An Overview of Bioinformatics Tools for DNA Meta-Barcoding Analysis of Microbial Communities of Bioaerosols: Digest for Microbiologists
Abstract
1. Introduction
2. Methods and Software
2.1. Controls and Bio-informatic Management of Controls
2.2. Mock Microbial Communities
2.3. Negative Controls
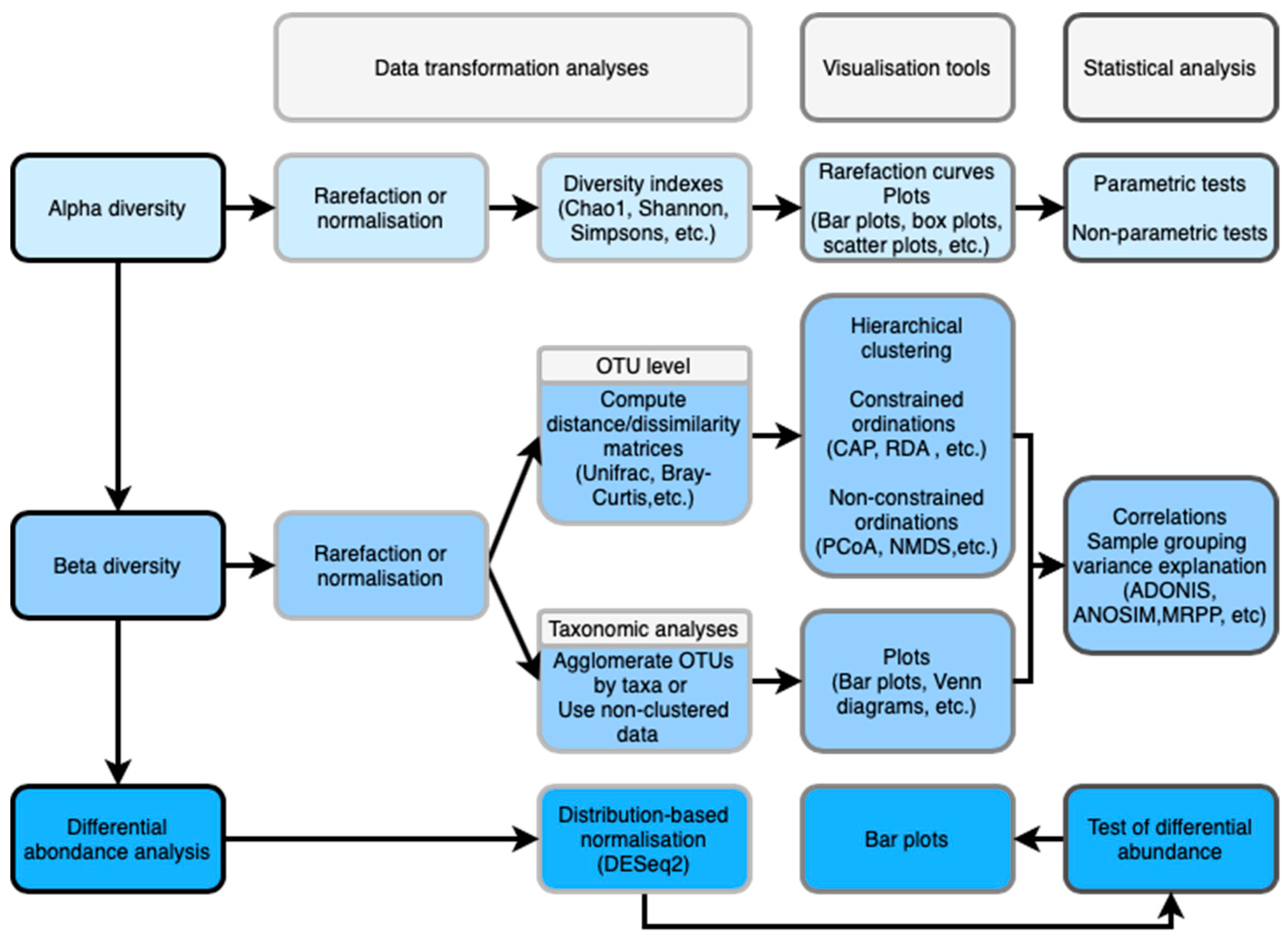
2.4. Data Transformation
3. Sequencing Depth
Alpha and Beta Diversity
4. Visualization Tools
4.1. Alpha and Beta Diversity
4.2. Additional Visualization Tools
5. Statistical Analysis
5.1. Parametric VS. Nonparametric Statistics
5.2. Comparisons Using Alpha and Beta Diversity Measures
5.3. Statistical Significance of Sample Groupings
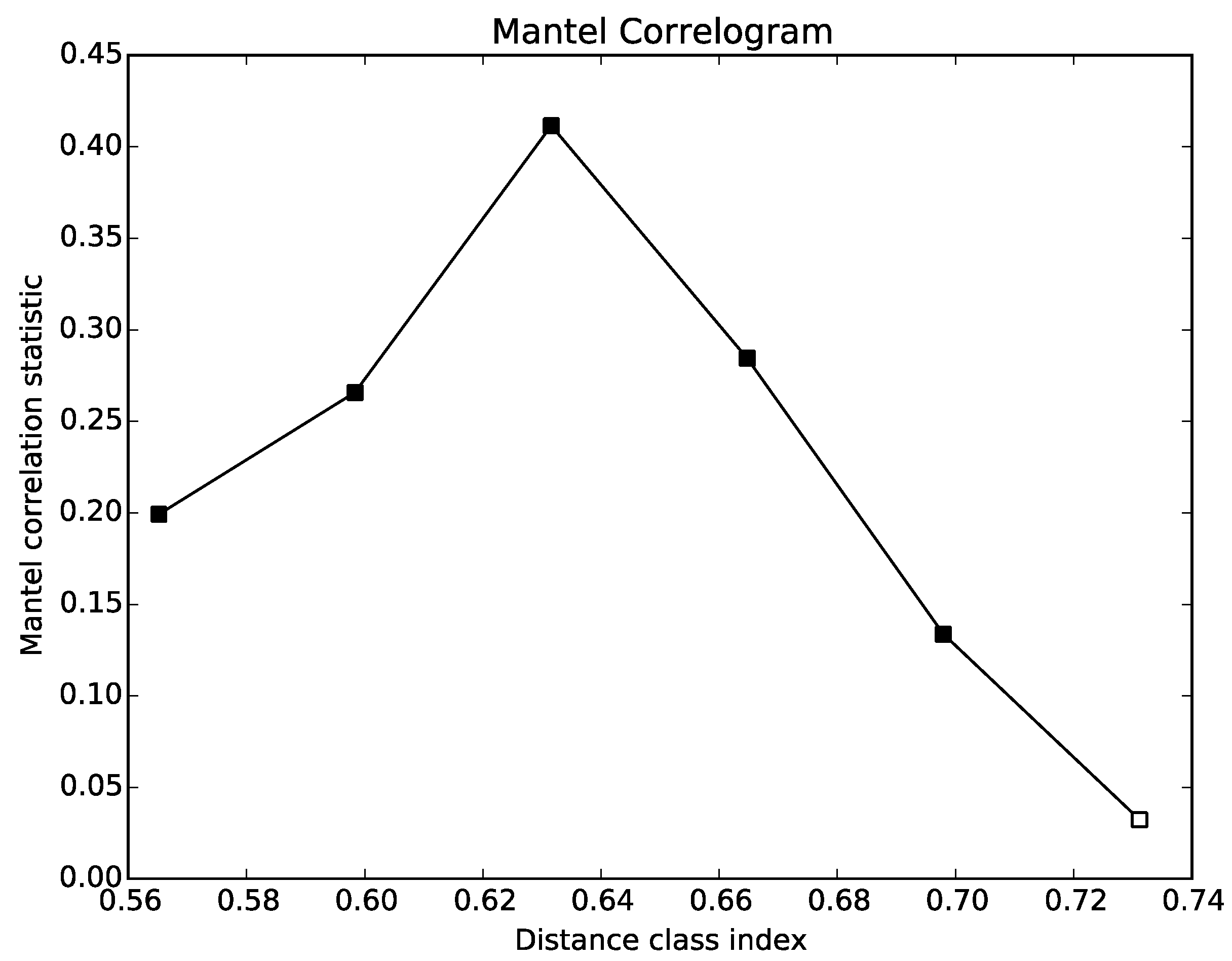
5.4. Correlations
6. Taxonomic Analyses
7. Differential Abundance
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mardis, E.R. Next-Generation DNA Sequencing Methods. Annu. Rev. Genom. Hum. Genet. 2008, 9, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Novais, R.C.; Thorstenson, Y. The evolution of Pyrosequencing® for microbiology: From genes to genomes. J. Microbiol. Methods 2011, 86, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; A Walters, W.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108, 4516–4522. [Google Scholar] [CrossRef]
- Bolhuis, H.; Cretoiu, M.S.; Stal, L.J. Molecular ecology of microbial mats. FEMS Microbiol. Ecol. 2014, 90, 335–350. [Google Scholar] [PubMed]
- Huttenhower, C.; Knight, R.; Brown, C.T.; Caporaso, J.G.; Clemente, J.C.; Gevers, D.; Franzosa, E.A.; Kelley, S.T.; Knights, D.; E Ley, R.; et al. Advancing the microbiome research community. Cell 2014, 159, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, J.K.; Rienzi, S.D.; Poole, A.C.; Koren, O.; Walters, W.A.; Caporaso, J.G.; Knight, R.; Ley, R.E. Conducting a Microbiome Study. Cell 2014, 158, 250–262. [Google Scholar] [CrossRef]
- Yoon, S.S.; Kim, E.-K.; Lee, W.-J. Functional genomic and metagenomic approaches to understanding gut microbiota–animal mutualism. Curr. Opin. Microbiol. 2015, 24, 38–46. [Google Scholar] [CrossRef]
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current understanding of the human microbiome. Nat. Med. 2018, 24, 392–400. [Google Scholar] [CrossRef]
- Benìtez-Pàez, A.; Sanz, Y. Milti-locus and long amplicon sequencing approach to study microbial diversity at species level using the MinIon™ portable nanopore sequencer. GigaScience 2017, 6, 1–12. [Google Scholar] [CrossRef]
- Filippis, F.D.; Laiola, M.; Blaiotta, G.; Ercolini, D. Different Amplicon Targets for Sequencing-Based Studies of Fungal Diversity. Appl. Environ. Microbiol. 2017, 83, e00905-17. [Google Scholar] [CrossRef] [PubMed]
- Pollock, J.; Glendinning, L.; Wisedchanwet, T.; Watson, M. The Madness of Microbiome: Attempting to Find Consensus "Best Practice" for 16S Microbiome Studies. Appl. Environ. Microbiol. 2018, 84, e02627-17. [Google Scholar] [CrossRef] [PubMed]
- Naqib, A.; Poggi, S.; Wang, W.; Hyde, M.; Kunstman, K.; Green, S.J. Making and Sequencing Heavily Multiplexed, High-Throughput 16S Ribosomal RNA Gene Amplicon Libraries Using a Flexible, Two-Stage PCR Protocol. Gene Expr. Anal. Methods Mol. Biol. 2018, 1783, 149–169. [Google Scholar]
- Hugenholtz, P.; Pace, N.R. Identifying microbial diversity in the natural environment: A molecular phylogenetic approach. Trends Biotechnol. 1996, 14, 190–197. [Google Scholar] [CrossRef]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Bolchacova, E.; Voigt, K.; Crous, P.W.; et al. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef]
- Venter, J.C.; Remington, K.; Heidelberg, J.F.; Halpern, A.L.; Rusch, D.; Eisen, J.A.; Wu, D.; Paulsen, I.; Nelson, K.E.; Nelson, W.; et al. Environmental Genome Shotgun Sequencing of the Sargasso Sea. Science 2004, 304, 66–74. [Google Scholar] [CrossRef]
- Delmont, T.O.; Robe, P.; Cecillon, S.; Clark, I.M.; Constancias, F.; Simonet, P.; Hirsch, P.; Vogel, T.M. Accessing the Soil Metagenome for Studies of Microbial Diversity. Appl. Environ. Microbiol. 2011, 77, 1315–1324. [Google Scholar] [CrossRef]
- Walter, J.; E Ley, R. The Human Gut Microbiome: Ecology and Recent Evolutionary Changes. Annu. Rev. Microbiol. 2011, 65, 411–429. [Google Scholar] [CrossRef]
- Philippot, L.; Raaijmakers, J.M.; Lemanceau, P.; Van Der Putten, W.H. Going back to the roots: The microbial ecology of the rhizosphere. Nat. Rev. Microbiol. 2013, 11, 789–799. [Google Scholar] [CrossRef]
- Peccia, J.; Hospodsky, D.; Bibby, K. New Directions: A revolution in DNA sequencing now allows for the meaningful integration of biology with aerosol science. Atmos. Environ. 2010, 45, 1896–1897. [Google Scholar] [CrossRef]
- Nehmé, B.; Gilbert, Y.; Létourneau, V.; Forster, R.J.; Veillette, M.; Villemur, R.; Duchaine, C. Culture-Independent characterization of archaeal biodiversity in swine confinement building bioaerosols. Appl. Environ. Microbiol. 2009, 75, 5445–5450. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, I.; Bertolini, V.; Ambrosini, R.; Bestetti, G.; Franzetti, A. Unravelling the bacterial diversity in the atmosphere. Appl. Microbiol. Biotechnol. 2013, 97, 4727. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, N.; Nazaroff, W.W.; Peccia, J. Assessing the aerodynamic diameters of taxon-specific fungal bioaerosols by quantitative PCR and next-generation DNA sequencing. J. Aerosol Sci. 2014, 78, 1–10. [Google Scholar] [CrossRef]
- Madsen, A.M.; Zervas, A.; Tendal, K.; Nielsen, J.L. Microbial diversity in bioaerosol samples causing ODTS compared to reference bioaerosol samples as measured using Illumina sequencing and MALDI-TOF. Environ. Res. 2015, 140, 255–267. [Google Scholar] [CrossRef]
- Mbareche, H.; Brisebois, E.; Veillette, M.; Duchaine, C. Bioaerosol sampling and detection methods based on molecular approaches: No pain no gain. Sci. Total. Environ. 2017, 599, 2095–2104. [Google Scholar] [CrossRef]
- Mbareche, H.; Veillette, M.; Bonifait, L.; Dubuis, M.-E.; Benard, Y.; Marchand, G.; Bilodeau, G.J.; Duchaine, C. A next generation sequencing approach with a suitable bioinformatics workflow to study fungal diversity in bioaerosols released from two different types of composting plants. Sci. Total Environ. 2017, 601, 1306–1314. [Google Scholar] [CrossRef]
- Dubuis, M.-E.; Mbareche, H.; Veillette, M.; Bakhiyi, B.; Zayed, J.; Lavoie, J.; Duchaine, C. Bioaerosols concentrations in working areas in biomethanization facilities. J. Air Waste Manag. Assoc. 2017, 67, 1258–1271. [Google Scholar] [CrossRef]
- Knight, R.; Jansson, J.K.; Field, D.; Fierer, N.; Desai, N.; Fuhrman, J.A.; Hugenholtz, P.; Van Der Lelie, D.; Meyer, F.; Stevens, R.; et al. Unlocking the potential of metagenomics through replicated experimental design. Nat. Biotechnol. 2012, 30, 513–520. [Google Scholar] [CrossRef]
- Hogeweg, P. The Roots of Bioinformatics in Theoretical Biology. PLoS Comput. Boil. 2011, 7, e1002021. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, communitt-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; I Gordon, J.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahe, F. VSEARCH: A versatile open source tool for metagenomics. Peer J. 2016, 4, e2584. [Google Scholar] [CrossRef] [PubMed]
- Jamison, D.C. Introduction, in Perl Programming for Biologists; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2003; pp. 1–5. [Google Scholar]
- Vincent, A.T.; Charette, S.J. Who qualifies to be a bioinformatician? Front. Genet. 2015, 6, 164. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R. Broadening the definition of a bioinformatician. Front. Genet. 2015, 6, 258. [Google Scholar] [CrossRef]
- The Human Microbiome Project Consortium. A framework for human microbiome research. Nature 2012, 486, 215–221. [Google Scholar] [CrossRef]
- Davenport, C.F.; Tümmler, B. Advances in computational analysis of metagenome sequences. Environ. Microbiol. 2013, 15, 1–5. [Google Scholar] [CrossRef]
- Sinha, R.; The Microbiome Quality Control Project Consortium; Abu-Ali, G.; Vogtmann, E.; Fodor, A.A.; Ren, B.; Amir, A.; Schwager, E.; Crabtree, J.; Ma, B.; et al. Assessment of variation in microbial community amplicon sequencing by the Microbiome Quality Control (MBQC) project consortium. Nat. Biotechnol. 2017, 35, 1077–1086. [Google Scholar] [CrossRef]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Stevens, M.H.H. The Vegan Package. Community Ecology Package. Available online: http://cran.r-project.org/ http://r-forge.r-project.org/projects/vegan/ (accessed on 18 June 2020).
- McMurdie, P.J.; Holmes, S. phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; A Johnson, A.J.; Holmes, S. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Glassing, A.; Dowd, S.; Galandiuk, S.; Davis, B.; Chiodini, R.J. Inherent bacterial DNA contamination of extraction and sequencing reagents may affect interpretation of microbiota in low bacterial biomass samples. Gut Pathog. 2016, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Laurence, M.; Hatzis, C.; Brash, D.E. Common contaminants in nest-generation sequencing that hinder discovery of low-abundance microbes. PLoS ONE 2014, 9, e97876. [Google Scholar] [CrossRef] [PubMed]
- Czurda, S.; Smelik, S.; Preuner-Stix, S.; Nogueira, F.; Lion, T. Occurrence of Fungal DNA Contamination in PCR Reagents: Approaches to Control and Decontamination. J. Clin. Microbiol. 2015, 54, 148–152. [Google Scholar] [CrossRef]
- Minich, J.J.; Sanders, J.G.; Amir, A.; Humphrey, G.; Gilbert, J.A.; Knight, R. Quantifying and Understanding Well-to-Well Contamination in Microbiome Research. MSystems 2019, 4, e00186-19. [Google Scholar] [CrossRef]
- Pfeiffer, F.; Gröber, C.; Blank, M.; Händler, K.; Beyer, M.; Schultze, J.L.; Mayer, G. Systematic evaluation of error rates and causes in short samples in next-generation sequencing. Sci. Rep. 2018, 8, 10950. [Google Scholar] [CrossRef]
- Kebschull, J.M.; Zador, A.M. Sources of PCR-induced distortions in high-throughput sequencing data sets. Nucleic Acids Res. 2015, 43, e143. [Google Scholar] [CrossRef]
- Mbareche, H.; Veillette, M.; Pilote, J.; Létourneau, V.; Duchaine, C. Bioaerosols Play a Major Role in the Nasopharyngeal Microbiota Content in Agricultural Environment. Int. J. Environ. Res. Public Health 2019, 16, 1375. [Google Scholar] [CrossRef]
- Bakker, M.G. A fungal mock community control for amplicon sequencing experiments. Mol. Ecol. Resour. 2018, 18, 541–556. [Google Scholar] [CrossRef]
- Hardwick, S.A.; Chen, W.Y.; Wong, T.; Kanakamedala, B.S.; Deveson, I.W.; Ongley, S.E.; Santini, N.S.; Marcellin, E.; Smith, M.A.; Nielsen, L.K.; et al. Synthetic microbe communities provide internal reference standards for metagenome sequencing and analysis. Nat. Commun. 2018, 9, 3096. [Google Scholar] [CrossRef] [PubMed]
- Salter, S.J.; Cox, M.J.; Turek, E.; Calus, S.; Cookson, W.O.C.; Moffatt, M.F.; Turner, P.; Parkhill, J.; Loman, N.J.; Walker, A.W. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Boil. 2014, 12, 87. [Google Scholar] [CrossRef] [PubMed]
- Davis, N.M.; Proctor, D.M.; Holmes, S.P.; Relman, D.A.; Callahan, B.J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 2018, 6, 226. [Google Scholar] [CrossRef] [PubMed]
- Karstens, L.; Asquith, M.; Davin, S.; Fair, D.; Gregory, W.T.; Wolfe, A.J.; Braun, J.; McWeeney, S. Controlling for Contaminants in Low-Biomass 16S rRNA Gene Sequencing Experiments. mSystems 2019, 4, e00290-19. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, V.; Gaïa, N.; Girard, M.; Schrenzel, J. Decontamination of 16S rRNA gene amplicon sequence datasets based on bacterial load assessment by qPCR. BMC Microbiol. 2016, 16, 73. [Google Scholar] [CrossRef]
- Sims, D.; Sudbery, I.; Ilott, N.E.; Heger, A.; Ponting, C.P. Sequencing depth and coverage: Key considerations in genomic analyses. Nat. Rev. Genet. 2014, 15, 121–132. [Google Scholar] [CrossRef]
- Dillies, M.-A.; Rau, A.; Aubert, J.; Hennequet-Antier, C.; Jeanmougin, M.; Servant, N.; Keime, C.; Marot, G.; Castel, D.; Estelle, J.; et al. A comprehensive evaluation of normalization methods for Illumina high-throughput RNA sequencing data analysis. Brief. Bioinform. 2013, 14, 671–683. [Google Scholar] [CrossRef]
- Paulson, J.N.; Stine, O.O.; Bravo, H.O.; Pop, M. Robust methods for differential analysis in marker gene surveys. Nat. Methods 2013, 10, 1200–1202. [Google Scholar] [CrossRef]
- Weiss, S.J.; Xu, Z.; Amir, A.; Peddada, S.; Bittinger, K.; Gonzalez, A.; Lozupone, C.; Zaneveld, J.R.; Vazquez-Baeza, Y.; Birmingham, A.; et al. Effect of library size variance, sparity, and compositionality on the analysis of microbiome data. Peer J. PrePrints 2015, 3, e1157v1. [Google Scholar]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 002832. [Google Scholar] [CrossRef]
- McMurdie, P.J.; Holmes, S. Waste Not, Want Not: Why Rarefying Microbiome Data Is Inadmissible. PLoS Comput. Boil. 2014, 10, e1003531. [Google Scholar] [CrossRef] [PubMed]
- Costea, P.I.; Zeller, G.; Sunagawa, S.; Bork, P. A fair comparison. Nat. Methods 2014, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, R.H. Evolution and Measurement of Species Diversity. Taxon 1972, 21, 213–251. [Google Scholar] [CrossRef]
- Magurran, A.E.; McGill, B.J. Biological Diversity; Anne, E., Ed.; Oxford University Press: Oxford, UK, 2011. [Google Scholar]
- Mbareche, H.; Veillette, M.; Pilote, J.; Létourneau, V.; Duchaine, C. Corrélation Entre la Diversité Bactérienne des Bioaérosols de Bâtiments de Porcheries et des Nasopharynx des Travailleurs Exposés. In Proceedings of the Journées de la recherché 2018 de l’Institut Universitaire de Cardiologie et de Pneumologie de Québec, Québec, QC, Canada, 30 May 2018. [Google Scholar]
- Faith, D.P.; Baker, A.M. Phylogenetic Diversity (PD) and Biodiversity Conservation: Some Bioinformatics Challenges. Evol. Bioinform. 2006, 2, 121–128. [Google Scholar] [CrossRef]
- Nilsson, R.H.; Kristiansson, E.; Ryberg, M.; Hallenberg, N.; Larsson, K.-H. IntraspecificITSVariability in the KingdomFungias Expressed in the International Sequence Databases and Its Implications for Molecular Species Identification. Evol. Bioinform. 2008, 4, 193–201. [Google Scholar] [CrossRef]
- Tuomisto, H. A diversity of beta diversities: Straightening up a concept gone awry. Part 1. Defining beta diversity as a function of alpha and gamma diversity. Ecography 2010, 33, 2–22. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Knight, R. Global patterns in bacterial diversity. Proc. Natl. Acad. Sci. USA 2007, 104, 11436–11440. [Google Scholar] [CrossRef]
- Kuczynski, J.; Liu, Z.; Lozupone, C.; McDonald, D.; Fierer, N.; Knight, R. Microbial community resemblance methods differ in their ability to detect biologically relevant patterns. Nat. Methods 2010, 7, 813–819. [Google Scholar] [CrossRef]
- Leprieur, F.; Albouy, C.; De Bortoli, J.; Cowman, P.F.; Bellwood, D.R.; Mouillot, D. Quantifying phylogenetic beta diversity: Distinguishing between ‘true’ turnover of lineages and phylogenetic diversity gradients. PLoS ONE 2012, 7, e42760. [Google Scholar] [CrossRef]
- Wang, J.; Shen, J.; Wu, Y.; Tu, C.; Soininen, J.; Stegen, J.; He, J.-Z.; Liu, X.; Zhang, L.; Zhang, E. Phylogenetic beta diversity in bacterial assemblages across ecosystems: Deterministic versus stochastic processes. ISME J. 2013, 7, 1310–1321. [Google Scholar] [CrossRef]
- Navas-Molina, J.A.; Peralta-Sánchez, J.M.; González, A.; McMurdie, P.J.; Vázquez-Baeza, Y.; Xu, Z.; Ursell, L.K.; Lauber, C.; Zhou, H.; Song, S.J.; et al. Advancing Our Understanding of the Human Microbiome Using QIIME. Meth. Enzymol. 2013, 531, 371–444. [Google Scholar] [PubMed]
- Lozupone, C.; Hamady, M.; Knight, R. UniFrac – An online tool for comparing microbial community diversity in a phylogenetic context. BMC Bioinform. 2006, 7, 371. [Google Scholar] [CrossRef] [PubMed]
- Bray, J.R.; Curtis, J.T. An Ordination of the Upland Forest Communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Rokach, L.; Maimon, O. Clustering methods. Data Mining and Knowledge Discovery Handbook; Springer: New York, NY, USA, 2005. [Google Scholar]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analyses for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Ramette, A. Multivariate analyses in microbial ecology. FEMS Microbiol. Ecol. 2007, 62, 142–160. [Google Scholar] [CrossRef]
- Zur, E.F.; Ieno, E.N.; Smith, G.M. Analyzing Ecological Data; Springer: New York, NY, USA, 2007. [Google Scholar]
- Buttigieg, P.L.; Ramette, A. A guide to statistical analysis in microbial ecology: A community-focused, living review of multivariate data analyses. FEMS Microbiol. Ecol. 2014, 90, 543–550. [Google Scholar] [CrossRef]
- Ter Braak, C.J.; Verdonschot, P.F.M. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- McArdle, B.H.; Anderson, M.J. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Boil. Evol. 1985, 4, 406–425. [Google Scholar]
- Saltelli, A.; Marivoet, J. Non-parametric statistics in sensitivity analysis for model output: A comparison of selected techniques. Reliab. Eng. Syst. Saf. 1990, 28, 229–253. [Google Scholar] [CrossRef]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar]
- Razali, N.M. Power comparisons of Shapiro-Wilk, Kolmogorov-Smirnov, Lilliefors, Anderson-Darling tests. J. Stat. Mod. Anal. 2011, 2, 21–23. [Google Scholar]
- García, S.; Fernández, A.; Luengo, J.; Herrera, F. Advanced nonparametric tests for multiple comparisons in the design of experiments in computational intelligence and data mining: Experimental analysis of power. Inf. Sci. 2010, 180, 2044–2064. [Google Scholar] [CrossRef]
- Zimmerman, D.W. A note one the influence of outliers on parametric and nonparametric tests. J. Gen. Psychol. 1994, 121, 391–401. [Google Scholar] [CrossRef]
- Kwak, S.G.; Kim, J.H. Central limit theorem: The cornerstone of modern statistics. Korean J. Anesthesiol. 2017, 70, 144–156. [Google Scholar] [CrossRef]
- Erceg-Hurn, D.M.; Mirosevich, V.M. Modern robust statistical methods: An easy way to maximize the accuracy and power of your research. Am. Psychol. 2008, 63, 591–601. [Google Scholar] [CrossRef]
- Legendre, P.; Anderson, M.J. Distance-based redundancy analysis: Testing multispecies responses in multifactorial ecological experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- Anderson, M.J. PERMANOVA: A Fortran Computer Program for Permutational Multivariate ANALysis of Variance. Ph.D. Thesis, Department of Statistics, University of Auckland, Auckland, New Zealand, 2005; p. 24. [Google Scholar]
- Berry, K.J.; Wong, R.K.W. Approximate mrpp p-values obtained from four exact moments. Commun. Stat. Simul. Comput. 1986, 15, 581–589. [Google Scholar] [CrossRef]
- Anderson, M.J.; Walsh, D.C.I. Permanova, Anosim, and the Mantel test in the face of heterogeneous dispersions: What null hypothesis are you testing? Ecol. Monogr. 2013, 83, 557–574. [Google Scholar] [CrossRef]
- McDonald, J.H. Handbook of Biological Statistics, 3rd ed.; Sparky House Publishing: Baltimore, MD, USA, 2014. [Google Scholar]
- Poff, N.L.; Olden, J.D.; Merritt, D.M.; Pepin, D.M. Homogenization of regional river dynamics by dams and global biodiversity implications. Proc. Natl. Acad. Sci. USA 2007, 104, 5732–5737. [Google Scholar] [CrossRef]
- Clarke, K.R.; Somerfield, P.J.; Gorley, R.N. Testing of null hypotheses in exploratory community analyses: Similarity profiles and biota-environment linkage. J. Exp. Mar. Boil. Ecol. 2008, 366, 56–69. [Google Scholar] [CrossRef]
- Junior, G.B.; Paiva, A.C.; Silva, A.C.; Oliveira, A.C. Classification of breast tissues using Moran’s index and Geary’s coefficient as texture signatures and SVM. Comput. Boil. Med. 2009, 39, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Legendre, P.; Fortin, M.J. Comparison of the Mantel test and alternative approaches for detecting complex multivariate relationships in the spatial analyses of genetic analyses. Mol. Ecol. Resour. 2010, 10, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Sokal, R.R.; Rohlf, F.J. The Principles and Practices of Statistics in Biological Research, 4th ed.; Stony Brook University; WH Freeman and Company: New York, NY, USA, 1995. [Google Scholar]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, A Chimera-Checked 16S rRNA Gene Database and Workbench Compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [PubMed]
- Kõljalg, U.; Larsson, K.-H.; Abarenkov, K.; Nilsson, R.H.; Alexander, I.J.; Eberhardt, U.; Erland, S.; Høiland, K.; Kjøller, R.; Larsson, E.; et al. UNITE: A database providing web-based methods for the molecular identification of ectomycorrhizal fungi. New Phytol. 2005, 166, 1063–1068. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Gevers, D.; Kugathasan, S.; Denson, L.A.; Vázquez-Baeza, Y.; Van Treuren, W.; Ren, B.; Schwager, E.; Knights, D.; Song, S.J.; Yassour, M.; et al. The treatment-naïve microbiome in new-onset Crohn’s disease. Cell Host Microbe 2014, 15, 382–392. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar] [CrossRef]
- Shade, A.; Handelsman, J. Beyond the Venn diagram: The hunt for a core microbiome. Environ. Microbiol. 2011, 14, 4–12. [Google Scholar] [CrossRef]
- Anders, S.; Huber, W. Differential expression analysis sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Hollander, M.; Wolfe, D.A.; Chicken, E. Nonparametric Statistical Methods, 2nd ed.; John Wiley and Sons Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Callahan, B.J.; McMurdie, P.J.; Holmes, S.P. Exact sequence variants should replace operational taxonomic units in marker-gene data analyses. ISME J. 2017, 11, 2639–2643. [Google Scholar] [CrossRef]

| Methods | Type of Statistics | Type of Variables | Comment |
|---|---|---|---|
| Adonis | Nonparametric | Categorical and Numerical | Semi-metric and non-metric dissimilarities |
| ANOSIM | Nonparametric | Categorical | - |
| BIO-ENV | N/A | Numerical (continuous or discrete) | Rank-correlation between Euclidean distances and distance matrix |
| Moran’s I | N/A | Numerical | Identify spatial configuration in samples |
| MRPP | Nonparametric | Categorical | - |
| PERMANOVA | Nonparametric | Categorical | Uses an ANOVA experimental design and returns pseudo-F and a p-value |
| PERMDISP | Parametric and nonparametric | Categorical | Analysis of multivariate homogeneity of variances |
| db-RDA | Nonparametric | Categorical | A category in the metadata can be specified to explain the variability between samples |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mbareche, H.; Dumont-Leblond, N.; Bilodeau, G.J.; Duchaine, C. An Overview of Bioinformatics Tools for DNA Meta-Barcoding Analysis of Microbial Communities of Bioaerosols: Digest for Microbiologists. Life 2020, 10, 185. https://doi.org/10.3390/life10090185
Mbareche H, Dumont-Leblond N, Bilodeau GJ, Duchaine C. An Overview of Bioinformatics Tools for DNA Meta-Barcoding Analysis of Microbial Communities of Bioaerosols: Digest for Microbiologists. Life. 2020; 10(9):185. https://doi.org/10.3390/life10090185
Chicago/Turabian StyleMbareche, Hamza, Nathan Dumont-Leblond, Guillaume J. Bilodeau, and Caroline Duchaine. 2020. "An Overview of Bioinformatics Tools for DNA Meta-Barcoding Analysis of Microbial Communities of Bioaerosols: Digest for Microbiologists" Life 10, no. 9: 185. https://doi.org/10.3390/life10090185
APA StyleMbareche, H., Dumont-Leblond, N., Bilodeau, G. J., & Duchaine, C. (2020). An Overview of Bioinformatics Tools for DNA Meta-Barcoding Analysis of Microbial Communities of Bioaerosols: Digest for Microbiologists. Life, 10(9), 185. https://doi.org/10.3390/life10090185






