Influence of Extraction Techniques and Solvents on the Antioxidant and Biological Potential of Different Parts of Scorzonera undulata
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Polyphenolic Extract Preparation
2.2.1. Maceration
2.2.2. Ultrasound
2.3. Total Phenolic Content (TPC)
2.4. Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
Derivatization Method
2.5. Liquid Chromatography-Electrospray Ionization-Tandem Mass Spectrometry (LC-ESI–MS) Analysis
2.6. Antioxidant Activity (DPPH Radical Scavenging Activity)
2.7. Biological Activities (Antienzymatic Activities)
2.7.1. Anti-Xanthine Oxidase Activity
2.7.2. Anti-5-Lipoxygenase Activity
2.7.3. Anti-α-Glucosidase Activity
2.7.4. Anti-α-Amylase Activity
2.8. Statistical Anylasis
3. Results and Discussion
3.1. Extraction Yields
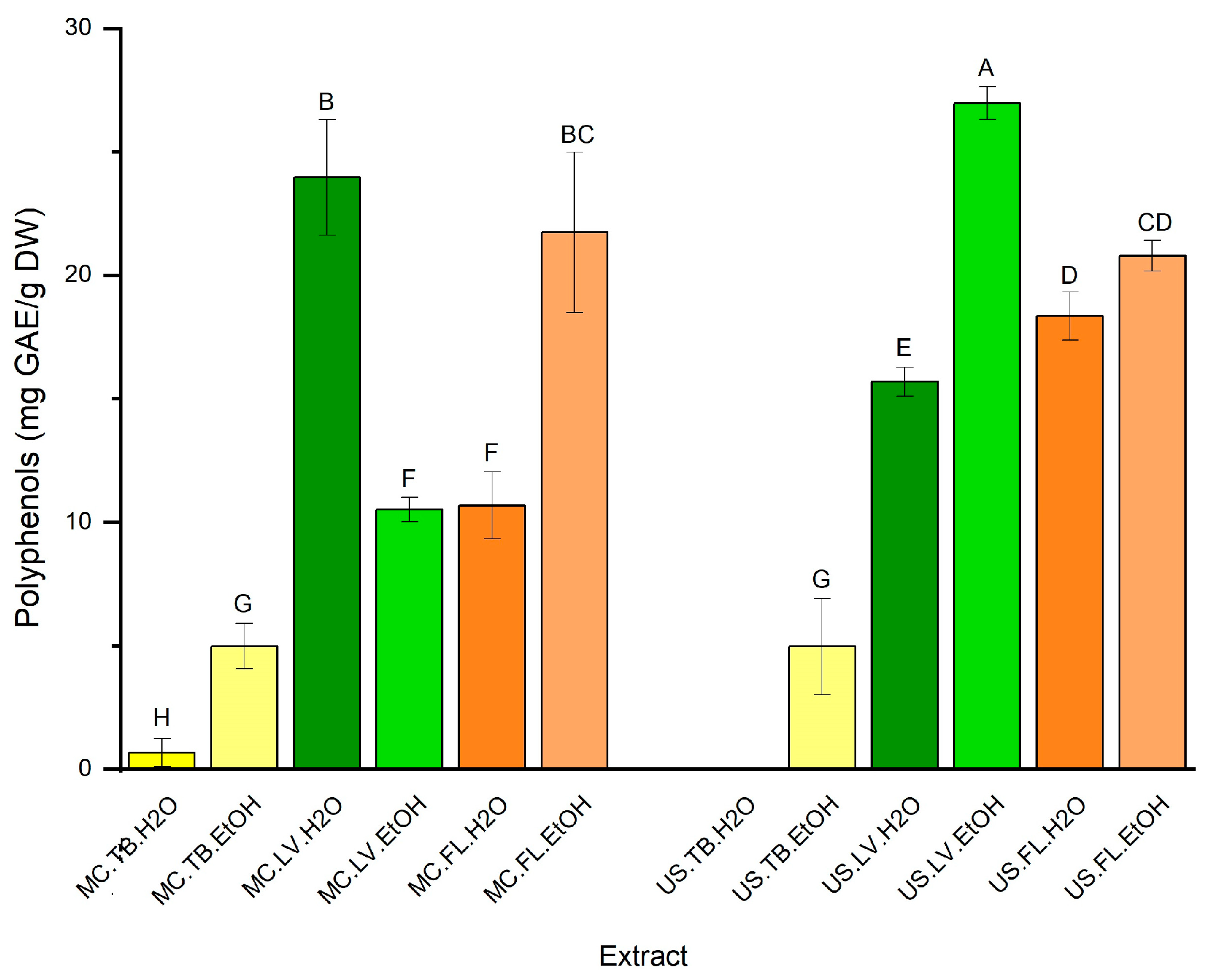
3.2. Polyphenol Content (TPC)
3.3. Identification of Volatile Compounds by GC-MS Analysis
3.4. Identification of Phenolic Compounds by LC-MS Analysis
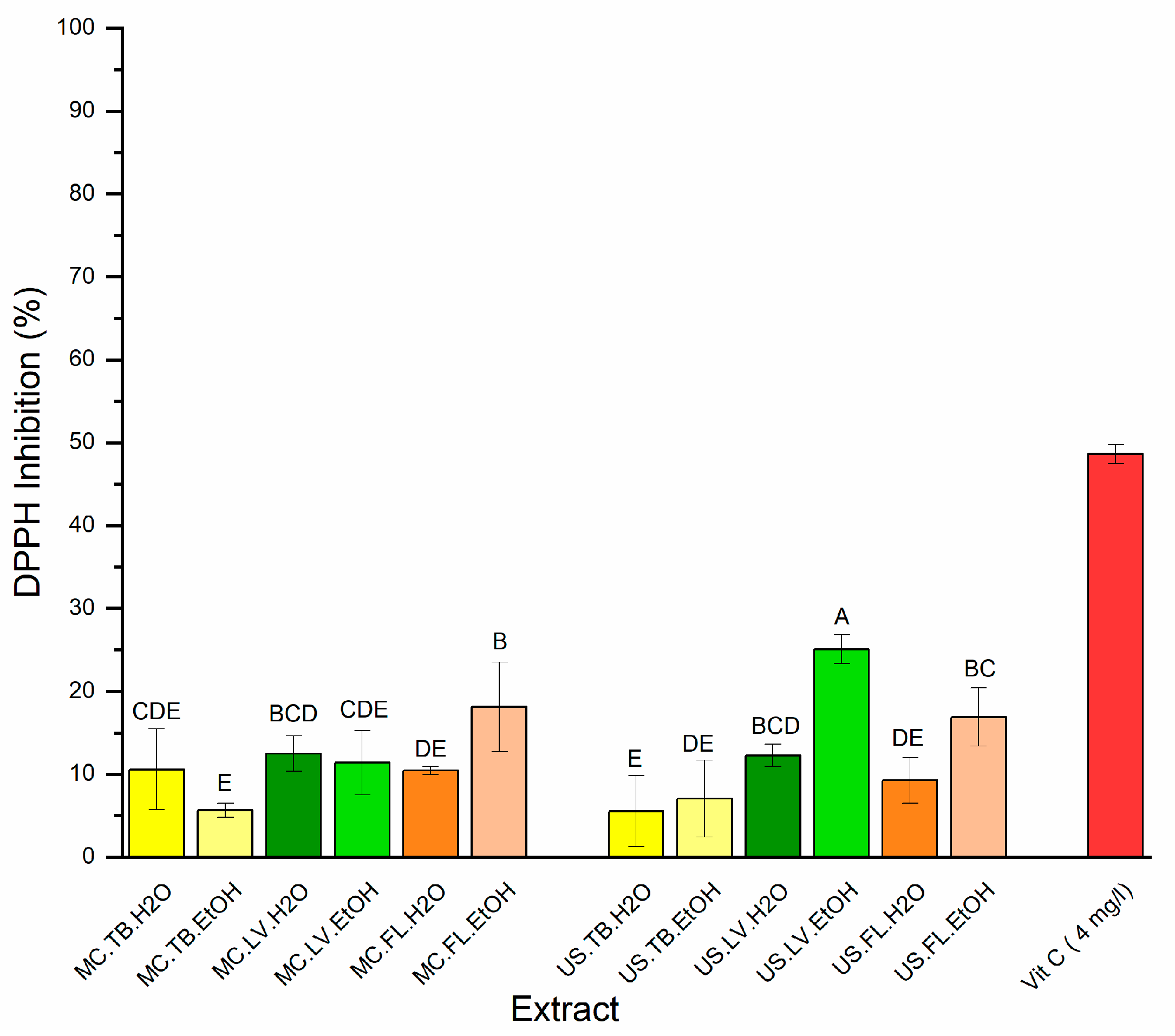
3.5. Antioxidant Activity
3.6. Biological Activities (Antienzymatic Activities)
3.7. Statistical Anylasis
3.7.1. Correlation Coefficient Analysis
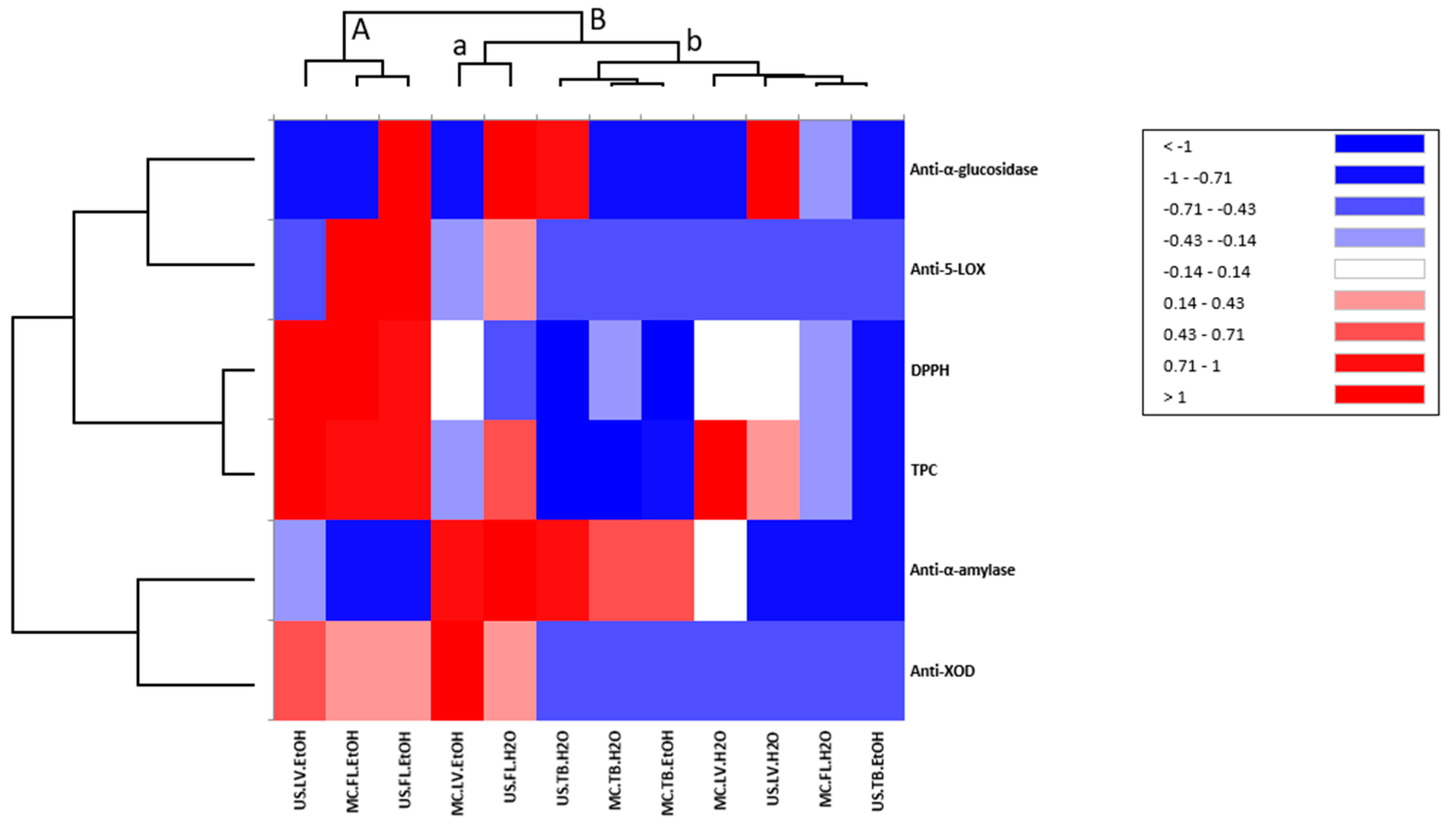
3.7.2. Heat Map Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lendzion, K.; Gornowicz, A.; Bielawski, K.; Bielawska, A. Phytochemical Composition and Biological Activities of Scorzonera Species. Int. J. Mol. Sci. 2021, 22, 5128. [Google Scholar] [CrossRef] [PubMed]
- Zaika, M.A.; Kilian, N.; Jones, K.; Krinitsina, A.A.; Nilova, M.V.; Speranskaya, A.S.; Sukhorukov, A.P. Scorzonera Sensu Lato (Asteraceae, Cichorieae)–Taxonomic Reassessment in the Light of New Molecular Phylogenetic and Carpological Analyses. PhytoKeys 2020, 137, 1. [Google Scholar] [CrossRef] [PubMed]
- Petkova, N. Characterization of Inulin from Black Salsify (Scorzonera hispanica L.) for Food and Pharmaceutical Purposes. Asian J. Pharm. Clin. Res. 2018, 11, 221–225. [Google Scholar] [CrossRef]
- Zhu, Y.; Wu, Q.-X.; Hu, P.-Z.; Wu, W.-S. Biguaiascorzolides A and B: Two Novel Dimeric Guaianolides with a Rare Skeleton, from Scorzonera Austriaca. Food Chem. 2009, 114, 1316–1320. [Google Scholar] [CrossRef]
- Yaylı, N.; Öksüz, E.; Korkmaz, B.; Erik, I.; Fandaklı, S.; Faiz, Ö.; Coşkunçelebi, K. Volatile and Phenolic Contents, Antimicrobial and Tyrosinase Activities of Two Endemic Species Scorzonera Pisidica and Scorzonera Sandrasica L. Grown in Turkey. Rec. Nat. Prod. 2022, 16, 46–57. [Google Scholar] [CrossRef]
- Bellassouad, K.; Feki, A.E.; Ayadi, H. Effect of Extraction Solvents on the Biomolecules and Antioxidant Properties of Scorzonera Undulata (Asteraceae): Application of Factorial Design Optimization Phenolic Extraction. Acta Sci. Pol. Technol. Aliment. 2015, 14, 313–330. [Google Scholar]
- Abdelkader, H.B.; Salah, K.B.H.; Liouane, K.; Boussaada, O.; Gafsi, K.; Mahjoub, M.A.; Aouni, M.; Hellal, A.N.; Mighri, Z. Antimicrobial Activity of Rhaponticum Acaule and Scorzonera Undulata Growing Wild in Tunisia. Afr. J. Microbiol. Res. 2010, 4, 1954–1958. [Google Scholar]
- Oreopoulou, A.; Tsimogiannis, D.; Oreopoulou, V. Extraction of Polyphenols from Aromatic and Medicinal Plants: An Overview of the Methods and the Effect of Extraction Parameters. In Polyphenols in Plants; Academic Press: Cambridge, MA, USA, 2019; pp. 243–259. [Google Scholar]
- Handa, S.S.; Khanuja, S.P.S.; Longo, G.; Rakesh, D.D. Extraction Technologies for Medicinal and Aromatic Plants; No. 66; United Nations Industrial Development Organization and the International Centre for Science and High Technology: Milan, Italy, 2008; pp. 21–25. [Google Scholar]
- Dobros, N.; Zawada, K.D.; Paradowska, K. Phytochemical Profiling, Antioxidant and Anti-Inflammatory Activity of Plants Belonging to the Lavandula Genus. Molecules 2023, 28, 256. [Google Scholar] [CrossRef]
- Esclapez, M.D.; García-Pérez, J.V.; Mulet, A.; Cárcel, J.A. Ultrasound-Assisted Extraction of Natural Products. Food Eng. Rev. 2011, 3, 108–120. [Google Scholar] [CrossRef]
- Talmaciu, A.I.; Volf, I.; Popa, V.I. A Comparative Analysis of the ‘Green’ Techniques Applied for Polyphenols Extraction from Bioresources. Chem. Biodivers. 2015, 12, 1635–1651. [Google Scholar] [CrossRef]
- Gharby, S.; Oubannin, S.; Ait Bouzid, H.; Bijla, L.; Ibourki, M.; Gagour, J.; Koubachi, J.; Sakar, E.H.; Majourhat, K.; Lee, L.-H. An Overview on the Use of Extracts from Medicinal and Aromatic Plants to Improve Nutritional Value and Oxidative Stability of Vegetable Oils. Foods 2022, 11, 3258. [Google Scholar] [CrossRef]
- Sarı, A.; Şahin, H.; Özsoy, N.; Çelik, B.Ö. Phenolic Compounds and in Vitro Antioxidant, Anti-Inflammatory, Antimicrobial Activities of Scorzonera Hieraciifolia Hayek Roots. S. Afr. J. Bot. 2019, 125, 116–119. [Google Scholar] [CrossRef]
- Llorent-Martínez, E.J.; Zengin, G.; Sinan, K.I.; Polat, R.; Canlı, D.; Picot-Allain, M.C.N.; Mahomoodally, M.F. Impact of Different Extraction Solvents and Techniques on the Biological Activities of Cirsium Yildizianum (Asteraceae: Cynareae). Ind. Crops Prod. 2020, 144, 112033. [Google Scholar] [CrossRef]
- Ben Khadher, T.; Aydi, S.; Mars, M.; Bouajila, J. Study on the Chemical Composition and the Biological Activities of Vitis Vinifera Stem Extracts. Molecules 2022, 27, 3109. [Google Scholar] [CrossRef]
- Rahmani, R.; Bouajila, J.; Jouaidi, M.; Debouba, M. African Mustard (Brassica tournefortii) as Source of Nutrients and Nutraceuticals Properties. J. Food Sci. 2020, 85, 1856–1871. [Google Scholar] [CrossRef]
- Dawra, M.; El Rayess, Y.; El Beyrouthy, M.; Nehme, N.; El Hage, R.; Taillandier, P.; Bouajila, J. Biological Activities and Chemical Characterization of the Lebanese Endemic Plant Origanum Ehrenbergii Boiss. Flavour Fragr. J. 2021, 36, 339–351. [Google Scholar] [CrossRef]
- Znati, M.; Ben Jannet, H.; Cazaux, S.; Souchard, J.P.; Harzallah Skhiri, F.; Bouajila, J. Antioxidant, 5-Lipoxygenase Inhibitory and Cytotoxic Activities of Compounds Isolated from the Ferula Lutea Flowers. Molecules 2014, 19, 16959–16975. [Google Scholar] [CrossRef]
- Kim, K.Y.; Nam, K.A.; Kurihara, H.; Kim, S.M. Potent α-Glucosidase Inhibitors Purified from the Red Alga Grateloupia Elliptica. Phytochemistry 2008, 69, 2820–2825. [Google Scholar] [CrossRef]
- Gois Ruivo da Silva, M.; Skrt, M.; Komes, D.; Poklar Ulrih, N.; Pogačnik, L. Enhanced Yield of Bioactivities from Onion (Allium cepa L.) Skin and Their Antioxidant and Anti-α-Amylase Activities. Int. J. Mol. Sci. 2020, 21, 2909. [Google Scholar] [CrossRef]
- Galvan D’Alessandro, L. Eco-Procédés Pour La Récupération Sélective d’antioxydants à Partir d’Aronia Melanocarpa et Ses Co-Produits. Ph.D. Thesis, Lille University, Lille, France, 4 December 2013. [Google Scholar]
- Palmieri, S.; Pellegrini, M.; Ricci, A.; Compagnone, D.; Lo Sterzo, C. Chemical Composition and Antioxidant Activity of Thyme, Hemp and Coriander Extracts: A Comparison Study of Maceration, Soxhlet, UAE and RSLDE Techniques. Foods 2020, 9, 1221. [Google Scholar] [CrossRef]
- Melguizo-Melguizo, D.; Diaz-de-Cerio, E.; Quirantes-Piné, R.; Švarc-Gajić, J.; Segura-Carretero, A. The Potential of Artemisia Vulgaris Leaves as a Source of Antioxidant Phenolic Compounds. J. Funct. Foods 2014, 10, 192–200. [Google Scholar] [CrossRef]
- Dall’Acqua, S.; Ak, G.; Sut, S.; Ferrarese, I.; Zengin, G.; Yıldıztugay, E.; Mahomoodally, M.F.; Sinan, K.I.; Lobine, D. Phenolics from Scorzonera Tomentosa L: Exploring the Potential Use in Industrial Applications via an Integrated Approach. Ind. Crops Prod. 2020, 154, 112751. [Google Scholar] [CrossRef]
- Rafińska, K.; Pomastowski, P.; Rudnicka, J.; Krakowska, A.; Maruśka, A.; Narkute, M.; Buszewski, B. Effect of Solvent and Extraction Technique on Composition and Biological Activity of Lepidium Sativum Extracts. Food Chem. 2019, 289, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Veličković, V.; Đurović, S.; Radojković, M.; Cvetanović, A.; Švarc-Gajić, J.; Vujić, J.; Trifunović, S.; Mašković, P.Z. Application of Conventional and Non-Conventional Extraction Approaches for Extraction of Erica Carnea L.: Chemical Profile and Biological Activity of Obtained Extracts. J. Supercrit. Fluids 2017, 128, 331–337. [Google Scholar] [CrossRef]
- Choi, S.J.; Kim, J.K.; Kim, H.K.; Harris, K.; Kim, C.-J.; Park, G.G.; Park, C.-S.; Shin, D.-H. 2, 4-Di-Tert-Butylphenol from Sweet Potato Protects against Oxidative Stress in PC12 Cells and in Mice. J. Med. Food 2013, 16, 977–983. [Google Scholar] [CrossRef]
- Nda-Umar, U.I.; Ramli, I.B.; Muhamad, E.N.; Azri, N.; Amadi, U.F.; Taufiq-Yap, Y.H. Influence of Heterogeneous Catalysts and Reaction Parameters on the Acetylation of Glycerol to Acetin: A Review. Appl. Sci. 2020, 10, 7155. [Google Scholar] [CrossRef]
- Dahmani, M.M.; Laoufi, R.; Selama, O.; Arab, K. Gas Chromatography Coupled to Mass Spectrometry Characterization, Anti-Inflammatory Effect, Wound-Healing Potential, and Hair Growth-Promoting Activity of Algerian Carthamus caeruleus L. (Asteraceae). Indian J. Pharmacol. 2018, 50, 123. [Google Scholar] [CrossRef]
- Benali, T.; Bakrim, S.; Ghchime, R.; Benkhaira, N.; El Omari, N.; Balahbib, A.; Taha, D.; Zengin, G.; Hasan, M.M.; Bibi, S. Pharmacological Insights into the Multifaceted Biological Properties of Quinic Acid. Biotechnol. Genet. Eng. Rev. 2022, 21, 1–30. [Google Scholar] [CrossRef]
- Sarıaltın, S.Y.; Acıkara, Ö.B. Assessment of Correlation Analysis, Phytochemical Profile, and Biological Activities of Endemic Scorzonera Species from Turkey. Chem. Biodivers. 2022, 19, e202200007. [Google Scholar] [CrossRef]
- Brahim, H. Valorisation et Identification Structurale Des Principes Actifs de La Plante de La Famille Asteraceae. Ph.D. Thesis, Université Mentouri Constantine, Constantine, Algeria, 2017. [Google Scholar]
- Sahin, H.; Sari, A.; Ozsoy, N.; Celik, B.O. Phenolic Compounds and Bioactivity of Scorzonera Pygmaea Sibth. & Sm. Aerial Parts: In Vitro Antioxidant, Anti-Inflammatory and Antimicrobial Activities. J. Fac. Pharm. Istanb. Univ. 2020, 50, 294–300. [Google Scholar]
- Raccuia, S.A.; Melilli, M.G. Seasonal Dynamics of Biomass, Inulin, and Water-Soluble Sugars in Roots of Cynara cardunculus L. F. Crop. Res. 2010, 116, 147–153. [Google Scholar] [CrossRef]
- Shoaib, M.; Shehzad, A.; Omar, M.; Rakha, A.; Raza, H.; Sharif, H.R.; Shakeel, A.; Ansari, A.; Niazi, S. Inulin: Properties, Health Benefits and Food Applications. Carbohydr. Polym. 2016, 147, 444–454. [Google Scholar] [CrossRef]
- Song, H.-P.; Zhang, H.; Fu, Y.; Mo, H.; Zhang, M.; Chen, J.; Li, P. Screening for Selective Inhibitors of Xanthine Oxidase from Flos Chrysanthemum Using Ultrafiltration LC–MS Combined with Enzyme Channel Blocking. J. Chromatogr. B 2014, 961, 56–61. [Google Scholar] [CrossRef]
- Mehmood, A.; Ishaq, M.; Zhao, L.; Safdar, B.; Rehman, A.; Munir, M.; Raza, A.; Nadeem, M.; Iqbal, W.; Wang, C. Natural Compounds with Xanthine Oxidase Inhibitory Activity: A Review. Chem. Biol. Drug Des. 2019, 93, 387–418. [Google Scholar] [CrossRef]
- Sinha, S.; Doble, M.; Manju, S.L. 5-Lipoxygenase as a Drug Target: A Review on Trends in Inhibitors Structural Design, SAR and Mechanism Based Approach. Bioorg. Med. Chem. 2019, 27, 3745–3759. [Google Scholar] [CrossRef]
- Benites, J.; Moiteiro, C.; Miguel, G.; Rojo, L.; López, J.; Venancio, F.; Ramalho, L.; Feio, S.; Dandlen, S.; Casanova, H. Composition and Biological Activity of the Essential Oil of Peruvian Lantana Camara. J. Chil. Chem. Soc. 2009, 54, 379–384. [Google Scholar] [CrossRef]
- Maina, S.; Ryu, D.H.; Bakari, G.; Misinzo, G.; Nho, C.W.; Kim, H.-Y. Variation in Phenolic Compounds and Antioxidant Activity of Various Organs of African Cabbage (Cleome gynandra L.) Accessions at Different Growth Stages. Antioxidants 2021, 10, 1952. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Antioxidant Constituents of Three Selected Red and Green Color Amaranthus Leafy Vegetable. Sci. Rep. 2019, 9, 18233. [Google Scholar] [CrossRef]
- Wang, L.; Clardy, A.; Hui, D.; Gao, A.; Wu, Y. Antioxidant and Antidiabetic Properties of Chinese and Indian Bitter Melons (Momordica charantia L.). Food Biosci. 2019, 29, 73–80. [Google Scholar] [CrossRef]
- Dobrinas, S.; Soceanu, A.; Popescu, V.; Carazeanu Popovici, I.; Jitariu, D. Relationship between Total Phenolic Content, Antioxidant Capacity, Fe and Cu Content from Tea Plant Samples at Different Brewing Times. Processes 2021, 9, 1311. [Google Scholar] [CrossRef]


| Water | Ethanol | |||||
|---|---|---|---|---|---|---|
| TB | LV | FL | TB | LV | FL | |
| Maceration | 0.81 | 0.57 | 0.12 | 0.16 | 0.14 | 0.19 |
| Ultrasound | 1.16 | 0.47 | 0.29 | 0.14 | 0.12 | 0.15 |
| N° | Compound Name | RT | MF | MW | Maceration | Ultrasound | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2O | Ethanol | H2O | Ethanol | |||||||||||||
| TB | LV | FL | TB | LV | FL | TB | LV | FL | TB | LV | FL | |||||
| 1 | 1-Hexadecanol | 14.64 | C16H34O | 242.4 | X | |||||||||||
| 2 | 2,4-Di-tert-butylphenol | 16.33 | C14H22O | 206.3 | X | X | X | X | X | X | X | X | X | X | ||
| 3 | 10-Heneicosene | 16.41 | C21H42 | 294.6 | X | |||||||||||
| 4 | 1-Hexadecanol, 2-methyl- | 16.42 | C17H36O | 256.5 | X | |||||||||||
| 5 | Octadecane,1,1′-[(1-methyl-1,2 ethanediyl)bis(oxy)]bis- | 17.52 | C39H80O2 | 581.1 | X | |||||||||||
| 6 | 2(4H)-Benzofuranone,5,6,7,7a-tetrahydro-4,4,7a-trimethyl-, (R)- | 17.64 | C11H16O2 | 180.2 | X | |||||||||||
| 7 | Phenol,2,2′-methylenebis[6-(1,1-dimethylethyl)-4-methyl- | 17.91 | C23H32O2 | 340.4 | X | X | X | X | X | |||||||
| 8 | 7,9-Di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione | 21.07 | C17H24O3 | 276.3 | ||||||||||||
| 9 | 3,7,11,15-Tetramethyl-2-hexadecen-1-ol | 22.13 | C20H40O | 296.5 | X | |||||||||||
| 10 | 9-Hexadecenoic acid, 9-octadecenyl ester, (Z,Z)- | 28.27 | C34H64O2 | 504.8 | X | X | X | |||||||||
| 11 | Z-10-Methyl-11-tetradecen-1-ol propionate | 28.29 | C18H34O2 | 282.5 | X | X | ||||||||||
| 12 | Heneicosane, 11-(-ethylpropyl) | 19.46 | C26H54 | 366.7 | X | |||||||||||
| 13 | Heneicosane | 21.32 | C21H44 | 296.5 | X | |||||||||||
| 14 | Pentacosane | 21.33 | C25H52 | 352.6 | X | |||||||||||
| N° | Compound | RT | MF | MW | Maceration | Ultrasound | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2O | Ethanol | H2O | Ethanol | |||||||||||||
| TB | LV | FL | TB | LV | FL | TB | LV | FL | TB | LV | FL | |||||
| 1 | 2,3-Butanediol | 7.85 | C4H10O2 | 90.1 | X | |||||||||||
| 2 | Lactic acid | 8.47 | C3H6O3 | 90.0 | X | X | X | X | X | X | X | X | X | X | ||
| 3 | Hexanoic acid | 8.82 | C6H12O2 | 116.1 | X | |||||||||||
| 4 | 2-Monoacetin | 10.42 | C9H14O6 | 218.2 | X | X | X | X | ||||||||
| 5 | Glycerol | 10.79 | C3H8O3 | 92,0 | X | X | X | X | X | X | X | X | X | X | X | |
| 6 | Butanedioic acid | 13.06 | C4H6O4 | 118.0 | X | X | X | X | ||||||||
| 7 | Decanoic acid | 14.90 | C10H20O2 | 172,2 | X | X | ||||||||||
| 8 | Malic acid | 15.27 | C4H6O5 | 134.0 | X | X | ||||||||||
| 9 | α-d-Fructofuranoe | 18.19 | C12H22O11 | 342.3 | X | X | X | X | X | X | X | X | ||||
| 10 | α-d-Fructopyranose | 18.55 | C6H12O6 | 180.1 | X | X | X | X | ||||||||
| 11 | Palmitic Acid | 20.94 | C16H32O2 | 256,4 | X | X | X | X | X | X | X | X | X | X | X | |
| N° | m/z | MF | MW | RT | Maceration | Ultrasound | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TB | LV | FL | TB | LV | FL | ||||||
| Phenolic acid concentration (mg/100 g DW) | |||||||||||
| 1 | Quinic acid | 191 | C7H12O6 | 192.1 | 1.59 | 34.19 ± 6.07 E | 165.55 ± 7.04 B | 81.70 ± 3.10 C | 40.13 ± 10.05 E | 187.69 ± 14.00 A | 63.97 ± 9.04 D |
| 2 | 1,3-dicaffeoylquinic acid | 515 | C25H24O12 | 516.4 | 1.61 | 0.28 ± 0.06 C | 4.41 ± 0.07 A | 3.40 ± 0.47 B | 0.48 ± 0.10 C | 2.97 ± 0.47 B | 3.08 ± 0.29 B |
| 3 | Gallic acid | 169 | C7H6O5 | 170.1 | 1.64 | 0.05 ± 0.04 A | 0.14 ± 0.07 A | 0.09 ± 0.03 A | 0.09 ± 0.09 A | 0.18 ± 0.07 A | 0.12 ± 0.08 A |
| 4 | Protocatechuic acid | 153 | C7H6O4 | 154.1 | 4.61 | 0.10 ± 0.03 D | 0.98 ± 0.07 B | 0.40 ± 0.15 C | 1.54 ± 0.10 A | 1.17 ± 0.17 B | 0.49 ± 0.05 C |
| 5 | Chlorogenic acid | 353 | C16H18O9 | 354.3 | 8.6 | 1.13 ± 0.08 E | 131.34 ± 23.21 C | 196.34 ± 11.5 B | 1.15 ± 0.10 E | 31.71 ± 4.22 D | 254.43 ± 25.02A |
| 6 | Caffeic acid | 179 | C9H8O4 | 180.1 | 10.27 | nd | nd | 0.49 ± 0.20 B | nd | nd | 1.52 ± 0.09 A |
| 7 | Syringic acid | 197 | C9H10O5 | 198.1 | 11.89 | nd | 5.28 ± 0.07 B | 3.86 ± 0.88 C | nd | nd | 10.40 ± 0.78 A |
| 8 | p-coumaric acid | 163 | C9H8O3 | 164.1 | 16.2 | 0.02 ± 0.01 B | nd | 0.03 ± 0.01 B | 0.02 ± 0.02 B | 0.29 ± 0.07 A | 0.03 ± 0.02 B |
| 9 | Ferulic acid | 193 | C10H10O4 | 194.1 | 19.54 | 0.03 ± 0.02 BC | 0.19 ± 0.07 A | 0.05 ± 0.01 BC | 0.02 ± 0.01 BC | nd | 0.06 ± 0.01 B |
| 10 | Rosmarinic acid | 359 | C18H16O8 | 360.3 | 24.30 | nd | nd | 0.03 ± 0.02 A | nd | nd | nd |
| 11 | Cinnamic acid | 147 | C9H8O2 | 148.1 | 29.22 | 0.15 ± 0.06 B | 0.51 ± 0.07 A | nd | nd | nd | nd |
| 1 | Rutin | 609 | C27H30O16 | 610.5 | 21.66 | 0.06 ± 0.02 B | 0.33 ± 0.09 A | nd | 0.02 ± 0.00 C | 0.17 ± 0.07 B | 0.27 ± 0.09 A |
| 2 | Quercetin | 301 | C15H10O7 | 302.2 | 29.12 | nd | 0.19 ± 0.07 A | nd | nd | nd | nd |
| 3 | Hyperoside | 463 | C21H20O12 | 464.3 | 21.71 | 0.07 ± 0.03 E | 0.93 ± 0.09 A | 0.45 ± 0.04 C | 0.04 ± 0.04 E | 0.23 ± 0.07 D | 0.80 ± 0.02 B |
| 4 | Luteolin-7-O-Glucoside | 447 | C21H20O11 | 448.3 | 22.32 | 0.18 ± 0.02 D | 245.11 ± 22.35 A | 31.29 ± 0.47 C | nd | 32.84 ± 6.22 C | 63.32 ± 4.22 B |
| 5 | Naringin | 579 | C27H32O14 | 580.5 | 23.94 | 0.18 ± 0.06 C | 37.35 ± 15.02 A | 10.01 ± 0.88 BC | nd | 5.66 ± 0.66 BC | 12.67 ± 1.33 B |
| 6 | Quercetrin | 447 | C15H10O7 | 302.2 | 24.47 | 0.11 ± 0.1 D | 5.91 ± 1.12 C | 22.33 ± 2.44 B | 0.15 ± 0.10 D | 2.91 ± 0.40 CD | 43.96 ± 4.55 A |
| 7 | Apigenin | 431 | C15H10O5 | 270.0 | 24.58 | 0.06 ± 0.08 D | 60.05 ± 2.47 A | 43.35 ± 8.54 B | 0.01 ± 0.00 D | 7.61 ± 1.01 C | 62.40 ± 4.88 A |
| 8 | kaempferol | 285 | C15H10O6 | 286.2 | 29.22 | nd | 153.49 ± 5.22 A | 46.01 ± 14.20 C | nd | 145.90 ± 2.33 AB | 135.62 ± 15.54 B |
| 9 | Naringenin | 271 | C15H12O5 | 272.2 | 31.39 | 0.03 ± 0.02 D | 0.18 ± 0.07 C | 0.13 ± 0.03 CD | 0.06 ± 0.04 D | 0.70 ± 0.07 A | 0.37 ± 0.09 B |
| 10 | Apigenin-7-O-glucoside | 269 | C21H20O10 | 432.4 | 31.95 | 0.01 ± 0.00E | 1.79 ± 0.10 C | 1.40 ± 0.12 D | 0.01 ± 0.01 E | 3.47 ± 0.12 A | 2.99 ± 0.47 B |
| Anti-XOD | Anti-5-LOX | Anti-α-Glucosidase | Anti-α-Amylase | |||
|---|---|---|---|---|---|---|
| Extract | ||||||
| Maceration | H2O | TB | nd | nd | nd | 18.42 ± 5.12 BC |
| LV | nd | nd | nd | 12.11 ± 0.25 CD | ||
| FL | nd | nd | 2.49 ± 1.96 B | nd | ||
| Ethanol | TB | nd | nd | nd | 17.24 ± 4.58 BC | |
| LV | 38.26 ± 0.79 A | 1.23 ± 3.43 B | nd | 20.50 ± 0.56 B | ||
| FL | 9.44 ± 7.68 B | 11.39 ± 1.03 A | nd | nd | ||
| Ultrasound | H2O | TB | nd | nd | 7.24 ± 2.06 AB | 20.08 ± 0.04 B |
| LV | 1.65 ± 0.62 C | nd | 8.71 ± 2.30 A | nd | ||
| FL | 11.72 ± 5.67 B | 3.48 ± 1.74 B | 9.77 ± 4.10 A | 31.34 ± 7.50 A | ||
| Ethanol | TB | nd | nd | nd | nd | |
| LV | 14.05 ± 2.43 B | nd | nd | 8.52 ± 1.28 D | ||
| FL | 11.05 ± 2.37 B | 14.05 ± 5.81 A | 9.71 ± 3.88 A | nd | ||
| Reference | Allopurinol (1 µg/mL) | NDGA (4 µg/mL) | Acarbose (50 µg/mL) | Acarbose (50 µg/mL) | ||
| 52.26 ± 8.56 | 44.08 ± 5.30 | 60.22 ± 2.50 | 32.24 ± 7.23 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Idoudi, S.; Othman, K.B.; Bouajila, J.; Tourrette, A.; Romdhane, M.; Elfalleh, W. Influence of Extraction Techniques and Solvents on the Antioxidant and Biological Potential of Different Parts of Scorzonera undulata. Life 2023, 13, 904. https://doi.org/10.3390/life13040904
Idoudi S, Othman KB, Bouajila J, Tourrette A, Romdhane M, Elfalleh W. Influence of Extraction Techniques and Solvents on the Antioxidant and Biological Potential of Different Parts of Scorzonera undulata. Life. 2023; 13(4):904. https://doi.org/10.3390/life13040904
Chicago/Turabian StyleIdoudi, Sourour, Khadija Ben Othman, Jalloul Bouajila, Audrey Tourrette, Mehrez Romdhane, and Walid Elfalleh. 2023. "Influence of Extraction Techniques and Solvents on the Antioxidant and Biological Potential of Different Parts of Scorzonera undulata" Life 13, no. 4: 904. https://doi.org/10.3390/life13040904
APA StyleIdoudi, S., Othman, K. B., Bouajila, J., Tourrette, A., Romdhane, M., & Elfalleh, W. (2023). Influence of Extraction Techniques and Solvents on the Antioxidant and Biological Potential of Different Parts of Scorzonera undulata. Life, 13(4), 904. https://doi.org/10.3390/life13040904











