Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses
Abstract
1. Introduction
2. Botanical and Ethnomedicinal Use
2.1. Taxonomy and Synonym
2.2. Botanical and Ecology Description
2.3. Geographic Distribution
2.4. Ethnomedicinal Use
| Area of Study/Country | Local Name | Part Used | Mode of Preparation | Traditional Use | References |
|---|---|---|---|---|---|
| Taza region (Morocco) | Babonj | Flower | Infusion Decoction | Diabetes | [72] |
| Beni Mellal (Morocco) | Babounj | Flower | Infusion | Diabetes mellitus | [73] |
| Daraa-Tafilalet region, Errachidia province (Morocco) | Not specified | Whole plant | Infusion | Nervous disorders | [74] |
| Tarfaya Province (Morocco) | Babounj | Leaves | Decoction | Antispasmodic | [75] |
| Fez (Morocco) | Babounj | Flower | Infusion Decoction | Colic, diarrhea, nervousness, depression, angina, canker sore, painful menstruation, fever, abscess, infections | [7] |
| Hatay Province (Turkey) | Babuneç Papatya | Flower head | Infusion | Cold, colic spasm, sedative | [4] |
| Granada province (southern Spain) | Not specified | Flowery plant | Infusion | Gastralgia, digestive disorder, conjunctivitis, dysmenorrhea, cold, cough, gases, female genital infection, kidney stones, eye infection, headache, insomnia | [76] |
| Alt Empordà region (Catalonia, Spain) | Camamilla, Camamilla de jardí | Not specified | Decoction Infusion | Antiseptic, ocular antiseptic, conjunctivitis, digestive, gastric and intestinal anti-inflammatory, stomachache, nausea, antiemetic | [77] |
| Trás-os-Montes (northern Portugal) | Camomila | Flowering top | Infusion Decoction | Depression, nervousness, stress, insomnia, neuralgia, sciatic pain, digestive, stomachache, gases, intestinal colic, infection of mouth, throat, and ear, cellulitis, asthma | [6] |
| Island of Procida (Campania, southern Italy) | Cammumilla | Whole plant Stem Inflorescence | Infusion Decoction In the bath | Cold, cough, sprain, broken bones, irritability, tired eyes, conjunctivitis, abdominal colic, gastrointestinal pain, muscular pain, menstrual pain | [3] |
| National Park of Cilento and Vallo di Diano (southern Italy) | Hammamilla | Aerial parts | Infusion | Sedative, dye yellow, bleach hair | [79] |
| Monte Sicani Regional Park Central Western Sicily (southern Italy) | Kamilica | Flower | Infusion | Sedative, digestive | [78] |
| Bulgaria | Not specified | Flower head | Decoction Inhalation Compress | Throat pain, cold, swollen eyes, cleansing face, genitalia | [86] |
| Southeastern Albania | Kamilica Koromil | Flowering tops | Tea | Intestinal discomfort, diarrhea, cough | [85] |
| Pirot County (eastern Serbia) | Bela rada, kamilica, podrumce | Flower | Infusion | Stomach disorder | [80] |
| Rtanj Mt. (eastern Serbia) | Kamilica | Flower | Infusion | Immune system strengthening, cold, sedative, conjunctivitis, anti-inflammatory | [84] |
| Negotin Krajina (eastern Serbia) | Kamilica | Flower Leaves | Infusion | Digestive disorder, vaginal disorder, eye care skin care, aroma for shampoos | [81] |
| Jablanica district (southeastern Serbia) | Kamilica | Herb | Infusion | Skin and mucus inflammation, digestive disorder, cough, anxiety, eyewash, mouthwash | [82] |
| Zlatibor district (southwestern Serbia) | Not specified | Herb | Infusion | Cold and stomach disorder | [83] |
| Pčinja district (southeastern Serbia) | Kamilica | Herb | Infusion | Skin inflammation, burns, digestive disorder, liver disorder, cough, anxiety, insomnia, eyewash, mouthwash | [5] |
| Peloponnisos (Greece) | Hamomili | Inflorescences |
Infusion Compress Vapor inhalation | Stomach disorder, constipation, ulcer, colic, allergy, insomnia, migraine, stress, skin problems (inflammation, dermatitis, acne, burn, eczema, itching, wound antiseptic), catarrh, sore throat, eye infection, aphthae, gingivitis, eyewash, mouthwash | [8] |
3. Phytochemical Interest
| Country/Source | Part Used | Compounds Groups | Main Compounds | References |
|---|---|---|---|---|
| Essential oils | ||||
| Morocco | Aerial parts | Terpenoids Coumarin | Chamazulene (26.11%), cis-β-farnesene (11.64%), eucalyptol (8.19%), trans-caryophyllene (5.95%), galaxolide (5.31%) Coumarin (6.01%) | [87] |
| Egypt | Flower | Terpenoids | α-Bisabolol oxide A (33–50.5%), cis-tonghaosu (10–18.7%), α-bisabolol oxide B (8.2–15.4%), α-bisabolone oxide A (5.4–14.6%), chamazulene (1.9–5.2%) | [13] |
| Flower | Terpenoids | Cis-β-Farnesene (27%), D-limonene (15.25%), α-bisabolol oxide A (14.9%) | [88] | |
| Turkey | Flower | Terpenoids | α-Bisabolol (27.36–38.6%), β-farnesene (25.05–30.15%), chamazulene (13.5–13.93%), Germancrene D (4.35–6.11%) | [89] |
| Iran | Flower | Terpenoids | α-Bisabolol oxide A (29.7–33.7%), chamazulene (18.76–20%), α-bisabolol oxide B (8.881–14.8%), α-bisabolone oxide A (6.64–8.3%), α-bisabolol (0.91–2.01%) | [93] |
| Flower | Terpenoids | α-Bisabolol oxide A (37.2–44.5%), α-bisabolone oxide A (11.7–16.5%), trans-β-farnesene (13.3–15.4%), menthol (0–13%), cis-spiroether (5.6–9.9%), α-bisabolol oxide B (3–7.1%) | [15] | |
| Flower | Terpenoids | Bisabolol oxide A (7.31–51.31%), bisabolone oxide (8.35–39.97%), bisabolol oxide B (3.18–35.7%), trans-β-farnesene (2.05–19.68%), spathulenol (0–9.46%) | [11] | |
| Aerial part | Terpenoids | α-Bisabolol (17.51%), cis-trans-farnesol (8.72%), β-bisabolene (8.37%), trans-β-farnesene (5.48%), guaiazulene (4.36%), α-pinene (3.68%), limonene (3.24%) | [96] | |
| Flower | Terpenoids | Chamazulene (31.2%), 1,8-cineole (15.2%), β-pinene (10.1%), α-pinene (8.14%), α-bisabolol (7.45%), terpinen-4-ol (4.11%) | [95] | |
| Aerial part | Terpenoids | Cis-pinocamphone (not detected–73.54%) α-bisabolol oxide A (7.97–62.16%), chamazulene (1.67–15.08%), trans-β-farnesene (1.24–12.87%) | [14] | |
| Flower | Terpenoids | α-Bisabolone oxide A (45.64–65.41%), trans-γ-bisabolene (not detected—42.76%), cis-γ-bisabolene (not detected—40.08%), α-bisabolol oxide B (not detected—21.88%), chamazulene (not detected—19.22%) | [10] | |
| Flower | Terpenoids | Trans-β-farnesene (24.19%), guaiazulene (10.57%), α-bisabolol oxide A (10.21%), α-farnesene (8.7%), α-bisabolol (7.27%) | [94] | |
| Flower | Terpenoids | α-Bisabolone oxide A (11.9–63.5%), α-bisabolol oxide A (10.6–37.9%), α-bisabolol oxide B (2.4–23.9%) | [12] | |
| Hungary | Flower | Terpenoids | α-Bisabolol oxide A (40.7%), chamazulene (14.3%), α-bisabolone oxide A (12.8%), α-bisabolol oxide B (8.7%) | [12] |
| Germany | Flower | Terpenoids | α-Bisabolol oxide A (39.1%), α-bisabolone oxide A (17.4%), α-bisabolol oxide B (17.1%), cis-Enyne-dicycloether (10.3%) | [12] |
| Flower | Terpenoids | Bisabolol oxide A (54.1%), cis-enyne-dicycloether (19%), bisabolol oxide B (6.7%), bisabolone oxide A (4.5%) | [9] | |
| Estonia | Flower | Terpenoids | Bisabolol oxide A (27.5–47.9%), bisabolone oxide A (1.6–17.1%), cis-enyne-dicycloether (11.7–14.9%), bisabolol oxide B (9.9–12.3%) | [9] |
| Greece | Flower | Terpenoids | Bisabolol oxide A (41.9%), cis-enyne-dicycloether (11.4%), bisabolol oxide B (6.4%) | [9] |
| Scotland | Flower | Terpenoids | Bisabolol oxide A (55.6%), cis-enyne-dicycloether (14%), bisabolol oxide B (8%), bisabolone oxide A (7.6%) | [9] |
| England | Flower | Terpenoids | Bisabolol oxide A (56%), cis-enyne-dicycloether (13.3%), bisabolol oxide B (7.1%), bisabolone oxide A (4.3%) | [9] |
| Latvia | Flower | Terpenoids | Bisabolol oxide A (51.9%), cis-enyne-dicycloether (13%), bisabolol oxide B (7.5%), trans-β-farnesene (5.3%) | [9] |
| Moldova | Flower | Terpenoids | α-Bisabolol (44.2%), cis-enyne-dicycloether (13.2%), bisabolone oxide A (12.4%), bisabolol oxide A (9.3%), bisabolol oxide B (6.6%) | [9] |
| Russia | Flower | Terpenoids | α-Bisabolol (23.9%), bisabolol oxide A (16.4%), cis-enyne-dicycloether (14.4%), bisabolol oxide B (10.5%), trans-Nerolidol (7.4%) | [9] |
| Czech | Flower | Terpenoids | α-Bisabolol (37%), cis-enyne-dicycloether (26.1%), chamazulene (9.8%), trans-β-farnesene (4.5%) | [9] |
| Armenia | Flower | Terpenoids | Bisabolol oxide B (27.2%), chamazulene (15.3%), bisabolol oxide A (12.6%), cis-enyne-dicycloether (12.6%), bisabolone oxide A (11.2%) | [9] |
| Ukraine | Flower | Terpenoids | Bisabolone oxide A (24.8%), α-bisabolol (17.1%), bisabolol oxide A (12.3%), bisabolol oxide B (11%), cis-enyne-dicycloether (8.8%) | [9] |
| Romania | Flower | Terpenoids | Bisabolol oxide A (70.2%), β-farnesene (6.47%), α-bisabolol oxide B (6.21%), cis-lanceol (5.071%) | [90] |
| Brazil | Flower | Terpenoids | α-Bisabolol oxide B (26.08%), β-farnesene (16.35%), bisabolol oxide A (14.7%), α-bisabolol (7.91%) | [91] |
| Italy | Aerial parts | Terpenoids | Cis-tonghaosu (11.8–45.9%), α-bisabolol oxide B (3.7–28.1%), α-bisabolol oxide A (2.7–19%), spathulenol (3.6–12.8%) | [92] |
| Australia | Flower | Terpenoids | Bisabolol oxide B (27.5%), bisabolol oxide A (27%), α-bisabolol (6.6%), cis-spiroether (6.1%), farnesene (4.5%), chamazulene (3.5%), trans-spiroether (0.6%) | [99] |
| USA | Commercial | Terpenoids | Trans-β-Farnesene (42.59%), bisabolol oxide A (21.2%), (E,E)-α-farnesene (8.32%), α-bisabolone oxide A (4.53%), α-bisabolol oxide B (4.43%), germacrene D (2.93%) | [100] |
| Extracts | ||||
| Egypt | Flower and roots Powder | Flavonoids Phenolic acids | Luteolin O-acylhexoside (2801.99 ppm), quercetin (1765.01 ppm) Ellagic acid (1582.81 ppm), catechol (1104.49 ppm), chlorogenic acid (937.48 ppm) | [102] |
| Flower Aqueous | Flavonoids Phenolic acids | Myricetin (1587.82 ppm), quercetin (927.72 ppm), naringenin (400.99 ppm) Benzoic acid (414.88 ppm), rosmarinic acid (370.59 ppm) | [53] | |
| Slovakia | Leaf rosettes Methanol | Phenolic acids | Ferulic acid (196.8–512.5 μg/g), caffeic acid (66.8–106.1 μg/g), vanillic acid (45.6–71.1 μg/g), chlorogenic acid (12.6–26.2 μg/g), p-coumaric acid (14.4–26.1 μg/g) | [18] |
| Flower or leaves Methanol | Coumarin | E-GMCA (9.82–17.8 mg/g), Z-GMCA (5.84–16.1 mg/g), herniarin (0.41–2.06 mg/g), daphnin (0.142–0.257 mg/g), skimmin (0.13–0.23 mg/g), umbelliferone (0.02–0.06 mg/g), daphnetin (trace-0.02 mg/g) | [103] | |
| Leaves Methanol | Coumarin Phenolic acids | E-GMCA (6.86–9.62 mg/g), Z-GMCA (1.22–6.6 mg/g) Vanillic acid (29.27–62.46 µg/g), caffeic acid (7.44–14.14 µg/g) | [17] | |
| Iran | Flower Methanol | Flavonoids | Luteolin (2.2 mg/g), apigenin (1.19 mg/g) | [10] |
| Not specified Ethanol | Alkane | n-Heptacosane (33.53%), 2,6,10,14,18,22-tetracosahexaene (16.71%), 1,2,2-trimethylcyclopropylamine (13.76%), 7-methoxy-2,3,4,5,6,7-hexahydro (6.13%), 1,2-benzenedicarboxylic acid (5.99%), Phenol, 4-(2-aminoethyl) (5.26%), hex-5-enylamine (4.48), 3-fluorophenethylamine (0.2%) | [104] | |
| Pakistan | Not specified Aqueous | Amino acids | l-Proline (185 mg/mL), l-asparagine (97 mg/mL), aminobutyric acid (52 mg/mL), l-aspartic acid (45 mg/mL), l-alanine (43 mg/mL), l-glutamic acid (42 mg/mL) | [16] |
| China | Roots, stems, Leaves 70% aqueous methanol | Caffeoylquinic acids | Isochlorogenic acid A (0.1–5.15 mg/g), chlorogenic acid (0.03–4.08 mg/g), isochlorogenic acid C (0.06–3.17 mg/g), isochlorogenic acid B (0.03–2.45 mg/g), neochlorogenic acid (0.02–1.68 mg/g), cryptochlorogenic acid (0.005–0.33 mg/g) | [105] |
| Flower Aqueous | Amino acids | Proline (4.24 mg/g), alanine (3.79 mg/g), isoleucine + leucine (2.59 mg/g), arginine + threonine (2.53 mg/g) | [106] | |
4. Pharmacological Interest
4.1. Antioxidant Activity
| Part Used | Main Component | Experimental Method | Key Results | References |
|---|---|---|---|---|
| Essential oils | ||||
| Leaves | Not specified | DPPH β-carotene-linoleic acid assay |
IC50 = 4.18 µg/mL Relative antioxidant activity = 12.69% | [111] |
| Leaves and flowers |
Enyne-dicycloether (36.13–47.6%) Bisabolol oxide A (47.1%) β-Farnesene (30.2–37.62%) | ABTS | TEAC = 13.81–27.56 μmol TE/mL | [109] |
| Flower | α-Bisabolone oxide A (35.74%) | DPPH Ferrous ion chelating ability β-carotene bleaching | IC50 = 793.89 µg/mL IC50 = 1448.68 µg/mL 34.21% | [108] |
| Flower | Trans-β-Farnesene (29.8%) | DPPH | EC50 = 2.07 mg/mL | [107] |
| Aerial parts | Cis-tonghaosu (11.8–45.9%) α-Bisabolol oxide B (3.7–28.1%) | DPPH FRAP | TEAC = ~30–273.5 μmol TE/100 g DW TEAC = ~35–657.1 µmol TE/100 g DW | [92] |
| Extracts | ||||
|
Aerial parts Methanol | Phenol content (~390–2689.2 mg GAE/100 g) | DPPH FRAP | TEAC = ~260–881.1 µmol TE/100 g DW TEAC = 137.2–1200.3 µmol TE/100 g DW | [92] |
|
Flower Ethyl acetate Na phosphate buffer Leaf Ethyl acetate Na phosphate buffer Stem Ethyl acetate Na phosphate buffer Root Ethyl acetate Na phosphate buffer | Phenol content 11.29 mg GAE/g DW 21.78 mg GAE/g DW 9.21 mg GAE/g DW 10.56 mg GAE/g DW 8.13 mg GAE/g DW 13.45 mg GAE/g DW 4.16 mg GAE/g DW 4.41 mg GAE/g DW | ABTS | LAA = 3.51 mg TE/g DW HAA = 17.57 mg TE/g DW LAA = 1.47 mg TE/g DW HAA = 9.28 mg TE/g DW LAA = 1.49 mg TE/g DW HAA = 12.27 mg TE/g DW LAA = 1.48 mg TE/g DW HAA = 18.02 mg TE/g DW | [110] |
|
Roots Methanol Ethanol | Not specified | DPPH | IC50 = 82.8% IC50 = 37.67% | [114] |
|
Flower 70% ethanol Water | Phenol content (117.31–151.45 mg CAE/mL) | DPPH Reducing power | IC50 = 0.0211–0.0606 mg/mL EC50 = 0.578–0.922 mg/mL | [118] |
|
Flower Water | Apigenin (231–1501 mg/kg) Luteolin-7-O-glucoside (166–1101 mg/kg) | ABTS DPPH Hydroxyl radical scavenging Lipid peroxidation inhibition | IC50 = 7.3–16.8 µg/mL IC50 = 10–45 µg/mL IC50 = 38.1–43.1 µg/mL IC50 = 28.7–35 µg/mL | [118] |
|
Plant material Water | Not specified | DPPH | 41.3–49.5% | [121] |
|
Flowering plant Ethanol | Phenol content (284.6 ± 16 mg GAE/g DW) | DPPH | IC50 = 56.4 µg/mL | [49] |
|
Flower Methanol Ethanol Diethyl ether Hexane | Phenol content (3.7 mg GAE/g DW) (3.5 mg GAE/g DW) (3.3 mg GAE/g DW) (2.4 mg GAE/g DW) | DPPH | EC50 = 0.0022 µmol EC50 = 0.0026 µmol EC50 = 0.0039 µmol EC50 = 0.0041 µmol | [116] |
|
Aerial parts Ethanol (70%) | Phenol content (78.4 mg GAE/g DW) | DPPH | IC50 = 50 µg/mL | [116] |
|
Leaves Methanol | Not specified | DPPH | IC50 = 65.8 μg/mL | [112] |
|
Whole herb Stem Flower Methanol | Phenol content Whole herb (37.1 mg/kg DW) Stem (23.6 mg/kg DW) Flower (31.9 mg/kg DW) | DPPH | Whole herb: IC50 ~2.5 μg/mL Stem: IC50 ~2.4 μg/mL Flower: IC50 ~2.35 μg/mL | [113] |
| Linoleic acid emulsion (30 h) | Whole herb: 63% Stem: 69% Flower: 60% | |||
| FRAP | Whole herb: absorbance ~1.8 Stem: absorbance ~0.88 Flower: absorbance ~0.9 | |||
| Ferrous ions (Fe2+) chelating capacity | Whole herb: 73% Stem: 67% Flower: 85% | |||
| Superoxide radical scavenging activity | Whole herb: IC50 = 2.1 μg/mL Stem: IC50 = 2.8 μg/mL Flower: IC50 = 2.2 μg/mL | |||
|
Leaves Methanol | Not specified | DPPH β-carotene-linoleic acid assay |
IC50 = 1.83 µg/mL Relative antioxidant activity = 11.37% | [111] |
|
Flower Ethanol 50% | Umbelliferone content (11.80 mg/100 g) Herniarin content (82.79 mg/100 g) | DPPH | 45.4–61.5% | [20] |
|
Flowering parts Water |
Phenol content (0.041- 0.165 mg GAE/mL) | DPPH | Inhibition = 2.53–4.62 µg TE/mL | [119] |
|
Inflorescences Ethanol (74.7%) | Flavonoid content (4.11%) | DPPH | IC50 = 18.19 µg/mL | [120] |
|
Flower Methanol (80%) | Phenol content (656.1 mg GAE/g FR) | DPPH FRAP | IC50 = 84.2 μg/mL IC50 = 13 mmol Fe+2/100 g | [46] |
|
Flower Water | Phenol content (0.207 mg GAE/g) | Lipid peroxidation inhibition | Inhibition = 44.15% | [122] |
|
Inflorescence Not specified | Flavonoid content (66.2–35.6 mg/g) | Lipid peroxidation inhibition | Inhibition = 10–100% | [123] |
|
Whole Plant Ethanol 80% | Not specified | Superoxide dismutase Malondialdehyde | ~2.2–3.1 U/mL plasma ~112–126 μmol | [125] |
|
Flower and root Powder | Luteolin O-acylhexoside (2801.99 ppm) | Lipid peroxidation inhibition Catalase Acetylcholine esterase Glutathione | 291.35–301.67 nmol 63.14–68.33 nmol 4.65–5.46 nmol 11.2–13.2 mg/g | [102] |
| Other | ||||
| Cell suspension culture |
Phenol content (5.54–9.51 mg GAE/g DW) | DPPH Peroxidase Superoxide dismutase Ascorbate peroxidase | Inhibition = 55.1–76.72% ~2.75–3.75 unite/mg protein ~0.27–0.43 unite/mg protein ~2000–5000 unite/mg protein | [124] |
| Cell suspension culture | Total soluble sugar (63.71–96.04 mg/g FW) | Peroxidase Superoxide dismutase Catalase | ~4.5–8 unite/mg FW ~0.5–0.75 unite/mg FW ~0.002–0.008 unite/mg FW | [19] |
4.2. Antibacterial Activity
| Part Used | Main Component | Experimental Method | Tested Organism | Key Results | References |
|---|---|---|---|---|---|
| Essential oils | |||||
| Leaves | Not specified | Disc diffusion Micro-dilution | Gram-positive Bacillus cereus LMG 13569 Listeria innocua LMG 1135668 Staphylococcus aureus ATCC 9244 Staphylococcus camorum LMG 13567 Streptococcus pyogenes Gram-negative Enterococcus faecalis CIP 103907 Escherichia coli CIP 11609 Salmonella enterica CIP 105150 Shigella dysenteriae CIP 5451 Proteus mirabilis 104588 CIP Pseudomonas aeruginosa | Φ = 17 mm; MIC = 4; MBC = 4 µg/mL Φ = 20 mm; MIC = 2; MBC = 2 µg/mL Φ = 21 mm; MIC = 2; MBC = 2 µg/mL Φ = 22 mm; MIC = 2; MBC = 2 µg/mL Φ = 24 mm; MIC = 2; MBC = 2 µg/mL Φ = 14 mm; MIC = 4; MBC = 8 µg/mL Φ = 14 mm; MIC = 4; MBC = 8 µg/mL Φ = 20 mm; MIC = 2; MBC = 2 µg/mL Φ = 25 mm; MIC = 1; MBC = 1 µg/mL Φ = 17 mm; MIC = 4; MBC = 4 µg/mL Φ = 30 mm; MIC = 1; MBC = 1 µg/mL | [111] |
| Aerial parts | Chamazulene (26.11%) | Disc diffusion Micro-dilution | Gram-positive Staphylococcus aureus Bacillus subtilis Gram-negative Escherichia coli (ATB:57) B6N Pseudomonas aeruginosa | Φ = 14.13 mm; MIC = 8.33 µL/mL Φ = 15.2 mm; MIC = 6.25 µL/mL Φ = 13.27 mm; MIC = 8.33 µL/mL Φ = 13.07 mm; MIC = 8.33 µL/mL | [87] |
| Flower | Not specified | Disc diffusion (0.78–100%) | Gram-positive Staphylococcus aureus ATCC-25923 Gram-negative Escherichia coli ATCC-25922 Pseudomonas aeruginosa ATCC | Φ = 8.55–38.34 mm; MIC = 6.25% Φ = 9.31–12.32 mm; MIC = 1.56% No inhibition | [132] |
| Aerial parts (95% flowers) | Trans-β-farnesene (18.7–38.5%) α-Bisabolol (38.3%) α-Bisabolol oxide A (25%) | Macro-dilution | Gram-positive Staphylococcus aureus ATCC 6538 Gram-negative Escherichia coli ATCC 25922 Salmonella abony ATCC 6017 Pseudomonas aeruginosa ATCC 9027 | MBC = 2000–8000 µg/mL MBC = 2000–8000 µg/mL MBC = 2000–8000 µg/mL MBC = 4000–8000 µg/mL | [27] |
| Aerial parts | α-Bisabolol oxide (38%) | Disc diffusion Micro-dilution | Gram-positive Staphylococcus aureus Bacillus cereus Bacillus subtilis Gram-negative Shigella shiga Shigella sonnei Pseudomonas aeruginosa Proteus sp. | Φ = 30 mm; MIC = 0.011; MBC = 0.13 µg/mL Φ = 36 mm; MIC = 0.022; MBC = 1.5 µg/mL Φ = 32 mm; MIC = 0.03; MBC = 1.5 µg/mL Φ = 25 mm; MIC = 0.14; MBC = 3 µg/mL Φ = 19 mm; MIC = 0.2; MBC = 3 µg/mL Φ = 19 mm; MIC = 4; MBC = 8 µg/mL Φ = 16 mm; MIC = 0.15; MBC = 3 µg/mL | [97] |
| Flower | α-Bisabolone oxide A (35.74%) | Disc diffusion Micro-dilution | Gram-positive Staphylococcus aureus ATCC 25923 Enterococcus faecalis ATCC 14506 Gram-negative Escherichia coli ATCC 25922 Klebsiella pneumoniae ATCC 13883 Proteus vulgaris ATCC 33420 Pseudomonas aeruginosa ATCC27853 | Φ = 71.59%; MIC = 0. 25 mg/mL Φ = 106.7%; MIC = 0.12 mg/mL Φ = 99.66%; MIC = 0.17 mg/mL Φ = 75.04%; MIC = 0.15 mg/mL Φ = 89.15%; MIC = 0.21 mg/mL Φ = 108.77%; MIC = 0.04 mg/mL | [108] |
| Flower | Guaiazulene (25.6%) | (0.2–0.5 µg/mL) Disc diffusion Biofilm formation and adherence assay Quantitative assay of alginate | Gram-negative Pseudomonas aeruginosa 8821M | No inhibition Biofilm production = 0.17–0.64 µg/mL Alginate production = 190.33–549.33 µg/mL | [129] |
| Flower | Guaiazulene (25.6%) | Disc diffusion Macro-dilution | Gram-positive Streptococcus pyogenes PTCC 1447 Streptococcus mutans PTCC 1601 Streptococcus salivarius PTCC 1448 Streptococcus faecalis ATCC 29212 Streptococcus sanguis PTCC 1449 | Φ = 9 mm; MIC = 0.1; MBC = 0.2 µg/mL Φ = 10 mm; MIC = 0.5; MBC = 1.5 µg/mL Φ = 9 mm; MIC = 0.5; MBC = 0.8 µg/mL Φ = 0.8 mm; MIC = 4; MBC = 7 µg/mL Φ = 8 mm; MIC = 0.5; MBC = 1 µg/mL | [129] |
| Commercial | Bisabolol and trans-β-farnesene | Macro-dilution | Gram-positive Staphylococcusaureus MRSA (16 strains) Staphylococcus aureus (2 ATCC strains) Staphylococcus epidermidis ATCC 12228 Enterococci faecalis ATCC 51299 Vancomycin-resistant enterococci (9 strains) | MIC = 2–>4; MBC = 2–>4% MIC = MBC >4% MIC = MBC >4% MIC = MBC >4% MIC = MBC >4% | [129] |
| Aerial parts | Trans-β-farnesene (42.2%) | Micro-dilution | Gram-positive Staphylococcus aureus ATCC 29213 Bacillus cereus ATCC 14579 Gram-negative Escherichia coli ATCC 10798 Pseudomonas aeruginosa ATCC 27853 | MIC = 313 μg/mL MIC = 625 μg/mL MIC = 625 μg/mL MIC = 313 μg/mL | [131] |
| Flower | Not specified | Disc diffusion Broth dilution Ex vivo | Gram-negative Enterococcusfaecalis | Reduction = 2.91 CFU at day 14 | [130] |
| Flower | Trans-β-Farnesene (29.8%) | Disc diffusion | Gram-positive Staphylococcus aureus WDCM 00032 Listeria monocytogenes WDCM 00020 Salmonella enterica WDCM 00030 Gram-negative Escherichia coli WDCM 00013 Pseudomonas aeruginosa WDCM 00024 | Φ = 40 mm Φ = 13.33 mm Φ = 25 mm Φ = 31 mm No inhibition | [130] |
| Not specified | Chamazulene (31.48%) | Micro-dilution | Gram-positive Staphylococcus aureus (16 strains) Gram-negative Escherichia coli (16 strains) | MIC 90% = 2.9 mg/mL MIC 90% = 28.2 mg/mL | [126] |
| Flower | α-Bisabolol oxide A (48.22%) | Disc diffusion Micro-dilution | Gram-positive Bacillus cereus ATCC 11778 Staphylococcus aureus ATCC 13565 Gram-negative Escherichia coli O157 ATCC 1659 Salmonella typhi ATCC 13076 | Φ ~12–22 mm; MIC = 10 µg/mL Φ ~12–26 mm; MIC = 10 µg/mL Φ ~7–19.5 mm; MIC = 12.5 µg/mL Φ ~10–21 mm; MIC = 12.5 µg/mL | [116] |
| Extracts | |||||
|
Commercial Aerial parts Methanol Ethanol Petroleum ether | Not specified | Well diffusion | Gram-positive Staphylococcus aureus ATCC 25923 Bacillus subtilis NCTC 8236 Gram-negative Escherichia coli ATCC 25922 Pseudomonas aeruginosa ATCC 27853 | Methanol: No inhibition Ethanol: Φ = 19 mm Petroleum ether: Φ = 25 mm Methanol: Φ = 17 mm Ethanol: Φ = 17 nm Petroleum ether: Φ = 26 mm Methanol: Φ = 17 mm Ethanol: Φ = 20 mm Petroleum ether: Φ = 23 mm Methanol: Φ = 17 mm Ethanol: Φ = 18 mm Petroleum ether: Φ = 22 mm | [101] |
| Flower Ethanol | Not specified | Broth microdilution | Gram-positive Staphylococcus aureus MRSA (30 strains) | MIC = 64–128 μg/mL | [140] |
| Leaves Methanol | Not specified | Disc diffusion Micro-dilution | Gram-positive Bacillus cereus LMG 13569 Listeria innocua LMG 1135668 Staphylococcus aureus ATCC 9244 Staphylococcus camorum LMG 13567 Streptococcus pyogenes Enterococcus faecalis CIP 103907 Gram-negative Escherichia coli CIP 11609 Salmonella enteric CIP 105150 Shigella dysenteriae CIP 5451 Proteus mirabilis 104588 CIP Pseudomonas aeruginosa | Φ = 17 mm; MIC = 100; MBC = 100 µg/mL Φ = 20 mm; MIC = 100; MBC ˃ 100 µg/mL Φ = 16 mm; MIC = 100; MBC = 100 µg/mL Φ = 19 mm; MIC = 100; MBC = 100 µg/mL Φ = 18 mm; MIC = 25; MBC = 50 µg/mL Φ = 13 mm; MIC = 100; MBC = 100 µg/mL Φ = 17 mm; MIC = 25; MBC = 25 µg/mL Φ = 17 mm; MIC = 100; MBC = 100 µg/mL Φ = 22 mm; MIC = 25; MBC = 25 µg/mL Φ = 15 mm; MIC = 50; MBC = 50 µg/mL Φ = 20 mm; MIC = 25; MBC = 25 µg/mL | [111] |
|
Flower Ethanol | Not specified | Well diffusion (3.12–50 mg/mL) Micro-dilution | Gram-positive Staphylococcus aureus MRSA (14 strains) Staphylococcus aureus MRSA (6 strains) Staphylococcus aureus ATCC 29213 Gram-negative Pseudomonas aeruginosa ATCC 27,853 and multidrug-resistant (16 strains) | Φ = 10.3–12.7 mm at 25–50 mg/mL MIC = 6.25; MBC = 12.5 mg/mL Φ = 12.3 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL Φ = 12.1 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL No inhibition | [24] |
|
Leaves Ethanol | Not specified | Well diffusion (3.12–50 mg/mL) Micro-dilution | Gram-positive Staphylococcus aureus MRSA (7 strains) Staphylococcus aureus ATCC 29213 Gram-negative Pseudomonas aeruginosa ATCC 27,853 and multidrug-resistant (16 strains) | Φ = 10.1 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL Φ = 9.8 mm at 50 mg/mL MIC–MBC > 50 mg/mL No zone; MIC = 12.5; MBC = 25 mg/mL | [24] |
|
Flower Ethanol | Phenylindolizine (32.82%) | Well diffusion Micro-dilution | Gram-positive Listeria monocytogenes ATCC 19117 Staphylococcus aureus ATCC 25923 Gram-negative Enterococcus faecalis Klebsiella pneumoniae Escherichia coli ATCC 25922 Enterobacter cloacae Acinetobacter baumannii | Φ = 15 mm; MIC = 6.75 mg/mL Inhibition Inhibition Inhibition No inhibition No inhibition No inhibition | [139] |
|
Leaves and flower Methanol Aqueous Chloroform | Phenol content 13.11 mg GAE/g DW 23.96 mg GAE/g DW 9.68 mg GAE/g DW | Disc diffusion | Gram-positive Staphylococcus aureus ATCC 6538 Bacillus sp. Gram-negative Escherichia coli ATCC 4157 Pseudomonas sp. ATCC 9027 | Methanol: Φ = 6 mm Aqueous: Φ = 10 mm Chloroform: No inhibition Methanol: Φ = 9.66 mm Aqueous: Φ = 11.66 mm Chloroform: Φ = 9.33 mm Methanol: Φ = 6 mm Aqueous: Φ = 10.66 mm Chloroform: No inhibition Methanol: Φ = 22.5 mm Aqueous: Φ = 9 mm Chloroform: Φ = 10.33 mm | [134] |
|
Flower Ethanol Cyclohexane | Not specified | Disc diffusion Broth dilution | Gram-positive Staphylococcus aureus ATCC 25923 Gram-negative Pseudomonas aeruginosa ATCC 27853 Escherichia coli ATCC 25922 Salmonella Typhimurium ATCC 14028 | No inhibition Ethanol: Φ = 10 mm; MIC = 1000 mg/mL No inhibition No inhibition | [137] |
|
Flower Ethanol | Phenol content (151.45 mg CAE/mL) | Micro-dilution | Gram-positive Escherichia coli | MIC = 39.1 µg/mL | [118] |
|
Not specified Methanol | 4-Amino- 1,5-pentandioic acid | Well diffusion (50 µL) | Gram-negative Proteus mirabilis | Φ = 6.01 mm | [141] |
|
Not specified Methanol Ethanol Aqueous | Not specified | Well diffusion (250–1000 mg/mL) | Gram-positive Staphylococcus aureus Gram-negative Escherichia coli Proteus sp. Klebsiella sp. | Methanol: Φ = 12 mm at 1000 mg/mL Ethanol: Φ = 15 mm at 1000 mg/mL No inhibition No inhibition No inhibition | [133] |
|
Flower Ethanol | Not specified | Well diffusion (10–100 μg/mL) | Gram-positive Staphylococcus aureus | Φ = 0–28 mm | [138] |
|
Not specified Methanol | Phenol contents (1.24 mg GAE/g) | Disc-diffusion Macro-dilution | Gram-positive Staphylococcus aureus MTCC 7443 Streptococcus mutans MTCC 497 Streptococcus mitis MTCC 2695 Streptococcus oralis MTCC 2696 Lactobacillus acidophilus MTCC 10307 Gram-negative Pseudomonas aeruginosa MTCC 7453 | Φ = 16.2 mm; MIC = 3.12 µg/mL Φ = 19.8 mm; MIC = 0.39 µg/mL Φ = 16.7 mm; MIC = 3.12 µg/mL Φ = 16.03 mm; MIC = 3.12 µg/mL Φ = 9.8 mm; MIC = 0.39 µg/mL No inhibition | [26] |
|
Flower Ethanol (70, 96, 99.8%) Ethyl acetate | Not specified | Micro-dilution | Gram-negative Helicobacter pylori ATCC 43504 | Ethanol: MIC = 62.5; MBC = 125–250 µg/mL Ethyl acetate: MIC = 31.3; MBC = 125 µg/mL | [136] |
|
Not specified Methanol | Not specified | Well diffusion (12.5–200 mg/mL) | Gram-positive Staphylococcus aureus (2 strains) Enterococcus faecalis (3 strains) Enterococcus durans Sp. 33 Gram-negative Proteus mirabilis (3 strains) Salmonella S7 Serratia U11 Providensia alcalifaciens Stenotrophomonas maltophilia | Φ = 9–19 mm at 50–200 mg/mL Φ = 9.5–14 mm at 100–200 mg/mL Φ = 10–13 mm at 100–200 mg/mL Φ = 8–16 mm at 100–200 mg/mL Φ = 8–13 mm at 50–200 mg/mL Φ = 12 mm at 200 mg/mL Φ = 8–12 mm at 100–200 mg/mL Φ = 8 mm at 200 mg/mL | [135] |
|
Flower Methanol | Phenol contents (656.1 mg CAE/g FR) | Diffusion (50 mg/mL) Micro-dilution | Gram-positive Staphylococcus aureus ATCC 6538 p Streptococcus epidermidis ATCC 12228 Gram-negative Pseudomonas aeruginosa ATCC 9027 | Φ = 1.3 mm; MIC = 62.5 μg/mL Φ = 1 mm; MIC = 125 μg/mL Φ = 0.3 mm; MIC = 500 μg/mL | [46] |
|
Aerial parts Aqueous | Not specified | Well diffusion (5–40 mg/mL) | Gram-positive Staphylococcus aureus Gram-negative Escherichia coli | Φ = 0.6–3.55 mm Φ = 0.6–3.6 mm | [143] |
|
Stems Leaves Aqueous | Not specified | Disc diffusion | Gram-positive Staphylococcus aureus Bacillus subtilis Gram-negative Escherichia coli Pseudomonas aeruginosa | Stems: Φ = 22.7 mm Leaves: Φ = 21.8 mm Stems: Φ = 9.2 mm Leaves: Φ = 23.9 mm Stems: Φ = 9.9 mm Leaves: Φ = 23.7 mm Stems: Φ = 27.4 mm Leaves: Φ = 24.9 mm | [145] |
|
Leaves Flower Ethanol | Not specified | Well diffusion Micro-dilution | Gram-negative Pseudomonas aeruginosa multidrug-resistant | Leaves: No zone MIC = 12.5; MBC = 25 mg/mL Flowers: No inhibition | [25] |
|
Not specified Aqueous | Not specified | Disc diffusion (15–25%) | Gram-positive Enterococcus faecalis ATCC 24212 | Φ = 20.62 mm at 25% | [144] |
|
Flower Methanol Ethanol Hexane Diethyl ether | Phenol content (3.7 mg GAE/g) (3.5 mg GAE/g) (2.4 mg GAE/g) (3.3 mg GAE/g) | Disc diffusion (7.5–20 µg/disc) Micro-dilution | Gram-positive Bacillus cereus ATCC 11778 Staphylococcus aureus ATCC 13565 Gram-negative Escherichia coli O157 ATCC 1659 Salmonella typhi ATCC 13076 | Methanol: Φ = 9–20 mm; MIC = 12.5 µg/mL Ethanol: Φ = 10–20 mm; MIC = 12.5 µg/mL Hexane: Φ = 9–21 mm; MIC = 12.5 µg/mL Diethyl ether: Φ = 7–18 mm; MIC=15 µg/mL Methanol: Φ = 11–19 mm; MIC=12.5 µg/mL Ethanol: Φ = 13–23 mm; MIC = 12.5 µg/mL Hexane: Φ = 10–23 mm; MIC = 10 µg/mL Diethyl ether: Φ = 8–19 mm; MIC = 15 µg/mL Methanol: Φ = 8–18 mm; MIC = 15 µg/mL Ethanol: Φ = 9–19 mm; MIC = 15 µg/mL Hexane: Φ = 8–19 mm; MIC = 15 µg/mL Diethyl ether: Φ = 7–15 mm; MIC = 17.5 µg/mL Methanol: Φ = 11–20 mm; MIC = 15 µg/mL Ethanol: Φ = 8–17 mm; MIC = 15 µg/mL Hexane: Φ = 8–19 mm; MIC = 15 µg/mL Diethyl ether: Φ = 6–16 mm; MIC = 15 µg/mL | [115] |
|
Flower Ethanol | Not specified | Well diffusion (3.12–50 mg/mL) Micro-dilution | Gram-positive Staphylococcus aureus MRSA (14 strains) Staphylococcus aureus MRSA (6 strains) Staphylococcus aureus ATCC 29213 | Φ = 10.3–12.7 mm at 25–50 mg/mL MIC = 6.25; MBC = 12.5 mg/mL Φ = 12.3 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL Φ = 12.1 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL | [23] |
|
Leaves Ethanol | Not specified | Well diffusion (3.12–50 mg/mL) Micro-dilution | Gram-positive Staphylococcus aureus MRSA (7 strains) Staphylococcus aureus ATCC 29213 | Φ = 10.1 mm at 50 mg/mL MIC = 12.5; MBC = 25 mg/mL Φ = 9.8 mm at 50 mg/mL MIC–MBC > 50 mg/mL | [23] |
|
Leaves Methanol | Not specified | Well diffusion Micro-dilution | Gram-positive Propionibacterium acnes ATCC 11827 Staphylococcus aureus ATCC 6538P Bacillus subtilis MTCC 736 Kocuria sp KM 24375 Gram-negative Escherichia coli ATCC 8739 Pseudomonas aeruginosa ATCC 9027 | Φ = 6 mm; MIC = 0.156 mg/mL No inhibition No inhibition No inhibition No inhibition No inhibition | [142] |
4.3. Antifungal Activity
| Part Used | Main Component | Experimental Method | Tested Organism | Key Results | References |
|---|---|---|---|---|---|
| Essential Oil | |||||
| Leaves | Not specified | Disc diffusion Micro-dilution | Candida albicans ATCC 10231 Candida albicans Aspergillus niger Aspergillus sp. | Φ = 20 mm; MIC = MFC = 1 µg/mL Φ = 19 mm; MIC = MFC = 2 µg/mL Φ = 17 mm; MIC = MFC = 2 µg/mL Φ = 14 mm; MIC = 16; MFC > 16 µg/mL | [111] |
| Flower | Cis-β-farnesene (27%) | Agar dilution (25–100 µL/mL) | Aspergillus flavus AFl375 Aspergillus niger FC24771 Aspergillus terreus Y.H. Yeh V0103 Fusarium culmorum CBS 128537 | Φ = 10.66–52.33% Φ = 89.66–100% Φ = 87–84% Φ = 91–86.66% | [88] |
| Aerial parts (95% flowers) | Trans-β-farnesene (18.7–38.5%) α-Bisabolol (38.3%) α-Bisabolol oxide A (25%) | Macro-dilution | Candida albicans ATCC 10231 | MFC = 2000–4000 µg/mL | [27] |
| Flower | α-Bisabolol oxide A (48.22%) | Disc diffusion (7.5–20 µg/disc) Micro-dilution | Candida albicans ATCC 10231 Aspergillus flavus ATCC 16875 | Φ ~14–26 mm; MIC = 19 µg/mL Φ ~9–23 mm; MIC = 12.5 µg/mL | [115] |
| Aerial parts | Trans-β-farnesene (42.2%) | Micro-dilution | Candida albicans ATCC 10231 Aspergillus niger ATCC 16888 | MIC = 313 μg/mL MIC = 625 μg/mL | [131] |
| Flower | α-Pinene (22.10%) | Broth dilution | Candida albicans (30 resistant) Candida albicans (30 susceptible) | MIC = 1700; MFC = 2300 μg/mL MIC = 1550; MFC = 2200 μg/mL | [146] |
| Extracts | |||||
| Leaves Methanol | Not specified | Disc diffusion Micro-dilution | Candida albicans ATCC 10231 Candida albicans Aspergillus niger Aspergillus sp. | Φ = 15 mm; MIC = MFC = 100 µg/mL Φ = 15 mm; MIC = MFC = 100 µg/mL Φ = 14 mm; MIC = 100; MFC > 100 µg/mL Φ = 13 mm; MIC = 200; MFC > 200 µg/mL | [111] |
| Leaves and flower Methanol Aqueous Chloroform | Phenols | Disc diffusion | Candida albicans ATCC 24433 Fusarium sp. | Chloroform: Φ = 6 mm Other extracts: No inhibition Chloroform: Φ = 6 mm Other extracts: No inhibition | [134] |
| Flower Ethanol | Phenol content (151.45 mg CAE/mL) | Micro-dilution | Aspergillus niger | MIC = 39.1 µg/mL | [117] |
| Not specified Methanol | 4-Amino- 1,5-pentandioic acid | Well diffusion | Aspergillus terreus | Φ = 5.89 mm | [141] |
| Flower Alcohol 70% | Not specified | Spectrophotometer device | Saccharomyces cerevisiae | Growth decrease = 48% at 3000 μg/mL | [148] |
| Aerial part Aqueous | Not specified | Well diffusion | Candida albicans | Φ = 0.26–2.56 mm; MIC = 5–40% | [143] |
| Seeds Aqueous Sulfated derivatives | Phenol content (16.4–19.7 mg GAE/g) (19.2–22.4 mg GAE/g) | Disc diffusion | Penicillium citrinum Aspergillus niger | Aqueous: Φ = 10–12 mm Sulfated derivatives: Φ = 10–12 mm Aqueous: Φ = 10 mm Sulfated derivatives: Φ = 7–10 mm | [149] |
| Not specified Aqueous | Not specified | Disc diffusion (15–25%) | Candida albicans ATCC 24433 | Φ = 24.16 mm at concentration of 25% | [144] |
| Flower Methanol Ethanol Hexane Diethyl ether | Not specified | Disc diffusion (7.5–20 µg/disc) Micro-dilution | Candida albicans ATCC 10231 Aspergillus flavus ATCC 16875 | Methanol: Φ = 15–23 mm; MIC = 10 µg/mL Ethanol: Φ = 8–21 mm; MIC = 12.5 µg/mL Hexane: Φ = 9–23 mm; MIC = 10 µg/mL Diethyl ether: Φ = 8–20 mm; MIC = 15 µg/mL Methanol: Φ = 18–24 mm; MIC = 12.5 µg/mL Ethanol: Φ = 11–18 mm; MIC = 15 µg/mL Hexane: Φ = 8–20 mm; MIC = 12.5 µg/mL Diethyl ether: Φ = 6–21 mm; MIC = 17.5µg/mL | [115] |
| Other | |||||
| Flower | Peptide AMP1 | Broth microdilution | Candida albicans Aspergillus sp. | MIC = 3.33–6.66 μmol MIC = 6.66–13.32 μmol | [150] |
4.4. Antiparasitic and Insecticidal Activities
4.5. Antidiabetic Activity
4.6. Anti-Tumoral Activity
4.7. Anti-Inflammatory Activity
5. Phyotherapeutical Applications
6. Other Applications
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azino-bis-(3-ethylbenzothiazoline-6-sulfonic acid) |
| ATCC | American Type Culture Collection |
| BHT | butylated hydroxytoluene |
| CAE | Chlorogenic Acid Equivalent |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| DW | Dry Weight |
| EC50 | Effective control to 50% growth inhibition |
| EO | Essential Oil |
| FRAP | Ferric Reducing Antioxidant Power |
| FW | Fresh Weight |
| GAE | Gallic Acid Equivalent |
| GMCA | β-d-glucopyranosyloxy-4-methoxycinnamic acid |
| IC50 | Half maximal Inhibitory Concentration |
| LC50 | Lethal Concentration necessary to kill 50% of the population |
| LD50 | Lethal Dose necessary to kill 50% of the population |
| MBC | Minimum Bactericidal Concentration |
| MFC | Minimum Fungicidal Concentration |
| MIC | Minimum Inhibitory Concentration |
| MRSA | Methicillin-Resistant Staphylococcus aureus |
| QE | Quercetin Equivalent |
| TE | Trolox Equivalent |
| TEAC | Trolox Equivalent Antioxidant Capacity |
References
- Lim, T.K. Matricaria chamomilla. In Edible Medicinal and Non-Medicinal Plants; Springer: Dordrecht, The Netherlands, 2014; Volume 7, pp. 397–431. [Google Scholar] [CrossRef]
- Singh, O.; Khanam, Z.; Misra, N.; Srivastava, M.K. Chamomile (Matricaria chamomilla L.): An overview. Pharmacogn. Rev. 2011, 5, 82–95. [Google Scholar] [CrossRef] [PubMed]
- Menale, B.; De Castro, O.; Di Iorio, E.; Ranaldi, M.; Muoio, R. Discovering the ethnobotanical traditions of the island of Procida (Campania, southern Italy). Plant Biosyst. An Int. J. Deal. All Asp. Plant Biol. 2021, 1–19. [Google Scholar] [CrossRef]
- Güzel, Y.; Güzelşemme, M.; Miski, M. Ethnobotany of medicinal plants used in Antakya: A multicultural district in Hatay Province of Turkey. J. Ethnopharmacol. 2015, 174, 118–152. [Google Scholar] [CrossRef] [PubMed]
- Živković, J.; Ilić, M.; Šavikin, K.; Zdunić, G.; Ilić, A.; Stojković, D. Traditional use of medicinal plants in South-Eastern Serbia (Pčinja District): Ethnopharmacological investigation on the current status and comparison with half a century old data. Front. Pharmacol. 2020, 11, 1020. [Google Scholar] [CrossRef] [PubMed]
- Neves, J.M.; Matos, C.; Moutinho, C.; Queiroz, G.; Gomes, L.R. Ethnopharmacological notes about ancient uses of medicinal plants in Trás-os-Montes (northern of Portugal). J. Ethnopharmacol. 2009, 124, 270–283. [Google Scholar] [CrossRef]
- Mikou, K.; Rachiq, S.; Jarrar Oulidi, A. Étude ethnobotanique des plantes médicinales et aromatiques utilisées dans la ville de Fès au Maroc. Phytotherapie 2016, 14, 35–43. [Google Scholar] [CrossRef]
- Petrakou, K.; Iatrou, G.; Lamari, F.N. Ethnopharmacological survey of medicinal plants traded in herbal markets in the Peloponnisos, Greece. J. Herb. Med. 2020, 19, 100305. [Google Scholar] [CrossRef]
- Orav, A.; Raal, A.; Arak, E. Content and composition of the essential oil of Chamomilla recutita (L.) Rauschert from some European countries. Nat. Prod. Res. 2010, 24, 48–55. [Google Scholar] [CrossRef]
- Piri, E.; Sourestani, M.M.; Khaleghi, E.; Mottaghipisheh, J.; Zomborszki, Z.P.; Hohmann, J.; Csupor, D. Chemo-diversity and antiradical potential of twelve Matricaria chamomilla L. populations from Iran: Pproof of ecological effects. Molecules 2019, 24, 1315. [Google Scholar] [CrossRef]
- Ghasemi, M.; Jelodar, N.B.; Modarresi, M.; Bagheri, N.; Jamali, A. Increase of chamazulene and α-bisabolol contents of the essential oil of german chamomile (Matricaria chamomilla L.) using salicylic acid treatments under normal and heat stress conditions. Foods 2016, 5, 56. [Google Scholar] [CrossRef]
- Mavandi, P.; Assareh, M.H.; Dehshiri, A.; Rezadoost, H.; Abdossi, V. Flower biomass, essential oil production and chemotype identification of some Iranian Matricaria chamomilla Var. recutita (L.) accessions and commercial varieties. J. Essent. Oil-Bear. Plants 2019, 22, 1228–1240. [Google Scholar] [CrossRef]
- Abbas, A.M.; Seddik, M.A.; Gahory, A.A.; Salaheldin, S.; Soliman, W.S. Differences in the aroma profile of chamomile (Matricaria chamomilla L.) after different drying conditions. Sustainability 2021, 13, 5083. [Google Scholar] [CrossRef]
- Homami, S.S.; Jaimand, K.; Rezaee, M.B.; Afzalzadeh, R. Comparative studies of different extraction methods of essential oil from Matricaria recutita L. in Iran. J. Chil. Chem. Soc. 2016, 61, 2982–2984. [Google Scholar] [CrossRef]
- Zarezadeh, S.; Riahi, H.; Shariatmadari, Z.; Sonboli, A. Effects of cyanobacterial suspensions as bio-fertilizers on growth factors and the essential oil composition of chamomile, Matricaria chamomilla L. J. Appl. Phycol. 2020, 32, 1231–1241. [Google Scholar] [CrossRef]
- Qureshi, M.N.; Stecher, G.; Bonn, G.K. Quality control of herbs: Determination of amino acids in Althaea officinalis, Matricaria chamomilla and Taraxacum officinale. Pak. J. Pharm. Sci. 2014, 27, 459–462. [Google Scholar]
- Petrulova, V.; Vilkova, M.; Kovalikova, Z.; Sajko, M.; Repcak, M. Ethylene Induction of non-enzymatic metabolic antioxidants in Matricaria chamomilla. Molecules 2020, 25, 5720. [Google Scholar] [CrossRef]
- Kováčik, J.; Klejdus, B.; Hedbavny, J.; Štork, F.; Bačkor, M. Comparison of cadmium and copper effect on phenolic metabolism, mineral nutrients and stress-related parameters in Matricaria chamomilla plants. Plant Soil 2009, 320, 231–242. [Google Scholar] [CrossRef]
- Hassanpour, H.; Ghanbarzadeh, M. Induction of cell division and antioxidative enzyme activity of Matricaria chamomilla L. cell line under clino-rotation. Plant Cell. Tissue Organ Cult. 2021, 146, 215–224. [Google Scholar] [CrossRef]
- Molnar, M.; Mendešević, N.; Šubarić, D.; Banjari, I.; Jokić, S. Comparison of various techniques for the extraction of umbelliferone and herniarin in Matricaria chamomilla processing fractions. Chem. Cent. J. 2017, 11, 78. [Google Scholar] [CrossRef]
- Kolodziejczyk-czepas, J.; Bijak, M.; Saluk, J.; Ponczek, M.B.; Zbikowska, H.M.; Nowak, P.; Tsirigotis-maniecka, M. Radical scavenging and antioxidant effects of Matricaria chamomilla polyphenolic—Polysaccharide conjugates. Int. J. Biol. Macromol. 2015, 72, 1152–1158. [Google Scholar] [CrossRef]
- Owlia, P.; Rosooli, I.; Saderi, H.; Aliahmadi, M. Retardation of biofilm formation with reduced productivity of alginate as a result of Pseudomonas aeruginosa exposure to Matricaria chamomilla essentiel oil. J. Pharmacogn. Mag. 2007, 10, 83. [Google Scholar]
- Sadat, S.S.; Ahani Azari, A.; Mazandarani, M. Evaluation of antibacterial activity of ethanolic extract of Matricaria chamomilla, Malva sylvestris and Capsella bursa-pastoris against methicillin-resistant Staphylococcus aureus. J. Med. Microbiol. Infect. Dis. 2021, 8, 127–131. [Google Scholar] [CrossRef]
- Ahani Azari, A.; Danesh, A. Antibacterial effect of Matricaria chamomilla alcoholic extract against drug-resistant isolates of Staphylococcus aureus and Pseudomonas aeruginosa. Infect. Epidemiol. Microbiol. 2021, 7, 29–35. [Google Scholar] [CrossRef]
- Poudineh, F.; Azari, A.A.; Fozouni, L. Antibacterial activity of ethanolic extract of Matricaria chamomilla, Malva cylvestris, and Capsella bursa-pastoris against multidrug-resistant Pseudomonas aeruginosa strains. Hamadan Univ. Med. Sci. 2021, 8, 23–26. [Google Scholar] [CrossRef]
- Lavanya, J.; Periyar Selvam, S.; Jeevitha Priya, M.; Jacintha, P.; Aradana, M. Antioxidant and antimicrobial activity of selected medicinal plants against human oral pathogens. Int. J. Pharm. Pharm. Sci. 2016, 8, 71–78. [Google Scholar] [CrossRef]
- Höferl, M.; Wanner, J.; Tabanca, N.; Ali, A.; Gochev, V.; Schmidt, E.; Kaul, V.K.; Singh, V.; Jirovetz, L. Biological activity of Matricaria chamomilla essential oils of various chemotypes. Planta Med. Int. Open 2020, 7, e114–e121. [Google Scholar] [CrossRef]
- Al-Mekhlafi, F.A.; Abutaha, N.; Al-Doaiss, A.A.; Ahmed Al- Keridis, L.; Alsayadi, A.I.; Ali El Hadi Mohamed, R.; Wadaan, M.A.; Elfaki Ibrahim, K.; Al-Khalifa, M.S. Target and non-target effects of Foeniculum vulgare and Matricaria chamomilla combined extract on Culex pipiens mosquitoes. Saudi J. Biol. Sci. 2021, 28, 5773–5780. [Google Scholar] [CrossRef]
- Villa-Rodriguez, J.A.; Kerimi, A.; Abranko, L.; Tumova, S.; Ford, L.; Blackburn, R.S.; Rayner, C.; Williamson, G. Acute metabolic actions of the major polyphenols in chamomile: An in vitro mechanistic study on their potential to attenuate postprandial hyperglycaemia. Sci. Rep. 2018, 8, 5471. [Google Scholar] [CrossRef]
- Ali, E.M. Phytochemical composition, antifungal, antiaflatoxigenic, antioxidant, and anticancer activities of Glycyrrhiza glabra L. and Matricaria chamomilla L. essential oils. J. Med. Plants Res. 2013, 7, 2197–2207. [Google Scholar] [CrossRef]
- Nikseresht, M.; Kamali, A.M.; Rahimi, H.R.; Delaviz, H.; Toori, M.A.; Kashani, I.R.; Mahmoudi, R. The hydroalcoholic extract of Matricaria chamomilla suppresses migration and invasion of human breast cancer MDA-MB-468 and MCF-7 cell lines. Pharmacogn. Res. 2017, 9, 87–95. [Google Scholar] [CrossRef]
- Asadi, Z.; Ghazanfari, T.; Hatami, H. Anti-inflammatory effects of Matricaria chamomilla extracts on BALB/c mice macrophages and lymphocytes. Iran. J. Allergy Asthma Immunol. 2020, 19, 63–73. [Google Scholar] [CrossRef]
- Asgharzade, S.; Rabiei, Z.; Rafieian-Kopaei, M. Effects of Matricaria chamomilla extract on motor coordination impairment induced by scopolamine in rats. Asian Pac. J. Trop. Biomed. 2015, 5, 829–833. [Google Scholar] [CrossRef]
- Golkhani, S.; Vahdati, A.; Modaresi, M. The effects of Matricaria chamomilla extract during neonatal period of rats on pituitary-gonadal hormone axis and changes in testicular tissue of male progenies. Middle East J. Fam. Med. 2017, 15, 126–132. [Google Scholar] [CrossRef][Green Version]
- Rafraf, M.; Zemestani, M.; Asghari-Jafarabadi, M. Effectiveness of chamomile tea on glycemic control and serum lipid profile in patients with type 2 diabetes. J. Endocrinol. Investig. 2015, 38, 163–170. [Google Scholar] [CrossRef]
- Bayliak, M.M.; Dmytriv, T.R.; Melnychuk, A.V.; Strilets, N.V.; Storey, K.B.; Lushchak, V.I. Chamomile as a potential remedy for obesity and metabolic syndrome. Excli J. 2021, 20, 1261–1286. [Google Scholar] [CrossRef]
- Awaad, A.A.; El-Meligy, R.M.; Zain, G.M.; Safhi, A.A.; AL Qurain, N.A.; Almoqren, S.S.; Zain, Y.M.; Sesh Adri, V.D.; Al-Saikhan, F.I. Experimental and clinical antihypertensive activity of Matricaria chamomilla extracts and their angiotensin-converting enzyme inhibitory activity. Phyther. Res. 2018, 32, 1564–1573. [Google Scholar] [CrossRef]
- Silveira, E.S.; Bezerra, S.B.; Ávila, K.S.; Rocha, T.M.; Pinheiro, R.G.; de Queiroz, M.G.R.; Magalhães, P.J.C.; Santos, F.A.; Leal, L.K.A.M. Gastrointestinal effects of standardized Brazilian phytomedicine (Arthur de Carvalho Drops®) containing Matricaria recutita, Gentiana lutea and Foeniculum vulgare. Pathophysiology 2019, 26, 349–359. [Google Scholar] [CrossRef]
- Morshedi, M.; Gol, A.; Mohammadzadeh, A. The effect of Matricaria chamomilla on the treatment of ibuprofen-induced gastric ulcers in male rats. Hormozgan Med. J. 2016, 20, 270–275. [Google Scholar]
- Sebai, H.; Jabri, M.A.; Souli, A.; Rtibi, K.; Selmi, S.; Tebourbi, O.; El-Benna, J.; Sakly, M. Antidiarrheal and antioxidant activities of chamomile (Matricaria recutita L.) decoction extract in rats. J. Ethnopharmacol. 2014, 152, 327–332. [Google Scholar] [CrossRef]
- Chandrashekhar, V.M.; Halagali, K.S.; Nidavani, R.B.; Shalavadi, M.H.; Biradar, B.S.; Biswas, D.; Muchchandi, I.S. Anti-allergic activity of German chamomile (Matricaria recutita L.) in mast cell mediated allergy model. J. Ethnopharmacol. 2011, 137, 336–340. [Google Scholar] [CrossRef]
- Dos Santos, D.S.; de Barreto, R.S.S.; Serafini, M.R.; Gouveia, D.N.; Marques, R.S.; de Nascimento, L.C.; de Nascimento, J.C.; Guimarães, A.G. Phytomedicines containing Matricaria species for the treatment of skin diseases: A biotechnological approach. Fitoterapia 2019, 138, 104267. [Google Scholar] [CrossRef] [PubMed]
- Bigagli, E.; Cinci, L.; D’Ambrosio, M.; Luceri, C. Pharmacological activities of an eye drop containing Matricaria chamomilla and Euphrasia officinalis extracts in UVB-induced oxidative stress and inflammation of human corneal cells. J. Photochem. Photobiol. B Biol. 2017, 173, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Aitken-Saavedra, J.; Chaves Tarquinio, S.B.; De Oliveira Da Rosa, W.L.; Fernandes Da Silva, A.; Almeida MacHado, B.M.E.; Santos Castro, I.; Oliveira Wennesheimer, A.; Morales-Bozo, I.; Uchoa Vasconcelos, A.C.; Neutzling Gomes, A.P. Effect of a homemade salivary substitute prepared using chamomile Matricaria chamomilla L. flower and flax Linum usitatissimum L. seed to relieve primary burning mouth syndrome: A preliminary report. J. Altern. Complement. Med. 2020, 26, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Saidi, R.; Heidari, H.; Sedehi, M.; Safdarian, B. Evaluating the effect of Matricaria chamomilla and Melissa officinalis on pain intensity and satisfaction with pain management in patients after orthopedic surgery. J. HerbMed Pharmacol. 2020, 9, 339–345. [Google Scholar] [CrossRef]
- Niknam, S.; Tofighi, Z.; Faramarzi, M.A.; Abdollahifar, M.A.; Sajadi, E.; Dinarvand, R.; Toliyat, T. Polyherbal combination for wound healing: Matricaria chamomilla L. and Punica granatum L. DARU J. Pharm. Sci. 2021, 29, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Abd-Allah, D.; Salah-Eldin, A. Amerolative influence of chamomile (Matricaria recutita L.) on synthetic food additive induced probable toxicity in male albino rats. J. Food Dairy Sci. 2021, 12, 161–170. [Google Scholar] [CrossRef]
- Jabri, M.A.; Aissani, N.; Tounsi, H.; Sakly, M.; Marzouki, L.; Sebai, H. Protective effect of chamomile (Matricaria recutita L.) decoction extract against alcohol-induced injury in rat gastric mucosa. Pathophysiology 2017, 24, 1–8. [Google Scholar] [CrossRef]
- Afrigan, L.; Jafari Anarkooli, I.; Sohrabi, D.; Abdanipour, A.; Yazdinezhad, A.; Sayyar, Z.; Ghorbanlou, M.; Arianmanesh, M. The effect of hydroethanolic extract of Matricaria chamomilla on the reproductive system of male rats exposed to formaldehyde. Andrologia 2019, 51, e13362. [Google Scholar] [CrossRef]
- Al-Niaeem, K.S.; Abdulrahman, N.M.A.; Attee, R.S. Using of anise (Pimpinella anisum) and chamomile (Matricaria chamomilla) powders for common carp Cyprinus carpio L. anesthesia. Biol. Appl. Environ. Res. 2019, 3, 111–117. [Google Scholar]
- Alsaadi, S.A.R.A.; Al-Perkhdri, A.S.A.; Al-Hadeedy, I.Y.H. Effects of Matricaria chamomilla flower aqueous extract on some hematological, biochemical parameters and carcass traits in Iraqi local rabbits. Plant Arch. 2020, 20, 1044–1049. [Google Scholar]
- Caleja, C.; Barros, L.; Antonio, A.L.; Oliveira, M.B.P.P.; Ferreira, I.C.F.R. A comparative study between natural and synthetic antioxidants: Evaluation of their performance after incorporation into biscuits. Food Chem. 2017, 216, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Ghoniem, A.A.; El-Hai, K.M.A.; El-Khateeb, A.Y.; Eldadamony, N.M.; Mahmoud, S.F.; Elsayed, A. Enhancing the potentiality of Trichoderma harzianum against pythium pathogen of beans using chamomile (Matricaria chamomilla, L.) flower extract. Molecules 2021, 26, 1178. [Google Scholar] [CrossRef] [PubMed]
- Shadizadeh, S.S.; Kharrat, R. Experimental investigation of Matricaria chamomilla extract effect on oil-water interfacial tension: Usable for chemical enhanced oil recovery. Pet. Sci. Technol. 2015, 33, 901–907. [Google Scholar] [CrossRef]
- Ugi, B.U.; Abeng, F.E.; Obeten, M.E.; Uwah, I.E. Management of aqueous corrosion of federated mild steel (Local constructional steel) at elevated temperatures employing environmentally friendly inhibitors: Matricaria chamomilla plant. Int. J. Chem. Sci. 2019, 3, 6–12. [Google Scholar]
- Das, S.; Horváth, B.; Šafranko, S.; Jokić, S.; Széchenyi, A.; Koszegi, T. Antimicrobial activity of chamomile essential oil: Effect of different formulations. Molecules 2019, 24, 4321. [Google Scholar] [CrossRef] [PubMed]
- Kareem, P.A. Silver nanoparticles synthesized by using Matricaria chamomilla extract and effect on bacteria isolated from dairy products. Diyala J. Pure Sci. 2018, 14, 176–187. [Google Scholar] [CrossRef]
- Karam, T.K.; Ortega, S.; Ueda Nakamura, T.; Auzély-Velty, R.; Nakamura, C.V. Development of chitosan nanocapsules containing essential oil of Matricaria chamomilla L. for the treatment of cutaneous leishmaniasis. Int. J. Biol. Macromol. 2020, 162, 199–208. [Google Scholar] [CrossRef]
- Caleja, C.; Ribeiro, A.; Barros, L.; Barreira, J.C.M.; Antonio, A.L.; Oliveira, M.B.P.P.; Barreiro, M.F.; Ferreira, I.C.F.R. Cottage cheeses functionalized with fennel and chamomile extracts: Comparative performance between free and microencapsulated forms. Food Chem. 2016, 199, 720–726. [Google Scholar] [CrossRef]
- Dadashpour, M.; Firouzi-Amandi, A.; Pourhassan-Moghaddam, M.; Maleki, M.J.; Soozangar, N.; Jeddi, F.; Nouri, M.; Zarghami, N.; Pilehvar-Soltanahmadi, Y. Biomimetic synthesis of silver nanoparticles using Matricaria chamomilla extract and their potential anticancer activity against human lung cancer cells. Mater. Sci. Eng. C 2018, 92, 902–912. [Google Scholar] [CrossRef]
- Alshehri, A.A.; Malik, M.A. Phytomediated photo-induced green synthesis of silver nanoparticles using Matricaria chamomilla L. and its catalytic activity against rhodamine B. Biomolecules 2020, 10, 1604. [Google Scholar] [CrossRef]
- Dogru, E.; Demirbas, A.; Altinsoy, B.; Duman, F.; Ocsoy, I. Formation of Matricaria chamomilla extract-incorporated Ag nanoparticles and size-dependent enhanced antimicrobial property. J. Photochem. Photobiol. B Biol. 2017, 174, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Caleja, C.; Barros, L.; Antonio, A.L.; Carocho, M.; Oliveira, M.B.P.P.; Ferreira, I.C.F.R. Fortification of yogurts with different antioxidant preservatives: A comparative study between natural and synthetic additives. Food Chem. 2016, 210, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.E.; Ali, S.M.E.H.; EL-Rhman, S.A.E.A.; El Mohamed, M.A. Brine-shrimp lethality bioassay of different extracts of the medicinal plant matricaria (chamomilla) flowers. Res. Sq. 2020, 10–15. [Google Scholar] [CrossRef]
- Franke, R.; Schilcher, H. Chamomile: Industrial Profiles; CRC Press: Boca Raton, FL, USA, 2005; 304p. [Google Scholar]
- Leung, A.; Foster., S.F. Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics; John Wiley and Sons: New York, NY, USA, 1996; Volume 38, 649p. [Google Scholar]
- Franz, C.; Bauer, R.; Carle, R.; Tedesco, D. Study on the assessment of plants/herbs, plant/herb extracts and their naturally or synthetically produced components as ‘additives’ for use in animal production. EFSA Support. Publ. 2007, 4, 070828. [Google Scholar]
- Alberts, W.G. German Chamomile Production; Directorate Agricultural Information Services. Department of Agriculture; Private Bag X: Pretoria, South Africa, 2009; Volume 144, 19p. [Google Scholar]
- Misra, N.; Luthra, R.; Singh, K.L.; Kumar, S.; Kiran, L. Recent Advances in Biosynthesis of Alkaloids; Elsevier: Amsterdam, The Netherlands, 1999; pp. 25–59. [Google Scholar]
- Mckay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phyther. Res. An Int. J. Devoted to Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2006, 20, 519–530. [Google Scholar] [CrossRef]
- Fennane, M. Etude phytoécologique des tétraclinaies marocaines. Thèse de Doctorat. Aix-Marseille 3. Phytosociologie des tétraclinaies marocaines. Bull. L’institutSci. Rabat 1987, 12, 99–148. [Google Scholar]
- Mrabti, H.N.; Bouyahya, A.; Naceiri Mrabti, N.; Jaradat, N.; Doudach, L.; Faouzi, M.E.A. Ethnobotanical survey of medicinal plants used by traditional healers to treat diabetes in the Taza region of Morocco. Evid.-Based Complement. Altern. Med. 2021, 2021, 5515634. [Google Scholar] [CrossRef]
- Mrabti, H.N.; Jaradat, N.; Kachmar, M.R.; Ed-Dra, A.; Ouahbi, A.; Cherrah, Y.; El Abbes Faouzi, M. Integrative herbal treatments of diabetes in Beni Mellal region of Morocco. J. Integr. Med. 2019, 17, 93–99. [Google Scholar] [CrossRef]
- Eddouks, M.; Ajebli, M.; Hebi, M. Ethnopharmacological survey of medicinal plants used in Daraa-Tafilalet region (Province of Errachidia), Morocco. J. Ethnopharmacol. 2017, 198, 516–530. [Google Scholar] [CrossRef]
- Idm’hand, E.; Msanda, F.; Cherifi, K. Ethnobotanical study and biodiversity of medicinal plants used in the Tarfaya Province, Morocco. Acta Ecol. Sin. 2020, 40, 134–144. [Google Scholar] [CrossRef]
- Benítez, G.; González-Tejero, M.R.; Molero-Mesa, J. Pharmaceutical ethnobotany in the western part of Granada province (southern Spain): Ethnopharmacological synthesis. J. Ethnopharmacol. 2010, 129, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Parada, M.; Carrió, E.; Bonet, M.À.; Vallès, J. Ethnobotany of the Alt Empordà region (Catalonia, Iberian Peninsula). Plants used in human traditional medicine. J. Ethnopharmacol. 2009, 124, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Tuttolomondo, T.; Licata, M.; Leto, C.; Savo, V.; Bonsangue, G.; Letizia Gargano, M.; Venturella, G.; La Bella, S. Ethnobotanical investigation on wild medicinal plants in the Monti Sicani Regional Park (Sicily, Italy). J. Ethnopharmacol. 2014, 153, 568–586. [Google Scholar] [CrossRef] [PubMed]
- Di Novella, R.; Di Novella, N.; De Martino, L.; Mancini, E.; De Feo, V. Traditional plant use in the National Park of Cilento and Vallo di Diano, Campania, Southern, Italy. J. Ethnopharmacol. 2013, 145, 328–342. [Google Scholar] [CrossRef]
- Marković, M.S.; Pljevljakušić, D.S.; Nikolić, B.M.; Miladinović, D.L.; Djokić, M.M.; Rakonjac, L.B.; Stankov Jovanović, V.P. Ethnoveterinary knowledge in Pirot County (Serbia). S. Afr. J. Bot. 2021, 137, 278–289. [Google Scholar] [CrossRef]
- Janaćković, P.; Gavrilović, M.; Savić, J.; Marin, P.D.; Stevanović, Z.D. Traditional knowledge on plant use from Negotin Krajina (Eastern Serbia): An ethnobotanical study. Indian J. Tradit. Knowl. 2019, 18, 25–33. [Google Scholar]
- Živković, J.; Ilić, M.; Zdunić, G.; Jovanović-Lješković, N.; Menković, N.; Šavikin, K. Traditional use of medicinal plants in Jablanica district (South-Eastern Serbia): Ethnobotanical survey and comparison with scientific data. Genet. Resour. Crop Evol. 2021, 68, 1655–1674. [Google Scholar] [CrossRef]
- Šavikin, K.; Zdunić, G.; Menković, N.; Živković, J.; Ćujić, N.; Tereščenko, M.; Bigović, D. Ethnobotanical study on traditional use of medicinal plants in South-Western Serbia, Zlatibor district. J. Ethnopharmacol. 2013, 146, 803–810. [Google Scholar] [CrossRef]
- Zlatković, B.K.; Bogosavljević, S.S.; Radivojević, A.R.; Pavlović, M.A. Traditional use of the native medicinal plant resource of Mt. Rtanj (Eastern Serbia): Ethnobotanical evaluation and comparison. J. Ethnopharmacol. 2014, 151, 704–713. [Google Scholar] [CrossRef]
- Pieroni, A. Traditional uses of wild food plants, medicinal plants, and domestic remedies in Albanian, Aromanian and Macedonian villages in South-Eastern Albania. J. Herb. Med. 2017, 9, 81–90. [Google Scholar] [CrossRef]
- Kozuharova, E.; Lebanova, H.; Getov, I.; Benbassat, N.; Napier, J. Descriptive study of contemporary status of the traditional knowledge on medicinal plants in Bulgaria. Afr. J. Pharm. Pharmacol. 2013, 7, 185–198. [Google Scholar] [CrossRef]
- El-Assri, E.-M.; Eloutassi, N.; El Barnossi, A.; Bakkari, F.; Hmamou, A.; Bouia, A. Wild chamomile (Matricaria recutita L) from the Taounate Province, Morocco: Extraction and valorisation of the antibacterial activity of its essential oils. Trop. J. Nat. Prod. Res. 2021, 5, 883–888. [Google Scholar] [CrossRef]
- EL-Hefny, M.; Abo Elgat, W.A.A.; Al-Huqail, A.A.; Ali, H.M. Essential and recovery oils from Matricaria chamomilla flowers as environmentally friendly fungicides against four fungi isolated from cultural heritage objects. Processes 2019, 7, 809. [Google Scholar] [CrossRef]
- Ayran, İ.; Çelik, S.A.; Kan, A.; Kan, Y. Essential oil yield and compositions of chamomile (Matricaria chamomilla L.) culıivated in different province of Turkey. Int. J. Agric. Environ. Food Sci. 2018, 2, 202–203. [Google Scholar] [CrossRef]
- Berechet, M.D.; Manaila, E.; Stelescu, M.D.; Craciun, G. The composition of essential oils obtained from Achillea millefolium and Matricaria chamomilla L., Originary from Romania. Rev. Chim. 2017, 68, 2787–2795. [Google Scholar] [CrossRef]
- Demarque, D.P.; Sabóia, J.F.; Fabri, J.R.; Carollo, C.A. Allelopathic activity of Matricaria chamomilla essential oil by the bioautography test. Allelopath. J. 2012, 29, 171–176. [Google Scholar]
- Formisano, C.; Delfine, S.; Oliviero, F.; Tenore, G.C.; Rigano, D.; Senatore, F. Correlation among environmental factors, chemical composition and antioxidative properties of essential oil and extracts of chamomile (Matricaria chamomilla L.) collected in Molise (South-central Italy). Ind. Crops Prod. 2015, 63, 256–263. [Google Scholar] [CrossRef]
- Baghalian, K.; Haghiry, A.; Naghavi, M.R.; Mohammadi, A. Effect of saline irrigation water on agronomical and phytochemical characters of chamomile (Matricaria recutita L.). Sci. Hortic. 2008, 116, 437–441. [Google Scholar] [CrossRef]
- Ayoughi, F.; Marzegar, M.; Sahari, M.A.; Naghdibadi, H. Chemical compositions of essential oils of Artemisia dracunculus L. and endemic Matricaria chamomilla L. and an evaluation of their antioxidative effects. J. Agric. Sci. Technol. 2011, 13, 79–88. [Google Scholar]
- Farhoudi, R. Chemical constituents and antioxidant properties of Matricaria recutita and Chamaemelum nobile essential oil growing wild in the South West of Iran. J. Essent. Oil-Bear. Plants 2013, 16, 531–537. [Google Scholar] [CrossRef]
- Hosseinzadeh, Z.; Piryae, M.; Asl, M.B.; Abolghasemi, M.M. ZnO polythiophene SBA-15 nanoparticles as a solid-phase microextraction fiber for fast determination of essential oils of Matricaria chamomilla. Nanochem. Res. 2018, 3, 124–130. [Google Scholar] [CrossRef]
- Kazemi, M. Chemical composition and antimicrobial activity of essential oil of Matricaria recutita. Int. J. Food Prop. 2015, 18, 1784–1792. [Google Scholar] [CrossRef]
- Mahmoudi, A.; Karami, M.; Ebadi, M.T.; Ayyari, M. Effects of infrared drying and air flow rate on qualitative parameters. Iran. J. Med. Aromat. Plants Res. 2020, 36, 709–723. [Google Scholar]
- Ganzera, M.; Schneider, P.; Stuppner, H. Inhibitory effects of the essential oil of chamomile (Matricaria recutita L.) and its major constituents on human cytochrome P450 enzymes. Life Sci. 2006, 78, 856–861. [Google Scholar] [CrossRef]
- Heuskin, S.; Godin, B.; Leroy, P.; Capella, Q.; Wathelet, J.P.; Verheggen, F.; Haubruge, E.; Lognay, G. Fast gas chromatography characterisation of purified semiochemicals from essential oils of Matricaria chamomilla L. (Asteraceae) and Nepeta cataria L. (Lamiaceae). J. Chromatogr. A 2009, 1216, 2768–2775. [Google Scholar] [CrossRef]
- Abdalla, R.M.; Abdelgadir, A.E. Antibacterial activity and phytochemical constituents of Cinnamomum verum and Matricaria chamomilla from Sudan. Bio Bull. 2016, 2, 8–12. [Google Scholar]
- Elsemelawy, S.A. Antidiabetic and antioxidative activity of chamomile (Matricaria chamomilla L.) powder on diabetic rats. J. Stud. Searches Specif. Educ. 2017, 3, 97–112. [Google Scholar]
- Petrul’ová-Poracká, V.; Repčák, M.; Vilková, M.; Imrich, J. Coumarins of Matricaria chamomilla L.: Aglycones and glycosides. Food Chem. 2013, 141, 54–59. [Google Scholar] [CrossRef]
- Sayyar, Z.; Yazdinezhad, A.; Hassan, M.; Jafari Anarkooli, I. Protective effect of Matricaria chamomilla ethanolic extract on hippocampal neuron damage in rats exposed to formaldehyde. Oxid. Med. Cell. Longev. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, P.; Ma, Y.; Wang, K.; Chang, X.; Bai, Y.; Zhang, D.; Yang, L. Simultaneous quantitative determination of six caffeoylquinic acids in Matricaria chamomilla L. with high-performance liquid chromatography. J. Chem. 2019, 2019, 4352832. [Google Scholar] [CrossRef]
- Ma, X.; Zhao, D.; Li, X.; Meng, L. Chromatographic method for determination of the free amino acid content of chamomile flowers. Pharmacogn. Mag. 2015, 11, 176. [Google Scholar] [CrossRef] [PubMed]
- Stanojevic, L.P.; Marjanovic-Balaban, Z.R.; Kalaba, V.D.; Stanojevic, J.S.; Cvetkovic, D.J. Chemical composition, antioxidant and antimicrobial activity of chamomile flowers essential oil (Matricaria chamomilla L.). J. Essent. Oil Bear. Plants 2016, 19, 2017–2028. [Google Scholar] [CrossRef]
- Mahdavi, B.; Ghorat, F.; Nasrollahzadeh, M.S.; Hosseyni-Tabar, M.; Rezaei-Seresht, H. Chemical composition, antioxidant, antibacterial, cytotoxicity, and hemolyses activity of essential oils from flower of Matricaria chamomilla var. chamomilla. Anti-Infect. Agents 2020, 18, 224–232. [Google Scholar] [CrossRef]
- Melo-Guerrero, M.C.; Ortiz-Jurado, D.E.; Hurtado-Benavides, A.M. Comparison of the composition and antioxidant activity of the chamomile essential oil (Matricaria chamomilla L.) obtained by supercritical fluids extraction and other green techniques. Rev. Acad. Colomb. Cienc. Exactas Fis. Nat. 2020, 44, 845–856. [Google Scholar] [CrossRef]
- El Mihyaoui, A.; Candela, M.E.; Cano, A.; Hernández-Ruiz, J.; Lamarti, A.; Arnao, M.B. Comparative study of wild chamomile plants from the north-west of Morocco: Bioactive components and total antioxidant activity. J. Med. Plants Res. 2021, 5, 431–441. [Google Scholar] [CrossRef]
- Abdoul-Latif, F.M.; Mohamed, N.; Edou, P.; Ali, A.A.; Djama, S.O.; Obame, L.C.; Bassolé, I.H.N.; Dicko, M.H. Antimicrobial and antioxidant activities of essential oil and methanol extract of Matricaria chamomilla L. from Djibouti. J. Med. Plants Res. 2011, 5, 1512–1517. [Google Scholar]
- El-Agbar, Z.A.; Shakya, A.K.; Khalaf, N.A.; Al-Haroon, M. Comparative antioxidant activity of some edible plants. Turk. J. Biol. 2008, 32, 193–196. [Google Scholar]
- Elmastaș, M.; Inkiliç, S.Ç.; Aboul-Enein, H.Y. Antioxidant capacity and determination of total phenolic compounds in daisy (Matricaria chamomilla, Fam. Asteraceae). World J. Anal. Chem. 2015, 3, 9–14. [Google Scholar] [CrossRef]
- Munir, N.; Iqbal, A.S.; Altaf, I.; Bashir, R.; Sharif, N.; Saleem, F.; Naz, S. Evaluation of antioxidant and antimicrobial potential of two endangered plant species Atropa belladonna and Matricaria chamomilla. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 111–117. [Google Scholar] [CrossRef]
- Roby, M.H.H.; Sarhan, M.A.; Selim, K.A.-H.; Khalel, K.I. Antioxidant and antimicrobial activities of essential oil and extracts of fennel (Foeniculum vulgare L.) and chamomile (Matricaria chamomilla L.). Ind. Crops Prod. 2013, 44, 437–445. [Google Scholar] [CrossRef]
- Alibabaei, Z.; Rabiei, Z.; Rahnama, S.; Mokhtari, S.; Rafieian-kopaei, M. Matricaria chamomilla extract demonstrates antioxidant properties against elevated rat brain oxidative status induced by amnestic dose of scopolamine. Biomed. Aging Pathol. 2014, 4, 355–360. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Mašković, P.; Savić, S.; Nikolić, L. Antioxidant and biological activity of chamomile extracts obtained by different techniques: Perspective of using superheated water for isolation of biologically active compounds. Ind. Crops Prod. 2015, 65, 582–591. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Zeković, Z.; Jerković, J.; Zengin, G.; Gašić, U.; Tešić, Ž.; Mašković, P.; Soares, C.; Fatima Barroso, M.; et al. The influence of the extraction temperature on polyphenolic profiles and bioactivity of chamomile (Matricaria chamomilla L.) subcritical water extracts. Food Chem. 2019, 271, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulou, N.S.; Megremi, S.F.; Tarantilis, P. Evaluation of antioxidant activity, toxicity, and phenolic profile of aqueous extracts of chamomile (Matricaria chamomilla L.) and sage (Salvia officinalis L.) prepared at different temperatures. Appl. Sci. 2020, 10, 2270. [Google Scholar] [CrossRef]
- Pereira, S.V.; Reis, R.A.S.P.; Garbuio, D.C.; De Freitas, L.A.P. Dynamic maceration of Matricaria chamomilla inflorescences: Optimal conditions for flavonoids and antioxidant activity. Rev. Bras. Farmacogn. 2018, 28, 111–117. [Google Scholar] [CrossRef]
- de Franco, E.P.D.; Contesini, F.J.; Lima da Silva, B.; Alves de Piloto Fernandes, A.M.; Wielewski Leme, C.; Gonçalves Cirino, J.P.; Bueno Campos, P.R.; de Oliveira Carvalho, P. Enzyme-assisted modification of flavonoids from Matricaria chamomilla: Antioxidant activity and inhibitory effect on digestive enzymes. J. Enzym. Inhib. Med. Chem. 2020, 35, 42–49. [Google Scholar] [CrossRef]
- Singh, K.G.; Sonia, S.; Konsoor, N. In vitro and ex-vivo studies on the antioxidant, anti-inflammatory and antiarthritic properties of Camellia sinensis, Hibiscus rosa sinensis, Matricaria chamomilla, Rosa sp., Zingiber officinale tea extracts. Int. J. Pharm. Sci. Res. 2018, 9, 3543–3551. [Google Scholar] [CrossRef]
- Nemţanu, M.R.; Kikuchi, I.S.; de Jesus Andreoli Pinto, T.; Mazilu, E.; Setnic, S.; Bucur, M.; Duliu, O.G.; Meltzer, V.; Pincu, E. Electron beam irradiation of Matricaria chamomilla L. for microbial decontamination. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2008, 266, 2520–2523. [Google Scholar] [CrossRef]
- Hassanpour, H.; Niknam, V. Establishment and assessment of cell suspension cultures of Matricaria chamomilla as a possible source of apigenin under static magnetic field. Plant Cell. Tissue Organ Cult. 2020, 142, 583–593. [Google Scholar] [CrossRef]
- Nargesi, S.; Moayeri, A.; Ghorbani, A.; Seifinejad, Y.; Shirzadpour, E. The effects of Matricaria chamomilla L. hydroalcoholic extract on atherosclerotic plaques, antioxidant activity, lipid profile and inflammatory indicators in rats. Biomed. Res. Ther. 2018, 5, 2752–2761. [Google Scholar] [CrossRef]
- Silva, N.C.C.; Barbosa, L.; Seito, L.N.; Fernandes, A., Jr. Antimicrobial activity and phytochemical analysis of crude extracts and essential oils from medicinal plants. Nat. Prod. Res. 2012, 26, 1510–1514. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial activity of some essential oils—Present status and future perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef] [PubMed]
- Owlia, P.; Rasooli, I.; Shahed, H. Antistreptococcal and antioxidant activity of essentiel oil from Matricaria chamomilla L. Res. J. Biol. Sci. 2007, 2, 155–160. [Google Scholar]
- Sakkas, H.; Economou, V.; Gousia, P.; Bozidis, P.; Sakkas, V.A.; Petsios, S.; Mpekoulis, G.; Ilia, A.; Papadopoulou, C. Antibacterial efficacy of commercially available essential oils tested against drug-resistant gram-positive pathogens. Appl. Sci. 2018, 8, 2201. [Google Scholar] [CrossRef]
- Shakya, V.K.; Luqman, S.; Tikku, A.P.; Chandra, A.; Singh, D.K. A relative assessment of essential oil of Chrysopogon zizanioides and Matricaria chamomilla along with calcium hydroxide and chlorhexidine gel against Enterococcus faecalis in ex vivo root canal models. J. Conserv. Dent. 2019, 22, 34. [Google Scholar] [CrossRef]
- Satyal, P.; Shrestha, S.; Setzer, W.N. Composition and bioactivities of an (E)-β-farnesene chemotype of chamomile (Matricaria chamomilla) essential oil from Nepal. Nat. Prod. Commun. 2015, 10, 1453–1457. [Google Scholar] [CrossRef]
- Hartmann, K.C.; Onofre, S.B. Atividade antimicrobiana de óleos essenciais da camoantimicrobial activity of the essential oils of chamomile (Matricaria chamomilla L.). Rev. Saúde Pesqui. 2010, 3, 279–284. [Google Scholar] [CrossRef]
- Ismail, M.C.; Waleed, S.; Ibrahim, K.; Fakhri, N.U. Synergistic interaction between chamomile flower (Matricaria chamomilla L.) extracts and tetracycline against wound infection bacteria. J. Al-Nahrain Univ. Sci. 2013, 16, 191–195. [Google Scholar] [CrossRef]
- Boudıeb, K.; Kaki, S.A.S.A.; Oulebsir-Mohandkaci, H.; Bennacer, A. Phytochemical characterization and antimicrobial potentialities of two medicinal plants, Chamaemelum nobile (L.) All and Matricaria chamomilla (L.). Int. J. Innov. Approaches Sci. Res. 2018, 2, 126–139. [Google Scholar] [CrossRef]
- Mariod, A.A.; Kakil, E.S.; Elneel, Y.F.H. Antibacterial activity of Zingiber officinale, Matricaria chamomilla and Nigella sativa extractions on the growth of pathogenic bacteria isolated from different clinical specimens. Acta Sci. Nutr. Health 2019, 3, 26–32. [Google Scholar]
- Malm, A.; Glowniak-Lipa, A.; Korona-Glowniak, I.; Baj, T. Anti-Helicobacter pylori activity in vitro of chamomile flowers, coneflower herbs, peppermint leaves and thyme herbs—A preliminary report. Curr. Issues Pharm. Med. Sci. 2015, 28, 30–32. [Google Scholar] [CrossRef]
- Carvalho, A.F.; Silva, D.M.; Silva, T.R.C.; Scarcelli, E.; Manhani, M.R. Avaliação da atividade antibacteriana de extratos etanólico e de ciclohexano a partir das flores de camomila (Matricaria chamomilla L.). Rev. Bras. Plantas Med. 2014, 16, 521–526. [Google Scholar] [CrossRef]
- Khashan, A.A.; Hamad, M.A.; Jadaan, M.S. In vivo antimicrobial activity of Matricaria chamomilla extract against pathogenic bacteria induced skin infections in mice. Syst. Rev. Pharm. 2020, 11, 672–676. [Google Scholar] [CrossRef]
- Bayoub, K.; Baibai, T.; Mountassif, D.; Retmane, A.; Soukri, A. Antibacterial activities of the crude ethanol extracts of medicinal plants against Listeria monocytogenes and some other pathogenic strains. Afr. J. Biotechnol. 2010, 9, 4251–4258. [Google Scholar]
- Abdi, P.; Mahdavi Ourtakand, M.; Honarmand Jahromy, S. The Effect of Matricaria chamomilla alcoholic extract on phenotype detection of efflux pumps of methicillin resistant Staphylococcus aureus (MRSA) isolated from skin lesions. Iran. J. Med. Microbiol. 2019, 13, 220–231. [Google Scholar] [CrossRef]
- Hameed, R.H.; Mohammed, G.J.; Hameed, I.H. Matricaria chamomilla: Bioactive compounds of methanolic fruit extract using GC-MS and FTIR techniques and determination of its antimicrobial properties. Indian J. Public Health Res. Dev. 2018, 9, 188–194. [Google Scholar] [CrossRef]
- Vora, J.; Srivastava, A.; Modi, H. Antibacterial and antioxidant strategies for acne treatment through plant extracts. Inform. Med. Unlocked 2018, 13, 128–132. [Google Scholar] [CrossRef]
- Omran, A.M. Antimicrobial and phytochemical study of Matricaria chamomilla L., Menthalongi folia L. and Salvia officinalis L. Plant Arch. 2018, 18, 387–397. [Google Scholar]
- Rahman, H.; Chandra, A. Microbiologic evaluation of Matricaria and chlorhexidine against E. faecalis and C. albicans. Indian J. Dent. 2015, 6, 60. [Google Scholar] [CrossRef]
- Peerzada, T.; Gupta, J. Distribution of phytochemicals in stems and leaves of Cichorium intybus and Matricaria chamomilla: Assessment of their antioxidant and antimicrobial potential. J. Biotechnol. Comput. Biol. Bionanotechnol. 2018, 99, 119–128. [Google Scholar] [CrossRef]
- Sharifzadeh, A.; Shokri, H. Antifungal activity of essential oils from Iranian plants against fluconazole-resistant and fluconazole-susceptible Candida albicans. Avicenna J. Phytomed. 2016, 6, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Mekonnen, A.; Yitayew, B.; Tesema, A.; Taddese, S. In vitro antimicrobial activity of essential oil of Thymus schimperi, Matricaria chamomilla, Eucalyptus globulus, and Rosmarinus officinalis. Int. J. Microbiol. 2016, 2016, 9545693. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour, M.; Mobini-dehkordi, M.; Saffar, B.; Teimori, H. Antiproliferative effects of Matricaria chamomilla on Saccharomyces cerevisiae. J. HerbMed Pharmacol. 2013, 2, 49–51. [Google Scholar]
- Osman, M.Y.; Taie, H.A.A.; Helmy, W.A.; Amer, H. Screening for antioxidant, antifungal, and antitumor activities of aqueous extracts of chamomile (Matricaria chamomilla). Egypt. Pharm. J. 2016, 15, 55–61. [Google Scholar] [CrossRef]
- Seyedjavadi, S.S.; Khani, S.; Zare-Zardini, H.; Halabian, R.; Goudarzi, M.; Khatami, S.; Imani Fooladi, A.A.; Amani, J.; Razzaghi-Abyaneh, M. Isolation, functional characterization, and biological properties of MCh-AMP1, a novel antifungal peptide from Matricaria chamomilla L. Chem. Biol. Drug Des. 2019, 93, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Hajaji, S.; Sifaoui, I.; López-Arencibia, A.; Reyes-Batlle, M.; Jiménez, I.A.; Bazzocchi, I.L.; Valladares, B.; Akkari, H.; Lorenzo-Morales, J.; Piñero, J.E. Leishmanicidal activity of α-bisabolol from Tunisian chamomile essential oil. Parasitol. Res. 2018, 2855–2867. [Google Scholar] [CrossRef] [PubMed]
- Hajaji, S.; Alimi, D.; Jabri, M.A.; Abuseir, S.; Gharbi, M.; Akkari, H. Anthelmintic activity of Tunisian chamomile (Matricaria recutita L.) against Haemonchus contortus. J. Helminthol. 2018, 92, 168–177. [Google Scholar] [CrossRef]
- Mansour, S.A.; El-Sharkawy, A.Z.; Abdel-Hamid, N.A. Toxicity of essential plant oils, in comparison with conventional insecticides, against the desert locust, Schistocerca gregaria (Forskål). Ind. Crops Prod. 2015, 63, 92–99. [Google Scholar] [CrossRef]
- Al-Jabr, A.M. Toxicity and repellency of seven plant essential oils to Oryzaephilus surinamensis (Coleoptera: Silvanidae) and Tribolium castaneum (Coleoptera: Tenebrioidae). Sci. J. King Faisal Univ. 2006, 7, 49–60. [Google Scholar]
- Abd El-Moneim, M.; Ali, F.S.; Turky, A.F. Control of Tetranychus urticae Koch by extracts of three essential oils of chamomile, marjoram and Eucalyptus. Asian Pac. J. Trop. Biomed. 2012, 2, 24–30. [Google Scholar] [CrossRef]
- Khodadad, P.; Mehdi, R. Biological activities of chamomile (Matricaria chamomile) flowers’ extract against the survival and egg laying of the cattle fever tick (Acari Ixodidae). J. Zhejiang Univ. Sci. B 2007, 8, 693–696. [Google Scholar] [CrossRef]
- Romero, M.D.C.; Valero, A.; Martín-Sánchez, J.; Navarro-Moll, M.C. Activity of Matricaria chamomilla essential oil against anisakiasis. Phytomedicine 2012, 19, 520–523. [Google Scholar] [CrossRef] [PubMed]
- Hajaji, S.; Sifaoui, I.; López-Arencibia, A.; Reyes-Batlle, M.; Jiménez, I.A.; Bazzocchi, I.L.; Valladares, B.; Pinero, J.E.; Lorenzo-Morales, J.; Akkari, H. Correlation of radical-scavenging capacity and amoebicidal activity of Matricaria recutita L. (Asteraceae). Exp. Parasitol. 2017, 183, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Váradyová, Z.; Pisarčíková, J.; Babják, M.; Hodges, A.; Mravčáková, D.; Kišidayová, S.; Königová, A.; Vadlejch, J.; Várady, M. Ovicidal and larvicidal activity of extracts from medicinal-plants against Haemonchus contortus. Exp. Parasitol. 2018, 195, 71–77. [Google Scholar] [CrossRef]
- Al-Mekhlafi, F.A.; Abutaha, N.; Al-Malki, A.M.; Al-Wadaan, M. Inhibition of the growth and development of mosquito larvae of Culexpipiens L. (Diptera: Culicidae) treated with extract from flower of Matricaria chamomilla (Asteraceae). Entomol. Res. 2020, 50, 138–145. [Google Scholar] [CrossRef]
- Villa-Rodriguez, J.A.; Kerimi, A.; Abranko, L.; Williamson, G. German chamomile (Matricaria chamomilla) extract and its major ppolyphenols inhibit intestinal α-glycosidases in vitro. FASEB J. 2015, 29, LB323. [Google Scholar] [CrossRef]
- Hwang, S.H.; Wang, Z.; Quispe, Y.N.G.; Lim, S.S.; Yu, J.M. Evaluation of aldose reductase, protein glycation, and antioxidant inhibitory activities of bioactive flavonoids in Matricaria recutita L. and their structure-activity relationship. J. Diabetes Res. 2018, 3276162. [Google Scholar] [CrossRef]
- Franco, R.R.; da Silva Carvalho, D.; de Moura, F.B.R.; Justino, A.B.; Silva, H.C.G.; Peixoto, L.G.; Espindola, F.S. Antioxidant and anti-glycation capacities of some medicinal plants and their potential inhibitory against digestive enzymes related to type 2 diabetes mellitus. J. Ethnopharmacol. 2018, 215, 140–146. [Google Scholar] [CrossRef]
- Romeilah, R.M. Anticancer and antioxidant activities of Matricaria chamomilla L. and Marjorana hortensis essential oils. Res. J. Med. Med. Sci. 2009, 4, 332–339. [Google Scholar]
- Kamali, A.M.; Nikseresht, M.; Delaviz, H.; Barmak, M.J.; Servatkhah, M.; Ardakani, M.T.; Mahmoudi, R. In vitro cytotoxic activity of Matricaria chamomilla root extract in Human breast cancer cell line MCF-7. Life Sci. J. 2014, 11, 403–406. [Google Scholar]
- Fraihat, A.; Alatrash, L.; Abbasi, R.; Abu-Irmaileh, B.; Hamed, S.; Mohammad, M.; Abu-Rish, E.; Bustanji, Y. Inhibitory effects of methanol extracts of selected plants on the proliferation of two human melanoma cell lines. Trop. J. Pharm. Res. 2018, 17, 1081–1086. [Google Scholar] [CrossRef]
- Wu, Y.; Xu, Y.; Yao, L. Anti-inflammatory and anti-allergic effects of German chamomile (Matricaria chamomilla L.). J. Essent. Oil Bear. Plants 2012, 15, 37–41. [Google Scholar] [CrossRef]
- Ortiz, M.I.; Cariño-Cortés, R.; Ponce-Monter, H.A.; González-García, M.P.; Castañeda-Hernández, G.; Salinas-Caballero, M. Synergistic interaction of Matricaria chamomilla extract with diclofenac and indomethacin on carrageenan-induced aw inflammation in rats. Drug Dev. Res. 2017, 78, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Ionita, R.; Postu, P.A.; Mihasan, M.; Gorgan, D.L.; Hancianu, M.; Cioanca, O.; Hritcu, L. Ameliorative effects of Matricaria chamomilla L. hydroalcoholic extract on scopolamine-induced memory impairment in rats: A behavioral and molecular study. Phytomedicine 2018, 47, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Rabiei, A.; Moghtadaei, M.; Mokhtari, S.; Rafieian-Kopaei, M. Matricaria chamomilla essential oil with physical training improves passive avoidance memory. Iran. J. Physiol. Pharmacol. 2018, 83, 83–91. [Google Scholar]
- Moshfegh, A.; Setorki, M. Neuroprotective effect of Matricaria chamomilla extract on motor dysfunction induced by transient global cerebral ischemia and reperfusion in Rat. Zahedan J. Res. Med. Sci. 2017, 19, 69. [Google Scholar] [CrossRef]
- Can, Ö.D.; Özkaya, Ü.D.; Kıyanb, H.T.; Demircib, B. Psychopharmacological profile of chamomile (Matricaria recutita L.) essential oil in mice. Phytomedicine 2012, 19, 306–310. [Google Scholar] [CrossRef]
- Abdullahzadeh, M.; Matourypour, P.; Naji, S.A. Investigation effect of oral chamomilla on sleep quality in elderly people in Isfahan: A randomized control trial. J. Educ. Health Promot. 2017, 6, 53. [Google Scholar] [CrossRef]
- Sorme, F.M.; Tabarrai, M.; Alimadady, H.; Rahimi, R.; Sepidarkish, M.; Karimi, M. Efficacy of Matricaria chamomilla L. in infantile colic: A double blind, placebo controlled randomized trial. J. Pharm. Res. Int. 2019, 31, 1–11. [Google Scholar] [CrossRef]
- Momeni, H.; Sadeghi, H.; Salehi, A. Comparison of Matricaria chamomilla oil, Trachyspermum copticum oil and Clonidin on withdrawal syndrome in narcotics anonymous. J. Sabzevar Univ. Med. Sci. 2018, 25, 185–194. [Google Scholar]
- Amsterdam, J.D.; Li, Q.S.; Xie, S.X.; Mao, J.J. Putative antidepressant effect of chamomile (Matricaria chamomilla L.) oral extract in subjects with comorbid generalized anxiety disorder and depression. J. Altern. Complement. Med. 2020, 26, 813–819. [Google Scholar] [CrossRef]
- Ebrahimi, H.; Mardani, A.; Basirinezhad, M.H.; Hamidzadeh, A.; Eskandari, F. The effects of Lavender and Chamomile essential oil inhalation aromatherapy on depression, anxiety and stress in older community-dwelling people: A randomized controlled trial. Explore 2021, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gholami, A.; Tabaraei, Y.; Ghorat, F.; Khalili, H. The effect of inhalation of Matricaria chamomile essential oil on patients’anxiety before esophagogastroduodenoscopy. Govaresh 2018, 22, 232–238. [Google Scholar]
- Shoara, R.; Hashempur, M.H.; Ashraf, A.; Salehi, A.; Dehshahri, S.; Habibagahi, Z. Efficacy and safety of topical Matricaria chamomilla L. (chamomile) oil for knee osteoarthritis: A randomizedcontrolled clinical trial. Complement. Ther. Clin. Pract. 2015, 21, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Zargaran, A.; Borhani-Haghighi, A.; Salehi-Marzijarani, M.; Faridi, P.; Daneshamouz, S.; Azadi, A.; Sadeghpour, H.; Sakhteman, A.; Mohagheghzadeh, A. Evaluation of the effect of topical chamomile (Matricaria chamomilla L.) oleogel as pain relief in migraine without aura: A randomized, double-blind, placebo-controlled, crossover study. Neurol. Sci. 2018, 39, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Aradmehr, M.; Azhari, S.; Ahmadi, S.; Azmoude, E. The effect of chamomile cream on episiotomy pain in primiparous women: A randomized clinical trial. J. Caring Sci. 2017, 6, 19–28. [Google Scholar] [CrossRef]
- Heidarifard, S.; Amir Ali Akbari, S.; Mojab, F.; Shakeri, N. Effect of Matricaria Camomilla aroma on severity of first stage labor ain. J. Clin. Nurs. Midwifery 2015, 4, 23–31. [Google Scholar]
- Saghafi, N.; Rhkhshandeh, H.; Pourmoghadam, N.; Pourali, L.; Ghazanfarpour, M.; Behrooznia, A.; Vafisani, F. Effectiveness of Matricaria chamomilla (chamomile) extract on pain control of cyclic mastalgia: A double-blind randomised controlled trial. J. Obstet. Gynaecol. 2018, 38, 81–84. [Google Scholar] [CrossRef]
- Khalesi, Z.B.; Beiranvand, S.P.; Bokaie, M. Efficacy of chamomile in the treatment of premenstrual syndrome: A systematic review. J. Pharmacopunct. 2019, 22, 204–209. [Google Scholar] [CrossRef]
- Niazi, A.; Moradi, M. The effect of chamomile on pain and menstrual bleeding in primary dysmenorrhea: A systematic review. Int. J. Community Based Nurs. Midwifery 2021, 9, 174–186. [Google Scholar] [CrossRef]
- Johari, H.; Sharifi, E.; Mardan, M.; Kafilzadeh, F.; Hemayatkhah, V.; Kargar, H.; Nikpoor, N. The effects of a hydroalcoholic extract of Matricaria chamomilla flower on the pituitary-gonadal axis and ovaries of rats. Int. J. Endocrinol. Metab. 2011, 9, 330–334. [Google Scholar] [CrossRef]
- Soltani, M.; Moghimian, M.; Abtahi-Eivari, S.H.; Shoorei, H.; Khaki, A.; Shokoohi, M. Protective effects of Matricaria chamomilla extract on torsion/detorsion-induced tissue damage and oxidative stress in adult rat testis. Int. J. Fertil. Steril. 2018, 12, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Soltani, M.; Moghimian, M.; Abtahi, H.; Shokoohi, M. The protective effect of Matricaria chamomilla extract on histological damage and oxidative stress induced by torsion/detorsion in adult rat ovary. Int. J. Women’s Health Reprod. Sci. 2017, 5, 187–192. [Google Scholar] [CrossRef]
- Alahmadi, A.A.; Alzahrani, A.A.; Ali, S.S.; Alahmadi, B.A.; Arab, R.A.; El-Shitany, N.A.E.-A. Both Matricaria chamomilla and metformin extract improved the function and histological structure of thyroid gland in polycystic ovary syndrome rats through antioxidant mechanism. Biomolecules 2020, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Alahmadi, A.A.; Alahmadi, B.A.; Wahman, L.F.; El-Shitany, N.A. Chamomile flower extract ameliorates biochemical and histological kidney dysfunction associated with polycystic ovary syndrome. Saudi J. Biol. Sci. 2021, 28, 6158–6166. [Google Scholar] [CrossRef]
- Ali, A.A.; Alattar, S.A. Study the protective effect of Matricaria chamomilla flower extract against the toxicity of Entamoeba histolytica induces liver and renal dysfunctions in adult albino male rats. Iraqi J. Sci. 2018, 59, 832–838. [Google Scholar] [CrossRef]
- Farouk, M.; Abokora, S.; Ali, A.-F. Protective effect of Matricaria Chamomilla extract on hepatic-renal toxicity of ceftriaxone in rats. Int. J. Pharmacol. Toxicol. 2017, 5, 44. [Google Scholar] [CrossRef]
- Cemek, M.; Kaǧa, S.; Şimşek, N.; Büyükokuroǧlu, M.E.; Konuk, M. Antihyperglycemic and antioxidative potential of Matricaria chamomilla L. in streptozotocin-induced diabetic rats. J. Nat. Med. 2008, 62, 284–293. [Google Scholar] [CrossRef]
- Najla, O.A.; Olfat, A.K.; Kholoud, S.R.; Enas, N.D.; Hanan, I.S.A. Hypoglycemic and biochemical effects of Matricaria chamomilla leave extract in streptozotocin-induced diabetic rats. J. Health Sci. 2012, 2, 43–48. [Google Scholar] [CrossRef]
- Fazili, A.; Gholami, S.; Sheikhpour, M.; Pousti, P. Therapeutic effects of in vivo-differentiated stem cell and Matricaria chamomilla L. Oil in diabetic rabbit. J. Diabetes Metab. Disord. 2020, 19, 453–460. [Google Scholar] [CrossRef]
- Ramadan, K.S.; Emam, M.A. Biochemical evaluation of antihyperglycemic and antioxidative effects of Matricaria chamomilla leave extract studied in streptozotocin-induced diabetic rats. Int. J. Res. Manag. Technol. 2012, 2, 298–302. [Google Scholar]
- El Joumaa, M.M.; Taleb, R.I.; Rizk, S.; Jamilah, M.B. Protective effect of Matricaria chamomilla extract against 1, 2-dimethylhydrazine-induced colorectal cancer in mice. J. Complement. Integr. Med. 2020, 17. [Google Scholar] [CrossRef] [PubMed]
- Nouri, M.H.K.; Abad, A.N.A. Antinociceptive effect of Matricaria chamomilla on vincristine-induced peripheral neuropathy in mice. Afr. J. Pharm. Pharmacol. 2012, 6, 24–29. [Google Scholar] [CrossRef]
- Ortiz, M.I.; Fernández-martínez, E.; Soria-jasso, L.E.; Lucas-gómez, I.; Villagómez-ibarra, R.; González-garcía, M.P.; Castañeda-hernández, G.; Salinas-caballero, M. Isolation, identification and molecular docking as cyclooxygenase (COX) inhibitors of the main constituents of Matricaria chamomilla L. extract and its synergistic interaction with diclofenac on nociception and gastric damage in rats. Biomed. Pharmacother. 2016, 78, 248–256. [Google Scholar] [CrossRef]
- Borhan, F.; Naji, A.; Vardanjani, M.M.; Sasani, L. Effects of Matricaria chamomilla on the severity of nausea and vomiting due to chemotherapy. Sci. J. Hamadan Nurs. Midwifery Fac. 2017, 25, 140–146. [Google Scholar] [CrossRef][Green Version]
- Ghamchini, V.M.; Salami, M.; Mohammadi, G.R.; Moradi, Z.; Kavosi, A.; Movahedi, A.; Bidkhori, M.; Aryaeefar, M.R. The effect of chamomile tea on anxiety and depression in cancer patients treated with chemotherapy. J. Young Pharm. 2019, 11, 309–312. [Google Scholar] [CrossRef]
- Khadem, E.; Shirazi, M.; Janani, L.; Rahimi, R.; Amiri, P.; Ghorat, F. Effect of topical chamomile oil on postoperative bowel activity after cesarean section: A randomized controlled trial. J. Res. Pharm. Pract. 2018, 7, 128. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, D.S.; Oh, J.; Geum, J.-H.; Kim, J.-E.; Choi, S.-Y.; Kim, J.H.; Cho, J.Y. Matricaria chamomilla (chamomile) ameliorates muscle atrophy in mice by targeting protein catalytic pathways, myogenesis, and mitochondrial dysfunction. Am. J. Chin. Med. 2021, 49, 1493–1514. [Google Scholar] [CrossRef]
- Ortiz-Bautista, R.J.; García-González, L.L.; Ocádiz-González, M.A.; Flores-Tochihuitl, J.; García-Villaseñor, A.; González-Hernández, M.; Muñoz-Hernández, L.; del Ortiz-Figueroa, M.C.; Ramírez-Anaya, M.; Reyna-Téllez, S.; et al. Matricaria chamomilla (aqueous extract) improves atopic dermatitis-like lesions in a murine model. Rev. Med. Inst. Mex. Seguro Soc. 2017, 55, 587–593. [Google Scholar]
- Garbossa, W.A.C.; Campos, P.M.B.G.M. Euterpe oleracea, Matricaria chamomilla, and Camellia sinensis as promising ingredients for development of skin care formulations. Ind. Crops Prod. 2016, 83, 1–10. [Google Scholar] [CrossRef]
- Jiménez Delgado, J.; Madrigal Rojas, J.; Salazar Barrantes, S. Tratamiento con manzanilla (Matricaria chamomilla), para reducción de las ojeras. Rev. Médica Univ. Costa Rica 2009, 3, 7833. [Google Scholar] [CrossRef][Green Version]
- Morales-Bozo, I.; Ortega-Pinto, A.; Rojas Alcayaga, G.; Aitken Saavedra, J.P.; Salinas Flores, O.; Lefimil Puente, C.; Lozano Moraga, C.; Manríquez Urbina, J.M.; Urzúa Orellana, B. Evaluation of the effectiveness of a chamomile (Matricaria chamomilla) and linseed (Linum usitatissimum) saliva substitute in the relief of xerostomia in elders. Gerodontology 2017, 34, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Goes, P.; Dutra, C.S.; Lisboa, M.R.P.; Gondim, D.V.; Leitão, R.; Brito, G.A.C.; Rego, R.O. Clinical efficacy of a 1% Matricaria chamomile L. mouthwash and 0.12% chlorhexidine for gingivitis control in patients undergoing orthodontic treatment with fixed appliances. J. Oral Sci. 2016, 58, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Braga, A.S.; de Simas, L.L.M.; Pires, J.G.; Souza, B.M.; de Melo, F.P.d.S.R.; Saldanha, L.L.; Dokkedal, A.L.; Magalhães, A.C. Antibiofilm and anti-caries effects of an experimental mouth rinse containing Matricaria chamomilla L. extract under microcosm biofilm on enamel. J. Dent. 2020, 99, 103415. [Google Scholar] [CrossRef] [PubMed]
- Tadbir, A.A.; Pourshahidi, S.; Ebrahimi, H.; Hajipour, Z.; Memarzade, M.R.; Shirazian, S. The effect of Matricaria chamomilla (chamomile) extract in orabase on minor aphthous stomatitis, a randomized clinical trial. J. Herb. Med. 2015, 5, 71–76. [Google Scholar] [CrossRef]
- Can, E.; Kizak, V.; Özçiçek, E.; Seyhaneyildiz Can, Ş. The efficacy of chamomile (Matricaria chamomilla) oil as a promising anaesthetic agent for two freshwater aquarium fish species. Isr. J. Aquac. Bamidgeh 2017, 69, 1–8. [Google Scholar]
- Rogosic, J.; Saric, T.; Zupan, I. Effect of Achillea Millefolium L. and Matricaria chamomilla L. on consumption of Juniperus Oxycedrus L. and J. Phoenicea L. by goats. Ann. Anim. Sci. 2015, 15, 119–127. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Antonio, A.L.; Ciric, A.; João, C.M.; Sokovic, M.; Oliveira, M.B.P.P.; Santos-buelga, C.; Ferreira, I.C.F.R. Development of a functional dairy food: Exploring bioactive and preservation effects of chamomile (Matricaria recutita L.). J. Funct. Foods 2015, 16, 114–124. [Google Scholar] [CrossRef]
- Majeed, H.; Bian, Y.-Y.; Ali, B.; Jamil, A.; Majeed, U.; Khan, Q.F.; Iqbal, K.J.; Shoemaker, C.F.; Fang, Z. Essential oil encapsulations: Uses, procedures, and trends. RSC Adv. 2015, 5, 58449–58463. [Google Scholar] [CrossRef]
- Negahdary, M.; Omidi, S.; Eghbali-Zarch, A.; Mousavi, S.A.; Mohseni, G.; Moradpour, Y.; Rahimi, G. Plant synthesis of silver nanoparticles using Matricaria chamomilla plant and evaluation of its antibacterial and antifungal effects. Biomed. Res. 2015, 26, 794–799. [Google Scholar]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M.F.B.; Rocha, J.E.; Coutinho, H.D.M.; Salehi, B.; Tabanelli, G.; Montanari, C.; Contreras, M.M.; et al. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Hajizadeh-Sharafabad, F.; Varshosaz, P.; Jafari-Vayghan, H.; Alizadeh, M.; Maleki, V. Chamomile (Matricaria recutita L.) and diabetes mellitus, current knowledge and the way forward: A systematic review. Complemen. Therap. Med. 2020, 48, 102284. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, R.; Singh, S.; Kumar, V.; Kumar, A.; Kumari, A.; Rathore, S.; Kumar, R.; Singh, S. A Comprehensive Review on Biology, Genetic Improvement, Agro and Process Technology of German Chamomile (Matricaria chamomilla L.). Plants 2022, 11, 29. [Google Scholar] [CrossRef] [PubMed]
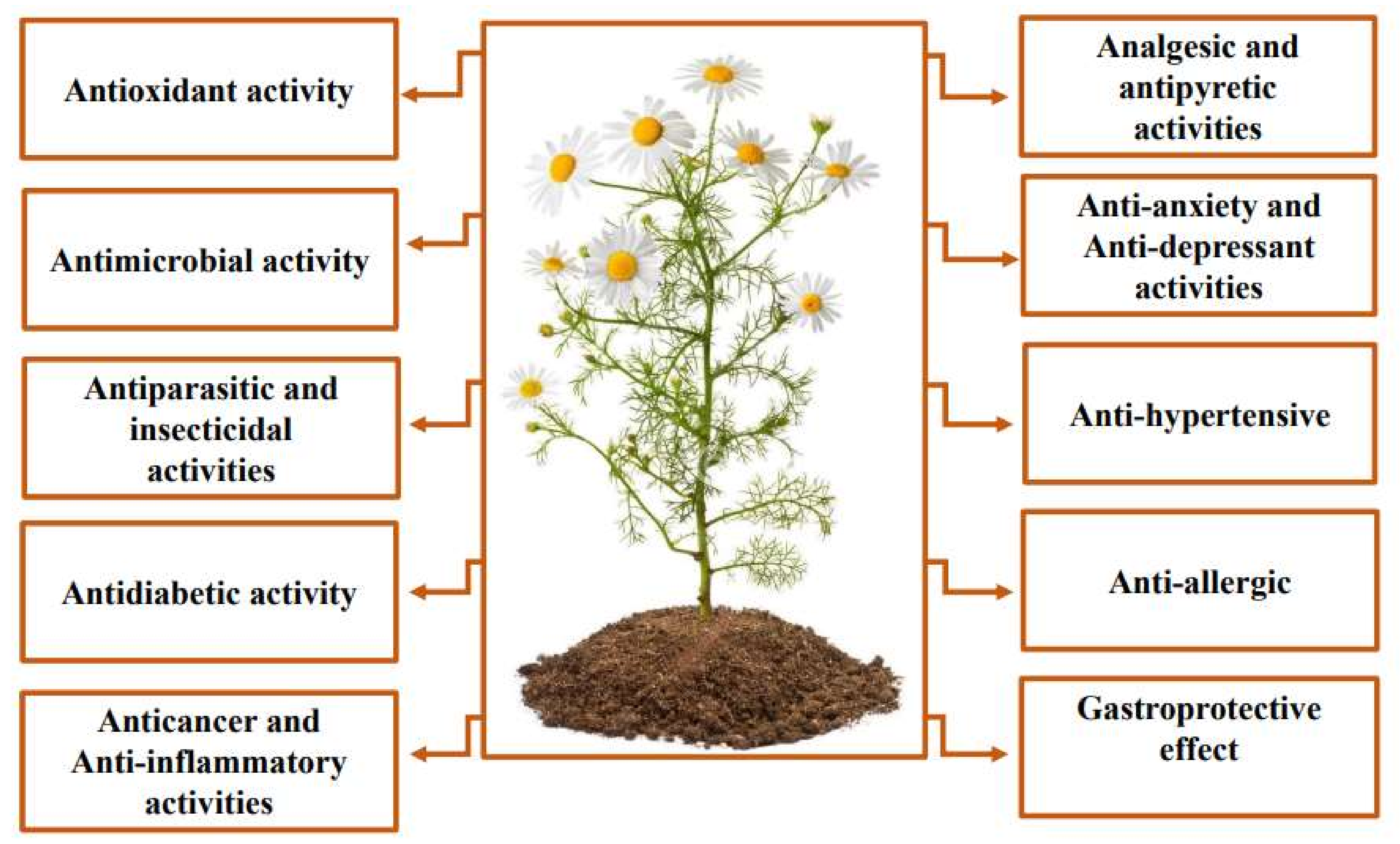
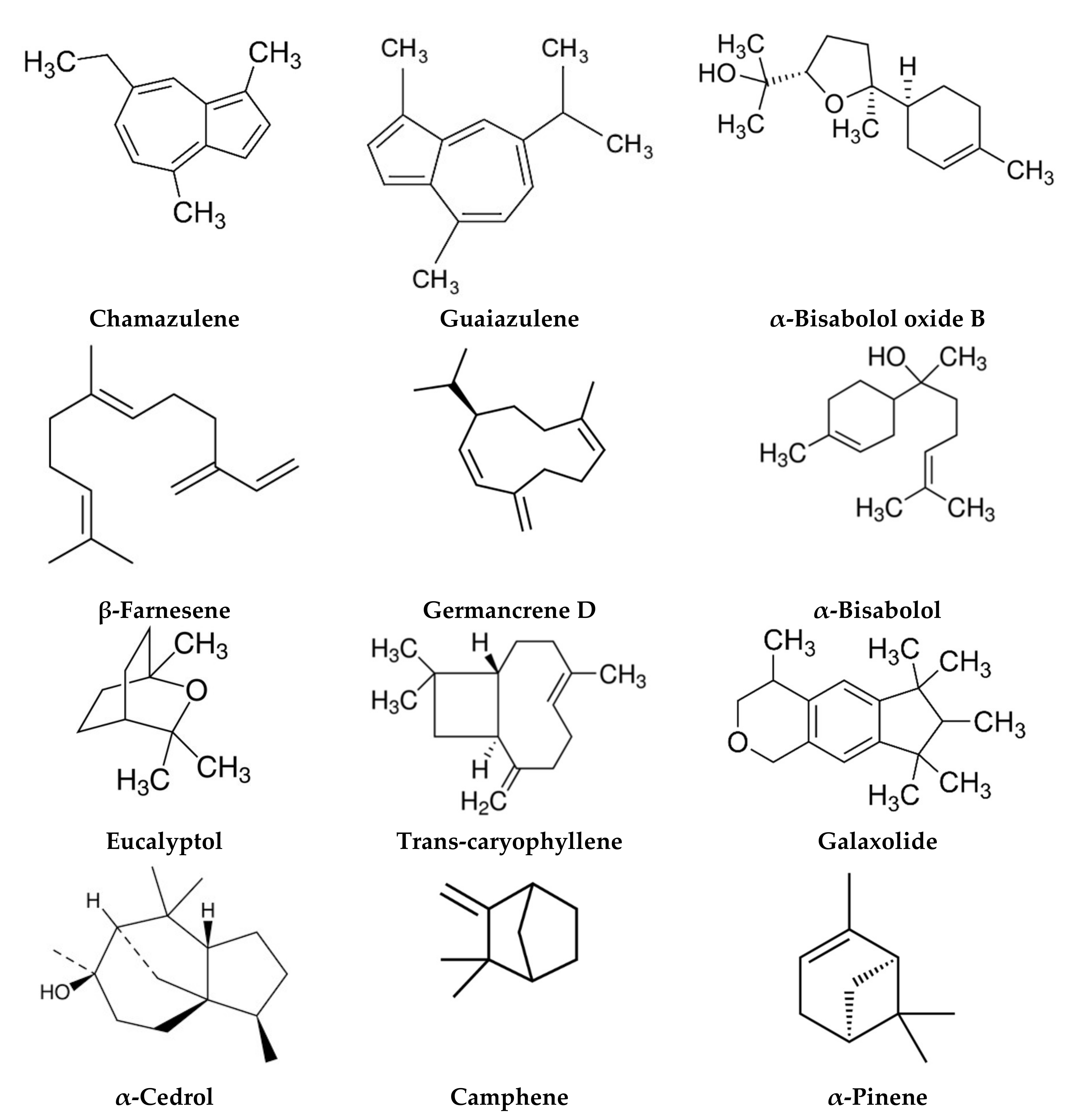
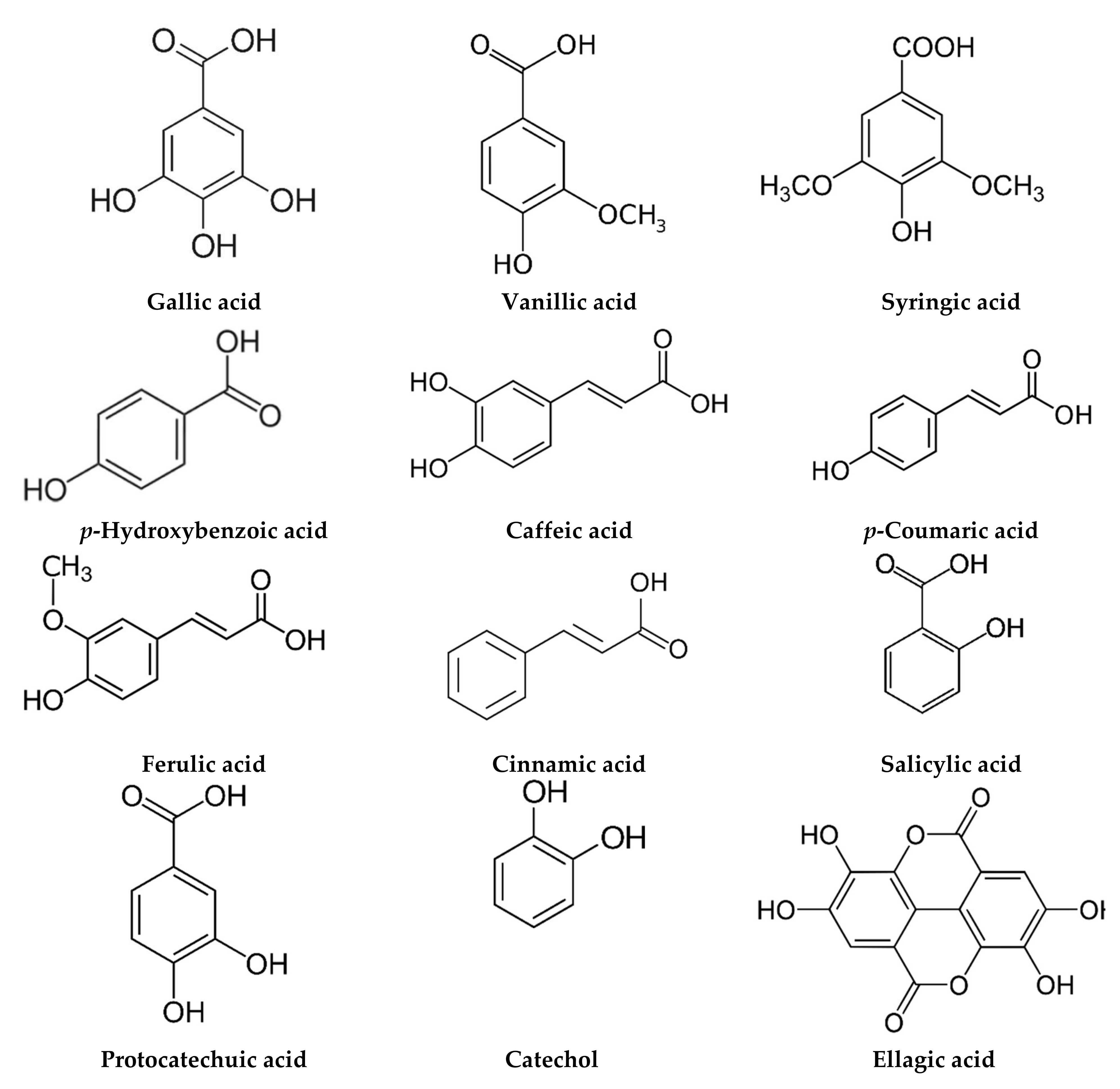
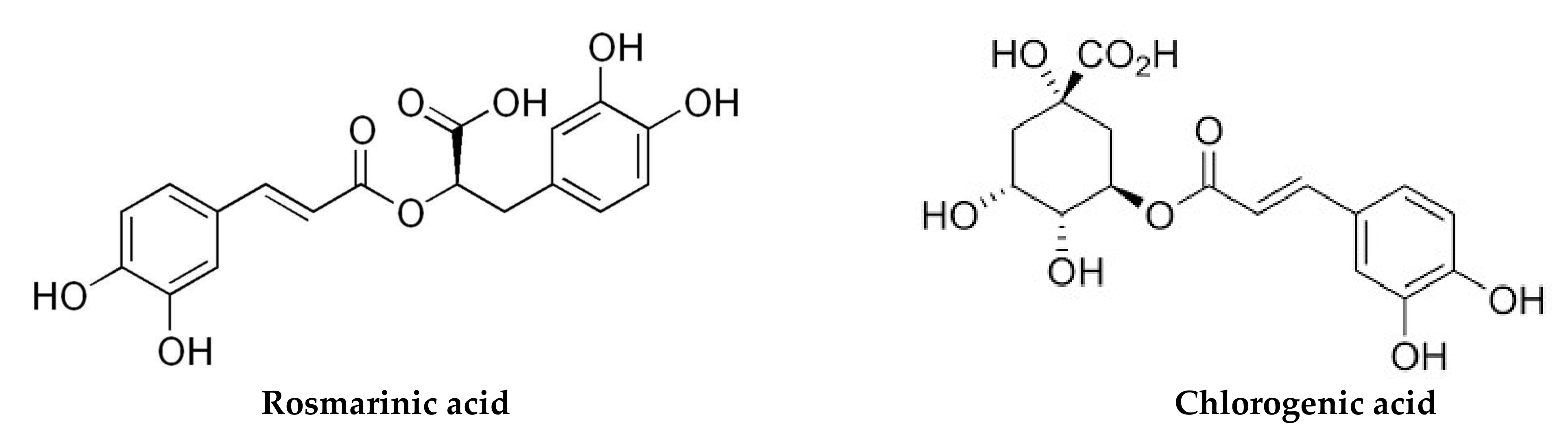
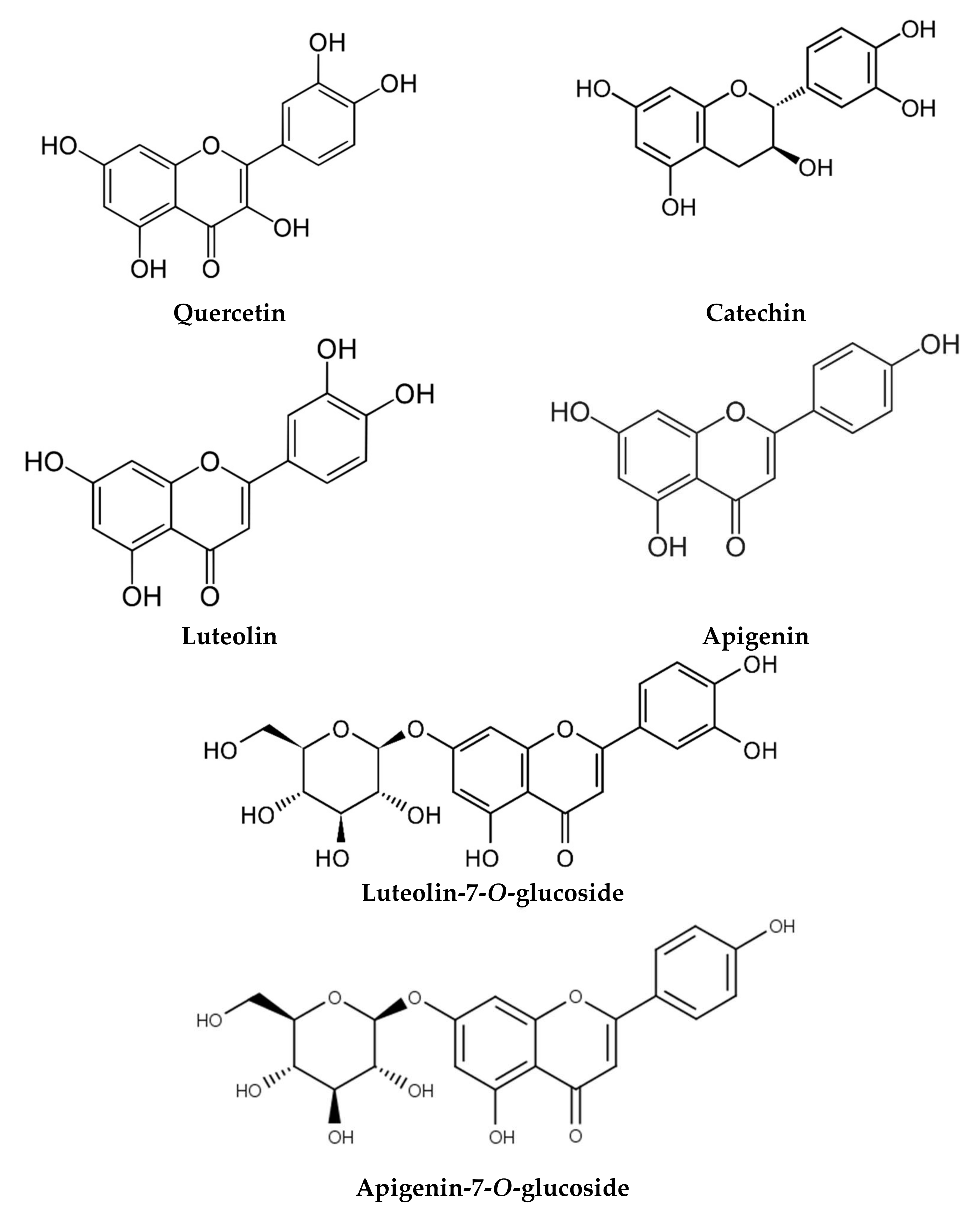
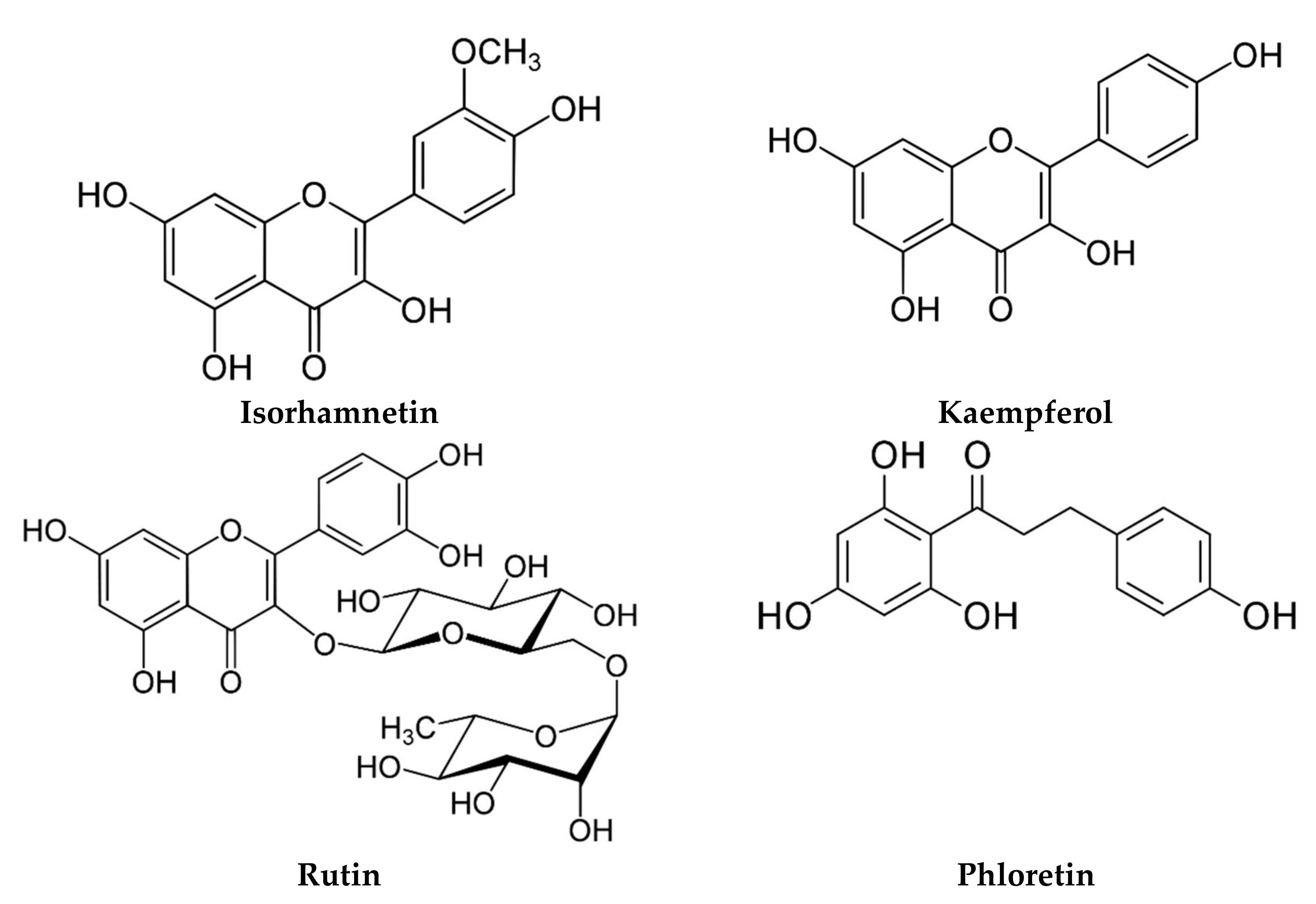
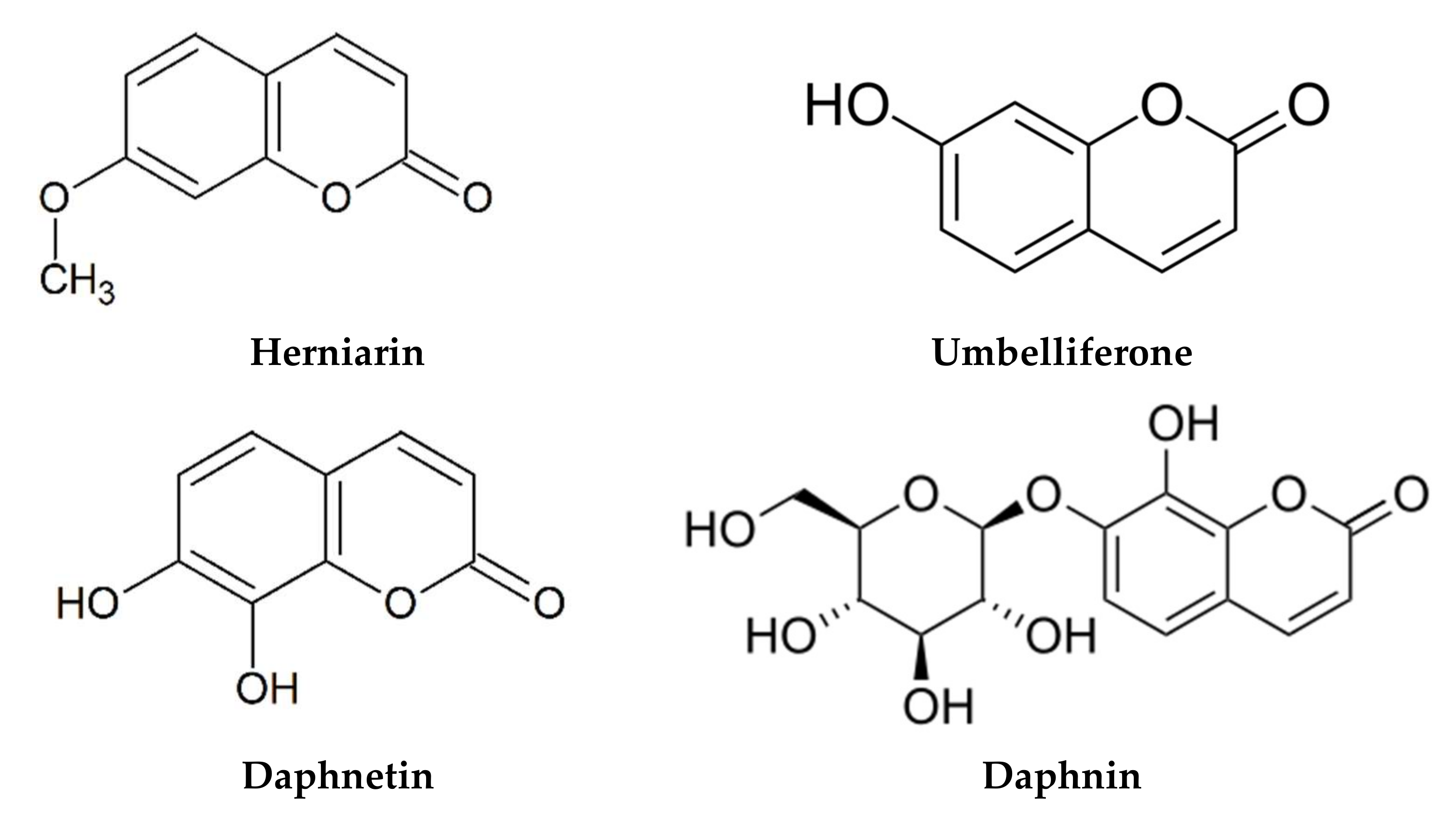
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Mihyaoui, A.; Esteves da Silva, J.C.G.; Charfi, S.; Candela Castillo, M.E.; Lamarti, A.; Arnao, M.B. Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses. Life 2022, 12, 479. https://doi.org/10.3390/life12040479
El Mihyaoui A, Esteves da Silva JCG, Charfi S, Candela Castillo ME, Lamarti A, Arnao MB. Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses. Life. 2022; 12(4):479. https://doi.org/10.3390/life12040479
Chicago/Turabian StyleEl Mihyaoui, Amina, Joaquim C. G. Esteves da Silva, Saoulajan Charfi, María Emilia Candela Castillo, Ahmed Lamarti, and Marino B. Arnao. 2022. "Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses" Life 12, no. 4: 479. https://doi.org/10.3390/life12040479
APA StyleEl Mihyaoui, A., Esteves da Silva, J. C. G., Charfi, S., Candela Castillo, M. E., Lamarti, A., & Arnao, M. B. (2022). Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses. Life, 12(4), 479. https://doi.org/10.3390/life12040479








