Muscle Hypertrophy and Architectural Changes in Response to Eight-Week Neuromuscular Electrical Stimulation Training in Healthy Older People
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Experimental Design
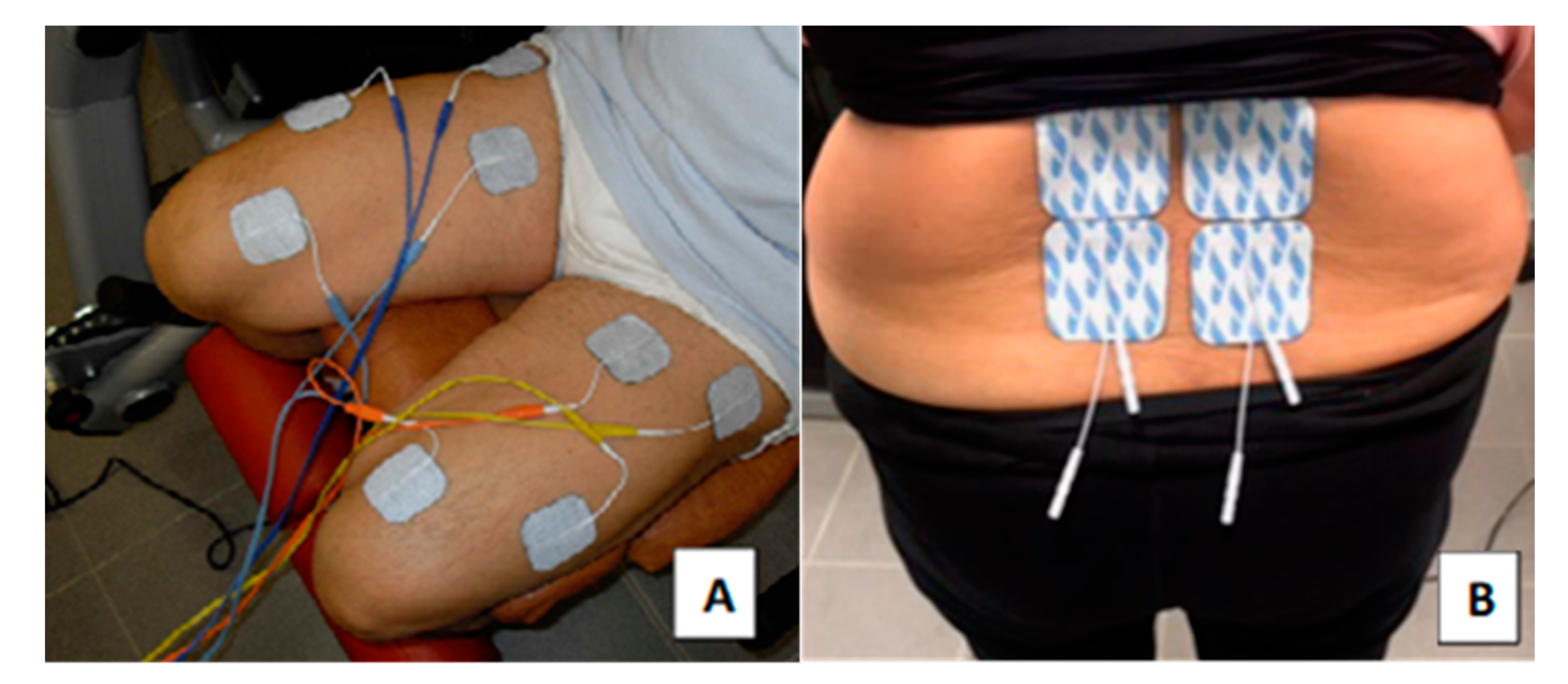
4.3. Combined NMES Training
4.4. NMES of the Quadriceps Muscle Group
4.5. NMES Training of Lumbar Paraspinal Muscles
4.6. Functional Tests
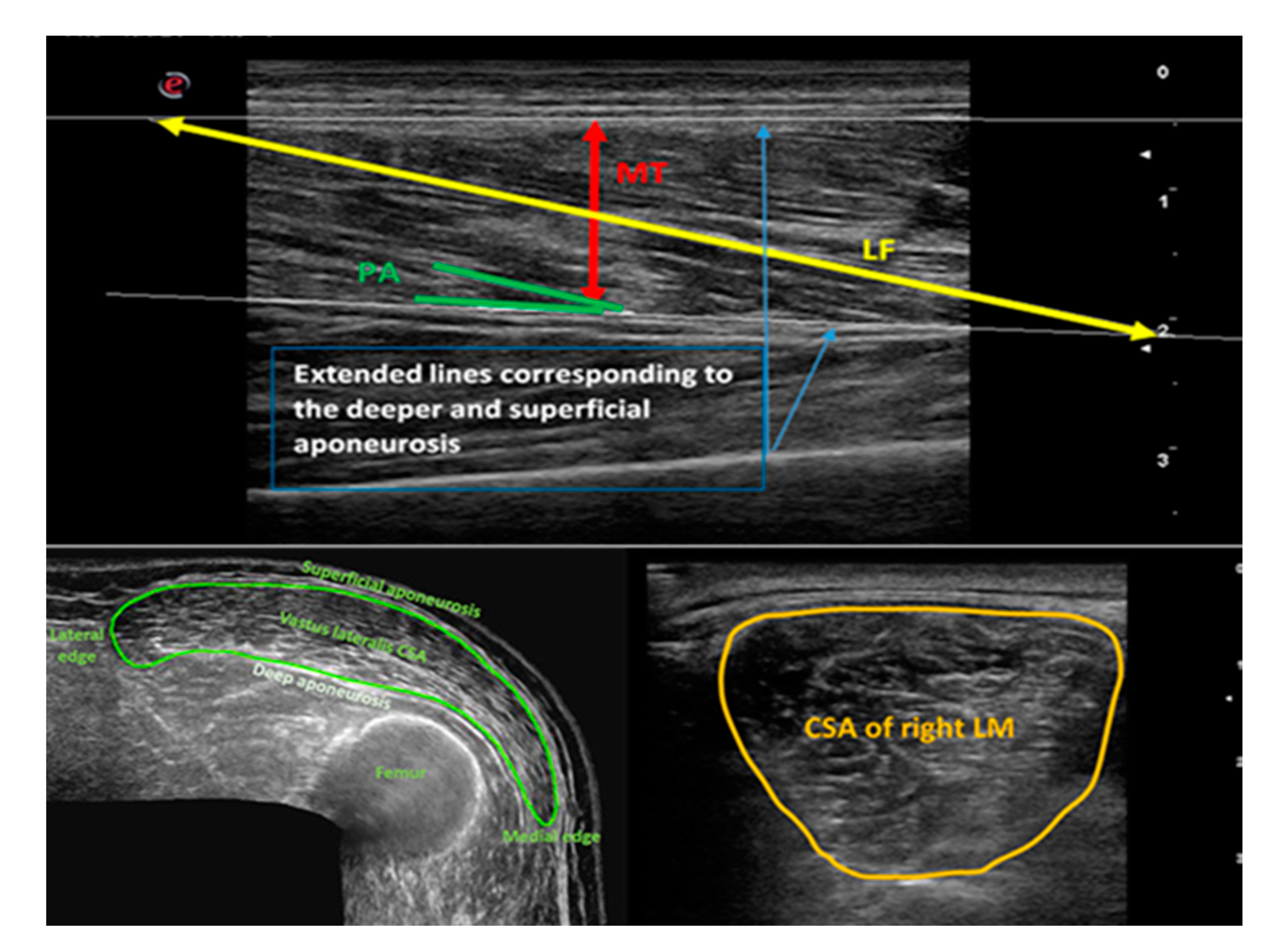
4.7. The Procedure of Ultrasound Scanning
4.8. Analysis of Ultrasound Images
4.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Faulkner, J.A.; Larkin, L.M.; Claflin, D.R.; Brooks, S.V. Age-related changes in the structure and function of skeletal muscles. Clin. Exp. Pharmacol. Physiol. 2007, 34, 1091–1096. [Google Scholar] [CrossRef]
- Morley, J.E. Sarcopenia in the elderly. Fam. Pract. 2012, 29, i44–i48. [Google Scholar] [CrossRef]
- Metter, E.J.; Lynch, N.; Conwit, R.; Lindle, R.; Tobin, J.; Hurley, B. Muscle quality and age: Cross-sectional and longitudinal comparisons. J. Gerontol. A Biol. Sci. Med. Sci. 1999, 54, B207–B218. [Google Scholar] [CrossRef]
- von Haehling, S.; Morley, J.E.; Anker, S.D. An overview of sarcopenia: Facts and numbers on prevalence and clinical impact. J. Cachexia Sarcopenia Muscle 2010, 1, 129–133. [Google Scholar] [CrossRef]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Young, A.; Stokes, M.; Crowe, M. The size and strength of the quadriceps muscles of old and young men. Clin. Physiol. 1985, 5, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Overend, T.J.; Cunningham, D.A.; Kramer, J.F.; Lefcoe, M.S.; Paterson, D.H. Knee extensor and knee flexor strength: Cross-sectional area ratios in young and elderly men. J. Gerontol. 1992, 47, M204–M210. [Google Scholar] [CrossRef] [PubMed]
- Fortin, M.; Videman, T.; Gibbons, L.E.; Battie, M.C. Paraspinal muscle morphology and composition: A 15-yr longitudinal magnetic resonance imaging study. Med. Sci. Sports Exerc. 2014, 46, 893–901. [Google Scholar] [CrossRef]
- Lexell, J.; Taylor, C.C.; Sjostrom, M. What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 1988, 84, 275–294. [Google Scholar] [CrossRef]
- Valentin, S.; Licka, T.; Elliott, J. Age and side-related morphometric MRI evaluation of trunk muscles in people without back pain. Man. Ther. 2015, 20, 90–95. [Google Scholar] [CrossRef]
- Chen, Z.N.; Yao, X.M.; Lv, Y.; He, B.J.; Ye, J.C.; Shao, R.X.; Jiang, H.W. Morphology of the lumbar multifidus muscle in lumbar disc herniation at different durations and at different ages. Exp. Ther. Med. 2018, 15, 4119–4126. [Google Scholar] [CrossRef]
- Jakobsson, F.; Borg, K.; Edstrom, L. Fibre-type composition, structure and cytoskeletal protein location of fibres in anterior tibial muscle. Comparison between young adults and physically active aged humans. Acta Neuropathol. 1990, 80, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Stone, M.H.; Stone, M.; Sands, W.A. Principles and Practice of Resistance Training; Human Kinetics, Inc.: Champaign, IL, USA, 2007. [Google Scholar]
- Doucet, B.M.; Lam, A.; Griffin, L. Neuromuscular electrical stimulation for skeletal muscle function. Yale J. Biol. Med. 2012, 85, 201–215. [Google Scholar] [PubMed]
- Maffiuletti, N.A.; Gondin, J.; Place, N.; Stevens-Lapsley, J.; Vivodtzev, I.; Minetto, M.A. Clinical Use of Neuromuscular Electrical Stimulation for Neuromuscular Rehabilitation: What Are We Overlooking? Arch. Phys. Med. Rehabil. 2018, 99, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Adams, V. Electromyostimulation to fight atrophy and to build muscle: Facts and numbers. J. Cachexia Sarcopenia Muscle 2018, 9, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Dirks, M.L.; Wall, B.T.; Snijders, T.; Ottenbros, C.L.; Verdijk, L.B.; van Loon, L.J. Neuromuscular electrical stimulation prevents muscle disuse atrophy during leg immobilization in humans. Acta. Physiol. (Oxf.) 2014, 210, 628–641. [Google Scholar] [CrossRef] [PubMed]
- Babault, N.; Cometti, G.; Bernardin, M.; Pousson, M.; Chatard, J.C. Effects of electromyostimulation training on muscle strength and power of elite rugby players. J. Strength Cond. Res. 2007, 21, 431–437. [Google Scholar] [CrossRef][Green Version]
- Maffiuletti, N.A.; Roig, M.; Karatzanos, E.; Nanas, S. Neuromuscular electrical stimulation for preventing skeletal-muscle weakness and wasting in critically ill patients: A systematic review. BMC. Med. 2013, 11, 137. [Google Scholar] [CrossRef]
- Wall, B.T.; Dirks, M.L.; Verdijk, L.B.; Snijders, T.; Hansen, D.; Vranckx, P.; Burd, N.A.; Dendale, P.; van Loon, L.J. Neuromuscular electrical stimulation increases muscle protein synthesis in elderly type 2 diabetic men. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E614–E623. [Google Scholar] [CrossRef]
- Gondin, J.; Brocca, L.; Bellinzona, E.; D’Antona, G.; Maffiuletti, N.A.; Miotti, D.; Pellegrino, M.A.; Bottinelli, R. Neuromuscular electrical stimulation training induces atypical adaptations of the human skeletal muscle phenotype: A functional and proteomic analysis. J. Appl. Physiol. (1985) 2011, 110, 433–450. [Google Scholar] [CrossRef]
- Saini, A.; Faulkner, S.; Al-Shanti, N.; Stewart, C. Powerful signals for weak muscles. Ageing Res. Rev. 2009, 8, 251–267. [Google Scholar] [CrossRef] [PubMed]
- Di Filippo, E.S.; Mancinelli, R.; Marrone, M.; Doria, C.; Verratti, V.; Toniolo, L.; Dantas, J.L.; Fulle, S.; Pietrangelo, T. Neuromuscular electrical stimulation improves skeletal muscle regeneration through satellite cell fusion with myofibers in healthy elderly subjects. J. Appl. Physiol. (1985) 2017, 123, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Kern, H.; Barberi, L.; Lofler, S.; Sbardella, S.; Burggraf, S.; Fruhmann, H.; Carraro, U.; Mosole, S.; Sarabon, N.; Vogelauer, M.; et al. Electrical stimulation counteracts muscle decline in seniors. Front. Aging Neurosci. 2014, 6, 189. [Google Scholar] [CrossRef] [PubMed]
- Veldman, M.P.; Gondin, J.; Place, N.; Maffiuletti, N.A. Effects of Neuromuscular Electrical Stimulation Training on Endurance Performance. Front. Physiol. 2016, 7, 544. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.M.; Bickel, C.S. Recruitment patterns in human skeletal muscle during electrical stimulation. Phys. Ther. 2005, 85, 358–364. [Google Scholar] [CrossRef]
- Talbot, L.A.; Gaines, J.M.; Ling, S.M.; Metter, E.J. A home-based protocol of electrical muscle stimulation for quadriceps muscle strength in older adults with osteoarthritis of the knee. J. Rheumatol. 2003, 30, 1571–1578. [Google Scholar]
- Marqueste, T.; Hug, F.; Decherchi, P.; Jammes, Y. Changes in neuromuscular function after training by functional electrical stimulation. Muscle Nerve 2003, 28, 181–188. [Google Scholar] [CrossRef]
- Maffiuletti, N.A. Physiological and methodological considerations for the use of neuromuscular electrical stimulation. Eur. J. Appl. Physiol. 2010, 110, 223–234. [Google Scholar] [CrossRef]
- Allen, G.M. Shoulder ultrasound imaging-integrating anatomy, biomechanics and disease processes. Eur. J. Radiol. 2008, 68, 137–146. [Google Scholar] [CrossRef]
- Bianchi, S.; Marcelis, S. Musculoskeletal sonography. In Musculoskeletal Diseases: Diagnostic Imaging and Interventional Techniques; Springer: New York, NY, USA, 2005. [Google Scholar]
- Jain, M.; Samuels, J. Musculoskeletal ultrasound as a diagnostic and prognostic tool in rheumatoid arthritis. Bull. NYU. Hosp. Jt. Dis. 2011, 69, 215–219. [Google Scholar]
- Naredo, E.; Uson, J.; Jimenez-Palop, M.; Martinez, A.; Vicente, E.; Brito, E.; Rodriguez, A.; Cornejo, F.J.; Castaneda, S.; Martinez, M.J.; et al. Ultrasound-detected musculoskeletal urate crystal deposition: Which joints and what findings should be assessed for diagnosing gout? Ann. Rheum. Dis. 2014, 73, 1522–1528. [Google Scholar] [CrossRef] [PubMed]
- Nofsinger, C.; Konin, J.G. Diagnostic ultrasound in sports medicine: Current concepts and advances. Sports. Med. Arthrosc. Rev. 2009, 17, 25–30. [Google Scholar] [CrossRef]
- Padua, L.; Liotta, G.; Di Pasquale, A.; Granata, G.; Pazzaglia, C.; Caliandro, P.; Martinoli, C. Contribution of ultrasound in the assessment of nerve diseases. Eur. J. Neurol. 2012, 19, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Pillen, S.; van Alfen, N. Skeletal muscle ultrasound. Neurol. Res. 2011, 33, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, M.; Kinel, E.; D’Amico, G.; Roncoletta, P. A 3D Spine and Full Skeleton Model for Opto-Electronic Stereo- Photogrammetric Multi-Sensor Biomechanical Analysis in Posture and Gait. In Innovations in Spinal Deformities and Postural Disorders; Bettany-Saltikov, J., Ed.; IntechOpen Limited: London, UK, 2017. [Google Scholar] [CrossRef]
- de Boer, M.D.; Seynnes, O.R.; di Prampero, P.E.; Pisot, R.; Mekjavic, I.B.; Biolo, G.; Narici, M.V. Effect of 5 weeks horizontal bed rest on human muscle thickness and architecture of weight bearing and non-weight bearing muscles. Eur. J. Appl. Physiol. 2008, 104, 401–407. [Google Scholar] [CrossRef]
- Narici, M.V.; Maganaris, C.N.; Reeves, N.D.; Capodaglio, P. Effect of aging on human muscle architecture. J. Appl. Physiol. (1985) 2003, 95, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- Seynnes, O.R.; de Boer, M.; Narici, M.V. Early skeletal muscle hypertrophy and architectural changes in response to high-intensity resistance training. J. Appl. Physiol. (1985) 2007, 102, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Altubasi, I.M. The Effect of Neuromuscular Electrical Stimulation (NMES) in Inducing Muscle Hypertrophy and Improvement in Muscle Torque within the Quadriceps Muscle of Elderly People. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, 2012. [Google Scholar]
- Langeard, A.; Bigot, L.; Chastan, N.; Gauthier, A. Does neuromuscular electrical stimulation training of the lower limb have functional effects on the elderly? A systematic review. Exp. Gerontol. 2017, 91, 88–98. [Google Scholar] [CrossRef]
- Abe, T.; Loenneke, J.P.; Thiebaud, R.S. Morphological and functional relationships with ultrasound measured muscle thickness of the lower extremity: A brief review. Ultrasound 2015, 23, 166–173. [Google Scholar] [CrossRef]
- Cooper, C.; Fielding, R.; Visser, M.; van Loon, L.J.; Rolland, Y.; Orwoll, E.; Reid, K.; Boonen, S.; Dere, W.; Epstein, S.; et al. Tools in the assessment of sarcopenia. Calcif. Tissue Int. 2013, 93, 201–210. [Google Scholar] [CrossRef]
- Reeves, N.D.; Maganaris, C.N.; Narici, M.V. Ultrasonographic assessment of human skeletal muscle size. Eur. J. Appl. Physiol. 2004, 91, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Franchi, M.V.; Longo, S.; Mallinson, J.; Quinlan, J.I.; Taylor, T.; Greenhaff, P.L.; Narici, M.V. Muscle thickness correlates to muscle cross-sectional area in the assessment of strength training-induced hypertrophy. Scand. J. Med. Sci. Sports 2018, 28, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Buckinx, F.; Landi, F.; Cesari, M.; Fielding, R.A.; Visser, M.; Engelke, K.; Maggi, S.; Dennison, E.; Al-Daghri, N.M.; Allepaerts, S.; et al. Pitfalls in the measurement of muscle mass: A need for a reference standard. J. Cachexia Sarcopenia Muscle 2018, 9, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Mijnarends, D.M.; Meijers, J.M.; Halfens, R.J.; ter Borg, S.; Luiking, Y.C.; Verlaan, S.; Schoberer, D.; Cruz Jentoft, A.J.; van Loon, L.J.; Schols, J.M. Validity and reliability of tools to measure muscle mass, strength, and physical performance in community-dwelling older people: A systematic review. J. Am. Med. Dir. Assoc. 2013, 14, 170–178. [Google Scholar] [CrossRef]
- Frontera, W.R.; Meredith, C.N.; O’Reilly, K.P.; Knuttgen, H.G.; Evans, W.J. Strength conditioning in older men: Skeletal muscle hypertrophy and improved function. J. Appl. Physiol. (1985) 1988, 64, 1038–1044. [Google Scholar] [CrossRef]
- Charette, S.L.; McEvoy, L.; Pyka, G.; Snow-Harter, C.; Guido, D.; Wiswell, R.A.; Marcus, R. Muscle hypertrophy response to resistance training in older women. J. Appl. Physiol. (1985) 1991, 70, 1912–1916. [Google Scholar] [CrossRef]
- Fiatarone, M.A.; Marks, E.C.; Ryan, N.D.; Meredith, C.N.; Lipsitz, L.A.; Evans, W.J. High-intensity strength training in nonagenarians. Effects on skeletal muscle. JAMA 1990, 263, 3029–3034. [Google Scholar] [CrossRef]
- Hides, J.A.; Stokes, M.J.; Saide, M.; Jull, G.A.; Cooper, D.H. Evidence of lumbar multifidus muscle wasting ipsilateral to symptoms in patients with acute/subacute low back pain. Spine (Phila Pa 1976) 1994, 19, 165–172. [Google Scholar] [CrossRef]
- Lee, S.W.; Chan, C.K.; Lam, T.S.; Lam, C.; Lau, N.C.; Lau, R.W.; Chan, S.T. Relationship between low back pain and lumbar multifidus size at different postures. Spine (Phila Pa 1976) 2006, 31, 2258–2262. [Google Scholar] [CrossRef]
- Sajer, S.; Guardiero, G.S.; Scicchitano, B.M. Myokines in Home-Based Functional Electrical Stimulation-Induced Recovery of Skeletal Muscle in Elderly and Permanent Denervation. Eur. J. Transl. Myol. 2018, 28, 7905. [Google Scholar] [CrossRef]
- Coghlan, S.; Crowe, L.; McCarthypersson, U.; Minogue, C.; Caulfield, B. Neuromuscular electrical stimulation training results in enhanced activation of spinal stabilizing muscles during spinal loading and improvements in pain ratings. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2011, 2011, 7622–7625. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, M.; Kinel, E.; Roncoletta, P. Normative 3D opto-electronic stereo-photogrammetric posture and spine morphology data in young healthy adult population. PLoS ONE 2017, 12, e0179619. [Google Scholar] [CrossRef] [PubMed]
- Narici, M.; Franchi, M.; Maganaris, C. Muscle structural assembly and functional consequences. J. Exp. Biol. 2016, 219, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Ema, R.; Akagi, R.; Wakahara, T.; Kawakami, Y. Training-induced changes in architecture of human skeletal muscles: Current evidence and unresolved issues. J. Phys. Fit. Sports Med. 2016, 5, 37–46. [Google Scholar] [CrossRef]
- Franchi, M.V.; Atherton, P.J.; Reeves, N.D.; Fluck, M.; Williams, J.; Mitchell, W.K.; Selby, A.; Beltran Valls, R.M.; Narici, M.V. Architectural, functional and molecular responses to concentric and eccentric loading in human skeletal muscle. Acta Physiol. (Oxf.) 2014, 210, 642–654. [Google Scholar] [CrossRef]
- Reeves, N.D.; Maganaris, C.N.; Longo, S.; Narici, M.V. Differential adaptations to eccentric versus conventional resistance training in older humans. Exp. Physiol. 2009, 94, 825–833. [Google Scholar] [CrossRef]
- Raj, I.S.; Bird, S.R.; Westfold, B.A.; Shield, A.J. Effects of eccentrically biased versus conventional weight training in older adults. Med. Sci. Sports Exerc. 2012, 44, 1167–1176. [Google Scholar] [CrossRef]
- Scanlon, T.C.; Fragala, M.S.; Stout, J.R.; Emerson, N.S.; Beyer, K.S.; Oliveira, L.P.; Hoffman, J.R. Muscle architecture and strength: Adaptations to short-term resistance training in older adults. Muscle Nerve 2014, 49, 584–592. [Google Scholar] [CrossRef]
- Noorkoiv, M.; Nosaka, K.; Blazevich, A.J. Neuromuscular adaptations associated with knee joint angle-specific force change. Med. Sci. Sports Exerc. 2014, 46, 1525–1537. [Google Scholar] [CrossRef]
- Steffl, M.; Bohannon, R.W.; Sontakova, L.; Tufano, J.J.; Shiells, K.; Holmerova, I. Relationship between sarcopenia and physical activity in older people: A systematic review and meta-analysis. Clin. Interv. Aging 2017, 12, 835–845. [Google Scholar] [CrossRef]
- Kalyani, R.R.; Corriere, M.; Ferrucci, L. Age-related and disease-related muscle loss: The effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014, 2, 819–829. [Google Scholar] [CrossRef]
- Stokes, M.; Hides, J.; Elliott, J.; Kiesel, K.; Hodges, P. Rehabilitative ultrasound imaging of the posterior paraspinal muscles. J. Orthop. Sports Phys. Ther. 2007, 37, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Reference values for the timed up and go test: A descriptive meta-analysis. J. Geriatr. Phys. Ther. 2006, 29, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Reference values for the five-repetition sit-to-stand test: A descriptive meta-analysis of data from elders. Percept. Mot. Ski. 2006, 103, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Bickel, C.S.; Slade, J.M.; Haddad, F.; Adams, G.R.; Dudley, G.A. Acute molecular responses of skeletal muscle to resistance exercise in able-bodied and spinal cord-injured subjects. J. Appl. Physiol. (1985) 2003, 94, 2255–2262. [Google Scholar] [CrossRef]
- Strasser, E.M.; Stattner, S.; Karner, J.; Klimpfinger, M.; Freynhofer, M.; Zaller, V.; Graf, A.; Wessner, B.; Bachl, N.; Roth, E.; et al. Neuromuscular electrical stimulation reduces skeletal muscle protein degradation and stimulates insulin-like growth factors in an age- and current-dependent manner: A randomized, controlled clinical trial in major abdominal surgical patients. Ann. Surg. 2009, 249, 738–743. [Google Scholar] [CrossRef]
- Barber, L.; Scicchitano, B.M.; Musaro, A. Molecular and Cellular Mechanisms of Muscle Aging and Sarcopenia and Effects of Electrical Stimulation in Seniors. Eur. J. Transl. Myol. 2015, 25, 231–236. [Google Scholar] [CrossRef]
- Matheny, R.W., Jr.; Nindl, B.C.; Adamo, M.L. Minireview: Mechano-growth factor: A putative product of IGF-I gene expression involved in tissue repair and regeneration. Endocrinology 2010, 151, 865–875. [Google Scholar] [CrossRef]
- Mancinelli, R.; Toniolo, L.; Di Filippo, E.S.; Doria, C.; Marrone, M.; Maroni, C.R.; Verratti, V.; Bondi, D.; Maccatrozzo, L.; Pietrangelo, T.; et al. Neuromuscular Electrical Stimulation Induces Skeletal Muscle Fiber Remodeling and Specific Gene Expression Profile in Healthy Elderly. Front. Physiol. 2019, 10, 1459. [Google Scholar] [CrossRef]
- Paillard, T. Muscle plasticity of aged subjects in response to electrical stimulation training and inversion and/or limitation of the sarcopenic process. Ageing Res. Rev. 2018, 46, 1–13. [Google Scholar] [CrossRef]
- Kemmler, W.; Weissenfels, A.; Teschler, M.; Willert, S.; Bebenek, M.; Shojaa, M.; Kohl, M.; Freiberger, E.; Sieber, C.; von Stengel, S. Whole-body electromyostimulation and protein supplementation favorably affect sarcopenic obesity in community-dwelling older men at risk: The randomized controlled FranSO study. Clin. Interv. Aging 2017, 12, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, L.M.; Weinberg, J.; Keaney, J.F., Jr. Common Statistical Pitfalls in Basic Science Research. J. Am. Heart Assoc. 2016, 5, e004142. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Robert, M.; Li, Y. Statistical methods and common problems in medical or biomedical science research. Int. J. Physiol. Pathophysiol. Pharmacol. 2017, 9, 157–163. [Google Scholar] [PubMed]
- Marmon, A.R.; Snyder-Mackler, L. Quantifying Neuromuscular Electrical Stimulation Dosage after Knee Arthroplasty. J. Life. Sci. (Lib.) 2011, 5, 581–583. [Google Scholar]
- Ando, R.; Taniguchi, K.; Saito, A.; Fujimiya, M.; Katayose, M.; Akima, H. Validity of fascicle length estimation in the vastus lateralis and vastus intermedius using ultrasonography. J. Electromyogr. Kinesiol. 2014, 24, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, T.; Oda, T.; Chino, K.; Kanehisa, H.; Fukunaga, T.; Kawakami, Y. Use of Three-Dimensional Ultrasonography for the Analysis of the Fascicle Length of Human Gastrocnemius Muscle during Contractions. Int. J. Sport. Stud. Hlth. 2005, 3, 226–234. [Google Scholar] [CrossRef]
- Kawakami, Y.; Abe, T.; Fukunaga, T. Muscle-fiber pennation angles are greater in hypertrophied than in normal muscles. J. Appl. Physiol. (1985) 1993, 74, 2740–2744. [Google Scholar] [CrossRef]
- Reeves, N.D.; Narici, M.V.; Maganaris, C.N. In vivo human muscle structure and function: Adaptations to resistance training in old age. Exp. Physiol. 2004, 89, 675–689. [Google Scholar] [CrossRef]
- World Medical, A. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Tanaka, H.; Monahan, K.D.; Seals, D.R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Baek, S.O.; Ahn, S.H.; Jones, R.; Cho, H.K.; Jung, G.S.; Cho, Y.W.; Tak, H.J. Activations of deep lumbar stabilizing muscles by transcutaneous neuromuscular electrical stimulation of lumbar paraspinal regions. Ann. Rehabil. Med. 2014, 38, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, J.H.; Jung, G.S.; Baek, S.O.; Jones, R.; Ahn, S.H. The effects of transcutaneous neuromuscular electrical stimulation on the activation of deep lumbar stabilizing muscles of patients with lumbar degenerative kyphosis. J. Phys. Ther. Sci. 2016, 28, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Glaviano, N.R.; Saliba, S. Can the Use of Neuromuscular Electrical Stimulation Be Improved to Optimize Quadriceps Strengthening? Sports Health 2016, 8, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Roselyn, C. Ortho 2: Iliac crest, L4-5 interspace, & Spinous process of YouTube. Available online: https://www.youtube.com/watch?v=HAMg0P-mu5s (accessed on 15 May 2004).
- Wallmann, H.W.; Evans, N.S.; Day, C.; Neelly, K.R. Interrater reliability of the Five-Times-Sit-to-Stand Test. Home Health Care Manag. Pract. 2013, 25, 13–17. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The timed “Up & Go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [CrossRef]
- Ticinesi, A.; Narici, M.V.; Lauretani, F.; Nouvenne, A.; Colizzi, E.; Mantovani, M.; Corsonello, A.; Landi, F.; Meschi, T.; Maggio, M. Assessing sarcopenia with vastus lateralis muscle ultrasound: An operative protocol. Aging Clin. Exp. Res. 2018, 30, 1437–1443. [Google Scholar] [CrossRef]
- Stokes, M.; Rankin, G.; Newham, D.J. Ultrasound imaging of lumbar multifidus muscle: Normal reference ranges for measurements and practical guidance on the technique. Man. Ther. 2005, 10, 116–126. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
| Intraclass Correlation Coefficient | 95% CI | ||
|---|---|---|---|
| Lower Limit | Upper Limit | ||
| CSA of VL | 0.988 | 0.849 | 0.998 |
| MT of VL | 0.994 | 0.948 | 0.999 |
| FL of VL | 0.971 | 0.752 | 0.995 |
| PA of VL | 0.948 | 0.577 | 0.990 |
| CSA of LM left | 0.970 | 0.854 | 0.994 |
| CSA of LM right | 0.994 | 0.974 | 0.999 |
| NMES n = 8 | CG n = 8 | p Value * | |
|---|---|---|---|
| Age (y) | 69.3 (3.2) | 68.0 (2.3) | 0.588 |
| Weight (kg) | 67.6 (15.9) | 80.1 (10.7) | 0.066 |
| Height (cm) | 160.1 (8.4) | 170.4 (9.2) | 0.058 |
| BMI (kg/m2) | 26.5 (3.8) | 27.9 (2.0) | 0.294 |
| NMES n = 8 | CG n = 8 | |||
|---|---|---|---|---|
| Pre- | Post- | Pre- | Post- | |
| MU Parameters | ||||
| CSA of VL (cm2) | 10.7 (4.6) | 12.2 (4.3) * | 12.8 (2.6) | 12.5 (2.6) * |
| MT (cm) | 1.7 (0.4) | 1.9 (0.4) * | 1.9 (0.2) | 1.8 (0.3) * |
| FL (cm) | 7.2 (0.5) | 7.3 (0.8) | 7.7 (0.7) | 7.5 (0.7) * |
| PA (in°) | 15.0 (3.9) | 15.0 (4.1) | 13.9 (3.2) | 13.6 (3.0) * |
| CSA of LM left (cm2) | 9.8 (2.7) | 10.4 (2.3) * | 10.7 (3.4) | 11.2 (4.0) |
| CSA of LM right (cm2) | 9.1 (2.1) | 9.2 (2.6) * | 10.3 (3.5) | 10.3 (3.4) |
| Functional Parameters | ||||
| FTSST (in secs) | 6.5 (2.1) | 6.5 (2.6) | 6.5 (0.4) | 6.6 (0.7) |
| TUG (in secs) | 4.7 (1.1) | 4.4 (1.5) | 4.6 (0.6) | 4.6 (0.5) |
| NMES n = 8 | CG n = 8 | p Value * | r | |
|---|---|---|---|---|
| MU Parameters | ||||
| CSA of VL | 11.3 (15.1) | −1.4 (4.0) | 0.006 | 0.68 |
| MT | 6.9 (6.9) | −2.2 (4.7) | 0.001 | 0.84 |
| FL | 2.1 (5.6) | −1.1 (4.2) | 0.046 | 0.50 |
| PA | 4.0 (9.0) | −0.6 (2.9) | 0.006 | 0.68 |
| CSA of LM left | 6.4 (8.2) | −0.6 (1.9) | 0.036 | 0.53 |
| CSA of LM right | 6.5 (9.8) | −0.8 (2.3) | 0.005 | 0.71 |
| Functional Parameters # | ||||
| FTSST | −0.03 (23.0) | 2.1 (11.7) | 0.345 | 0.24 |
| TUG | −10.9 (13.8) | 2.6 (6.5) | 0.046 | 0.50 |
| Combined NMES Training | Week | Duration | Frequency and Pulse | Intensity | Pre-Load | Load |
|---|---|---|---|---|---|---|
| Quadriceps | 1–8 w (3/w) | 18′ | 75 Hz Rectangular biphasic waves (400 µs) | Maximum tolerable intensity corresponded to the individual’s pain threshold | 5′ pedaling | 40 contractions of 6.25 s (rise time 1.5 s, steady time 4 s, fall time 0.75 s) |
| Lumbar paraspinal muscles | 1–8 w (3/w) | 15′ | 50 Hz Symmetrical biphasic waves (300 µs) | 5′ pedaling | 45 contractions of 10 s (rise time 1 s, steady time 8 s, fall time 1 s) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jandova, T.; Narici, M.V.; Steffl, M.; Bondi, D.; D’Amico, M.; Pavlu, D.; Verratti, V.; Fulle, S.; Pietrangelo, T. Muscle Hypertrophy and Architectural Changes in Response to Eight-Week Neuromuscular Electrical Stimulation Training in Healthy Older People. Life 2020, 10, 184. https://doi.org/10.3390/life10090184
Jandova T, Narici MV, Steffl M, Bondi D, D’Amico M, Pavlu D, Verratti V, Fulle S, Pietrangelo T. Muscle Hypertrophy and Architectural Changes in Response to Eight-Week Neuromuscular Electrical Stimulation Training in Healthy Older People. Life. 2020; 10(9):184. https://doi.org/10.3390/life10090184
Chicago/Turabian StyleJandova, Tereza, Marco V. Narici, Michal Steffl, Danilo Bondi, Moreno D’Amico, Dagmar Pavlu, Vittore Verratti, Stefania Fulle, and Tiziana Pietrangelo. 2020. "Muscle Hypertrophy and Architectural Changes in Response to Eight-Week Neuromuscular Electrical Stimulation Training in Healthy Older People" Life 10, no. 9: 184. https://doi.org/10.3390/life10090184
APA StyleJandova, T., Narici, M. V., Steffl, M., Bondi, D., D’Amico, M., Pavlu, D., Verratti, V., Fulle, S., & Pietrangelo, T. (2020). Muscle Hypertrophy and Architectural Changes in Response to Eight-Week Neuromuscular Electrical Stimulation Training in Healthy Older People. Life, 10(9), 184. https://doi.org/10.3390/life10090184










