How to Improve the Management of Acute Ischemic Stroke by Modern Technologies, Artificial Intelligence, and New Treatment Methods
Abstract
1. Introduction
2. Stroke Management
2.1. Prehospital Diagnosis
2.1.1. Stroke Symptoms Recognition and First Aid Call
2.1.2. After First Aid Arrival
2.2. Team Activation, Consultation, and Transfer
2.3. Prenotification—Stroke Team Activation before Patient Arrival
2.4. Hospital Diagnosis
2.4.1. Patient Registration
2.4.2. Brain Imaging
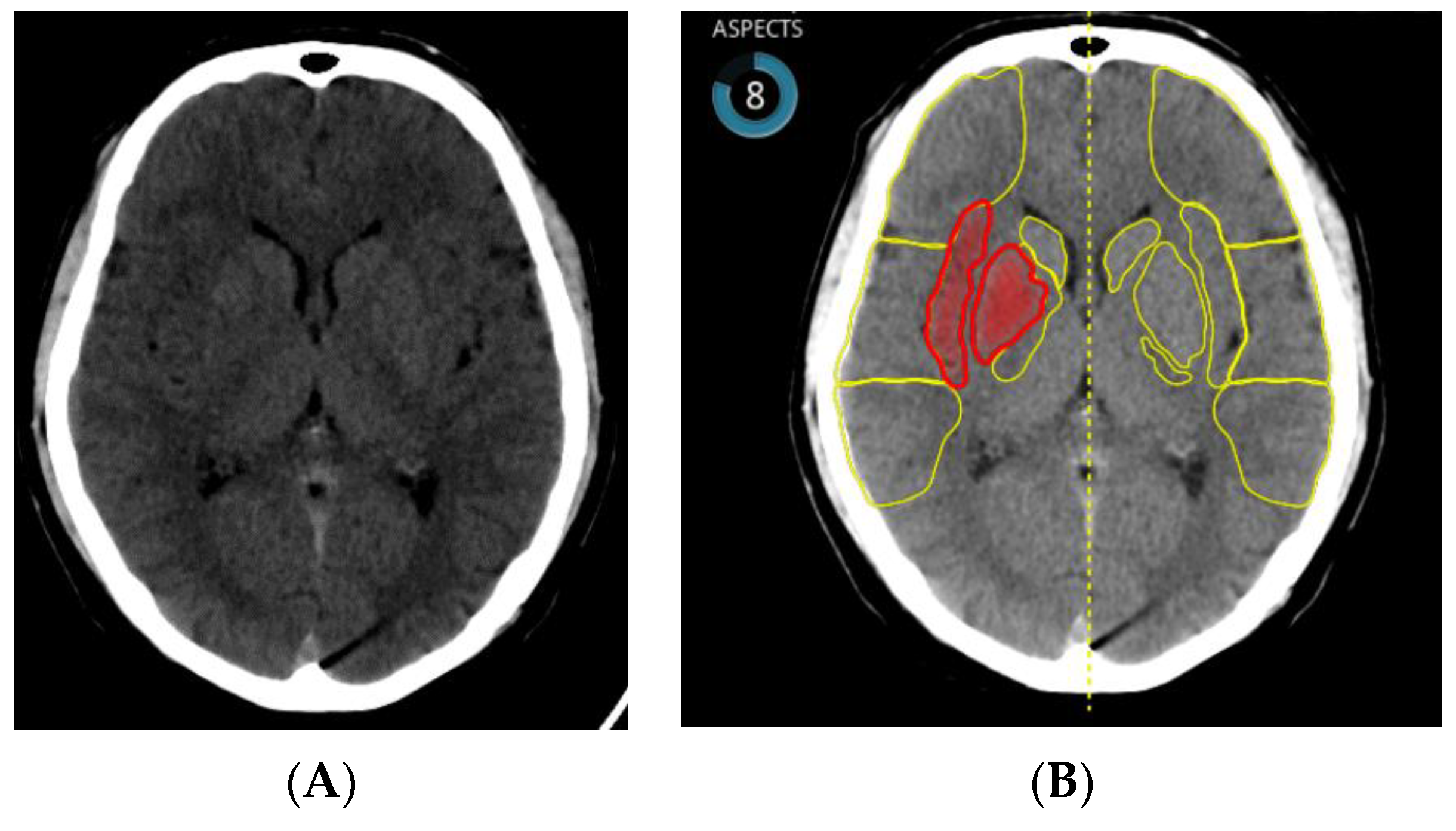
ASPECTS
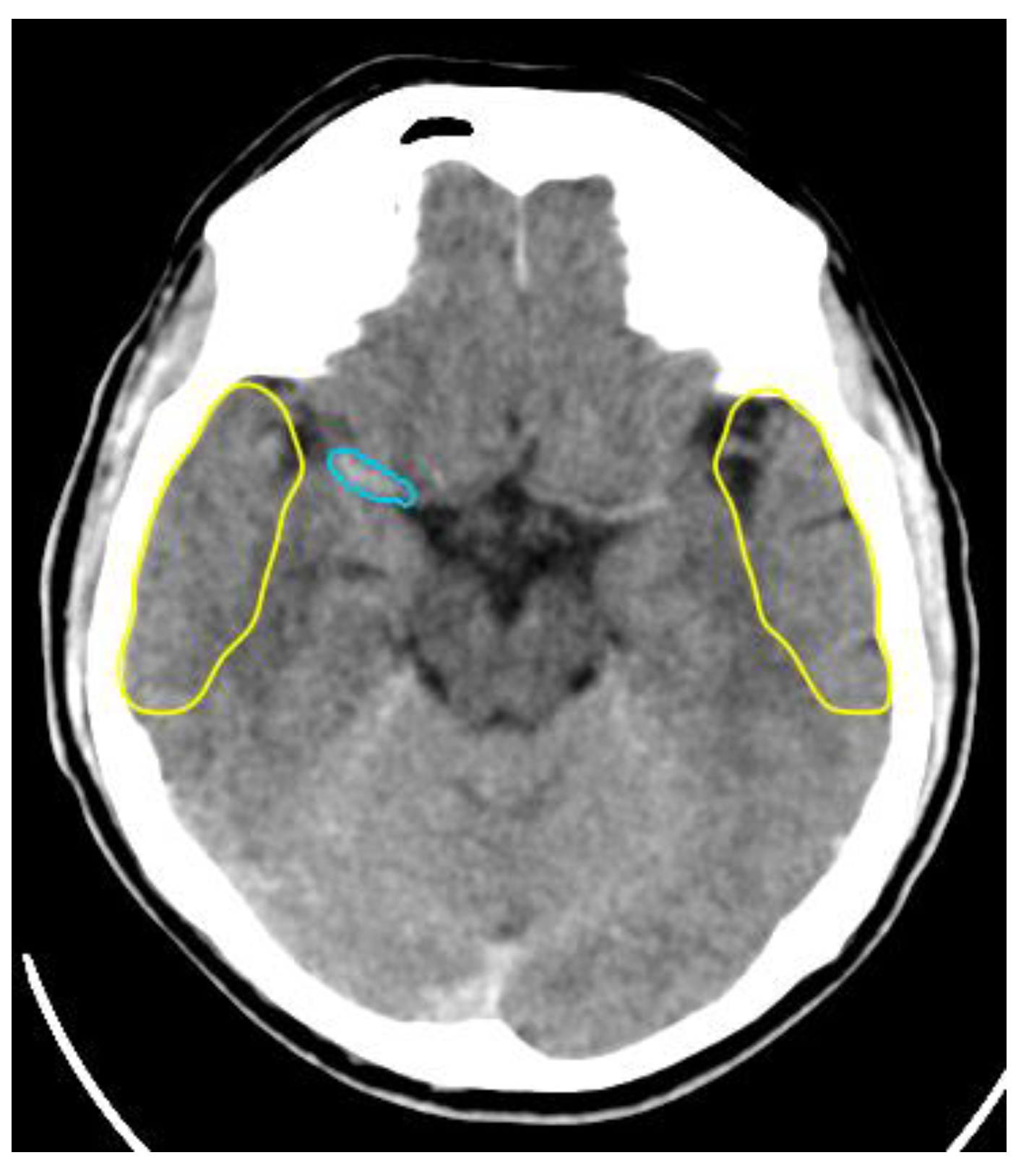
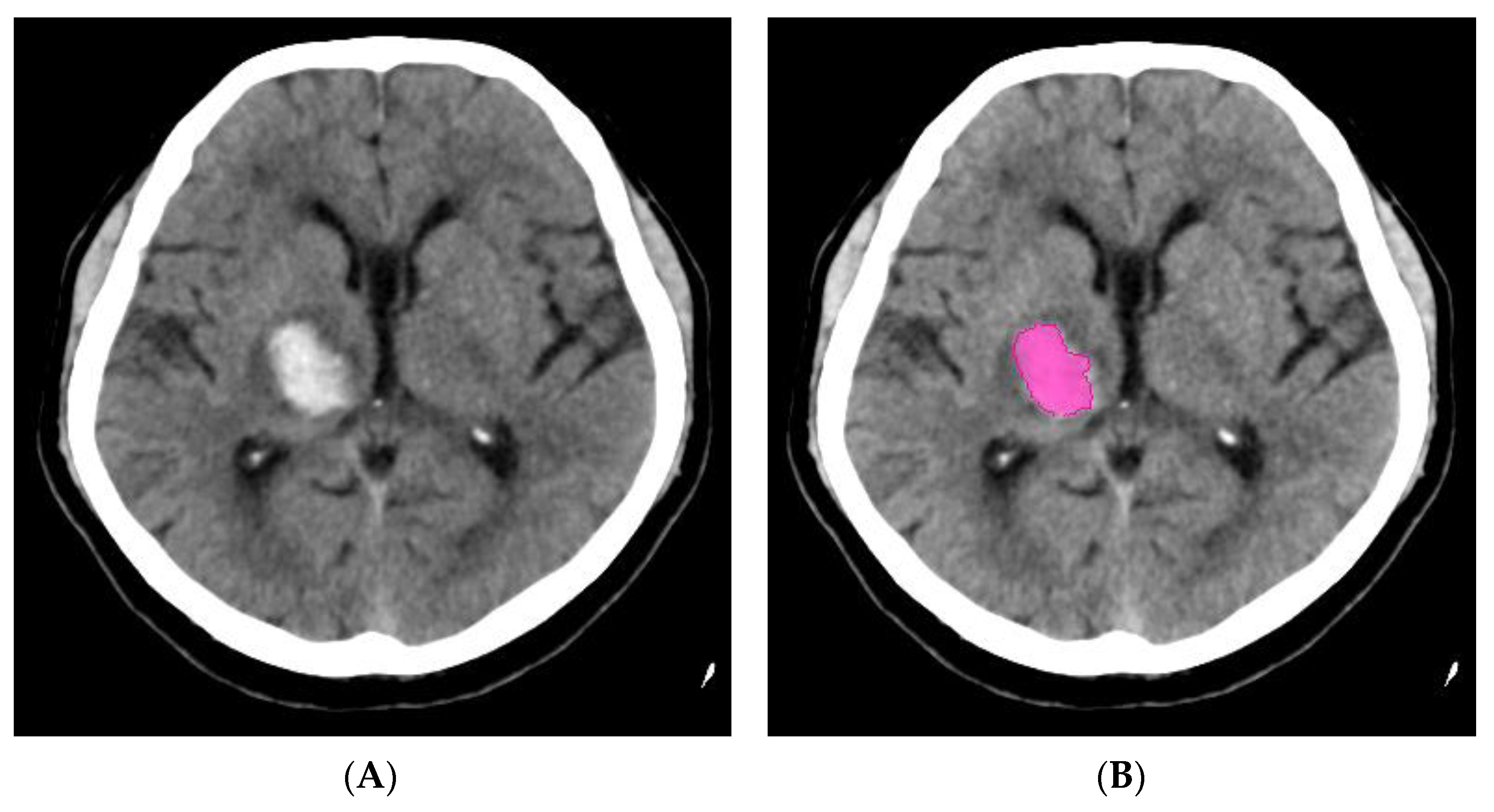
Artery Occlusion Detection
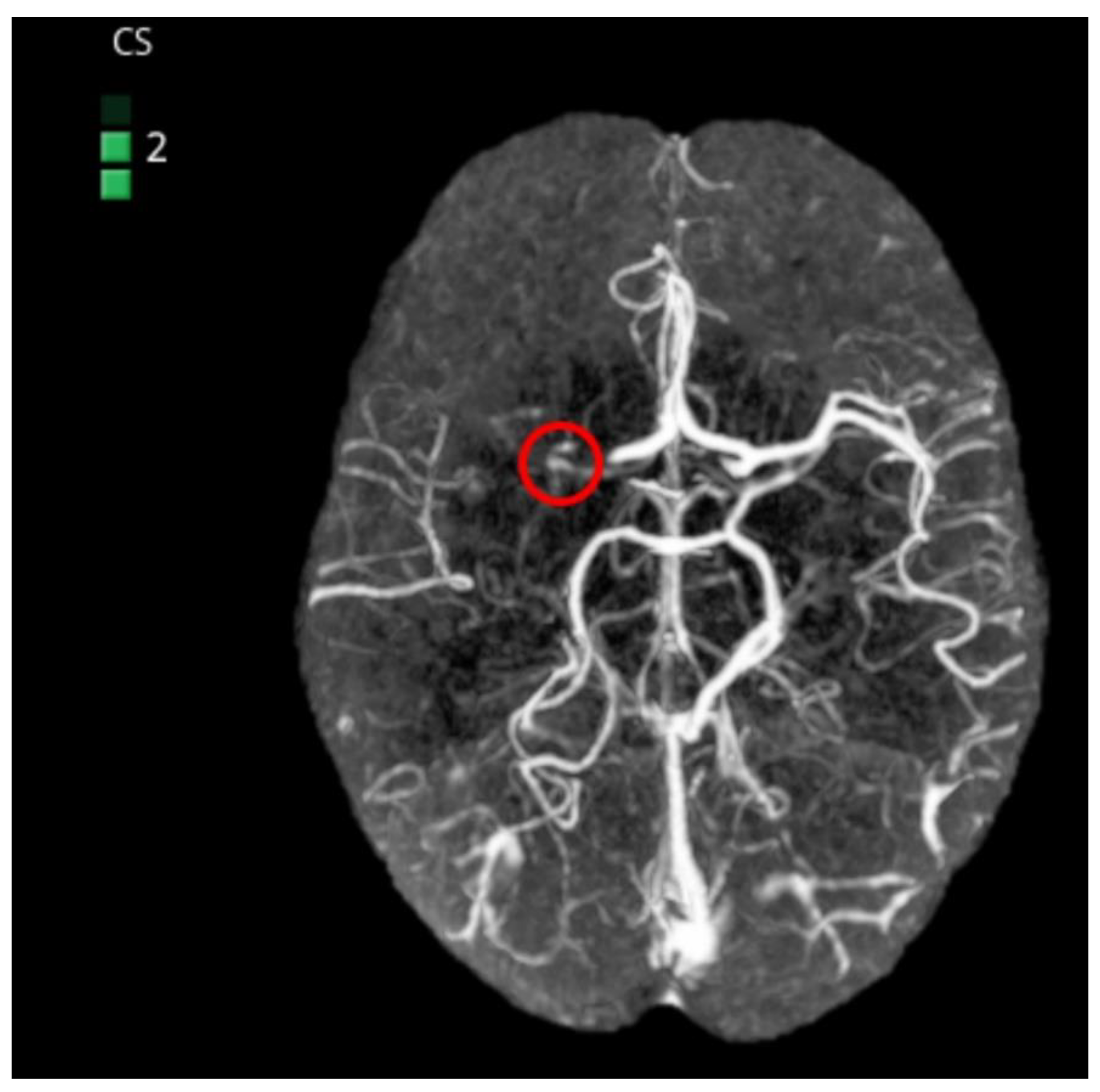
Collateral Score
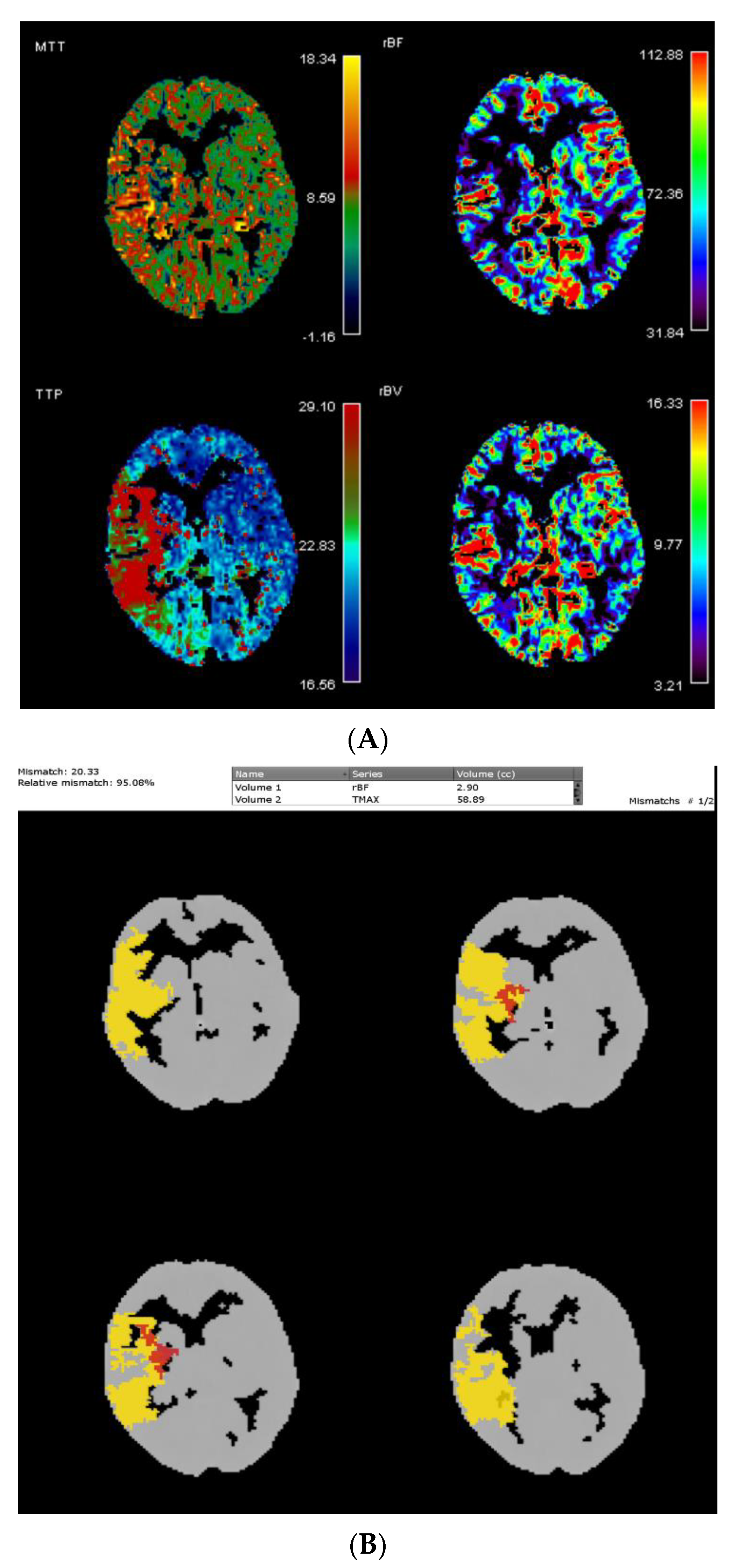
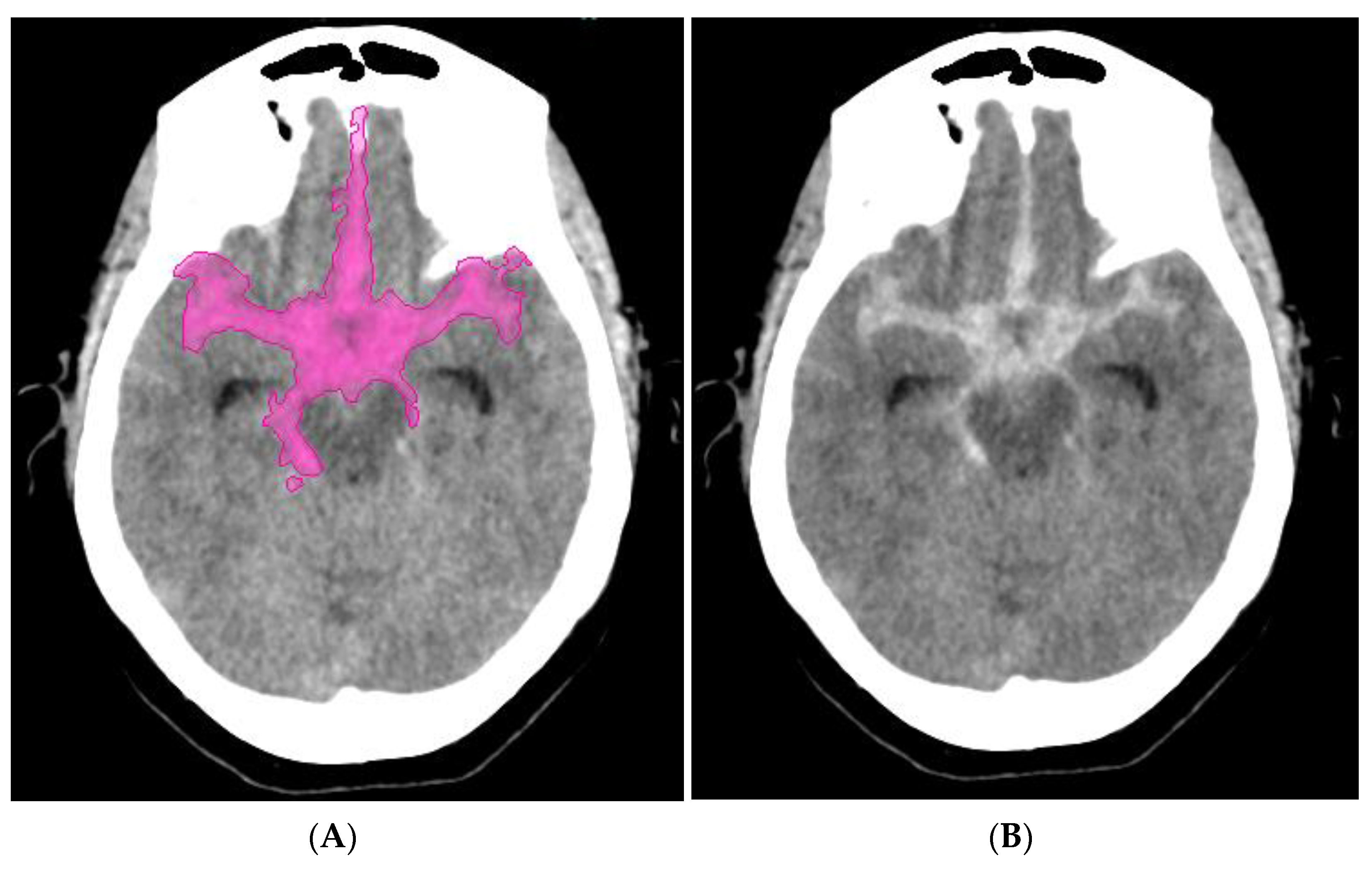
Perfusion Maps
Differential Diagnosis
Prognosis
2.5. Inclusion Criteria and Techniques of the Treatment
2.5.1. First-Pass Effect
2.5.2. Aspiration vs. Stent Retriever
2.5.3. Potential Hemorrhagic Procedural Complications
2.5.4. Neurointerventional Robotics
2.6. Intensive Care, Rehabilitation, and Medical Control
3. Safety and Ethical Problems
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Benjamin:, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart disease and stroke statistics-2018 update: A report from the american heart association [published correction appears in circulation. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef]
- Feigin, V.L.; Forouzanfar, M.H.; Krishnamurthi, R.; Mensah, G.A.; Connor, M.; Bennett, D.A.; Moran, A.E.; Sacco, R.L.; Anderson, L.; Truelsen, T.; et al. Global and regional burden of stroke during 1990-2010: Findings from the Global Burden of Disease Study 2010. Lancet 2014, 383, 245–254. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; De Ferranti, S.; Després, J.-P.; Fullerton, H.J.; et al. Heart disease and stroke statistics-2016 update: A report from the american heart association. Circulation 2016, 133, e38–e360. [Google Scholar] [CrossRef]
- Norrving, B.; Barrick, J.; Davalos, A.; Dichgans, M.; Cordonnier, C.; Guekht, A.; Kutluk, K.; Mikulik, R.; Wardlaw, J.; Richard, E.; et al. Action plan for stroke in Europe 2018—2030. Eur. Stroke J. 2018, 3, 309–336. [Google Scholar] [CrossRef] [PubMed]
- Saver, J.L. Time is brain-quantified. Stroke 2006, 37, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Bechstein, M.; Elsheikh, S.; Wodarg, F.; Taschner, C.A.; Hanning, U.; Buhk, J.-H.; McDonough, R.; Goebell, E.; Fiehler, J.; Bester, M. Republished: Interhospital teleproctoring of endovascular intracranial aneurysm treatment using a dedicated live-streaming technology: First experiences during the COVID-19 pandemic. J. Neurointerv. Surg. 2021, 13, e1. [Google Scholar] [CrossRef] [PubMed]
- Fiehler, J.; Brouwer, P.; Díaz, C.; Hirsch, J.A.; Kulcsar, Z.; Liebeskind, D.; Linfante, I.; Lylyk, P.; Mack, W.J.; Milburn, J.; et al. COVID-19 and neurointerventional service worldwide: A survey of the European Society of Minimally Invasive Neurological Therapy (ESMINT), the Society Of Neurointerventional Surgery (SNIS), the Sociedad Iberolatinoamericana de Neuroradiologia Diagnostica y Terapeutica (SILAN), the Society of Vascular and Interventional Neurology (SVIN), and the World Federation of Interventional and Therapeutic Neuroradiology (WFITN). J. Neurointerv. Surg. 2020, 12, 726–730. [Google Scholar] [CrossRef]
- Goyal, M.; Sutherland, G.R.; Lama, S.; Cimflova, P.; Kashani, N.; Mayank, A.; Psychogios, M.-N.; Spelle, L.; Costalat, V.; Sakai, N.; et al. Neurointerventional robotics: Challenges and opportunities. Clin. Neuroradiol. 2020, 30, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.M.; Cancelliere, N.M.; Nicholson, P.; Radovanovic, I.; Drake, K.E.; Sungur, J.-M.; Krings, T.; Turk, A. First-in-human, robotic-assisted neuroendovascular intervention. J. Neurointerv. Surg. 2020, 12, 338–340. [Google Scholar] [CrossRef]
- Cresswell, K.; Williams, R.; Sheikh, A. Using cloud technology in health care during the COVID-19 pandemic. Lancet Digit. Health 2021, 3, e4–e5. [Google Scholar] [CrossRef]
- Turc, G.; Bhogal, P.; Fischer, U.; Khatri, P.; Lobotesis, K.; Mazighi, M.; Schellinger, P.D.; Toni, D.; De Vries, J.; White, P.; et al. European Stroke Organisation (ESO)—European Society for Minimally Invasive Neurological Therapy (ESMINT) guidelines on mechanical thrombectomy in acute ischemic stroke. J. Neurointerv. Surg. 2019, 11, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Rudd, A.; Bladin, C.; Carli, P.; De Silva, D.; Field, T.; Jauch, E.; Kudenchuk, P.; Kurz, M.; Lærdal, T.; Ong, M.; et al. Utstein recommendation for emergency stroke care. Int. J. Stroke 2020, 15, 555–564. [Google Scholar] [CrossRef] [PubMed]
- El-Ghanem, M.; Al-Mufti, F.; Thulasi, V.; Singh, I.P.; Gandhi, C. Expanding the treatment window for ischemic stroke through the application of novel system-based technology. Neurosurg. Focus. 2017, 42, E7. [Google Scholar] [CrossRef]
- Duong, M.T.; Rauschecker, A.M.; Rudie, J.D.; Chen, P.-H.; Cook, T.S.; Bryan, R.N.; Mohan, S. Artificial intelligence for precision education in radiology. Br. J. Radiol. 2019, 92, 20190389. [Google Scholar] [CrossRef] [PubMed]
- Forney, M.C.; McBride, A.F. Artificial intelligence in radiology residency training. Semin. Musculoskelet. Radiol. 2020, 24, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Soun, J.; Chow, D.; Nagamine, M.; Takhtawala, R.; Filippi, C.; Yu, W.; Chang, P. Artificial intelligence and acute stroke imaging. Am. J. Neuroradiol. 2021, 42, 2–11. [Google Scholar] [CrossRef]
- Mokli, Y.; Pfaff, J.; Dos Santos, D.P.; Herweh, C.; Nagel, S. Computer-aided imaging analysis in acute ischemic stroke—background and clinical applications. Neurol. Res. Pract. 2019, 1, 23. [Google Scholar] [CrossRef]
- Mouridsen, K.; Thurner, P.; Zaharchuk, G. Artificial intelligence applications in stroke. Stroke 2020, 51, 2573–2579. [Google Scholar] [CrossRef]
- Heo, J.; Yoon, J.G.; Park, H.; Kim, Y.D.; Nam, H.S.; Heo, J.H. Machine learning-based model for prediction of outcomes in acute stroke. Stroke 2019, 50, 1263–1265. [Google Scholar] [CrossRef]
- Ishii, E.; Ebner, D.K.; Kimura, S.; Agha-Mir-Salim, L.; Uchimido, R.; Celi, L.A. The advent of medical artificial intelligence: Lessons from the Japanese approach. J. Intensive Care 2020, 8, 35. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, T.; Zhang, Z.; Lin, B.; Mei, Y.; Zhang, Y.; Chen, G. The current status of stroke-related smartphone applications available to adopt in China: A systematic review study. Medicine 2020, 99, e20656. [Google Scholar] [CrossRef] [PubMed]
- Bandini, A.; Green, J.; Richburg, B.; Yunusova, Y. Automatic detection of orofacial impairment in stroke. Interspeech 2018, 2018, 1711–1715. [Google Scholar] [CrossRef]
- Van Gaal, S.; Demchuk, A. Clinical and technological approaches to the prehospital diagnosis of large vessel occlusion. Stroke 2018, 49, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, R.G.; Silva, G.S.; Lima, F.O.; Yeh, Y.-C.; Fleming, C.; Branco, D.; Yancey, A.H.; Ratcliff, J.J.; Wages, R.K.; Doss, E.; et al. The FAST-ED app: A smartphone platform for the field triage of patients with stroke. Stroke 2017, 48, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Malka, R.; Miller, M.; Guarin, D.; Fullerton, Z.; Hadlock, T.; Banks, C. Reliability between in-person and still photograph assessment of facial function in facial paralysis using the eFACE facial grading system. Facial Plast. Surg. Aesthet. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Torab-Miandoab, A.; Samad-Soltani, T.; Shams-Vahdati, S.; Rezaei-Hachesu, P. An intelligent system for improving adherence to guidelines on acute stroke. Turk. J. Emerg. Med. 2020, 20, 118–134. [Google Scholar] [CrossRef]
- Lin, M.P.; Sanossian, N.; Liebeskind, D.S. Imaging of prehospital stroke therapeutics. Expert Rev. Cardiovasc. Ther. 2015, 13, 1001–1015. [Google Scholar] [CrossRef]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the american heart association/american stroke association. Stroke 2019, 50, e344–e418. [Google Scholar] [CrossRef]
- Bayona, H.; Ropero, B.; Salazar, A.J.; Pérez, J.C.; Granja, M.F.; Martínez, C.F.; Useche, J.N. Comprehensive telestroke network to optimize health care delivery for cerebrovascular diseases: Algorithm development. J. Med. Internet Res. 2020, 22, e18058. [Google Scholar] [CrossRef]
- Hubert, G.J.; Santo, G.; Vanhooren, G.; Zvan, B.; Campos, S.T.; Alasheev, A.; Abilleira, S.; Corea, F. Recommendations on telestroke in Europe. Eur. Stroke J. 2019, 4, 101–109. [Google Scholar] [CrossRef]
- Phan, T.G.; Beare, R.; Srikanth, V.; Ma, H. Googling location for operating base of mobile stroke unit in metropolitan Sydney. Front. Neurol. 2019, 10, 810. [Google Scholar] [CrossRef] [PubMed]
- Stemi. Available online: https://www.stemiglobal.com (accessed on 1 February 2021).
- Noone, M.L.; Moideen, F.; Krishna, R.B.; Kumar, V.P.; Karadan, U.; Chellenton, J.; Salam, K. Mobile app based strategy improves door-to-needle time in the treatment of acute ischemic stroke. J. Stroke Cerebrovasc. Dis. 2020, 29, 105319. [Google Scholar] [CrossRef]
- Tian, M.; Ajay, V.S.; Dunzhu, D.; Hameed, S.S.; Li, X.; Liu, Z.; Li, C.; Chen, H.; Cho, K.; Li, R.; et al. A cluster-randomized, controlled trial of a simplified multifaceted management program for individuals at high cardiovascular risk (SimCard Trial) in Rural Tibet, China, and Haryana, India. Circulation 2015, 132, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Dubey, D.; Amritphale, A.; Sawhney, A.; Amritphale, N.; Dubey, P.; Pandey, A. Smart phone applications as a source of information on stroke. J. Stroke 2014, 16, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Forcier, M.B.; Gallois, H.; Mullan, S.; Joly, Y. Integrating artificial intelligence into health care through data access: Can the GDPR act as a beacon for policymakers? J. Law Biosci. 2019, 6, 317–335. [Google Scholar] [CrossRef]
- Mansour, O.Y.; Ramadan, I.; Elfatatry, A.; Hamdi, M.; Abudu, A.; Hassan, T.; Eldeeb, H.; Marouf, H.; Mogahed, M.; Farouk, M.; et al. Using ESN-smartphone application to maximize ais reperfusion therapy in Alexandria Stroke Network: A stroke chain of survival organizational model. Front. Neurol. 2021, 12, 597717. [Google Scholar] [CrossRef]
- Stroke Clock App. Available online: https://www.sublimestudio.ch/en/portfolio/stroke-clock-app-en/ (accessed on 20 May 2021).
- Fousse, M.; Grün, D.; Helwig, S.A.; Walter, S.; Bekhit, A.; Wagenpfeil, S.; Lesmeister, M.; Kettner, M.; Roumia, S.; Mühl-Benninghaus, R.; et al. Effects of a feedback-demanding stroke clock on acute stroke management: A randomized study. Stroke. 2020, 51, 2895–2900. [Google Scholar] [CrossRef]
- Bourcier, R.; Goyal, M.; Liebeskind, D.S.; Muir, K.W.; Desal, H.; Siddiqui, A.H.; Dippel, D.W.J.; Majoie, C.B.; Van Zwam, W.H.; Jovin, T.G.; et al. Association of time from stroke onset to groin puncture with quality of reperfusion after mechanical thrombectomy: A meta-analysis of individual patient data from 7 randomized clinical trials. JAMA Neurol. 2019, 76, 405–411. [Google Scholar] [CrossRef]
- Katsanos, A.H. Updates in stroke treatment, diagnostic methods and predictors of outcome. J. Clin. Med. 2020, 9, 2789. [Google Scholar] [CrossRef]
- Psychogios, M.-N.; Maier, I.L.; Tsogkas, I.; Hesse, A.C.; Brehm, A.; Behme, D.; Schnieder, M.; Schregel, K.; Papageorgiou, I.; Liebeskind, D.S.; et al. One-stop management of 230 consecutive acute stroke patients: Report of procedural times and clinical outcome. J. Clin. Med. 2019, 8, 2185. [Google Scholar] [CrossRef]
- Bash, S.; Tanenbaum, L.N. Deep learning: Promising to revolutionize image reconstruction. Appl. Radiol. 2021, 50, 32–37. [Google Scholar]
- Nagendran, M.; Chen, Y.; Lovejoy, C.A.; Gordon, A.C.; Komorowski, M.; Harvey, H.; Topol, E.J.; Ioannidis, J.P.A.; Collins, G.S.; Maruthappu, M. Artificial intelligence versus clinicians: Systematic review of design, reporting standards, and claims of deep learning studies. BMJ 2020, 368, m689. [Google Scholar] [CrossRef]
- Xie, W.; Cao, X.; Dong, H.; Liu, Y. The use of smartphone-based triage to reduce the rate of outpatient error registration: Cross-sectional study. JMIR mHealth uHealth. 2019, 7, e15313. [Google Scholar] [CrossRef]
- Hosny, A.; Parmar, C.; Quackenbush, J.; Schwartz, L.H.; Aerts, H.J.W.L. Artificial intelligence in radiology. Nat. Rev. Cancer. 2018, 18, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Sala, E.; Ursprung, S. Artificial intelligence in radiology: The computer’s helping hand need guidance. Radiol. Artif. Intell. 2020, 2, e200207. [Google Scholar] [CrossRef]
- Lotan, E. Emerging artificial intelligence imaging applications for stroke interventions. Am. J. Neuroradiol. 2021, 42, 255–256. [Google Scholar] [CrossRef]
- Liaw, N.; Liebeskind, D. Emerging therapies in acute ischemic stroke. F1000Research 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Nagel, S.; Herweh, C.; Pfaff, J.A.R.; Schieber, S.; Schönenberger, S.; Möhlenbruch, M.A.; Bendszus, M.; Ringleb, P.A. Simplified selection criteria for patients with longer or unknown time to treatment predict good outcome after mechanical thrombectomy. J. Neurointerv. Surg. 2019, 11, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Van Horn, N.; Kniep, H.; Broocks, G.; Meyer, L.; Flottmann, F.; Bechstein, M.; Götz, J.; Thomalla, G.; Bendszus, M.; Bonekamp, S.; et al. ASPECTS interobserver agreement of 100 investigators from the TENSION Study. Clin. Neuroradiol. 2021. [Google Scholar] [CrossRef]
- Seker, F.; Pfaff, J.; Nagel, S.; Vollherbst, D.; Gerry, S.; Möhlenbruch, M.A.; Bendszus, M.; Herweh, C. CT Reconstruction levels affect automated and reader-based ASPECTS ratings in acute ischemic stroke. J. Neuroimaging 2019, 29, 62–64. [Google Scholar] [CrossRef]
- Nagel, S.; Sinha, D.; Day, D.; Reith, W.; Chapot, R.; Papanagiotou, P.; Warburton, E.A.; Guyler, P.; Tysoe, S.; Fassbender, K.; et al. e-ASPECTS software is non-inferior to neuroradiologists in applying the ASPECT score to computed tomography scans of acute ischemic stroke patients. Int. J. Stroke 2017, 12, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Austein, F.; Wodarg, F.; Jürgensen, N.; Huhndorf, M.; Meyne, J.; Lindner, T.; Jansen, O.; Larsen, N.; Riedel, C. Automated versus manual imaging assessment of early ischemic changes in acute stroke: Comparison of two software packages and expert consensus. Eur. Radiol. 2019, 29, 6285–6292. [Google Scholar] [CrossRef]
- Murray, N.M.; Unberath, M.; Hager, G.D.; Hui, F.K. Artificial intelligence to diagnose ischemic stroke and identify large vessel occlusions: A systematic review. J. Neurointerv. Surg. 2020, 12, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Lee, Y.; Tsai, D.Y.; Matsuyama, E.; Kinoshita, T.; Ishii, K. An automated detection method for the MCA dot sign of acute stroke in unenhanced CT. Radiol. Phys. Technol. 2014, 7, 79–88. [Google Scholar] [CrossRef]
- Chatterjee, A.; Somayaji, N.R.; Kabakis, I.M. Abstract WMP16: Artificial intelligence detection of cerebrovascular large vessel occlusion—nine month, 650 patient evaluation of the diagnostic accuracy and performance of the Viz.ai LVO algorithm. Stroke 2019, 50, AWMP16. [Google Scholar] [CrossRef]
- Olive-Gadea, M.; Crespo, C.; Granes, C.; Hernandez-Perez, M.; De La Ossa, N.P.; Laredo, C.; Urra, X.; Soler, J.C.; Soler, A.; Puyalto, P.; et al. Deep learning based software to identify large vessel occlusion on noncontrast computed tomography. Stroke 2020, 51, 3133–3137. [Google Scholar] [CrossRef]
- Hanning, U.; Sporns, P.B.; Psychogios, M.N.; Jeibmann, A.; Minnerup, J.; Gelderblom, M.; Schulte, K.; Nawabi, J.; Broocks, G.; Meyer, L.; et al. Imaging-based prediction of histological clot composition from admission CT imaging. J. Neurointerv. Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Rabinstein, A.A. Update on treatment of acute ischemic stroke. Continuum 2020, 26, 268–286. [Google Scholar] [CrossRef]
- Grunwald, I.Q.; Kulikovski, J.; Reith, W.; Gerry, S.; Namias, R.; Politi, M.; Papanagiotou, P.; Essig, M.; Mathur, S.; Joly, O.; et al. Collateral automation for triage in stroke: Evaluating automated scoring of collaterals in acute stroke on computed tomography scans. Cerebrovasc. Dis. 2019, 47, 217–222. [Google Scholar] [CrossRef]
- Shah, V.S.; Eaton, R.G.; Cua, S.; Dornbos, D.; Hoang, N.; Schunemann, V.; Nimjee, S.; Youssef, P.; Powers, C.J. Scoring of middle cerebral artery collaterals predicts RAPID CT-perfusion analysis and short-term outcomes in acute ischemic stroke patients undergoing thrombectomy. World Neurosurg. 2020, 135, e494–e499. [Google Scholar] [CrossRef] [PubMed]
- Faizy, T.D.; Kabiri, R.; Christensen, S.; Mlynash, M.; Kuraitis, G.; Meyer, L.; Marks, M.P.; Broocks, G.; Flottmann, F.; Lansberg, M.G.; et al. Venous outflow profiles are linked to cerebral edema formation at noncontrast head CT after treatment in acute ischemic stroke regardless of collateral vessel status at CT angiography. Radiology 2021, 299, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Scheldeman, L.; Wouters, A.; Boutitie, F.; Dupont, P.; Christensen, S.; Cheng, B.; Ebinger, M.; Endres, M.; Fiebach, J.B.; Gerloff, C.; et al. Different mismatch concepts for magnetic resonance imaging-guided thrombolysis in unknown onset stroke. Ann. Neurol. 2020, 87, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Koopman, M.S.; Berkhemer, O.A.; Geuskens, R.R.E.G.; Emmer, B.J.; Van Walderveen, M.A.A.; Jenniskens, S.F.M.; Van Zwam, W.H.; Van Oostenbrugge, R.J.; Van Der Lugt, A.; Dippel, D.W.J.; et al. Comparison of three commonly used CT perfusion software packages in patients with acute ischemic stroke. J. Neurointerv. Surg. 2019, 11, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Rava, R.; Snyder, K.; Mokin, M.; Waqas, M.; Allman, A.; Senko, J.; Podgorsak, A.; Bhurwani, M.S.; Hoi, Y.; Siddiqui, A.; et al. Assessment of a bayesian vitrea CT perfusion analysis to predict final infarct and penumbra volumes in patients with acute ischemic stroke: A comparison with RAPID. Am. J. Neuroradiol. 2020, 41, 206–212. [Google Scholar] [CrossRef]
- McDougall, C.; Chan, L.; Sachan, S.; Guo, J.; Sah, R.; Menon, B.; Demchuk, A.; Hill, M.; Forkert, N.; D’Esterre, C.; et al. Dynamic CTA-derived perfusion maps predict final infarct volume: The simple perfusion reconstruction algorithm. Am. J. Neuroradiol. 2020, 41, 2034–2040. [Google Scholar] [CrossRef]
- Reid, M.; Famuyide, A.O.; Forkert, N.D.; Talai, A.S.; Evans, J.W.; Sitaram, A.; Hafeez, M.; Najm, M.; Menon, B.K.; Demchuk, A.; et al. Accuracy and reliability of multiphase CTA perfusion for identifying ischemic core. Clin. Neuroradiol. 2019, 29, 543–552. [Google Scholar] [CrossRef]
- Dundamadappa, S.; Iyer, K.; Agrawal, A.; Choi, D.J. Multiphase CT angiography: A useful technique in acute stroke imaging-collaterals and beyond. Am. J. Neuroradiol. 2021, 42, 221–227. [Google Scholar] [CrossRef]
- Ospel, J.; Volny, O.; Qiu, W.; Najm, M.; Kashani, N.; Goyal, M.; Menon, B. Displaying multiphase CT angiography using a time-variant color map: Practical considerations and potential applications in patients with acute stroke. Am. J. Neuroradiol. 2020, 41, 200–205. [Google Scholar] [CrossRef]
- Verdolotti, T.; Pilato, F.; Cottonaro, S.; Monelli, E.; Giordano, C.; Guadalupi, P.; Benenati, M.; Ramaglia, A.; Costantini, A.M.; Alexandre, A.; et al. ColorViz, a new and rapid tool for assessing collateral circulation during stroke. Brain Sci. 2020, 10, 882. [Google Scholar] [CrossRef]
- Meier, R.; Lux, P.; Med, B.; Jung, S.; Fischer, U.; Gralla, J.; Reyes, M.; Wiest, R.; McKinley, R.; Kaesmacher, J. Neural network–derived perfusion maps for the assessment of lesions in patients with acute ischemic stroke. Radiol. Artif. Intell. 2019, 1, 5. [Google Scholar] [CrossRef]
- Lee, H.; Yune, S.; Mansouri, M.; Kim, M.; Tajmir, S.H.; Guerrier, C.E.; Ebert, S.A.; Pomerantz, S.R.; Romero, J.M.; Kamalian, S.; et al. An explainable deep-learning algorithm for the detection of acute intracranial haemorrhage from small datasets. Nat. Biomed. Eng. 2019, 3, 173–182. [Google Scholar] [CrossRef]
- Ye, H.; Gao, F.; Yin, Y.; Guo, D.; Zhao, P.; Lu, Y.; Wang, X.; Bai, J.; Cao, K.; Song, Q.; et al. Precise diagnosis of intracranial hemorrhage and subtypes using a three-dimensional joint convolutional and recurrent neural network. Eur. Radiol. 2019, 29, 6191–6201. [Google Scholar] [CrossRef]
- Chilamkurthy, S.; Ghosh, R.; Tanamala, S.; Biviji, M.; Campeau, N.G.; Venugopal, V.K.; Mahajan, V.; Rao, P.; Warier, P. Deep learning algorithms for detection of critical findings in head CT scans: A retrospective study. Lancet 2018, 392, 2388–2396. [Google Scholar] [CrossRef]
- Desai, V.; Flanders, A.E.; Lakhani, P. Application of deep learning in neuroradiology: Automated detection of basal ganglia hemorrhage using 2D-convolutional neural networks. arXiv 2017, arXiv:1710.03823. Available online: https://arxiv.org/ftp/arxiv/papers/1710/1710.03823.pdf (accessed on 20 February 2021).
- Yeo, M.; Tahayori, B.; Kok, H.K.; Maingard, J.; Kutaiba, N.; Russell, J.; Thijs, V.; Jhamb, A.; Chandra, R.V.; Brooks, M.; et al. Review of deep learning algorithms for the automatic detection of intracranial hemorrhages on computed tomography head imaging. J. Neurointerv. Surg. 2021, 13, 369–378. [Google Scholar] [CrossRef]
- Ginat, D.T. Analysis of head CT scans flagged by deep learning software for acute intracranial hemorrhage. Neuroradiology 2020, 62, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Kaka, H.; Zhang, E.; Khan, N. Artificial intelligence and deep learning in neuroradiology: Exploring the New Frontier. Can. Assoc. Radiol. J. 2021, 72, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Lui, Y.W.; Chang, P.D.; Zaharchuk, G.; Barboriak, D.P.; Flanders, A.E.; Wintermark, M.; Hess, C.P.; Filippi, C.G. Artificial intelligence in neuroradiology: Current status and future directions. Am. J. Neuroradiol. 2020, 41, E52–E59. [Google Scholar] [CrossRef]
- Ho, K.C.; Scalzo, F.; Sarma, K.V.; Speier, W.; El-Saden, S.; Arnold, C. Predicting ischemic stroke tissue fate using a deep convolutional neural network on source magnetic resonance perfusion images. J. Med. Imaging 2019, 6, 026001. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Xie, Y.; Thamm, T.; Gong, E.; Ouyang, J.; Huang, C.; Christensen, S.; Marks, M.P.; Lansberg, M.G.; Albers, G.W.; et al. Use of deep learning to predict final ischemic stroke lesions from initial magnetic resonance imaging. JAMA Netw. Open. 2020, 3, e200772. [Google Scholar] [CrossRef]
- Debs, N.; Cho, T.-H.; Rousseau, D.; Berthezène, Y.; Buisson, M.; Eker, O.; Mechtouff, L.; Nighoghossian, N.; Ovize, M.; Frindel, C. Impact of the reperfusion status for predicting the final stroke infarct using deep learning. Neuroimage. Clin. 2021, 29, 102548. [Google Scholar] [CrossRef]
- Pfaff, J.; Herweh, C.; Schieber, S.; Schönenberger, S.; Bösel, J.; Ringleb, P.; Möhlenbruch, M.; Bendszus, M.; Nagel, S. e-ASPECTS correlates with and is predictive of outcome after mechanical thrombectomy. Am. J. Neuroradiol. 2017, 38, 1594–1599. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Guo, D.; Lou, M.; Liebeskind, D.; Scalzo, F. Prediction of hemorrhagic transformation severity in acute stroke from source perfusion MRI. IEEE Trans. Biomed. Eng. 2018, 65, 2058–2065. [Google Scholar] [CrossRef]
- Bacchi, S.; Oakden-Rayner, L.; Zerner, T.; Kleinig, T.; Patel, S.; Jannes, J. Deep learning natural language processing successfully predicts the cerebrovascular cause of transient ischemic attack-like presentations. Stroke 2019, 50, 758–760. [Google Scholar] [CrossRef]
- Cimflova, P.; Volny, O.; Mikulik, R.; Tyshchenko, B.; Belaskova, S.; Vinklarek, J.; Cervenak, V.; Krivka, T.; Vanicek, A.P.J.; Krajina, A. Detection of ischemic changes on baseline multimodal computed tomography: Expert reading vs. Brainomix and RAPID software. J. Stroke Cerebrovasc. Dis. 2020, 29, 104978. [Google Scholar] [CrossRef]
- Goyal, M.; Menon, B.K.; Van Zwam, W.H.; Dippel, D.W.; Mitchell, P.J.; Demchuk, A.M.; Dávalos, A.; Majoie, C.B.; Van Der Lugt, A.; De Miquel, M.A.; et al. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 2016, 387, 1723–1731. [Google Scholar] [CrossRef]
- Albers, G.W.; Marks, M.P.; Kemp, S.; Christensen, S.; Tsai, J.P.; Ortega-Gutierrez, S.; McTaggart, R.A.; Torbey, M.T.; Kim-Tenser, M.; Leslie-Mazwi, T.; et al. Thrombectomy for stroke at 6 to 16 h with selection by perfusion imaging. N. Engl. J. Med. 2018, 378, 708–718. [Google Scholar] [CrossRef]
- Nogueira, R.G.; Jadhav, A.P.; Haussen, D.C.; Bonafe, A.; Budzik, R.F.; Bhuva, P.; Yavagal, D.R.; Ribo, M.; Cognard, C.; Hanel, R.A.; et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N. Engl. J. Med. 2018, 378, 11–21. [Google Scholar] [CrossRef]
- Bendszus, M.; Bonekamp, S.; Berge, E.; Boutitie, F.; Brouwer, P.; Gizewski, E.; Krajina, A.; Pierot, L.; Randall, G.; Simonsen, C.Z.; et al. A randomized controlled trial to test efficacy and safety of thrombectomy in stroke with extended lesion and extended time window. Int. J. Stroke 2019, 14, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.; Ospel, J.M.; Menon, B.K.; Hill, M.D. MeVO: The next frontier? J. Neurointerv. Surg. 2020, 12, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Meyer, L.; Stracke, C.P.; Jungi, N.; Wallocha, M.; Broocks, G.; Sporns, P.B.; Maegerlein, C.; Dorn, F.; Zimmermann, H.; Naziri, W.; et al. Thrombectomy for primary distal posterior cerebral artery occlusion stroke: The TOPMOST study. JAMA Neurol. 2021, 78, 434–444. [Google Scholar] [CrossRef]
- Zi, W.; Qiu, Z.; Wu, D.; Li, F.; Liu, H.; Liu, W.; Huang, W.; Shi, Z.; Bai, Y.; Liu, Z.; et al. Assessment of endovascular treatment for acute basilar artery occlusion via a nationwide prospective registry. JAMA Neurol. 2020, 77, 561–573. [Google Scholar] [CrossRef]
- Liu, X.; Dai, Q.; Ye, R.; Zi, W.; Liu, Y.; Wang, H.; Zhu, W.; Ma, M.; Yin, Q.; Li, M.; et al. Endovascular treatment versus standard medical treatment for vertebrobasilar artery occlusion (BEST): An open-label, randomised controlled trial. Lancet Neurol. 2020, 19, 115–122. [Google Scholar] [CrossRef]
- Zaidat, O.O.; Castonguay, A.C.; Linfante, I.; Gupta, R.; Martin, C.O.; Holloway, W.E.; Mueller-Kronast, N.; English, J.D.; Dabus, G.; Malisch, T.W.; et al. First pass effect: A new measure for stroke thrombectomy devices. Stroke 2018, 49, 660–666. [Google Scholar] [CrossRef]
- Kaesmacher, J.; Dobrocky, T.; Heldner, M.R.; Bellwald, S.; Mosimann, P.J.; Mordasini, P.; Bigi, S.; Arnold, M.; Gralla, J.; Fischer, U. Systematic review and meta-analysis on outcome differences among patients with TICI2b versus TICI3 reperfusions: Success revisited. J. Neurol. Neurosurg. Psychiatry 2018, 89, 910–917. [Google Scholar] [CrossRef]
- Brinjikji, W.; Starke, R.M.; Murad, M.H.; Fiorella, D.; Pereira, V.M.; Goyal, M.; Kallmes, D.F. Impact of balloon guide catheter on technical and clinical outcomes: A systematic review and meta-analysis. J. Neurointerv. Surg. 2018, 10, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.-H.; Kim, B.M.; Kang, D.-H.; Heo, J.H.; Nam, H.S.; Kim, Y.D.; Hwang, Y.-H.; Kim, Y.-W.; Kim, D.J.; Kwak, H.S.; et al. Balloon guide catheter is beneficial in endovascular treatment regardless of mechanical recanalization modality. Stroke 2019, 50, 1490–1496. [Google Scholar] [CrossRef]
- Zaidat, O.O.; Mueller-Kronast, N.H.; Hassan, A.E.; Haussen, D.C.; Jadhav, A.P.; Froehler, M.T.; Jahan, R.; Aziz-Sultan, M.A.; Klucznik, R.P.; Saver, J.L.; et al. Impact of balloon guide catheter use on clinical and angiographic outcomes in the STRATIS stroke thrombectomy registry. Stroke 2019, 50, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Ospel, J.M.; Volny, O.; Jayaraman, M.; McTaggart, R.; Goyal, M. Optimizing fast first pass complete reperfusion in acute ischemic stroke—the BADDASS approach (BAlloon guiDe with large bore Distal Access catheter with dual aspiration with Stent-retriever as Standard approach). Expert. Rev. Med. Devices 2019, 16, 955–963. [Google Scholar] [CrossRef]
- Haussen, D.C.; Al-Bayati, A.R.; Grossberg, J.A.; Bouslama, M.; Barreira, C.; Bianchi, N.; Frankel, M.R.; Nogueira, R.G. Longer stent retrievers enhance thrombectomy performance in acute stroke. J. Neurointerv. Surg. 2019, 11, 6–8. [Google Scholar] [CrossRef]
- Maus, V.; Brehm, A.; Tsogkas, I.; Henkel, S.; Psychogios, M.N. Stent retriever placement in embolectomy: The choice of the post-bifurcational trunk influences the first-pass reperfusion result in M1 occlusions. J. Neurointerv. Surg. 2019, 11, 237–240. [Google Scholar] [CrossRef]
- Bernava, G.; Rosi, A.; Boto, J.; Brina, O.; Kulcsar, Z.; Czarnetzki, C.; Carrera, E.; Schaller, K.; Lovblad, K.-O.; Machi, P. Direct thromboaspiration efficacy for mechanical thrombectomy is related to the angle of interaction between the aspiration catheter and the clot. J. Neurointerv. Surg. 2020, 12, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Haussen, D.C.; Rebello, L.C.; Nogueira, R.G. Optimizating clot retrieval in acute stroke: The push and fluff technique for closed-cell stentrievers. Stroke 2015, 46, 2838–2842. [Google Scholar] [CrossRef] [PubMed]
- Fennell, V.S.; Nagesh, S.V.S.; Meess, K.M.; Gutierrez, L.; James, R.H.; Springer, M.E.; Siddiqui, A.H. What to do about fibrin rich ‘tough clots’? Comparing the solitaire stent retriever with a novel geometric clot extractor in an in vitro stroke model. J. Neurointerv. Surg. 2018, 10, 907–910. [Google Scholar] [CrossRef]
- Gupta, R.; Saver, J.L.; Levy, E.I.; Zaidat, O.O.; Yavagal, D.R.; Liebeskind, D.S.; Khaldi, A.; Gross, B.A.; Lang, M.J.; Narayanan, S.; et al. A new class of radially adjustable stentrievers for acute ischemic stroke: Primary results of the multicenter tiger trial. Stroke. 2021, 52, 1534–1544. [Google Scholar] [CrossRef]
- Almandoz, J.E.D.; Kayan, Y.; Young, M.L.; Fease, J.L.; Scholz, J.M.; Milner, A.M.; Hehr, T.H.; Roohani, P.; Mulder, M.; Tarrel, R.M. Comparison of clinical outcomes in patients with acute ischemic strokes treated with mechanical thrombectomy using either Solumbra or ADAPT techniques. J. Neurointerv. Surg. 2016, 8, 1123–1128. [Google Scholar] [CrossRef] [PubMed]
- Massari, F.; Henninger, N.; Lozano, J.D.; Patel, A.; Kuhn, A.L.; Howk, M.; Perras, M.; Brooks, C.; Gounis, M.J.; Kan, P.; et al. ARTS (Aspiration-Retriever Technique for Stroke): Initial clinical experience. Interv. Neuroradiol. 2016, 22, 325–332. [Google Scholar] [CrossRef]
- McTaggart, R.A.; Tung, E.L.; Yaghi, S.; Cutting, S.M.; Hemendinger, M.; Gale, H.I.; Baird, G.L.; Haas, R.A.; Jayaraman, M. Continuous aspiration prior to intracranial vascular embolectomy (CAPTIVE): A technique which improves outcomes. J. Neurointerv. Surg. 2017, 9, 1154–1159. [Google Scholar] [CrossRef] [PubMed]
- Maus, V.; Behme, D.; Kabbasch, C.; Borggrefe, J.; Tsogkas, I.; Nikoubashman, O.; Wiesmann, M.; Knauth, M.; Mpotsaris, A.; Psychogios, M.N. Maximizing first-pass complete reperfusion with SAVE. Clin. Neuroradiol. 2018, 28, 327–338. [Google Scholar] [CrossRef]
- Maegerlein, C.; Berndt, M.T.; Mönch, S.; Kreiser, K.; Boeckh-Behrens, T.; Lehm, M.; Wunderlich, S.; Zimmer, C.; Friedrich, B. Further development of combined techniques using stent retrievers, aspiration catheters and BGC: The PROTECTPLUS Technique. Clin. Neuroradiol. 2020, 30, 59–65. [Google Scholar] [CrossRef]
- Baek, J.-H.; Kim, B.M.; Heo, J.H.; Nam, H.S.; Kim, Y.D.; Park, H.; Bang, O.Y.; Yoo, J.; Kim, D.J.; Jeon, P.; et al. Number of stent retriever passes associated with futile recanalization in acute stroke. Stroke 2018, 49, 2088–2095. [Google Scholar] [CrossRef] [PubMed]
- Labeyrie, M.-A.; Civelli, V.; Reiner, P.; Aymard, A.; Saint-Maurice, J.-P.; Zetchi, A.; Houdart, E. Prevalence and treatment of spontaneous intracranial artery dissections in patients with acute stroke due to intracranial large vessel occlusion. J. Neurointerv. Surg. 2018, 10, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Patro, S.N.; Iancu, D. Dual-stent retrieval for mechanical thrombectomy of refractory clot in acute stroke as a rescue technique. Can. Med Assoc. J. 2017, 189, E634–E637. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Kim, B.M.; Bang, O.Y.; Baek, J.-H.; Heo, J.H.; Nam, H.S.; Kim, Y.D.; Yoo, J.; Kim, D.J.; Jeon, P.; et al. Rescue stenting for failed mechanical thrombectomy in acute ischemic stroke: A multicenter experience. Stroke 2018, 49, 958–964. [Google Scholar] [CrossRef]
- Chen, V.H.; Lee, G.K.; Tan, C.-H.; Leow, A.S.; Tan, Y.-K.; Goh, C.; Gopinathan, A.; Yang, C.; Chan, B.P.; Sharma, V.K.; et al. Intra-arterial adjunctive medications for acute ischemic stroke during mechanical thrombectomy: A meta-analysis. Stroke 2021, 49, 958–964. [Google Scholar] [CrossRef]
- Hofmeister, J.; Bernava, G.; Rosi, A.; Vargas, M.I.; Carrera, E.; Montet, X.; Burgermeister, S.; Poletti, P.-A.; Platon, A.; Lovblad, K.-O.; et al. Clot-based radiomics predict a mechanical thrombectomy strategy for successful recanalization in acute ischemic stroke. Stroke 2020, 51, 2488–2494. [Google Scholar] [CrossRef] [PubMed]
- Bernsen, M.L.E.; Goldhoorn, R.-J.B.; Lingsma, H.F.; Van Oostenbrugge, R.J.; Van Zwam, W.H.; Uyttenboogaart, M.; Roos, Y.B.; Martens, J.M.; Hofmeijer, J.; Dippel, D.W.; et al. Importance of occlusion site for thrombectomy technique in stroke: Comparison between aspiration and stent retriever. Stroke 2021, 52, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Lapergue, B.; Blanc, R.; Gory, B.; Labreuche, J.; Duhamel, A.; Marnat, G.; Saleme, S.; Costalat, V.; Bracard, S.; Desal, H.; et al. Effect of endovascular contact aspiration vs. stent retriever on revascularization in patients with acute ischemic stroke and large vessel occlusion: The ASTER randomized clinical trial. JAMA 2017, 318, 443–452. [Google Scholar] [CrossRef]
- Turk, A.S.; Siddiqui, A.; Fifi, J.T.; De Leacy, R.A.; Fiorella, D.J.; Gu, E.; Levy, E.I.; Snyder, K.V.; Hanel, R.A.; Aghaebrahim, A.; et al. Aspiration thrombectomy versus stent retriever thrombectomy as first-line approach for large vessel occlusion (COMPASS): A multicentre, randomised, open label, blinded outcome, non-inferiority trial. Lancet 2019, 393, 998–1008. [Google Scholar] [CrossRef]
- Alawieh, A.M.; Eid, M.; Anadani, M.; Sattur, M.; Maier, I.L.; Feng, W.; Goyal, N.; Starke, R.M.; Rai, A.; Fargen, K.M.; et al. Thrombectomy technique predicts outcome in posterior circulation stroke-insights from the STAR collaboration. Neurosurgery 2020, 87, 982–991. [Google Scholar] [CrossRef]
- Ye, G.; Lu, J.; Qi, P.; Yin, X.; Wang, L.; Wang, D. Firstline a direct aspiration first pass technique versus firstline stent retriever for acute basilar artery occlusion: A systematic review and meta-analysis. J. Neurointerv. Surg. 2019, 11, 740–746. [Google Scholar] [CrossRef]
- Barchetti, G.; Cagnazzo, F.; Raz, E.; Barbagallo, G.; Toccaceli, G.; Peschillo, S. Mechanical thrombectomy of distal occlusions using a direct aspiration first pass technique compared with new generation of Mini-0.017 microcatheter compatible-stent retrievers: A meta-analysis. World Neurosurg. 2020, 134, 111–119. [Google Scholar] [CrossRef]
- Schramm, P.; Navia, P.; Papa, R.; Zamarro, J.; Tomasello, A.; Weber, W.; Fiehler, J.; Michel, P.; Pereira, V.M.; Krings, T.; et al. ADAPT technique with ACE68 and ACE64 reperfusion catheters in ischemic stroke treatment: Results from the PROMISE study. J. Neurointerv. Surg. 2019, 11, 226–231. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, D.; Griffin, E.; Doyle, K.; Power, S.; Brennan, P.; Sheehan, M.; O’Hare, A.; Looby, S.; Santos, A.D.S.; Rossi, R.; et al. A standardized aspiration-first approach for thrombectomy to increase speed and improve recanalization rates. Am. J. Neuroradiol. 2019, 40, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Chivot, C.; Renier, J.; Deramond, H.; Bouzerar, R.; Yzet, T. Direct aspiration for thrombectomy in ischemic stroke: Impact of dwell time. Interv. Neuroradiol. 2020, 26, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Alawieh, A.; Pierce, A.K.; Vargas, J.; Turk, A.S.; Turner, R.D.; Chaudry, M.I.; Spiotta, A.M. The golden 35 min of stroke intervention with ADAPT: Effect of thrombectomy procedural time in acute ischemic stroke on outcome. J. Neurointerv. Surg. 2018, 10, 213–220. [Google Scholar] [CrossRef]
- Keulers, A.; Nikoubashman, O.; Mpotsaris, A.; Wilson, S.D.; Wiesmann, M. Preventing vessel perforations in endovascular thrombectomy: Feasibility and safety of passing the clot with a microcatheter without microwire: The wireless microcatheter technique. J. Neurointerv. Surg. 2019, 11, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Zeleňák, K.; Vorčák, M.; Sykora, J.; Trabalková, Z.; Zeleňáková, J.; Kantorová, E.; Nosáľ, V. Management of ruptured hidden mirror intracranial aneurysm during mechanical thrombectomy. Interdiscip. Neurosurg. 2019, 17, 60–63. [Google Scholar] [CrossRef]
- Nozaki, T.; Noda, M.; Ishibashi, T.; Morita, A. Ruptured hidden intracranial aneurysm during mechanical thrombectomy: A case report. Surg. Neurol. Int. 2020, 11, 446. [Google Scholar] [CrossRef]
- Ryska, P.; Lojik, M.; Kocer, N.; Zelenak, K.; Cesak, T.; Cimflova, P.; Bhorkar, A.M.; Vitkova, E.; Krajina, A. Mechanical thrombectomy performed in thrombosed fusiform aneurysm after surgery for craniopharyngioma in adult: Case report. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2021. [Google Scholar] [CrossRef] [PubMed]
- Patel, T.M.; Shah, S.C.; Pancholy, S.B. Long distance tele-robotic-assisted percutaneous coronary intervention: A report of first-in-human experience. EClinicalMedicine 2019, 14, 53–58. [Google Scholar] [CrossRef]
- Kim, J.; Chae, M.; Chang, H.J.; Kim, Y.A.; Park, E. Predicting cardiac arrest and respiratory failure using feasible artificial intelligence with simple trajectories of patient data. J. Clin. Med. 2019, 8, 1336. [Google Scholar] [CrossRef]
- Zhang, X.; Yue, Z.; Wang, J. Robotics in lower-limb rehabilitation after stroke. Behav. Neurol. 2017, 2017, 3731802. [Google Scholar] [CrossRef]
- Weber, L.M.; Stein, J. The use of robots in stroke rehabilitation: A narrative review. NeuroRehabilitation 2018, 43, 99–110. [Google Scholar] [CrossRef]
- Lin, D.J.; Finklestein, S.P.; Cramer, S.C. New directions in treatments targeting stroke recovery. Stroke 2018, 49, 3107–3114. [Google Scholar] [CrossRef]
- Sofia–Hanson Robotics. Available online: https://www.hansonrobotics.com/sophia/ (accessed on 19 March 2021).
- Zhang, Y.; Fan, D.; Ji, H.; Qiao, S.; Li, X. Treatment adherence and secondary prevention of ischemic stroke among discharged patients using mobile phone- and wechat-based improvement services: Cohort study. JMIR mHealth uHealth 2020, 8, e16496. [Google Scholar] [CrossRef] [PubMed]
- European Society of Radiology (ESR). What the radiologist should know about artificial intelligence—An ESR white paper. Insights Imaging 2019, 10, 44. [Google Scholar] [CrossRef] [PubMed]
- Tang, A.; Tam, R.; Cadrin-Chênevert, A.; Guest, W.; Chong, J.; Barfett, J.; Chepelev, L.; Cairns, R.; Mitchell, J.R.; Cicero, M.D.; et al. Canadian Association of Radiologists White Paper on Artificial Intelligence in Radiology. Can. Assoc. Radiol. J. 2018, 69, 120–135. [Google Scholar] [CrossRef]
- Parker, W.; Jaremko, J.L.; Cicero, M.; Azar, M.; El-Emam, K.; Gray, B.G.; Hurrell, C.; Lavoie-Cardinal, F.; Desjardins, B.; Lum, A.; et al. Canadian Association of Radiologists White Paper on De-Identification of Medical Imaging: Part 1, General Principles. Can. Assoc. Radiol. J. 2021, 72, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.; Jaremko, J.L.; Cicero, M.; Azar, M.; El-Emam, K.; Gray, B.G.; Hurrell, C.; Lavoie-Cardinal, F.; Desjardins, B.; Lum, A.; et al. Canadian Association of Radiologists White Paper on De-identification of medical imaging: Part 2, practical considerations. Can. Assoc. Radiol. J. 2021, 72, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Geis, J.R.; Brady, A.P.; Wu, C.C.; Spencer, J.; Ranschaert, E.; Jaremko, J.L.; Langer, S.G.; Kitts, A.B.; Birch, J.; Shields, W.F.; et al. Ethics of artificial intelligence in radiology: Summary of the joint European and North American Multisociety Statement. Radiology 2019, 293, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Brady, A.P.; Neri, E. Artificial Intelligence in Radiology-Ethical Considerations. Diagnostics 2020, 10, 231. [Google Scholar] [CrossRef] [PubMed]
- Lindqwister, A.L.; Hassanpour, S.; Lewis, P.J.; Sin, J.M. AI-RADS: An artificial intelligence curriculum for residents. Acad. Radiol. 2020, 1076. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeleňák, K.; Krajina, A.; Meyer, L.; Fiehler, J.; ESMINT Artificial Intelligence and Robotics Ad hoc Committee; Behme, D.; Bulja, D.; Caroff, J.; Chotai, A.A.; Da Ros, V.; et al. How to Improve the Management of Acute Ischemic Stroke by Modern Technologies, Artificial Intelligence, and New Treatment Methods. Life 2021, 11, 488. https://doi.org/10.3390/life11060488
Zeleňák K, Krajina A, Meyer L, Fiehler J, ESMINT Artificial Intelligence and Robotics Ad hoc Committee, Behme D, Bulja D, Caroff J, Chotai AA, Da Ros V, et al. How to Improve the Management of Acute Ischemic Stroke by Modern Technologies, Artificial Intelligence, and New Treatment Methods. Life. 2021; 11(6):488. https://doi.org/10.3390/life11060488
Chicago/Turabian StyleZeleňák, Kamil, Antonín Krajina, Lukas Meyer, Jens Fiehler, ESMINT Artificial Intelligence and Robotics Ad hoc Committee, Daniel Behme, Deniz Bulja, Jildaz Caroff, Amar Ajay Chotai, Valerio Da Ros, and et al. 2021. "How to Improve the Management of Acute Ischemic Stroke by Modern Technologies, Artificial Intelligence, and New Treatment Methods" Life 11, no. 6: 488. https://doi.org/10.3390/life11060488
APA StyleZeleňák, K., Krajina, A., Meyer, L., Fiehler, J., ESMINT Artificial Intelligence and Robotics Ad hoc Committee, Behme, D., Bulja, D., Caroff, J., Chotai, A. A., Da Ros, V., Gentric, J.-C., Hofmeister, J., Kass-Hout, O., Kocatürk, Ö., Lynch, J., Pearson, E., & Vukasinovic, I. (2021). How to Improve the Management of Acute Ischemic Stroke by Modern Technologies, Artificial Intelligence, and New Treatment Methods. Life, 11(6), 488. https://doi.org/10.3390/life11060488






