Development of a Mucoadhesive Liquid Crystal System for the Administration of Rifampicin Applicable in Tuberculosis Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Liquid Crystal Development
2.3. Incorporation of Rifampicin in Liquid Crystal Systems
2.4. Physico-Chemical Characterization of Liquid Crystal Systems
2.4.1. Characterization of Liquid Crystals by Polarized Light Microscopy
2.4.2. Continuous Rheological Analysis
2.4.3. Oscillatory Rheological Analysis
2.4.4. Assessment of Mucoadhesive Forces
2.4.5. X-ray Scattering at Low Angle (SAXS)
2.4.6. Differential Exploratory Calorimetry Analyses
2.5. Cytotoxicity Tests (Agar Diffusion Method)
2.6. Bacterial Killing Kinetics of the RIF Carrying LCS, a Bacterial Inoculum of Mtb
2.7. In Vivo Assay
Mouse Model Infection with Mtb
3. Results
3.1. Liquid Crystal Development
3.2. Incorporation of Rifampicin in Liquid Crystal Systems
3.3. Physico-Chemical Characterization of Liquid Crystal Systems
3.3.1. Characterization of Liquid Crystals by Polarized Light Microscopy
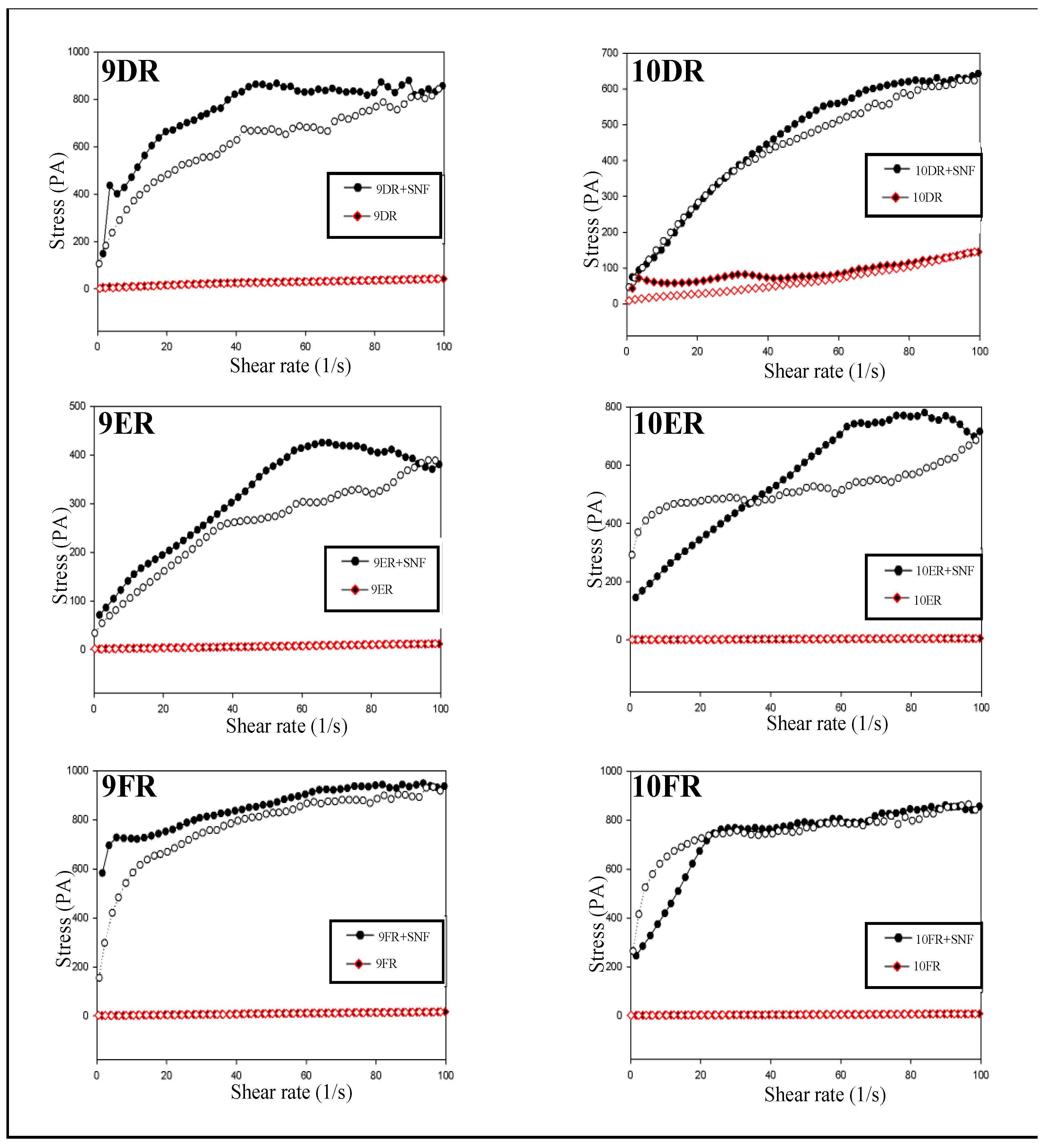
3.3.2. Continuous Rheological Analysis
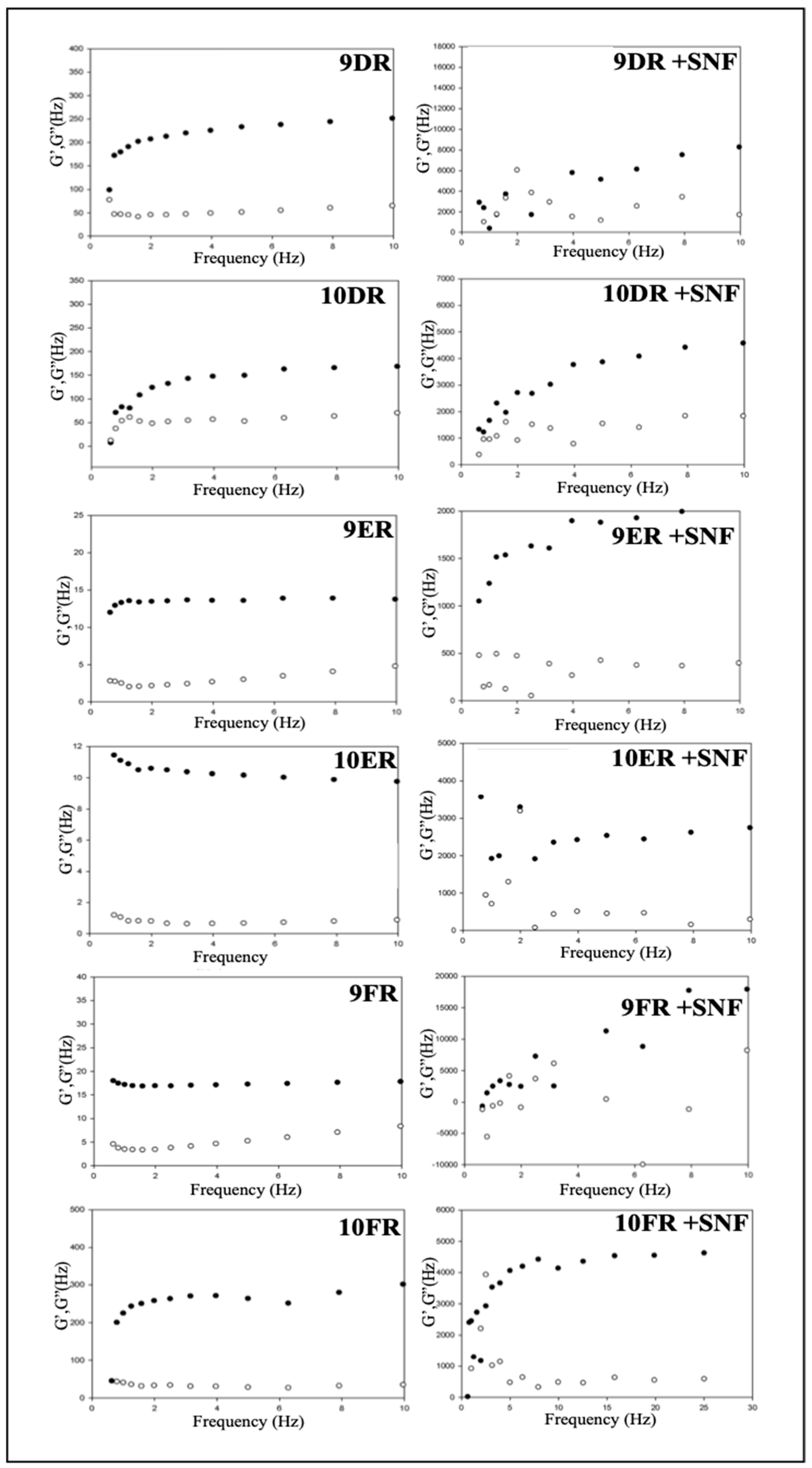
3.3.3. Oscillatory Rheological Analysis
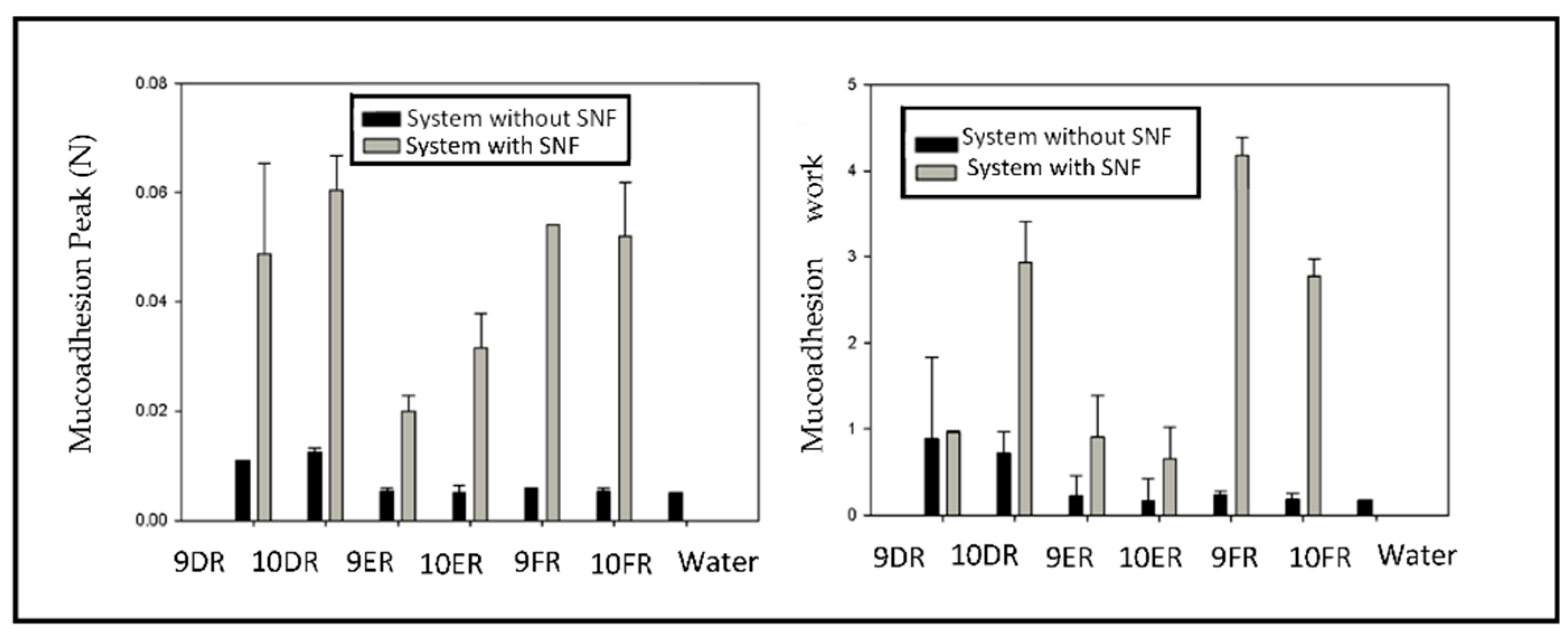
3.3.4. Assessment of Mucoadhesive Forces
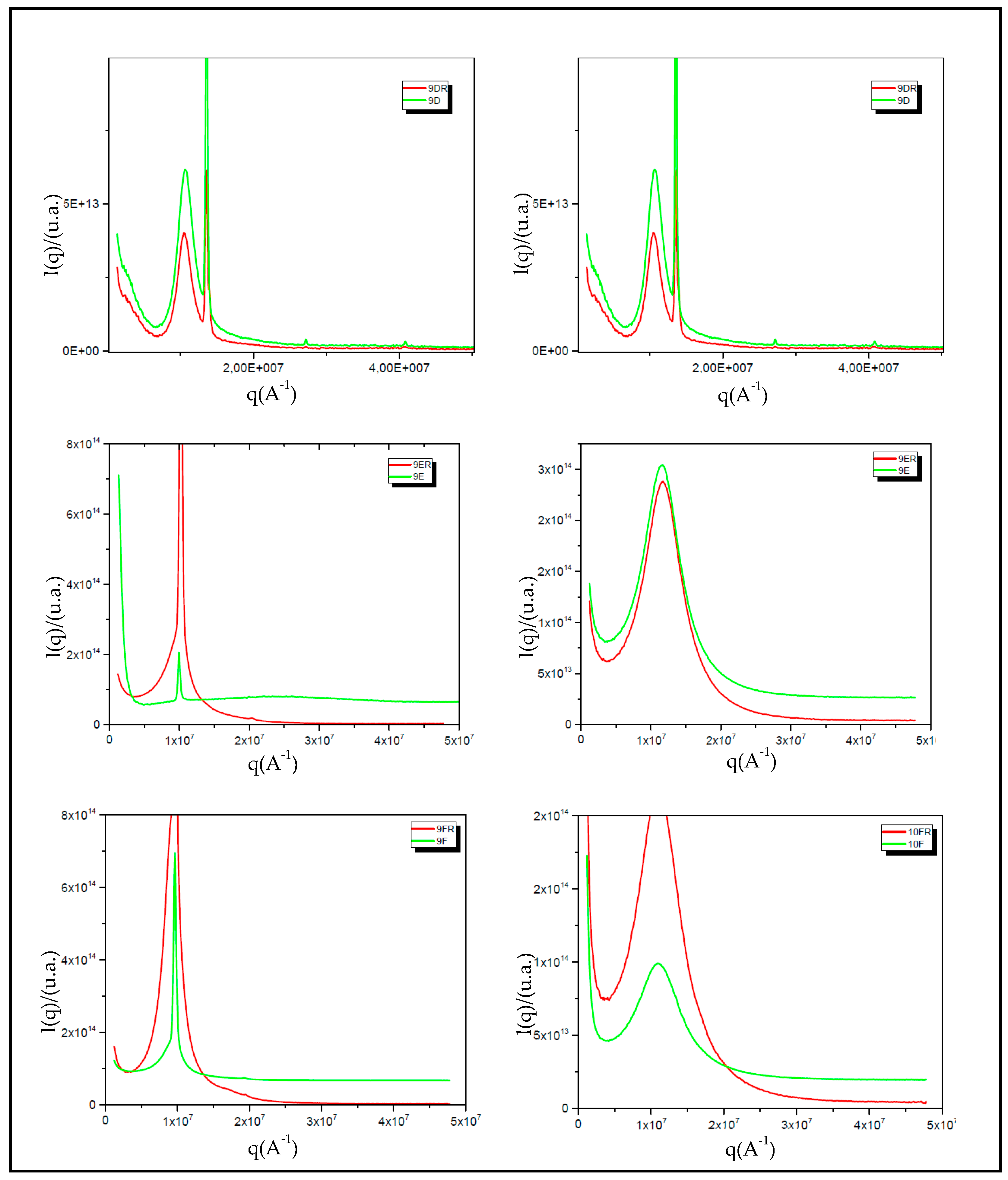
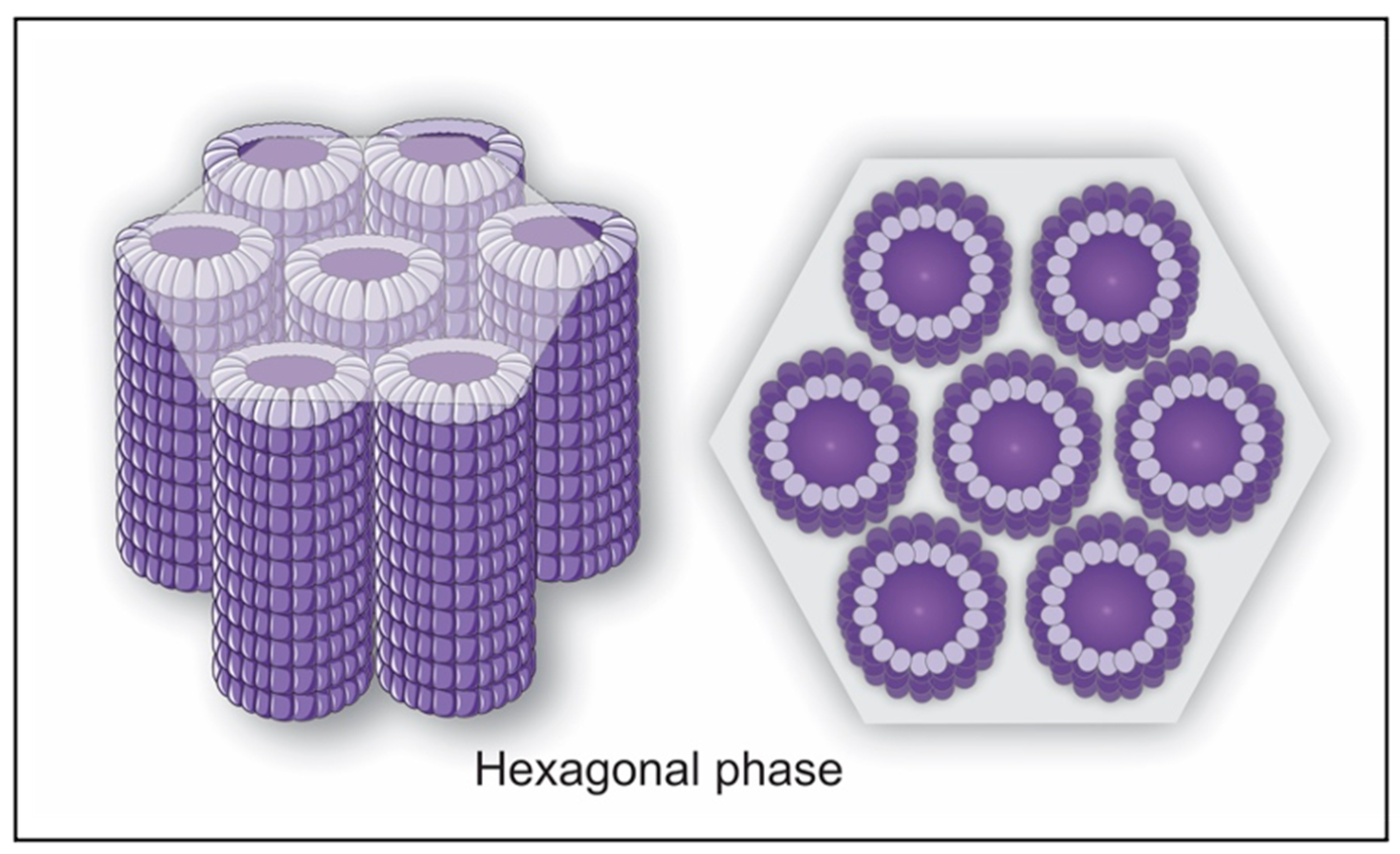
3.3.5. X-ray Scattering at Low Angle (SAXS)
3.3.6. Differential Exploratory Calorimetry Analyses (DSC)
3.4. Cytoxicity Tests (Agar Diffusion Method)
3.5. Bacterial Kinetics
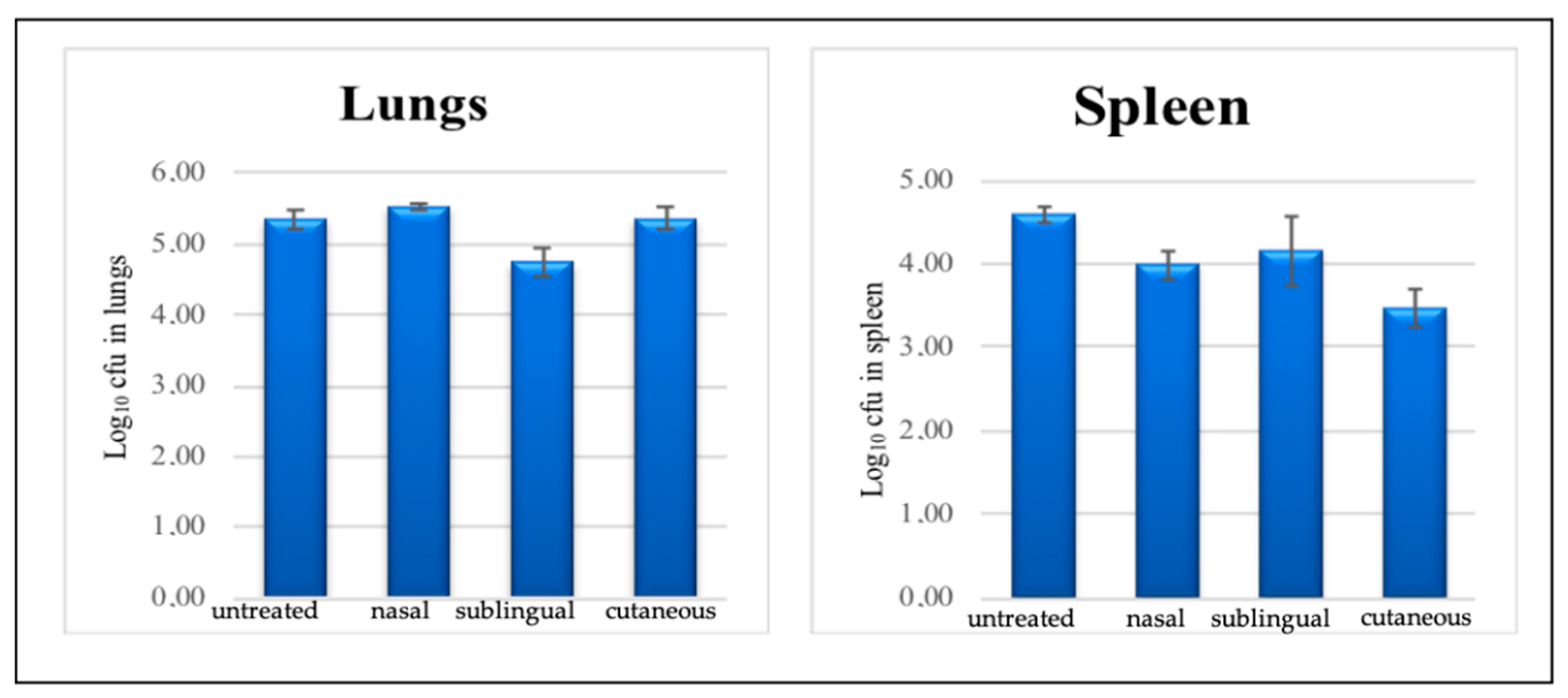
3.6. In Vivo Assay
Mouse Model Infection with M. tuberculosis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Word Health Organization. 2021, Volume 1, p. 1. Available online: https://www.who.int/ (accessed on 15 February 2022).
- Ahmed, M.; Ramadan, W.; Rambhu, D.; Shakeel, F. Potential of nanoemulsions for intravenous delivery of rifampicin. Pharmazie 2008, 63, 806–811. [Google Scholar] [PubMed]
- Chokshi, N.V.; Khatri, H.N.; Patel, M.M. Formulation, optimization, and characterization of rifampicin-loaded solid lipid nanoparticles for the treatment of tuberculosis. Drug Dev. Ind. Pharm. 2018, 44, 1975–1989. [Google Scholar] [CrossRef]
- Floss, H.G.; Yu, T.W. Rifamycin—Mode of action, resistance, and biosynthesis. Chem. Rev. 2005, 105, 621–632. [Google Scholar] [CrossRef]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mainardes, R.M. Liposomes and Micro/Nanoparticles as Colloidal Carriers for Nasal Drug Delivery. Curr. Drug Deliv. 2006, 3, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Makai, M.; Csányi, E.; Németh, Z.; Pálinkás, J.; Eros, I. Structure and drug release of lamellar liquid crystals containing glycerol. Int. J. Pharm. 2003, 256, 95–107. [Google Scholar] [CrossRef]
- Carvalho, F.C. Surfactant systems for nasal zidovudine delivery: Structural, rheological and mucoadhesive properties. J. Pharm. Pharmacol. 2010, 62, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Singh, S. Phase transitions in liquid crystals. Phys. Rep. 2000, 324, 107–269. [Google Scholar] [CrossRef]
- Laffleur, F. Mucoadhesive polymers for buccal drug delivery. Drug Dev. Ind. Pharm. 2014, 40, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Zahir-Jouzdani, F.; Wolf, J.D.; Atyabi, F.; Bernkop-Schnürch, A. In situ gelling and mucoadhesive polymers: Why do they need each other? Expert Opin. Drug Deliv. 2018, 15, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- David, S.R. Development and evaluation of curcumin liquid crystal systems for cervical cancer. Sci. Pharm. 2020, 88, 15. [Google Scholar] [CrossRef] [Green Version]
- Calixto, G.M.F.; Victorelli, F.D.; Dovigo, L.N.; Chorilli, M. Polyethyleneimine and Chitosan Polymer-Based Mucoadhesive Liquid Crystalline Systems Intended for Buccal Drug Delivery. AAPS Pharm. Sci.Tech. 2018, 19, 820–836. [Google Scholar] [CrossRef] [Green Version]
- Nalone, L.; Marques, C.; Costa, S.; Souto, E.B.; Severino, P. Liquid crystalline drug delivery systems. Drug Deliv. Trends 2020, 3, 141–149. [Google Scholar] [CrossRef]
- Venugopal, J. Continuous Nanostructures for the Controlled Release of Drugs. Curr. Pharm. Des. 2009, 15, 1799–1808. [Google Scholar] [CrossRef] [PubMed]
- Sturegård, E. Little difference between minimum inhibitory concentrations of Mycobacterium tuberculosis wild-type organisms determined with BACTEC MGIT 960 and Middlebrook 7H10. Clin. Microbiol. Infect. 2015, 21, 148.e5–148.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosevear, F.B. The microscopy of the liquid crystalline neat and middle phases of soaps and synthetic detergents. J. Am. Oil Chem. Soc. 1954, 31, 628–639. [Google Scholar] [CrossRef]
- Eccleston, G.M.; Beattie, L. Microstructural changes during the storage of systems containing cetostearyl alcohol/polyoxyethylene alkyl ether surfactants. Drug Dev. Ind. Pharm. 1988, 14, 2499–2518. [Google Scholar] [CrossRef]
- Li, Y. In situ phase transition of microemulsions for parenteral injection yielding lyotropic liquid crystalline carriers of the antitumor drug bufalin. Colloids Surf. B Biointerfaces 2019, 173, 217–225. [Google Scholar] [CrossRef] [Green Version]
- Bhowmik, P.K. Thermotropic liquid-crystalline properties of viologens containing 4-n-alkylbenzenesulfonates. Crystals 2019, 9, 77. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Zhou, J.; Xu, Y. Study of the in vitro cytotoxicity testing of medical devices. Biomed. Rep. 2015, 3, 617–620. [Google Scholar] [CrossRef] [Green Version]
- Patil, S.S.; Mahadik, K.R.; Paradkar, A.R. Liquid crystalline phase as a probe for crystal engineering of lactose: Carrier for pulmonary drug delivery. Eur. J. Pharm. Sci. 2015, 68, 43–50. [Google Scholar] [CrossRef]
- Jones, D.S.; Brown, A.F.; Woolfson, A.D. Rheological characterization of bioadhesive, antimicrobial, semisolids designed for the treatment of periodontal diseases: Transient and dynamic viscoelastic and continuous shear analysis. J. Pharm. Sci. 2001, 90, 1978–1990. [Google Scholar] [CrossRef]
- Metzner, A.B.; Prilutski, G.M. Rheological Properties of Polymeric Liquid Crystals. J. Rheol. 1986, 30, 661–691. [Google Scholar] [CrossRef]
- Savic, S. An alkyl polyglucoside-mixed emulsifier as stabilizer of emulsion systems: The influence of colloidal structure on emulsions skin hydration potential. J. Colloid Interface Sci. 2011, 358, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Fresno, M.J.C.; Ramírez, A.D.; Jiménez, M.M. Systematic study of the flow behaviour and mechanical properties of Carbopol® UltrezTM 10 hydroalcoholic gels. Eur. J. Pharm. Biopharm. 2002, 54, 329–335. [Google Scholar] [CrossRef]
- Unsal, E.; Broens, M.; Armstrong, R.T. Pore Scale Dynamics of Microemulsion Formation. Langmuir 2016, 32, 7096–7108. [Google Scholar] [CrossRef] [PubMed]
- Tse-Ve-Koon, K.; Tremblay, N.; Constantin, D.; Freyssingeas, É. Structure, thermodynamics and dynamics of the isotropic phase of spherical non-ionic surfactant micelles. J. Colloid Interface Sci. 2013, 393, 161–1730. [Google Scholar] [CrossRef]
- Zabara, A.; Mezzenga, R. Controlling molecular transport and sustained drug release in lipid-based liquid crystalline mesophases. J. Control. Release 2014, 188, 31–43. [Google Scholar] [CrossRef]
- Saxena, A.; Kaloti, M.; Bohidar, H.B. Rheological properties of binary and ternary protein-polysaccharide co-hydrogels and comparative release kinetics of salbutamol sulphate from their matrices. Int. J. Biol. Macromol. 2011, 48, 263–270. [Google Scholar] [CrossRef]
- Carvalho, F.C.; Bruschi, M.L.; Evangelista, R.C.; Gremião, M.P.D. Mucoadhesive drug delivery systems. Braz. J. Pharm. Sci. 2010, 46, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Holmqvist, P.; Alexandridis, P.; Lindman, B. Modification of the microstructure in poloxamer Block copolymer-water-"oil" systems by varying the ‘oil’ type. Macromolecules 1997, 30, 6788–6797. [Google Scholar] [CrossRef]
- Osorio, R.M. Cytotoxicity of endodontic materials. J. Endod. 1998, 24, 91–96. [Google Scholar] [CrossRef]
- Farkas, E.; Kiss, D.; Zelkó, R. Study on the release of chlorhexidine base and salts from different liquid crystalline structures. Int. J. Pharm. 2007, 340, 71–75. [Google Scholar] [CrossRef]
- Oyafuso, M.H.; Carvalho, F.C.; Chiavacci, L.A.; Gremião, M.P.D.; Chorilli, M. Design and characterization of silicone and surfactant based systems for topical drug delivery. J. Nanosci. Nanotechnol. 2015, 15, 817–826. [Google Scholar] [CrossRef]
- Henriksen, J.R.; Andresen, T.L.; Feldborg, L.N.; Duelund, L.; Ipsen, J.H. Understanding detergent effects on lipid membranes: A model study of lysolipids. Biophys. J. 2010, 98, 2199–2205. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N. Pharmacokinetics and dose response of anti-TB drugs in rat infection model of tuberculosis. Tuberculosis 2014, 94, 282–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shim, C.K.; Lee, J.U. Comparative study of rifampicin pharmacokinetics administered orally and intravenously in the fasted and non-fasted rats. Arch. Pharmacal Res. 1985, 8, 177–186. [Google Scholar] [CrossRef]
- Ugwoke, M.I.; Agu, R.U.; Verbeke, N.; Kinget, R. Nasal mucoadhesive drug delivery: Background, applications, trends and future perspectives. Adv. Drug Deliv. Rev. 2005, 57, 1640–1665. [Google Scholar] [CrossRef]
- Thorne, R.G.; Pronk, G.J.; Padmanabhan, V.; Frey, W.H. Delivery of insulin-like growth factor-I to the rat brain and spinal cord along olfactory and trigeminal pathways following intranasal administration. Neuroscience 2004, 127, 481–496. [Google Scholar] [CrossRef]




| Grade | Cytotoxicity | Halo Size |
|---|---|---|
| 0 | Absence | Absence of discoloration around of sample |
| 1 | Light | Discoloration zone limited to the area under the sample |
| 2 | Mild | Discoloration zone around the sample up to 0.5 cm |
| 3 | Moderate | Discoloration zone from 0.5 to 1.0 cm around the sample |
| 4 | Severe | Discoloration zone greater than 1.0 cm |
| System | Composition | Aspect | |||||
|---|---|---|---|---|---|---|---|
| PPD (%) | Oleilamine (%) | PPD: Oleilamine 1:1 (w:w) (%) | Water (%) | Procetyl® AWS (%) | |||
| 9D | 30 | _ | _ | 20 | 50 | STVI | L. crystal |
| 10D | 40 | _ | _ | 10 | 50 | STVI | L. crystal |
| 9E | _ | 30 | _ | 20 | 50 | STBV | L. crystal |
| 10E | _ | 40 | _ | 10 | 50 | STBV | Precursor liquid crystal system |
| 9F | _ | _ | 30 | 20 | 50 | SOVI | Precursor liquid crystal system |
| 10F | _ | _ | 40 | 10 | 50 | SOBV | L. crystal |
| System | Composition | |||||
|---|---|---|---|---|---|---|
| PPD (%) | Oleilamine (%) | PPD: Oleilamine 1:1 (w:w) (%) | Water (%) | Procetyl® AWS (%) | Rifampicim (µg·mL−1) | |
| 9DR | 30 | _ | _ | 20 | 50 | 0.150 |
| 10DR | 40 | _ | _ | 10 | 50 | 0.150 |
| 9ER | _ | 30 | _ | 20 | 50 | 0.150 |
| 10ER | _ | 40 | _ | 10 | 50 | 0.150 |
| 9FR | _ | _ | 30 | 20 | 50 | 0.150 |
| 10FR | _ | _ | 40 | 10 | 50 | 0.150 |
| System | n | k | |
|---|---|---|---|
| Liquid Crystal System | 9D | 1.95 | 0.059 |
| 10D | 0.958 | 1.832 | |
| 9E | 0.917 | 0.185 | |
| 10E | 0.901 | 0.077 | |
| 9F | 0.787 | 0.434 | |
| 10F | 0.177 | 3.337 | |
| Liquid Crystal System with rifampicin | 9DR | 0.540 | 3.452 |
| 10DR | 0.382 | 20.824 | |
| 9ER | 0.758 | 0.354 | |
| 10ER | 0.925 | 0.069 | |
| 9FR | 0.829 | 0.337 | |
| 10FR | 0.697 | 0.264 | |
| Liquid Crystal System + SNF | 9D | 0.145 | 703.144 |
| 10D | 0.509 | 83.275 | |
| 9E | 0.415 | 55.683 | |
| 10E | 0.745 | 26.151 | |
| 9F | 0.486 | 77.446 | |
| 10F | 0.470 | 129.193 | |
| Liquid Crystal System with rifampicin + SNF | 9DR | 0.229 | 319.585 |
| 10DR | 0.533 | 59.849 | |
| 9ER | 0.445 | 58.022 | |
| 10ER | 0.491 | 86.391 | |
| 9FR | 0.119 | 547.658 | |
| 10FR | 0.245 | 292.803 | |
| System | R | S | n | |
|---|---|---|---|---|
| Initial System (IS) | 9D | 0.987 | 164.282 | 0.155 |
| 10D | 0.982 | 853.439 | 0.116 | |
| 9E | 0.624 | 14.854 | 0.022 | |
| 10E | 0 | 9.791 | 8 × 10−13 | |
| 9F | 0.470 | 117.978 | 0.025 | |
| 10F | 0 | 330.185 | 1.3 × 10−11 | |
| IS + Rifampicin | 9DR | 0.954 | 176.103 | 0.155 |
| 10DR | 0.957 | 86.734 | 0.292 | |
| 9ER | 0.862 | 12.844 | 0.045 | |
| 10ER | 0 | 9.922 | 6 × 10−3 | |
| 9FR | 0.801 | 16.532 | 0.053 | |
| 10FR | 0.867 | 205.799 | 0.157 | |
| Initial System (IS) + SNF | 9D | 0.669 | 5970.146 | 0.232 |
| 10D | 0.902 | 3050.039 | 0.170 | |
| 9E | 0.841 | 914.508 | 0.113 | |
| 10E | 0.819 | 910.679 | 0.134 | |
| 9F | 0.743 | 1575.645 | 0.107 | |
| 10F | 0.691 | 2003.491 | 0.059 | |
| SI + Rifampicin + SNF | 9DR | 0.454 | 3468.944 | 0.242 |
| 10DR | 0.725 | 2468.397 | 0.181 | |
| 9ER | 0.949 | 58.022 | 0.445 | |
| 10ER | 0.517 | 1947.099 | 0.132 | |
| 9FR | 0.091 | 8295.673 | 0.062 | |
| 10FR | 0.662 | 2380.354 | 0.174 | |
| System | qmax1 | qmax2 | qmax3 | d2/d1 | d3/d1 | Structure | |
|---|---|---|---|---|---|---|---|
| Initial System (IS) | 9D | 13,502,052.0 | 27,209,302.3 | 40,779,753.8 | 2 | 3 | Hexagonal |
| 10D | 13,577,291.0 | 21,767,587.8 | 43,661,650.7 | 2 | 3 | Hexagonal | |
| 9E | 9,913,284.3 | 19,887,965.9 | - | 2 | Lamellar | ||
| 10E | 11,524,167.8 | - | - | Microemulsion | |||
| 9F | 9,610,123.12 | 19,288,645.7 | - | 2 | Lamellar | ||
| 10F | 10,897,172.8 | - | - | Microemulsion | |||
| IS + Rifampicin | 9DR | 13,555,631.6 | 27,173,962.6 | 41,005,472.0 | 2 | 3 | Hexagonal |
| 10DR | 13,578,263.8 | 27,088,719.0 | 43,661,650.7 | 2 | 3 | Hexagonal | |
| 9ER | 10,257,637.9 | 20,251,938.0 | - | 2 | Lamellar | ||
| 10ER | 11,737,346.1 | - | - | Microemulsion | |||
| 9FR | 9,664,842.6 | 19,418,604.7 | - | 2 | Lamellar | ||
| 10FR | 10,997,492 | - | - | Microemulsion | |||
| System | Halo (mm) | Cytotoxicity |
|---|---|---|
| 9D | 8.7 | Moderate |
| 10D | 8.8 | Moderate |
| 9E | 12 | Moderate |
| 10E | 7.7 | Moderate |
| 9F | 6.7 | Moderate |
| 10F | 5.9 | Moderate |
| 9DR | 5 | Light |
| 10DR | 5.8 | Moderate |
| 9ER | 8.5 | Moderate |
| 10ER | 7.4 | Moderate |
| 9FR | 6.8 | Moderate |
| 10FR | 6.6 | Moderate |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, K.P.; Rodero, C.F.; Ribeiro, C.M.; Gremião, M.P.D.; Peccinini, R.G.; Pavan, F.R.; Pearce, C.; Gonzalez-Juarrero, M.; Chorilli, M. Development of a Mucoadhesive Liquid Crystal System for the Administration of Rifampicin Applicable in Tuberculosis Therapy. Life 2022, 12, 1138. https://doi.org/10.3390/life12081138
Santos KP, Rodero CF, Ribeiro CM, Gremião MPD, Peccinini RG, Pavan FR, Pearce C, Gonzalez-Juarrero M, Chorilli M. Development of a Mucoadhesive Liquid Crystal System for the Administration of Rifampicin Applicable in Tuberculosis Therapy. Life. 2022; 12(8):1138. https://doi.org/10.3390/life12081138
Chicago/Turabian StyleSantos, Kaio Pini, Camila Fernanda Rodero, Camila Maríngolo Ribeiro, Maria P. D. Gremião, Rosângela Gonçalves Peccinini, Fernando Rogerio Pavan, Camron Pearce, Mercedes Gonzalez-Juarrero, and Marlus Chorilli. 2022. "Development of a Mucoadhesive Liquid Crystal System for the Administration of Rifampicin Applicable in Tuberculosis Therapy" Life 12, no. 8: 1138. https://doi.org/10.3390/life12081138
APA StyleSantos, K. P., Rodero, C. F., Ribeiro, C. M., Gremião, M. P. D., Peccinini, R. G., Pavan, F. R., Pearce, C., Gonzalez-Juarrero, M., & Chorilli, M. (2022). Development of a Mucoadhesive Liquid Crystal System for the Administration of Rifampicin Applicable in Tuberculosis Therapy. Life, 12(8), 1138. https://doi.org/10.3390/life12081138








