Ex-Vivo Skin Explant Culture Is a Model for TSLP-Mediated Skin Barrier Immunity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Serum Isolation
2.3. Epidermal Ear Sheets
2.4. Histological Analysis and Immunofluorescence Microscopy
2.5. Epidermal Single Cell Suspension and Flow Cytometry Analysis
2.6. Skin Explant Cultures
2.7. ELISA
2.8. Skin Protein Isolation
2.9. Statistical Methods
3. Results
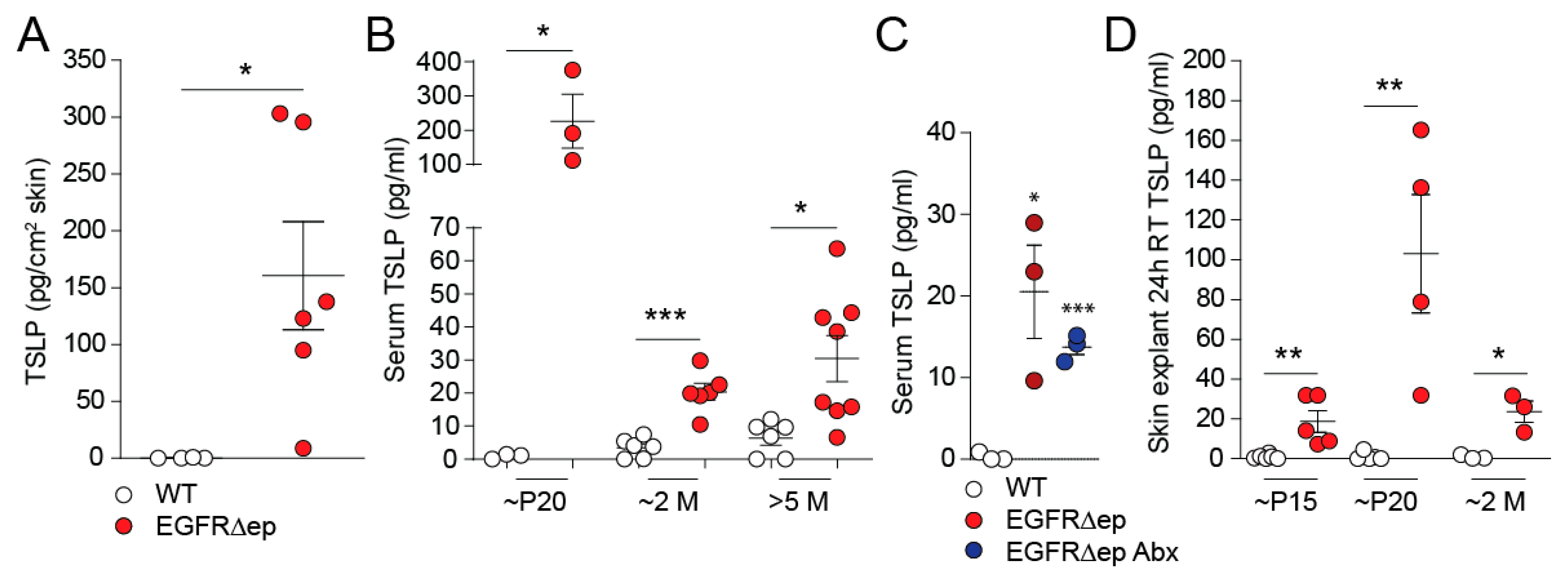
3.1. TSLP Is Induced and Released from EGFR∆ep Skin In Vivo and Ex Vivo
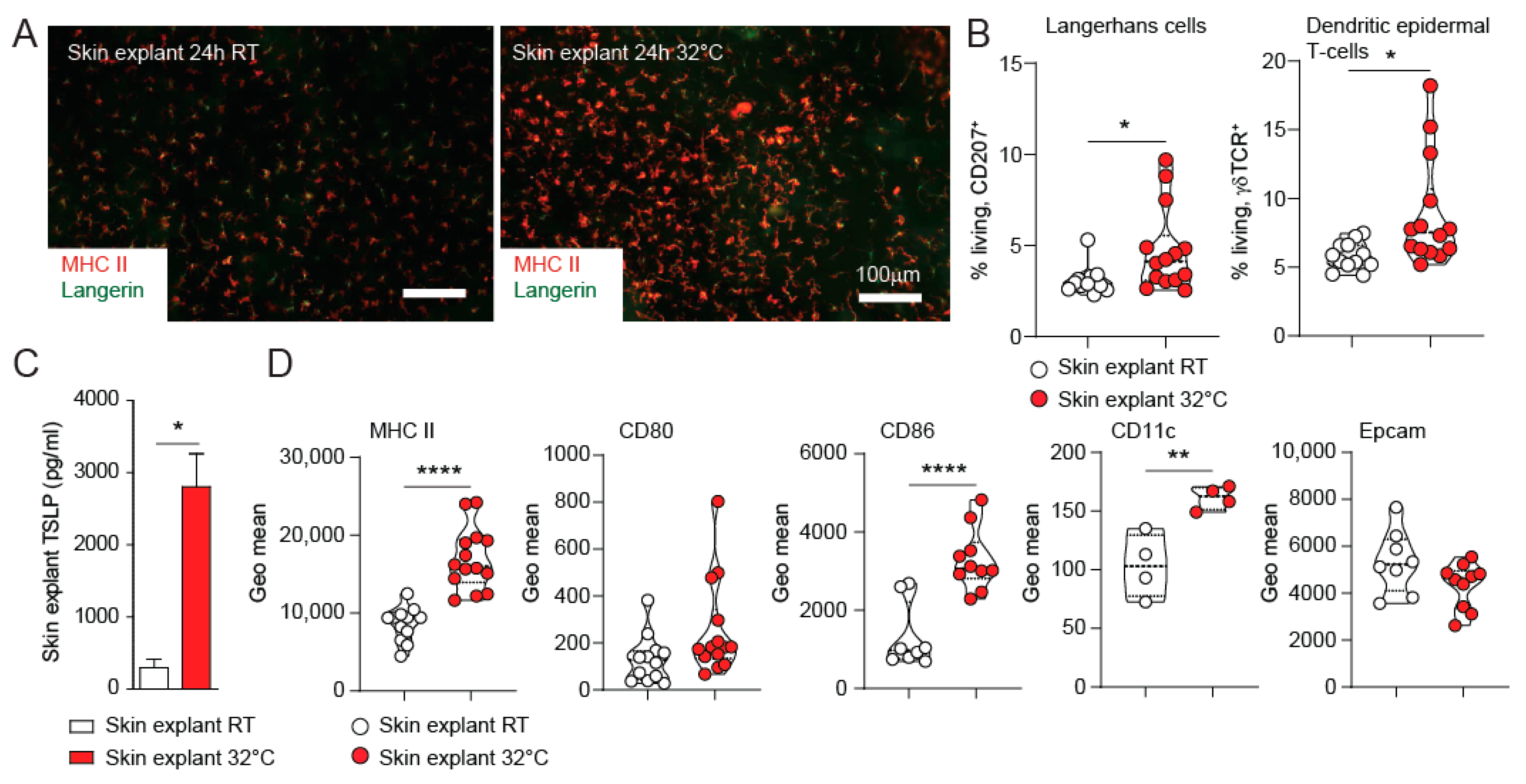
3.2. TSLP Expression from Skin Explants Cultured at 32 °C Represents an Ex-Vivo Model for Skin Barrier Immunity
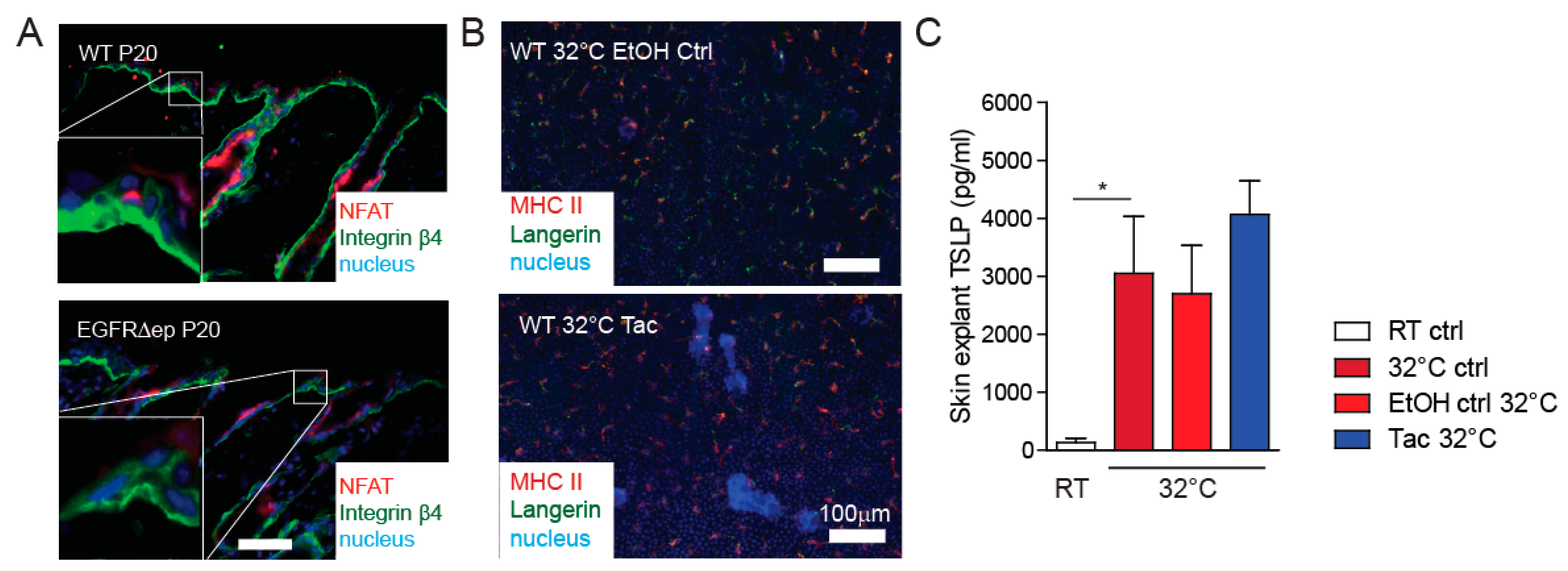
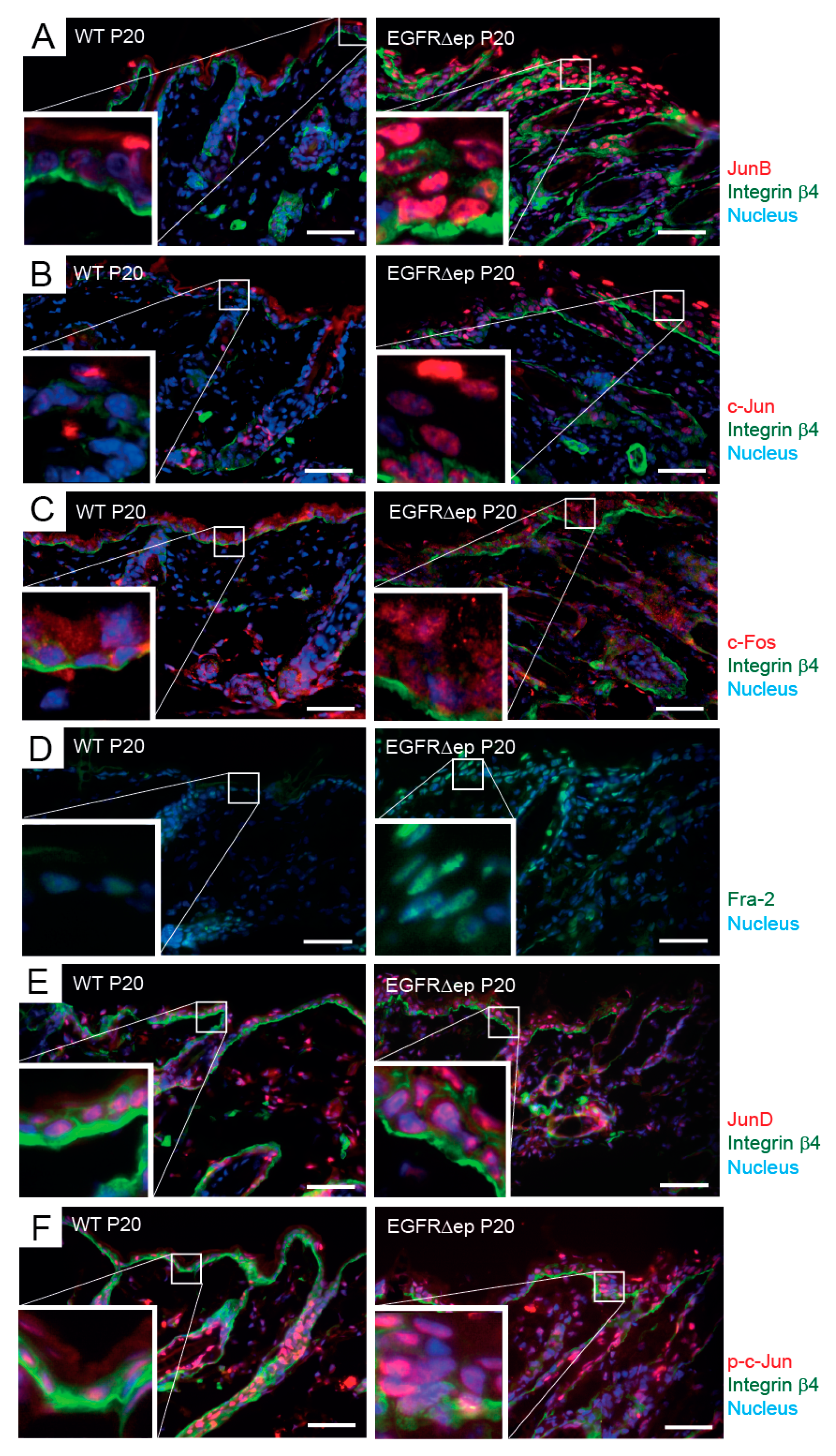
3.3. TSLP Transcriptional Regulator NFAT Is Not Up-Regulated in EGFR∆ep Mice, and an NFAT Pathway Inhibitor Does Not Block TSLP Expression Ex Vivo
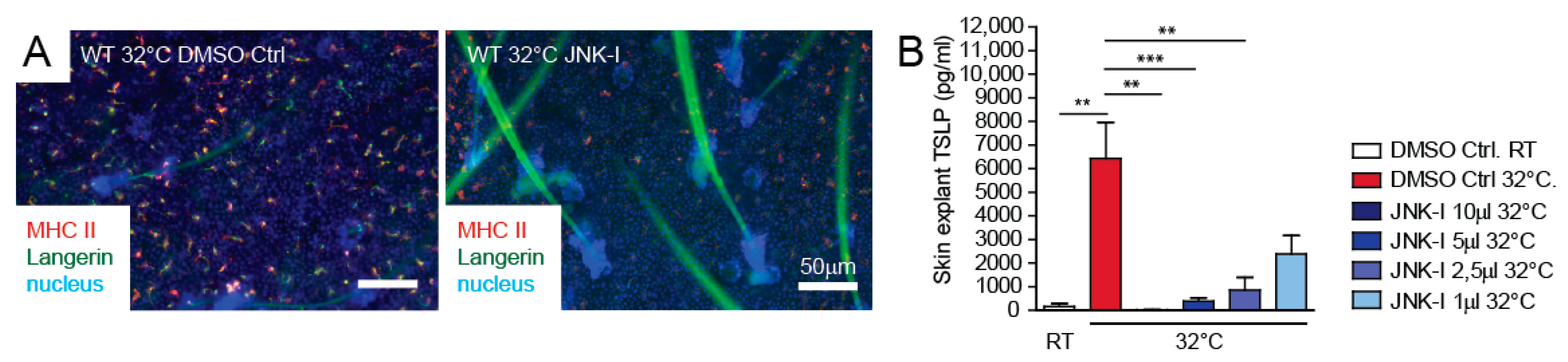
3.4. JNK Pathway Inhibitor Blocks TSLP Expression Ex Vivo
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Y.E.; Fischbach, M.A.; Belkaid, Y. Skin microbiota-host interactions. Nature 2018, 553, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Bauer, T.; Zagórska, A.; Jurkin, J.; Yasmin, N.; Köffel, R.; Richter, S.; Gesslbauer, B.; Lemke, G.; StrobL, H. Identification of Axl as a downstream effector of TGF-beta1 during Langerhans cell differentiation and epidermal homeostasis. J. Exp. Med. 2012, 209, 2033–2047. [Google Scholar] [CrossRef] [Green Version]
- Lichtenberger, B.M.; Gerber, P.A.; Holcmann, M.; Buhren, B.A.; Amberg, N.; Smolle, V.; Schrumpf, H.; Boelke, E.; Ansari, P.; Mackenzie, C.; et al. Epidermal EGFR Controls Cutaneous Host Defense and Prevents Inflammation. Sci. Transl. Med. 2013, 5, 199ra111. [Google Scholar] [CrossRef]
- Corren, J.; Ziegler, S.F. TSLP: From allergy to cancer. Nat. Immunol. 2019, 20, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Ebner, S.; Nguyen, V.A.; Forstner, M.; Wang, Y.-H.; Wolfram, D.; Liu, Y.-J.; Romani, N. Thymic stromal lymphopoietin converts human epidermal Langerhans cells into antigen-presenting cells that induce proallergic T cells. J. Allergy Clin. Immunol. 2007, 119, 982–990. [Google Scholar] [CrossRef]
- Weidinger, S.; Beck, L.A.; Bieber, T.; Kabashima, K.; Irvine, A.D. Nature Reviews Disease Primers Atopic dermatitis. Nat. Rev. Dis. Primers 2018, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Pastore, S.; Giustizieri, M.L.; Mascia, F.; Giannetti, A.; Kaushansky, K.; Girolomoni, G. Dysregulated Activation of Activator Protein 1 in Keratinocytes of Atopic Dermatitis Patients with Enhanced Expression of Granulocyte/Macrophage-Colony Stimulating Factor. J. Investig. Dermatol. 2000, 115, 1134–1143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zenz, R.; Eferl, R.; Kenner, L.; Florin, L.; Hummerich, L.; Mehic, D.; Scheuch, H.; Angel, P.; Tschachler, E.; Wagner, E.F. Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature 2005, 437, 369–375. [Google Scholar] [CrossRef]
- Novoszel, P.; Holcmann, M.; Stulnig, G.; De Sa Fernandes, C.; Zyulina, V.; Borek, I.; Linder, M.; Bogusch, A.; Drobits, B.; Bauer, T.; et al. Psoriatic skin inflammation is promoted by c-Jun/AP-1-dependent CCL2 and IL-23 expression in dendritic cells. EMBO Mol. Med. 2021, 13, e12409. [Google Scholar] [CrossRef] [PubMed]
- Klufa, J.; Bauer, T.; Hanson, B.; Herbold, C.; Starkl, P.; Lichtenberger, B.; Srutkova, D.; Schulz, D.; Vujic, I.; Mohr, T.; et al. Hair eruption initiates and commensal skin microbiota aggravate adverse events of anti-EGFR therapy. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging functions of the EGFR in cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef]
- Holcmann, M.; Sibilia, M. Mechanisms underlying skin disorders induced by EGFR inhibitors. Mol. Cell. Oncol. 2015, 2, e1004969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, S.R.; Thé, L.; Batia, L.M.; Beattie, K.; Katibah, G.E.; McClain, S.P.; Pellegrino, M.; Estandian, D.M.; Bautista, D.M. The Epithelial Cell-Derived Atopic Dermatitis Cytokine TSLP Activates Neurons to Induce Itch. Cell 2013, 155, 285–295. [Google Scholar] [CrossRef] [Green Version]
- Murthy, A.; Shao, Y.W.; Narala, S.R.; Molyneux, S.; Zuniga-Pflucker, J.C.; Khokha, R. Notch Activation by the Metalloproteinase ADAM17 Regulates Myeloproliferation and Atopic Barrier Immunity by Suppressing Epithelial Cytokine Synthesis. Immunity 2012, 36, 105–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, X.; Guo, Y.; Jiang, C.; Chang, Q.; Zhang, S.; Luo, T.; Zhang, B.; Jia, X.; Hung, M.-C.; Dong, C.; et al. JNK1 negatively controls antifungal innate immunity by suppressing CD23 expression. Nat. Med. 2017, 23, 337–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raguz, J.; Jeric, I.; Niault, T.; Nowacka, J.D.; Kuzet, S.E.; Rupp, C.; Fischer, I.; Biggi, S.; Borsello, T.; Baccarini, M. Epidermal RAF prevents allergic skin disease. eLife 2016, 5, e14012. [Google Scholar] [CrossRef]
- A Ascierto, P.; Schadendorf, D.; Berking, C.; Agarwala, S.S.; Van Herpen, C.M.; Queirolo, P.; Blank, C.U.; Hauschild, A.; Beck, J.T.; St-Pierre, A.; et al. MEK162 for patients with advanced melanoma harbouring NRAS or Val600 BRAF mutations: A non-randomised, open-label phase 2 study. Lancet Oncol. 2013, 14, 249–256. [Google Scholar] [CrossRef]
- Yoo, J.; Omori, M.; Gyarmati, D.; Zhou, B.; Aye, T.; Brewer, A.; Comeau, M.R.; Campbell, D.J.; Ziegler, S.F. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med. 2005, 202, 541–549. [Google Scholar] [CrossRef]
- Ratzinger, G.; Stoitzner, P.; Ebner, S.; Lutz, M.B.; Layton, G.T.; Rainer, C.; Senior, R.M.; Shipley, J.M.; Fritsch, P.; Schuler, G.; et al. Matrix Metalloproteinases 9 and 2 Are Necessary for the Migration of Langerhans Cells and Dermal Dendritic Cells from Human and Murine Skin. J. Immunol. 2002, 168, 4361–4371. [Google Scholar] [CrossRef] [Green Version]
- Stoitzner, P.; Zanella, M.; Ortner, U.; Lukas, M.; Tagwerker, A.; Janke, K.; Lutz, M.B.; Schuler, G.; Echtenacher, B.; Ryffel, B.; et al. Migration of langerhans cells and dermal dendritic cells in skin organ cultures: Augmentation by TNF-alpha and IL-1beta. J. Leukoc. Biol. 1999, 66, 462–470. [Google Scholar] [CrossRef]
- Weinlich, G.; Heine, M.; Stössel, H.; Zanella, M.; Stoitzner, P.; Ortner, U.; Smolle, J.; Koch, F.; Sepp, N.T.; Schuler, G.; et al. Entry into afferent lymphatics and maturation in situ of migrating murine cutaneous dendritic cells. J. Investig. Dermatol. 1998, 110, 441–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eberlin, S.; Silva, M.S.D.; Facchini, G.; Silva, G.H.D.; Pinheiro, A.L.T.A.; Eberlin, S.; Pinheiro, A.D.S. The Ex Vivo Skin Model as an Alternative Tool for the Efficacy and Safety Evaluation of Topical Products. Altern. Lab. Anim. 2020, 48, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Lemke, G. Biology of the TAM receptors. Cold Spring Harb. Perspect. Biol. 2013, 5, a009076. [Google Scholar] [CrossRef] [PubMed]
| Antibodies | SOURCE | IDENTIFIER |
|---|---|---|
| Armenian hamster anti-mouse TCR γ/δ Antibody, FITC | Biolegend | Cat# 118106, RRID:AB_313830 |
| Rat anti-mouse/human CD11b Antibody, Pacific Blue | Biolegend | Cat# 101224, RRID:AB_755986 |
| Armenian hamster anti-mouse CD11c Antibody, PE/Cy5 | Biolegend | Cat# 117316, RRID:AB_493566 |
| Rat anti-mouse I-A/I-E Antibody, APC/Cy7 | Biolegend | Cat# 107628, RRID:AB_2069377 |
| Rat anti-mouse CD45 Antibody, APC | Biolegend | Cat# 103112, RRID:AB_312977 |
| Rat anti-Langerin/CD207 Antibody (929F3.01), Alexa Fluor 488 | Dendritics | Cat# DDX0362A488, RRID:AB_1148740 |
| Rat anti-mouse I-A/I-E Antibody, PE | Biolegend | Cat# 107608, RRID:AB_313323 |
| Rat anti-mouse/human Langerin/CD207, Alexa Fluor 488 | Dendritics | Cat# DDX0362 929F3.01 |
| Rat anti-mouse EpCAM/CD326, FITC | Biolegend | Cat# 118208, RRID:AB_1134107 |
| Rat anti-mouse CD45, PE | Biolegend | Cat# 103106, RRID:AB_312971 |
| Rat anti-mouse CD45, APC | Biolegend | Cat# 103112, RRID:AB_312977 |
| Anti-beta III Tubulin Antibody (TUJ-1) | abcam | ab14545 |
| JunD (329) | Santa cruz | sc-74 |
| NFATc1 | Santa cruz-sc-7294 | 7A6 |
| Fra2 CN10 | home made | |
| Phospho-cJun Ser73 | Cell Signalling | D47G9 |
| cJun | Cell Signalling | 60A8 |
| JunB | Cell Signalling | C37F9 |
| cFos | THP-Medical Products | Y075085 |
| CD104-Integrin β4 chain | BD Pharmingen™ | Cat# 553745 |
| Donkey anti-rabbit 594/488 Alexa Fluor | Invitrogen-Life Technologies | Cat# A21206 |
| Donkey anti-rat 594/488 Alexa Fluor | Invitrogen-Life Technologies | Cat# 21208 |
| Donkey anti-Guinea Pig 594 Alexa Fluor | Invitrogen-Life Technologies | Cat# A11076 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bauer, T.; Gubi, D.; Klufa, J.; Novoszel, P.; Holcmann, M.; Sibilia, M. Ex-Vivo Skin Explant Culture Is a Model for TSLP-Mediated Skin Barrier Immunity. Life 2021, 11, 1237. https://doi.org/10.3390/life11111237
Bauer T, Gubi D, Klufa J, Novoszel P, Holcmann M, Sibilia M. Ex-Vivo Skin Explant Culture Is a Model for TSLP-Mediated Skin Barrier Immunity. Life. 2021; 11(11):1237. https://doi.org/10.3390/life11111237
Chicago/Turabian StyleBauer, Thomas, Daniela Gubi, Jörg Klufa, Philipp Novoszel, Martin Holcmann, and Maria Sibilia. 2021. "Ex-Vivo Skin Explant Culture Is a Model for TSLP-Mediated Skin Barrier Immunity" Life 11, no. 11: 1237. https://doi.org/10.3390/life11111237
APA StyleBauer, T., Gubi, D., Klufa, J., Novoszel, P., Holcmann, M., & Sibilia, M. (2021). Ex-Vivo Skin Explant Culture Is a Model for TSLP-Mediated Skin Barrier Immunity. Life, 11(11), 1237. https://doi.org/10.3390/life11111237






