Enhancing Alkane Production in Cyanobacterial Lipid Droplets: A Model Platform for Industrially Relevant Compound Production
Abstract
:1. Introduction
2. Results
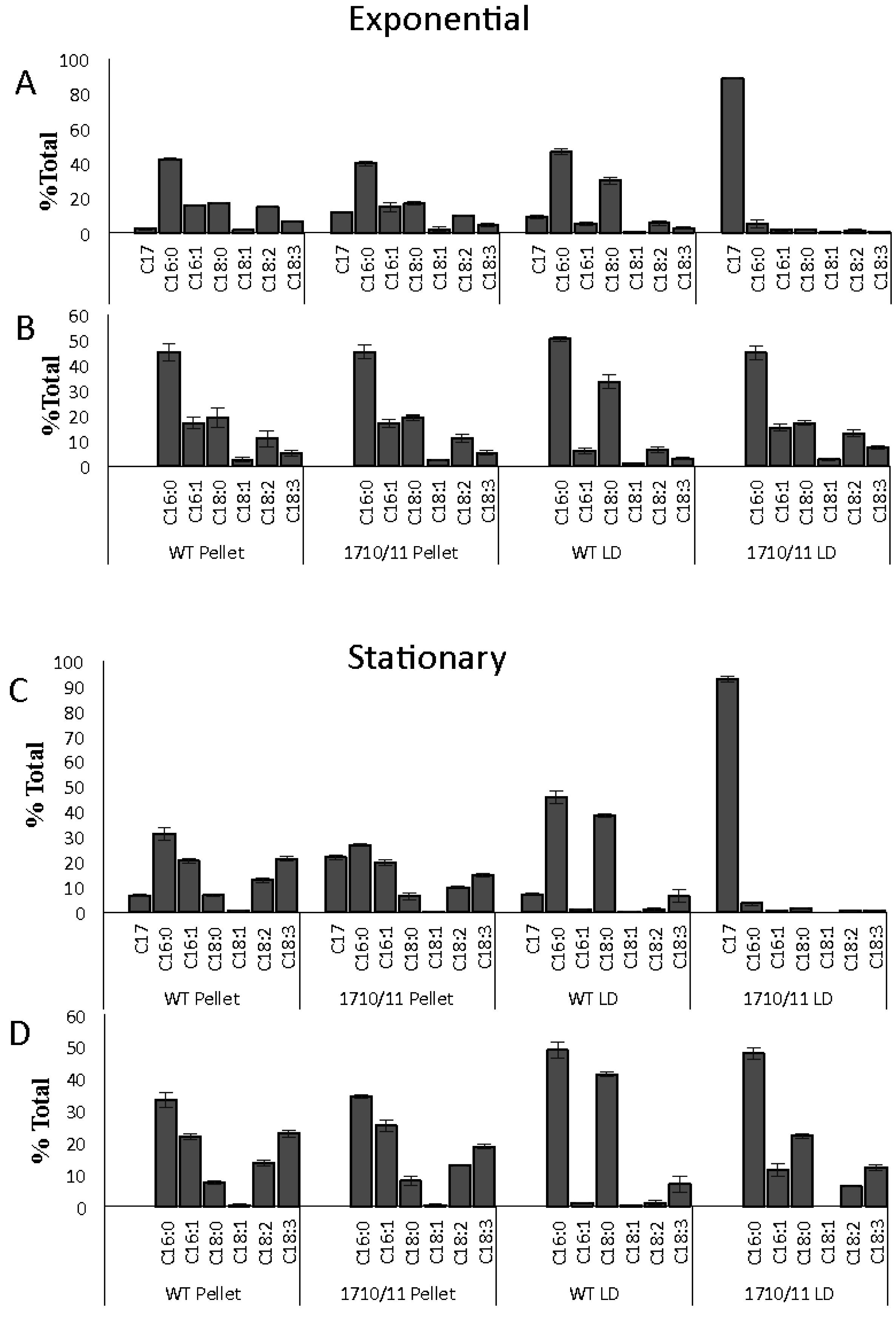
2.1. Multiple Copies of Aar/Adc Leads to Enhanced C17 Alkane Production Specifically in Lipid Droplets
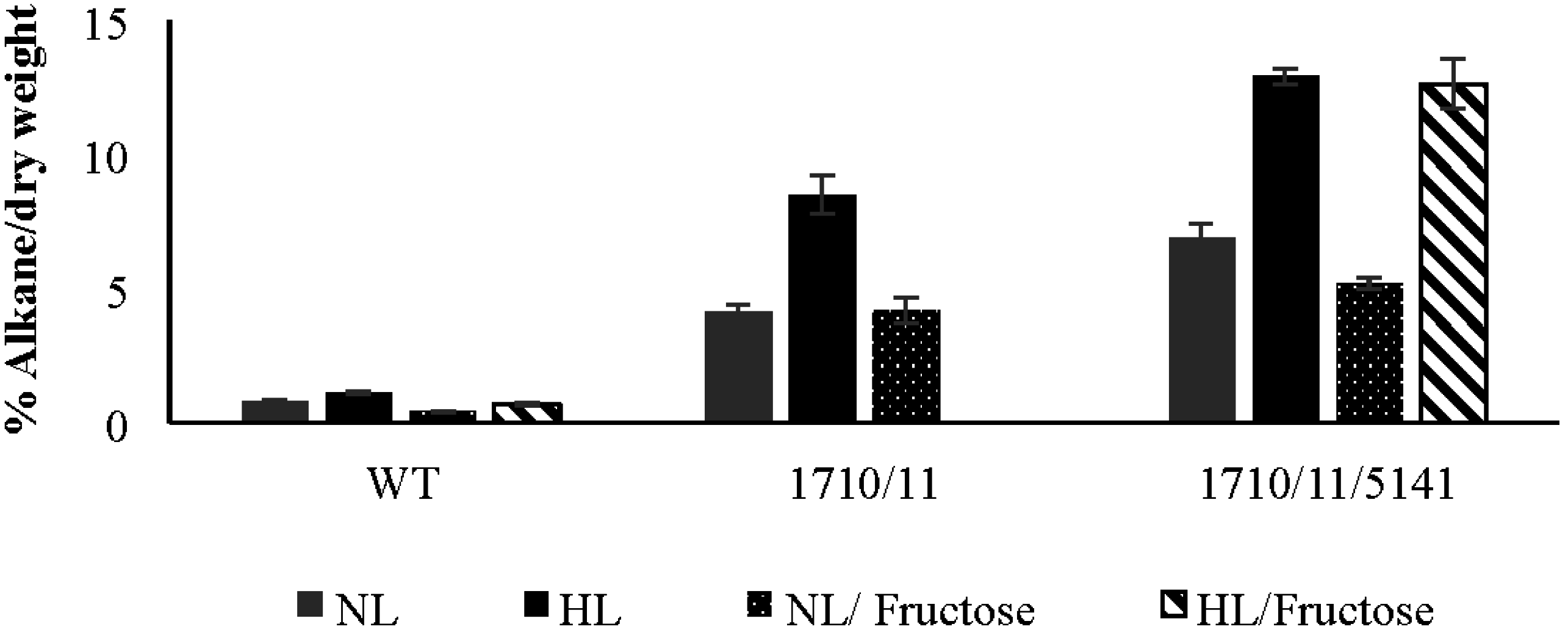
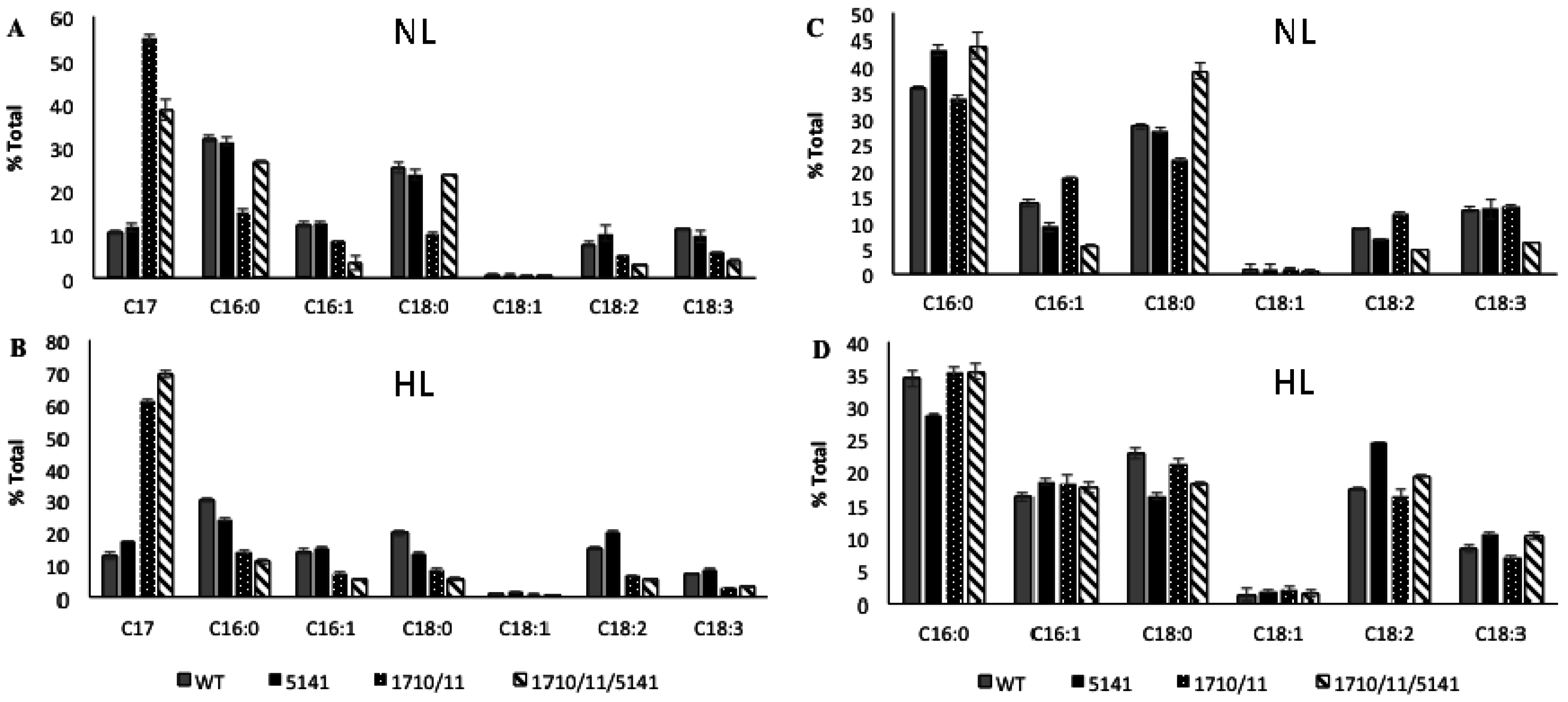
2.2. Phenotypic Effects of Aar/Adc Over-Expression
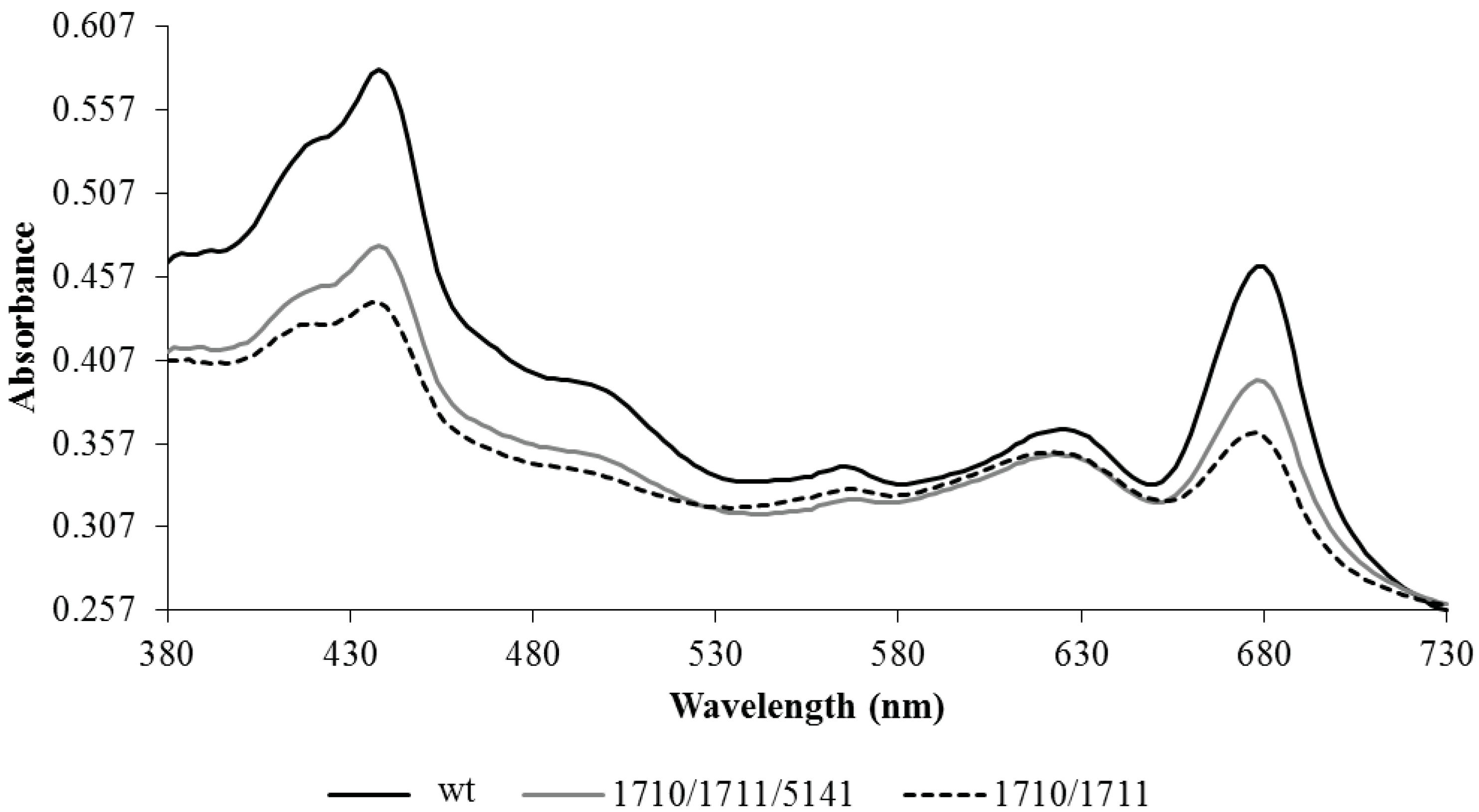
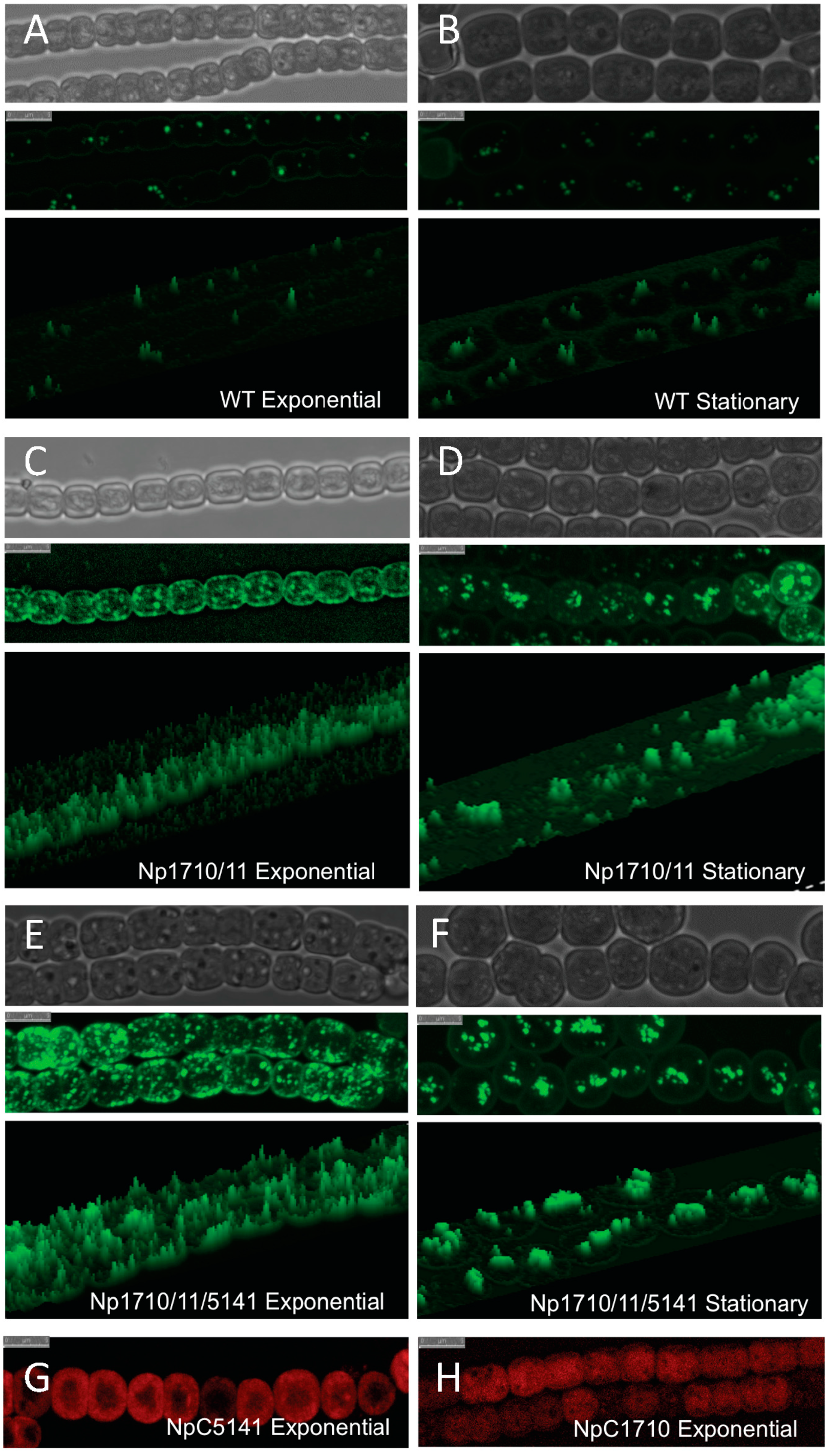
2.3. Discovery and Use of a Novel Gene Enhancing Alkane Production
2.4. Exogenously Supplied Carbon Enhances Growth but Not Alkane Production
2.5. Cellular Localization of Proteins Enhancing Alkane Production
3. Discussion
4. Materials and Methods
4.1. Strains and Growth Conditions
4.2. Plasmid and Strain Construction
4.3. Microscopy
4.4. Relative Heptadecane and Fatty Acids Composition of Whole Cells, LDs, and Cell Pellets
4.5. Heptadecane Quantitation
4.6. Whole-Cell Spectral Analysis
Acknowledgments
Author Contributions
Conflict of Interests
References
- Lu, Z. A perspective: Photosynthetic production of fatty acid-based biofuels in genetically engineered cyanobacteria. Biotechnol. Adv. 2010, 28, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Ducat, D.C.; Way, J.C.; Silver, P.A. Engineering cyanobacteria to generate high-value products. Trends Biotechnol. 2011, 29, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.M.P.; Stsumi, S. Cyanobacterial biofuel production. J. Biotechnol. 2012, 162, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Rosgaard, L.; de Porcellinis, A.J.; Jacobsen, J.H.; Frigaard, N.-U.; Sajyragu, Y. Bioengineering of carbon fixation, biofuels, and biochemicals in cyanobacteria and plants. J. Biotechnol. 2013, 162, 134–147. [Google Scholar] [CrossRef]
- Huntley, M.E.; Redalje, D.G. CO2 mitigation and renewable oil from photosynthetic microbes: A new appraisal. Mitig. Adapt. Strateg. Glob. Chang. 2007, 12, 573–608. [Google Scholar] [CrossRef]
- Dismukes, G.C.; Carrieri, D.; Bennette, N.; Ananyev, G.M.; Posewitz, M.C. Aquatic phototrophs: Efficient alternatives to land-based crops for biofuels. Curr. Opin. Biotechnol. 2008, 19, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Kessler, F.; Vidi, P.-A. Plastoglobule lipid bodies: Their functions in chloroplasts and their potential for applications. Adv. Biochem. Engin/Biotechnol. 2007, 107, 153–172. [Google Scholar]
- Austin, J.R.; Frost, E.; Vidi, P.-A.; Kessler, F.; Staehelina, L.A. Plastoglobules are lipoprotein subcompartments of the chloroplast that are permanently coupled to thylakoid membranes and contain biosynthetic enzymes. Plant Cell 2006, 18, 1693–1703. [Google Scholar] [CrossRef] [PubMed]
- Rey, P.; Gillet, B.; Römer, S.; Eymery, F.; Massimino, J.; Peltier, G.; Kuntz, M. Over-expression of a pepper plastid lipid-associated protein in tobacco leads to changes in plastid ultrastructure and plant development upon stress. Plant J. 2000, 21, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Khandelia, H.; Duelund, L.; Pakkanen, K.I.; Ipsen, J.H. Triglyceride blisters in lipid bilayers: Implications for lipid droplet biogenesis and the mobile lipid signal in cancer cell membranes. PLoS One 2010, 5, e12811. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.J.; Vance, J. Mechanisms of lipid-body formation. TIBS 1999, 24, 109–115. [Google Scholar] [PubMed]
- Murphy, D.J. The dynamic roles of intracellular lipid droplets: From archaea to mammals. Protoplasma 2012, 249, 541–585. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.T.; Ullrich, N.; Joo, S.; Waffenschidt, S.; Goodenough, U. Algal lipid bodies: Stress induction, purification, and biochemical characterization in wild-type and starchless chlamydomonas reinhardtii. Eukaryot. Cell 2009, 8, 1856–1868. [Google Scholar] [CrossRef] [PubMed]
- Peramuna, A.; Summers, M.L. Composition and occurrence of lipid droplets in the cyanobacterium Nostoc punctiforme. Arch. Microbiol. 2014, 196, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Christi, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Demirbas, A. Progress and recent trends in biodiesel fuels. Energy Convers. Man. 2009, 50, 14–34. [Google Scholar] [CrossRef]
- Daroch, M.; Geng, S.; Wang, G. Recent advances in liquid biofuel production from algal feedstocks. Appl. Energy 2013, 102, 1371–1381. [Google Scholar] [CrossRef]
- Biodeisel Handling and Use Guide. Available online: http://www.biodiesel.org/docs/using-hotline/nrel-handling-and-use.pdf?sfvrsn=4 (accessed on 18 March 2015).
- Ladygina, N.; Dedyukhina, E.G.; Vainshtein, M.B. A review on microbial synthesis of hydrocarbons. Process Biochem. 2006, 41, 1001–1014. [Google Scholar] [CrossRef]
- Winters, K.; Parker, P.L.; Van Baalen, C. Hydrocarbons of blue-green algae: Geochemical signifficance. Science 1969, 163, 467–468. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M.; Srebnik, M. Variability of hydrocarbon and fatty acid components in cultures of the filamentous cyanobacterium Scytonema sp. isolated from microbial community “black cover” of limestone walls in Jerusalem. Biochem. (Moscow) 2002, 67, 1276–1282. [Google Scholar] [CrossRef]
- Schirmer, A.; Rude, M.A.; Li, X.; Popova, E.; del Cardayre, S.B. Microbial biosynthesis of alkanes. Science 2010, 329, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, X.; Lu, X. Engineering cyanobacteria to improve photosynthetic production of alka(e)nes. Biotechnol. Biofuels 2013, 6, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Nardini, M.; Dijkstra, B.W. α/β hydrolase fold enzymes: The family keeps growing. Curr. Opin. Struct. Biol. 1999, 9, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Marchler-Bauer, A.; Lu, S.; Anderson, J.B.; Chitsaz, F.; Derbyshire, M.K.; DeWeese-Scott, C.K.; Fong, J.H.; Geer, L.Y.; Geer, R.C.; Gonzales, N.R.; et al. Cdd: A conserved domain database for the functional annotation of proteins. Nucleic. Acids Res. 2011, 39, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Meeks, J.C.; Elhai, J. Regulation of cellular differentiation in filamentous cyanobacteria in free-living and plant-associated symbiotic growth states. Microbiol. Mol. Biol. Rev. 2002, 66, 94–121. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.M.; Yao, L.; Gao, Q.Q.; Wang, W.H.; Qi, F.X.; Lu, X.F. Photosynthesis driven conversion of carbon dioxide to fatty alcohols and hydrocarbons in cyanobacteria. Metab. Eng. 2011, 13, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Howard, T.P.; Middelhaufe, S.; Moore, K.; Edner, C.; Kolak, D.M.; Taylor, G.N.; Parker, D.A.; Lee, R.; Smirnoff, N.; Aves, S.J.; et al. Synthesis of customized petroleum-replica fuel molecules by targeted modification of free fatty acid pools in Escherichia coli. Proc. Natl. Acad. Sci. USA 2013, 110, 7636–7641. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, T.J.; Simon, S.A.; MacDonald, R.C. The organization of n-alkanes in lipid bilayers. Biochim. Biophys. Acta-Biomembranes 1980, 597, 445–463. [Google Scholar] [CrossRef]
- Kern, J.; Zouni, A. Lipids in the structure of photosystem I, photosystem II and cytochrome b6f complex. In Lipids in Photosynthesis: Essential and Regulatory Functions; Wada, H., Murata, N., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 203–242. [Google Scholar]
- Kanervo, E.; Tasaka, Y.; Murata, N.; Aro, E.-M. Membrane lipid unsaturation modulates processing of the photosystem II reaction-center protein D1 at low temperatures. Plant Phys. 1997, 114, 841–849. [Google Scholar] [CrossRef]
- Kaczmarzyk, D.; Fulda, M. Fatty acid activation in cyanobacteria mediated by acyl-acyl carrier protein synthetase enables fatty acid recycling. Plant Phys. 2010, 152, 1598–1610. [Google Scholar] [CrossRef]
- Allen, M.; Arnon, D.I. Studies on nitrogen-fixing blue-green algae. I. Growth and nitrogen fixation by Anabaena cylindrica lemm. Plant Physiol. 1955, 30, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1989. [Google Scholar]
- Summers, M.L.; Wallis, J.G.; Campbell, E.L.; Meeks, J.C. Genetic evidence of a major role for glucos-6-phosphate dehydrogenase in nitrogen fixation and dark growth of the cyanobacterium Nostoc sp. Strain ATCC 29133. J. Bacteriol. 1995, 177, 6184–6194. [Google Scholar] [PubMed]
- Thanbichler, M.; Iniesta, A.A.; Shapiro, L. A comprehensive set of plasmids for vanillate- and xylose-inducible gene expression in Caulobacter crescentus. Nucleic Acids Res. 2007, 35, e137. [Google Scholar] [CrossRef] [PubMed]
- Araujo, P.; Nguyen, T.-T.; Froyland, L.; Wang, J.; Kang, J.X. Evaluation of a rapid method for the quantitative analysis of fatty acids in various matrices. J. Cromatogr. 2008, 1212, 106–113. [Google Scholar] [CrossRef]
- Nultch, W.; Agel, G. Fluence rate and wavelength dependence of photobleaching in the cyanobacterium Anabaena variabilis. Arch. Microbiol. 1986, 144, 268–271. [Google Scholar] [CrossRef]
- Kehoe, D.M.; Gutu, A. Responding to color: The regulation of complementary chromatic adaptation. Annu. Rev. Plant Biol. 2006, 57, 127–150. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peramuna, A.; Morton, R.; Summers, M.L. Enhancing Alkane Production in Cyanobacterial Lipid Droplets: A Model Platform for Industrially Relevant Compound Production. Life 2015, 5, 1111-1126. https://doi.org/10.3390/life5021111
Peramuna A, Morton R, Summers ML. Enhancing Alkane Production in Cyanobacterial Lipid Droplets: A Model Platform for Industrially Relevant Compound Production. Life. 2015; 5(2):1111-1126. https://doi.org/10.3390/life5021111
Chicago/Turabian StylePeramuna, Anantha, Ray Morton, and Michael L. Summers. 2015. "Enhancing Alkane Production in Cyanobacterial Lipid Droplets: A Model Platform for Industrially Relevant Compound Production" Life 5, no. 2: 1111-1126. https://doi.org/10.3390/life5021111
APA StylePeramuna, A., Morton, R., & Summers, M. L. (2015). Enhancing Alkane Production in Cyanobacterial Lipid Droplets: A Model Platform for Industrially Relevant Compound Production. Life, 5(2), 1111-1126. https://doi.org/10.3390/life5021111




