The Role of Extracellular Matrix and Hydrogels in Mesenchymal Stem Cell Chondrogenesis and Cartilage Regeneration
Abstract
1. Introduction
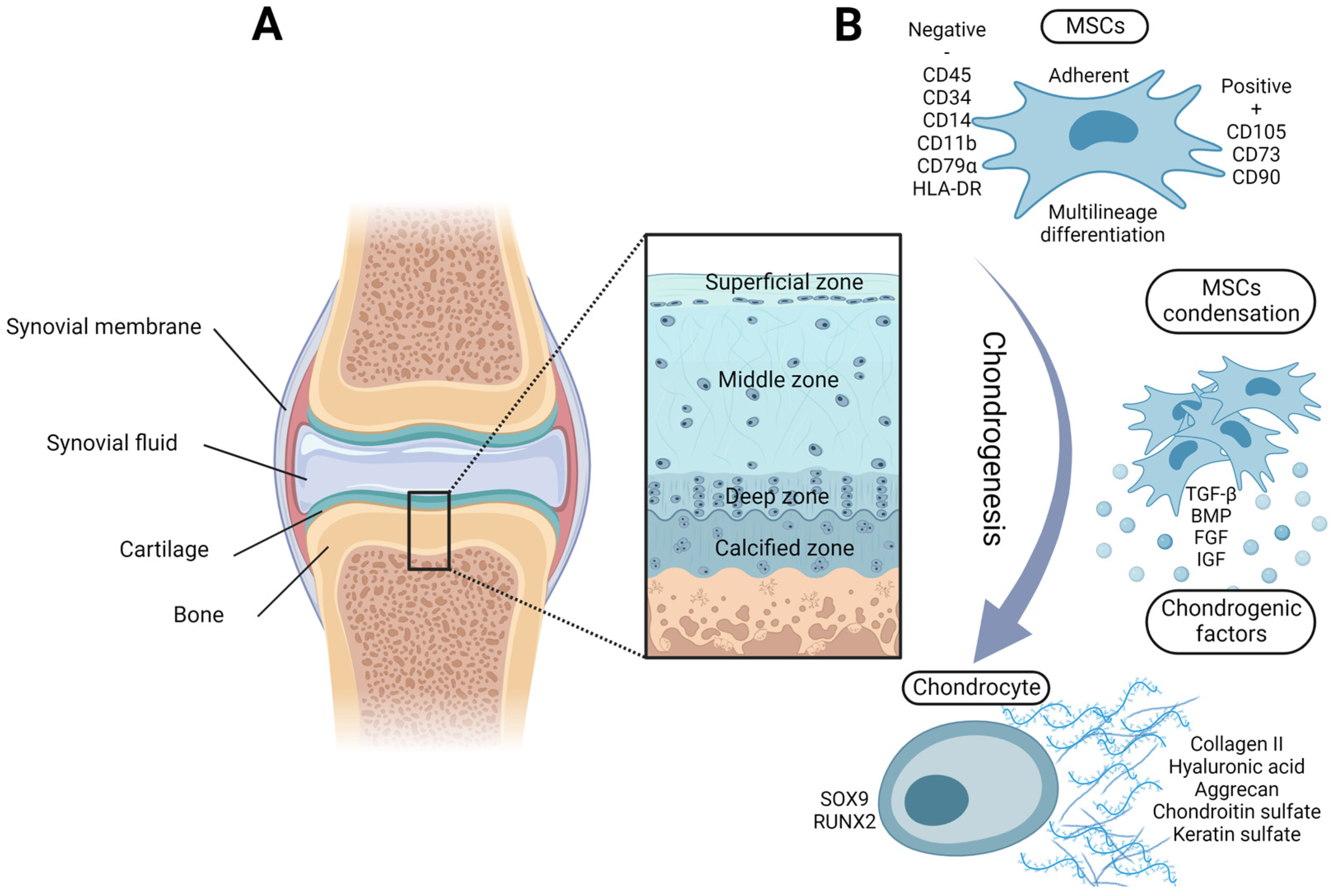
2. Chondrogenic Potential of Adult MSCs
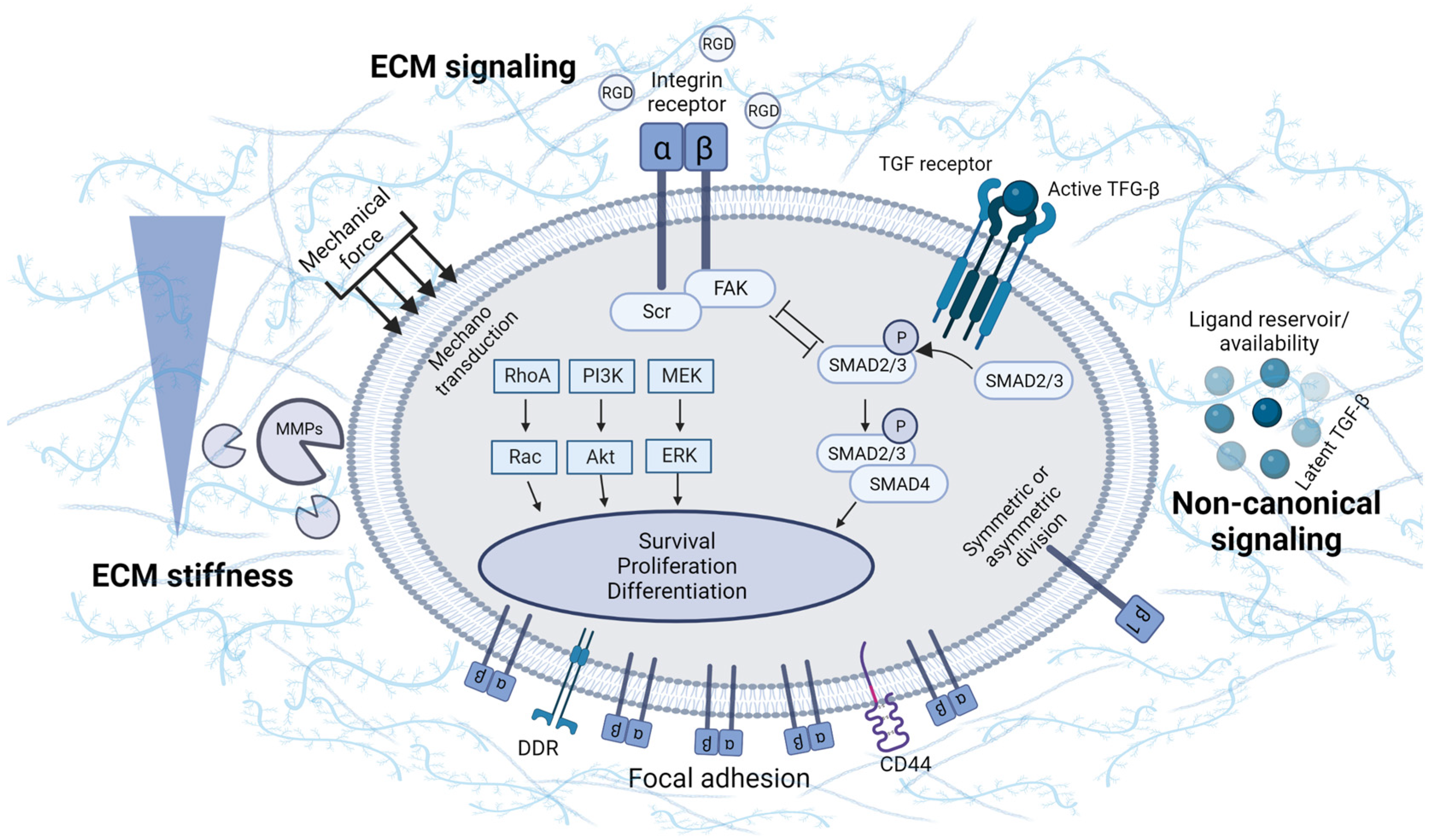
3. Involvement of ECM Physical Properties and Signaling in MSC Chondrogenesis
4. Hydrogels
4.1. Natural Polymers for Hydrogels
4.2. Synthetic Polymers for Hydrogels
4.3. Injectable and Functionalized Hydrogels
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The Basic Science of Articular Cartilage: Structure, Composition, and Function. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Burrage, P.S.; Mix, K.S.; Brinckerhoff, C.E. Matrix Metalloproteinases: Role in Arthritis. Front. Biosci. 2006, 11, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Bhosale, A.M.; Richardson, J.B. Articular Cartilage: Structure, Injuries and Review of Management. Br. Med. Bull. 2008, 87, 77–95. [Google Scholar] [CrossRef] [PubMed]
- Debreova, M.; Culenova, M.; Smolinska, V.; Nicodemou, A.; Csobonyeiova, M.; Danisovic, L. Rheumatoid Arthritis: From Synovium Biology to Cell-Based Therapy. Cytotherapy 2022, 24, 365–375. [Google Scholar] [CrossRef]
- Johnstone, B.; Yoo, J.U. Autologous Mesenchymal Progenitor Cells in Articular Cartilage Repair. Clin. Orthop. Relat. Res. 1999, 367, 156–162. [Google Scholar] [CrossRef]
- Convery, F.R.; Akeson, W.H.; Keown, G.H. The Repair of Large Osteochondral Defects. An Experimental Study in Horses. Clin. Orthop. Relat. Res. 1972, 82, 253–262. [Google Scholar] [CrossRef]
- Litwic, A.; Edwards, M.H.; Dennison, E.M.; Cooper, C. Epidemiology and Burden of Osteoarthritis. Br. Med. Bull. 2013, 105, 185–199. [Google Scholar] [CrossRef]
- Hootman, J.M.; Helmick, C.G. Projections of US Prevalence of Arthritis and Associated Activity Limitations. Arthritis Rheum. 2006, 54, 226–229. [Google Scholar] [CrossRef]
- Pilz, V.; Hanstein, T.; Skripitz, R. Projections of Primary Hip Arthroplasty in Germany until 2040. Acta Orthop. 2018, 89, 308–313. [Google Scholar] [CrossRef]
- Gaffo, A.; Saag, K.G.; Curtis, J.R. Treatment of Rheumatoid Arthritis. Am. J. Heal. Pharm. 2006, 63, 2451–2465. [Google Scholar] [CrossRef]
- Caplan, A.I. Mesenchymal Stem Cells. J. Orthop. Res. 1991, 9, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Friedenstein, A.J.; Piatetzky-Shapiro, I.I.; Petrakova, K.V. Osteogenesis in Transplants of Bone Marrow Cells. J. Embryol. Exp. Morphol. 1966, 16, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human Adipose Tissue Is a Source of Multipotent Stem Cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Jones, E.A.; English, A.; Henshaw, K.; Kinsey, S.E.; Markham, A.F.; Emery, P.; McGonagle, D. Enumeration and Phenotypic Characterization of Synovial Fluid Multipotential Mesenchymal Progenitor Cells in Inflammatory and Degenerative Arthritis. Arthritis Rheum. 2004, 50, 817–827. [Google Scholar] [CrossRef] [PubMed]
- De Bari, C.; Dell’Accio, F.; Tylzanowski, P.; Luyten, F.P. Multipotent Mesenchymal Stem Cells from Adult Human Synovial Membrane. Arthritis Rheum. 2001, 44, 1928–1942. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; McNeill, E.; Tian, H.; Soker, S.; Andersson, K.E.; Yoo, J.J.; Atala, A. Urine Derived Cells Are a Potential Source for Urological Tissue Reconstruction. J. Urol. 2008, 180, 2226–2233. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal Criteria for Defining Multipotent Mesenchymal Stromal Cells. The International Society for Cellular Therapy Position Statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- García-Bernal, D.; García-Arranz, M.; Yáñez, R.M.; Hervás-Salcedo, R.; Cortés, A.; Fernández-García, M.; Hernando-Rodríguez, M.; Quintana-Bustamante, Ó.; Bueren, J.A.; García-Olmo, D.; et al. The Current Status of Mesenchymal Stromal Cells: Controversies, Unresolved Issues and Some Promising Solutions to Improve Their Therapeutic Efficacy. Front. Cell Dev. Biol. 2021, 9, 650–664. [Google Scholar] [CrossRef]
- Dounchis, J.S.; Goomer, R.S.; Harwood, F.L.; Khatod, M.; Coutts, R.D.; Amiel, D. Chondrogenic Phenotype of Perichondrium-Derived Chondroprogenitor Cells Is Influenced by Transforming Growth Factor-Beta 1. J. Orthop. Res. 1997, 15, 803–807. [Google Scholar] [CrossRef]
- Park, J.; Gelse, K.; Frank, S.; von der Mark, K.; Aigner, T.; Schneider, H. Transgene-Activated Mesenchymal Cells for Articular Cartilage Repair: A Comparison of Primary Bone Marrow-, Perichondrium/Periosteum- and Fat-Derived Cells. J. Gene Med. 2006, 8, 112–125. [Google Scholar] [CrossRef]
- Goldring, M.B.; Tsuchimochi, K.; Ijiri, K. The Control of Chondrogenesis. J. Cell. Biochem. 2006, 97, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Umansky, R. The Effect of Cell Population Density on the Developmental Fate of Reaggregating Mouse Limb Bud Mesenchyme. Dev. Biol. 1966, 13, 31–56. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, P.B.; Solursh, M.; Reiter, R.S. Stage-Related Capacity for Limb Chondrogenesis in Cell Culture. Dev. Biol. 1977, 60, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Uzieliene, I.; Bironaite, D.; Bernotas, P.; Sobolev, A.; Bernotiene, E. Mechanotransducive Biomimetic Systems for Chondrogenic Differentiation In Vitro. Int. J. Mol. Sci. 2021, 22, 9690. [Google Scholar] [CrossRef]
- Danišovič, Ľ.; Varga, I.; Polák, Š. Growth Factors and Chondrogenic Differentiation of Mesenchymal Stem Cells. Tissue Cell 2012, 44, 69–73. [Google Scholar] [CrossRef]
- Goldring, M.B. Chondrogenesis, Chondrocyte Differentiation, and Articular Cartilage Metabolism in Health and Osteoarthritis. Ther. Adv. Musculoskelet. Dis. 2012, 4, 269–285. [Google Scholar] [CrossRef]
- Pelttari, K.; Winter, A.; Steck, E.; Goetzke, K.; Hennig, T.; Ochs, B.G.; Aigner, T.; Richter, W. Premature Induction of Hypertrophy during in Vitro Chondrogenesis of Human Mesenchymal Stem Cells Correlates with Calcification and Vascular Invasion after Ectopic Transplantation in SCID Mice. Arthritis Rheum. 2006, 54, 3254–3266. [Google Scholar] [CrossRef]
- Mueller, M.B.; Tuan, R.S. Functional Characterization of Hypertrophy in Chondrogenesis of Human Mesenchymal Stem Cells. Arthritis Rheum. 2008, 58, 1377–1388. [Google Scholar] [CrossRef]
- Dudics, V.; Kunstár, A.; Kovács, J.; Lakatos, T.; Géher, P.; Gömör, B.; Monostori, E.; Uher, F. Chondrogenic Potential of Mesenchymal Stem Cells from Patients with Rheumatoid Arthritis and Osteoarthritis: Measurements in a Microculture System. Cells Tissues Organs 2009, 189, 307–316. [Google Scholar] [CrossRef]
- Scharstuhl, A.; Schewe, B.; Benz, K.; Gaissmaier, C.; Bühring, H.-J.; Stoop, R. Chondrogenic Potential of Human Adult Mesenchymal Stem Cells Is Independent of Age or Osteoarthritis Etiology. Stem. Cells 2007, 25, 3244–3251. [Google Scholar] [CrossRef]
- Danišovič, L.; Lesný, P.; Havlas, V.; Teyssler, P.; Syrová, Z.; Kopáni, M.; Fujeríková, G.; Trč, T.; Syková, E.; Jendelová, P. Chondrogenic Differentiation of Human Bone Marrow and Adipose Tissue-Derived Mesenchymal Stem Cells. J. Appl. Biomed. 2007, 5, 139–150. [Google Scholar] [CrossRef]
- Hennig, T.; Lorenz, H.; Thiel, A.; Goetzke, K.; Dickhut, A.; Geiger, F.; Richter, W. Reduced Chondrogenic Potential of Adipose Tissue Derived Stromal Cells Correlates with an Altered TGFβ Receptor and BMP Profile and Is Overcome by BMP-6. J. Cell. Physiol. 2007, 211, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.H.; Rogan, H.; Kajiyama, G.; Goodman, S.B.; Smith, R.L.; Maloney, W.; Yang, F. Interaction between Osteoarthritic Chondrocytes and Adipose-Derived Stem Cells Is Dependent on Cell Distribution in Three-Dimension and Transforming Growth Factor-Β3 Induction. Tissue Eng. Part A 2015, 21, 992–1002. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Chen, P.; Wu, Y.; Xiong, S.; Sun, H.; Xia, Q.; Shi, L.; Liu, H.; Ouyang, H.W. Programmed Application of Transforming Growth Factor Β3 and Rac1 Inhibitor NSC23766 Committed Hyaline Cartilage Differentiation of Adipose-Derived Stem Cells for Osteochondral Defect Repair. Stem. Cells Transl. Med. 2014, 3, 1242–1251. [Google Scholar] [CrossRef]
- Shirasawa, S.; Sekiya, I.; Sakaguchi, Y.; Yagishita, K.; Ichinose, S.; Muneta, T. In Vitro Chondrogenesis of Human Synovium-Derived Mesenchymal Stem Cells: Optimal Condition and Comparison with Bone Marrow-Derived Cells. J. Cell. Biochem. 2006, 97, 84–97. [Google Scholar] [CrossRef]
- McCarthy, H.E.; Bara, J.J.; Brakspear, K.; Singhrao, S.K.; Archer, C.W. The Comparison of Equine Articular Cartilage Progenitor Cells and Bone Marrow-Derived Stromal Cells as Potential Cell Sources for Cartilage Repair in the Horse. Vet. J. 2012, 192, 345–351. [Google Scholar] [CrossRef]
- Jones, E.A.; Crawford, A.; English, A.; Henshaw, K.; Mundy, J.; Corscadden, D.; Chapman, T.; Emery, P.; Hatton, P.; McGonagle, D. Synovial Fluid Mesenchymal Stem Cells in Health and Early Osteoarthritis: Detection and Functional Evaluation at the Single-Cell Level. Arthritis Rheum. 2008, 58, 1731–1740. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, W.J.; Hwang, S.C.; Choe, Y.; Kim, S.; Bok, E.; Lee, S.; Kim, S.J.; Kim, H.O.; Ock, S.A.; et al. Chronic Inflammation-Induced Senescence Impairs Immunomodulatory Properties of Synovial Fluid Mesenchymal Stem Cells in Rheumatoid Arthritis. Stem Cell Res. Ther. 2021, 12, 502. [Google Scholar] [CrossRef]
- Arufe, M.C.; De La Fuente, M.C.; Fuentes-Boquete, I.; De Toro, F.J.; Blanco, F.J. Differentiation of Synovial CD-105+ Human Mesenchymal Stem Cells into Chondrocyte-like Cells through Spheroid Formation. J. Cell. Biochem. 2009, 108, 145–155. [Google Scholar] [CrossRef]
- Gullo, F.; De Bari, C. Prospective Purification of a Subpopulation of Human Synovial Mesenchymal Stem Cells with Enhanced Chondro-Osteogenic Potency. Rheumatology 2013, 52, 1758–1768. [Google Scholar] [CrossRef][Green Version]
- Chen, L.; Li, L.; Xing, F.; Peng, J.; Peng, K.; Wang, Y.; Xiang, Z. Human Urine-Derived Stem Cells: Potential for Cell-Based Therapy of Cartilage Defects. Stem. Cells Int. 2018, 2018, 4686259. [Google Scholar] [CrossRef] [PubMed]
- Culenova, M.; Nicodemou, A.; Novakova, Z.V.; Debreova, M.; Smolinská, V.; Bernatova, S.; Ivanisova, D.; Novotna, O.; Vasicek, J.; Varga, I.; et al. Isolation, Culture and Comprehensive Characterization of Biological Properties of Human Urine-Derived Stem Cells. Int. J. Mol. Sci. 2021, 22, 12503. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Xing, F.; Zou, M.; Gong, M.; Li, L.; Xiang, Z. Comparison of Chondrogenesis-Related Biological Behaviors between Human Urine-Derived Stem Cells and Human Bone Marrow Mesenchymal Stem Cells from the Same Individual. Stem. Cell Res. Ther. 2021, 12, 366. [Google Scholar] [CrossRef] [PubMed]
- Pei, M.; Pei, Y.A.; Zhou, S.; Mikaeiliagah, E.; Erickson, C.; Giertych, B.; Akhter, H.; Wang, L.; Stewart, A.; Parenti, J.; et al. Matrix from Urine Stem Cells Boosts Tissue-Specific Stem Cell Mediated Functional Cartilage Reconstruction. Bioact. Mater. 2023, 23, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Pei, M.; Li, J.; Zhang, Y.; Liu, G.; Wei, L.; Zhang, Y. Expansion on a Matrix Deposited by Nonchondrogenic Urine Stem Cells Strengthens the Chondrogenic Capacity of Repeated-Passage Bone Marrow Stromal Cells. Cell Tissue Res. 2014, 356, 391–403. [Google Scholar] [CrossRef]
- Hynes, R.O. The Extracellular Matrix: Not Just Pretty Fibrils. Science 2009, 326, 1216–1219. [Google Scholar] [CrossRef]
- Marinkovic, M.; Tran, O.N.; Block, T.J.; Rakian, R.; Gonzalez, A.O.; Dean, D.D.; Yeh, C.K.; Chen, X.D. Native Extracellular Matrix, Synthesized Ex Vivo by Bone Marrow or Adipose Stromal Cells, Faithfully Directs Mesenchymal Stem Cell Differentiation. Matrix Biol. Plus 2020, 8, 100044. [Google Scholar] [CrossRef]
- Sun, Y.; Yan, L.; Chen, S.; Pei, M. Functionality of Decellularized Matrix in Cartilage Regeneration: A Comparison of Tissue versus Cell Sources. Acta Biomater. 2018, 74, 56–73. [Google Scholar] [CrossRef]
- Rothrauff, B.B.; Yang, G.; Tuan, R.S. Tissue-Specific Bioactivity of Soluble Tendon-Derived and Cartilage-Derived Extracellular Matrices on Adult Mesenchymal Stem Cells. Stem Cell Res. Ther. 2017, 8, 133. [Google Scholar] [CrossRef]
- Matsiko, A.; Gleeson, J.P.; O’Brien, F.J. Scaffold Mean Pore Size Influences Mesenchymal Stem Cell Chondrogenic Differentiation and Matrix Deposition. Tissue Eng. Part A 2015, 21, 486–497. [Google Scholar] [CrossRef]
- Nava, M.M.; Draghi, L.; Giordano, C.; Pietrabissa, R. The Effect of Scaffold Pore Size in Cartilage Tissue Engineering. J. Appl. Biomater. Funct. Mater. 2016, 14, e223–e229. [Google Scholar] [CrossRef] [PubMed]
- Mammoto, A.; Mammoto, T.; Ingber, D.E. Mechanosensitive Mechanisms in Transcriptional Regulation. J. Cell Sci. 2012, 125, 3061–3073. [Google Scholar] [CrossRef] [PubMed]
- Pardo-Saganta, A.; Calvo, I.A.; Saez, B.; Prosper, F. Role of the Extracellular Matrix in Stem Cell Maintenance. Curr. Stem Cell Reports 2019, 5, 1–10. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in Mechanotransduction. Nature 2011, 474, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.L.; Cooke, M.E.; Alliston, T. ECM Stiffness Primes the TGFβ Pathway to Promote Chondrocyte Differentiation. Mol. Biol. Cell 2012, 23, 3731–3742. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Chu, J.S.; Tsou, A.D.; Diop, R.; Tang, Z.; Wang, A.; Li, S. The Effect of Matrix Stiffness on the Differentiation of Mesenchymal Stem Cells in Response to TGF-β. Biomaterials 2011, 32, 3921–3930. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.M.; Matsiko, A.; Haugh, M.G.; Gleeson, J.P.; O’Brien, F.J. Mesenchymal Stem Cell Fate Is Regulated by the Composition and Mechanical Properties of Collagen-Glycosaminoglycan Scaffolds. J. Mech. Behav. Biomed. Mater. 2012, 11, 53–62. [Google Scholar] [CrossRef]
- Bian, L.; Hou, C.; Tous, E.; Rai, R.; Mauck, R.L.; Burdick, J.A. The Influence of Hyaluronic Acid Hydrogel Crosslinking Density and Macromolecular Diffusivity on Human MSC Chondrogenesis and Hypertrophy. Biomaterials 2013, 34, 413–421. [Google Scholar] [CrossRef]
- Wang, T.; Lai, J.H.; Han, L.H.; Tong, X.; Yang, F. Chondrogenic Differentiation of Adipose-Derived Stromal Cells in Combinatorial Hydrogels Containing Cartilage Matrix Proteins with Decoupled Mechanical Stiffness. Tissue Eng.-Part A 2014, 20, 2131–2139. [Google Scholar] [CrossRef]
- Underhill, C. CD44: The Hyaluronan Receptor. J. Cell Sci. 1992, 103, 293–298. [Google Scholar] [CrossRef]
- Vogel, W.; Gish, G.D.; Alves, F.; Pawson, T. The Discoidin Domain Receptor Tyrosine Kinases Are Activated by Collagen. Mol. Cell 1997, 1, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Song, E.K.; Park, T.J. Integrin Signaling in Cartilage Development. Animal Cells Syst. 2014, 18, 365–371. [Google Scholar] [CrossRef]
- Bang, O.S.; Kim, E.J.; Chung, J.G.; Lee, S.R.; Park, T.K.; Kang, S.S. Association of Focal Adhesion Kinase with Fibronectin and Paxillin Is Required for Precartilage Condensation of Chick Mesenchymal Cells. Biochem. Biophys. Res. Commun. 2000, 278, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.G.; Ko, J.; Lee, H.R.; Do, S.H.; Park, K. Mesenchymal Cells Condensation-Inducible Mesh Scaffolds for Cartilage Tissue Engineering. Biomaterials 2016, 85, 18–29. [Google Scholar] [CrossRef]
- Jin, E.J.; Choi, Y.A.; Kyun Park, E.; Bang, O.S.; Kang, S.S. MMP-2 Functions as a Negative Regulator of Chondrogenic Cell Condensation via down-Regulation of the FAK-Integrin Β1 Interaction. Dev. Biol. 2007, 308, 474–484. [Google Scholar] [CrossRef]
- Zeng, G.Q.; Chen, A.B.; Li, W.; Song, J.H.; Gao, C.Y. High MMP-1, MMP-2, and MMP-9 Protein Levels in Osteoarthritis. Genet. Mol. Res. 2015, 14, 14811–14822. [Google Scholar] [CrossRef]
- Suh, H.N.; Han, H.J. Collagen I Regulates the Self-Renewal of Mouse Embryonic Stem Cells through A2β1 Integrin- and DDR1-Dependent Bmi-1. J. Cell. Physiol. 2011, 226, 3422–3432. [Google Scholar] [CrossRef]
- Li, Y.Y.; Choy, T.H.; Ho, F.C.; Chan, P.B. Scaffold Composition Affects Cytoskeleton Organization, Cell-Matrix Interaction and the Cellular Fate of Human Mesenchymal Stem Cells upon Chondrogenic Differentiation. Biomaterials 2015, 52, 208–220. [Google Scholar] [CrossRef]
- Zhang, T.; Wen, F.; Wu, Y.; Goh, G.S.H.; Ge, Z.; Tan, L.P.; Hui, J.H.P.; Yang, Z. Cross-Talk between TGF-Beta/SMAD and Integrin Signaling Pathways in Regulating Hypertrophy of Mesenchymal Stem Cell Chondrogenesis under Deferral Dynamic Compression. Biomaterials 2015, 38, 72–85. [Google Scholar] [CrossRef]
- Todorovic, V.; Jurukovski, V.; Chen, Y.; Fontana, L.; Dabovic, B.; Rifkin, D.B. Latent TGF-β Binding Proteins. Int. J. Biochem. Cell Biol. 2005, 37, 38–41. [Google Scholar] [CrossRef]
- Zhu, Y.; Oganesian, A.; Keene, D.R.; Sandell, L.J. Type IIA Procollagen Containing the Cysteine-Rich Amino Propeptide Is Deposited in the Extracellular Matrix of Prechondrogenic Tissue and Binds to TGF-Β1 and BMP-2. J. Cell Biol. 1999, 144, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Abreu, J.G.; Ketpura, N.I.; Reversade, B.; De Robertis, E.M. Connective-Tissue Growth Factor (Ctgf) Modulates Cell Signalling by Bmp and TGF-β. Nat. Cell Biol. 2002, 4, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Olsen, S.K.; Goetz, R. A Protein Canyon in the FGF-FGF Receptor Dimer Selects from an à La Carte Menu of Heparan Sulfate Motifs. Curr. Opin. Struct. Biol. 2005, 15, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Hydrogels for Tissue Engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Banerjee, R. Biopolymer-Based Hydrogels for Cartilage Tissue Engineering. Chem. Rev. 2011, 111, 4453–4474. [Google Scholar] [CrossRef] [PubMed]
- Pierschbacher, M.D.; Ruoslahti, E. Cell Attachment Activity of Fibronectin Can Be Duplicated by Small Synthetic Fragments of the Molecule. Nature 1984, 309, 30–33. [Google Scholar] [CrossRef]
- Wu, S.C.; Chen, C.H.; Chang, J.K.; Fu, Y.C.; Wang, C.K.; Eswaramoorthy, R.; Lin, Y.S.; Wang, Y.H.; Lin, S.Y.; Wang, G.J.; et al. Hyaluronan Initiates Chondrogenesis Mainly via CD44 in Human Adipose-Derived Stem Cells. J. Appl. Physiol. 2013, 114, 1610–1618. [Google Scholar] [CrossRef]
- Bian, L.; Guvendiren, M.; Mauck, R.L.; Burdick, J.A. Hydrogels That Mimic Developmentally Relevant Matrix and N-Cadherin Interactions Enhance MSC Chondrogenesis. Proc. Natl. Acad. Sci. USA 2013, 110, 10117–10122. [Google Scholar] [CrossRef]
- Burdick, J.A.; Chung, C. Influence of Three-Dimensional Hyaluronic Acid Microenvironments on Mesenchymal Stem Cell Chondrogenesis. Tissue Eng. Part A 2009, 15, 243–254. [Google Scholar] [CrossRef]
- Wagenbrenner, M.; Mayer-Wagner, S.; Rudert, M.; Holzapfel, B.M.; Weissenberger, M. Combinations of Hydrogels and Mesenchymal Stromal Cells (MSCs) for Cartilage Tissue Engineering—A Review of the Literature. Gels 2021, 7, 217. [Google Scholar] [CrossRef]
- Lee, K.B.L.; Hui, J.H.P.; Song, I.C.; Ardany, L.; Lee, E.H. Injectable Mesenchymal Stem Cell Therapy for Large Cartilage Defects—A Porcine Model. Stem Cells 2007, 25, 2964–2971. [Google Scholar] [CrossRef] [PubMed]
- Saw, K.Y.; Hussin, P.; Loke, S.C.; Azam, M.; Chen, H.C.; Tay, Y.G.; Low, S.; Wallin, K.L.; Ragavanaidu, K. Articular Cartilage Regeneration with Autologous Marrow Aspirate and Hyaluronic Acid: An Experimental Study in a Goat Model. Arthrosc.-J. Arthrosc. Relat. Surg. 2009, 25, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Chiang, E.-R.; Ma, H.-L.; Wang, J.-P.; Liu, C.-L.; Chen, T.-H.; Hung, S.-C. Allogeneic Mesenchymal Stem Cells in Combination with Hyaluronic Acid for the Treatment of Osteoarthritis in Rabbits. PLoS ONE 2016, 11, e0149835. [Google Scholar] [CrossRef] [PubMed]
- Mokbel, A.N.; El Tookhy, O.S.; Shamaa, A.A.; Rashed, L.A.; Sabry, D.; El Sayed, A.M. Homing and Reparative Effect of Intra-Articular Injection of Autologus Mesenchymal Stem Cells in Osteoarthritic Animal Model. BMC Musculoskelet. Disord. 2011, 12, 259. [Google Scholar] [CrossRef]
- Sato, M.; Uchida, K.; Nakajima, H.; Miyazaki, T.; Guerrero, A.; Watanabe, S.; Roberts, S.; Baba, H. Direct Transplantation of Mesenchymal Stem Cells into the Knee Joints of Hartley Strain Guinea Pigs with Spontaneous Osteoarthritis. Arthritis Res. Ther. 2012, 14, R31. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.B.L.; Wang, V.T.Z.; Chan, Y.H.; Hui, J.H.P. A Novel, Minimally-Invasive Technique of Cartilage Repair in the Human Knee Using Arthroscopic Microfracture and Injections of Mesenchymal Stem Cells and Hyaluronic Acid—A Prospective Comparative Study on Safety and Short-Term Efficacy. Ann. Acad. Med. 2012, 41, 511–517. [Google Scholar]
- Lv, X.; He, J.; Zhang, X.; Luo, X.; He, N.; Sun, Z.; Xia, H.; Liu, V.; Zhang, L.; Lin, X.; et al. Comparative Efficacy of Autologous Stromal Vascular Fraction and Autologous Adipose-Derived Mesenchymal Stem Cells Combined with Hyaluronic Acid for the Treatment of Sheep Osteoarthritis. Cell Transplant. 2018, 27, 1111–1125. [Google Scholar] [CrossRef]
- Feng, C.; Luo, X.; He, N.; Xia, H.; Lv, X.; Zhang, X.; Li, D.; Wang, F.; He, J.; Zhang, L.; et al. Efficacy and Persistence of Allogeneic Adipose-Derived Mesenchymal Stem Cells Combined with Hyaluronic Acid in Osteoarthritis After Intra-Articular Injection in a Sheep Model. Tissue Eng. Part A 2018, 24, 219–233. [Google Scholar] [CrossRef]
- Brand, D.D.; Latham, K.A.; Rosloniec, E.F. Collagen-Induced Arthritis. Nat. Protoc. 2007, 2, 1269–1275. [Google Scholar] [CrossRef]
- Chun, S.Y.; Kim, H.T.; Kwon, S.Y.; Kim, J.; Kim, B.S.; Yoo, E.S.; Kwon, T.G. The Efficacy and Safety of Collagen-I and Hypoxic Conditions in Urine-Derived Stem Cell Ex Vivo Culture. Tissue Eng. Regen. Med. 2016, 13, 403–415. [Google Scholar] [CrossRef]
- Nöth, U.; Rackwitz, L.; Heymer, A.; Weber, M.; Baumann, B.; Steinert, A.; Schütze, N.; Jakob, F.; Eulert, J. Chondrogenic Differentiation of Human Mesenchymal Stem Cells in Collagen Type I Hydrogels. J. Biomed. Mater. Res. Part A 2007, 83, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Rutgers, M.; Saris, D.B.; Vonk, L.A.; Van Rijen, M.H.; Akrum, V.; Langeveld, D.; Van Boxtel, A.; Dhert, W.J.; Creemers, L.B. Effect of Collagen Type I or Type II on Chondrogenesis by Cultured Human Articular Chondrocytes. Tissue Eng. Part A 2013, 19, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Sevastianov, V.I.; Basok, Y.B.; Kirsanova, L.A.; Grigoriev, A.M.; Kirillova, A.D.; Nemets, E.A.; Subbot, A.M.; Gautier, S.V. A Comparison of the Capacity of Mesenchymal Stromal Cells for Cartilage Regeneration Depending on Collagen-Based Injectable Biomimetic Scaffold Type. Life 2021, 11, 756. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.; Kim, S.; Lin, B.; Wu, B.M.; Lee, M. Cartilaginous Extracellular Matrix-Modified Chitosan Hydrogels for Cartilage Tissue Engineering. ACS Appl. Mater. Interfaces 2014, 6, 20110–20121. [Google Scholar] [CrossRef] [PubMed]
- Salamon, A.; van Vlierberghe, S.; van Nieuwenhove, I.; Baudisch, F.; Graulus, G.J.; Benecke, V.; Alberti, K.; Neumann, H.G.; Rychly, J.; Martins, J.C.; et al. Gelatin-Based Hydrogels Promote Chondrogenic Differentiation of Human Adipose Tissue-Derived Mesenchymal Stem Cells in Vitro. Materials 2014, 7, 1342–1359. [Google Scholar] [CrossRef]
- Awad, H.A.; Wickham, M.Q.; Leddy, H.A.; Gimble, J.M.; Guilak, F. Chondrogenic Differentiation of Adipose-Derived Adult Stem Cells in Agarose, Alginate, and Gelatin Scaffolds. Biomaterials 2004, 25, 3211–3222. [Google Scholar] [CrossRef]
- Choi, S.M.; Lee, K.-M.; Ryu, S.B.; Park, Y.J.; Hwang, Y.G.; Baek, D.; Choi, Y.; Park, K.H.; Park, K.D.; Lee, J.W. Enhanced Articular Cartilage Regeneration with SIRT1-Activated MSCs Using Gelatin-Based Hydrogel. Cell Death Dis. 2018, 9, 866. [Google Scholar] [CrossRef]
- Wei, Y.; Hu, Y.; Hao, W.; Han, Y.; Meng, G.; Zhang, D.; Wu, Z.; Wang, H. A Novel Injectable Scaffold for Cartilage Tissue Engineering Using Adipose-Derived Adult Stem Cells. J. Orthop. Res. 2008, 26, 27–33. [Google Scholar] [CrossRef]
- Varghese, S.; Hwang, N.S.; Canver, A.C.; Theprungsirikul, P.; Lin, D.W.; Elisseeff, J. Chondroitin Sulfate Based Niches for Chondrogenic Differentiation of Mesenchymal Stem Cells. Matrix Biol. 2008, 27, 12–21. [Google Scholar] [CrossRef]
- Petrovova, E.; Tomco, M.; Holovska, K.; Danko, J.; Kresakova, L.; Vdoviakova, K.; Simaiova, V.; Kolvek, F.; Hornakova, P.; Toth, T.; et al. PHB/CHIT Scaffold as a Promising Biopolymer in the Treatment of Osteochondral Defects—An Experimental Animal Study. Polymers 2021, 13, 1232. [Google Scholar] [CrossRef]
- Naderi-Meshkin, H.; Andreas, K.; Matin, M.M.; Sittinger, M.; Bidkhori, H.R.; Ahmadiankia, N.; Bahrami, A.R.; Ringe, J. Chitosan-Based Injectable Hydrogel as a Promising in Situ Forming Scaffold for Cartilage Tissue Engineering. Cell Biol. Int. 2014, 38, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Ragetly, G.; Griffon, D.J.; Chung, Y.S. The Effect of Type II Collagen Coating of Chitosan Fibrous Scaffolds on Mesenchymal Stem Cell Adhesion and Chondrogenesis. Acta Biomater. 2010, 6, 3988–3997. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Zhu, F.; Li, X.; Liang, Q.; Zhuo, Z.; Huang, J.; Duan, L.; Xiong, J.; Wang, D. Repair of Osteochondral Defects Using Injectable Chitosan-Based Hydrogel Encapsulated Synovial Fluid-Derived Mesenchymal Stem Cells in a Rabbit Model. Mater. Sci. Eng. C 2019, 99, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Hersel, U.; Dahmen, C.; Kessler, H. RGD Modified Polymers: Biomaterials for Stimulated Cell Adhesion and Beyond. Biomaterials 2003, 24, 4385–4415. [Google Scholar] [CrossRef]
- Culenova, M.; Birova, I.; Alexy, P.; Galfiova, P.; Nicodemou, A.; Moncmanova, B.; Plavec, R.; Tomanova, K.; Mencik, P.; Ziaran, S.; et al. In Vitro Characterization of Poly(Lactic Acid)/Poly(Hydroxybutyrate)/Thermoplastic Starch Blends for Tissue Engineering Application. Cell Transplant. 2021, 30, 096368972110210. [Google Scholar] [CrossRef] [PubMed]
- Mahboudi, H.; Sadat Hosseini, F.; Kehtari, M.; Hassannia, H.; Enderami, S.E.; Nojehdehi, S. The Effect of PLLA/PVA Nanofibrous Scaffold on the Chondrogenesis of Human Induced Pluripotent Stem Cells. Int. J. Polym. Mater. Polym. Biomater. 2020, 69, 669–677. [Google Scholar] [CrossRef]
- Williams, C.G.; Kim, T.K.; Taboas, A.; Malik, A.; Manson, P.; Elisseeff, J. In Vitro Chondrogenesis of Bone Marrow-Derived Mesenchymal Stem Cells in a Photopolymerizing Hydrogel. Tissue Eng. 2003, 9, 679–688. [Google Scholar] [CrossRef]
- Qiong Liu, S.; Tian, Q.; Wang, L.; Hedrick, J.L.; Po Hui, J.H.; Yan Yang, Y.; Ee, P.L.R. Injectable Biodegradable Polyethylene Glycol/RGD Peptide Hybrid Hydrogels for In Vitro Chondrogenesis of Human Mesenchymal Stern Cellsa. Macromol. Rapid Commun. 2010, 31, 1148–1154. [Google Scholar] [CrossRef]
- Hwang, N.S.; Varghese, S.; Li, H.; Elisseeff, J. Regulation of Osteogenic and Chondrogenic Differentiation of Mesenchymal Stem Cells in PEG-ECM Hydrogels. Cell Tissue Res. 2011, 344, 499–509. [Google Scholar] [CrossRef]
- Li, J.; Huang, Y.; Song, J.; Li, X.; Zhang, X.; Zhou, Z.; Chen, D.; Ma, P.X.; Peng, W.; Wang, W.; et al. Cartilage Regeneration Using Arthroscopic Flushing Fluid-Derived Mesenchymal Stem Cells Encapsulated in a One-Step Rapid Cross-Linked Hydrogel. Acta Biomater. 2018, 79, 202–215. [Google Scholar] [CrossRef]
- Liu, H.; Ding, J.; Li, C.; Wang, C.; Wang, Y.; Wang, J.; Chang, F. Hydrogel Is Superior to Fibrin Gel as Matrix of Stem Cells in Alleviating Antigen-Induced Arthritis. Polymers 2016, 8, 182. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; De Bank, P.A.; Shakesheff, K.M. Injectable Scaffolds for Tissue Regeneration. J. Mater. Chem. 2004, 14, 1915–1923. [Google Scholar] [CrossRef]
- Nagahama, K.; Takahashi, A.; Ohya, Y. Biodegradable Polymers Exhibiting Temperature-Responsive Sol-Gel Transition as Injectable Biomedical Materials. React. Funct. Polym. 2013, 73, 979–985. [Google Scholar] [CrossRef]
- Kim, H.K.; Shim, W.S.; Kim, S.E.; Lee, K.H.; Kang, E.; Kim, J.H.; Kim, K.; Kwon, I.C.; Lee, D.S. Injectable in Situ-Forming PH/Thermo-Sensitive Hydrogel for Bone Tissue Engineering. Tissue Eng. Part A 2009, 15, 923–933. [Google Scholar] [CrossRef]
- Park, H.; Woo, E.K.; Lee, K.Y. Ionically Cross-Linkable Hyaluronate-Based Hydrogels for Injectable Cell Delivery. J. Control. Release 2014, 196, 146–153. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, X.; Ma, C.; Yi, H.; Ali, Z.; Mou, X.; Li, S.; Deng, Y.; He, N. Injectable Hydrogels for Cartilage and Bone Tissue Engineering. Bone Res. 2017, 5, 17014. [Google Scholar] [CrossRef]
- Lin, H.; Cheng, A.W.M.; Alexander, P.G.; Beck, A.M.; Tuan, R.S. Cartilage Tissue Engineering Application of Injectable Gelatin Hydrogel with In Situ Visible-Light-Activated Gelation Capability in Both Air and Aqueous Solution. Tissue Eng. Part A 2014, 20, 2402–2411. [Google Scholar] [CrossRef]
- Levinson, C.; Lee, M.; Applegate, L.A.; Zenobi-Wong, M. An Injectable Heparin-Conjugated Hyaluronan Scaffold for Local Delivery of Transforming Growth Factor Β1 Promotes Successful Chondrogenesis. Acta Biomater. 2019, 99, 168–180. [Google Scholar] [CrossRef]
- Jooybar, E.; Abdekhodaie, M.J.; Alvi, M.; Mousavi, A.; Karperien, M.; Dijkstra, P.J. An Injectable Platelet Lysate-Hyaluronic Acid Hydrogel Supports Cellular Activities and Induces Chondrogenesis of Encapsulated Mesenchymal Stem Cells. Acta Biomater. 2019, 83, 233–244. [Google Scholar] [CrossRef]
- Dehghan-Baniani, D.; Chen, Y.; Wang, D.; Bagheri, R.; Solouk, A.; Wu, H. Injectable in Situ Forming Kartogenin-Loaded Chitosan Hydrogel with Tunable Rheological Properties for Cartilage Tissue Engineering. Colloids Surfaces B Biointerfaces 2020, 192, 111059. [Google Scholar] [CrossRef]
- Zhao, Y.; Gao, C.; Liu, H.; Liu, H.; Feng, Y.; Li, Z.; Liu, H.; Wang, J.; Yang, B.; Lin, Q. Infliximab-Based Self-Healing Hydrogel Composite Scaffold Enhances Stem Cell Survival, Engraftment, and Function in Rheumatoid Arthritis Treatment. Acta Biomater. 2021, 121, 653–664. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strecanska, M.; Danisovic, L.; Ziaran, S.; Cehakova, M. The Role of Extracellular Matrix and Hydrogels in Mesenchymal Stem Cell Chondrogenesis and Cartilage Regeneration. Life 2022, 12, 2066. https://doi.org/10.3390/life12122066
Strecanska M, Danisovic L, Ziaran S, Cehakova M. The Role of Extracellular Matrix and Hydrogels in Mesenchymal Stem Cell Chondrogenesis and Cartilage Regeneration. Life. 2022; 12(12):2066. https://doi.org/10.3390/life12122066
Chicago/Turabian StyleStrecanska, Magdalena, Lubos Danisovic, Stanislav Ziaran, and Michaela Cehakova. 2022. "The Role of Extracellular Matrix and Hydrogels in Mesenchymal Stem Cell Chondrogenesis and Cartilage Regeneration" Life 12, no. 12: 2066. https://doi.org/10.3390/life12122066
APA StyleStrecanska, M., Danisovic, L., Ziaran, S., & Cehakova, M. (2022). The Role of Extracellular Matrix and Hydrogels in Mesenchymal Stem Cell Chondrogenesis and Cartilage Regeneration. Life, 12(12), 2066. https://doi.org/10.3390/life12122066







