The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Rearing Conditions and Diet Preparation
2.2. Growth Performance and Feed Utilization Indices
2.3. Blood Sampling
2.4. Hematological Analysis
2.5. Determination of Phagocytic Activity
2.6. Biochemical Assays
2.7. Determination of Malondialdehyde (MDA) and Antioxidants
2.8. Determination of Digestive Enzymes Activity
2.9. Gene Expression
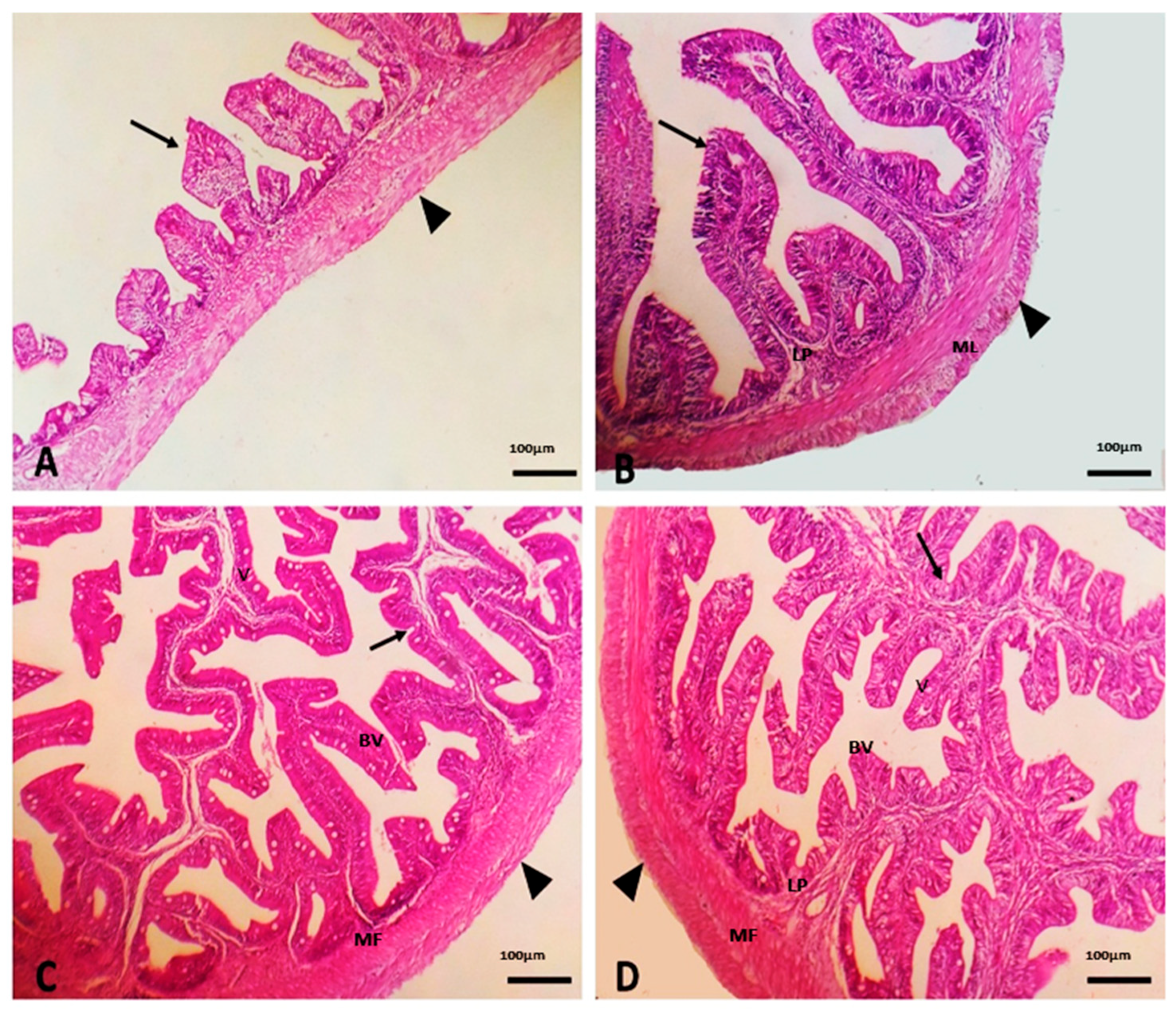
2.10. Morphometry of Intestinal Villi
2.11. Experimental Infection
2.12. Statistical Model and Analysis Procedure
3. Results
3.1. Effect of Dietary SC on Growth Performance
3.2. Effect of Dietary SC on Whole Body and Muscle Composition
3.3. Effect of Dietary SC on Intestinal Morphology
3.4. Effect of Dietary SC on Hematological and Immunological Variables
3.5. Effect of Dietary SC on Circulating Biochemical Parameters
3.6. Effect of Dietary SC on Redox Status
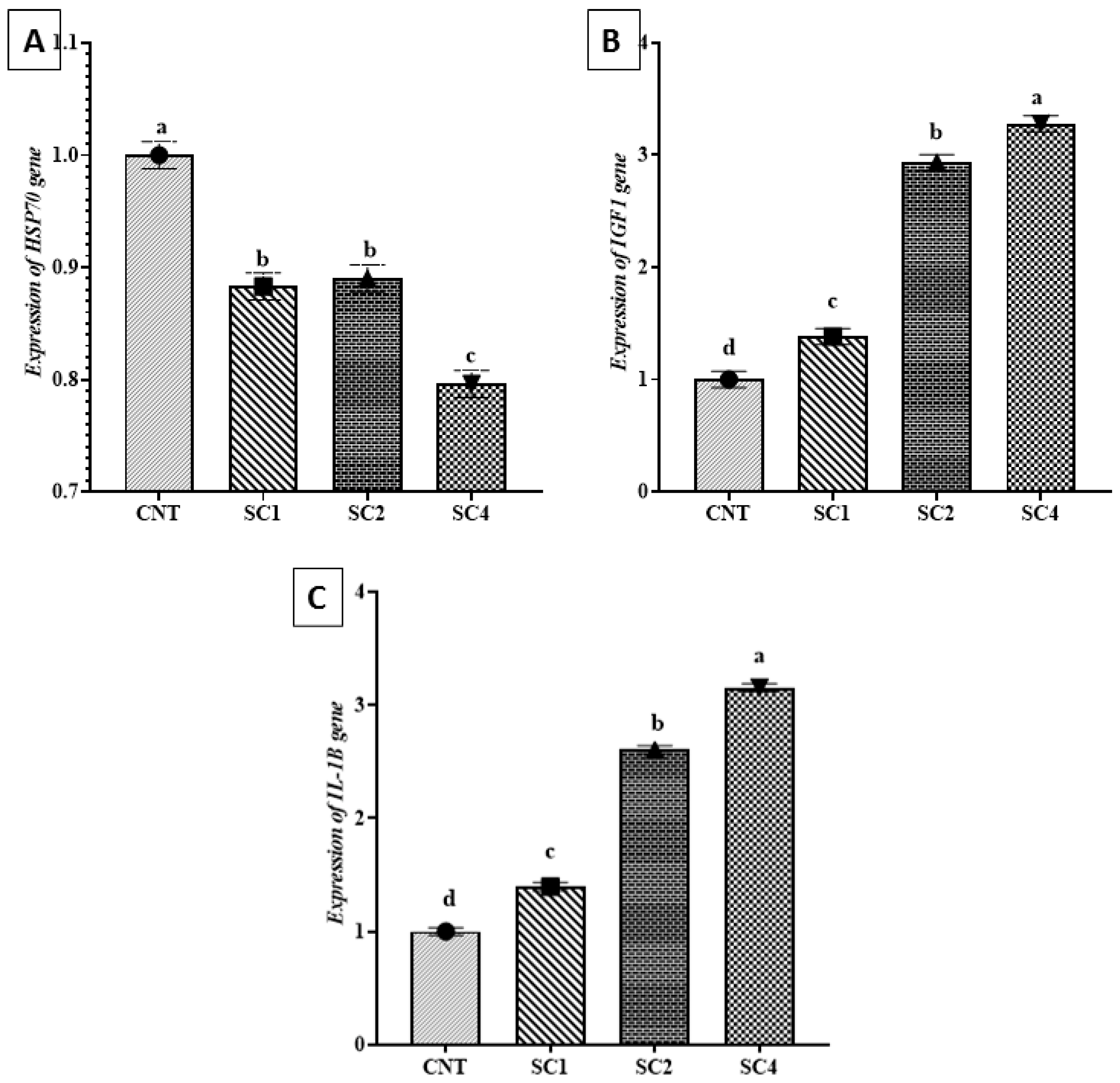
3.7. Effect of Dietary SC on HSP70, IGF1, and IL-1β Expression
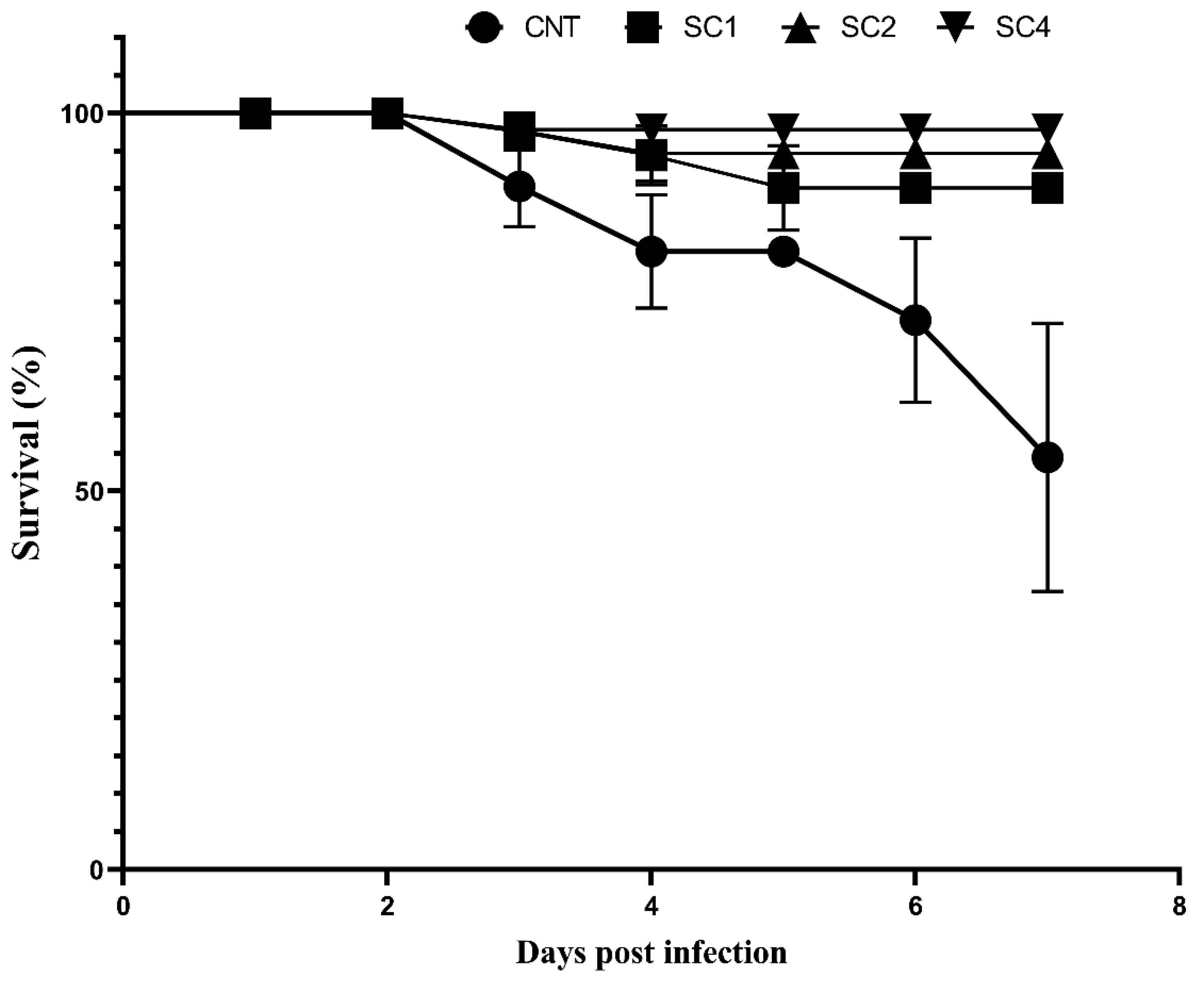
3.8. SC Increased Survival Rate in Sea Bream Challenged with V. parahaemolyticus
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Naiel, M.A.; Negm, S.S.; Abd El-hameed, S.A.; Abdel-Latif, H.M. Dietary organic selenium improves growth, serum biochemical indices, immune responses, antioxidative capacity, and modulates transcription of stress-related genes in Nile tilapia reared under sub-optimal temperature. J. Therm. Biol. 2021, 99, 102999. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, M.; Cui, J.; Du, Y.; Teng, X.; Zhang, Z. Heat shock proteins took part in oxidative stress-mediated inflammatory injury via NF-κB pathway in excess manganese-treated chicken livers. Ecotoxicol. Environ. Saf. 2021, 226, 112833. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Miao, Z.; Teng, X.; Xu, S. Chlorpyrifos triggers epithelioma papulosum cyprini cell pyroptosis via miR-124-3p/CAPN1 axis. J. Hazard. Mater. 2022, 424, 127318. [Google Scholar] [CrossRef] [PubMed]
- Miao, Z.; Zhang, K.; Bao, R.; Li, J.; Tang, Y.; Teng, X. Th1/Th2 imbalance and heat shock protein mediated inflammatory damage triggered by manganese via activating NF-κB pathway in chicken nervous system in vivo and in vitro. Environ. Sci. Pollut. Res. 2021, 28, 44361–44373. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, Y.; Teng, X.; Luan, P.; Teng, X.; Yin, X. Immunosuppression participated in complement activation-mediated inflammatory injury caused by 4-octylphenol via TLR7/IκBα/NF-κB pathway in common carp (Cyprinus carpio) gills. Aquat. Toxicol. 2022, 249, 106211. [Google Scholar] [CrossRef]
- Naiel, M.A.; Ismael, N.E.; Abd El-hameed, S.A.; Amer, M.S. The antioxidative and immunity roles of chitosan nanoparticle and vitamin C-supplemented diets against imidacloprid toxicity on Oreochromis niloticus. Aquaculture 2020, 523, 735219. [Google Scholar] [CrossRef]
- Naiel, M.E.; Shehata, A.M.; El-kholy, A.I.; El-Naggar, K.; Farag, M.R.; Alagawany, M. The mitigating role of probiotics against the adverse effects of suboptimal temperature in farmed fish: A review. Aquaculture 2022, 550, 737877. [Google Scholar] [CrossRef]
- Luis Balcázar, J.; Decamp, O.; Vendrell, D.; De Blas, I.; Ruiz-Zarzuela, I. Health and nutritional properties of probiotics in fish and shellfish. Microb. Ecol. Health Dis. 2006, 18, 65–70. [Google Scholar] [CrossRef]
- Nayak, S.K. Probiotics and immunity: A fish perspective. Fish Shellfish Immunol. 2010, 29, 2–14. [Google Scholar] [CrossRef]
- Abdel-Latif, H.M.; El-Ashram, S.; Yilmaz, S.; Naiel, M.A.; Kari, Z.A.; Hamid, N.K.A.; Dawood, M.A.; Nowosad, J.; Kucharczyk, D. The effectiveness of Arthrospira platensis and microalgae in relieving stressful conditions affecting finfish and shellfish species: An overview. Aquac. Rep. 2022, 24, 101135. [Google Scholar] [CrossRef]
- Abu-Elala, N.; Marzouk, M.; Moustafa, M. Use of different (Saccharomyces cerevisiae) biotic forms as immune-modulator and growth promoter for (Oreochromis niloticus) challenged with some fish pathogens. Int. J. Vet. Sci. Med. 2013, 1, 21–29. [Google Scholar] [CrossRef] [Green Version]
- Abu-Elala, N.M.; Younis, N.A.; AbuBakr, H.O.; Ragaa, N.M.; Borges, L.L.; Bonato, M.A. Efficacy of dietary yeast cell wall supplementation on the nutrition and immune response of Nile tilapia. Egypt. J. Aquat. Res. 2018, 44, 333–341. [Google Scholar] [CrossRef]
- Oliva-Teles, A.; Gonçalves, P. Partial replacement of fishmeal by brewers yeast (Saccaromyces cerevisae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 2001, 202, 269–278. [Google Scholar] [CrossRef]
- Korni, F.M.; Sleim, A.S.A.; Abdellatief, J.I.; Abd-elaziz, R.A. Prevention of vibriosis in sea bass, Dicentrarchus labrax using ginger nanoparticles and Saccharomyces cerevisiae. J. Fish Pathol. 2021, 34, 185–199. [Google Scholar]
- Waché, Y.; Auffray, F.; Gatesoupe, F.-J.; Zambonino, J.; Gayet, V.; Labbé, L.; Quentel, C. Cross effects of the strain of dietary Saccharomyces cerevisiae and rearing conditions on the onset of intestinal microbiota and digestive enzymes in rainbow trout, Onchorhynchus mykiss, fry. Aquaculture 2006, 258, 470–478. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Tawwab, M.; Abdel-Rahman, A.M.; Ismael, N.E. Evaluation of commercial live bakers’ yeast, Saccharomyces cerevisiae as a growth and immunity promoter for Fry Nile tilapia, Oreochromis niloticus (L.) challenged in situ with Aeromonas hydrophila. Aquaculture 2008, 280, 185–189. [Google Scholar] [CrossRef]
- Abdel-Tawwab, M. Interactive effects of dietary protein and live bakery yeast, Saccharomyces cerevisiae on growth performance of Nile tilapia, Oreochromis niloticus (L.) fry and their challenge against Aeromonas hydrophila infection. Aquac. Int. 2012, 20, 317–331. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Koshio, S.; Ishikawa, M.; Yokoyama, S.; El Basuini, M.F.; Hossain, M.S.; Nhu, T.H.; Moss, A.S.; Dossou, S.; Wei, H. Dietary supplementation of β-glucan improves growth performance, the innate immune response and stress resistance of red sea bream,(Pagrus major). Aquac. Nutr. 2015, 23, 148–159. [Google Scholar] [CrossRef]
- Li, P.; Lawrence, A.L.; Castille, F.L.; Gatlin, D.M. Preliminary evaluation of a purified nucleotide mixture as a dietary supplement for Pacific white shrimp Litopenaeus vannamei (Boone). Aquac. Res. 2007, 38, 887–890. [Google Scholar] [CrossRef]
- Esteban, M. Glucan receptor but not mannose receptor is involved in the phagocytosis of Saccharomyces cerevisiae by seabream (Sparus aurata L.) blood leucocytes. Fish Shellfish Immunol. 2004, 16, 447–451. [Google Scholar] [CrossRef]
- Staykov, Y.; Spring, P.; Denev, S.; Sweetman, J. Effect of a mannan oligosaccharide on the growth performance and immune status of rainbow trout (Oncorhynchus mykiss). Aquac. Int. 2007, 15, 153–161. [Google Scholar] [CrossRef]
- Dimitroglou, A.; Davies, S.; Sweetman, J. The effect of dietary mannan oligosaccharides on the intestinal histology of rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 150, S63. [Google Scholar] [CrossRef]
- Misra, C.K.; Das, B.K.; Mukherjee, S.C.; Pattnaik, P. Effect of long term administration of dietary β-glucan on immunity, growth and survival of Labeo rohita fingerlings. Aquaculture 2006, 255, 82–94. [Google Scholar] [CrossRef]
- Barros, M.M.; Falcon, D.R.; de Oliveira Orsi, R.; Pezzato, L.E.; Fernandes, A.C., Jr.; Guimarães, I.G.; Fernandes, A., Jr.; Padovani, C.R.; Sartori, M.M.P. Non-specific immune parameters and physiological response of Nile tilapia fed β-glucan and vitamin C for different periods and submitted to stress and bacterial challenge. Fish Shellfish Immunol. 2014, 39, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Dimitroglou, A.; Merrifield, D.L.; Spring, P.; Sweetman, J.; Moate, R.; Davies, S.J. Effects of mannan oligosaccharide (MOS) supplementation on growth performance, feed utilisation, intestinal histology and gut microbiota of gilthead sea bream (Sparus aurata). Aquaculture 2010, 300, 182–188. [Google Scholar] [CrossRef]
- Welker, T.L.; Lim, C.; Yildirim-Aksoy, M.; Shelby, R.; Klesius, P.H. Immune Response and Resistance to Stress and Edwardsiella ictaluri Challenge in Channel Catfish, Ictalurus punctatus, Fed Diets Containing Commercial Whole-Cell Yeast or Yeast Subcomponents. J. World Aquac. Soc. 2007, 38, 24–35. [Google Scholar] [CrossRef]
- Izquierdo, M.; Turkmen, S.; Montero, D.; Zamorano, M.; Afonso, J.; Karalazos, V.; Fernández-Palacios, H. Nutritional programming through broodstock diets to improve utilization of very low fishmeal and fish oil diets in gilthead sea bream. Aquaculture 2015, 449, 18–26. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemists (AOAC). In Van Nostrand’s Encyclopedia of Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar] [CrossRef]
- Dielbandhoesing, S.; Zhang, H.; Caro, L.; Van Der Vaart, J.; Klis, F.; Verrips, C.; Brul, S. Specific cell wall proteins confer resistance to nisin upon yeast cells. Appl. Environ. Microbiol. 1998, 64, 4047–4052. [Google Scholar] [CrossRef] [Green Version]
- Faggio, C.; Piccione, G.; Marafioti, S.; Arfuso, F.; Trischitta, F.; Fortino, G.; Fazio, F. Monthly variations of haematological parameters of Sparus aurata and Dicentrarchus labrax reared in Mediterranean land off-shore tanks. Cah. Biol. Mar. 2014, 55, 437–443. [Google Scholar]
- Feldman, B.F.; Zinkl, J.G.; Jain, N.C. Schalm’s Veterinary Hematology; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000. [Google Scholar]
- Stoskopf, M. Shark Pharmacology and Toxicology. In Fish Medicine; WB Saunders Company: Philadelphia, PA, USA, 1993; pp. 809–839. [Google Scholar]
- Dacie, J.; Lewis, S. Practical Haematology, 8th ed.; Churchill Livinstone, Longman group Ltd.: Edinburgh, UK, 1991; p. 390. [Google Scholar]
- Thrall, M.; Baker, D.; Lassen, E. Veterinary Haematology and Clinical Chemistry; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004. [Google Scholar]
- Reyes-Becerril, M.; López-Medina, T.; Ascencio-Valle, F.; Esteban, M.Á. Immune response of gilthead seabream (Sparus aurata) following experimental infection with Aeromonas hydrophila. Fish Shellfish Immunol. 2011, 31, 564–570. [Google Scholar] [CrossRef]
- Cai, W.-Q.; Li, S.-F.; Ma, J.-Y. Diseases resistance of Nile tilapia (Oreochromis niloticus), blue tilapia (Oreochromis aureus) and their hybrid (female Nile tilapia× male blue tilapia) to Aeromonas sobria. Aquaculture 2004, 229, 79–87. [Google Scholar] [CrossRef]
- Rosenfeld, G. Corante pancromatico para hematologia e citologia humana. Nova combinaçao dos componentes do May-Grünwald e do Giemsa num so corante de emprego rapido. Mem. Inst. Butantan 1947, 20, 328–334. [Google Scholar]
- Nishikimi, M.; Rao, N.A.; Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Aebi, H. [13] Catalase in vitro. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1984; Volume 105, pp. 121–126. [Google Scholar]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Moss, D.; Henderson, A. Digestive enzymes of pancreatic origin. Tietz Textb. Clin. Chem. 1999, 3, 689–708. [Google Scholar]
- Caraway, W. α-amylase colorimetric method. Am. J. Clin. Pathol. 1959, 32, 97–99. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Gewaily, M.S.; Shukry, M.; Abdel-Kader, M.F.; Alkafafy, M.; Farrag, F.A.; Moustafa, E.M.; Doan, H.V.; Abd-Elghany, M.F.; Abdelhamid, A.F.; Dawood, M.A. Dietary Lactobacillus plantarum relieves Nile tilapia (Oreochromis niloticus) juvenile from oxidative stress, immunosuppression and inflammation induced by deltamethrin and Aeromonas hydrophila. Front. Mar. Sci. 2021, 8, 203. [Google Scholar]
- Cruickshank, R.; Duguid, J.; Marmion, B.; Swain, R. Medical Microbiology: The Pratice of Medical Microbiology. In Medical Microbiology: The Pratice of Medical Microbiology; Churchill Livingstone: Edinburgh, UK, 1975; p. 587. [Google Scholar]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Vieira, F.; Buglione, C.; Mourino, J.; Jatobá, A.; Martins, M.; Schleder, D.; Andreatta, E.; Barraco, M.; Vinatea, L. Effect of probiotic supplemented diet on marine shrimp survival after challenge with Vibrio harveyi. Arq. Bras. Med. Veterinária Zootec. 2010, 62, 631–638. [Google Scholar] [CrossRef] [Green Version]
- Das, A.; Nakhro, K.; Chowdhury, S.; Kamilya, D. Effects of potential probiotic Bacillus amyloliquifaciens FPTB16 on systemic and cutaneous mucosal immune responses and disease resistance of catla (Catla catla). Fish Shellfish Immunol. 2013, 35, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Merrifield, D.L.; Bradley, G.; Baker, R.T.M.; Davies, S.J. Probiotic applications for rainbow trout (Oncorhynchus mykiss Walbaum) II. Effects on growth performance, feed utilization, intestinal microbiota and related health criteria postantibiotic treatment. Aquac. Nutr. 2010, 16, 496–503. [Google Scholar] [CrossRef]
- Darafsh, F.; Soltani, M.; Abdolhay, H.; Shamsaei Mehrejan, M. Efficacy of dietary supplementation of Bacillus licheniformis and Bacillus subtilis probiotics and Saccharomyces cerevisiae (yeast) on the hematological, immune response, and biochemical features of Persian sturgeon (Acipenser persicus) fingerlings. Iran. J. Fish. Sci. 2020, 19, 2024–2038. [Google Scholar]
- Ayiku, S.; Shen, J.-F.; Tan, B.-P.; Dong, X.-H.; Liu, H.-Y. Effects of dietary yeast culture on shrimp growth, immune response, intestinal health and disease resistance against Vibrio harveyi. Fish Shellfish Immunol. 2020, 102, 286–295. [Google Scholar] [CrossRef]
- Huang, L.; Ran, C.; He, S.; Ren, P.; Hu, J.; Zhao, X.; Zhou, Z. Effects of dietary Saccharomyces cerevisiae culture or live cells with Bacillus amyloliquefaciens spores on growth performance, gut mucosal morphology, hsp70 gene expression, and disease resistance of juvenile common carp (Cyprinus carpio). Aquaculture 2015, 438, 33–38. [Google Scholar] [CrossRef]
- Lu, J.; Qi, C.; Limbu, S.M.; Han, F.; Yang, L.; Wang, X.; Qin, J.G.; Chen, L. Dietary mannan oligosaccharide (MOS) improves growth performance, antioxidant capacity, non-specific immunity and intestinal histology of juvenile Chinese mitten crabs (Eriocheir sinensis). Aquaculture 2019, 510, 337–346. [Google Scholar] [CrossRef]
- Zhang, R.; Jiang, Y.; Zhou, L.; Chen, Y.; Wen, C.; Liu, W.; Zhou, Y. Effects of dietary yeast extract supplementation on growth, body composition, non-specific immunity, and antioxidant status of Chinese mitten crab (Eriocheir sinensis). Fish Shellfish Immunol. 2019, 86, 1019–1025. [Google Scholar] [CrossRef]
- Jin, M.; Xiong, J.; Zhou, Q.-C.; Yuan, Y.; Wang, X.-X.; Sun, P. Dietary yeast hydrolysate and brewer’s yeast supplementation could enhance growth performance, innate immunity capacity and ammonia nitrogen stress resistance ability of Pacific white shrimp (Litopenaeus vannamei). Fish Shellfish Immunol. 2018, 82, 121–129. [Google Scholar] [CrossRef]
- Chaitanawisuti, N.; Choeychom, C.; Piyatiratitivorakul, S. Effect of dietary supplementation of brewers yeast and nucleotide singularly on growth, survival and vibriosis resistance on juveniles of the gastropod spotted babylon (Babylonia areolata). Aquac. Int. 2010, 19, 489–496. [Google Scholar] [CrossRef]
- Adel, M.; Lazado, C.C.; Safari, R.; Yeganeh, S.; Zorriehzahra, M.J. Aqualase-sup®, a yeast-based in-feed probiotic, modulates intestinal microbiota, immunity and growth of rainbow trout (Oncorhynchus mykiss). Aquac. Res. 2016, 48, 1815–1826. [Google Scholar] [CrossRef]
- Jarmołowicz, S.; ZakĘŚ, Z.; Siwicki, A.; Kowalska, A.; Hopko, M.; GŁĄbski, E.; DEMSKA-ZAKĘŚ, K.; Partyka, K. Effects of brewer’s yeast extract on growth performance and health of juvenile pikeperch Sander lucioperca (L.). Aquac. Nutr. 2012, 18, 457–464. [Google Scholar] [CrossRef]
- He, S.; Zhou, Z.; Liu, Y.; Shi, P.; Yao, B.; Ringø, E.; Yoon, I. Effects of dietary Saccharomyces cerevisiae fermentation product (DVAQUA®) on growth performance, intestinal autochthonous bacterial community and non-specific immunity of hybrid tilapia (Oreochromis niloticus ♀×O. aureus ♂) cultured in cages. Aquaculture 2009, 294, 99–107. [Google Scholar] [CrossRef]
- Triantaphyllopoulos, K.A.; Cartas, D.; Miliou, H. Factors influencing (GH) and (IGF-I) gene expression on growth in teleost fish: How can aquaculture industry benefit? Rev. Aquac. 2019, 12, 1637–1662. [Google Scholar] [CrossRef]
- Larsen, D.A.; Beckman, B.R.; Dickhoff, W.W. The effect of low temperature and fasting during the winter on metabolic stores and endocrine physiology (insulin, insulin-like growth factor-I, and thyroxine) of coho salmon, Oncorhynchus kisutch. Gen. Comp. Endocrinol. 2001, 123, 308–323. [Google Scholar] [CrossRef]
- Dawood, M.A.; Magouz, F.I.; Essa, M.; Mansour, M. Impact of yeast fermented poultry by-product meal on growth, digestive enzyme activities, intestinal morphometry and immune response traits of common carp (Cyprinus carpio). Ann. Anim. Sci. 2020, 20, 939–959. [Google Scholar] [CrossRef]
- Ran, C.; Huang, L.; Liu, Z.; Xu, L.; Yang, Y.; Tacon, P.; Auclair, E.; Zhou, Z. A comparison of the beneficial effects of live and heat-inactivated baker’s yeast on Nile tilapia: Suggestions on the role and function of the secretory metabolites released from the yeast. PLoS ONE 2015, 10, e0145448. [Google Scholar] [CrossRef]
- Yang, X.; He, Y.; Chi, S.; Tan, B.; Lin, S.; Dong, X.; Yang, Q.; Liu, H.; Zhang, S. Supplementation with Saccharomyces cerevisiae hydrolysate in a complex plant protein, low-fishmeal diet improves intestinal morphology, immune function and Vibrio harveyi disease resistance in Epinephelus coioides. Aquaculture 2020, 529, 735655. [Google Scholar] [CrossRef]
- Islam, S.M.; Rohani, M.F.; Shahjahan, M. Probiotic yeast enhances growth performance of Nile tilapia (Oreochromis niloticus) through morphological modifications of intestine. Aquac. Rep. 2021, 21, 100800. [Google Scholar] [CrossRef]
- Chen, X.-Q.; Zhao, W.; Xie, S.-W.; Xie, J.-J.; Zhang, Z.-H.; Tian, L.-X.; Liu, Y.-J.; Niu, J. Effects of dietary hydrolyzed yeast (Rhodotorula mucilaginosa) on growth performance, immune response, antioxidant capacity and histomorphology of juvenile Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2019, 90, 30–39. [Google Scholar] [CrossRef]
- Yuan, X.-Y.; Jiang, G.-Z.; Wang, C.-C.; Abasubong, K.P.; Zou, Q.; Zhou, Y.-Y.; Liu, W.-B. Effects of partial replacement of fish meal by yeast hydrolysate on antioxidant capability, intestinal morphology, and inflammation-related gene expression of juvenile Jian carp (Cyprinus carpio var. Jian). Fish Physiol. Biochem. 2018, 45, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, G.; Ouraji, H.; Khalesi, M.K.; Sudagar, M.; Barari, A.; Zarei Dangesaraki, M.; Jani Khalili, K.H. Effects of a prebiotic, Immunogen (sup®), on feed utilization, body composition, immunity and resistance to (Aeromonas hydrophila) infection in the common carp (Cyprinus carpio, Linnaeus) fingerlings. J. Anim. Physiol. Anim. Nutr. 2011, 96, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Naiel, M.A.; Gewida, A.G.; Merwad, A.-R.M.; Abdel-Hamid, E.A.; Negm, S.S.; Alagawany, M.; Farag, M.R. The effects of various organic fertilizers with or without adsorbents on the productivity, antioxidant status and immune responses of Nile tilapia raised in cement ponds. Aquaculture 2022, 548, 737593. [Google Scholar] [CrossRef]
- Soltanzadeh, S.; Esmaeili Fereidouni, A.; Ouraji, H.; Khalili, K.J. Growth performance, body composition, hematological, and serum biochemical responses of beluga (Huso huso) juveniles to different dietary inclusion levels of faba bean (Vicia faba) meal. Aquac. Int. 2015, 24, 395–413. [Google Scholar] [CrossRef]
- Zhang, P.; Cao, S.; Zou, T.; Han, D.; Liu, H.; Jin, J.; Yang, Y.; Zhu, X.; Xie, S.; Zhou, W. Effects of dietary yeast culture on growth performance, immune response and disease resistance of gibel carp (Carassius auratus gibelio CAS III). Fish Shellfish Immunol. 2018, 82, 400–407. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Eweedah, N.M.; Khalafalla, M.M.; Khalid, A.; Asely, A.E.; Fadl, S.E.; Amin, A.A.; Paray, B.A.; Ahmed, H.A. Saccharomyces cerevisiae increases the acceptability of Nile tilapia (Oreochromis niloticus) to date palm seed meal. Aquac. Rep. 2020, 17, 100314. [Google Scholar] [CrossRef]
- Park, Y.; Kim, J.H.; Lee, H.-S.; Jung, E.Y.; Lee, H.; Noh, D.O.; Suh, H.J. Thermal stability of yeast hydrolysate as a novel anti-obesity material. Food Chem. 2013, 136, 316–321. [Google Scholar] [CrossRef]
- Jung, E.Y.; Cho, M.K.; Hong, Y.-H.; Kim, J.H.; Park, Y.; Chang, U.J.; Suh, H.J. Yeast hydrolysate can reduce body weight and abdominal fat accumulation in obese adults. Nutrition 2014, 30, 25–32. [Google Scholar] [CrossRef]
- Eslamloo, K.; Falahatkar, B.; Yokoyama, S. Effects of dietary bovine lactoferrin on growth, physiological performance, iron metabolism and non-specific immune responses of Siberian sturgeon Acipenser baeri. Fish Shellfish Immunol. 2012, 32, 976–985. [Google Scholar] [CrossRef]
- Naiel, M.A.; Khames, M.K.; Abdel-Razek, N.; Gharib, A.A.; El-Tarabily, K.A. The dietary administration of miswak leaf powder promotes performance, antioxidant, immune activity, and resistance against infectious diseases on Nile tilapia (Oreochromis niloticus). Aquac. Rep. 2021, 20, 100707. [Google Scholar] [CrossRef]
- Li, J.; Xu, Y.; Jin, L.; Li, X. Effects of a probiotic mixture (Bacillus subtilis;YB-1 and Bacillus cereus;YB-2) on disease resistance and non-specific immunity of sea cucumber,(Apostichopus japonicus, Selenka). Aquac. Res. 2014, 46, 3008–3019. [Google Scholar] [CrossRef]
- Wang, L.; Ge, C.; Wang, J.; Dai, J.; Zhang, P.; Li, Y. Effects of different combinations of Bacillus on immunity and antioxidant activities in common carp. Aquac. Int. 2017, 25, 2091–2099. [Google Scholar] [CrossRef]
- Jiao, W.; Han, Q.; Xu, Y.; Jiang, H.; Xing, H.; Teng, X. Impaired immune function and structural integrity in the gills of common carp (Cyprinus carpio L.) caused by chlorpyrifos exposure: Through oxidative stress and apoptosis. Fish Shellfish Immunol. 2019, 86, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-P.; Wu, Z.-H.; Jian, J.-C.; Zhang, X.-Z. Effect of marine red yeast Rhodosporidium paludigenum on growth and antioxidant competence of Litopenaeus vannamei. Aquaculture 2010, 309, 62–65. [Google Scholar] [CrossRef]
- Saurabh, S.; Sahoo, P.K. Lysozyme: An important defence molecule of fish innate immune system. Aquac. Res. 2008, 39, 223–239. [Google Scholar] [CrossRef]
- Negm, S.S.; Ismael, N.E.; Ahmed, A.I.; Asely, A.M.E.; Naiel, M.A. The efficiency of dietary Sargassum aquifolium on the performance, innate immune responses, antioxidant activity, and intestinal microbiota of Nile Tilapia (Oreochromis niloticus) raised at high stocking density. J. Appl. Phycol. 2021, 33, 4067–4082. [Google Scholar] [CrossRef]
- Michaud, E.; Mastrandrea, C.; Rochereau, N.; Paul, S. Human Secretory IgM: An Elusive Player in Mucosal Immunity. Trends Immunol. 2020, 41, 141–156. [Google Scholar] [CrossRef]
- Ravindra; Pradhan, P.K.; Paria, A.; Pande, V.; Verma, D.K.; Arya, P.; Rathore, G.; Sood, N. Expression of immune genes in Indian major carp, Catla catla challenged with Flavobacterium columnare. Fish Shellfish Immunol. 2019, 94, 599–606. [Google Scholar] [CrossRef]
- Yang, X.; He, Y.; Lin, S.; Dong, X.; Yang, Q.; Liu, H.; Zhang, S.; Chi, S.; Tan, B. Saccharomyces cerevisiae extracts improved the effects of a low fishmeal, complex plant protein diet in the orange-spotted grouper, Epinephelus coioides. Aquac. Rep. 2021, 19, 100574. [Google Scholar] [CrossRef]
- Zhang, J.; Cui, J.; Wang, Y.; Lin, X.; Teng, X.; Tang, Y. Complex molecular mechanism of ammonia-induced apoptosis in chicken peripheral blood lymphocytes: miR-27b-3p, heat shock proteins, immunosuppression, death receptor pathway, and mitochondrial pathway. Ecotoxicol. Environ. Saf. 2022, 236, 113471. [Google Scholar] [CrossRef]
- Rungrassamee, W.; Klanchui, A.; Maibunkaew, S.; Karoonuthaisiri, N. Bacterial dynamics in intestines of the black tiger shrimp and the Pacific white shrimp during Vibrio harveyi exposure. J. Invertebr. Pathol. 2016, 133, 12–19. [Google Scholar] [CrossRef]
| Ingredient | % |
|---|---|
| Fish meal (Crude protein, 70%) | 58 |
| Wheat flour | 27.4 |
| Fish oil | 11.2 |
| Lecithin | 1 |
| a Minerals + Vitamin mix | 0.4 |
| Fish protein soluble | 2 |
| Total | 100 |
| Chemical composition | |
| Crude protein | 45.1 |
| Crude Lipids | 18.3 |
| Ash | 8.8 |
| Starch | 20.3 |
| b Gross Energy, GE, kJ/g | 22.7 |
| Gene | Forward (5→3′) | Reverse (5→3′) | GenBank Accession No. |
|---|---|---|---|
| β-actin | CGACGGACAGGTCATCACCA | AGAAGCATTTGCGGTGGACG | AF384096.1 |
| IGF1 | AGTGCGATGTGCTGTATC | CAGCTCACAGCTTTGGAAG- | EF563837.1 |
| HSP70 | AATGTTCTGCGCATCATCAA | CCAACCTTTTTGTCCAATCC | EU805481.1 |
| IL-1β | GGGCTGAACAACAGCACTCTC | TTAACACTCTCCACCCTCCA | 115592467 |
| Parameters | Control | Dietary Treatments | SEM | p-Value | ||
|---|---|---|---|---|---|---|
| SC1 | SC2 | SC4 | ||||
| IBW (g) | 31.233 | 32.600 | 31.233 | 31.833 | 1.254 | 0.0963 |
| FBW (g) | 109.737 b | 126.053 a | 129.460 a | 129.767 a | 1.287 | <0.001 |
| DWG (g/day) | 0.700 c | 0.834 b | 0.877 a | 0.874 a | 0.012 | <0.001 |
| SGR (g/d−1%) | 1.120 c | 1.207 b | 1.270 a | 1.254 a | 0.013 | <0.001 |
| FCR (g/g) | 2.624 b | 2.525 bc | 2.502 c | 1.916 a | 0.038 | <0.001 |
| PER (g/g) | 1.312 a | 1.179 b | 1.125 c | 1.136 bc | 0.017 | <0.001 |
| Dietary Treatments | Dry Matter | Moisture | Crude Protein | Crude Fat | Ash |
|---|---|---|---|---|---|
| Control | 33.630 c | 66.370 a | 40.820 b | 30.273 d | 18.433 a |
| SC1 | 35.990 b | 64.010 b | 46.970 ab | 32.916 c | 17.870 b |
| SC2 | 36.273 b | 63.726 b | 47.060 ab | 35.823 b | 17.020 c |
| SC4 | 36.993 a | 63.006 c | 52.260 a | 37.396 a | 16.970 c |
| SEM | 0.192 | 0.192 | 2.991 | 0.366 | 0.070 |
| p-value | <0.001 | <0.001 | 0.0387 | <0.001 | <0.001 |
| Control | Dietary Treatments | SEM | p-Value | |||
|---|---|---|---|---|---|---|
| SC1 | SC2 | SC4 | ||||
| Hematological variables | ||||||
| RBCs (×106 µL) | 3.610 | 3.733 | 3.583 | 4.003 | 0.504 | 0.4326 |
| HGB (g/dL) | 10.986 c | 11.636 b | 11.950 a | 12.200 a | 0.080 | <0.001 |
| PCV (×103 µL) | 35.33 c | 36.66 bc | 38.33 ab | 40.00 a | 0.645 | 0.0046 |
| MCV (fL) | 89.07 c | 97.22 b | 97.71 ab | 98.20 a | 0.153 | 0.0077 |
| MCH (pg/dL) | 30.44 b | 30.45 b | 30.79 ab | 31.15 a | 0.158 | 0.0397 |
| MCHC (g/dL) | 31.00 | 31.88 | 31.40 | 31.26 | 0.120 | 0.4421 |
| Immunological variables | ||||||
| WBCs (×106 µL) | 19.273 c | 26.140 b | 27.717 b | 30.707 a | 0.867 | <0.001 |
| Monocytes (%) | 8.00 | 9.00 | 9.00 | 9.33 | 0.527 | 0.3700 |
| Lymphocytes (%) | 70.33 a | 81.00 a | 78.00 a | 56.00 b | 4.847 | 0.0307 |
| Basophils (%) | 0.666 | 1.000 | 1.000 | 0.666 | 0.235 | 0.5954 |
| Eosinophils (%) | 1.333 | 1.333 | 1.000 | 1.000 | 0.372 | 0.8473 |
| Lysozyme (%) | 6.850 d | 8.960 c | 9.303 b | 10.303 a | 0.087 | <0.001 |
| Phagocytic activity (%) | 8.160 c | 11.136 b | 11.206 b | 12.036 a | 0.098 | <0.001 |
| Phagocytic index | 1.000 b | 1.103 ab | 1.213 a | 1.243 a | 0.044 | 0.0164 |
| IgM (ng/mL) | 3.040 c | 4.340 b | 4.610 b | 5.220 a | 0.153 | <0.001 |
| Parameters | Control | Dietary Treatments | SEM | p-Value | ||
|---|---|---|---|---|---|---|
| SC1 | SC2 | SC4 | ||||
| TP (g/dL) | 4.906 d | 5.020 c | 5.206 b | 5.300 a | 0.015 | <0.001 |
| Alb (g/dL) | 1.310 b | 1.334 b | 1.340 b | 1.400 a | 0.017 | 0.031 |
| Globulin (g/dL) | 3.596 d | 3.683 c | 3.863 b | 3.906 a | 0.007 | <0.001 |
| Glucose (mg/dL) | 12.336 d | 14.010 c | 15.070 a | 15.970 a | 0.114 | <0.001 |
| TG (mg/dL) | 88.020 c | 91.256 b | 97.560 a | 99.733 a | 1.607 | <0.001 |
| Chol (mg/dL) | 91.430 c | 97.220 b | 104.523 a | 107.860 a | 1.982 | <0.001 |
| Urea (mg/dL) | 1.680 a | 1.670 a | 1.560 b | 1.583 b | 0.007 | <0.001 |
| AST (IU/L) | 20.026 | 20.006 | 18.763 | 18.836 | 0.582 | 0.310 |
| ALT (IU/L) | 30.186 | 29.213 | 29.380 | 28.626 | 0.359 | 0.0822 |
| Amylase (U/L) | 40.623 d | 46.600 c | 52.183 b | 58.270 a | 1.037 | <0.001 |
| Lipase (U/L) | 34.550 c | 49.980 b | 51.407 ab | 52.883 a | 0.758 | <0.001 |
| Cortisol (ng/mL) | 26.080 | 25.333 | 25.626 | 24.893 | 0.337 | 0.0674 |
| Parameters | Control | Dietary Treatments | SEM | p-Value | ||
|---|---|---|---|---|---|---|
| SC1 | SC2 | SC4 | ||||
| MDA (mmol/L) | 14.650 a | 11.303 b | 10.040 c | 9.773 c | 0.327 | <0.001 |
| CAT (U/mL) | 11.130 b | 11.343 b | 11.410 b | 12.513 a | 0.277 | 0.0423 |
| SOD (U/mL) | 9.470 c | 10.100 b | 10.290 b | 10.716 a | 0.105 | 0.0002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Bab, A.F.F.; Saghir, S.A.M.; El-Naser, I.A.A.; El-Kheir, S.M.M.A.; Abdel-Kader, M.F.; Alruhaimi, R.S.; Alqhtani, H.A.; Mahmoud, A.M.; Naiel, M.A.E.; El-Raghi, A.A. The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata). Life 2022, 12, 1013. https://doi.org/10.3390/life12071013
El-Bab AFF, Saghir SAM, El-Naser IAA, El-Kheir SMMA, Abdel-Kader MF, Alruhaimi RS, Alqhtani HA, Mahmoud AM, Naiel MAE, El-Raghi AA. The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata). Life. 2022; 12(7):1013. https://doi.org/10.3390/life12071013
Chicago/Turabian StyleEl-Bab, Ahmed F. Fath, Sultan A. M. Saghir, Ibrahim Atta Abu El-Naser, Salwa M. M. Abo El-Kheir, Marwa F. Abdel-Kader, Reem S. Alruhaimi, Haifa A. Alqhtani, Ayman M. Mahmoud, Mohammed A. E. Naiel, and Ali Ali El-Raghi. 2022. "The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata)" Life 12, no. 7: 1013. https://doi.org/10.3390/life12071013
APA StyleEl-Bab, A. F. F., Saghir, S. A. M., El-Naser, I. A. A., El-Kheir, S. M. M. A., Abdel-Kader, M. F., Alruhaimi, R. S., Alqhtani, H. A., Mahmoud, A. M., Naiel, M. A. E., & El-Raghi, A. A. (2022). The Effect of Dietary Saccharomyces cerevisiae on Growth Performance, Oxidative Status, and Immune Response of Sea Bream (Sparus aurata). Life, 12(7), 1013. https://doi.org/10.3390/life12071013








