Domesticated LTR-Retrotransposon gag-Related Gene (Gagr) as a Member of the Stress Response Network in Drosophila
Abstract
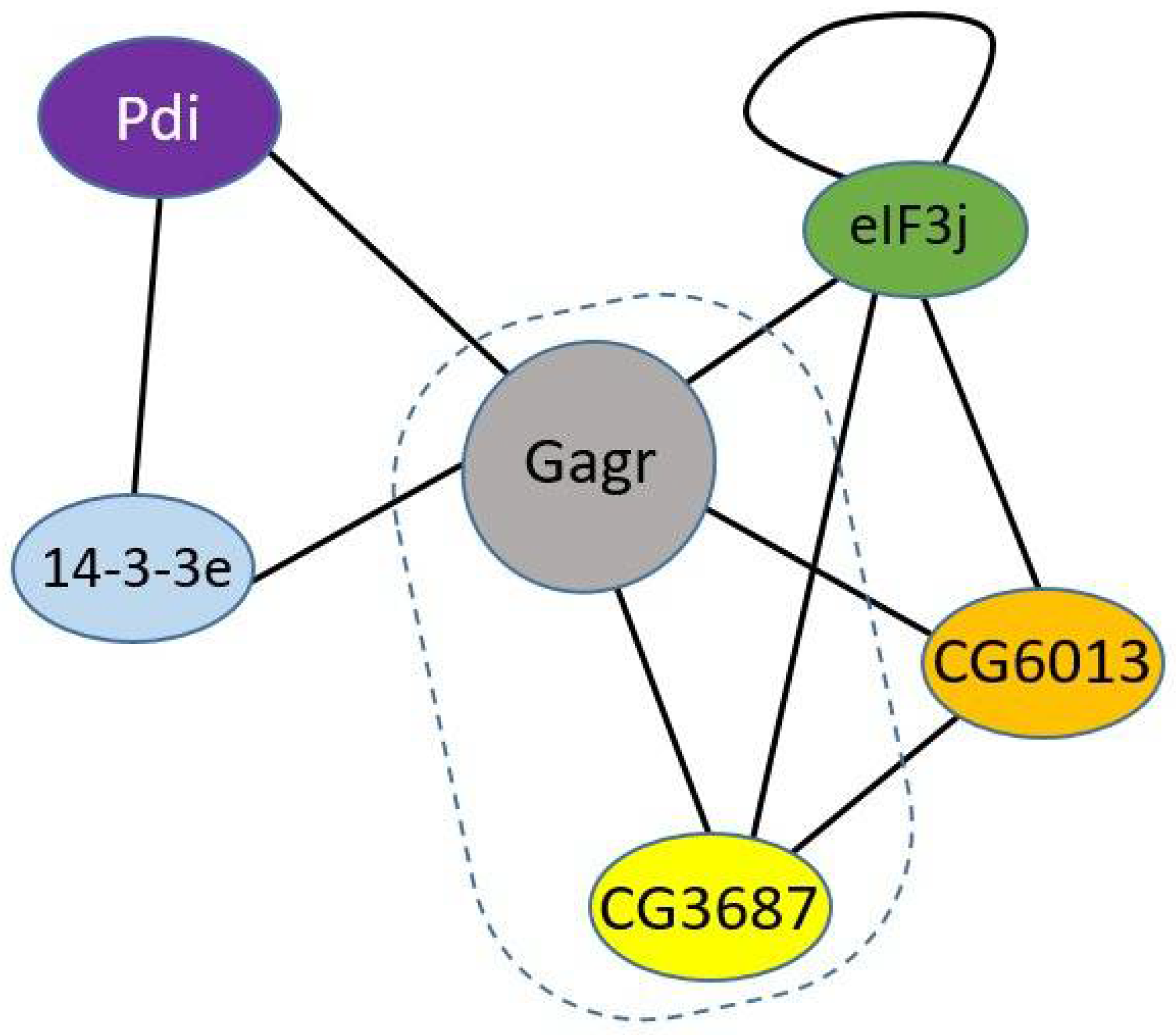
1. Introduction
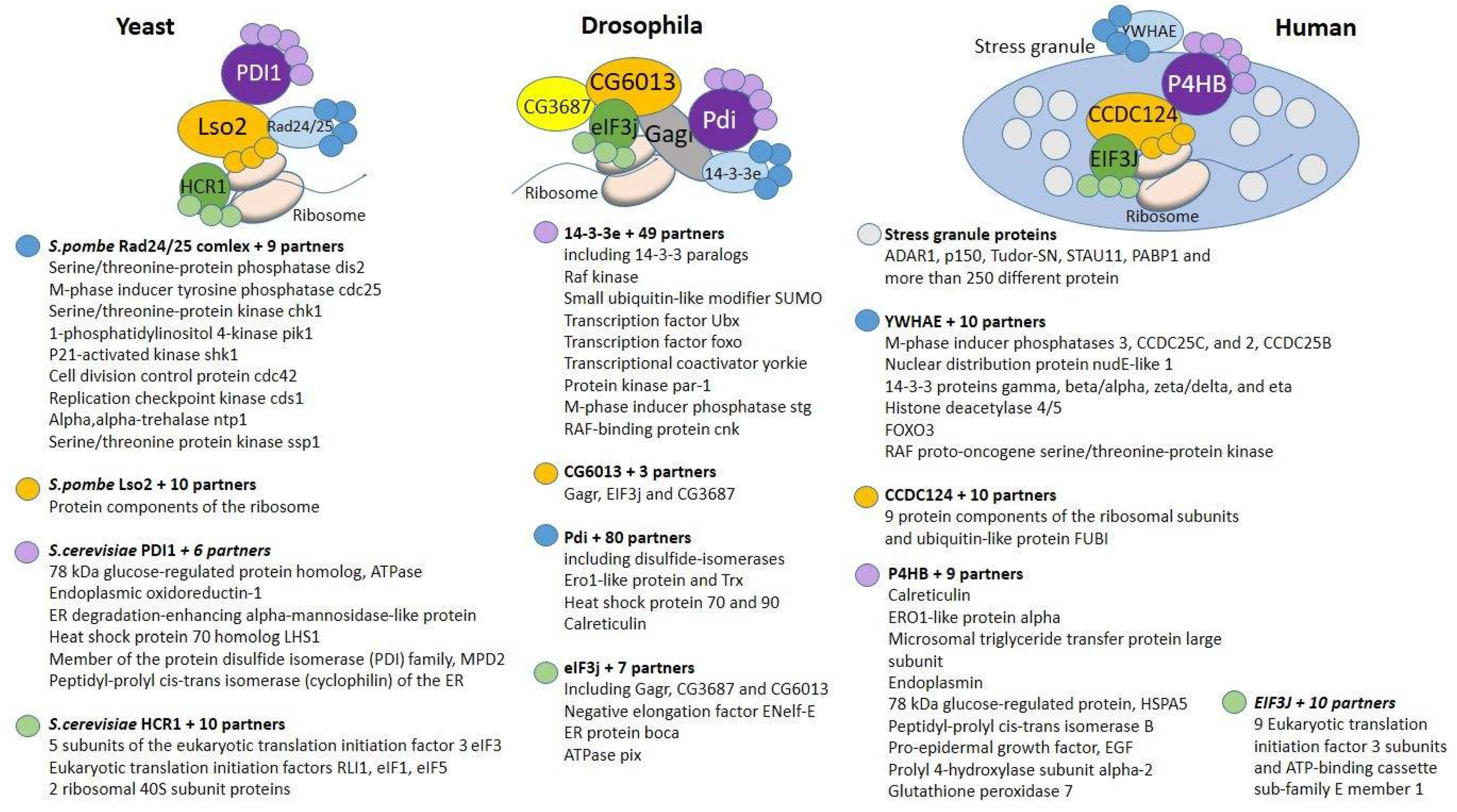
2. Components of the Complex That Are Conserved in Eukaryotes
2.1. 14-3-3e
2.2. Pdi
2.3. eIF3j
2.4. CG6013
3. Components of the Complex of Interactions That Have Arisen in Insects
3.1. CG3687
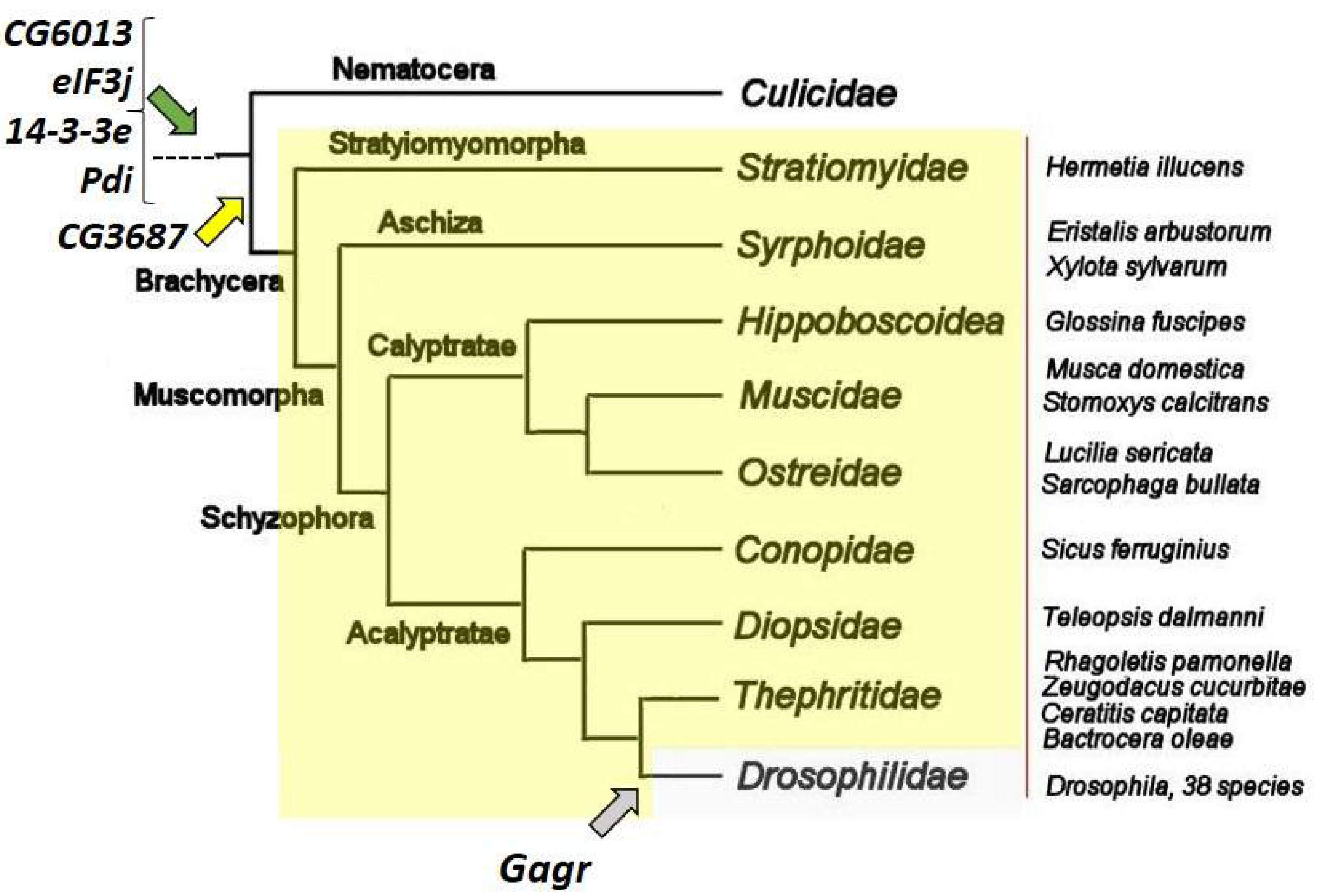
3.2. Gagr
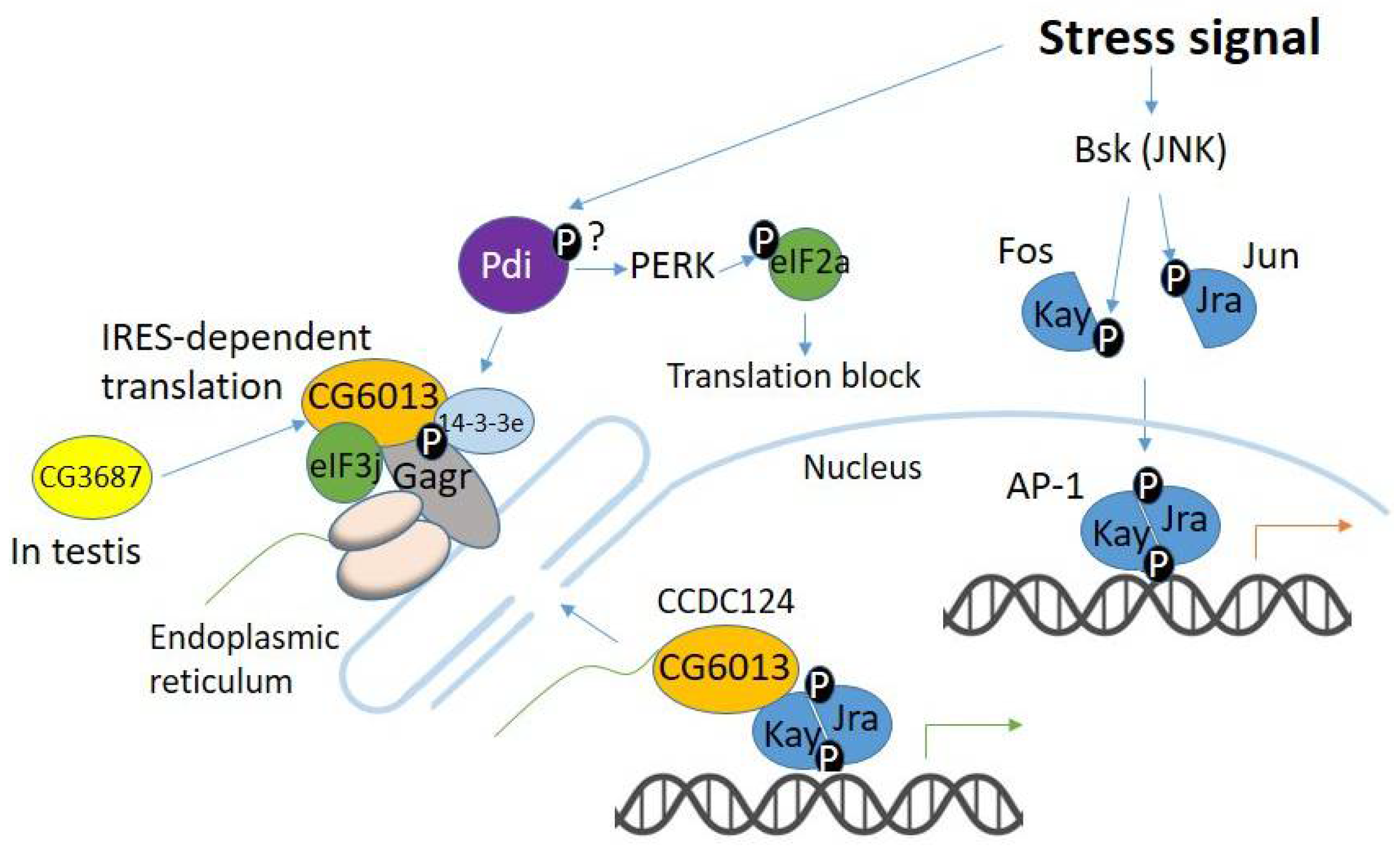
4. Evolution of Gagr and Its Partners in the Context of Stress-Signaling Pathways
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Miller, W.J.; Mcdonald, J.F.; Pinsker, W. Molecular domestication of mobile elements. Genetica 1997, 100, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Dupressoir, A.; Marceau, G.; Vernochet, C.; Benit, L.; Kanellopoulos, C.; Sapin, V.; Heidmann, T. Syncytin-A and Syncytin-B, two fusogenic placenta-specific murine envelope genes of retroviral origin conserved in Muridae. Proc. Natl. Acad. Sci. USA 2005, 102, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Emera, D.; Wagner, G.P. Transposable element recruitments in the mammalian placenta: Impacts and mechanisms. Brief. Funct. Genom. 2012, 11, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Henke, C.; Strissel, P.L.; Schubert, M.-T.; Mitchell, M.; Stolt, C.C.; Faschingbauer, F.; Beckmann, M.W.; Strick, R. Selective expression of sense and antisense transcripts of the sushi-ichi related retrotransposon-derived family during mouse placentogenesis. Retrovirology 2015, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Kämmerer, U.; Germeyer, A.; Stengel, S.; Kapp, M.; Denner, J. Human endogenous retrovirus K (HERV-K) is expressed in villous and extravillous cytotrophoblast cells of the human placenta. J. Reprod. Immunol. 2011, 91, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mallet, F.; Bouton, O.; Prudhomme, S.; Cheynet, V.; Oriol, G.; Bonnaud, B.; Lucotte, G.; Duret, L.; Mandrand, B. The endogenous retroviral locus ERVWE1 is a bona fide gene involved in hominoid placental physiology. Proc. Natl. Acad. Sci. USA 2004, 101, 1731–1736. [Google Scholar] [CrossRef]
- Benit, L.; Parseval, N.D.; Casella, J.F.; Callebaut, I.; Cordonnier, A.; Heidmann, T. Cloning of a new murine endogenous retrovirus, MuERV-L, with strong similarity to the human HERV-L element and with a gag coding sequence closely related to the Fv1 restriction gene. J. Virol. 1997, 71, 5652–5657. [Google Scholar] [CrossRef]
- Kaneko-Ishino, T.; Ishino, F. The role of genes domesticated from LTR retrotransposons and retroviruses in mammals. Front. Microbiol. 2012, 3, 262. [Google Scholar] [CrossRef]
- Yap, M.W.; Colbeck, E.; Ellis, S.A.; Stoye, J.P. Evolution of the retroviral restriction gene Fv1: Inhibition of non-MLV retroviruses. PLoS Pathog. 2014, 10, e1003968. [Google Scholar] [CrossRef]
- Schrader, L.; Schmitz, J. The impact of transposable elements in adaptive evolution. Mol. Ecol. 2019, 28, 1537–1549. [Google Scholar] [CrossRef]
- Nefedova, L.; Kim, A. Mechanisms of LTR-Retroelement Transposition: Lessons from Drosophila melanogaster. Viruses 2017, 16, 81. [Google Scholar] [CrossRef] [PubMed]
- Nefedova, L.N.; Kuzmin, I.V.; Makhnovskii, P.A.; Kim, A.I. Domesticated retroviral GAG gene in Drosophila: New functions for an old gene. Virology 2014, 450–451, 196–204. [Google Scholar] [CrossRef]
- Makhnovskii, P.; Balakireva, Y.; Nefedova, L.; Lavrenov, A.; Kuzmin, I.; Kim, A. Domesticated gag gene of drosophila LTR retrotransposons is involved in response to oxidative stress. Genes 2020, 11, 396. [Google Scholar] [CrossRef] [PubMed]
- Silverman, N.; Zhou, R.; Erlich, R.L.; Hunter, M.; Bernstein, E.; Schneider, D.; Maniatis, T. Immune activation of NF-kappaB and JNK requires Drosophila TAK1. J. Biol. Chem. 2003, 278, 48928–48934. [Google Scholar] [CrossRef] [PubMed]
- Kemp, C.; Mueller, S.; Goto, A.; Barbier, V.; Paro, S.; Bonnay, F.; Dostert, C.; Troxler, L.; Hetru, C.; Meignin, C.; et al. Broad RNA interference-mediated antiviral immunity and virus-specific inducible responses in Drosophila. J. Immunol. 2013, 190, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Guruharsha, K.G.; Rual, J.F.; Zhai, B.; Mintseris, J.; Vaidya, P.; Vaidya, N.; Beekman, C.; Wong, C.; Rhee, D.Y.; Cenaj, O.; et al. A Protein Complex Network of Drosophila melanogaster. Cell 2011, 147, 690–703. [Google Scholar] [CrossRef]
- Van Hemert, M.J.; Steensma, H.Y.; van Heusden, G.P. 14-3-3 proteins: Key regulators of cell division, signalling and apoptosis. Bioessays 2001, 23, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Parua, P.K.; Young, E.T. Binding and transcriptional regulation by 14-3-3 (Bmh) proteins requires residues outside of the canonical motif. Eukaryot Cell 2014, 13, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Muslin, A.J.; Tanner, J.W.; Allen, P.M.; Shaw, A.S. Interaction of 14-3-3 with signaling proteins is mediated by the recognition of phosphoserine. Cell 1996, 84, 889–897. [Google Scholar] [CrossRef]
- Lalle, M.; Leptourgidou, F.; Camerini, S.; Pozio, E.; Skoulakis, E.M. Interkingdom complementation reveals structural conservation and functional divergence of 14-3-3 proteins. PLoS ONE 2013, 8, e78090. [Google Scholar] [CrossRef]
- Kaundal, A.; Ramu, V.S.; Oh, S.; Lee, S.; Pant, B.; Lee, H.K.; Rojas, C.M.; Senthil-Kumar, M.; Mysore, K.S. General Control Nonrepressible4 degrades 14-3-3 and the RIN4 complex to regulate stomatal aperture with implications on nonhost disease resistance and drought tolerance. Plant Cell 2017, 29, 2233–2248. [Google Scholar] [CrossRef] [PubMed]
- Shin, R.; Jez, J.M.; Basra, A.; Zhang, B.; Schachtman, D.P. 14-3-3 proteins fine-tune plant nutrient metabolism. FEBS Lett. 2011, 585, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, T.H.; Walworth, N.C. Association of Chk1 with 14-3-3 proteins is stimulated by DNA damage. Genes Dev. 1999, 13, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.C.; al-Khodairy, F.; Fotou, E.; Sheldrick, K.S.; Griffiths, D.J.; Carr, A.M. 14-3-3 protein homologs required for the DNA damage checkpoint in fission yeast. Science 1994, 265, 533–535. [Google Scholar] [CrossRef] [PubMed]
- Ozoe, F.; Kurokawa, R.; Kobayashi, Y.; Jeong, H.T.; Tanaka, K.; Sen, K.; Nakagawa, T.; Matsuda, H.; Kawamukai, M. The 14-3-3 proteins Rad24 and Rad25 negatively regulate Byr2 by affecting its localization in Schizosaccharomyces pombe. Mol. Cell. Biol. 2002, 22, 7105–7119. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stoica, C.; Carmichael, J.B.; Parker, H.; Pare, J.; Hobman, T.C. Interactions between the RNA interference effector protein Ago1 and 14-3-3 proteins: Consequences for cell cycle progression. J. Biol. Chem. 2006, 281, 37646–37651. [Google Scholar] [CrossRef]
- Braun, K.A.; Parua, P.K.; Dombek, K.M.; Miner, G.E.; Young, E.T. 14-3-3 (Bmh) proteins regulate combinatorial transcription following RNA polymerase II recruitment by binding at Adr1-dependent promoters in Saccharomyces cerevisiae. Mol. Cell. Biol. 2013, 33, 712–724. [Google Scholar] [CrossRef]
- Trembley, M.A.; Berrus, H.L.; Whicher, J.R.; Humphrey-Dixon, E.L. The yeast 14-3-3 proteins BMH1 and BMH2 differentially regulate rapamycin-mediated transcription. Biosci. Rep. 2014, 34, e00099. [Google Scholar] [CrossRef]
- Conklin, D.S.; Galaktionov, K.; Beach, D. 14-3-3 proteins associate with cdc25 phosphatases. Proc. Natl. Acad. Sci. USA 1995, 92, 7892–7896. [Google Scholar] [CrossRef]
- Wang, X.; Grammatikakis, N.; Siganou, A.; Calderwood, S.K. Regulation of molecular chaperone gene transcription involves the serine phosphorylation, 14-3-3 epsilon binding, and cytoplasmic sequestration of heat shock factor 1. Mol. Cell. Biol. 2003, 23, 6013–6026. [Google Scholar] [CrossRef]
- Ren, F.; Zhang, L.; Jiang, J. Hippo signaling regulates Yorkie nuclear localization and activity through 14-3-3 dependent and independent mechanisms. Dev. Biol. 2010, 337, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Galligan, J.J.; Petersen, D.R. The human protein disulfide isomerase gene family. Hum. Genom. 2012, 6, 6. [Google Scholar] [CrossRef]
- Kivirikko, K.I.; Myllyharju, J. Prolyl 4-hydroxylases and their protein disulfide isomerase subunit. Matrix Biol. 1998, 16, 357–368. [Google Scholar] [CrossRef]
- Moretti, A.I.S.; Laurindo, F.R.M. Protein disulfide isomerases: Redox connections in and out of the endoplasmic reticulum. Arch. Biochem. Biophys. 2017, 617, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, C.; Stevens, T.H. The yeast EUG1 gene encodes an endoplasmic reticulum protein that is functionally related to protein disulfide isomerase. Mol. Cell. Biol. 1992, 12, 4601–4611. [Google Scholar]
- Palm, D.; Streit, D.; Shanmugam, T.; Weis, B.L.; Ruprecht, M.; Simm, S.; Schleiff, E. Plant-specific ribosome biogenesis factors in Arabidopsis thaliana with essential function in rRNA processing. Nucleic Acids Res. 2019, 47, 1880–1895. [Google Scholar] [CrossRef]
- Lighthouse, D.V.; Buszczak, M.; Spradling, A.C. New components of the Drosophila fusome suggest it plays novel roles in signaling and transport. Dev. Biol. 2008, 17, 59–71. [Google Scholar] [CrossRef]
- Giansanti, M.G.; Belloni, G.; Gatti, M. Rab11 is required for membrane trafficking and actomyosin ring constriction in meiotic cytokinesis of Drosophila males. Mol. Biol. Cell. 2007, 18, 5034–5047. [Google Scholar] [CrossRef]
- Hetz, C.; Russelakis-Carneiro, M.; Wälchli, S.; Carboni, S.; Vial-Knecht, E.; Maundrell, K.; Castilla, J.; Soto, C. The disulfide isomerase Grp58 is a protective factor against prion neurotoxicity. J. Neurosci. 2005, 25, 2793–2802. [Google Scholar] [CrossRef]
- Wilkinson, B.; Gilbert, H.F. Protein disulfide isomerase. Biochim. Biophys. Acta 2004, 1699, 35–44. [Google Scholar] [CrossRef]
- Yu, J.; Li, T.; Liu, Y.; Wang, X.; Zhang, J.; Wang, X.; Shi, G.; Lou, J.; Wang, L.; Wang, C.C.; et al. Phosphorylation switches protein disulfide isomerase activity to maintain proteostasis and attenuate ER stress. EMBO J. 2020, 39, e103841. [Google Scholar] [CrossRef]
- Sovolyova, N.; Healy, S.; Samali, A.; Logue, S.E. Stressed to death–mechanisms of ER stress-induced cell death. Biol. Chem. 2014, 395, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Clive, D.R.; Greene, J.J. Cooperation of protein disulfide isomerase and redox environment in the regulation of NF−κB and AP1 binding to DNA. Cell Biochem. Funct. 1996, 14, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Laurindo, F.R.; Pescatore, L.A.; de Castro Fernandes, D. Protein disulfide isomerase in redox cell signaling and homeostasis. Free Radic. Biol. Med. 2012, 52, 1954–1969. [Google Scholar] [CrossRef] [PubMed]
- Valásek, L.; Hasek, J.; Nielsen, K.H.; Hinnebusch, A.G. Dual function of eIF3j/Hcr1p in processing 20 S pre-rRNA and translation initiation. J. Biol. Chem. 2001, 276, 43351–43360. [Google Scholar] [CrossRef]
- Fraser, C.S.; Lee, J.Y.; Mayeur, G.L.; Bushell, M.; Doudna, J.A.; Hershey, J.W. The j-subunit of human translation initiation factor eIF3 is required for the stable binding of eIF3 and its subcomplexes to 40 S ribosomal subunits in vitro. J. Biol. Chem. 2004, 279, 8946–8956. [Google Scholar] [CrossRef]
- Majzoub, K.; Hafirassou, M.L.; Meignin, C.; Goto, A.; Marzi, S.; Fedorova, A.; Verdier, Y.; Vinh, J.; Hoffmann, J.A.; Martin, F.; et al. RACK1 controls IRES-mediated translation of viruses. Cell 2014, 159, 1086–1095. [Google Scholar] [CrossRef]
- Komar, A.A.; Hatzoglou, M. Cellular IRES-mediated translation. Cell Cycle 2011, 10, 229–240. [Google Scholar] [CrossRef]
- Spriggs, K.A.; Stoneley, M.; Bushell, M.; Willis, A.E. Re-programming of translation following cell stress allows IRES-mediated translation to predominate. Biol. Cell 2008, 100, 27–38. [Google Scholar] [CrossRef]
- Johnson, A.G.; Grosely, R.; Petrov, A.N.; Puglisi, J.D. Dynamics of IRES-mediated translation. Phil. Trans. R Soc. 2017, 372, 20160177. [Google Scholar] [CrossRef]
- He, Y.; Chen, Y.; Song, W.; Zhu, L.; Dong, Z.; Ow, D.W. A Pap1–Oxs1 signaling pathway for disulfide stress in Schizosaccharomyces pombe. Nucleic Acid Res. 2017, 45, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.N.; Buschauer, R.; Mackens-Kiani, T.; Best, K.; Kratzat, H.; Berninghausen, O.; Becker, T.; Gilbert, W.; Cheng, J.; Beckmann, R. Structure and function of yeast Lso2 and human CCDC124 bound to hibernating ribosomes. PLoS Biol. 2020, 18, e3000780. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Dong, Z.; Ow, D.W. Protection from disulfide stress by inhibition of Pap1 nuclear export in Schizosaccharomyces pombe. Genetics 2018, 210, 857–868. [Google Scholar] [CrossRef]
- Wang, Y.J.; Vaidyanathan, P.P.; Rojas-Duran, M.F.; Udeshi, N.D.; Bartoli, K.M.; Carr, S.A.; Gilbert, W.V. Lso2 is a conserved ribosome-bound protein required for translational recovery in yeast. PLoS Biol. 2018, 16, e2005903. [Google Scholar] [CrossRef]
- Kudo, N.; Taoka, H.; Toda, T.; Horinouchi, S. A novel nuclear export signal sensitive to oxidative stress in the fission yeast transcription factor Pap1. J. Biol. Chem. 1999, 274, 15151–15158. [Google Scholar] [CrossRef]
- Castello, A.; Fischer, B.; Eichelbaum, K.; Horos, R.; Beckmann, B.M.; Strein, C.; Davey, N.E.; Humphreys, D.T.; Preiss, T.; Steinmetz, L.M.; et al. Insights into RNA biology from an atlas of mammalian mRNA-binding proteins. Cell 2012, 149, 1393–1406. [Google Scholar] [CrossRef]
- Li, S.; Wang, L.; Fu, B.; Berman, M.A.; Diallo, A.; Dorf, M.E. TRIM65 regulates microRNA activity by ubiquitination of TNRC6. Proc. Natl. Acad. Sci. USA 2014, 111, 6970–6975. [Google Scholar] [CrossRef]
- Malinová, A.; Cvačková, Z.; Matějů, D.; Hořejší, Z.; Abéza, C.; Vandermoere, F.; Bertrand, E.; Staněk, D.; Verheggen, C. Assembly of the U5 snRNP component PRPF8 is controlled by the HSP90/R2TP chaperones. J. Cell Biol. 2017, 216, 1579–1596. [Google Scholar] [CrossRef] [PubMed]
- Kırlı, K.; Karaca, S.; Dehne, H.J.; Samwer, M.; Pan, K.T.; Lenz, C.; Urlaub, H.; Görlich, D. A deep proteomics perspective on CRM1-mediated nuclear export and nucleocytoplasmic partitioning. Elife 2015, 4, e11466. [Google Scholar] [CrossRef] [PubMed]
- Telkoparan, P.; Erkek, S.; Yaman, E.; Alotaibi, H.; Bayık, D.; Tazebay, U.H. Coiled-coil domain containing protein 124 is a novel centrosome and midbody protein that interacts with the Ras-guanine nucleotide exchange factor 1B and is involved in cytokinesis. PLoS ONE 2013, 8, e69289. [Google Scholar] [CrossRef] [PubMed]
- Schnorrer, F.; Schönbauer, C.; Langer, C.C.; Dietzl, G.; Novatchkova, M.; Schernhuber, K.; Fellner, M.; Azaryan, A.; Radolf, M.; Stark, A.; et al. Systematic genetic analysis of muscle morphogenesis and function in Drosophila. Nature 2010, 464, 287–291. [Google Scholar] [CrossRef]
- Larkin, A.; Marygold, S.J.; Antonazzo, G.; Attrill, H.; dos Santos, G.; Garapati, P.V.; Goodman, J.L.; Gramates, L.S.; Millburn, G.; Strelets, V.B.; et al. FlyBase: Updates to the Drosophila melanogaster knowledge base. Nucleic Acids Res. 2021, 49, D899–D907. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Yeates, D.K.; Wiegmann, B.M. Congruence and controversy: Toward a higher-level phylogeny of the Diptera. Annu. Rev. Entomol. 1999, 44, 397–428. [Google Scholar] [CrossRef] [PubMed]
- Higgins, D.; Thompson, J.; Gibson, T.; Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar]
- Pagni, M.; Ioannidis, V.; Cerutti, L.; Zahn-Zabal, M.; Jongeneel, C.V.; Hau, J.; Martin, O.; Kuznetsov, D.; Falquet, L. MyHits: Improvements to an interactive resource for analyzing protein sequences. Nucleic Acids Res. 2007, 35, W433–W437. [Google Scholar] [CrossRef]
- Yan, J.; Kurgan, L.A. DRNApred, fast sequence-based method that accurately predicts and discriminates DNA- and RNA-binding residues. Nucleic Acids Res. 2017, 45, e84. [Google Scholar] [CrossRef] [PubMed]
- Celniker, S.E.; Dillon, L.A.; Gerstein, M.B.; Gunsalus, K.C.; Henikoff, S.; Karpen, G.H.; Kellis, M.; Lai, E.C.; Lieb, J.D.; MacAlpine, D.M.; et al. modENCODE Consortium. Unlocking the secrets of the genome. Nature 2009, 459, 927–930. [Google Scholar] [CrossRef] [PubMed]
- Dietzl, G.; Chen, D.; Schnorrer, F.; Su, K.C.; Barinova, Y.; Fellner, M.; Gasser, B.; Kinsey, K.; Oppel, S.; Scheiblauer, S.; et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 2007, 448, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Esmail, S.; Manolson, M.F. Advances in understanding N-glycosylation structure, function, and regulation in health and disease. Eur. J. Cell Biol. 2021, 100, 151186. [Google Scholar] [CrossRef]
- Braakman, I.; Hebert, D.N. Protein folding in the endoplasmic reticulum. Cold Spring Harb. Perspect. Biol. 2013, 5, a013201. [Google Scholar] [CrossRef] [PubMed]
- Tannous, A.; Patel, N.; Tamura, T.; Hebert, D.N. Reglucosylation by UDP-glucose:glycoprotein glucosyltransferase 1 delays glycoprotein secretion but not degradation. Mol. Biol. Cell 2015, 26, 390–405. [Google Scholar] [CrossRef] [PubMed]
- Tomancak, P.; Berman, B.P.; Beaton, A.; Weiszmann, R.; Kwan, E.; Hartenstein, V.; Celniker, S.E.; Rubin, G.M. Global analysis of patterns of gene expression during Drosophila embryogenesis. Genome Biol. 2007, 8, R145. [Google Scholar] [CrossRef] [PubMed]
- Dorus, S.; Busby, S.; Gerike, U.; Shabanowitz, J.; Hunt, D.F.; Karr, T.L. Genomic and functional evolution of the Drosophila melanogaster sperm proteome. Nat. Genet. 2006, 38, 1440–1445. [Google Scholar] [CrossRef] [PubMed]
- Markmiller, S.; Soltanieh, S.; Server, K.L.; Mak, R.; Jin, W.; Fang, M.Y.; Luo, E.C.; Krach, F.; Yang, D.; Sen, A.; et al. Context-dependent and disease-specific diversity in protein interactions within stress granules. Cell 2018, 172, 590–604. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P.; et al. The STRING database in 2021: Customizable protein–protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021, 49, D605–D612. [Google Scholar] [CrossRef]
- Santabarbara-Ruiz, P.; Lopez-Santillan, M.; Martinez-Rodriguez, I.; Binagui-Casas, A.; Perez, L.; Milan, M.; Corominas, M.; Serras, F. ROS-Induced JNK and p38 signaling is required for unpaired cytokine activation during Drosophila regeneration. PLoS Genet. 2015, 11, e1005595. [Google Scholar] [CrossRef]
- Johnson, G.L.; Nakamura, K. The c-jun kinase/stress-activated pathway: Regulation, function and role in human disease. Biochim. Biophys. Acta 2007, 1773, 1341–1348. [Google Scholar] [CrossRef]
- Zeitlinger, J.; Kockel, L.; Peverali, F.A.; Jackson, D.B.; Mlodzik, M.; Bohmann, D. Defective dorsal closure and loss of epidermal decapentaplegic expression in Drosophila fos mutants. EMBO J. 1997, 16, 7393–7401. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Dudzic, J.P.; Li, X.; Collas, E.J.; Boquete, J.P.; Lemaitre, B. Remote control of intestinal stem cell activity by haemocytes in Drosophila. PLoS Genet. 2016, 12, e1006089. [Google Scholar] [CrossRef]
- Fox, D.T.; Spradling, A.C. The Drosophila hindgut lacks constitutively active adult stem cells but proliferates in response to tissue damage. Cell Stem Cell 2009, 5, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Kanatsu-Shinohara, M.; Yamamoto, T.; Toh, H.; Kazuki, Y.; Kazuki, K.; Imoto, J.; Ikeo, K.; Oshima, M.; Shirahige, K.; Iwama, A.; et al. Aging of spermatogonial stem cells by Jnk-mediated glycolysis activation. Proc. Natl. Acad. Sci. USA 2019, 116, 16404–16409. [Google Scholar] [CrossRef] [PubMed]
- Herrera, S.C.; Bach, E.A. The Emerging Roles of JNK Signaling in Drosophila Stem Cell Homeostasis. Int. J. Mol. Sci. 2021, 22, 5519. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nefedova, L.; Gigin, A.; Kim, A. Domesticated LTR-Retrotransposon gag-Related Gene (Gagr) as a Member of the Stress Response Network in Drosophila. Life 2022, 12, 364. https://doi.org/10.3390/life12030364
Nefedova L, Gigin A, Kim A. Domesticated LTR-Retrotransposon gag-Related Gene (Gagr) as a Member of the Stress Response Network in Drosophila. Life. 2022; 12(3):364. https://doi.org/10.3390/life12030364
Chicago/Turabian StyleNefedova, Lidia, Alexey Gigin, and Alexander Kim. 2022. "Domesticated LTR-Retrotransposon gag-Related Gene (Gagr) as a Member of the Stress Response Network in Drosophila" Life 12, no. 3: 364. https://doi.org/10.3390/life12030364
APA StyleNefedova, L., Gigin, A., & Kim, A. (2022). Domesticated LTR-Retrotransposon gag-Related Gene (Gagr) as a Member of the Stress Response Network in Drosophila. Life, 12(3), 364. https://doi.org/10.3390/life12030364




