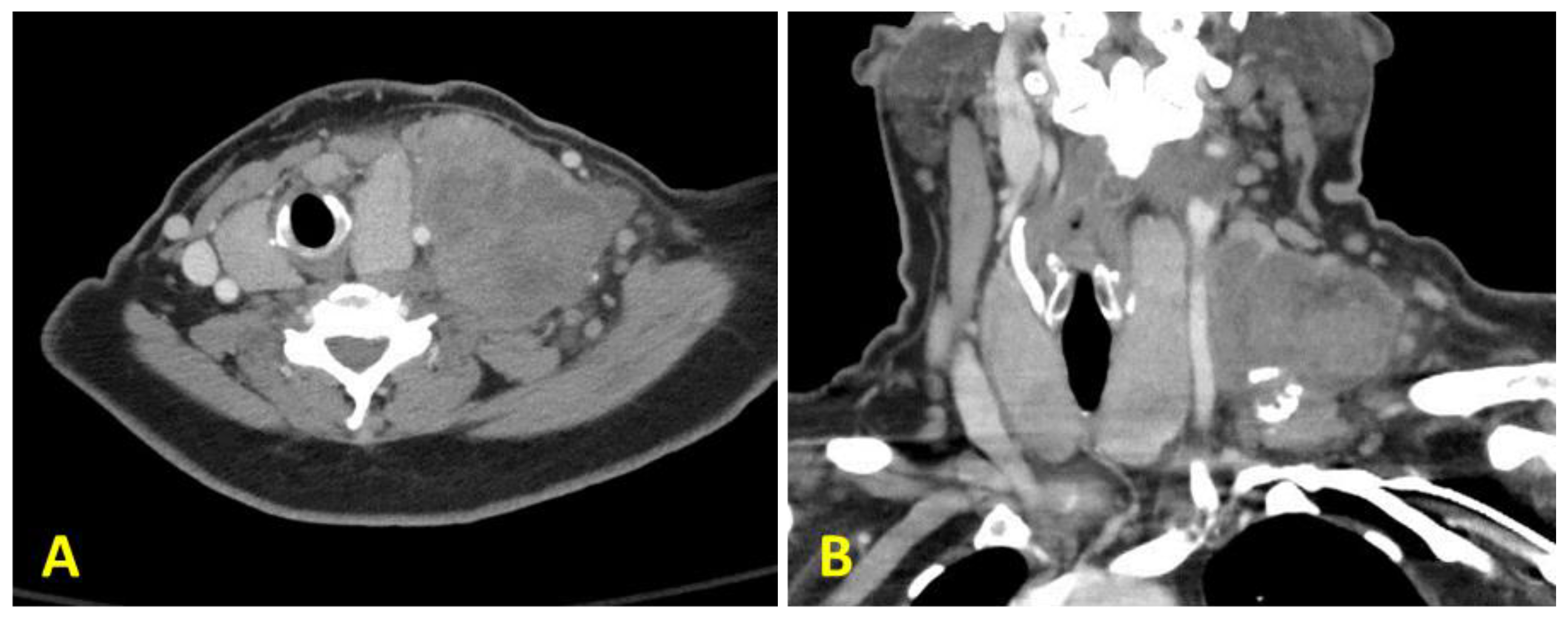
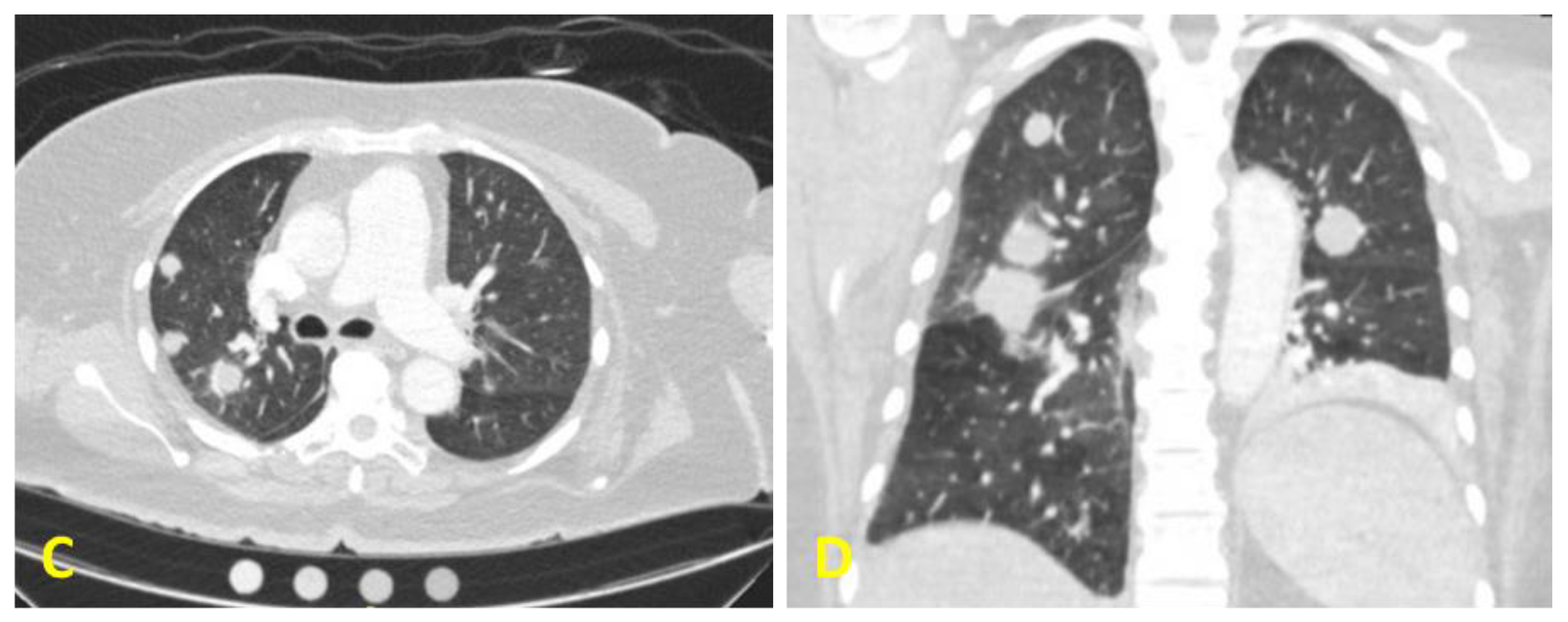
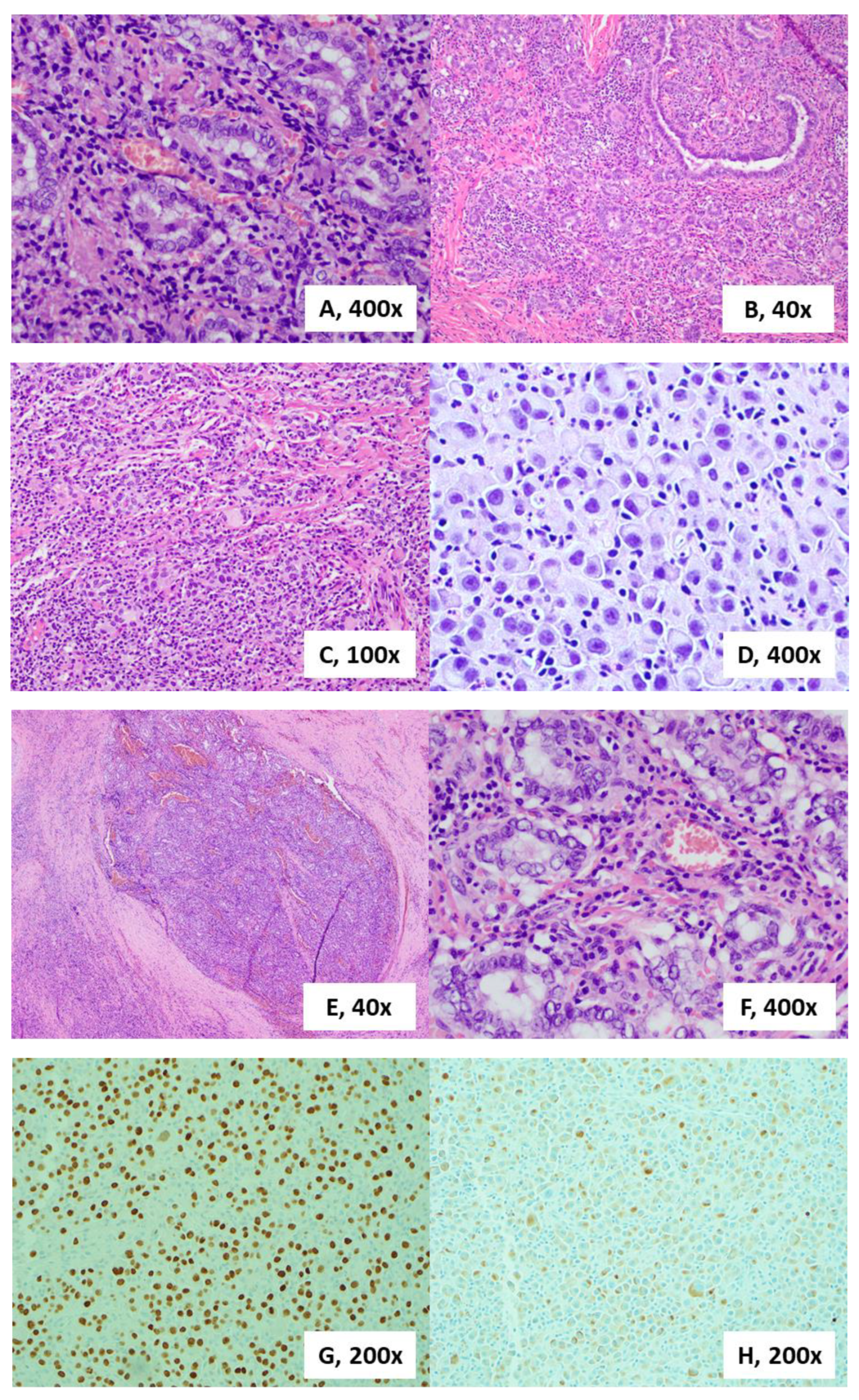
2. Case Presentation
3. Discussion
Author Contributions
Funding
Statement
Conflicts of Interest
References
- Nagaiah, G.; Hossain, A.; Mooney, C.J.; Parmentier, J.; Remick, S.C. Anaplastic thyroid cancer: A review of epidemiology, pathogenesis, and treatment. J. Oncol. 2011, 2011, 542358. [Google Scholar] [CrossRef] [PubMed]
- Giuffrida, D.; Gharib, H. Anaplastic thyroid carcinoma: Current diagnosis and treatment. Ann. Oncol. 2000, 11, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Kebebew, E.; Greenspan, F.S.; Clark, O.H.; Woeber, K.A.; McMillan, A. Anaplastic thyroid carcinoma: Treatment outcome and prognostic factors. Cancer 2005, 103, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Are, C.; Shaha, A.R. Anaplastic thyroid carcinoma: Biology, pathogenesis, prognostic factors, and treatment approaches. Ann. Surg. Oncol. 2006, 13, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Hundahl, S.A.; Fleming, I.D.; Fremgen, A.M.; Menck, H.R. A National Cancer Data Base report on 53,856 cases of thyroid carcinoma treated in the U.S., 1985–1995. Cancer 1998, 83, 2638–2648. [Google Scholar] [CrossRef]
- Kunstman, J.W.; Juhlin, C.C.; Goh, G.; Brown, T.C.; Stenman, A.; Healy, J.M.; Rubinstein, J.C.; Choi, M.; Kiss, N.; Nelson-Williams, C.; et al. Characterization of the mutational landscape of anaplastic thyroid cancer via whole-exome sequencing. Hum. Mol. Genet. 2015, 24, 2318–2329. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, M.N.; Kimura, E.T.; Gandhi, M.; Biddinger, P.W.; Knauf, J.A.; Basolo, F.; Zhu, Z.; Giannini, R.; Salvatore, G.; Fusco, A.; et al. BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J. Clin. Endocrinol. Metab. 2003, 88, 5399–5404. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; Zafereo, M.; Gunn, G.B.; Ferrarotto, R. Anaplastic thyroid carcinoma: Treatment in the age of molecular targeted therapy. J. Oncol. Pract. 2016, 12, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Swaak-Kragten, A.T.; de Wilt, J.H.; Schmitz, P.I.; Bontenbal, M.; Levendag, P.C. Multimodality treatment for anaplastic thyroid carcinoma–treatment outcome in 75 patients. Radiother. Oncol. 2009, 92, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, A.; Rao, A.; Ang, K.K.; Garden, A.S.; Morrison, W.H.; Rosenthal, D.I.; Evans, D.B.; Clayman, G.; Sherman, S.I.; Schwartz, D.L. Anaplastic thyroid cancer: Clinical outcomes with conformal radiotherapy. Head Neck 2010, 32, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Ain, K.B.; Egorin, M.J.; DeSimone, P.A.; Collaborative Anaplastic Thyroid Cancer Health Intervention Trials (CATCHIT) Group*. Treatment of anaplastic thyroid carcinoma with paclitaxel: Phase 2 trial using ninety-six-hour infusion. Thyroid 2000, 10, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Rosove, M.H.; Peddi, P.F.; Glaspy, J.A. BRAF V600E inhibition in anaplastic thyroid cancer. N. Engl. J. Med. 2013, 368, 684–685. [Google Scholar] [CrossRef] [PubMed]
- Landa, I.; Ibrahimpasic, T.; Boucai, L.; Sinha, R.; Knauf, J.A.; Shah, R.H.; Dogan, S.; Ricarte-Filho, J.C.; Krishnamoorthy, G.P.; Xu, B.; et al. Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J. Clin. Investig. 2016, 126, 1052–1066. [Google Scholar] [CrossRef] [PubMed]
- Onoda, N.; Ito, Y.; Ito, K.; Sugitani, I.; Takahashi, S.; Yamaguchi, I.; Kawakami, Y.; Tsukada, K. Phase II clinical trial of sorafenib in Japanese patients with anaplastic thyroid carcinoma and locally advanced or metastatic medullary thyroid carcinoma. Thyroid 2015, 25, A1202015. [Google Scholar]
- Keutgen, X.M.; Sadowski, S.M.; Kebebew, E. Management of anaplastic thyroid cancer. Gland Surg. 2015, 4, 44–51. [Google Scholar] [PubMed]
- Smallridge, R.C.; Ain, K.B.; Asa, S.L.; Bible, K.C.; Brierley, J.D.; Burman, K.D.; Kebebew, E.; Lee, N.Y.; Nikiforov, Y.E.; Sara Rosenthal, M.; et al. American Thyroid Association guidelines for management of patients with anaplastic thyroid cancer. Thyroid 2012, 22, 1104–1139. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.K.; Chan, J.K.; Tang, S.K. Paucicellular variant of anaplastic thyroid carcinoma: A mimic of Riedel’s thyroiditis. Amer. J. Clin. Path. 1996, 105, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Veran-Taguibao, S.; Labadie, K.P.; Wang, B.Y.; Nangia, C.; Lin, H.; Lin, F. Aggressive Thyroid Carcinoma Mimicking a Benign Histiocytic Proliferation-Utility of Tumor Markers by Immunohistochemical Analysis. Cancer Res. Rep. 2018, 1, 1–5. [Google Scholar]
- Oktay, M.H.; Smolkin, M.B.; Williams, M.; Cajigas, A. Metastatic anaplastic carcinoma of the thyroid mimicking squamous cell carcinoma: Report of a case of a challenging cytologic diagnosis. Acta Cytol. 2006, 50, 201–204. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mo, J.H.; Tan, D.; Zhao, X.S.; Tjoa, T.; Wang, B.Y. Anaplastic Thyroid Carcinoma Histologically Mimicking a Plasmacytoma. Diagnostics 2020, 10, 29. https://doi.org/10.3390/diagnostics10010029
Mo JH, Tan D, Zhao XS, Tjoa T, Wang BY. Anaplastic Thyroid Carcinoma Histologically Mimicking a Plasmacytoma. Diagnostics. 2020; 10(1):29. https://doi.org/10.3390/diagnostics10010029
Chicago/Turabian StyleMo, Joshua H., Donald Tan, Xiaohui S. Zhao, Tjoson Tjoa, and Beverly Y. Wang. 2020. "Anaplastic Thyroid Carcinoma Histologically Mimicking a Plasmacytoma" Diagnostics 10, no. 1: 29. https://doi.org/10.3390/diagnostics10010029
APA StyleMo, J. H., Tan, D., Zhao, X. S., Tjoa, T., & Wang, B. Y. (2020). Anaplastic Thyroid Carcinoma Histologically Mimicking a Plasmacytoma. Diagnostics, 10(1), 29. https://doi.org/10.3390/diagnostics10010029



