Physiological Doses of Hydroxytyrosol Modulate Gene Expression in Skeletal Muscle of Exercised Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Hydroxytyrosol Treatment
2.3. Quantitative Real-Time (qRT)-PCR
2.4. Western Blotting
2.5. Statistical Analysis
3. Results
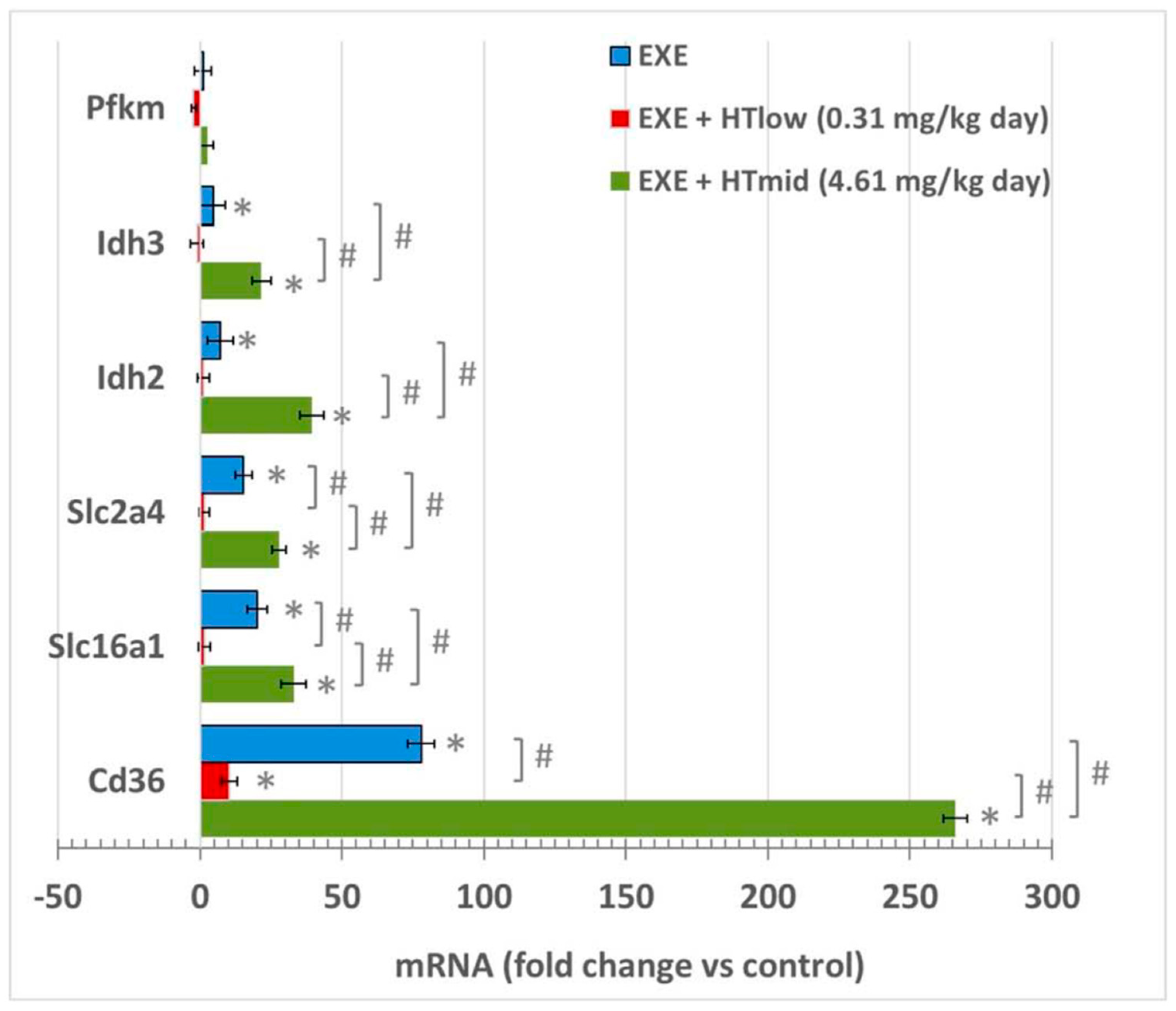
3.1. mRNA Expression in Soleus Muscles
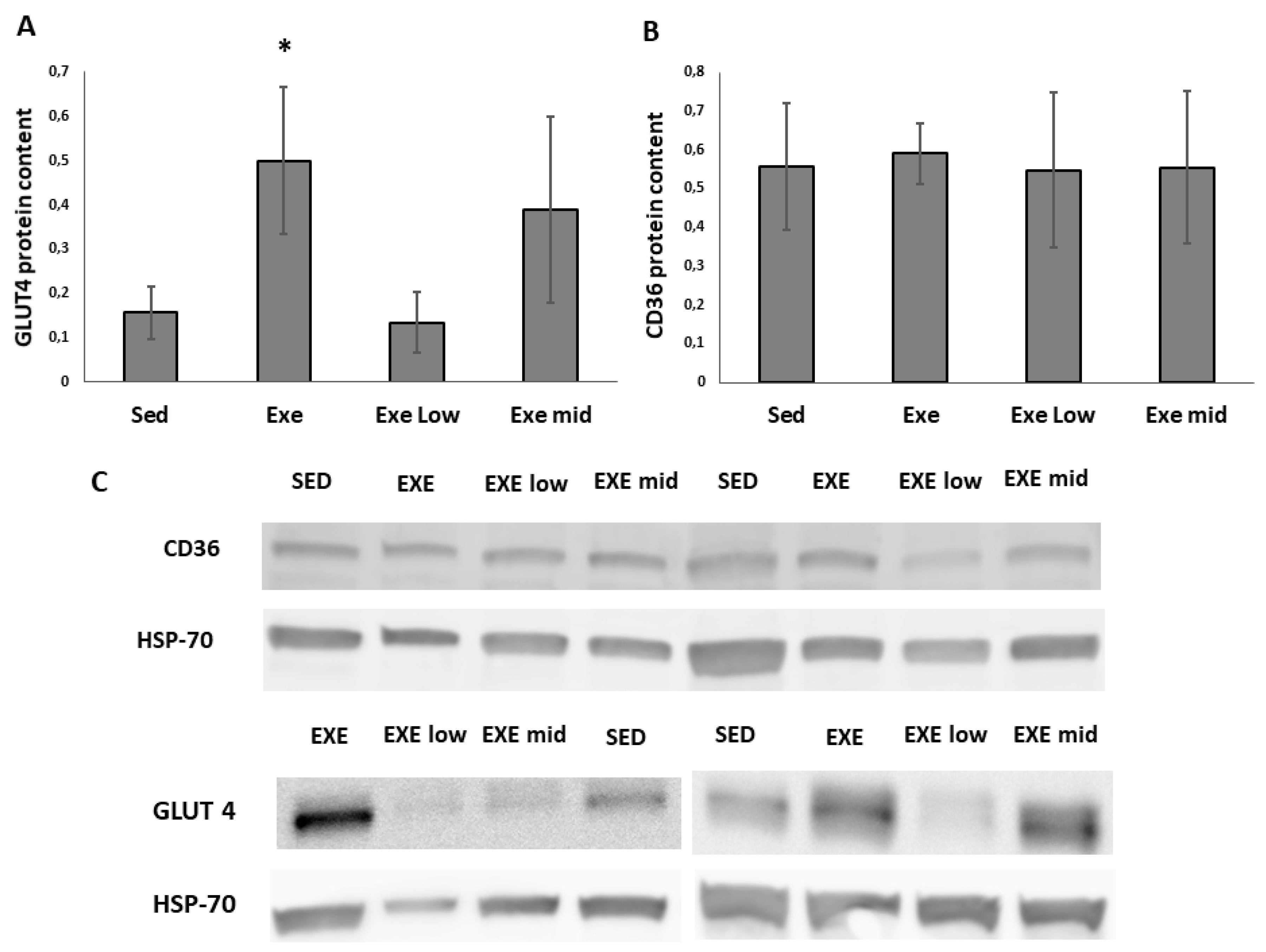
3.2. CD36 and GLUT4 Protein Content
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Lampousi, A.M.; Portillo, M.P.; Romaguera, D.; Hoffmann, G.; Boeing, H. Olive oil in the prevention and management of type 2 diabetes mellitus: A systematic review and meta-analysis of cohort studies and intervention trials. Nutr. Diabetes 2017, 7, e262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quiles, J.L.; Huertas, J.R.; Manas, M.; Battino, M.; Mataix, J. Physical exercise affects the lipid profile of mitochondrial membranes in rats fed with virgin olive oil or sunflower oil. Br. J. Nutr. 1999, 81, 21–24. [Google Scholar] [CrossRef] [Green Version]
- Huertas, J.R.; Battino, M.; Barzanti, V.; Maranesi, M.; Parenti-Castelli, G.; Littarru, G.P.; Turchetto, E.; Mataix, F.J.; Lenaz, G. Mitochondrial and microsomal cholesterol mobilization after oxidative stress induced by adriamycin in rats fed with dietary olive and corn oil. Life Sci. 1992, 26, 2111–2118. [Google Scholar] [CrossRef]
- Quiles, J.L.; Huertas, J.R.; Mañas, M.; Ochoa, J.J.; Battino, M.; Mataix, J. Oxidative stress induced by exercise and dietary fat modulates the coenzyme Q and vitamin A balance between plasma and mitochondria. Int. J. Vitam. Nutr. Res. 1999, 69, 243–249. [Google Scholar] [CrossRef]
- Musumeci, G.; Trovato, F.M.; Imbesi, R.; Castrogiovanni, P. Effects of dietary extra-virgin olive oil on oxidative stress resulting from exhaustive exercise in rat skeletal muscle: A morphological study. Acta Histochem. 2014, 116, 61–69. [Google Scholar] [CrossRef]
- Esquius, L.; Garcia-Retortillo, S.; Balagué, N.; Hristovski, R.; Javierre, C. Physiological-and performance-related effects of acute olive oil supplementation at moderate exercise intensity. J. Int. Soc. Sports Nutr. 2019, 16, 12. [Google Scholar] [CrossRef] [Green Version]
- Huertas, J.R.; Casuso, R.A.; Agustín, P.H.; Cogliati, S. Stay Fit, Stay Young: Mitochondria in Movement: The Role of Exercise in the New Mitochondrial Paradigm. Oxidative Med. Cell. Longev. 2019, 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cicerale, S.; Lucas, L.; Keast, R. Antimicrobial, antioxidant and anti-inflammatory phenolic activities in extra virgin olive oil. Curr. Opin. Biotechnol. 2012, 23, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Gorzynik-Debicka, M.; Przychodzen, P.; Cappello, F.; Kuban-Jankowska, A.; Marino Gammazza, A.; Knap, N.; Wozniak, M.; Gorska-Ponikowska, M. Potential health benefits of olive oil and plant polyphenols. Int. J. Mol. Sci. 2018, 19, 686. [Google Scholar] [CrossRef] [Green Version]
- Feng, Z.; Bai, L.; Yan, J.; Li, Y.; Shen, W.; Wang, Y.; Wertz, K.; Weber, P.; Zhang, Y.; Chen, Y. Mitochondrial dynamic remodeling in strenuous exercise-induced muscle and mitochondrial dysfunction: Regulatory effects of hydroxytyrosol. Free Radic. Biol. Med. 2011, 50, 1437–1446. [Google Scholar] [CrossRef]
- Cao, K.; Xu, J.; Zou, X.; Li, Y.; Chen, C.; Zheng, A.; Li, H.; Li, H.; Szeto, I.M.-Y.; Shi, Y. Hydroxytyrosol prevents diet-induced metabolic syndrome and attenuates mitochondrial abnormalities in obese mice. Free Radic. Biol. Med. 2014, 67, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Al Fazazi, S.; Casuso, R.A.; Aragón-Vela, J.; Casals, C.; Huertas, J.R. Effects of hydroxytyrosol dose on the redox status of exercised rats: The role of hydroxytyrosol in exercise performance. J. Int. Soc. Sports Nutr. 2018, 15, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casuso, R.A.; Al-Fazazi, S.; Hidalgo-Gutierrez, A.; López, L.C.; Plaza-Díaz, J.; Rueda-Robles, A.; Huertas, J.R. Hydroxytyrosol influences exercise-induced mitochondrial respiratory complex assembly into supercomplexes in rats. Free Radic. Biol. Med. 2019, 134, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Casuso, R.A.; Al Fazazi, S.; Ruiz-Ojeda, F.J.; Plaza-Diaz, J.; Rueda-Robles, A.; Aragón-Vela, J.; Huertas, J.R. Hydroxytyrosol Modifies Skeletal Muscle GLUT4/AKT/Rac1 Axis in Trained Rats. J. Cell. Physiol. 2021, 236, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Huertas, J.; Al Fazazi, S.; Hidalgo-Gutierrez, A.; López, L.; Casuso, R. Antioxidant effect of exercise: Exploring the role of the mitochondrial complex I superassembly. Redox Biol. 2017, 13, 477–481. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Medina-Remón, A.; Pérez-Jiménez, J.; Martínez-González, M.A.; Covas, M.I.; Corella, D.; Salas-Sal-vadó, J.; Gómez-Gracia, E.; Lapetra, J.; Arós, F.; et al. Dietary intake and major food sourc-es of polyphenols in a Spanish population at high cardiovascular risk: The PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 953–959. [Google Scholar] [CrossRef]
- de la Torre-Robles, A.; Rivas, A.; Lorenzo-Tovar, M.L.; Monteagudo, C.; Mariscal-Arcas, M.; Olea-Serrano, F. Estimation of the intake of phenol compounds from virgin olive oil of a population from southern Spain. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess 2014, 9, 1460–1469. [Google Scholar] [CrossRef]
- Murgia, M.; Nagaraj, N.; Deshmukh, A.S.; Zeiler, M.; Cancellara, P.; Moretti, I.; Reggiani, C.; Schiaffino, S.; Mann, M. Single muscle fiber proteomics reveals unexpected mitochondrial specialization. EMBO Rep. 2015, 16, 387–395. [Google Scholar] [CrossRef]
- Ren, J.-M.; Semenkovich, C.F.; Gulve, E.A.; Gao, J.; Holloszy, J.O. Exercise induces rapid increases in GLUT4 expression, glucose transport capacity, and insulin-stimulated glycogen storage in muscle. J. Biol. Chem. 1994, 269, 14396–14401. [Google Scholar] [CrossRef]
- McCullagh, K.; Poole, R.; Halestrap, A.; O’Brien, M.; Bonen, A. Role of the lactate transporter (MCT1) in skeletal muscles. Am. J. Physiol.-Endocrinol. Metab. 1996, 271, E143–E150. [Google Scholar] [CrossRef]
- McCullagh, K.; Poole, R.; Halestrap, A.; Tipton, K.; O’Brien, M.; Bonen, A. Chronic electrical stimulation increases MCT1 and lactate uptake in red and white skeletal muscle. Am. J. Physiol.-Endocrinol. Metab. 1997, 273, E239–E246. [Google Scholar] [CrossRef] [PubMed]
- Bonen, A. The expression of lactate transporters (MCT1 and MCT4) in heart and muscle. Eur. J. Appl. Physiol. 2001, 86, 6–11. [Google Scholar] [CrossRef]
- Bonen, A.; Han, X.-X.; Habets, D.D.; Febbraio, M.; Glatz, J.F.; Luiken, J.J. A null mutation in skeletal muscle FAT/CD36 reveals its essential role in insulin-and AICAR-stimulated fatty acid metabolism. Am. J. Physiol.-Endocrinol. Metab. 2007, 292, E1740–E1749. [Google Scholar] [CrossRef]
- Jeppesen, J.; Albers, P.H.; Rose, A.J.; Birk, J.B.; Schjerling, P.; Dzamko, N.; Steinberg, G.R.; Kiens, B. Contraction-induced skeletal muscle FAT/CD36 trafficking and FA uptake is AMPK independent. J. Lipid Res. 2011, 52, 699–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiaffino, S.; Reggiani, C.; Kostrominova, T.; Mann, M.; Murgia, M. Mitochondrial specialization revealed by single muscle fiber proteomics: Focus on the Krebs cycle. Scand. J. Med. Sci. Sports 2015, 25, 41–48. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Contreras, C.; Vazquez-Gomez, M.; Barbero, A.; Pesantez, J.L.; Zinellu, A.; Berlinguer, F.; Gonzalez-Añover, P.; Gonzalez, J.; Encinas, T.; Torres-Rovira, L. Polyphenols and IUGR Pregnancies: Effects of Maternal Hydroxytyrosol Supplementation on Placental Gene Expression and Fetal Antioxidant Status, DNA-Methylation and Phenotype. Int. J. Mol. Sci. 2019, 20, 1187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di, A.F.; Falconi, A.; Di, C.G.; Micioni, M.D.B.; Costa, A.; Caramuta, S.; Del, M.C.; Compagnone, D.; Dainese, E.; Cifani, C. Extravirgin olive oil up-regulates CB1 tumor suppressor gene in human colon cancer cells and in rat colon via epigenetic mechanisms. J. Nutr. Biochem. 2015, 26, 250–258. [Google Scholar]
- Arpón, A.; Milagro, F.; Razquin, C.; Corella, D.; Estruch, R.; Fitó, M.; Marti, A.; Martínez-González, M.; Ros, E.; Salas-Salvadó, J. Impact of consuming extra-virgin olive oil or nuts within a Mediterranean diet on DNA methylation in peripheral white blood cells within the PREDIMED-Navarra randomized controlled trial: A role for dietary lipids. Nutrients 2018, 10, 15. [Google Scholar] [CrossRef] [Green Version]
- Hao, J.; Shen, W.; Yu, G.; Jia, H.; Li, X.; Feng, Z.; Wang, Y.; Weber, P.; Wertz, K.; Sharman, E. Hydroxytyrosol promotes mitochondrial biogenesis and mitochondrial function in 3T3-L1 adipocytes. J. Nutr. Biochem. 2010, 21, 634–644. [Google Scholar] [CrossRef]
- Doege, H.; Bocianski, A.; Scheepers, A.; Axer, H.; Eckel, J.; Joost, H.G.; Schürmann, A. Characterization of human glucose transporter (GLUT) 11 (encoded by SLC2A11), a novel sugar-transport facilitator specifically expressed in heart and skeletal muscle. Biochem. J. 2001, 359, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Cogliati, S.; Cabrera-Alarcón, J.L.; Enriquez, J.A. Regulation and functional role of the electron transport chain supercomplexes. Biochem. Soc. Trans. 2021, BST20210460. [Google Scholar] [CrossRef] [PubMed]
- Balsa, E.; Soustek, M.S.; Thomas, A.; Cogliati, S.; García-Poyatos, C.; Martín-García, E.; Jedrychowski, M.; Gygi, S.P.; Enriquez, J.A.; Puigserver, P. ER and Nutrient Stress Promote Assembly of Respiratory Chain Supercomplexes through the PERK-eIF2α Axis. Mol. Cell 2019, 74, 877–890. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, D.; Sorrentino, V.; Auwerx, J. Cytosolic Proteostasis Networks of the Mitochondrial Stress Response. Trends Biochem. Sci. 2017, 9, 712–725. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 2020, 8, 421–438. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casuso, R.A.; Al Fazazi, S.; Plaza-Díaz, J.; Ruiz-Ojeda, F.J.; Rueda-Robles, A.; Aragón-Vela, J.; Huertas, J.R. Physiological Doses of Hydroxytyrosol Modulate Gene Expression in Skeletal Muscle of Exercised Rats. Life 2021, 11, 1393. https://doi.org/10.3390/life11121393
Casuso RA, Al Fazazi S, Plaza-Díaz J, Ruiz-Ojeda FJ, Rueda-Robles A, Aragón-Vela J, Huertas JR. Physiological Doses of Hydroxytyrosol Modulate Gene Expression in Skeletal Muscle of Exercised Rats. Life. 2021; 11(12):1393. https://doi.org/10.3390/life11121393
Chicago/Turabian StyleCasuso, Rafael A., Saad Al Fazazi, Julio Plaza-Díaz, Francisco J. Ruiz-Ojeda, Ascensión Rueda-Robles, Jerónimo Aragón-Vela, and Jesús R. Huertas. 2021. "Physiological Doses of Hydroxytyrosol Modulate Gene Expression in Skeletal Muscle of Exercised Rats" Life 11, no. 12: 1393. https://doi.org/10.3390/life11121393
APA StyleCasuso, R. A., Al Fazazi, S., Plaza-Díaz, J., Ruiz-Ojeda, F. J., Rueda-Robles, A., Aragón-Vela, J., & Huertas, J. R. (2021). Physiological Doses of Hydroxytyrosol Modulate Gene Expression in Skeletal Muscle of Exercised Rats. Life, 11(12), 1393. https://doi.org/10.3390/life11121393










