Natural Sulfurs Inhibit LPS-Induced Inflammatory Responses through NF-κB Signaling in CCD-986Sk Skin Fibroblasts
Abstract
1. Introduction
2. Material and Methods
2.1. Antibodies and Cell Culture Reagents
2.2. Cell Culture and Treatment
2.3. Cell Viability Assay
2.4. Real-Time qPCR
2.5. Western Blotting
2.6. Flow Cytometry Analysis for TLR4 and Cellular ROS Detection
2.7. Comet Assay
2.8. ELISA
2.9. Statistical Analyses
3. Results
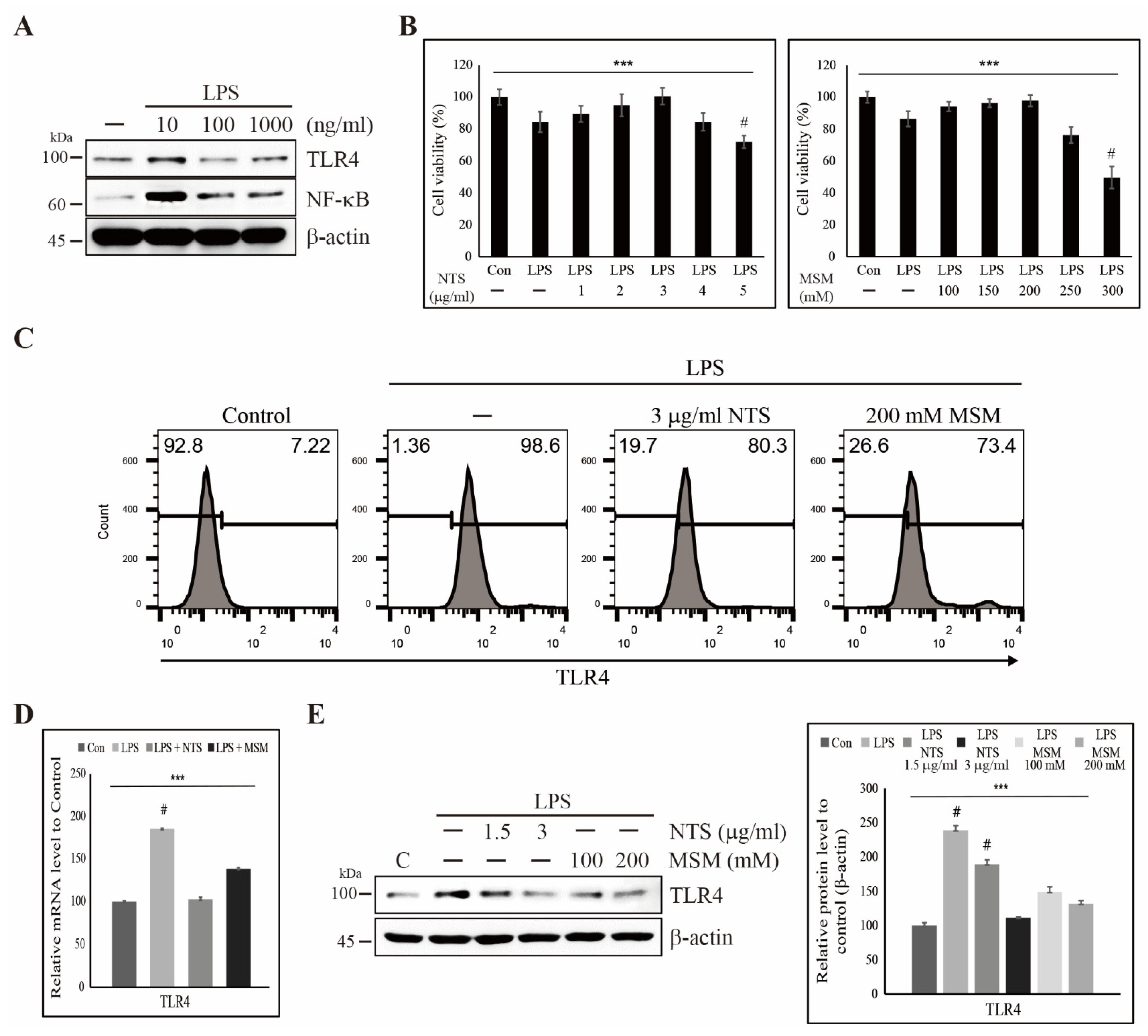
3.1. Sulfur Compound-Induced Cytoprotection Against LPS-Induced Cell Death and Inhibition of TLR4 Expression
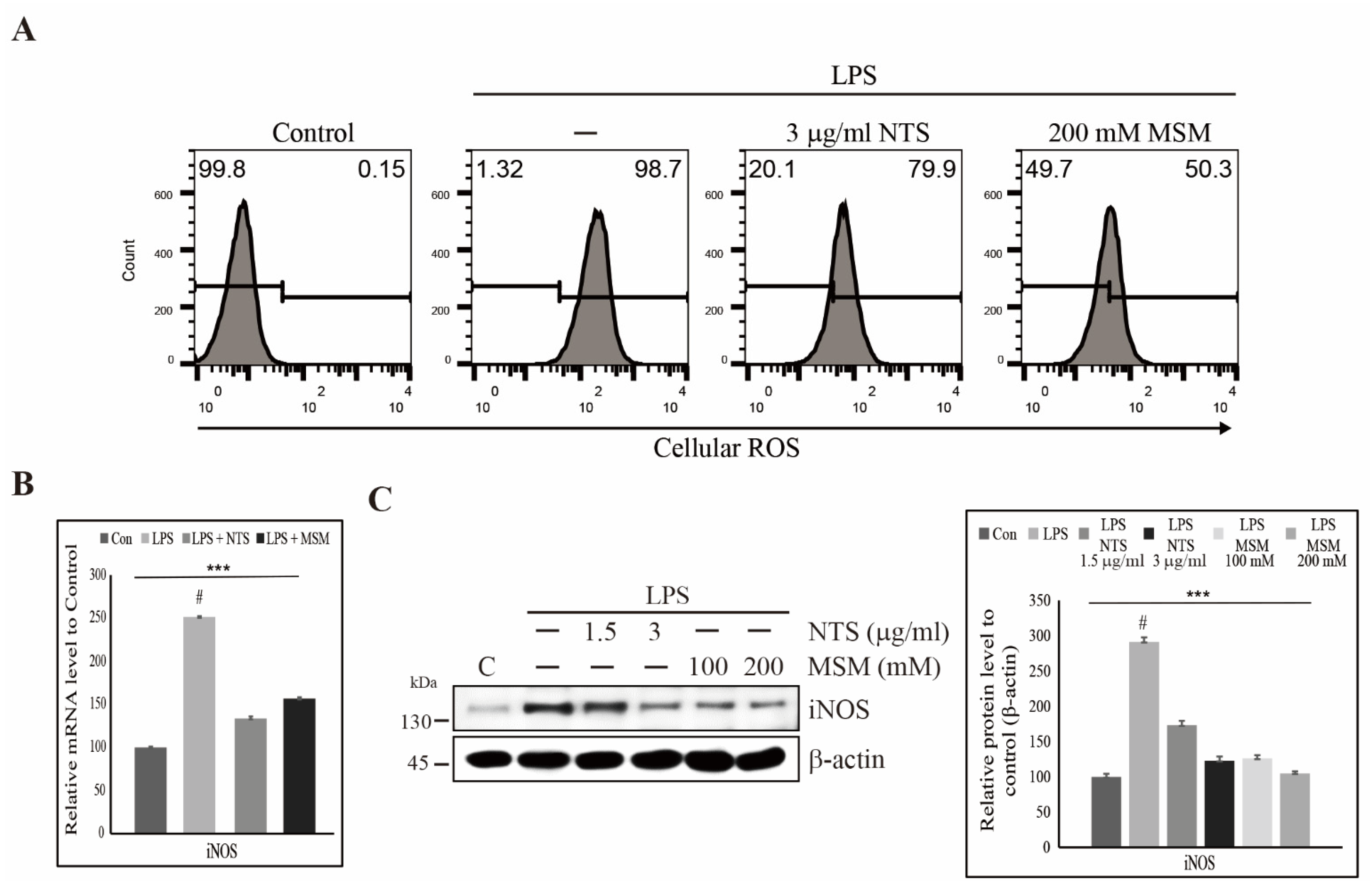
3.2. Suppression of LPS-Induced ROS by Sulfur Compounds in Human Skin Cells
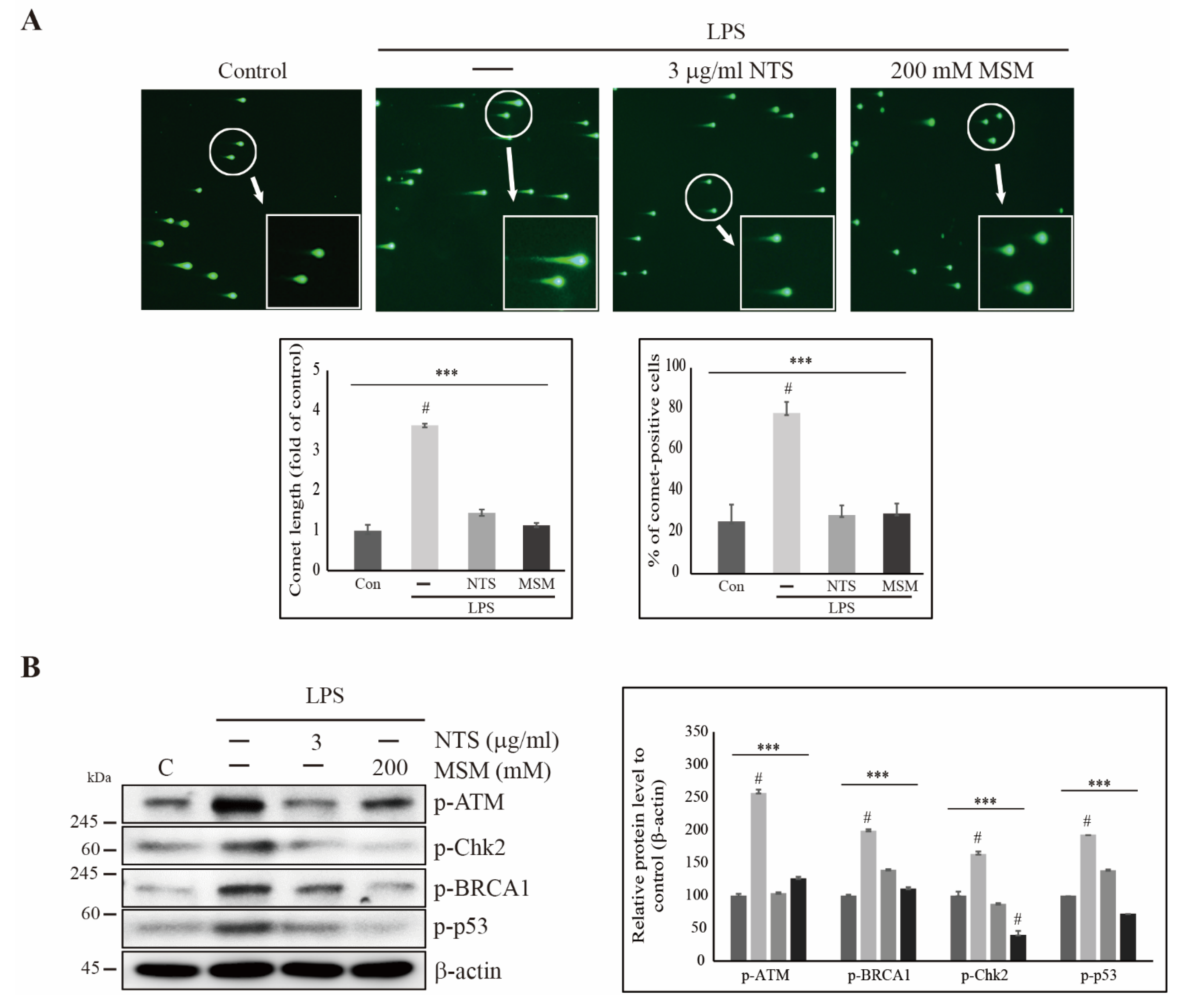
3.3. Sulfur Compounds Inhibited LPS-Induced DNA Damage in CCD-986Sk Cells
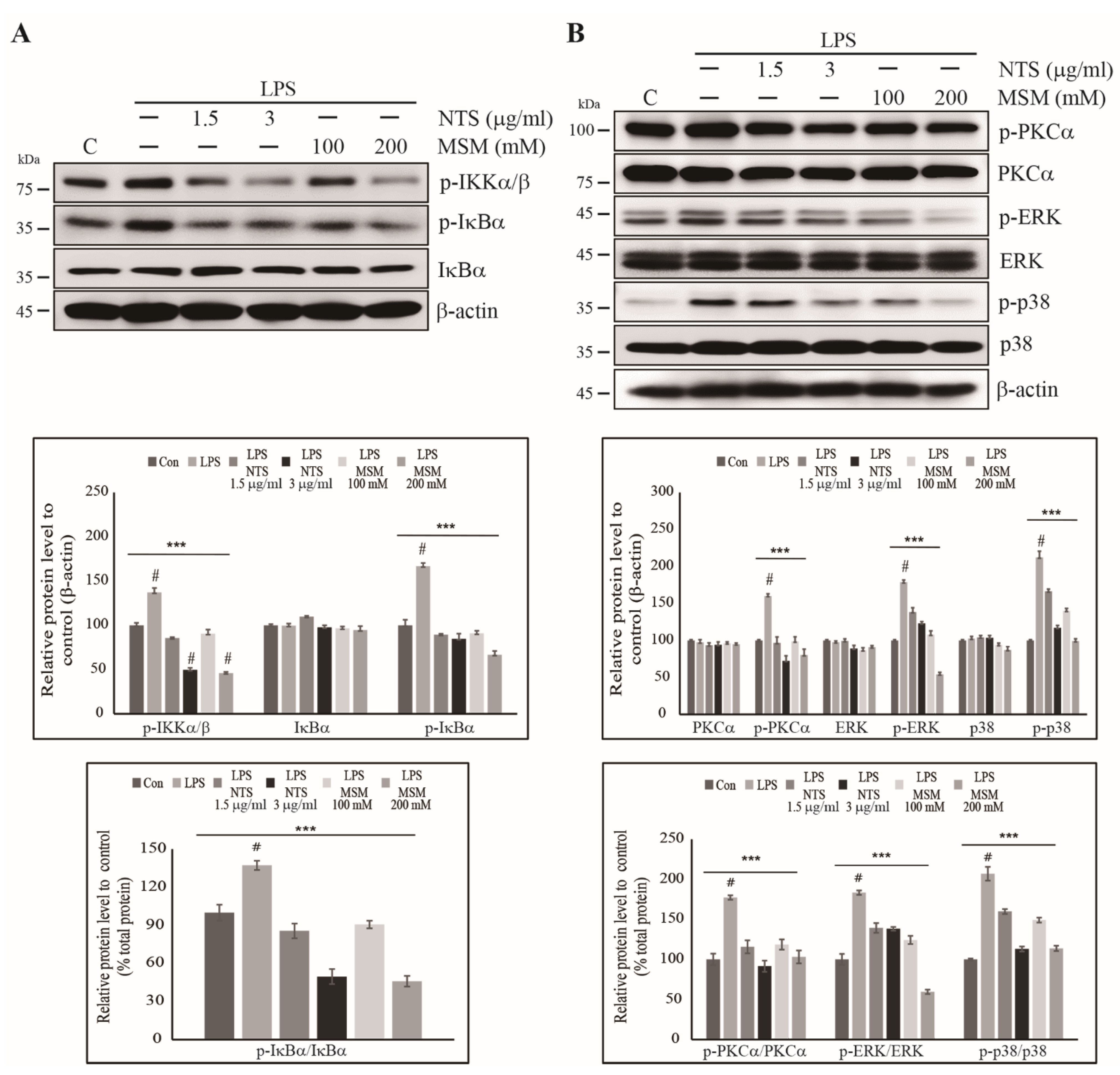
3.4. Inhibition of LPS-Induced Canonical Pathway and PKC-Mediated Upstream Targets of NF-κB by Sulfur Compounds
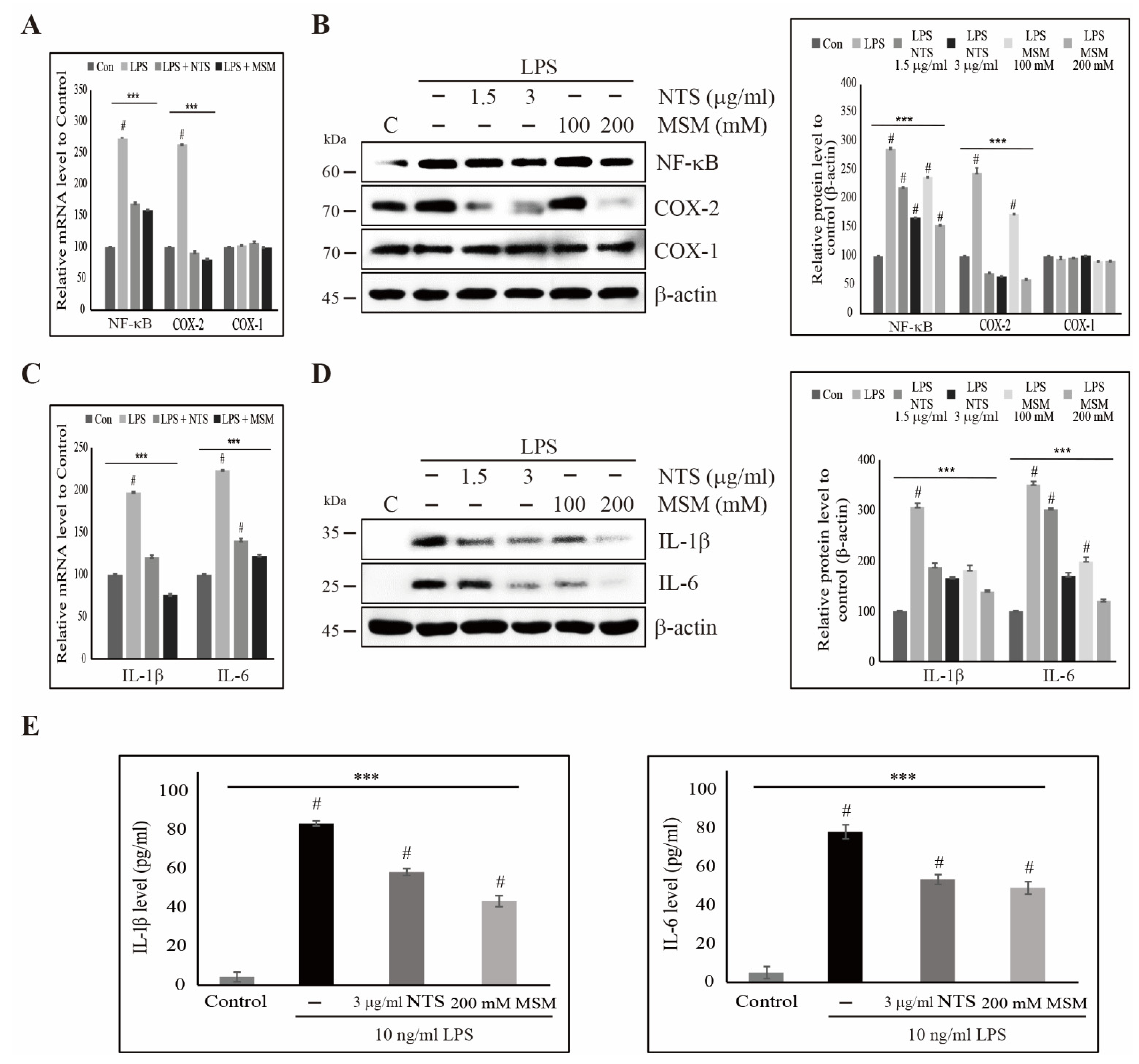
3.5. Sulfur Compounds Inhibited LPS-Induced COX-2 and Proinflammatory Cytokines
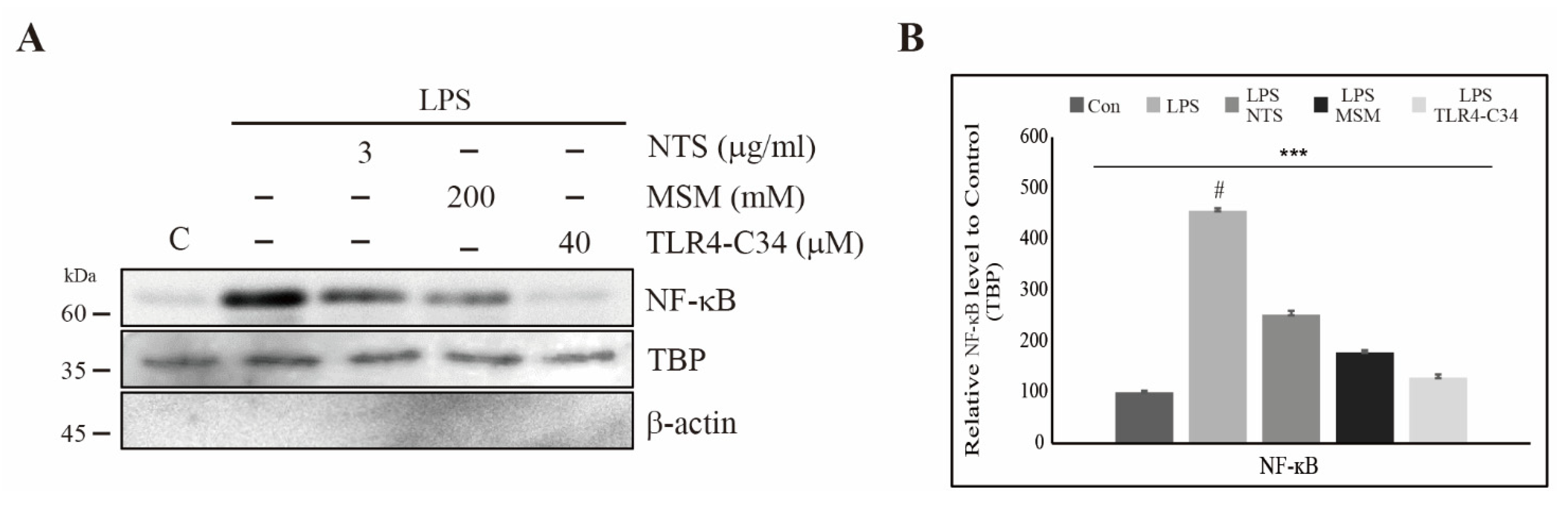
3.6. Downregulation of LPS-Induced Nuclear Translocation of NF-κB by Sulfur Compounds in CCD-986Sk Cells
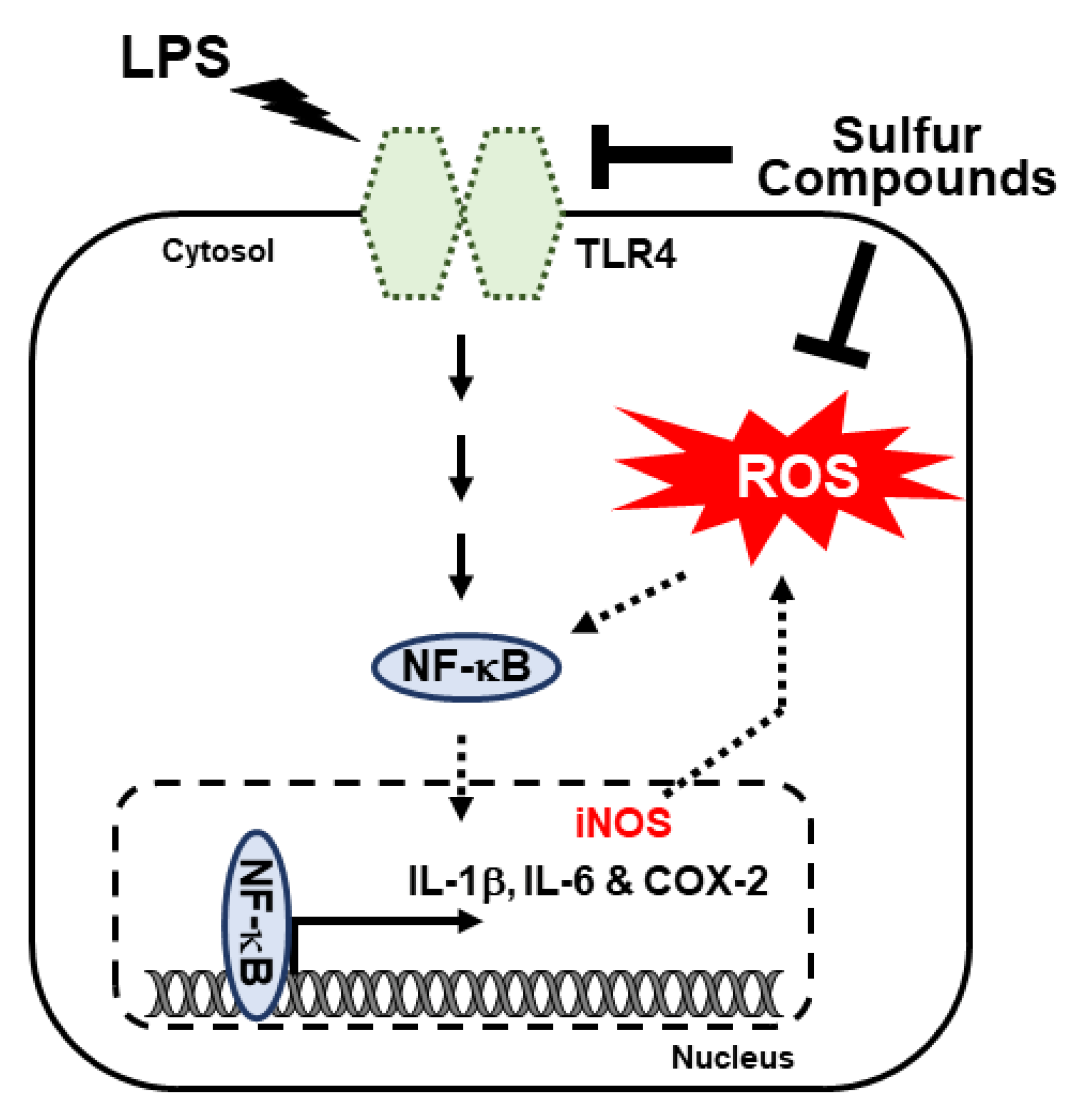
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gvirtz, R.; Ogen-Shtern, N.; Cohen, G. Kinetic cytokine secretion profile of lps-induced inflammation in the human skin organ culture. Pharmaceutics 2020, 12, 299. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, K.A.U.; Fuchs, E. Skin and its regenerative powers: An alliance between stem cells and their niche. Dev. Cell 2017, 43, 387–401. [Google Scholar] [CrossRef] [PubMed]
- Jordana, M.; Sarnstrand, B.; Sime, P.J.; Ramis, I. Immune-inflammatory functions of fibroblasts. Eur. Respir. J. 1994, 7, 2212–2222. [Google Scholar] [CrossRef] [PubMed]
- Palm, E.; Khalaf, H.; Bengtsson, T. Porphyromonas gingivalis downregulates the immune response of fibroblasts. BMC Microbiol. 2013, 13, 155. [Google Scholar] [CrossRef]
- Xi, X.; McMillan, D.H.; Lehmann, G.M.; Sime, P.J.; Libby, R.T.; Huxlin, K.R.; Feldon, S.E.; Phipps, R.P. Ocular fibroblast diversity: Implications for inflammation and ocular wound healing. Invest. Ophthalmol. Vis. Sci. 2011, 52, 4859–4865. [Google Scholar] [CrossRef]
- Bautista-Hernandez, L.A.; Gomez-Olivares, J.L.; Buentello-Volante, B.; Bautista-de Lucio, V.M. Fibroblasts: The unknown sentinels eliciting immune responses against microorganisms. Eur. J. Microbiol. Immunol. 2017, 7, 151–157. [Google Scholar] [CrossRef]
- Yucel, G.; Zhao, Z.; El-Battrawy, I.; Lan, H.; Lang, S.; Li, X.; Buljubasic, F.; Zimmermann, W.H.; Cyganek, L.; Utikal, J.; et al. Lipopolysaccharides induced inflammatory responses and electrophysiological dysfunctions in human-induced pluripotent stem cell derived cardiomyocytes. Sci. Rep. 2017, 7, 2935. [Google Scholar] [CrossRef]
- Piktel, E.; Wnorowska, U.; Ciesluk, M.; Deptula, P.; Pogoda, K.; Misztalewska-Turkowicz, I.; Paprocka, P.; Niemirowicz-Laskowska, K.; Wilczewska, A.Z.; Janmey, P.A.; et al. Inhibition of inflammatory response in human keratinocytes by magnetic nanoparticles functionalized with PBP10 peptide derived from the PIP2-binding site of human plasma gelsolin. J. Nanobiotechnology 2019, 17, 22. [Google Scholar] [CrossRef] [PubMed]
- Rafi, M.M.; Yadav, P.N.; Rossi, A.O. Glucosamine inhibits LPS-induced COX-2 and iNOS expression in mouse macrophage cells (RAW 264.7) by inhibition of p38-MAP kinase and transcription factor NF-kappaB. Mol. Nutr. Food Res. 2007, 51, 587–593. [Google Scholar] [CrossRef]
- Park, K.; Lee, J.H.; Cho, H.C.; Cho, S.Y.; Cho, J.W. Down-regulation of IL-6, IL-8, TNF-alpha and IL-1beta by glucosamine in HaCaT cells, but not in the presence of TNF-alpha. Oncol. Lett. 2010, 1, 289–292. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Gallo, R.L. Toll-like receptors in skin infections and inflammatory diseases. Infect. Disord Drug Targets 2008, 8, 144–155. [Google Scholar] [CrossRef]
- Uematsu, S.; Akira, S. Toll-like receptors and innate immunity. J. Mol. Med. 2006, 84, 712–725. [Google Scholar] [CrossRef]
- Beutler, B. Inferences, questions and possibilities in Toll-like receptor signalling. Nature 2004, 430, 257–263. [Google Scholar] [CrossRef]
- Chait, A.; Bornfeldt, K.E. Diabetes and atherosclerosis: Is there a role for hyperglycemia? J. Lipid Res. 2009, 50, S335–S339. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.G.; Hayden, M.S.; Ghosh, S. NF-kappaB, inflammation, and metabolic disease. Cell Metab. 2011, 13, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Bjorkbacka, H.; Kunjathoor, V.V.; Moore, K.J.; Koehn, S.; Ordija, C.M.; Lee, M.A.; Means, T.; Halmen, K.; Luster, A.D.; Golenbock, D.T.; et al. Reduced atherosclerosis in MyD88-null mice links elevated serum cholesterol levels to activation of innate immunity signaling pathways. Nat. Med. 2004, 10, 416–421. [Google Scholar] [CrossRef]
- Karin, M.; Delhase, M. The I kappa B kinase (IKK) and NF-kappa B: Key elements of proinflammatory signalling. Semin Immunol 2000, 12, 85–98. [Google Scholar] [CrossRef]
- Grech, A.P.; Amesbury, M.; Chan, T.; Gardam, S.; Basten, A.; Brink, R. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-kappaB activation in mature B cells. Immunity 2004, 21, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.W.; Chu, W.; Hu, Y.; Delhase, M.; Deerinck, T.; Ellisman, M.; Johnson, R.; Karin, M. The IKKbeta subunit of IkappaB kinase (IKK) is essential for nuclear factor kappaB activation and prevention of apoptosis. J. Exp. Med. 1999, 189, 1839–1845. [Google Scholar] [CrossRef] [PubMed]
- Beg, A.A.; Sha, W.C.; Bronson, R.T.; Baltimore, D. Constitutive NF-kappa B activation, enhanced granulopoiesis, and neonatal lethality in I kappa B alpha-deficient mice. Genes. Dev. 1995, 9, 2736–2746. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.C. The noncanonical NF-kappaB pathway. Immunol Rev. 2012, 246, 125–140. [Google Scholar] [CrossRef]
- Jo, E.S.; Sp, N.; Kang, D.Y.; Rugamba, A.; Kim, I.H.; Bae, S.W.; Liu, Q.; Jang, K.J.; Yang, Y.M. Sulfur Compounds Inhibit High Glucose-Induced Inflammation by Regulating NF-kappaB Signaling in Human Monocytes. Molecules 2020, 25, 2342. [Google Scholar] [CrossRef]
- Yamakawa, T.; Eguchi, S.; Matsumoto, T.; Yamakawa, Y.; Numaguchi, K.; Miyata, I.; Reynolds, C.M.; Motley, E.D.; Inagami, T. Intracellular signaling in rat cultured vascular smooth muscle cells: Roles of nuclear factor-kappaB and p38 mitogen-activated protein kinase on tumor necrosis factor-alpha production. Endocrinology 1999, 140, 3562–3572. [Google Scholar] [CrossRef]
- Olson, C.M.; Hedrick, M.N.; Izadi, H.; Bates, T.C.; Olivera, E.R.; Anguita, J. p38 mitogen-activated protein kinase controls NF-kappaB transcriptional activation and tumor necrosis factor alpha production through RelA phosphorylation mediated by mitogen- and stress-activated protein kinase 1 in response to Borrelia burgdorferi antigens. Infect. Immun. 2007, 75, 270–277. [Google Scholar] [CrossRef] [PubMed]
- van der Merwe, M.; Bloomer, R.J. The Influence of Methylsulfonylmethane on Inflammation-Associated Cytokine Release before and following Strenuous Exercise. J. Sports Med. 2016, 2016, 7498359. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.; Surh, J. Influence of Sulfur Fertilization on the Antioxidant Activities of Onion Juices Prepared by Thermal Treatment. Prev. Nutr. Food Sci. 2016, 21, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Caron, J.M.; Bannon, M.; Rosshirt, L.; Luis, J.; Monteagudo, L.; Caron, J.M.; Sternstein, G.M. Methyl sulfone induces loss of metastatic properties and reemergence of normal phenotypes in a metastatic cloudman S-91 (M3) murine melanoma cell line. PLoS ONE 2010, 5, e11788. [Google Scholar] [CrossRef]
- P, N.S.; Darvin, P.; Yoo, Y.B.; Joung, Y.H.; Kang, D.Y.; Kim, D.N.; Hwang, T.S.; Kim, S.Y.; Kim, W.S.; Lee, H.K.; et al. The combination of methylsulfonylmethane and tamoxifen inhibits the Jak2/STAT5b pathway and synergistically inhibits tumor growth and metastasis in ER-positive breast cancer xenografts. BMC Cancer 2015, 15, 474. [Google Scholar] [CrossRef]
- Preetha, N.S.; Kang, D.Y.; Darvin, P.; Kim, D.N.; Joung, Y.H.; Kim, S.Y.; Cho, K.H.; Do, C.H.; Do Park, K.; Lee, J.H.; et al. Induction of in vitro ketosis condition and suppression using methylsulfonylmethane by altering ANGPTL3 expression through STAT5b signaling mechanism. Anim. Cells Syst. 2015, 19, 30–38. [Google Scholar] [CrossRef]
- Miller, L.E. Methylsulfonylmethane decreases inflammatory response to tumor necrosis factor-alpha in cardiac cells. Am. J. Cardiovasc. Dis. 2018, 8, 31–38. [Google Scholar] [PubMed]
- Kang, D.Y.; Darvin, P.; Yoo, Y.B.; Joung, Y.H.; Sp, N.; Byun, H.J.; Yang, Y.M. Methylsulfonylmethane inhibits HER2 expression through STAT5b in breast cancer cells. Int. J. Oncol. 2016, 48, 836–842. [Google Scholar] [CrossRef] [PubMed]
- P, N.S.; Kang, D.Y.; Kim, B.J.; Joung, Y.H.; Darvin, P.; Byun, H.J.; Kim, J.G.; Park, J.U.; Yang, Y.M. Methylsulfonylmethane Induces G1 Arrest and Mitochondrial Apoptosis in YD-38 Gingival Cancer Cells. Anticancer. Res. 2017, 37, 1637–1646. [Google Scholar] [CrossRef]
- Lim, C.I.; Choe, H.S.; Kang, C.; Lee, B.K.; Ryu, K.S. Effects of dietary organic sulfur on performance, egg quality and cell-mediated immune response of laying hens. Korean, J. Poult Sci. 2018, 45, 97–107. [Google Scholar] [CrossRef][Green Version]
- Lee, J.S.; Kwon, J.K.; Han, S.H.; An, I.J.; Kim, S.J.; Lee, S.H.; Park, Y.S.; Park, B.K.; Kim, B.S.; Kim, S.K.; et al. Toxicity study of detoxication sulphur at 3 months post-treatment in rats. J. Fd. Hyg. Saf. 2010, 25, 263–268. [Google Scholar]
- Kang, D.Y.; Sp, N.; Jo, E.S.; Kim, H.D.; Kim, I.H.; Bae, S.W.; Jang, K.J.; Yang, Y.M. Nontoxic sulfur enhances growth hormone signaling through the JAK2/STAT5b/IGF1 pathway in C2C12 cells. Int. J. Mol. Med. 2020, 45, 931–938. [Google Scholar] [CrossRef]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Lee, D.Y.; Li, H.; Lim, H.J.; Lee, H.J.; Jeon, R.; Ryu, J.H. Anti-inflammatory activity of sulfur-containing compounds from garlic. J. Med. Food 2012, 15, 992–999. [Google Scholar] [CrossRef] [PubMed]
- Butawan, M.; Benjamin, R.L.; Bloomer, R.J. Methylsulfonylmethane: Applications and Safety of a Novel Dietary Supplement. Nutrients 2017, 9, 290. [Google Scholar] [CrossRef] [PubMed]
- Sp, N.; Kang, D.Y.; Kim, D.H.; Lee, H.G.; Park, Y.M.; Kim, I.H.; Lee, H.K.; Cho, B.W.; Jang, K.J.; Yang, Y.M. Methylsulfonylmethane inhibits cortisol-induced stress through p53-mediated SDHA/HPRT1 expression in racehorse skeletal muscle cells: A primary step against exercise stress. Exp. Ther Med. 2020, 19, 214–222. [Google Scholar] [CrossRef]
- Morton, J.I.; Siegel, B.V. Effects of oral dimethyl sulfoxide and dimethyl sulfone on murine autoimmune lymphoproliferative disease. Proc. Soc. Exp. Biol. Med. 1986, 183, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Joung, Y.H.; Darvin, P.; Kang, D.Y.; Sp, N.; Byun, H.J.; Lee, C.H.; Lee, H.K.; Yang, Y.M. Methylsulfonylmethane inhibits RANKL-induced osteoclastogenesis in BMMs by suppressing NF-kappaB and STAT3 activities. PLoS ONE 2016, 11, e0159891. [Google Scholar] [CrossRef]
- Yu, B.; Li, Q.; Zhou, M. LPSinduced upregulation of the TLR4 signaling pathway inhibits osteogenic differentiation of human periodontal ligament stem cells under inflammatory conditions. Int. J. Mol. Med. 2019, 43, 2341–2351. [Google Scholar] [CrossRef]
- Ngkelo, A.; Meja, K.; Yeadon, M.; Adcock, I.; Kirkham, P.A. LPS induced inflammatory responses in human peripheral blood mononuclear cells is mediated through NOX4 and Gialpha dependent PI-3kinase signalling. J. Inflamm. 2012, 9, 1. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, C.; Shan, S.; Liu, X.; Jiang, Z.; Ren, T. TLR4/ROS/miRNA-21 pathway underlies lipopolysaccharide instructed primary tumor outgrowth in lung cancer patients. Oncotarget 2016, 7, 42172–42182. [Google Scholar] [CrossRef]
- Wu, F.; Tyml, K.; Wilson, J.X. iNOS expression requires NADPH oxidase-dependent redox signaling in microvascular endothelial cells. J. Cell Physiol. 2008, 217, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Qiao, W.; Huang, Y.; Bian, Z.; Sun, X.; Wang, X.; Gao, Q.; Peng, Y.; Meng, L. Lipopolysaccharide-induced DNA damage response activates nuclear factor kappaB signalling pathway via GATA4 in dental pulp cells. Int. Endod. J. 2019, 52, 1704–1715. [Google Scholar] [CrossRef] [PubMed]
- Xiang, P.; Chen, T.; Mou, Y.; Wu, H.; Xie, P.; Lu, G.; Gong, X.; Hu, Q.; Zhang, Y.; Ji, H. NZ suppresses TLR4/NF-kappaB signalings and NLRP3 inflammasome activation in LPS-induced RAW264.7 macrophages. Inflamm. Res. 2015, 64, 799–808. [Google Scholar] [CrossRef]
- Zusso, M.; Lunardi, V.; Franceschini, D.; Pagetta, A.; Lo, R.; Stifani, S.; Frigo, A.C.; Giusti, P.; Moro, S. Ciprofloxacin and levofloxacin attenuate microglia inflammatory response via TLR4/NF-kB pathway. J. Neuroinflamm. 2019, 16, 148. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.C. The non-canonical NF-kappaB pathway in immunity and inflammation. Nat. Rev. Immunol. 2017, 17, 545–558. [Google Scholar] [CrossRef]
- Dasu, M.R.; Devaraj, S.; Jialal, I. High glucose induces IL-1beta expression in human monocytes: Mechanistic insights. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E337–E346. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Shen, S.; Ma, Y.; Kim, J.K.; Rodriguez-Agudo, D.; Heuman, D.M.; Hylemon, P.B.; Pandak, W.M.; Ren, S. 25-Hydroxycholesterol-3-sulfate attenuates inflammatory response via PPARgamma signaling in human THP-1 macrophages. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E788–E799. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Surh, Y.J. Nrf2 as a novel molecular target for chemoprevention. Cancer Lett. 2005, 224, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.W.; Kim, S.C.; Kwak, T.W.; Lee, J.R.; Jo, M.J.; Ahn, Y.T.; Kim, J.M.; An, W.G. Anti-inflammatory effects of bangpungtongsung-san, a traditional herbal prescription. Evid. Based Complement. Altern. Med. 2012, 2012, 892943. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sp, N.; Kang, D.Y.; Kim, H.D.; Rugamba, A.; Jo, E.S.; Park, J.-C.; Bae, S.W.; Lee, J.-M.; Jang, K.-J. Natural Sulfurs Inhibit LPS-Induced Inflammatory Responses through NF-κB Signaling in CCD-986Sk Skin Fibroblasts. Life 2021, 11, 427. https://doi.org/10.3390/life11050427
Sp N, Kang DY, Kim HD, Rugamba A, Jo ES, Park J-C, Bae SW, Lee J-M, Jang K-J. Natural Sulfurs Inhibit LPS-Induced Inflammatory Responses through NF-κB Signaling in CCD-986Sk Skin Fibroblasts. Life. 2021; 11(5):427. https://doi.org/10.3390/life11050427
Chicago/Turabian StyleSp, Nipin, Dong Young Kang, Hyoung Do Kim, Alexis Rugamba, Eun Seong Jo, Jong-Chan Park, Se Won Bae, Jin-Moo Lee, and Kyoung-Jin Jang. 2021. "Natural Sulfurs Inhibit LPS-Induced Inflammatory Responses through NF-κB Signaling in CCD-986Sk Skin Fibroblasts" Life 11, no. 5: 427. https://doi.org/10.3390/life11050427
APA StyleSp, N., Kang, D. Y., Kim, H. D., Rugamba, A., Jo, E. S., Park, J.-C., Bae, S. W., Lee, J.-M., & Jang, K.-J. (2021). Natural Sulfurs Inhibit LPS-Induced Inflammatory Responses through NF-κB Signaling in CCD-986Sk Skin Fibroblasts. Life, 11(5), 427. https://doi.org/10.3390/life11050427







