Efficient Arrangement of the Replication Fork Trap for In Vitro Propagation of Monomeric Circular DNA in the Chromosome-Replication Cycle Reaction
Abstract
1. Introduction
2. Materials and Methods
2.1. Replication Cycle Reaction
2.2. Purified Proteins
2.3. Plasmid Construction
3. Results
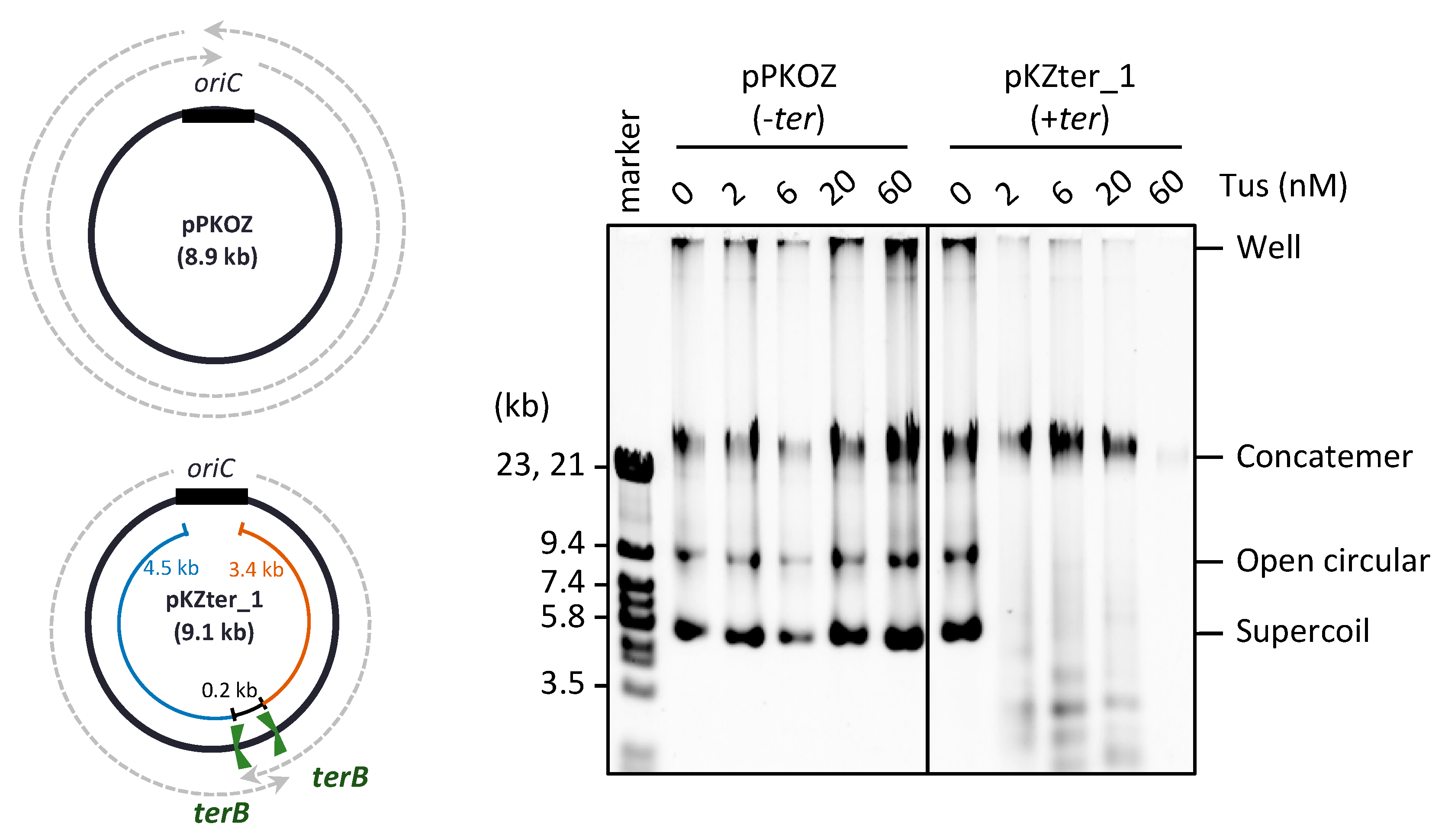
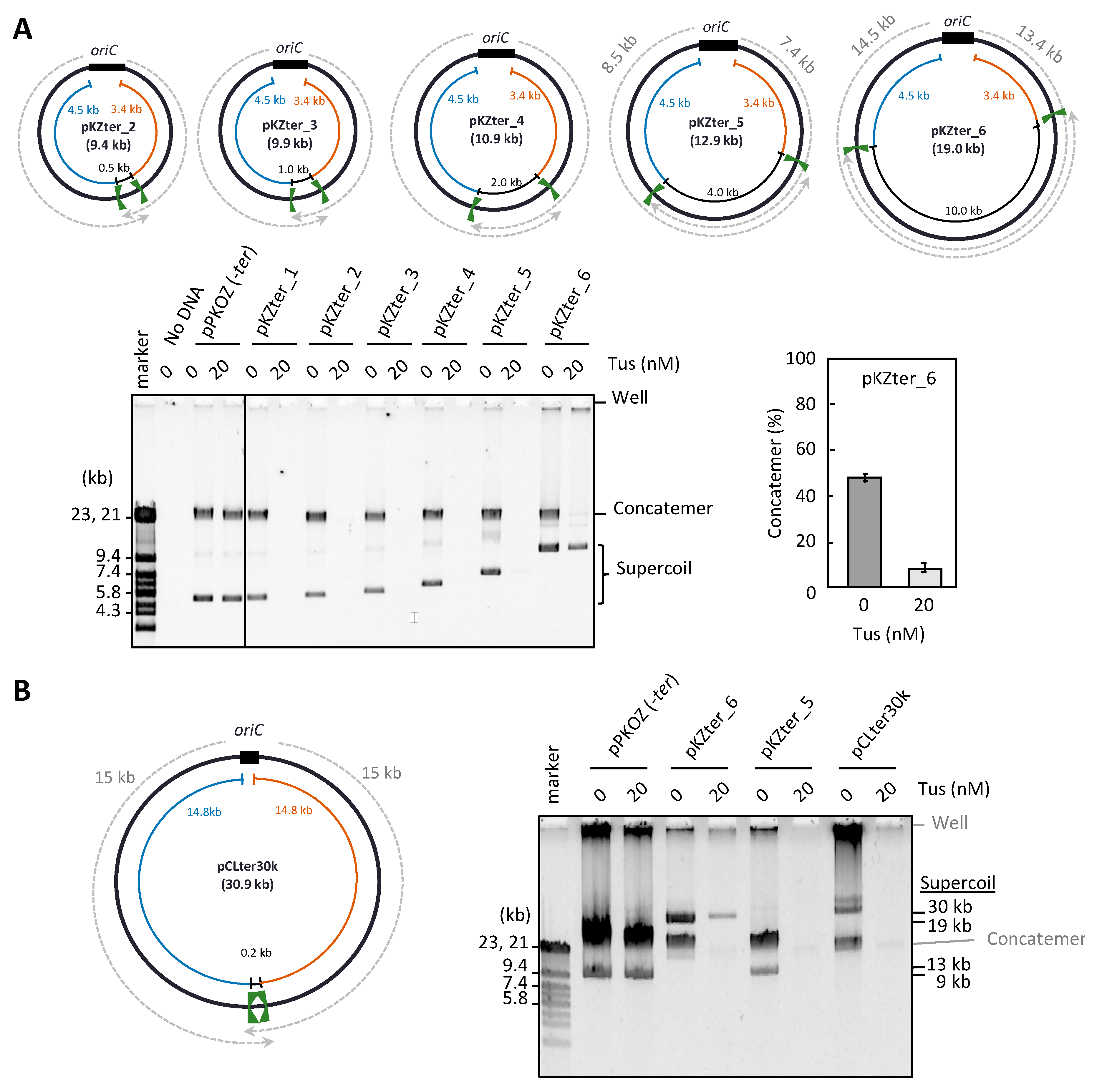
3.1. Chromosome-Like Arrangement of oriC-ter Sites Is Detrimental to the RCR Propagation of Circular DNA
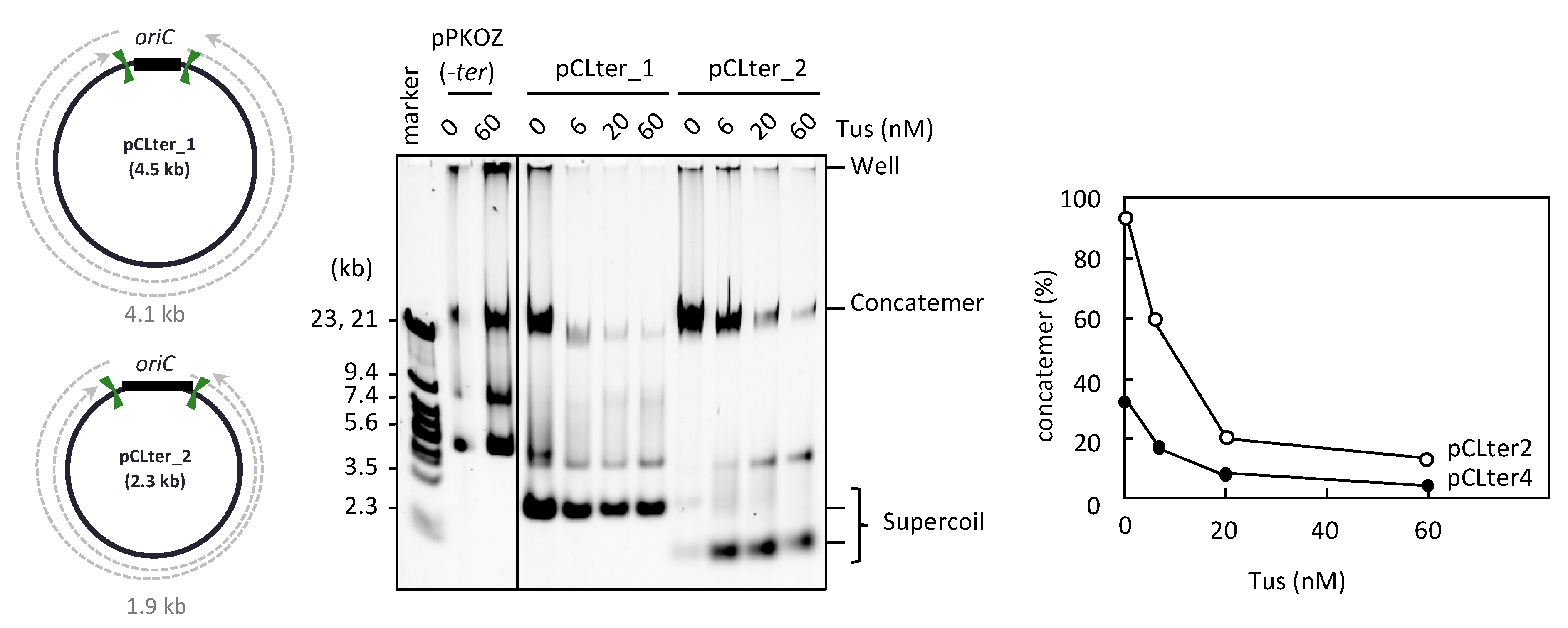
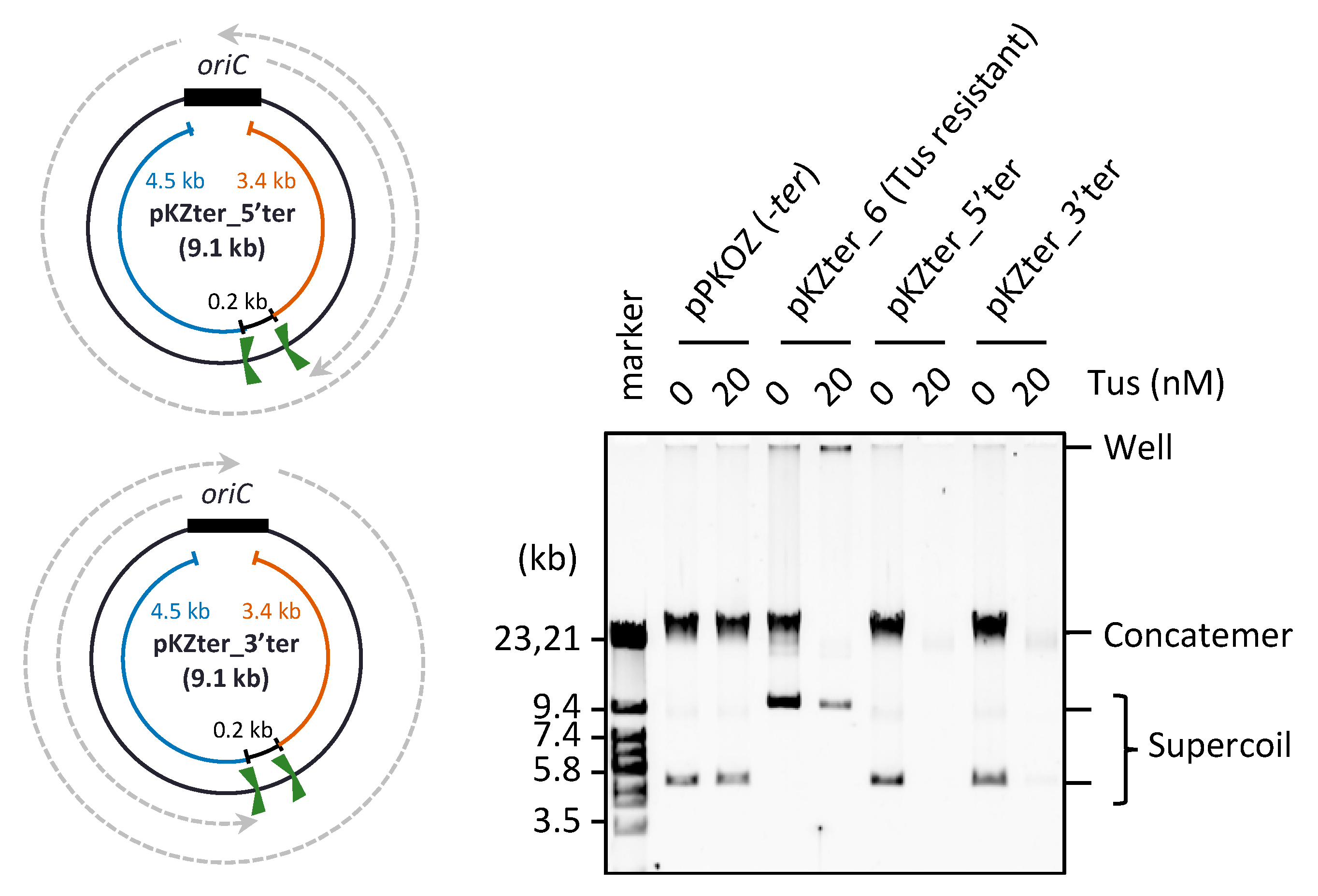
3.2. The Arrangement of ter Sites, Rather Than the Distance between Them, Is Critical for Repetition of the Replication Cycle
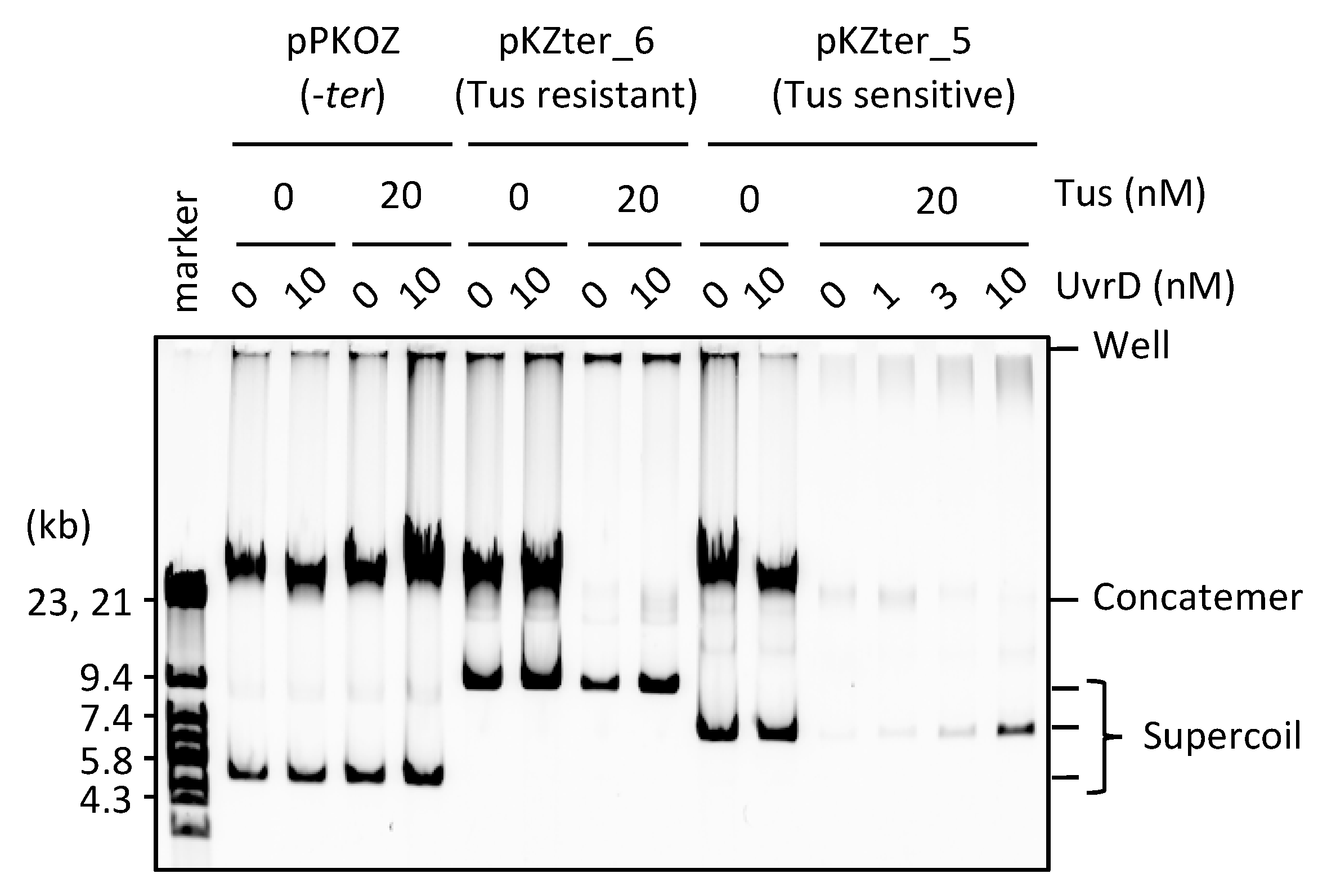
3.3. UvrD Helicase Rescues the Detrimental Effect of the Tus-ter Traps in RCR
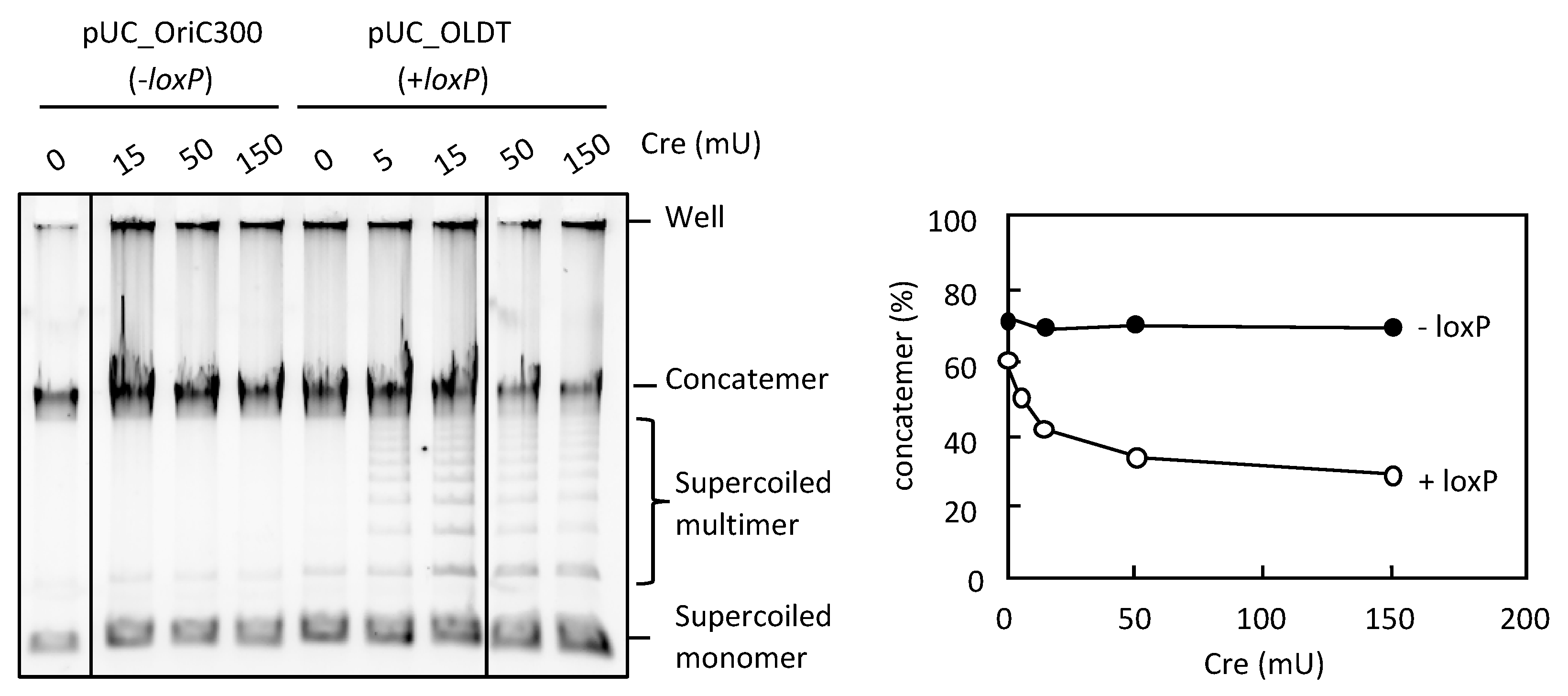
3.4. Resolution of the DNA Concatemers by the Cre-loxP System in RCR
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ichihashi, N.; Yomo, T. Constructive Approaches for Understanding the Origin of Self-Replication and Evolution. Life 2016, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Forster, A.C.; Church, G.M. Towards synthesis of a minimal cell. Mol. Syst. Biol. 2006, 2, 45. [Google Scholar] [CrossRef] [PubMed]
- Beattie, T.R.; Reyes-Lamothe, R. A Replisome’s journey through the bacterial chromosome. Front. Microbiol. 2015, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kornberg, A.; Baker, T.A. DNA Replication, 2nd ed.; W.H. Freeman: New York, NY, USA, 1992. [Google Scholar]
- Kaguni, J.M.; Kornberg, A. Replication initiated at the origin (oriC) of the E. coli chromosome reconstituted with purified enzymes. Cell 1984, 38, 183–190. [Google Scholar] [CrossRef]
- Funnell, B.E.; Baker, T.A.; Kornberg, A. Complete enzymatic replication of plasmids containing the origin of the Escherichia coli chromosome. J. Biol. Chem. 1986, 261, 5616–5624. [Google Scholar] [PubMed]
- Ozaki, S.; Katayama, T. DnaA structure, function, and dynamics in the initiation at the chromosomal origin. Plasmid 2009, 62, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Kaguni, J.M. Replication initiation at the Escherichia coli chromosomal origin. Curr. Opin. Chem. Biol. 2011, 15, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.; O’Donnell, M.M. Cellular DNA replicases: components and dynamics at the replication fork. Annu. Rev. Biochem. 2005, 74, 283–315. [Google Scholar] [CrossRef] [PubMed]
- McHenry, C.S. DNA Replicases from a Bacterial Perspective. Annu. Rev. Biochem. 2011, 80, 403–436. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, K.; Katayama, T.; Iwai, S.; Hidaka, M.; Horiuchi, T.; Maki, H. Fate of DNA replication fork encountering a single DNA lesion during oriC plasmid DNA replication in vitro. Genes Cells 2003, 8, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, H.; Marians, K.J. Initiation of bidirectional replication at the chromosomal origin is directed by the interaction between helicase and primase. J. Biol. Chem. 1999, 274, 27244–27248. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, H.; Marians, K.J. Primase couples leading- and lagging-strand DNA synthesis from oriC. J. Biol. Chem. 1994, 269, 6058–6063. [Google Scholar] [PubMed]
- Hiasa, H.; Marians, K.J. Tus prevents overreplication of oriC plasmid DNA. J. Biol. Chem. 1994, 269, 26959–26968. [Google Scholar] [PubMed]
- Neylon, C.; Kralicek, A.V.; Hill, T.M.; Dixon, N.E. Replication termination in Escherichia coli: structure and antihelicase activity of the Tus-ter complex. Microbiol. Mol. Biol. Rev. 2005, 69, 501–526. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.M.; Tecklenburg, M.L.; Pelletier, A.J.; Kuempel, P.L. tus, the trans-acting gene required for termination of DNA replication in Escherichia coli, encodes a DNA-binding protein. Proc. Natl. Acad. Sci. USA 1989, 86, 1593–1597. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Hidaka, M.; Horiuchi, T. Evidence of a ter specific binding protein essential for the termination reaction of DNA replication in Escherichia coli. EMBO J. 1989, 8, 2435–2441. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.M.; Marians, K.J. Escherichia coli Tus protein acts to arrest the progression of DNA replication forks in vitro. Proc. Natl. Acad. Sci. USA 1990, 87, 2481–2485. [Google Scholar] [CrossRef] [PubMed]
- Khatri, G.S.; MacAllister, T.; Sista, P.R.; Bastia, D. The replication terminator protein of E. coli is a DNA sequence-specific contra-helicase. Cell 1989, 59, 667–674. [Google Scholar] [CrossRef]
- Mulcair, M.D.; Schaeffer, P.M.; Oakley, A.J.; Cross, H.F.; Neylon, C.; Hill, T.M.; Dixon, N.E. A Molecular Mousetrap Determines Polarity of Termination of DNA Replication in E. coli. Cell 2006, 125, 1309–1319. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.H.U.M.; Kornberg, A.; Hidakat, M.; Kobayashit, T.; Horiuchit, T. Escherichia coli replication termination protein impedes the action of helicases. Proc. Natl. Acad. Sci. USA 1989, 86, 9104–9108. [Google Scholar] [CrossRef] [PubMed]
- Roecklein, B.; Pelletier, A.; Kuempel, P. The tus gene of Escherichia coli: autoregulation, analysis of flanking sequences and identification of a complementary system in Salmonella typhimurium. Res. Microbiol. 1991, 142, 169–175. [Google Scholar] [CrossRef]
- Su’etsugu, M.; Takada, H.; Katayama, T.; Tsujimoto, H. Exponential propagation of large circular DNA by reconstitution of a chromosome-replication cycle. Nucleic Acids Res. 2017, 45, 11525–11534. [Google Scholar] [CrossRef] [PubMed]
- Iwadate, Y.; Honda, H.; Sato, H.; Hashimoto, M.; Kato, J. Oxidative stress sensitivity of engineered Escherichia coli cells with a reduced genome. FEMS Microbiol. Lett. 2011, 322, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Heyneker, H.L. Single-step assembly of a gene and entire plasmid from large numbers of oligodeoxyribonucleotides. Genes 1995, 164, 49–53. [Google Scholar]
- Vicente, M.; Gomez, M.J.; Ayala, J.A. Regulation of transcription of cell division genes in the Eschericia coli dcw cluster. Cell. Mol. Life Sci. 1998, 54, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Smelkova, N.; Marians, K.J. Timely Release of Both Replication Forks from oriC Requires Modulation of Origin Topology. J. Biol. Chem. 2001, 276, 39186–39191. [Google Scholar] [CrossRef] [PubMed]
- Tanner, N.A.; Loparo, J.J.; Hamdan, S.M.; Jergic, S.; Dixon, N.E.; van Oijen, A.M. Real-time single-molecule observation of rolling-circle DNA replication. Nucleic Acids Res. 2009, 37, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Bidnenko, V.; Lestini, R.; Michel, B. The Escherichia coli UvrD helicase is essential for Tus removal during recombination-dependent replication restart from Ter sites. Mol. Microbiol. 2006, 62, 382–396. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, K.; Van Duyne, G.D. Cre–loxP biochemistry. Methods 2002, 28, 374–383. [Google Scholar] [CrossRef]
- Duggin, I.G.; Wake, R.G.; Bell, S.D.; Hill, T.M. The replication fork trap and termination of chromosome replication. Mol. Microbiol. 2008, 70, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Rocha, E.P.C.; Danchin, A. Gene essentiality determines chromosome organisation in bacteria. Nucleic Acids Res. 2003, 31, 6570–6577. [Google Scholar] [CrossRef] [PubMed]
- Krabbe, M.; Zabielski, J.; Bernander, R.; Nordström, K. Inactivation of the replication-termination system affects the replication mode and causes unstable maintenance of plasmid R1. Mol. Microbiol. 1997, 24, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, H.; Marians, K.J. Differential inhibition of the DNA translocation and DNA unwinding activities of DNA helicases by the Escherichia coli tus protein. J. Biol. Chem. 1992, 267, 11379–11385. [Google Scholar] [PubMed]
- Runyon, G.T.; Bear, D.G.; Lohman, T.M. Escherichia coli helicase II (UvrD) protein initiates DNA unwinding at nicks and blunt ends. Proc. Natl. Acad. Sci. USA 1990, 87, 6383–6387. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasebe, T.; Narita, K.; Hidaka, S.; Su’etsugu, M. Efficient Arrangement of the Replication Fork Trap for In Vitro Propagation of Monomeric Circular DNA in the Chromosome-Replication Cycle Reaction. Life 2018, 8, 43. https://doi.org/10.3390/life8040043
Hasebe T, Narita K, Hidaka S, Su’etsugu M. Efficient Arrangement of the Replication Fork Trap for In Vitro Propagation of Monomeric Circular DNA in the Chromosome-Replication Cycle Reaction. Life. 2018; 8(4):43. https://doi.org/10.3390/life8040043
Chicago/Turabian StyleHasebe, Tomonori, Kouhei Narita, Shiomi Hidaka, and Masayuki Su’etsugu. 2018. "Efficient Arrangement of the Replication Fork Trap for In Vitro Propagation of Monomeric Circular DNA in the Chromosome-Replication Cycle Reaction" Life 8, no. 4: 43. https://doi.org/10.3390/life8040043
APA StyleHasebe, T., Narita, K., Hidaka, S., & Su’etsugu, M. (2018). Efficient Arrangement of the Replication Fork Trap for In Vitro Propagation of Monomeric Circular DNA in the Chromosome-Replication Cycle Reaction. Life, 8(4), 43. https://doi.org/10.3390/life8040043



