The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss
Abstract
1. Introduction
2. Hair Structure and Hair Cycle
3. Physiological and Functional Roles of Wnt/β-Catenin Signaling for Improving Hair Growth
3.1. Wnt1a
3.2. Wnt3a
3.3. Wnt4
3.4. Wnt5a
3.5. Wnt10b
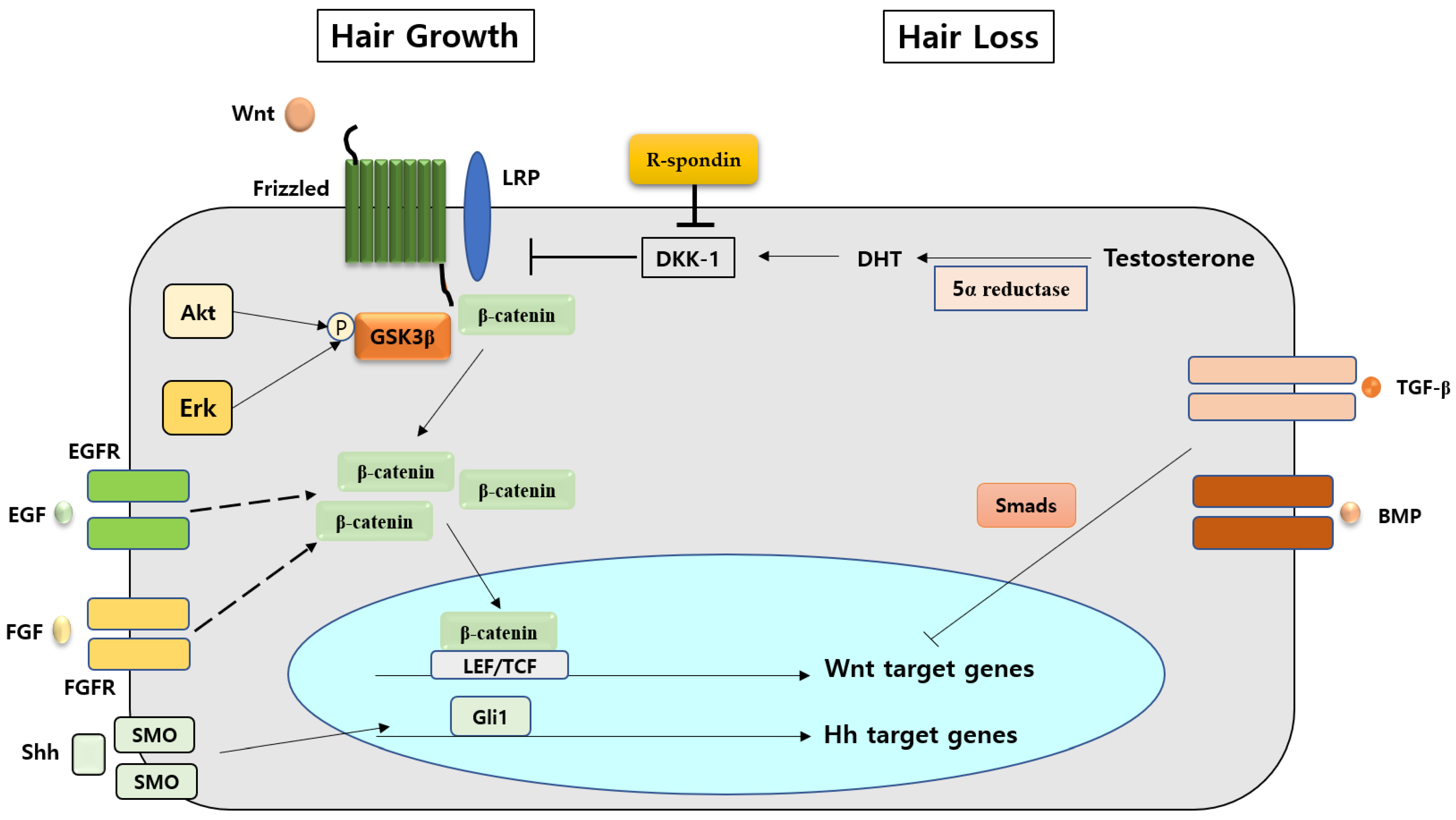
4. Various Factors Interacting with the Wnt/β-Catenin Signaling Pathway
5. Natural Products for Treating Hair Loss by Targeting the Wnt/β-Catenin Signaling Pathway
6. Materials and Methods
6.1. Search Strategy
6.2. Selection of Studies
6.3. Data Extraction
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wolff, H.; Fischer, T.W.; Blume-Peytavi, U. The Diagnosis and Treatment of Hair and Scalp Diseases. Dtsch. Arztebl Int. 2016, 113, 377–386. [Google Scholar] [CrossRef]
- York, K.; Meah, N.; Bhoyrul, B.; Sinclair, R. A Review of the Treatment of Male Pattern Hair Loss. Expert Opin. Pharmacother. 2020, 21, 603–612. [Google Scholar] [CrossRef]
- Hossein Mostafa, D.; Samadi, A.; Niknam, S.; Nasrollahi, S.A.; Guishard, A.; Firooz, A.J. Efficacy of Cetirizine 1% Versus Minoxidil 5% Topical Solution in the Treatment of Male Alopecia: A Randomized, Single-blind Controlled Study. Pharm Pharm Sci. 2021, 24, 191–199. [Google Scholar] [CrossRef]
- Rossi, A.; Cantisani, C.; Melis, L.; Iorio, A.; Scali, E.; Calvieri, S. Minoxidil use in Dermatology, Side Effects and Recent Patents. Recent. Pat. Inflamm. Allergy Drug Discov. 2012, 6, 130–136. [Google Scholar]
- Corona, G.; Tirabassi, G.; Santi, D.; Maseroli, E.; Gacci, M.; Dicuio, M.; Sforza, A.; Mannucci, E.; Maggi, M. Sexual Dysfunction in Subjects Treated with Inhibitors of 5alpha-Reductase for Benign Prostatic Hyperplasia: A Comprehensive Review and Meta-Analysis. Andrology 2017, 5, 671–678. [Google Scholar] [CrossRef]
- Kaplan-Marans, E.; Sandozi, A.; Martinez, M.; Lee, J.; Schulman, A.; Khurgin, J. Medications most Commonly Associated with Erectile Dysfunction: Evaluation of the Food and Drug Administration National Pharmacovigilance Database. J. Sex. Med. 2022, 10, 100543. [Google Scholar] [CrossRef]
- Irwig, M.S.; Kolukula, S. Persistent Sexual Side Effects of Finasteride for Male Pattern Hair Loss. J. Sex. Med. 2011, 8, 1747–1753. [Google Scholar] [CrossRef]
- Choi, B.Y. Targeting Wnt/Beta-Catenin Pathway for Developing Therapies for Hair Loss. Int. J. Mol. Sci. 2020, 21, 4915. [Google Scholar] [CrossRef]
- Hu, X.M.; Li, Z.X.; Zhang, D.Y.; Yang, Y.C.; Fu, S.A.; Zhang, Z.Q.; Yang, R.H.; Xiong, K. A Systematic Summary of Survival and Death Signalling during the Life of Hair Follicle Stem Cells. Stem Cell Res. Ther. 2021, 12, 453. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, J.; Burgeson, R.E.; Morgan, B.A. Wnt Signaling Maintains the Hair-Inducing Activity of the Dermal Papilla. Genes Dev. 2000, 14, 1181–1185. [Google Scholar] [CrossRef]
- Enshell-Seijffers, D.; Lindon, C.; Kashiwagi, M.; Morgan, B.A. Beta-Catenin Activity in the Dermal Papilla Regulates Morphogenesis and Regeneration of Hair. Dev. Cell 2010, 18, 633–642. [Google Scholar] [CrossRef]
- Soma, T.; Fujiwara, S.; Shirakata, Y.; Hashimoto, K.; Kishimoto, J. Hair-Inducing Ability of Human Dermal Papilla Cells Cultured Under Wnt/Beta-Catenin Signalling Activation. Exp. Dermatol. 2012, 21, 307–309. [Google Scholar] [CrossRef]
- Park, S.; Lee, J. Modulation of Hair Growth Promoting Effect by Natural Products. Pharmaceutics 2021, 13, 2163. [Google Scholar] [CrossRef]
- Bassino, E.; Gasparri, F.; Munaron, L. Protective Role of Nutritional Plants Containing Flavonoids in Hair Follicle Disruption: A Review. Int. J. Mol. Sci. 2020, 21, 523. [Google Scholar] [CrossRef]
- Shen, Y.L.; Li, X.Q.; Pan, R.R.; Yue, W.; Zhang, L.J.; Zhang, H. Medicinal Plants for the Treatment of Hair Loss and the Suggested Mechanisms. Curr. Pharm. Des. 2018, 24, 3090–3100. [Google Scholar] [CrossRef]
- Botchkarev, V.A.; Kishimoto, J. Molecular Control of Epithelial-Mesenchymal Interactions during Hair Follicle Cycling. J. Investig. Dermatol. Symp. Proc. 2003, 8, 46–55. [Google Scholar] [CrossRef]
- Houschyar, K.S.; Borrelli, M.R.; Tapking, C.; Popp, D.; Puladi, B.; Ooms, M.; Chelliah, M.P.; Rein, S.; Pforringer, D.; Thor, D.; et al. Molecular Mechanisms of Hair Growth and Regeneration: Current Understanding and Novel Paradigms. Dermatology 2020, 236, 271–280. [Google Scholar] [CrossRef]
- Lee, J.; Bscke, R.; Tang, P.C.; Hartman, B.H.; Heller, S.; Koehler, K.R. Hair Follicle Development in Mouse Pluripotent Stem Cell-Derived Skin Organoids. Cell Rep. 2018, 22, 242–254. [Google Scholar] [CrossRef]
- Llamas-Molina, J.M.; Carrero-Castano, A.; Ruiz-Villaverde, R.; Campos, A. Tissue Engineering and Regeneration of the Human Hair Follicle in Androgenetic Alopecia: Literature Review. Life 2022, 12, 117. [Google Scholar] [CrossRef]
- Madaan, A.; Verma, R.; Singh, A.T.; Jaggi, M. Review of Hair Follicle Dermal Papilla Cells as in Vitro Screening Model for Hair Growth. Int. J. Cosmet. Sci. 2018, 40, 429–450. [Google Scholar] [CrossRef]
- Taghiabadi, E.; Nilforoushzadeh, M.A.; Aghdami, N. Maintaining Hair Inductivity in Human Dermal Papilla Cells: A Review of Effective Methods. Skin Pharmacol. Physiol. 2020, 33, 280–292. [Google Scholar] [CrossRef]
- Lee, S.A.; Li, K.N.; Tumbar, T. Stem Cell-Intrinsic Mechanisms Regulating Adult Hair Follicle Homeostasis. Exp. Dermatol. 2021, 30, 430–447. [Google Scholar] [CrossRef]
- Ji, S.; Zhu, Z.; Sun, X.; Fu, X. Functional Hair Follicle Regeneration: An Updated Review. Signal. Transduct Target Ther. 2021, 6, 66. [Google Scholar] [CrossRef]
- Park, S. Hair Follicle Morphogenesis during Embryogenesis, Neogenesis, and Organogenesis. Front. Cell Dev. Biol. 2022, 10, 933370. [Google Scholar] [CrossRef]
- Lin, X.; Zhu, L.; He, J. Morphogenesis, Growth Cycle and Molecular Regulation of Hair Follicles. Front. Cell Dev. Biol. 2022, 10, 899095. [Google Scholar] [CrossRef]
- Veltri, A.; Lang, C.; Lien, W.H. Concise Review: Wnt Signaling Pathways in Skin Development and Epidermal Stem Cells. Stem Cells 2018, 36, 22–35. [Google Scholar] [CrossRef]
- Huelsken, J.; Vogel, R.; Erdmann, B.; Cotsarelis, G.; Birchmeier, W. Beta-Catenin Controls Hair Follicle Morphogenesis and Stem Cell Differentiation in the Skin. Cell 2001, 105, 533–545. [Google Scholar] [CrossRef]
- Leiros, G.J.; Attorresi, A.I.; Balana, M.E. Hair Follicle Stem Cell Differentiation is Inhibited through Cross-Talk between Wnt/Beta-Catenin and Androgen Signalling in Dermal Papilla Cells from Patients with Androgenetic Alopecia. Br. J. Dermatol. 2012, 166, 1035–1042. [Google Scholar] [CrossRef]
- Doumpas, N.; Lampart, F.; Robinson, M.D.; Lentini, A.; Nestor, C.E.; Cantu, C.; Basler, K. TCF/LEF Dependent and Independent Transcriptional Regulation of Wnt/Beta-Catenin Target Genes. EMBO J. 2019, 38, e98873. [Google Scholar] [CrossRef]
- Bafico, A.; Liu, G.; Yaniv, A.; Gazit, A.; Aaronson, S.A. Novel Mechanism of Wnt Signalling Inhibition Mediated by Dickkopf-1 Interaction with LRP6/Arrow. Nat. Cell Biol. 2001, 3, 683–686. [Google Scholar] [CrossRef]
- Mao, B.; Wu, W.; Li, Y.; Hoppe, D.; Stannek, P.; Glinka, A.; Niehrs, C. LDL-Receptor-Related Protein 6 is a Receptor for Dickkopf Proteins. Nature 2001, 411, 321–325. [Google Scholar] [CrossRef]
- Kwack, M.H.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Dickkopf 1 Promotes Regression of Hair Follicles. J. Investig. Dermatol. 2012, 132, 1554–1560. [Google Scholar] [CrossRef]
- Mahmoud, E.A.; Elgarhy, L.H.; Hasby, E.A.; Mohammad, L. Dickkopf-1 Expression in Androgenetic Alopecia and Alopecia Areata in Male Patients. Am. J. Dermatopathol. 2019, 41, 122–127. [Google Scholar] [CrossRef]
- Huang, S.; Zhu, X.; Tao, Y.; Sun, Q.; Wang, L.; Li, B.; He, L.; Guo, X.; Ma, G. Involvement of Epithelial Wntless in the Regulation of Postnatal Hair Follicle Morphogenesis. Arch. Dermatol. Res. 2015, 307, 835–839. [Google Scholar] [CrossRef]
- Myung, P.S.; Takeo, M.; Ito, M.; Atit, R.P. Epithelial Wnt Ligand Secretion is Required for Adult Hair Follicle Growth and Regeneration. J. Investig. Dermatol. 2013, 133, 31–41. [Google Scholar] [CrossRef]
- Huang, S.; Zhu, X.; Liu, Y.; Tao, Y.; Feng, G.; He, L.; Guo, X.; Ma, G. Wls is Expressed in the Epidermis and Regulates Embryonic Hair Follicle Induction in Mice. PLoS ONE 2012, 7, e45904. [Google Scholar] [CrossRef]
- Dong, L.; Hao, H.; Xia, L.; Liu, J.; Ti, D.; Tong, C.; Hou, Q.; Han, Q.; Zhao, Y.; Liu, H.; et al. Treatment of MSCs with Wnt1a-Conditioned Medium Activates DP Cells and Promotes Hair Follicle Regrowth. Sci. Rep. 2014, 4, 5432. [Google Scholar] [CrossRef]
- Stoffel, W.; Schmidt-Soltau, I.; Jenke, B.; Binczek, E.; Hammels, I. Hair Growth Cycle is Arrested in SCD1 Deficiency by Impaired Wnt3a-Palmitoleoylation and Retrieved by the Artificial Lipid Barrier. J. Investig. Dermatol. 2017, 137, 1424–1433. [Google Scholar] [CrossRef]
- Rajendran, R.L.; Gangadaran, P.; Seo, C.H.; Kwack, M.H.; Oh, J.M.; Lee, H.W.; Gopal, A.; Sung, Y.K.; Jeong, S.Y.; Lee, S.W.; et al. Macrophage-Derived Extracellular Vesicle Promotes Hair Growth. Cells 2020, 9, 856. [Google Scholar] [CrossRef]
- Park, J.M.; Jun, M.S.; Kim, J.A.; Mali, N.M.; Hsi, T.C.; Cho, A.; Kim, J.C.; Kim, J.Y.; Seo, I.; Kim, J.; et al. Restoration of Immune Privilege in Human Dermal Papillae Controlling Epithelial-Mesenchymal Interactions in Hair Formation. Tissue Eng. Regen. Med. 2022, 19, 105–116. [Google Scholar] [CrossRef]
- Hawkshaw, N.J.; Hardman, J.A.; Alam, M.; Jimenez, F.; Paus, R. Deciphering the Molecular Morphology of the Human Hair Cycle: Wnt Signalling during the Telogen-Anagen Transformation. Br. J. Dermatol. 2020, 182, 1184–1193. [Google Scholar] [CrossRef]
- Xing, Y.Z.; Wang, R.M.; Yang, K.; Guo, H.Y.; Deng, F.; Li, Y.H.; Ye, J.X.; He, L.; Lian, X.H.; Yang, T. Adenovirus-Mediated Wnt5a Expression Inhibits the Telogen-to-Anagen Transition of Hair Follicles in Mice. Int. J. Med. Sci. 2013, 10, 908–914. [Google Scholar] [CrossRef][Green Version]
- Xing, Y.; Ma, X.; Guo, H.; Deng, F.; Yang, J.; Li, Y. Wnt5a Suppresses Beta-Catenin Signaling during Hair Follicle Regeneration. Int. J. Med. Sci. 2016, 13, 603–610. [Google Scholar] [CrossRef]
- Tiwari, R.L.; Mishra, P.; Martin, N.; George, N.O.; Sakk, V.; Soller, K.; Nalapareddy, K.; Nattamai, K.; Scharffetter-Kochanek, K.; Florian, M.C.; et al. A Wnt5a-Cdc42 Axis Controls Aging and Rejuvenation of Hair-Follicle Stem Cells. Aging 2021, 13, 4778–4793. [Google Scholar] [CrossRef]
- Ouji, Y.; Yoshikawa, M.; Moriya, K.; Ishizaka, S. Effects of Wnt-10b on Hair Shaft Growth in Hair Follicle Cultures. Biochem. Biophys. Res. Commun. 2007, 359, 516–522. [Google Scholar] [CrossRef]
- Li, Y.H.; Zhang, K.; Yang, K.; Ye, J.X.; Xing, Y.Z.; Guo, H.Y.; Deng, F.; Lian, X.H.; Yang, T. Adenovirus-Mediated Wnt10b Overexpression Induces Hair Follicle Regeneration. J. Investig. Dermatol. 2013, 133, 42–48. [Google Scholar] [CrossRef]
- Zhang, H.; Nan, W.; Wang, S.; Zhang, T.; Si, H.; Yang, F.; Li, G. Epidermal Growth Factor Promotes Proliferation and Migration of Follicular Outer Root Sheath Cells Via Wnt/Beta-Catenin Signaling. Cell. Physiol. Biochem. 2016, 39, 360–370. [Google Scholar] [CrossRef]
- Xing, Y.; Xu, W.; Yang, K.; Lian, X.; Yang, T. Immunolocalization of Wnt5a during the Hair Cycle and its Role in Hair Shaft Growth in Mice. Acta Histochem. 2011, 113, 608–612. [Google Scholar] [CrossRef]
- Leiros, G.J.; Ceruti, J.M.; Castellanos, M.L.; Kusinsky, A.G.; Balana, M.E. Androgens Modify Wnt Agonists/Antagonists Expression Balance in Dermal Papilla Cells Preventing Hair Follicle Stem Cell Differentiation in Androgenetic Alopecia. Mol. Cell. Endocrinol. 2017, 439, 26–34. [Google Scholar] [CrossRef]
- Wu, P.; Zhang, Y.; Xing, Y.; Xu, W.; Guo, H.; Deng, F.; Ma, X.; Li, Y. The Balance of Bmp6 and Wnt10b Regulates the Telogen-Anagen Transition of Hair Follicles. Cell Commun. Signal. 2019, 17, 16. [Google Scholar] [CrossRef]
- Kitagawa, T.; Matsuda, K.; Inui, S.; Takenaka, H.; Katoh, N.; Itami, S.; Kishimoto, S.; Kawata, M. Keratinocyte Growth Inhibition through the Modification of Wnt Signaling by Androgen in Balding Dermal Papilla Cells. J. Clin. Endocrinol. Metab. 2009, 94, 1288–1294. [Google Scholar] [CrossRef]
- Lin, W.H.; Xiang, L.J.; Shi, H.X.; Zhang, J.; Jiang, L.P.; Cai, P.T.; Lin, Z.L.; Lin, B.B.; Huang, Y.; Zhang, H.L.; et al. Fibroblast Growth Factors Stimulate Hair Growth through Beta-Catenin and Shh Expression in C57BL/6 Mice. Biomed. Res. Int. 2015, 2015, 730139. [Google Scholar]
- Liu, S.; Leask, A. CCN2 Modulates Hair Follicle Cycling in Mice. Mol. Biol. Cell 2013, 24, 3939–3944. [Google Scholar] [CrossRef]
- Zhang, J.; He, X.C.; Tong, W.G.; Johnson, T.; Wiedemann, L.M.; Mishina, Y.; Feng, J.Q.; Li, L. Bone Morphogenetic Protein Signaling Inhibits Hair Follicle Anagen Induction by Restricting Epithelial Stem/Progenitor Cell Activation and Expansion. Stem Cells 2006, 24, 2826–2839. [Google Scholar] [CrossRef]
- Tripurani, S.K.; Wang, Y.; Fan, Y.X.; Rahimi, M.; Wong, L.; Lee, M.H.; Starost, M.F.; Rubin, J.S.; Johnson, G.R. Suppression of Wnt/Beta-Catenin Signaling by EGF Receptor is Required for Hair Follicle Development. Mol. Biol. Cell 2018, 29, 2784–2799. [Google Scholar] [CrossRef]
- Zhang, H.; Nan, W.; Wang, S.; Si, H.; Li, G. Balance between Fibroblast Growth Factor 10 and Secreted Frizzled-Relate Protein-1 Controls the Development of Hair Follicle by Competitively Regulating Beta-Catenin Signaling. Biomed. Pharmacother. 2018, 103, 1531–1537. [Google Scholar] [CrossRef]
- Suzuki, K.; Yamaguchi, Y.; Villacorte, M.; Mihara, K.; Akiyama, M.; Shimizu, H.; Taketo, M.M.; Nakagata, N.; Tsukiyama, T.; Yamaguchi, T.P.; et al. Embryonic Hair Follicle Fate Change by Augmented Beta-Catenin through Shh and Bmp Signaling. Development 2009, 136, 367–372. [Google Scholar] [CrossRef]
- Silva-Vargas, V.; Lo Celso, C.; Giangreco, A.; Ofstad, T.; Prowse, D.M.; Braun, K.M.; Watt, F.M. Beta-Catenin and Hedgehog Signal Strength can Specify Number and Location of Hair Follicles in Adult Epidermis without Recruitment of Bulge Stem Cells. Dev. Cell 2005, 9, 121–131. [Google Scholar] [CrossRef]
- Premanand, A.; Reena Rajkumari, B. Androgen Modulation of Wnt/Beta-Catenin Signaling in Androgenetic Alopecia. Arch. Dermatol. Res. 2018, 310, 391–399. [Google Scholar] [CrossRef]
- Park, P.J.; Moon, B.S.; Lee, S.H.; Kim, S.N.; Kim, A.R.; Kim, H.J.; Park, W.S.; Choi, K.Y.; Cho, E.G.; Lee, T.R. Hair Growth-Promoting Effect of Aconiti ciliare Tuber Extract Mediated by the Activation of Wnt/Beta-Catenin Signaling. Life Sci. 2012, 91, 935–943. [Google Scholar] [CrossRef]
- Kim, B.H.; Lee, M.J.; Lee, W.Y.; Pyo, J.; Shin, M.S.; Hwang, G.S.; Shin, D.; Kim, C.E.; Park, E.S.; Kang, K.S. Hair Growth Stimulation Effect of Centipeda minima Extract: Identification of Active Compounds and Anagen-Activating Signaling Pathways. Biomolecules 2021, 11, 976. [Google Scholar] [CrossRef]
- Mohanta, T.K.; Tamboli, Y.; Zubaidha, P.K. Phytochemical and Medicinal Importance of Ginkgo biloba L. Nat. Prod. Res. 2014, 28, 746–752. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, Q.; Nan, W.; Wang, Y.; Wang, S.; Yang, F.; Li, G. Ginkgolide B and Bilobalide Promote the Growth and Increase Beta-Catenin Expression in Hair Follicle Dermal Papilla Cells of American Minks. Biofactors 2019, 45, 950–958. [Google Scholar] [CrossRef]
- Li, Y.; Sheng, Y.; Liu, J.; Xu, G.; Yu, W.; Cui, Q.; Lu, X.; Du, P.; An, L. Hair-Growth Promoting Effect and Anti-Inflammatory Mechanism of Ginkgo biloba Polysaccharides. Carbohydr. Polym. 2022, 278, 118811. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.Y.; Choi, E.J.; Kim, J.A.; Hwang, Y.L.; Kim, C.D.; Lee, M.H.; Roh, S.S.; Kim, Y.H.; Han, I.; Kang, S. Malva verticillata Seed Extracts Upregulate the Wnt Pathway in Human Dermal Papilla Cells. Int. J. Cosmet. Sci. 2016, 38, 148–154. [Google Scholar] [CrossRef]
- Ryu, H.S.; Jeong, J.; Lee, C.M.; Lee, K.S.; Lee, J.N.; Park, S.M.; Lee, Y.M. Activation of Hair Cell Growth Factors by Linoleic Acid in Malva verticillata Seed. Molecules 2021, 26, 2117. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.; Ho, R.; Greff, S.; Filaire, E.; Ranouille, E.; Chazaud, C.; Herbette, G.; Butaud, J.F.; Berthon, J.Y.; Raharivelomanana, P. Hair Growth Activity of Three Plants of the Polynesian Cosmetopoeia and their Regulatory Effect on Dermal Papilla Cells. Molecules 2020, 25, 4360. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tang, G.; Li, X.; Sun, W.; Liang, Y.; Gan, D.; Liu, G.; Song, W.; Wang, Z. Study on the Chemical Constituents of Nut Oil from Prunus mira Koehne and the Mechanism of Promoting Hair Growth. J. Ethnopharmacol. 2020, 258, 112831. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, J.; Chen, W.; Li, X.; Fu, K.; Sun, W.; Liang, Y.; Xu, M.; Zhang, J.; Fan, G.; et al. Identification of Hair Growth Promoting Components in the Kernels of Prunus mira Koehne and their Mechanism of Action. Molecules 2022, 27, 5242. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.Y.; Choi, Y.H.; Kim, J.; Park, S.Y.; Nam, Y.J.; Lee, S.Y.; Jeon, J.H.; Jin, M.H.; Lee, S. Polygonum multiflorum Extract Support Hair Growth by Elongating Anagen Phase and Abrogating the Effect of Androgen in Cultured Human Dermal Papilla Cells. BMC Complement. Med. Ther. 2020, 20, 144–145. [Google Scholar] [CrossRef] [PubMed]
- Truong, V.L.; Bak, M.J.; Lee, C.; Jun, M.; Jeong, W.S. Hair Regenerative Mechanisms of Red Ginseng Oil and its Major Components in the Testosterone-Induced Delay of Anagen Entry in C57BL/6 Mice. Molecules 2017, 22, 1505. [Google Scholar] [CrossRef]
- Jin, G.R.; Zhang, Y.L.; Yap, J.; Boisvert, W.A.; Lee, B.H. Hair Growth Potential of Salvia plebeia Extract and its Associated Mechanisms. Pharm. Biol. 2020, 58, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Ruksiriwanich, W.; Khantham, C.; Muangsanguan, A.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R.; Sringarm, K.; Ferrer, E.; et al. Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract. Plants 2022, 11, 1499. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.N.; Park, D.K.; Park, H.J. Hair Growth-Promoting Activity of Hot Water Extract of Thuja orientalis. BMC Complement. Altern. Med. 2013, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Kawai, Y.; Masutani, T.; Tanaka, K.; Ito, K.; Iddamalgoda, A. Effects of Watercress Extract Fraction on R-Spondin 1-Mediated Growth of Human Hair. Int. J. Cosmet. Sci. 2022, 44, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Chen, J.; Zhang, Y.; Wang, S.; Zhong, W.; Yuan, H.; Wu, X.; Wang, C.; Zheng, Y.; Wei, Y.; et al. Alpinetin Promotes Hair Regeneration Via Activating Hair Follicle Stem Cells. Chin. Med. 2022, 17, 63. [Google Scholar] [CrossRef] [PubMed]
- Xing, F.; Yi, W.J.; Miao, F.; Su, M.Y.; Lei, T.C. Baicalin Increases Hair Follicle Development by Increasing Canonical Wnt/Betacatenin Signaling and Activating Dermal Papillar Cells in Mice. Int. J. Mol. Med. 2018, 41, 2079–2085. [Google Scholar] [PubMed]
- Shin, S.H.; Bak, S.S.; Kim, M.K.; Sung, Y.K.; Kim, J.C. Baicalin, a Flavonoid, Affects the Activity of Human Dermal Papilla Cells and Promotes Anagen Induction in Mice. Naunyn Schmiedebergs Arch. Pharmacol. 2015, 388, 583–586. [Google Scholar] [CrossRef]
- Zeng, Q.; Wang, Z.; Zhu, Z.; Hu, Y.; Wang, Y.; Xue, Y.; Wu, Y.; Guo, Y.; Liang, P.; Chen, H.; et al. Glycyrrhizin Micellar Nanocarriers for Topical Delivery of Baicalin to the Hair Follicles: A Targeted Approach Tailored for Alopecia Treatment. Int. J. Pharm. 2022, 625, 122109. [Google Scholar] [CrossRef]
- Kim, Y.E.; Choi, H.C.; Lee, I.C.; Yuk, D.Y.; Lee, H.; Choi, B.Y. 3-Deoxysappanchalcone Promotes Proliferation of Human Hair Follicle Dermal Papilla Cells and Hair Growth in C57BL/6 Mice by Modulating WNT/Beta-Catenin and STAT Signaling. Biomol. Ther. 2016, 24, 572–580. [Google Scholar] [CrossRef]
- Hiipakka, R.A.; Zhang, H.Z.; Dai, W.; Dai, Q.; Liao, S. Structure-Activity Relationships for Inhibition of Human 5alpha-Reductases by Polyphenols. Biochem. Pharmacol. 2002, 63, 1165–1176. [Google Scholar] [CrossRef]
- Katoulis, A.C.; Liakou, A.I.; Koumaki, D.; Vakirlis, E.; Tsantes, A.G.; Mortaki, D.; Bozi, E.; Ioannides, D. A Randomized, Single-Blinded, Vehicle-Controlled Study of a Topical Active Blend in the Treatment of Androgenetic Alopecia. Dermatol. Ther. 2020, 33, e13734. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.S.; Han, J.H.; Yoo, H.G.; Chung, J.H.; Cho, K.H.; Eun, H.C.; Kim, K.H. Human Hair Growth Enhancement in Vitro by Green Tea Epigallocatechin-3-Gallate (EGCG). Phytomedicine 2007, 14, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Kubo, C.; Ogawa, M.; Uehara, N.; Katakura, Y. Fisetin Promotes Hair Growth by Augmenting TERT Expression. Front. Cell Dev. Biol. 2020, 8, 566617. [Google Scholar] [CrossRef]
- Ogawa, M.; Udono, M.; Teruya, K.; Uehara, N.; Katakura, Y. Exosomes Derived from Fisetin-Treated Keratinocytes Mediate Hair Growth Promotion. Nutrients 2021, 13, 2087. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, S.R.; Choi, Y.H.; Shin, J.Y.; Kim, C.D.; Kang, N.G.; Park, B.C.; Lee, S. Quercitrin Stimulates Hair Growth with Enhanced Expression of Growth Factors Via Activation of MAPK/CREB Signaling Pathway. Molecules 2020, 25, 4004. [Google Scholar] [CrossRef]
- Bejaoui, M.; Villareal, M.O.; Isoda, H. Beta-Catenin-Mediated Hair Growth Induction Effect of 3,4,5-Tri-O-Caffeoylquinic Acid. Aging 2019, 11, 4216–4237. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ni, C.; Huang, Y.; Tang, Y.; Yang, K.; Shi, X.; Zhang, Y.; Li, Z.; Wang, J.; Zhu, Y.; et al. Hair Growth-Promoting Effect of Resveratrol in Mice, Human Hair Follicles and Dermal Papilla Cells. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1805–1814. [Google Scholar] [CrossRef]
- Cheon, H.I.; Bae, S.; Ahn, K.J. Flavonoid Silibinin Increases Hair-Inductive Property Via Akt and Wnt/Beta-Catenin Signaling Activation in 3-Dimensional-Spheroid Cultured Human Dermal Papilla Cells. J. Microbiol. Biotechnol. 2019, 29, 321–329. [Google Scholar] [CrossRef]
- Lim, K.M.; An, S.; Lee, O.K.; Lee, M.J.; Lee, J.P.; Lee, K.S.; Lee, G.T.; Lee, K.K.; Bae, S. Analysis of Changes in microRNA Expression Profiles in Response to the Troxerutin-Mediated Antioxidant Effect in Human Dermal Papilla Cells. Mol. Med. Rep. 2015, 12, 2650–2660. [Google Scholar] [CrossRef]
- Lee, Y.R.; Bae, S.; Kim, J.Y.; Lee, J.; Cho, D.H.; Kim, H.S.; An, I.S.; An, S. Monoterpenoid Loliolide Regulates Hair Follicle Inductivity of Human Dermal Papilla Cells by Activating the Akt/Beta-Catenin Signaling Pathway. J. Microbiol. Biotechnol. 2019, 29, 1830–1840. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.E.; Choi, H.C.; Nam, G.; Choi, B.Y. Costunolide Promotes the Proliferation of Human Hair Follicle Dermal Papilla Cells and Induces Hair Growth in C57BL/6 Mice. J. Cosmet. Dermatol. 2019, 18, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Chen, X.; Yi, H.; Han, L.; Ji, B.; Chen, H.; Deng, W.; Wan, M. Beta-Catenin is Involved in Oleanolic Acid-Dependent Promotion of Proliferation in Human Hair Matrix Cells in an in Vitro Organ Culture Model. Fitoterapia 2017, 121, 136–140. [Google Scholar] [CrossRef]
- Ahmed, N.S.; Ghatak, S.; El Masry, M.S.; Gnyawali, S.C.; Roy, S.; Amer, M.; Everts, H.; Sen, C.K.; Khanna, S. Epidermal E-Cadherin Dependent Beta-Catenin Pathway is Phytochemical Inducible and Accelerates Anagen Hair Cycling. Mol. Ther. 2017, 25, 2502–2512. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Yoon, J.; Shin, S.H.; Zahoor, M.; Kim, H.J.; Park, P.J.; Park, W.S.; Do, S.M.; Kim, H.Y.; Choi, K.Y. Valproic Acid Induces Hair Regeneration in Murine Model and Activates Alkaline Phosphatase Activity in Human Dermal Papilla Cells. PLoS ONE 2012, 7, e34152. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.J.; Choi, S.J.; Yoon, S.Y.; Lee, J.Y.; Park, W.S.; Park, P.J.; Kim, K.H.; Eun, H.C.; Kwon, O. Valproic Acid Promotes Human Hair Growth in in Vitro Culture Model. J. Dermatol. Sci. 2013, 72, 16–24. [Google Scholar] [CrossRef]
- Jo, S.J.; Shin, H.; Park, Y.W.; Paik, S.H.; Park, W.S.; Jeong, Y.S.; Shin, H.J.; Kwon, O. Topical Valproic Acid Increases the Hair Count in Male Patients with Androgenetic Alopecia: A Randomized, Comparative, Clinical Feasibility Study using Phototrichogram Analysis. J. Dermatol. 2014, 41, 285–291. [Google Scholar] [CrossRef]
- Li, S.; Chen, J.; Chen, F.; Wang, C.; Guo, X.; Wang, C.; Fan, Y.; Wang, Y.; Peng, Y.; Li, W. Liposomal Honokiol Promotes Hair Growth Via Activating Wnt3a/Beta-Catenin Signaling Pathway and Down Regulating TGF-Beta1 in C57BL/6N Mice. Biomed. Pharmacother. 2021, 141, 111793. [Google Scholar] [CrossRef]
- Kang, N.J.; Han, S.C.; Kang, H.J.; Ko, G.; Yoon, W.J.; Kang, H.K.; Yoo, E.S. Anti-Inflammatory Effect of 3-Bromo-4,5-Dihydroxybenzaldehyde, a Component of Polysiphonia morrowii, in Vivo and in Vitro. Toxicol. Res. 2017, 33, 325–332. [Google Scholar] [CrossRef]
- Kang, J.I.; Choi, Y.K.; Han, S.C.; Nam, H.; Lee, G.; Kang, J.H.; Koh, Y.S.; Hyun, J.W.; Yoo, E.S.; Kang, H.K. 5-Bromo-3,4-Dihydroxybenzaldehyde Promotes Hair Growth through Activation of Wnt/Beta-Catenin and Autophagy Pathways and Inhibition of TGF-Beta Pathways in Dermal Papilla Cells. Molecules 2022, 27, 2176. [Google Scholar] [CrossRef]
- Woo, H.; Lee, S.; Kim, S.; Park, D.; Jung, E. Effect of Sinapic Acid on Hair Growth Promoting in Human Hair Follicle Dermal Papilla Cells Via Akt Activation. Arch. Dermatol. Res. 2017, 309, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Kang, J.I.; Hyun, J.W.; Koh, Y.S.; Kang, J.H.; Hyun, C.G.; Yoon, K.S.; Lee, K.S.; Lee, C.M.; Kim, T.Y.; et al. Myristoleic Acid Promotes Anagen Signaling by Autophagy through Activating Wnt/Beta-Catenin and ERK Pathways in Dermal Papilla Cells. Biomol. Ther. 2021, 29, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, H.; Jing, J.; Yu, L.; Wu, X.; Lu, Z. Morroniside Regulates Hair Growth and Cycle Transition Via Activation of the Wnt/Beta-Catenin Signaling Pathway. Sci. Rep. 2018, 8, 13785. [Google Scholar] [CrossRef] [PubMed]
- Romoli, M.; Mazzocchetti, P.; D’Alonzo, R.; Siliquini, S.; Rinaldi, V.E.; Verrotti, A.; Calabresi, P.; Costa, C. Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences. Curr. Neuropharmacol. 2019, 17, 926–946. [Google Scholar] [CrossRef]
| Wnt Isoforms | Cell or Animal Type | Experimental Methods | Functional Roles | References |
|---|---|---|---|---|
| Wnt1a | C57BL/6 mice | Overexpression of Wnt1a-CM derived from BM-MSCs | ↑ Versican, Lef-1 ↑ Gli-1, Ptc-1 ↑ ALP ↑ Transition of telogen to anagen | [37] |
| Wnt3a | Nude mouse | Grafting of Chick embryo fibroblasts producing Wnt3a | ↑ Hair inductive activity | [10] |
| SCD1-null mice | - | ↓ Posttranslational O-palmitoleoylation of Wnt3a | [38] | |
| Balb/c mice DPCs | MAC-EV containing Wnt3a/Wnt7b | ↑ Hair Growth ↑ HF Number, Dermis thickness ↑ HF shaft elongation ↑ KGF, VEGF, Axin2, Lef1 ↑ Versican, ALP, p-AKT, Bcl-2 | [39] | |
| Human hair organ DPCs | WNT3a-CM | ↑ Hair growth | [40] | |
| Wnt4 | Male occipital scalp HFs from patients | ↑ WNT3, WNT4, WNT10B ↑ AXIN2, LEF1 ↓ SFRP1, DKK1 | [41] | |
| Wnt5 | C57BL/6J mice | Overexpression of adenovirus, Wnt5a | ↑ Telogen stage ↓ β-catenin ↓ Myc, Axin2 | [42,43] |
| C57BL/6 mice | Overexpression of adenovirus Wnt5a, and AdSimBC | ↓ β-catenin, Lef1 | [43] | |
| C57BL/6 mice Cdc42GAP−/− mice | overexpression of adenovirus Wnt5a, | ↑ small RhoGTPase Cdc42 | [44] | |
| Wnt10b | whisker hair follicle organ culture from C3H/HeN mice | Medium containing Wnt-10b | ↑ Shaft growth ↑ Keratin 15 in the bulge region | [45] |
| C57BL6/N mice | In vivo injection of AdWnt10b | ↑ Wnt/β-catenin ↑ Hair follicle regeneration | [46] | |
| C57BL/6 mice | Intradermal injection of AdWnt10b | ↑ β-catenin ↑ CD34 | [47] |
| Chemical Name (Active Material) | Cell or Animal Type | Working Concentration | Molecular Targets or Effects | References |
|---|---|---|---|---|
| Aconitie ciliare tuber | Human immortalized DPCs RvDP C57BL/6 mice | 5–50 μg/mL 10 mg/mL | ↑ Wnt/β-catenin ↑ ALP | [60] |
| Centipeda minima (L.) (CMX) A. Braun and Asch | Human DPCs | 0–12.5 μg/mL | ↑ Wnt5a, Frizzled, VEGF ↑ ERK, JNK | [61] |
| Ginkgo biloba extract (Ginkgolide B and bilobalide) | Human DPCs | 0–100 μM | ↑ Akt, ERK ↑ β-catenin ↓ DKK1 | [62,63,64] |
| Malva verticillata (Linoleic acid and oleic acid in n-hexane) | Human DPCs | 20 μg/mL | ↑ Wnt/β-catenin ↑ IGF-1, KGF, ↑ VEGF, HGF ↓ DKK1 | [65,66] |
| Polynesian cosmetopoeia, Bidens pilosa, Calophyllum inophyllum, and Fagraea berteroana | Human DPCs | 0–12.5 μg/mL | ↑ Wnt/β-catenin ↓ DKK1, TGF β | [67] |
| Polygonum multiflorum extract | C57BL6/N mice | 4.7 mg/12 cm2 | ↑ Shh ↑ β-catenin | [68,69] |
| Prunus mira Koehne | C57BL/6 mice | Crude oil 15.06–60.26 mg/cm2/day | ↑ Wnt 10b/β-catenin | [70] |
| Red ginseng oil (RGO) | C57BL/6 mice | 10% | ↑ Wnt/β-catenin ↑ Lef-1, Shh/Gli1 ↑ Cyclin D1, E ↓ TGF-β | [71] |
| Salvia plebeia (SP) R. Brown (Labiatae) | Human DPCs C57BL/6 mice | 0–31.3 μg/mL 1000 μg/mL | ↑ Wnt/β-catenin ↑ Akt, ERK ↑ HGF ↓ TGFβ, SMAD2/3 | [72] |
| Shallot (Allium ascalonicum L.) | Human DPCs | 0.1 mg/mL | ↑ Wnt/β-catenin ↑ VEGF ↑ Shh, SMO, Gil1 | [73] |
| Thuja orientalis | C57BL6/N mice | 5.05 mg/12 cm2/day | ↑ Wnt/β-catenin ↑ Shh | [74] |
| Watercress | Ex vivo organ culture of human hair follicles 44 male subjects with AGA | 2% WCE-containing Lotion (twice daily for 6 months) | ↑ R-spondin 1 ↓ DKK1 ↑ hair thickness (diameter) ↑ hair density | [75] |
| Category | Chemical Name (Active Material) | Cell or Animal Type | Working Concentration | Molecular Targets | References |
|---|---|---|---|---|---|
| Flavonoid | Alpinetin | C57BL/6 J mice | 3 mg/mL | ↑ Lgr5+ HFSCs ↑ Wnt/β-catenin ↑ K15+, Lef1+, Gli1+ ↓ caspase-3 | [76] |
| Flavone glycoside | Baicalin | Balb/c-nu mice transplanted with skin cells of C57BL/6 mice Human DPCs C57BL/6 mice | 50 μM or 100 μM GL-baicalin micelle (each 5 mg/ 1 mg) | ↑ Wnt/β-catenin ↑ Wnt3a ↑ Frizzled 7, ↑ Lef1, ALP, VEGF | [77,78,79] |
| Chalcone | 3-Deoxysappan chalcone (3-DSC) | Human DPCs C57BL/6 mice | 0.1–10 μM 3 mM | ↑ Wnt/β-catenin ↑ VEGF, FGF | [80] |
| Flavanol | Epigallocatechin-3-gallate (EGCG) | Human DPCs ORS cells 44 patients with AGA | 0.25–4 μM Lotion containing EGCG for 24 weeks (twice daily). | ↑ ERK, Akt ↑ Bcl-2/Bax ratio ↑ median anagen-to-telogen ratio | [81,82,83] |
| Flavonol | Fisetin | C57BL/6 mice | 0.1% | ↑ Wnt/β-catenin ↑ KGF | [84,85] |
| Flavone (Quercetin O-glycoside) | Quercitrin | Human DPCs Cultured hair follicles | 0.1–100 nM, 1–10 μM | ↑ NAD(P)H, ΔΨ ↑ Bcl-2 ↑ Akt, ERK, CREB ↑ FGF, KGF, VEGF | [86] |
| Tannin | 3,4,5-tri-O-caffeoyl quinic acid (TCQA) | Human DPCs C3H mice | 10 μM 1% | ↑ Wnt/β-catenin ↑ ALP | [87] |
| Stilbenoid | Resveratrol | C57BL/6 mice | 0.1% | ↑ Wnt/β-catenin KGF | [84,88] |
| Flavonoid | Silibinin | 3D spheroid derived from Human DPCs | 10 μM | ↑ Wnt5 ↑ ALP, Akt ↑ FGF7 | [89] |
| Flavonoid derivative of rutin | Troxerutin | Human DPCs | 10 μM | ↑ Wnt/β-catenin | [90] |
| Monoterpenoid hydroxyl lactone | Loliolide | Human DPCs spheroid | 20 μg/mL | ↑ β-catenin ↑ VEGF, IGF, KGF ↑ Akt, ALP | [91] |
| Sesquiterpene lactone | Costunolide | Human DPCs C57BL/6 mice | 0.1–3 μM | ↑ Wnt/β-catenin ↑ Gli1 ↓ 5α-reductase, TGF- β | [92] |
| Pentacyclic triterpenoid | Oleanolic acid | Human hair follicle organ culture | 1 or 10 μg/mL | ↑ Wnt/β-catenin | [93] |
| Vitamine E analog | Tocotrienol | C57BL/6 mice | 5 mg/cm2 | ↑ β-catenin ↓ E-cadherin | [94] |
| Organic weak acid | Valproic acid | Human DPCs, ORS C57BL/6, C3H mice 40 patients with AGA | 0.1 mM–700 mM A tonic spray containing 8.3% sodium valproate for 24 weeks | ↑ Wnt/β-catenin ↑ ALP ↑ The mean change in total hair count | [95,96,97] |
| Lignan | Honokiol | C57BL/6N mice | 20 mg/mL | ↑ Wnt3a/β-catenin ↓ TGF-β | [98] |
| bromophenol | 5-bromo-3,4-dihydroxybenzaldehyde (BDB) | Human DPCS | 0.01, 0.1, 1 μM | ↑ Wnt/β-catenin ↑ Atg5, Atg7, Atg16L, ↑ LC3II ↓ TGF-β | [99,100] |
| Cinnamic acid derivative | Sinapic acid | Human DPCs | 10, 50, 100 μM | ↑ Wnt/β-catenin ↑ VEGF, IFG-1 ↑ Akt, ERK | [101] |
| Omega-5 fatty acid | Myristoleic acid | Human DPCs | 1, 5 μM | ↑ Wnt/β-catenin ↑ Cyclin A, Cdc2, ↑ Cyclin B1 ↑ ERK, Akt | [102] |
| Iridoid glycoside | Morroniside | ORS cells C57BL/6 mice | 1 or 10 μM 100 μM | ↑ Wnt10b/β-catenin ↑ Lef1 | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, D.W. The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss. Life 2022, 12, 1856. https://doi.org/10.3390/life12111856
Shin DW. The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss. Life. 2022; 12(11):1856. https://doi.org/10.3390/life12111856
Chicago/Turabian StyleShin, Dong Wook. 2022. "The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss" Life 12, no. 11: 1856. https://doi.org/10.3390/life12111856
APA StyleShin, D. W. (2022). The Molecular Mechanism of Natural Products Activating Wnt/β-Catenin Signaling Pathway for Improving Hair Loss. Life, 12(11), 1856. https://doi.org/10.3390/life12111856






