Higher NLR Values Can Predict Gram-Negative Spontaneous Bacterial Peritonitis and a High In-Hospital Mortality Rate in Patients with Spontaneous Bacterial Peritonitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Institutional Protocol Approval
2.3. Outcomes
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Patient Group
3.2. Predictive Role of NLR in the Type of SBP
3.3. Predictive Role of NLR for In-Hospital Mortality
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J. Hepatol. 2018, 69, 406–460, Erratum in J. Hepatol. 2018, 69, 1207. [Google Scholar] [CrossRef] [PubMed]
- Bunchorntavakul, C.; Chamroonkul, N.; Chavalitdhamrong, D. Bacterial infections in cirrhosis: A critical review and practical guidance. World J. Hepatol. 2016, 8, 307–321. [Google Scholar] [CrossRef]
- Ascione, T.; Di Flumeri, G.; Boccia, G.; De Caro, F. Infections in patients affected by liver cirrhosis: An update. Infez. Med. 2017, 25, 91–97. [Google Scholar] [PubMed]
- Piano, S.; Singh, V.; Caraceni, P.; Maiwall, R.; Alessandria, C.; Fernandez, J.; Soares, E.C.; Kim, D.J.; Kim, S.E.; Marino, M.; et al. Epidemiology and Effects of Bacterial Infections in Patients with Cirrhosis Worldwide. Gastroenterology 2019, 156, 1368–1380.e10. [Google Scholar] [CrossRef] [PubMed]
- Voicu, M.N.; Popescu, F.; Florescu, D.N.; Rogoveanu, I.; Turcu-Stiolica, A.; Gheonea, D.I.; Iovanescu, V.F.; Iordache, S.; Cazacu, S.M.; Ungureanu, B.S. Clostridioides difficile Infection among Cirrhotic Patients with Variceal Bleeding. Antibiotics 2021, 10, 731. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aithal, G.P.; Palaniyappan, N.; China, L.; Härmälä, S.; Macken, L.; Ryan, J.M.; Wilkes, E.A.; Moore, K.; Leithead, J.A.; Hayes, P.C.; et al. Guidelines on the management of ascites in cirrhosis. Gut 2021, 70, 9–29. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ekpanyapong, S.; Reddy, K.R. Infections in Cirrhosis. Curr. Treat. Options Gastroenterol. 2019, 17, 254–270. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Biggins, S.W.; Angeli, P.; Garcia-Tsao, G.; Ginès, P.; Ling, S.C.; Nadim, M.K.; Wong, F.; Kim, W.R. Diagnosis, Evaluation, and Management of Ascites, Spontaneous Bacterial Peritonitis and Hepatorenal Syndrome: 2021 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2021, 74, 1014–1048. [Google Scholar] [CrossRef] [PubMed]
- Căruntu, F.A.; Benea, L. Spontaneous bacterial peritonitis: Pathogenesis, diagnosis, treatment. J. Gastrointest. Liver Dis. 2006, 15, 51–56. [Google Scholar] [PubMed]
- Long, B.; Gottlieb, M. Emergency medicine updates: Spontaneous bacterial peritonitis. Am. J. Emerg. Med. 2023, 70, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Fiore, M.; Di Franco, S.; Alfieri, A.; Passavanti, M.B.; Pace, M.C.; Kelly, M.E.; Damiani, G.; Leone, S. Spontaneous bacterial peritonitis caused by Gram-negative bacteria: An update of epidemiology and antimicrobial treatments. Expert. Rev. Gastroenterol. Hepatol. 2019, 13, 683–692. [Google Scholar] [CrossRef]
- Shizuma, T. Spontaneous bacterial and fungal peritonitis in patients with liver cirrhosis: A literature review. World J. Hepatol. 2018, 10, 254–266. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fiore, M.; Maraolo, A.E.; Gentile, I.; Borgia, G.; Leone, S.; Sansone, P.; Passavanti, M.B.; Aurilio, C.; Pace, M.C. Current concepts and future strategies in the antimicrobial therapy of emerging Gram-positive spontaneous bacterial peritonitis. World J. Hepatol. 2017, 9, 1166–1175. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nookala, V.K.; Sharzehi, K.; Lee, T.P. An Increase of Gram-Positive Bacteria in Spontaneous Bacterial Peritonitis. Gastroenterology 2012, 142, S951. [Google Scholar] [CrossRef]
- Oliveira, J.C.; Carrera, E.; Petry, R.C.; Deutschendorf, C.; Mantovani, A.; Barcelos, S.T.A.; Cassales, S.; Schacher, F.C.; Lopes, A.B.; Alvares-da-Silva, M.R. High Prevalence of Multidrug-Resistant Bacteria in Cirrhotic Patients with Spontaneous Bacterial Peritonitis: Is It Time to Change the Standard Antimicrobial Approach? Can. J. Gastroenterol. Hepatol. 2019, 2019, 6963910. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cazacu, S.M.; Parscoveanu, M.; Cartu, D.; Moraru, E.; Rogoveanu, I.; Ungureanu, B.S.; Iordache, S.; Florescu, D.N.; Iovanescu, V.F.; Dragomir, M.I. NLR48 is Better Than CRP, and mCTSI, and Similar to BISAP and SOFA Scores for Mortality Prediction in Acute Pancreatitis: A Comparison of 6 Scores. J. Inflamm. Res. 2023, 16, 4793–4804. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Qiu, L.; Jin, X.; Wang, J.J.; Tang, X.D.; Fang, X.; Li, S.J.; Wang, F.; Chen, X.L. Plasma Neutrophil-to-Lymphocyte Ratio on the Third Day Postburn is Associated with 90-Day Mortality Among Patients with Burns Over 30% of Total Body Surface Area in Two Chinese Burns Centers. J. Inflamm. Res. 2021, 14, 519–526. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wu, H.; Cao, T.; Ji, T.; Luo, Y.; Huang, J.; Ma, K. Predictive value of the neutrophil-to-lymphocyte ratio in the prognosis and risk of death for adult sepsis patients: A meta-analysis. Front. Immunol. 2024, 15, 1336456. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kosidło, J.W.; Wolszczak-Biedrzycka, B.; Matowicka-Karna, J.; Dymicka-Piekarska, V.; Dorf, J. Clinical Significance and Diagnostic Utility of NLR, LMR, PLR and SII in the Course of COVID-19: A Literature Review. J. Inflamm. Res. 2023, 16, 539–562. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ulloque-Badaracco, J.R.; Ivan Salas-Tello, W.; Al-Kassab-Córdova, A.; Alarcón-Braga, E.A.; Benites-Zapata, V.A.; Maguiña, J.L.; Hernandez, A.V. Prognostic value of neutrophil-to-lymphocyte ratio in COVID-19 patients: A systematic review and meta-analysis. Int. J. Clin. Pract. 2021, 75, e14596. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turgunova, L.; Mekhantseva, I.; Akhmaltdinova, L.; Kostinov, M.; Zhumadilova, Z.; Turmukhambetova, A. Association of sTREM-1 and Neutrophil-to-Lymphocyte Ratio as Prognostic Markers in COVID-19 Short- and Medium-term Mortality. J. Inflamm. Res. 2023, 16, 5807–5817. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sarkar, S.; Khanna, P.; Singh, A.K. The Impact of Neutrophil-Lymphocyte Count Ratio in COVID-19: A Systematic Review and Meta-Analysis. J. Intensive Care Med. 2022, 37, 857–869. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, L.; Zhang, W.; Wang, J.; Jin, Q.; Ma, D.; Huang, R. Neutrophil-to-lymphocyte ratio predicts 30-, 90-, and 180-day readmissions of patients with hepatic encephalopathy. Front. Med. 2023, 10, 1185182. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hong, C.M.; Su, T.H.; Hsu, S.J.; Tseng, T.C.; Liu, C.H.; Yang, H.C.; Kao, J.H.; Chen, P.J.; Cheng, P.N.; Hung, C.H.; et al. Addition of neutrophil-to-lymphocyte ratio to Pre-DAA FIB-4 does not increase prediction value for de novo liver complications in hepatitis C. J. Formos. Med. Assoc. 2024; Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.H.; Zhou, L.F.; Wang, C.X.; Zha, Y.; Fan, C.; Zhong, B.Y.; Zhu, X.L.; Wang, W.D. The Value of Neutrophil-to-Lymphocyte Ratio in Predicting Mortality After Transjugular Intrahepatic Portosystemic Shunt Placement. J. Inflamm. Res. 2024, 17, 5211–5221. [Google Scholar] [CrossRef]
- Seyedi, S.A.; Nabipoorashrafi, S.A.; Hernandez, J.; Nguyen, A.; Lucke-Wold, B.; Nourigheimasi, S.; Khanzadeh, S. Neutrophil to Lymphocyte Ratio and Spontaneous Bacterial Peritonitis among Cirrhotic Patients: A Systematic Review and Meta-analysis. Can. J. Gastroenterol. Hepatol. 2022, 2022, 8604060. [Google Scholar] [CrossRef] [PubMed]
- Efgan, M.G.; Acar, H.; Kanter, E.; Kırık, S.; Duman Şahan, T. Role of Systemic Immune Inflammation Index, Systemic Immune Response Index, Neutrophil Lymphocyte Ratio and Platelet Lymphocyte Ratio in Predicting Peritoneal Culture Positivity and Prognosis in Cases of Spontaneous Bacterial Peritonitis Admitted to the Emergency Department. Medicina 2024, 60, 1335. [Google Scholar] [CrossRef]
- Baweja, A.; Jhamb, R.; Kumar, R.; Garg, S.; Gogoi, P. Clinical utility of Neutrophil Lymphocyte Ratio (NLR) as a marker of Spontaneous Bacterial Peritonitis (SBP) in patients with cirrhosis—An exploratory study. Int. J. Sci. Res. Arch. 2021, 3, 31–42. [Google Scholar] [CrossRef]
- Awad, S.; Ahmed, E.; Mohamed, E. Role of Combined Blood Neutrophil- Lymphocyte Ratio and C-reactive Protein in Diagnosis of Spontaneous Bacterial Peritonitis. Benha J. Appl. Sci. 2020, 5, 43–49. [Google Scholar] [CrossRef]
- Huynh, N.C.; Vo, T.D. Validation of a new simple scoring system to predict spontaneous bacterial peritonitis in patients with cirrhosis and ascites. BMC Gastroenterol. 2023, 23, 272. [Google Scholar] [CrossRef]
- Abdel-Razik, A.M.N.; Abdel-Aziz, M.; Elsherbiny, W.; Zakaria, S.; Shabana, W.; Abed, S.; Elhelaly, R.; Elzehery, R.; Eldars, W.; Elbendary, M. Mansoura simple scoring system for prediction of spontaneous bacterial peritonitis: Lesson learnt. Eur. J. Gastroenterol. Hepatol. 2019, 31, 1017–1024. [Google Scholar] [CrossRef]
- Abdo, G.; Nir, U.; Rawajdey, R.; Abu Dahoud, W.; Massalha, J.; Hajouj, T.; Assadi, M.H.; William, N. A Novel Score-Based Approach by Using Routine Laboratory Tests for Accurate Diagnosis of Spontaneous Bacterial Peritonitis (SBP) in Cirrhotic Patients. EJIFCC 2023, 34, 297–304. [Google Scholar]
- Mousa, N.; Besheer, T.; Abdel-Razik, A.; Hamed, M.; Deiab, A.G.; Sheta, T.; Eldars, W. Can combined blood neutrophil to lymphocyte ratio and C-reactive protein be used for diagnosis of spontaneous bacterial peritonitis? Br. J. Biomed. Sci. 2018, 75, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Yang, H.; Zeng, L.; Yang, W.; Guo, F.; Cui, W.; Chen, C.; Zhao, J.; Wu, S.; Yang, N.; et al. Calculated inflammatory markers derived from complete blood count results, along with routine laboratory and clinical data, predict treatment failure of acute peritonitis in chronic peritoneal dialysis patients. Ren. Fail. 2023, 45, 2179856. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jonathan, J.; Pradian, E.; Zulfariansyah, A. Correlation Between Neutrophil-lymphocyte Count Ratio and Procalcitonin in Sepsis and Septic Shock. Maj. Kedokt. Bdg. 2019, 51, 165–171. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.; Hua, Q.; Wu, J.; Wang, Y. Diagnostic Value of Systemic Inflammatory Response Index for Catheter-Related Bloodstream Infection in Patients Undergoing Haemodialysis. J. Immunol. Res. 2022, 2022, 7453354. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liang, P.; Yu, F. Predictive Value of Procalcitonin and Neutrophil-to-Lymphocyte Ratio Variations for Bloodstream Infection with Septic Shock. Med. Sci. Monit. 2022, 28, e935966. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, X.; Yuan, X.; Wang, C. The clinical value of IL-3, IL-4, IL-12p70, IL17A, IFN-γ, MIP-1β, NLR, P-selectin, and TNF-α in differentiating bloodstream infections caused by gram-negative, gram-positive bacteria and fungi in hospitalized patients: An Observational Study. Medicine 2019, 98, e17315. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Roldgaard, M.; Benfield, T.; Tingsgård, S. Blood neutrophil to lymphocyte ratio is associated with 90-day mortality and 60-day readmission in Gram negative bacteremia: A multi-center cohort study. BMC Infect. Dis. 2024, 24, 255. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tang, W.; Zhang, W.; Li, X.; Cheng, J.; Liu, Z.; Zhou, Q.; Guan, S. Hematological parameters in patients with bloodstream infection: A retrospective observational study. J. Infect. Dev. Ctries. 2020, 14, 1264–1273. [Google Scholar] [CrossRef] [PubMed]
- Cazacu, S.M.; Zlatian, O.M.; Plesea, E.L.; Vacariu, A.I.; Cimpoeru, M.; Rogoveanu, I.; Bigea, C.C.; Iordache, S. Predominant Gram-Positive Etiology May Be Associated with a Lower Mortality Rate but with Higher Antibiotic Resistance in Spontaneous Bacterial Peritonitis: A 7-Year Study in a Tertiary Center in Romania. Life 2025, 15, 855. [Google Scholar] [CrossRef]
- Tay, P.W.L.; Xiao, J.; Tan, D.J.H.; Ng, C.; Lye, Y.N.; Lim, W.H.; Teo, V.X.Y.; Heng, R.R.Y.; Yeow, M.W.X.; Lum, L.H.W.; et al. An Epidemiological Meta-Analysis on the Worldwide Prevalence, Resistance, and Outcomes of Spontaneous Bacterial Peritonitis in Cirrhosis. Front. Med. 2021, 8, 693652. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meyyur Aravamudan, V.; Khan, S.R.; Hussain, I. Clostridium difficile Infection in Liver Cirrhosis Carries a Higher Risk of Mortality: A Comprehensive Literature Review. Cureus 2019, 11, e5463. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Mattos, A.A.; Costabeber, A.M.; Lionço, L.C.; Tovo, C.V. Multi-resistant bacteria in spontaneous bacterial peritonitis: A new step in management? World J. Gastroenterol. 2014, 20, 14079–14086. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Piroth, L.; Pechinot, A.; Di Martino, V.; Hansmann, Y.; Putot, A.; Patry, I.; Hadou, T.; Jaulhac, B.; Chirouze, C.; Rabaud, C.; et al. Evolving epidemiology and antimicrobial resistance in spontaneous bacterial peritonitis: A two-year observational study. BMC Infect. Dis. 2014, 14, 287. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pimentel, R.; Leitão, J.; Gregório, C.; Santos, L.; Carvalho, A.; Figueiredo, P. Spontaneous Bacterial Peritonitis in Cirrhotic Patients: A Shift in the Microbial Pattern? A Retrospective Analysis. GE-Port. J. Gastroenterol. 2021, 29, 256–266. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ratnasekera, I.U.; Johnson, A.; Powell, E.E.; Henderson, A.; Irvine, K.M.; Valery, P.C. Epidemiology of ascites fluid infections in patients with cirrhosis in Queensland, Australia from 2008 to 2017: A population-based study. Medicine 2022, 101, e29217. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alexopoulou, A.; Vasilieva, L.; Agiasotelli, D.; Siranidi, K.; Pouriki, S.; Tsiriga, A.; Toutouza, M.; Dourakis, S.P. Extensively drug-resistant bacteria are an independent predictive factor of mortality in 130 patients with spontaneous bacterial peritonitis or spontaneous bacteremia. World J. Gastroenterol. 2016, 22, 4049–4056. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Khoury, A.; Rattanasuwan, T.; Ebied, A.M. Shifting microorganism incidence in cirrhotic patients with ascites: A 5-year retrospective cross-sectional analysis. Dig. Med. Res. 2020, 3, 45. [Google Scholar] [CrossRef]
- Friedrich, K.; Nüssle, S.; Rehlen, T.; Stremmel, W.; Mischnik, A.; Eisenbach, C. Microbiology and resistance in first episodes of spontaneous bacterial peritonitis: Implications for management and prognosis. J. Gastroenterol. Hepatol. 2016, 31, 1191–1195. [Google Scholar] [CrossRef] [PubMed]
- Santoiemma, P.P.; Dakwar, O.; Angarone, M.P. A retrospective analysis of cases of Spontaneous Bacterial Peritonitis in cirrhosis patients. PLoS ONE 2020, 15, e0239470. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quickert, S.; Würstle, S.; Reuken, P.A.; Hagel, S.; Schneider, J.; Schmid, R.M.; Neugebauer, S.; Stallmach, A.; Bruns, T. Real-World Effectiveness of Piperacillin/Tazobactam with and without Linezolid for Spontaneous Bacterial Peritonitis. Dig. Dis. 2022, 40, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Wu, D.; Wei, L.; Liu, S.; Zhao, P.; Tu, B.; Xie, Y.; Liu, Y.; Wang, X.; Liu, L.; et al. Nosocomial and Community-Acquired Spontaneous Bacterial Peritonitis in patients with liver cirrhosis in China: Comparative Microbiology and Therapeutic Implications. Sci. Rep. 2017, 7, 46025, Erratum in Sci Rep. 2017, 7, 46868. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bert, F.; Andreu, M.; Durand, F.; Degos, F.; Galdbart, J.O.; Moreau, R.; Branger, C.; Lambert-Zechovsky, N.; Valla, D. Nosocomial and community-acquired spontaneous bacterial peritonitis: Comparative microbiology and therapeutic implications. Eur. J. Clin. Microbiol. Infect. Dis. 2003, 22, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Ning, N.Z.; Li, T.; Zhang, J.L.; Qu, F.; Huang, J.; Liu, X.; Li, Z.; Geng, W.; Fu, J.L.; Huan, W.; et al. Clinical and bacteriological features and prognosis of ascitic fluid infection in Chinese patients with cirrhosis. BMC Infect. Dis. 2018, 18, 253. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ding, X.; Yu, Y.; Chen, M.; Wang, C.; Kang, Y.; Lou, J. Causative agents and outcome of spontaneous bacterial peritonitis in cirrhotic patients: Community-acquired versus nosocomial infections. BMC Infect. Dis. 2019, 19, 463. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Popoiag, R.E.; Panaitescu, E.; Suceveanu, A.I.; Suceveanu, A.P.; Micu, S.I.; Mazilu, L.; Parepa, I.; Voinea, F.; Costea, D.O.; Enache, F.; et al. Spontaneous bacterial peritonitis mortality trends of cirrhotic patients in the last decade in Constanta County. Exp. Ther. Med. 2021, 22, 732. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Al-Ghamdi, H.; Al-Harbi, N.; Mokhtar, H.; Daffallah, M.; Memon, Y.; Aljumah, A.A.; Sanai, F.M. Changes in the patterns and microbiology of spontaneous bacterial peritonitis: Analysis of 200 cirrhotic patients. Acta Gastroenterol. Belg. 2019, 82, 261–266. [Google Scholar] [PubMed]
- Cazacu, S.M.; Alexandru, D.O.; Popescu, A.V.; Popa, P.; Rogoveanu, I.; Iovanescu, V.F. Is There a Role for the Neutrophil-to-Lymphocyte Ratio for Rebleeding and Mortality Risk Prediction in Acute Variceal Bleeding? A Comparative 5-Year Retrospective Study. Diseases 2025, 13, 265. [Google Scholar] [CrossRef]
- Metawea, M.I.; Moteleub, H.N.A.E. Diagnostic role of simple indices in HCV-related liver cirrhosis outcomes: A prospective cross-sectional study. Clin. Exp. Hepatol. 2022, 8, 29–35. [Google Scholar] [CrossRef]
- Wang, X.; Feng, H.; Hui, Y.; Yu, Z.; Zhao, T.; Mao, L.; Lin, L.; Wang, B.; Fan, X.; Yu, Q.; et al. Neutrophil-to-lymphocyte ratio is associated with malnutrition risk estimated by the Royal Free Hospital-Nutritional Prioritizing Tool in hospitalized cirrhosis. JPEN J. Parenter. Enter. Nutr. 2022, 46, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Chen, H.; Chen, Z.; Tan, J.; Wu, F.; Li, X. Prognostic value of neutrophil-to-lymphocyte ratio in patients with hepatocellular carcinoma receiving curative therapies: A systematic review and meta-analysis. BMC Cancer 2025, 25, 571. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Li, J.; Deng, H.; Li, H.; Su, C.; Guo, X. Neutrophil-to-lymphocyte ratio for the prognostic assessment of hepatocellular carcinoma: A systematic review and meta-analysis of observational studies. Oncotarget 2016, 7, 45283–45301. [Google Scholar] [CrossRef] [PubMed]
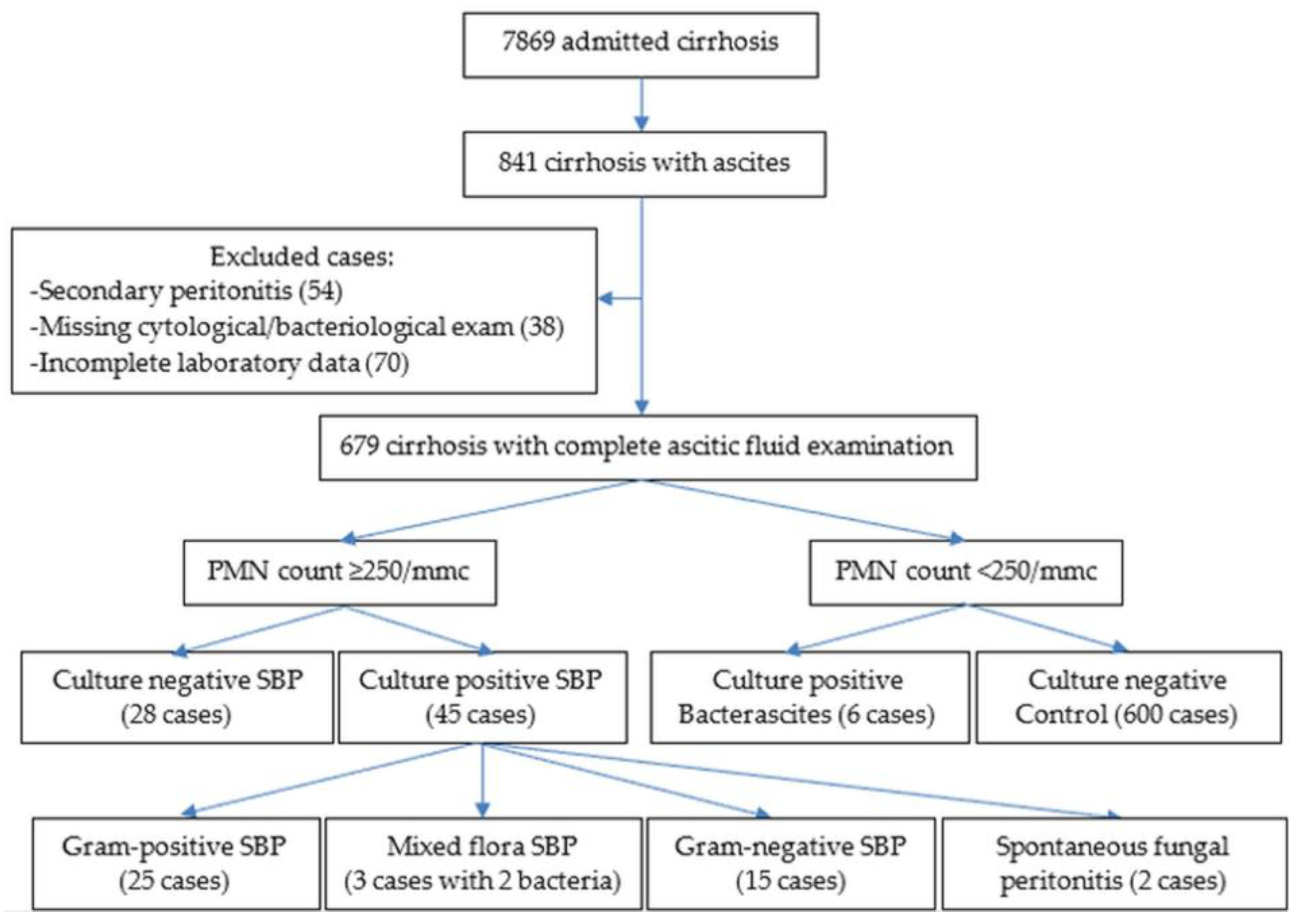
| SBP | Ascitic PMN count ≥ 250/mm3 |
| Culture-positive SBP | Ascitic PMN count ≥ 250/mm3 + a positive ascitic culture |
| Culture-negative SBP | Ascitic PMN count ≥ 250/mm3 + a negative ascitic culture |
| Bacterascitis | Ascitic PMN count < 250/mm3 + a positive ascitic culture |
| Control Group | Ascitic PMN count < 250/mm3 + a negative ascitic culture |
| Culture-Positive SBP (N = 45) | Culture-Negative SBP (N = 28) | Control (N = 600) | p-Value | |
|---|---|---|---|---|
| Age (years); mean± std dev (range) | 60.1 ± 12.1 | 59.4 ± 11.4 | 59.2 ± 11.3 | 0.883 |
| Gender Males/Females (% Males) | 29/16 (64.4) | 22/6 (78.6) | 392/208 (65.3) | 0.346 |
| Cirrhosis etiology (%) | ||||
| Alcoholic | 36 (80) | 15 (53.6) | 474 (79.0) | 0.017 |
| Hepatitis B or/and C | 6 (13.3) | 10 (35.7) | 69 11.5) | |
| Mixed (viral + alcoholic) | 3 (6.7) | 3 (10.7) | 53 (8.8) | |
| Other (autoimmune, biliary) | 0 (0) | 0 (0) | 4 (0.7) | |
| Systolic blood pressure (mean ± stdev) | 117.9 ± 18.7 | 115.0 ± 22.5 | 121.9 ± 20.8 | 0.272 |
| Pulse (count/min, mean ± stdev) | 89.7 ± 18.8 | 91.6 ± 19.4 | 87.5 ± 17.9 | 0.343 |
| Laboratory analyses (mean ± stdev) | ||||
| Hemoglobin (g/dL) | 9.98 ± 2.29 | 10.56 ± 2.25 | 10.39 ± 2.37 | 0.555 |
| Leucocyte (count/mm3) | 10,991 ± 6611 | 11,190 ± 5378 | 8713 ± 5182 | 0.000 |
| Neutrophil (count/mm3) | 8371 ± 6138 | 8082 ± 3960 | 6065 ± 4470 | <0.0001 |
| Lymphocyte (count/mm3) | 1416 ± 818 | 1861 ± 2487 | 1564 ± 989 | 0.670 |
| Platelet count (×1000/mm3) | 172,563 ± 116,111 | 158,860 ± 103,637 | 149,639 ± 82,947 | 0.619 |
| Urea (mg/dL) | 56.1 ± 43.9 | 74.0 ± 55.5 | 53.0 ± 45.8 | 0.036 |
| Creatinine (mg/dL) | 1.43 ± 2.73 | 1.61 ± 1.75 | 1.15 ± 1.31 | 0.050 |
| INR | 1.763 ± 0.503 | 1.672 ± 0.357 | 1.643 ± 0.639 | 0.062 |
| Albumin (g/dL) | 2.46 ± 0.60 | 2.41 ± 0.46 | 2.63 ± 0.56 | 0.033 |
| Total bilirubin (mg/dL) | 6.16 ± 6.16 | 7.03 ± 8.46 | 4.01 ± 4.69 | 0.018 |
| ALT (UI/dL) | 49.9 ± 95.4 | 38.8 ± 30.4 | 36.0 ± 34.7 | 0.341 |
| AST (UI/dL) | 119.8 ± 156.1 | 90.4 ± 80.7 | 85.0 ± 74.0 | 0.297 |
| Na (mmol/L) | 131.2 ± 6.3 | 131.7 ± 5.7 | 132.0 ± 5.9 | 0.598 |
| K (mmol/L) | 4.57 ± 0.88 | 4.38 ± 0.77 | 4.36 ± 0.80 | 0.233 |
| ESR (mm) | 57 ± 35 | 51 ± 27 | 51 ± 30 | 0.590 |
| Blood culture positive (%) | 1 (2.2) | 1 (3.6) | 15 (2.5) | 0.931 |
| Previous antibiotherapy (%) | 21 (46.7) | 12 (42.9) | 256 (42.7) | 0.872 |
| Child class (%) A | 1 (2.2) | 0 (0) | 23 (3.8) | 0.562 |
| B | 17 (37.8) | 10 (35.7) | 260 (43.3) | |
| C | 27 (60) | 18 (64.3) | 317 (52.8) | |
| Child/MELD score (Mean ± Stdev) | ||||
| CTP | 9.867 ± 1.804 | 10.179 ± 1.701 | 9.703 ± 2.020 | 0.302 |
| CTP-creatinine | 10.578 ± 2.360 | 11.393 ± 2.558 | 10.377 ± 2.298 | 0.109 |
| MELD-3 | 39.556 ± 23.812 | 35.857 ± 17.264 | 29.110 ± 12.378 | 0.000 |
| MELD-Na | 39.044 ± 19.862 | 36.214 ± 16.008 | 29.997 ± 11.400 | 0.000 |
| Complications (%) | ||||
| Encephalopathy | 14 (31.1) | 8 (28.6) | 198 (33.0) | 0.864 |
| Acute kidney injury | ||||
| Comorbidities (%) | ||||
| Hepatocellular carcinoma | 7 (15.6) | 3 (10.7) | 40 (6.7) | 0.072 |
| Portal vein thrombosis | 7 (15.6) | 4 (14.3) | 36 (6.0) | 0.016 |
| Acute variceal bleeding | 9 (20) | 4 (14.3) | 72 (12.0) | 0.286 |
| Pneumonia | 7 (9.7) | 3 (10.7) | 58 (15.6) | 0.447 |
| Clostridium difficile colitis | 3 (10.7) | 3 (10.7) | 10 (1.7) | 0.001 |
| Cardiovascular | 4 (19.0) | 5 (17.9) | 114 (8.9) | 0.238 |
| Diabetes | 5 (14.0) | 4 (14.3) | 84 11.1) | 0.861 |
| Previous chronic kidney disease | 5 (8.5) | 3 10.7) | 51 (11.1) | 0.781 |
| Mortality % in-hospital | 26.7 | 21.4 | 6.0 | <0.0001 |
| 30-day | 44.4 | 43.5 | 13.3 | <0.0001 |
| 90-day | 46.7 | 54.5 | 21.2 | <0.0001 |
| 1-year | 73.0 | 76.2 | 37.3 | <0.0001 |
| Non-Nosocomial Infection (%) | Nosocomial Infection (%) | |
|---|---|---|
| G-positive | 16 (59.3) | 17 (60.7) |
| Staphylococcus aureus | 11 (40.7) | 12 (41.8) |
| Streptococcus spp. | 2 (7.4) | 1 (3.6) |
| Enterococcus spp. | 2 (7.4) | 4 (14.3) |
| Staphylococcus coagulase-negative | 1 (3.7) | 0 (0) |
| G-negative | 9 (33.3) | 11 (39.3) |
| E. coli | 3 (11.1) | 3 (10.7) |
| Klebsiella spp. | 2 (7.4) | 3 (10.7) |
| Acinetobacter baumanii | 1 (3.7) | 3 (10.7) |
| Pseudomonas aeruginosa | 1 (3.7) | 1 (3.6) |
| Citrobacter | 1 (3.7) | 0 (0) |
| Proteus mirabilis | 1 (3.7) | 0 (0) |
| Enterobacter spp. | 0 (0) | 1 (3.6) |
| Candida | 2 (7.4) | 0 (0) |
| TOTAL | 27 | 28 |
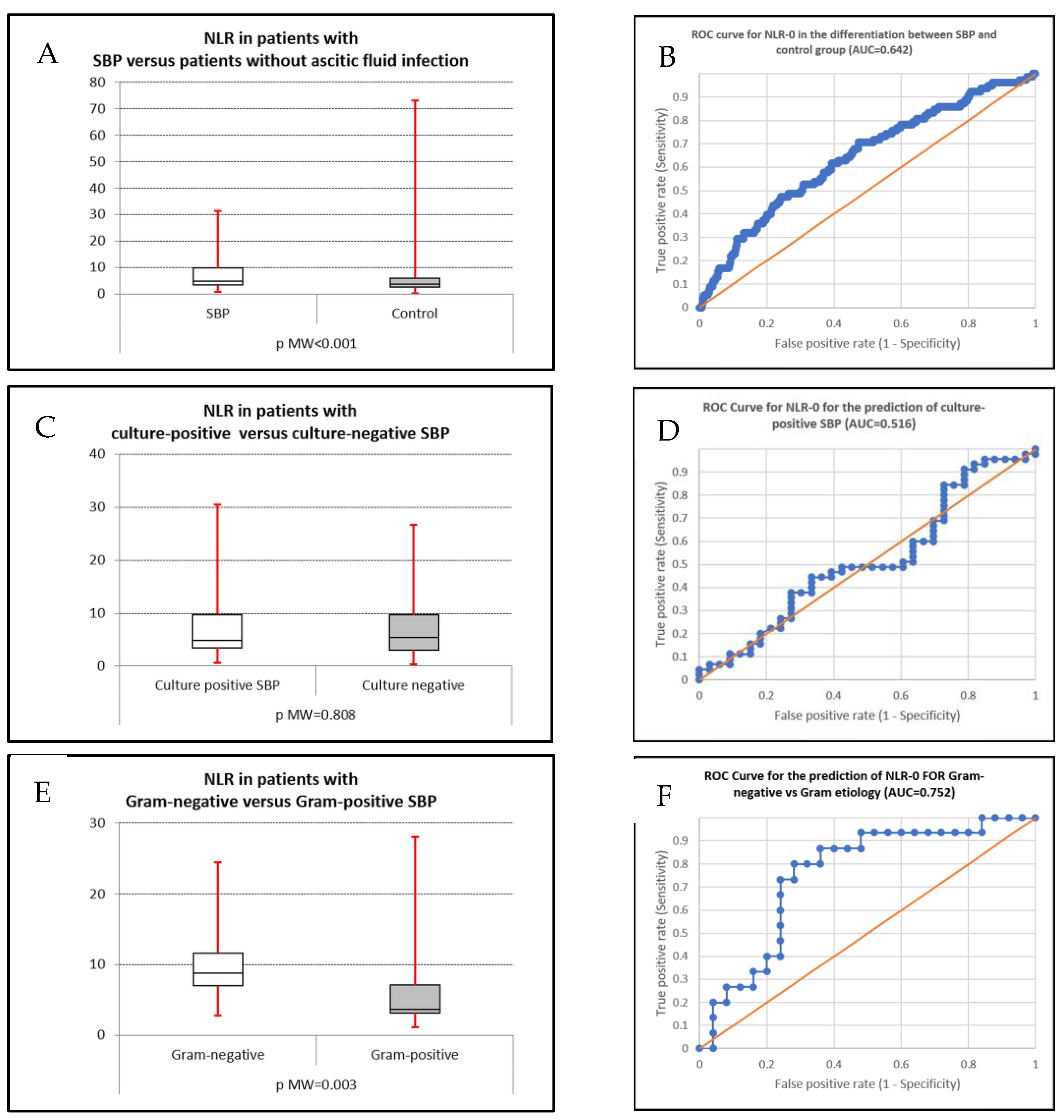
| Patient Group | Median (IQR) | AUC | p-Value | Cutoff | Sensitivity | Specificity |
|---|---|---|---|---|---|---|
| All SBP | 5.30 (3.35–9.81) | 0.642 | <0.001 | 3.817 | 70.5 | 52.8 |
| Control | 3.66 (2.49–5.94) | |||||
| Culture-positive SBP | 4.75 (3.36–9.77) | 0.516 | 0.808 | 2.292 | 91.1 | 21.2 |
| Culture-negative SBP | 5.33 (2.96–9.73) | |||||
| Gram-negative SBP | 8.79 (7.01–11.61) | 0.752 | 0.003 | 6.371 | 80 | 72 |
| Gram-positive SBP | 3.92 (3.20–7.07) | |||||
| Gram-negative SBP | 8.79 (7.01–11.61) | 0.814 | <0.001 | 6.741 | 80 | 79.9 |
| Control | 3.66 (2.49–5.94) |
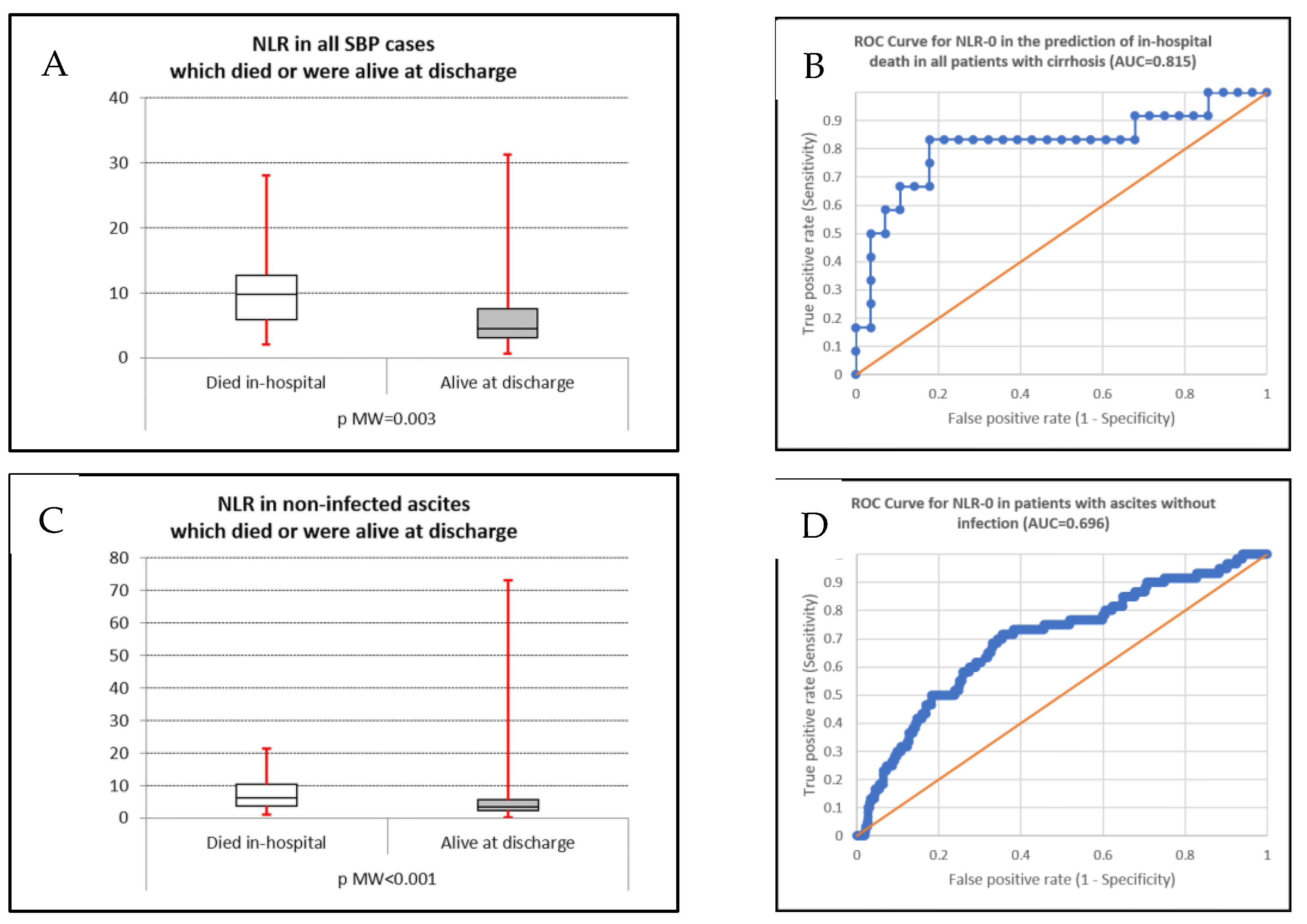
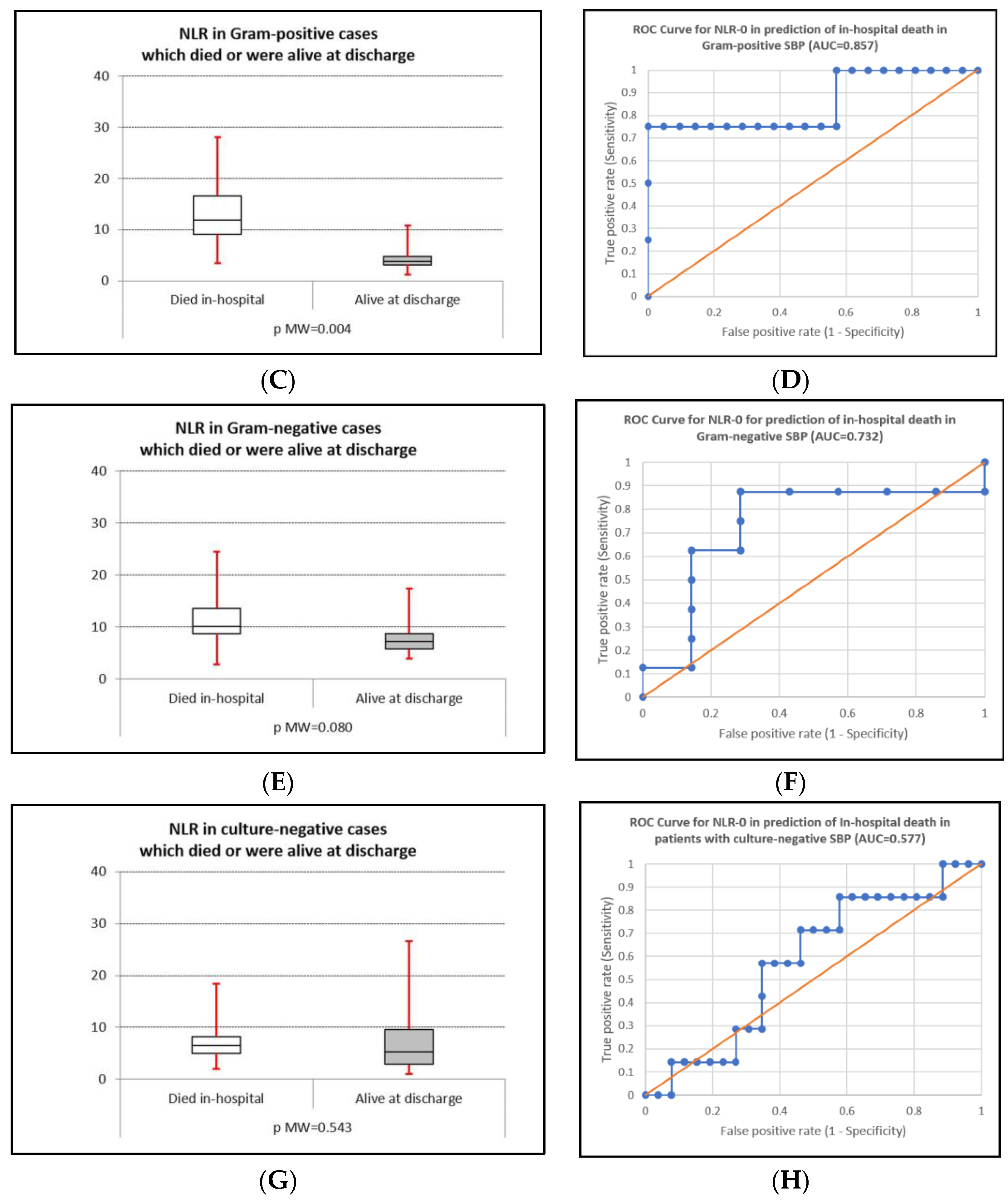
| Patient Group | Median (IQR) | AUC | p-Value | Cutoff | Sensitivity | Specificity | |
|---|---|---|---|---|---|---|---|
| All ascites | Dead | 7.09 (4.42–11.23) | 0.815 | <0.001 | 7.750 | 83.3 | 82.1 |
| Alive | 3.63 (2.45–5.80) | ||||||
| All SBP | Dead | 9.73 (5.92–12.75) | 0.717 | 0.003 | 6.371 | 73.7 | 67.8 |
| Alive | 4.54 (3.09–7.61) | ||||||
| Control | Dead | 6.22 (3.71–10.30) | 0.696 | <0.001 | 4.524 | 71.7 | 64.5 |
| Alive | 3.56 (2.37–5.59) | ||||||
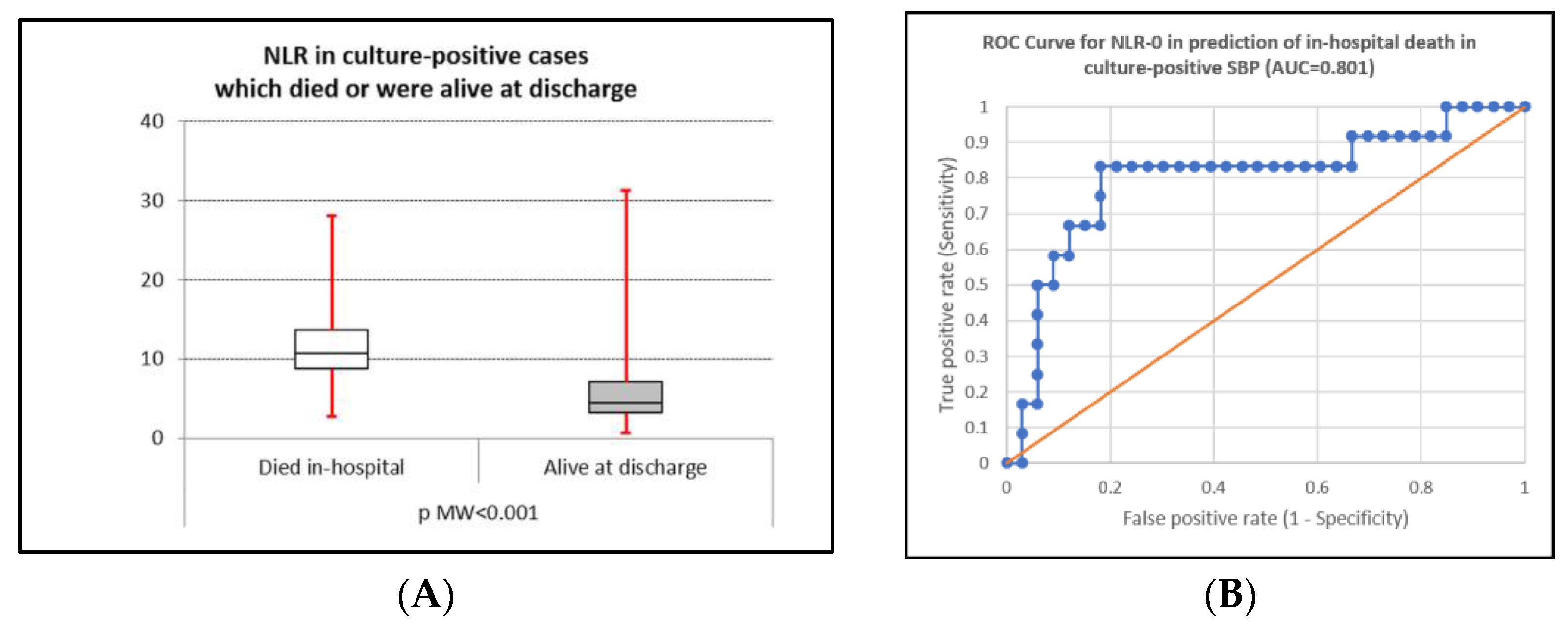
| Culture+ SBP | Dead | 10.70 (8.75–13.6) | 0.818 | <0.001 | 7.750 | 81.8 | 80.1 |
| Alive | 4.47 (3.20–7.07) | ||||||
| Culture− SBP | Dead | 6.50 (5.00–8.13) | 0.577 | 0.543 | 4.297 | 85.7 | 42.3 |
| Alive | 5.26 (2.90–9.65) | ||||||
| Gram+ SBP | Dead | 11.85 (9.05–16.6) | 0.857 | 0.004 | 10.821 | 75.0 | 100 |
| Alive | 3.82 (3.16–4.74) | ||||||
| Gram− SBP | Dead | 10.13 (8.75–13.6) | 0.732 | 0.080 | 7.750 | 87.5 | 71.4 |
| Alive | 7.22 (5.72–8.69) | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cazacu, S.M.; Zlatian, O.M.; Alexandru, D.O.; Plesea, E.L.; Vacariu, I.A.; Cimpoeru, M.; Rogoveanu, I.; Bigea, C.C.; Marginean, C.M.; Iordache, S. Higher NLR Values Can Predict Gram-Negative Spontaneous Bacterial Peritonitis and a High In-Hospital Mortality Rate in Patients with Spontaneous Bacterial Peritonitis. Life 2025, 15, 1363. https://doi.org/10.3390/life15091363
Cazacu SM, Zlatian OM, Alexandru DO, Plesea EL, Vacariu IA, Cimpoeru M, Rogoveanu I, Bigea CC, Marginean CM, Iordache S. Higher NLR Values Can Predict Gram-Negative Spontaneous Bacterial Peritonitis and a High In-Hospital Mortality Rate in Patients with Spontaneous Bacterial Peritonitis. Life. 2025; 15(9):1363. https://doi.org/10.3390/life15091363
Chicago/Turabian StyleCazacu, Sergiu Marian, Ovidiu Mircea Zlatian, Dragos Ovidiu Alexandru, Elena Leocadia Plesea, Ioan Alexandru Vacariu, Mihai Cimpoeru, Ion Rogoveanu, Camelia Cristiana Bigea, Cristina Maria Marginean, and Sevastita Iordache. 2025. "Higher NLR Values Can Predict Gram-Negative Spontaneous Bacterial Peritonitis and a High In-Hospital Mortality Rate in Patients with Spontaneous Bacterial Peritonitis" Life 15, no. 9: 1363. https://doi.org/10.3390/life15091363
APA StyleCazacu, S. M., Zlatian, O. M., Alexandru, D. O., Plesea, E. L., Vacariu, I. A., Cimpoeru, M., Rogoveanu, I., Bigea, C. C., Marginean, C. M., & Iordache, S. (2025). Higher NLR Values Can Predict Gram-Negative Spontaneous Bacterial Peritonitis and a High In-Hospital Mortality Rate in Patients with Spontaneous Bacterial Peritonitis. Life, 15(9), 1363. https://doi.org/10.3390/life15091363






