Bronchoscopic Management of COPD and Advances in Therapy
Abstract
1. Introduction
2. Bronchoscopic Lung Volume Reduction
2.1. History
2.2. Pre-Procedural Screening and Evaluation
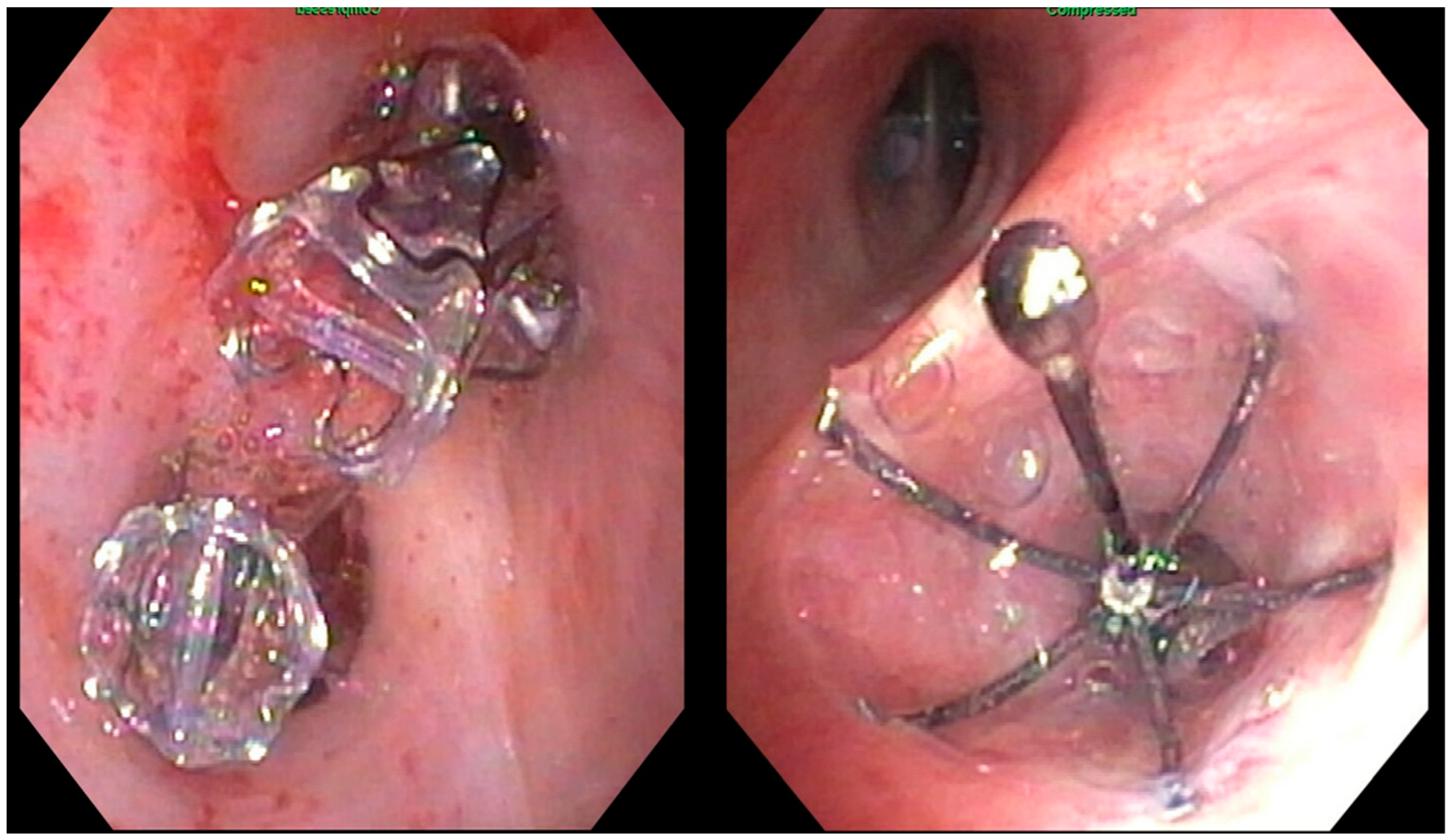
2.3. Endobronchial Valve Placement
2.4. Post-Procedural Outcomes
3. Investigational Therapies for BLVR
3.1. Endobronchial Coils
3.2. Thermal Vapor Ablation
3.3. Biologic Lung Reduction
3.4. Airway Bypass Stents
4. Investigational Therapies Focused on Mucus Hypersecretion and Inflammation
4.1. Targeted Lung Denervation
4.2. Bronchial Rheoplasty
Metered Cryospray and Balloon Deobstruction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Celli, B.R.; Wedzicha, J.A. Update on Clinical Aspects of Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2019, 381, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Safiri, S.; Carson-Chahhoud, K.; Noori, M.; Nejadghaderi, S.A.; Sullman, M.J.M.; Heris, J.A.; Ansarin, K.; Mansournia, M.A.; Collins, G.S.; Kolahi, A.-A.; et al. Burden of chronic obstructive pulmonary disease and its attributable risk factors in 204 countries and territories, 1990–2019: Results from the Global Burden of Disease Study 2019. BMJ 2022, 378, e069679. [Google Scholar] [CrossRef] [PubMed]
- Agustí, A.; Hogg, J.C. Update on the Pathogenesis of Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2019, 381, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, P.; Guenette, J.; Langer, D.; Laviolette, L.; Mainguy, V.; Maltais, F.; Ribeiro, F.; Saey, D. Pathogenesis of hyperinflation in chronic obstructive pulmonary disease. Int. J. Chronic Obstr. Pulm. Dis. 2014, 9, 187–201. [Google Scholar] [CrossRef]
- Rossi, A.; Aisanov, Z.; Avdeev, S.; Di Maria, G.; Donner, C.F.; Izquierdo, J.L.; Roche, N.; Similowski, T.; Watz, H.; Worth, H.; et al. Mechanisms, assessment and therapeutic implications of lung hyperinflation in COPD. Respir. Med. 2015, 109, 785–802. [Google Scholar] [CrossRef]
- Miravitlles, M.; Ribera, A. Understanding the impact of symptoms on the burden of COPD. Respir. Res. 2017, 18, 67. [Google Scholar] [CrossRef]
- Mineo, D.; Ambrogi, V.; Cufari, M.E.; Gambardella, S.; Pignotti, L.; Pompeo, E.; Mineo, T.C. Variations of Inflammatory Mediators and α1-Antitrypsin Levels after Lung Volume Reduction Surgery for Emphysema. Am. J. Respir. Crit. Care Med. 2010, 181, 806–814. [Google Scholar] [CrossRef]
- Clarenbach, C.F.; Sievi, N.A.; Brock, M.; Schneiter, D.; Weder, W.; Kohler, M. Lung Volume Reduction Surgery and Improvement of Endothelial Function and Blood Pressure in Patients with Chronic Obstructive Pulmonary Disease. A Randomized Controlled Trial. Am. J. Respir. Crit. Care Med. 2015, 192, 307–314. [Google Scholar] [CrossRef]
- Di Marco, F.; Sotgiu, G.; Santus, P.; O’Donnell, D.E.; Beeh, K.-M.; Dore, S.; Roggi, M.A.; Giuliani, L.; Blasi, F.; Centanni, S. Long-acting bronchodilators improve exercise capacity in COPD patients: A systematic review and meta-analysis. Respir. Res. 2018, 19, 18. [Google Scholar] [CrossRef]
- Somfay, A.; Porszasz, J.; Lee, S.; Casaburi, R. Dose-response effect of oxygen on hyperinflation and exercise endurance in nonhypoxaemic COPD patients. Eur. Respir. J. 2001, 18, 77–84. [Google Scholar] [CrossRef]
- Porszasz, J.; Emtner, M.; Goto, S.; Somfay, A.; Whipp, B.J.; Casaburi, R. Exercise Training Decreases Ventilatory Requirements and Exercise-Induced Hyperinflation at Submaximal Intensities in Patients With COPD. Chest 2005, 128, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Fishman, A.; Martinez, F.; Naunheim, K.; Piantadosi, S.; Wise, R.; Ries, A.; Weinmann, G.; Wood, D.E.; National Emphysema Treatment Trial Research Group. A Randomized Trial Comparing Lung-Volume–Reduction Surgery with Medical Therapy for Severe Emphysema. N. Engl. J. Med. 2003, 348, 2059–2073. [Google Scholar] [CrossRef] [PubMed]
- Criner, G.J.; Cordova, F.; Sternberg, A.L.; Martinez, F.J. The National Emphysema Treatment Trial (NETT): Part II: Lessons Learned about Lung Volume Reduction Surgery. Am. J. Respir. Crit. Care Med. 2011, 184, 881–893. [Google Scholar] [CrossRef]
- Valipour, A.; Slebos, D.-J.; Herth, F.; Darwiche, K.; Wagner, M.; Ficker, J.H.; Petermann, C.; Hubner, R.-H.; Stanzel, F.; Eberhardt, R. Endobronchial Valve Therapy in Patients with Homogeneous Emphysema. Results from the IMPACT Study. Am. J. Respir. Crit. Care Med. 2016, 194, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Vogelmeier, C.F.; Alter, P. Assessing Symptom Burden. Clin. Chest Med. 2020, 41, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.W.; Harding, G.; Berry, P.; Wiklund, I.; Chen, W.H.; Kline Leidy, N. Development and first validation of the COPD Assessment Test. Eur. Respir. J. 2009, 34, 648–654. [Google Scholar] [CrossRef]
- van Dijk, M.; Hartman, J.E.; ten Klooster, K.; Hacken, N.H.T.; Kerstjens, H.A.; Slebos, D.-J. Endobronchial Valve Treatment in Emphysema Patients with a Very Low DLCO. Respiration 2020, 99, 163–170. [Google Scholar] [CrossRef]
- Lenga, P.; Ruwwe-Glösenkamp, C.; Grah, C.; Pfannschmidt, J.; Rückert, J.; Eggeling, S.; Gläser, S.; Schmidt, B.; Schneider, P.; Kurz, S.; et al. Endoscopic lung volume reduction with endobronchial valves in very lowDLCOpatients: Results from the German Registry—Lungenemphysemregister e.V. ERJ Open Res. 2021, 7, 1–8. [Google Scholar] [CrossRef]
- Criner, G.J.; Sue, R.; Wright, S.; Dransfield, M.; Rivas-Perez, H.; Wiese, T.; Sciurba, F.C.; Shah, P.L.; Wahidi, M.M.; de Oliveira, H.G.; et al. A Multicenter Randomized Controlled Trial of Zephyr Endobronchial Valve Treatment in Heterogeneous Emphysema (LIBERATE). Am. J. Respir. Crit. Care Med. 2018, 198, 1151–1164. [Google Scholar] [CrossRef]
- Criner, G.J.; Delage, A.; Voelker, K.; Hogarth, D.K.; Majid, A.; Zgoda, M.; Lazarus, D.R.; Casal, R.; Benzaquen, S.B.; Holladay, R.C.; et al. Improving Lung Function in Severe Heterogenous Emphysema with the Spiration Valve System (EMPROVE). A Multicenter, Open-Label Randomized Controlled Clinical Trial. Am. J. Respir. Crit. Care Med. 2019, 200, 1354–1362. [Google Scholar] [CrossRef]
- Lee, S.W.; Lee, S.M.; Shin, S.Y.; Park, T.S.; Oh, S.Y.; Kim, N.; Hong, Y.; Lee, J.S.; Oh, Y.-M.; Seo, J.B. Improvement in Ventilation-Perfusion Mismatch after Bronchoscopic Lung Volume Reduction: Quantitative Image Analysis. Radiology 2017, 285, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Klooster, K.; Slebos, D.-J. Endobronchial Valves for the Treatment of Advanced Emphysema. Chest 2021, 159, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.V.; Slebos, D.-J.; Kirk, A.; Kornaszewska, M.; Carron, K.; Ek, L.; Broman, G.; Hillerdal, G.; Mal, H.; Pison, C.; et al. A Multicenter Randomized Controlled Trial of Zephyr Endobronchial Valve Treatment in Heterogeneous Emphysema (TRANSFORM). Am. J. Respir. Crit. Care Med. 2017, 196, 1535–1543. [Google Scholar] [CrossRef]
- Dass, C.M.; Goldbach, A.D.; Dako, F.M.; Kumaran, M.M.; Steiner, R.; Criner, G.J. Role of Imaging in Bronchoscopic Lung Volume Reduction Using Endobronchial Valve: State of the Art Review. J. Thorac. Imaging 2020, 36, 131–141. [Google Scholar] [CrossRef]
- Chandra, D.; Lipson, D.A.; Hoffman, E.A.; Hansen-Flaschen, J.; Sciurba, F.C.; DeCamp, M.M.; Reilly, J.J.; Washko, G.R. Perfusion Scintigraphy and Patient Selection for Lung Volume Reduction Surgery. Am. J. Respir. Crit. Care Med. 2010, 182, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Klooster, K.; Koster, T.D.; Ruwwe-Glösenkamp, C.; Theilig, D.; Doellinger, F.; Saccomanno, J.; Kerstjens, H.A.; Slebos, D.-J.; Hübner, R.-H. An Integrative Approach of the Fissure Completeness Score and Chartis Assessment in Endobronchial Valve Treatment for Emphysema. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 1325–1334. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Ram, S.; Labaki, W.W.; Han, M.K.; Galbán, C.J. CT-Based Commercial Software Applications: Improving Patient Care Through Accurate COPD Subtyping. Int. J. Chronic Obstr. Pulm. Dis. 2022, 17, 919–930. [Google Scholar] [CrossRef]
- Gevenois, P.A.; De Vuyst, P.; De Maertelaer, V.; Zanen, J.; Jacobovitz, D.; Cosio, M.G.; Yernault, J.C. Comparison of computed density and microscopic morphometry in pulmonary emphysema. Am. J. Respir. Crit. Care Med. 1996, 154, 187–192. [Google Scholar] [CrossRef]
- Coxson, H.O.; Rogers, R.M.; Whittall, K.P.; D'Yachkova, Y.; Paré, P.D.; Sciurba, F.C.; Hogg, J.C. A Quantification of the Lung Surface Area in Emphysema Using Computed Tomography. Am. J. Respir. Crit. Care Med. 1999, 159, 851–856. [Google Scholar] [CrossRef]
- Sciurba, F.C.; Ernst, A.; Herth, F.J.; Strange, C.; Criner, G.J.; Marquette, C.H.; Kovitz, K.L.; Chiacchierini, R.P.; Goldin, J.; McLennan, G. A Randomized Study of Endobronchial Valves for Advanced Emphysema. N. Engl. J. Med. 2010, 363, 1233–1244. [Google Scholar] [CrossRef]
- Schuhmann, M.; Raffy, P.; Yin, Y.; Gompelmann, D.; Oguz, I.; Eberhardt, R.; Hornberg, D.; Heussel, C.P.; Wood, S.; Herth, F.J.F. Computed Tomography Predictors of Response to Endobronchial Valve Lung Reduction Treatment. Comparison with Chartis. Am. J. Respir. Crit. Care Med. 2015, 191, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Koster, D.; Slebos, D.-J. The fissure: Interlobar collateral ventilation and implications for endoscopic therapy in emphysema. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Zantah, M.; Gangemi, A.J.; Criner, G.J. Bronchoscopic lung volume reduction: Status quo. Ann. Transl. Med. 2020, 8, 1469. [Google Scholar] [CrossRef] [PubMed]
- Herth, F.J.; Slebos, D.-J.; Criner, G.J.; Valipour, A.; Sciurba, F.; Shah, P.L. Endoscopic Lung Volume Reduction: An Expert Panel Recommendation—Update 2019. Respiration 2019, 97, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Klooster, K.; ten Hacken, N.H.T.; Hartman, J.E.; Kerstjens, H.A.M.; Van Rikxoort, E.M.; Slebos, D.-J. Endobronchial Valves for Emphysema without Interlobar Collateral Ventilation. N. Engl. J. Med. 2015, 373, 2325–2335. [Google Scholar] [CrossRef]
- Hartman, J.E.; Klooster, K.; Slebos, D.-J. From Bench to Bedside: Implementation of Endobronchial Valve Treatment for Patients with Advanced Emphysema in Routine Clinical Care. Respiration 2020, 99, 187–188. [Google Scholar] [CrossRef] [PubMed]
- Chew, J.; Mahadeva, R. The role of a multidisciplinary severe chronic obstructive pulmonary disease hyperinflation service in patient selection for lung volume reduction. J. Thorac. Dis. 2018, 10 (Suppl. 27), S3335–S3343. [Google Scholar] [CrossRef]
- Davey, C.; Zoumot, Z.; Jordan, S.; McNulty, W.H.; Carr, D.H.; Hind, M.D.; Hansell, D.M.; Rubens, M.B.; Banya, W.; Polkey, M.I.; et al. Bronchoscopic lung volume reduction with endobronchial valves for patients with heterogeneous emphysema and intact interlobar fissures (the BeLieVeR-HIFi study): A randomised controlled trial. Lancet 2015, 386, 1066–1073. [Google Scholar] [CrossRef]
- Li, S.; Wang, G.; Wang, C.; Gao, X.; Jin, F.; Yang, H.; Han, B.; Zhou, R.; Chen, C.; Chen, L.; et al. The REACH Trial: A Randomized Controlled Trial Assessing the Safety and Effectiveness of the Spiration® Valve System in the Treatment of Severe Emphysema. Respiration 2018, 97, 416–427. [Google Scholar] [CrossRef]
- Herth, F.J.; Noppen, M.; Valipour, A.; Leroy, S.; Vergnon, J.-M.; Ficker, J.H.; Egan, J.J.; Gasparini, S.; Agusti, C.; Holmes-Higgin, D.; et al. Efficacy predictors of lung volume reduction with Zephyr valves in a European cohort. Eur. Respir. J. 2012, 39, 1334–1342. [Google Scholar] [CrossRef]
- Wood, D.E.; McKenna, R.J.; Yusen, R.D.; Sterman, D.; Ost, D.; Springmeyer, S.C.; Gonzalez, H.X.; Mulligan, M.S.; Gildea, T.; Houck, W.V.; et al. A multicenter trial of an intrabronchial valve for treatment of severe emphysema. J. Thorac. Cardiovasc. Surg. 2007, 133, 65–73.e2. [Google Scholar] [CrossRef] [PubMed]
- Eberhardt, R.; Slebos, D.-J.; Herth, F.J.; Darwiche, K.; Wagner, M.; Ficker, J.H.; Petermann, C.; Hübner, R.-H.; Stanzel, F.; Shargill, N.S.; et al. Endobronchial Valve (Zephyr) Treatment in Homogeneous Emphysema: One-Year Results from the IMPACT Randomized Clinical Trial. Respiration 2021, 100, 1174–1185. [Google Scholar] [CrossRef] [PubMed]
- Kirk, A.; Slebos, D.-J.; Kornaszweska, M.; Shah, P.; Carron, K.; Mal, H.; Pison, C.; Downer, N.; Darwiche, K.; Rao, J.; et al. Durability of Zephyr Valve treatment: 24-month follow-up in the TRANSFORM Study. Eur. Respir. J. 2021, 58 (Suppl. 65), OA1201. [Google Scholar] [CrossRef]
- Patel, M.; Chowdhury, J.; Zhao, H.; Lu, X.M.; Roth, S.M.; Giovacchini, C.X.; Wahidi, M.M.; Criner, G. Meta-analysis and Systematic Review of Bronchoscopic Lung Volume Reduction Through Endobronchial Valves in Severe Emphysema. J. Bronchol. Interv. Pulmonol. 2022, 29, 224–237. [Google Scholar] [CrossRef] [PubMed]
- Koster, T.D.; Klooster, K.; Ten Hacken, N.H.T.; van Dijk, M.; Slebos, D.-J. Endobronchial valve therapy for severe emphysema: An overview of valve-related complications and its management. Expert Rev. Respir. Med. 2020, 14, 1235–1247. [Google Scholar] [CrossRef]
- van Dijk, M.; Sue, R.; Criner, G.J.; Gompelmann, D.; Herth, F.J.; Hogarth, D.K.; Klooster, K.; Kocks, J.W.; de Oliveira, H.G.; Shah, P.L.; et al. Expert Statement: Pneumothorax Associated with One-Way Valve Therapy for Emphysema: 2020 Update. Respiration 2021, 100, 969–978. [Google Scholar] [CrossRef]
- Roodenburg, S.A.; Klooster, K.; Hartman, J.E.; Koster, T.D.; van Dijk, M.; Slebos, D.J. Revision Bronchoscopy After Endobronchial Valve Treatment for Emphysema: Indications, Findings and Outcomes. Int. J. Chronic Obstr. Pulm. Dis. 2021, 16, 1127–1136. [Google Scholar] [CrossRef]
- Slebos, D.-J.; ten Hacken, N.H.; Hetzel, M.; Herth, F.J.; Shah, P.L. Endobronchial Coils for Endoscopic Lung Volume Reduction: Best Practice Recommendations from an Expert Panel. Respiration 2018, 96, 1–11. [Google Scholar] [CrossRef]
- Slebos, D.-J.; Cicenia, J.; Sciurba, F.C.; Criner, G.J.; Hartman, J.E.; Garner, J.; Deslée, G.; Delage, A.; Jantz, M.; Marquette, C.-H.; et al. Predictors of Response to Endobronchial Coil Therapy in Patients With Advanced Emphysema. Chest 2019, 155, 928–937. [Google Scholar] [CrossRef]
- Hartman, J.E.; Shah, P.L.; Sciurba, F.; Herth, F.J.; Slebos, D.; Gompelmann, D.; Schuhmann, M.; Eberhardt, R.; Harzheim, D.; Rump, B.; et al. Endobronchial coils for emphysema: Dual mechanism of action on lobar residual volume reduction. Respirology 2020, 25, 1160–1166. [Google Scholar] [CrossRef]
- Sciurba, F.C.; Criner, G.J.; Strange, C.; Shah, P.L.; Michaud, G.; Connolly, T.A.; Deslée, G.; Tillis, W.P.; Delage, A.; Marquette, C.-H.; et al. Effect of Endobronchial Coils vs Usual Care on Exercise Tolerance in Patients with Severe Emphysema: The RENEW Randomized Clinical Trial. JAMA 2016, 315, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Deslée, G.; Mal, H.; Dutau, H.; Bourdin, A.; Vergnon, J.M.; Pison, C.; Kessler, R.; Jounieaux, V.; Thiberville, L.; Leroy, S.; et al. Lung Volume Reduction Coil Treatment vs Usual Care in Patients with Severe Emphysema: The REVOLENS Randomized Clinical Trial. JAMA 2016, 315, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.L.; Zoumot, Z.; Singh, S.; Bicknell, S.R.; Ross, E.T.; Quiring, J.; Hopkinson, N.S.; Kemp, S.V. Endobronchial coils for the treatment of severe emphysema with hyperinflation (RESET): A randomised controlled trial. Lancet Respir. Med. 2013, 1, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Herth, F.J.; Slebos, D.-J.; Shah, P.L.; Hetzel, M.; Schmid-Bindert, G.; LaPrad, A.S.; Deslée, G.; Valipour, A. Protocol of a Randomized Controlled Study of the PneumRx Endobronchial Coil System versus Standard-of-Care Medical Management in the Treatment of Subjects with Severe Emphysema (ELEVATE). Respiration 2019, 98, 512–520. [Google Scholar] [CrossRef] [PubMed]
- 2023 GOLD Report—Global Initiative for Chronic Obstructive Lung Disease—GOLD. Available online: https://goldcopd.org/2023-gold-report-2/ (accessed on 24 February 2023).
- Gompelmann, D.; Shah, P.L.; Valipour, A.; Herth, F.J. Bronchoscopic Thermal Vapor Ablation: Best Practice Recommendations from an Expert Panel on Endoscopic Lung Volume Reduction. Respiration 2018, 95, 392–400. [Google Scholar] [CrossRef]
- Snell, G.; Herth, F.J.; Hopkins, P.; Baker, K.M.; Witt, C.; Gotfried, M.H.; Valipour, A.; Wagner, M.; Stanzel, F.; Egan, J.J.; et al. Bronchoscopic thermal vapour ablation therapy in the management of heterogeneous emphysema. Eur. Respir. J. 2011, 39, 1326–1333. [Google Scholar] [CrossRef]
- Herth, F.J.; Shah, P.; Valipour, A.; Eberhardt, R.; Grah, C.; Egan, J.; Ficker, J.; Wagner, M.; Witt, C.; Liebers, U.; et al. STEP-UP randomized controlled trial of vapor ablation in patients with severe emphysema: 12 month results. Eur. Respir. J. 2016, 48 (Suppl. 60), OA475. [Google Scholar] [CrossRef]
- Bakeer, M.; Abdelgawad, T.T.; Ali, R.E.-M.; El-Morsi, A.; El-Badrawy, M.K.; El-Sharawy, S. Low cost biological lung volume reduction therapy for advanced emphysema. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 1793–1800. [Google Scholar] [CrossRef]
- Herth, F.; Gompelmann, D.; Stanzel, F.; Bonnet, R.; Behr, J.; Schmidt, B.; Magnussen, H.; Ernst, A.; Eberhardt, R. Treatment of Advanced Emphysema with Emphysematous Lung Sealant (AeriSeal®). Respiration 2011, 82, 36–45. [Google Scholar] [CrossRef]
- Joglekar, M.M.; Slebos, D.-J.; Leijten, J.; Burgess, J.K.; Pouwels, S.D. Crosslink bio-adhesives for bronchoscopic lung volume reduction: Current status and future direction. Eur. Respir. Rev. 2021, 30, 210142. [Google Scholar] [CrossRef]
- Come, C.E.; Kramer, M.R.; Dransfield, M.T.; Abu-Hijleh, M.; Berkowitz, D.; Bezzi, M.; Bhatt, S.P.; Boyd, M.B.; Cases, E.; Chen, A.C.; et al. A randomised trial of lung sealant versus medical therapy for advanced emphysema. Eur. Respir. J. 2015, 46, 651–662. [Google Scholar] [CrossRef]
- Perotin, J.M.; Dewolf, M.; Launois, C.; Dormoy, V.; Deslee, G. Bronchoscopic management of asthma, COPD and emphysema. Eur. Respir. Rev. 2021, 30, 200029. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.; Slebos, D.-J.; Cardoso, P.; Cetti, E.; Voelker, K.; Levine, B.; Russell, M.; Goldin, J.; Brown, M.; Cooper, J.; et al. Bronchoscopic lung-volume reduction with Exhale airway stents for emphysema (EASE trial): Randomised, sham-controlled, multicentre trial. Lancet 2011, 378, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Dotan, Y.; So, J.Y.; Kim, V. Chronic Bronchitis: Where Are We Now? Chronic Obstr. Pulm. Dis. J. COPD Found. 2019, 6, 178–192. [Google Scholar] [CrossRef]
- Hartman, J.E.; Garner, J.L.; Shah, P.L.; Slebos, D.-J. New bronchoscopic treatment modalities for patients with chronic bronchitis. Eur. Respir. Rev. 2021, 30, 200281. [Google Scholar] [CrossRef]
- Valipour, A.; Shah, P.L.; Pison, C.; Ninane, V.; Janssens, W.; Perez, T.; Kessler, R.; Deslee, G.; Garner, J.; Abele, C.; et al. Safety and Dose Study of Targeted Lung Denervation in Moderate/Severe COPD Patients. Respiration 2019, 98, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Slebos, D.-J.; Klooster, K.; Koegelenberg, C.F.N.; Theron, J.; Styen, D.; Valipour, A.; Mayse, M.; Bolliger, C.T. Targeted lung denervation for moderate to severe COPD: A pilot study. Thorax 2015, 70, 411–419. [Google Scholar] [CrossRef]
- Valipour, A.; Asadi, S.; Pison, C.; Jondot, M.; Kessler, R.; Benneddif, K.; Deslee, G.; Verdier, M.; Slebos, D.-J.; Mayse, M. Long-term safety of bilateral targeted lung denervation in patients with COPD. Int. J. Chronic Obstr. Pulm. Dis. 2018, 13, 2163–2172. [Google Scholar] [CrossRef]
- Slebos, D.-J.; Shah, P.L.; Herth, F.J.F.; Pison, C.; Schumann, C.; Hübner, R.-H.; Bonta, P.I.; Kessler, R.; Gesierich, W.; Darwiche, K.; et al. Safety and Adverse Events after Targeted Lung Denervation for Symptomatic Moderate to Severe Chronic Obstructive Pulmonary Disease (AIRFLOW). A Multicenter Randomized Controlled Clinical Trial. Am. J. Respir. Crit. Care Med. 2019, 200, 1477–1486. [Google Scholar] [CrossRef]
- Valipour, A.; Shah, P.L.; Herth, F.J.; Pison, C.; Schumann, C.; Hübner, R.-H.; Bonta, P.I.; Kessler, R.; Gesierich, W.; Darwiche, K.; et al. Two-Year Outcomes for the Double-Blind, Randomized, Sham-Controlled Study of Targeted Lung Denervation in Patients with Moderate to Severe COPD: AIRFLOW-2. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 2807–2816. [Google Scholar] [CrossRef]
- Slebos, D.J.; Degano, B.; Valipour, A.; Shah, P.L.; Deslée, G.; Sciurba, F.C.; AIRFLOW-3 Trial Study Group. Design for a multicenter, randomized, sham-controlled study to evaluate safety and efficacy after treatment with the Nuvaira® lung denervation system in subjects with chronic obstructive pulmonary disease (AIRFLOW-3). BMC Pulm. Med. 2020, 20, 41. [Google Scholar] [CrossRef]
- Valipour, A.; Fernandez-Bussy, S.; Ing, A.J.; Steinfort, D.P.; Snell, G.I.; Williamson, J.P.; Saghaie, T.; Irving, L.B.; Dabscheck, E.J.; Krimsky, W.S.; et al. Bronchial Rheoplasty for Treatment of Chronic Bronchitis. Twelve-Month Results from a Multicenter Clinical Trial. Am. J. Respir. Crit. Care Med. 2020, 202, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Clinical Study of the RheOx Bronchial Rheoplasty System in Treating the Symptoms of Chronic Bronchitis. Available online: https://clinicaltrials.gov/ct2/show/NCT04677465?term=rhesolve&draw=2&rank=1 (accessed on 25 February 2023).
- Karakoca, Y.; Gogus, G.K.; Yapicier, O. Use of Resector Balloon Desobstruction in Patients With Severe Chronic Obstructive Pulmonary Disease: A Pilot Feasibility Study on a Novel Desobstruction Technique. J. Bronchol. Interv. Pulmonol. 2015, 22, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Garner, J.L.; Shaipanich, T.; Hartman, J.E.; Orton, C.M.; Caneja, C.; Klooster, K.; Thornton, J.; Sin, D.D.; Slebos, D.-J.; Shah, P.L. A Prospective Safety and Feasibility Study of Metered CryoSpray (MCS) for Patients with Chronic Bronchitis in COPD. Eur. Respir. J. 2020, 56, 2000556. [Google Scholar] [CrossRef] [PubMed]
- Buttery, S.C.; Lewis, A.; Latimer, L.; Kemp, S.V.; Shah, P.L.; Waller, D.; Lau, K.; Jordan, S.; Greening, N.J.; Rathinam, S.; et al. Comparative Effect of Lung volume reduction surgery for Emphysema and Bronchoscopic lung volume reduction with valve placement: The CELEB trial. Eur. Respir. J. 2022, 60 (Suppl. 66), 4567. [Google Scholar] [CrossRef]

| Trial | Trial Characteristics | Fissure Integrity and Heterogeneity | Follow-Up | FEV1 (mL) Change | FEV1 (%) Change | 6MWT Change | SGRQ Change |
|---|---|---|---|---|---|---|---|
| VENT (2010) | Multicenter prospective RCT (n = 321) | Not specified | 6 months | NR | +16.2% | +7.7% | NR |
| STELVIO (2015) ^ | Prospective RCT (n = 68) | Enrolled CV patients | 6 months 12 months (n = 64) | +140 NR | +17.8% +17% | +74 m +61 m | −14.7 −11 |
| BeLieVer-HiFi (2015) | Single-center, double-blind, sham-controlled RCT (n = 50) | Targeted heterogeneous patients | 3 months | +30 | +5.9% | +22 m | −0.8 * |
| IMPACT ^ (2016) | Prospective multicenter RCT (n = 93) | Targeted homogenous patients | 3 months | +120 | +16.9% | +40 m | −7.6 |
| TRANSFORM ^ (2017) | Prospective multicenter RCT (n = 97) | Targeted heterogenous, CV patients | 3 months | +230 | +29.3% | +78.7 m | −6.5 |
| LIBERATE (2018) | International multicenter RCT (n = 190) | Targeted heterogenous, CV patients | 12 months | +106 | +18% | +39.3 m | −7.05 |
| REACH (2019) | Prospective multicenter unblinded RCT (n = 107) | Targeted heterogenous, CV patients | 3 months | +101 | NR | +19.7 m * | −7.19 * |
| EMPROVE (2019) | International prospective RCT (n = 172) | Targeted heterogenous, CV patients | 6 months | +101 | NR | +6.9 m * | −13 |
| Trial | Duration of Follow-Up | Pneumothorax | COPD Exacerbation | Pneumonia | Respiratory Failure | Device-Related Deaths |
|---|---|---|---|---|---|---|
| VENT (2010) | 90 days | 4.2% | 9.3% | 3.3% | 1.4% | <1% |
| STELVIO (2015) | 6 months | 18% | 12% | 6% | NR | 1.5% |
| BeLieVer-HiFi (2015) | 90 days | 8% | 64% | 8% | NR | 8% |
| IMPACT (2016) | 3 months | 25.6% | 16.3% | 0 | 2.3% | 0 |
| TRANSFORM (2017) | 30 days | 20% | 4.6% | 4.6% | NR | 0 |
| LIBERATE (2018) | 45 days | 26.6% | 7.8% | <1% | 1.6% | 3.1% |
| REACH (2019) | 3 months | 7.6% | 19.7% | 1.5% | NR | 0 |
| EMPROVE (2019) | 6 months | 32 events * | 16.2% | 8.9% | 2/7% | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
DeMarco, B.; MacRosty, C.R. Bronchoscopic Management of COPD and Advances in Therapy. Life 2023, 13, 1036. https://doi.org/10.3390/life13041036
DeMarco B, MacRosty CR. Bronchoscopic Management of COPD and Advances in Therapy. Life. 2023; 13(4):1036. https://doi.org/10.3390/life13041036
Chicago/Turabian StyleDeMarco, Benjamin, and Christina R. MacRosty. 2023. "Bronchoscopic Management of COPD and Advances in Therapy" Life 13, no. 4: 1036. https://doi.org/10.3390/life13041036
APA StyleDeMarco, B., & MacRosty, C. R. (2023). Bronchoscopic Management of COPD and Advances in Therapy. Life, 13(4), 1036. https://doi.org/10.3390/life13041036






