Deregulated Serotonin Pathway in Women with Morbid Obesity and NAFLD
Abstract
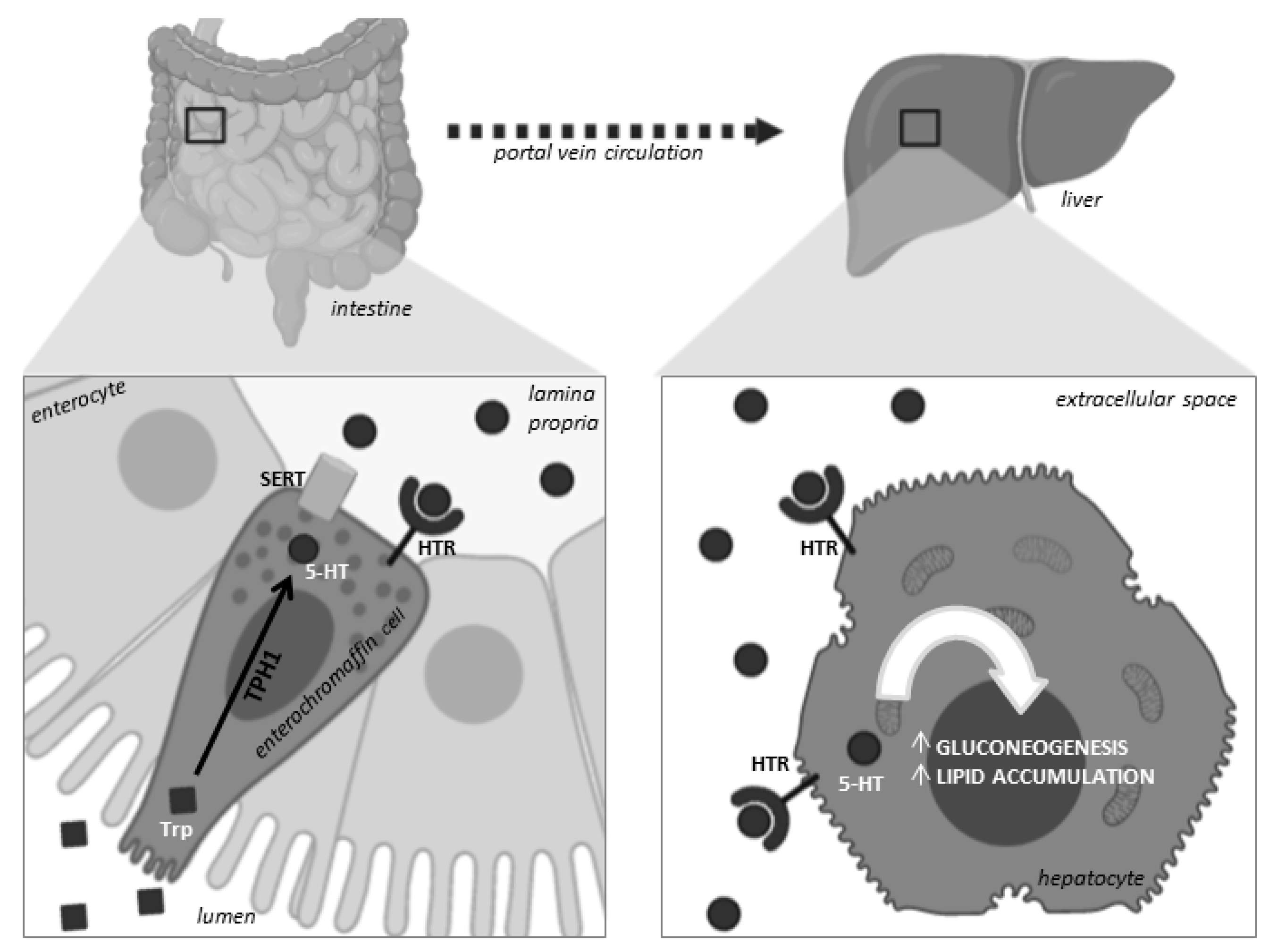
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Sample Size
2.3. Liver Pathological Features
2.4. Biochemical Analyses
2.5. mRNA Levels in the Liver and Jejunum
2.6. Statistical Analysis
3. Results
3.1. Baseline Characteristics of Subjects
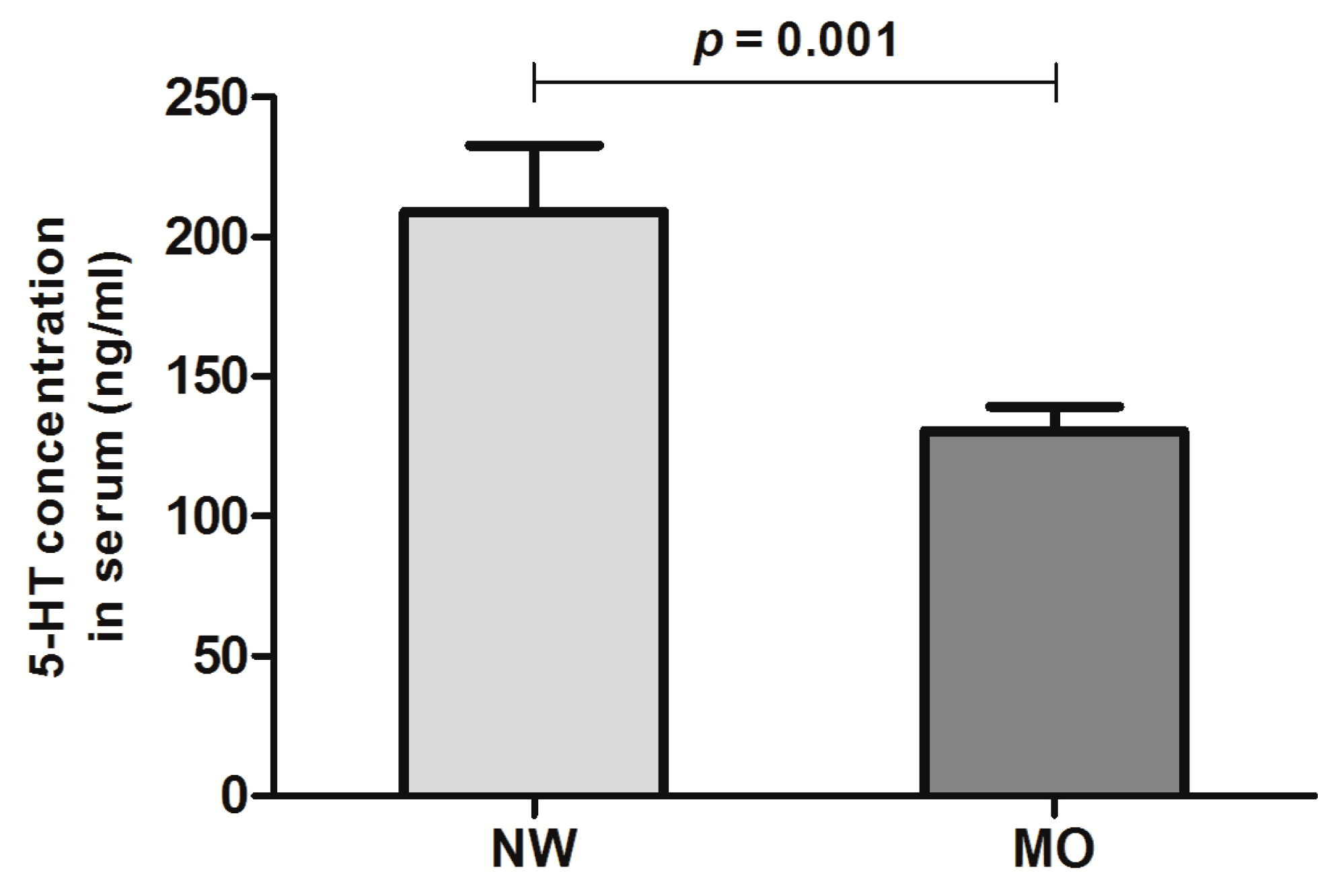
3.2. Peripheral Levels of Serotonin in the Studied Cohort
3.3. Correlations of Serum Levels of Serotonin with Metabolic Syndrome Presence
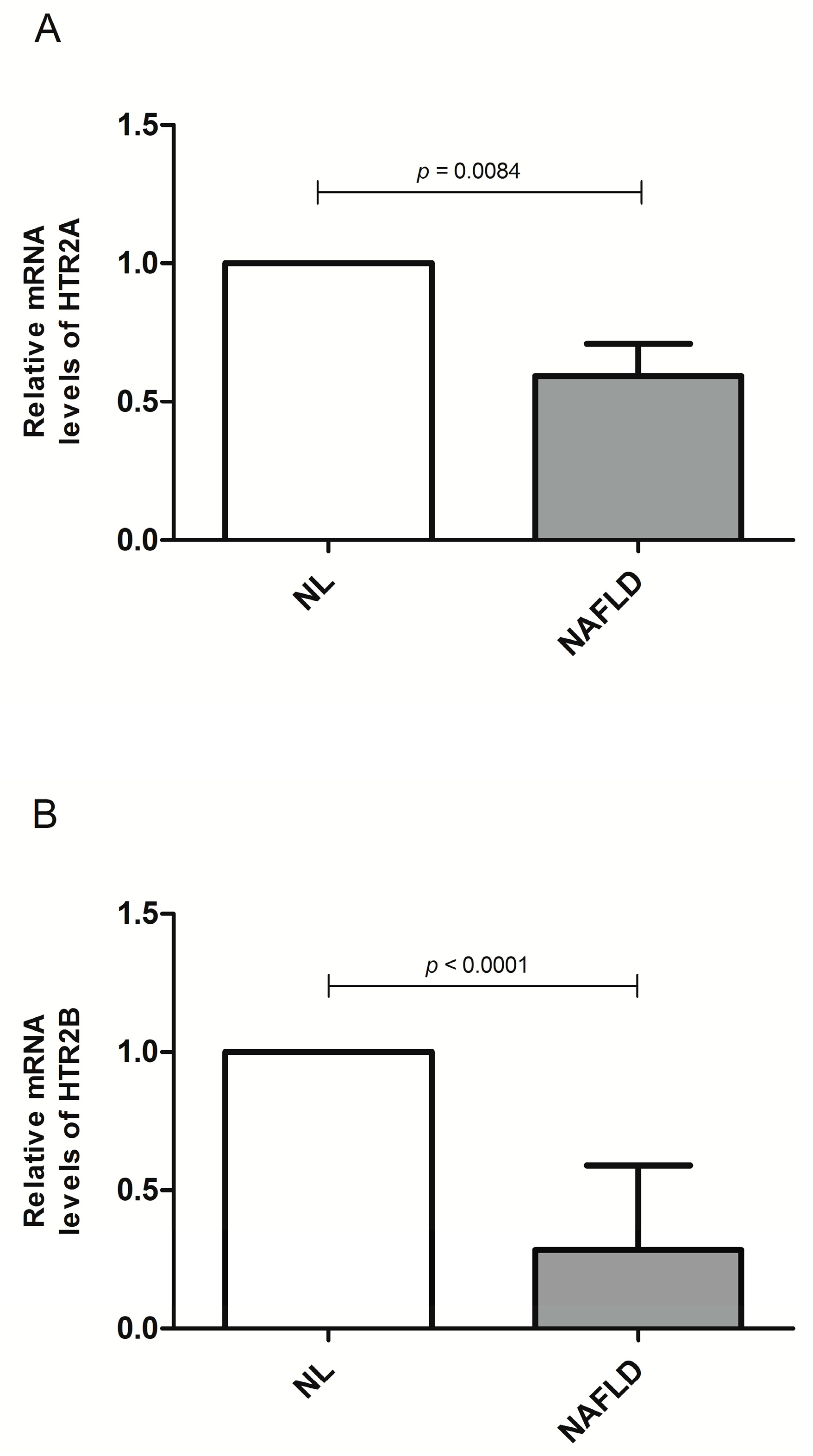
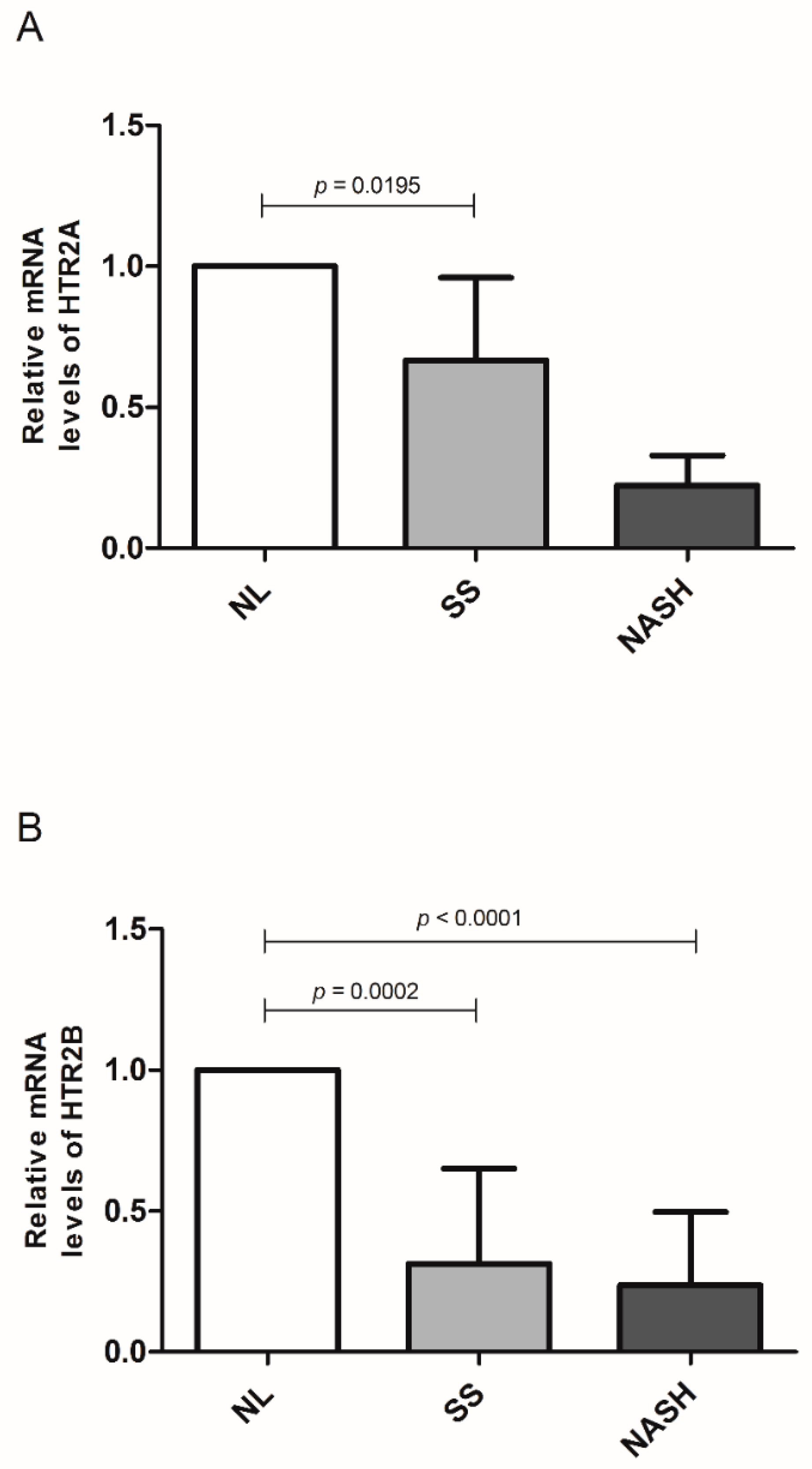
3.4. Evaluation of HTR2A, HTR2B, and HTR3 mRNA Levels in Liver and HTR3 and HTR4 in Jejunum According to Liver Histology
3.5. Correlations of Liver Expression of HTR2A, HTR2B, and HTR3 and Jejunal Expression of HTR3 and HTR4 with Serum 5-HT Levels and Metabolic Syndrome Presence
3.6. Correlations of Peripheral Levels of Serotonin with Inflammatory Cytokines
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ALP | alkaline phosphatase |
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| ATP | adenosine triphosphate |
| BMI | body mass index |
| CRP | C-reactive protein |
| DBP | diastolic blood pressure |
| DPP-4 | dipeptidyl peptidase-4 |
| EC | enterochromaffin cells |
| ELISA | enzyme-linked immunosorbent assay |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| GGT | gamma-glutamyltransferase |
| GI | gastrointestinal |
| GLP-1 | glucagon-like peptide-1 |
| HbA1c | glycosylated hemoglobin |
| HCT | hematocrit |
| HDL-C | high-density lipoprotein cholesterol |
| HepG2 | human hepatocellular carcinoma |
| HFD | high-fat diet |
| HGB | hemoglobin |
| HOMA2-IR | homeostasis model assessment of insulin resistance |
| HPLC | high-performance liquid chromatography |
| HSCs | hepatic stellate cells |
| 5-HT | 5-hydroxytryptamin (serotonin) |
| HTR2A | serotonin receptor 2A |
| HTR2B | serotonin receptor 2B |
| HTR3 | serotonin receptor 3 |
| HTR4 | serotonin receptor 4 |
| HTRx | serotonin receptor subtypes |
| IBS | irritable bowel syndrome |
| IL | interleukin |
| IR | insulin resistance |
| LDL-C | low-density lipoprotein-cholesterol |
| MO | morbid obesity |
| NAFLD | non-alcoholic fatty liver disease |
| NASH | non-alcoholic steatohepatitis |
| NL | normal liver |
| NW | normal weight |
| PGC1α/PPARy | peroxisome proliferator- activated receptor gamma coactivator 1-alpha |
| PLT | platelets |
| RBC | red blood cells |
| ROS | reactive oxygen species |
| RT-qPCR | real-time quantitative polymerase chain reaction |
| SBP | systolic blood pressure |
| SERT | serotonin transporter |
| SS | simple steatosis |
| T2DM | type 2 diabetes mellitus |
| TG | triglycerides |
| TNF-α | tumor necrosis factor alpha |
| TPH1 | tryptophan hydroxylase 1 |
| VLCD | very low calorie diet |
| WBC | white blood cells |
| WC | waist circumference |
References
- Younossi, Z.; Tacke, F.; Arrese, M.; Chander Sharma, B.; Mostafa, I.; Bugianesi, E.; Wai-Sun Wong, V.; Yilmaz, Y.; George, J.; Fan, J.; et al. Global Perspectives on Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis. Hepatology 2019, 69, 2672–2682. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Park, S.K.; Oh, C.-M.; Chung, P.-W.; Ryoo, J.-H. Non-Alcoholic Fatty Liver Disease and Its Association with Depression in Korean General Population. J. Korean Med. Sci. 2019, 34, e199. [Google Scholar] [CrossRef] [PubMed]
- Lonardo, A.; Ballestri, S. Perspectives of nonalcoholic fatty liver disease research: A personal point of view. Explor. Med. 2020, 1, 85–107. [Google Scholar] [CrossRef]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, J.R.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.; Kirk, E.P.; Mittendorfer, B.; Patterson, B.W.; Klein, S. Randomized trial of exercise effect on intrahepatic triglyceride content and lipid kinetics in nonalcoholic fatty liver disease. Hepatology 2012, 55, 1738–1745. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef] [PubMed]
- Auguet, T.; Bertran, L.; Binetti, J.; Aguilar, C.; Martínez, S.; Sabench, F.; Lopez-Dupla, J.M.; Porras, J.A.; Riesco, D.; Del Castillo, D.; et al. Relationship between IL-8 Circulating Levels and TLR2 Hepatic Expression in Women with Morbid Obesity and Nonalcoholic Steatohepatitis. Int. J. Mol. Sci. 2020, 21, 4189. [Google Scholar] [CrossRef]
- Aragonès, G.; Colom-Pellicer, M.; Aguilar, C.; Guiu-Jurado, E.; Martínez, S.; Sabench, F.; Antonio Porras, J.; Riesco, D.; Del Castillo, D.; Richart, C.; et al. Circulating microbiota-derived metabolites: A “liquid biopsy? Int. J. Obes. 2020, 44, 875–885. [Google Scholar] [CrossRef]
- Yamada, S.; Guo, X.; Wang, K.-Y.; Tanimoto, A.; Sasaguri, Y. Novel function of histamine signaling via histamine receptors in cholesterol and bile acid metabolism: Histamine H2 receptor protects against nonalcoholic fatty liver disease: Roles of HRs in cholesterol/BA metabolism. Pathol. Int. 2016, 66, 376–385. [Google Scholar] [CrossRef][Green Version]
- El-Merahbi, R.; Löffler, M.; Mayer, A.; Sumara, G. The roles of peripheral serotonin in metabolic homeostasis. FEBS Lett. 2015, 589, 1728–1734. [Google Scholar] [CrossRef]
- Berger, M.; Gray, J.A.; Roth, B.L. The Expanded Biology of Serotonin. Annu. Rev. Med. 2009, 60, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.A.; Sun, E.W.; Martin, A.M.; Keating, D.J. The ever-changing roles of serotonin. Int. J. Biochem. Cell Biol. 2020, 125, 105776. [Google Scholar] [CrossRef] [PubMed]
- Gershon, M.D.; Tack, J. The Serotonin Signaling System: From Basic Understanding To Drug Development for Functional GI Disorders. Gastroenterology 2007, 132, 397–414. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.M.; Tyler, K.; MacEachern, S.J.; Balemba, O.B.; Johnson, A.C.; Brooks, E.M.; Zhao, H.; Swain, G.M.; Moses, P.L.; Galligan, J.J.; et al. Activation of Colonic Mucosal 5-HT4 Receptors Accelerates Propulsive Motility and Inhibits Visceral Hypersensitivity. Gastroenterology 2012, 142, 844–854. [Google Scholar] [CrossRef]
- Mawe, G.M.; Hoffman, J.M. Serotonin signalling in the gut—Functions, dysfunctions and therapeutic targets. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 473–486. [Google Scholar] [CrossRef]
- Baganz, N.L.; Blakely, R.D. A Dialogue between the Immune System and Brain, Spoken in the Language of Serotonin. ACS Chem. Neurosci. 2013, 4, 48–63. [Google Scholar] [CrossRef]
- Peroutka, S.J.; Howell, T.A. The molecular evolution of G protein-coupled receptors: Focus on 5-hydroxytryptamine receptors. Neuropharmacology 1994, 33, 319–324. [Google Scholar] [CrossRef]
- Reeves, D.C.; Lummis, S.C.R. The molecular basis of the structure and function of the 5-HT 3 receptor: A model ligand-gated ion channel (Review). Mol. Membr. Biol. 2002, 19, 11–26. [Google Scholar] [CrossRef]
- Amireault, P.; Sibon, D.; Côté, F. Life without Peripheral Serotonin: Insights from Tryptophan Hydroxylase 1 Knockout Mice Reveal the Existence of Paracrine/Autocrine Serotonergic Networks. ACS Chem. Neurosci. 2013, 4, 64–71. [Google Scholar] [CrossRef]
- Mercado, C.P.; Quintero, M.V.; Li, Y.; Singh, P.; Byrd, A.K.; Talabnin, K.; Ishihara, M.; Azadi, P.; Rusch, N.J.; Kuberan, B.; et al. A serotonin-induced N-glycan switch regulates platelet aggregation. Sci. Rep. 2013, 3, 2795. [Google Scholar] [CrossRef]
- Stasi, C.; Bellini, M.; Bassotti, G.; Blandizzi, C.; Milani, S. Serotonin receptors and their role in the pathophysiology and therapy of irritable bowel syndrome. Tech. Coloproctol. 2014, 18, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Karsenty, G. The two faces of serotonin in bone biology. J. Cell Biol. 2010, 191, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.D.; Palanivel, R.; Mottillo, E.P.; Bujak, A.L.; Wang, H.; Ford, R.J.; Collins, A.; Blümer, R.M.; Fullerton, M.D.; Yabut, J.M.; et al. Inhibiting peripheral serotonin synthesis reduces obesity and metabolic dysfunction by promoting brown adipose tissue thermogenesis. Nat. Med. 2015, 21, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.-M.; Namkung, J.; Go, Y.; Shong, K.E.; Kim, K.; Kim, H.; Park, B.-Y.; Lee, H.W.; Jeon, Y.H.; Song, J.; et al. Regulation of systemic energy homeostasis by serotonin in adipose tissues. Nat. Commun. 2015, 6, 6794. [Google Scholar] [CrossRef]
- Sumara, G.; Sumara, O.; Kim, J.K.; Karsenty, G. Gut-Derived Serotonin Is a Multifunctional Determinant to Fasting Adaptation. Cell Metab. 2012, 16, 588–600. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Kanamori, H.; Seki, E.; Hoshi, M.; Ohtaki, H.; Yasuda, Y.; Ito, H.; Suetsugu, A.; Nagaki, M.; Moriwaki, H.; et al. l-Tryptophan-mediated Enhancement of Susceptibility to Nonalcoholic Fatty Liver Disease Is Dependent on the Mammalian Target of Rapamycin. J. Biol. Chem. 2011, 286, 34800–34808. [Google Scholar] [CrossRef]
- Zhang, Y.; Deng, R.; Yang, X.; Xu, W.; Liu, Y.; Li, F.; Zhang, J.; Tang, H.; Ji, X.; Bi, Y.; et al. Glucose potentiates β-cell function by inducing Tphl expression in rat islets. FASEB J. 2017, 31, 5342–5355. [Google Scholar] [CrossRef]
- Choi, W.; Namkung, J.; Hwang, I.; Kim, H.; Lim, A.; Park, H.J.; Lee, H.W.; Han, K.-H.; Park, S.; Jeong, J.-S.; et al. Serotonin signals through a gut-liver axis to regulate hepatic steatosis. Nat. Commun. 2018, 9, 4824. [Google Scholar] [CrossRef]
- Niture, S.; Gyamfi, M.A.; Kedir, H.; Arthur, E.; Ressom, H.; Deep, G.; Kumar, D. Serotonin induced hepatic steatosis is associated with modulation of autophagy and notch signaling pathway. Cell Commun. Signal. 2018, 16, 78. [Google Scholar] [CrossRef]
- Fu, J.; Li, C.; Zhang, G.; Tong, X.; Zhang, H.; Ding, J.; Ma, Y.; Cheng, R.; Hou, S.; An, S.; et al. Crucial Roles of 5-HT and 5-HT2 Receptor in Diabetes-Related Lipid Accumulation and Pro-Inflammatory Cytokine Generation in Hepatocytes. Cell Physiol. Biochem. 2018, 48, 2409–2428. [Google Scholar] [CrossRef]
- Van lelyveld, N.; Ter Linde, J.; Schipper, M.E.; Samsom, M. Regional differences in expression of TPH-1, SERT, 5-HT3 and 5-HT4 receptors in the human stomach and duodenum. Neurogastroenterol. Motil. 2007, 19, 342–348. [Google Scholar] [CrossRef]
- Aikiyo, S.; Kishi, K.; Kaji, N.; Mikawa, S.; Kondo, M.; Shimada, S.; Hori, M. Contribution of Serotonin 3A Receptor to Motor Function and Its Expression in the Gastrointestinal Tract. Digestion 2020, 2020, 1–11. [Google Scholar] [CrossRef]
- Hodge, S.; Bunting, B.P.; Carr, E.; Strain, J.J.; Stewart-Knox, B.J. Obesity, Whole Blood Serotonin and Sex Differences in Healthy Volunteers. Obes. Facts 2012, 5, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Young, R.L.; Lumsden, A.L.; Martin, A.M.; Schober, G.; Pezos, N.; Thazhath, S.S.; Isaacs, N.J.; Cvijanovic, N.; Sun, E.W.L.; Wu, T.; et al. Augmented capacity for peripheral serotonin release in human obesity. Int. J. Obes. 2018, 42, 1880–1889. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Kim, J.H.; Noh, S.; Hur, H.J.; Sung, M.J.; Hwang, J.-T.; Park, J.H.; Yang, H.J.; Kim, M.-S.; Kwon, D.Y.; et al. Metabolomic Analysis of Livers and Serum from High-Fat Diet Induced Obese Mice. J. Proteome Res. 2011, 10, 722–731. [Google Scholar] [CrossRef]
- Mustajoki, P.; Pekkarinen, T. Very low energy diets in the treatment of obesity. Obes. Rev. 2001, 2, 61–72. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F.; Bairey Merz, N.; Barnes, P.J.; Brinton, R.D.; Carrero, J.-J.; DeMeo, D.L.; De Vries, G.J.; Epperson, C.N.; Govindan, R.; Klein, S.L.; et al. Sex and gender: Modifiers of health, disease, and medicine. Lancet 2020, 396, 565–582. [Google Scholar] [CrossRef]
- Lonardo, A.; Suzuki, A. Sexual Dimorphism of NAFLD in Adults. Focus on Clinical Aspects and Implications for Practice and Translational Research. JCM 2020, 9, 1278. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.-C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Jamali, R.; Razavizade, M.; Arj, A.; Aarabi, M.H. Serum adipokines might predict liver histology findings in non-alcoholic fatty liver disease. WJG 2016, 22, 5096. [Google Scholar] [CrossRef]
- Ajmera, V.; Perito, E.R.; Bass, N.M.; Terrault, N.A.; Yates, K.P.; Gill, R.; Loomba, R.; Diehl, A.M.; Aouizerat, B.E.; NASH Clinical Research Network. Novel plasma biomarkers associated with liver disease severity in adults with nonalcoholic fatty liver disease. Hepatology 2017, 65, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Takaki, A.; Ikeda, F.; Yasunaka, T.; Onji, M.; Nouso, K.; Nakatsuka, A.; Wada, J.; Koike, K.; Miyahara, K.; et al. Serum-inducible protein (IP)-10 is a disease progression-related marker for non-alcoholic fatty liver disease. Hepatol. Int. 2017, 11, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Uysal, S.; Armutcu, F.; Aydogan, T.; Akin, K.; Ikizek, M.; Yigitoglu, M.R. Some inflammatory cytokine levels, iron metabolism and oxidan stress markers in subjects with nonalcoholic steatohepatitis. Clin. Biochem. 2011, 44, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Prakash, S.; Chhabra, S.; Singla, V.; Madan, K.; Gupta, S.D.; Panda, S.K.; Khanal, S.; Acharya, S.K. Association of pro-inflammatory cytokines, adipokines & oxidative. Indian, J. Med. Res. 2012, 136, 229. [Google Scholar]
- Ritze, Y.; Schollenberger, A.; Hamze Sinno, M.; Bühler, N.; Böhle, M.; Bárdos, G.; Sauer, H.; Mack, I.; Enck, P.; Zipfel, S.; et al. Gastric ghrelin, GOAT, leptin, and leptinR expression as well as peripheral serotonin are dysregulated in humans with obesity. Neurogastroenterol. Motil. 2016, 28, 806–815. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, R.L.; Senadheera, S.; Markus, I.; Liu, L.; Howitt, L.; Chen, H.; Murphy, T.V.; Sandow, S.L.; Bertrand, P.P. A Western Diet Increases Serotonin Availability in Rat Small Intestine. Endocrinology 2011, 152, 36–47. [Google Scholar] [CrossRef]
- Blum, I.; Vered, Y.; Graff, E.; Grosskopf, Y.; Don, R.; Harsat, A.; Raz, O. The influence of meal composition on plasma serotonin and norepinephrine concentrations. Metabolism 1992, 41, 137–140. [Google Scholar] [CrossRef]
- Watanabe, H.; Nakano, T.; Saito, R.; Akasaka, D.; Saito, K.; Ogasawara, H.; Minashima, T.; Miyazawa, K.; Kanaya, T.; Takakura, I.; et al. Serotonin Improves High Fat Diet Induced Obesity in Mice. PLoS ONE 2016, 11, e0147143. [Google Scholar] [CrossRef]
- Brand, T.; Anderson, G.M. The Measurement of Platelet-Poor Plasma Serotonin: A Systematic Review of Prior Reports and Recommendations for Improved Analysis. Clin. Chem. 2011, 57, 1376–1386. [Google Scholar] [CrossRef]
- Strasser, B.; Fuchs, D. Diet versus Exercise in Weight Loss and Maintenance: Focus on Tryptophan. Int. J. Tryptophan Res. 2016, 9, IJTR-S33385. [Google Scholar] [CrossRef]
- Young, R.L.; Lumsden, A.L.; Keating, D.J. Gut Serotonin Is a Regulator of Obesity and Metabolism. Gastroenterology 2015, 149, 253–255. [Google Scholar] [CrossRef]
- Martin, A.M.; Young, R.L.; Leong, L.; Rogers, G.B.; Spencer, N.J.; Jessup, C.F.; Keating, D.J. The Diverse Metabolic Roles of Peripheral Serotonin. Endocrinology 2017, 158, 1049–1063. [Google Scholar] [CrossRef] [PubMed]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 161, 264–276. [Google Scholar] [CrossRef]
- Ge, X.; Pan, J.; Liu, Y.; Wang, H.; Zhou, W.; Wang, X. Intestinal Crosstalk between Microbiota and Serotonin and its Impact on Gut Motility. Curr. Pharm. Biotechnol. 2018, 19, 190–195. [Google Scholar] [CrossRef]
- Cuevas-Sierra, A.; Ramos-Lopez, O.; Riezu-Boj, J.I.; Milagro, F.I.; Martinez, J.A. Diet, Gut Microbiota, and Obesity: Links with Host Genetics and Epigenetics and Potential Applications. Adv. Nutr. 2019, 10, S17–S30. [Google Scholar] [CrossRef] [PubMed]
- Banskota, S.; Ghia, J.-E.; Khan, W.I. Serotonin in the gut: Blessing or a curse. Biochimie 2019, 161, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Haub, S.; Ritze, Y.; Ladel, I.; Saum, K.; Hubert, A.; Spruss, A.; Trautwein, C.; Bischoff, S.C. Serotonin Receptor Type 3 Antagonists Improve Obesity-Associated Fatty Liver Disease in Mice. J. Pharmacol. Exp. Ther. 2011, 339, 790–798. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Friedman, S.L. Mechanisms of hepatic stellate cell activation. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Ruddell, R.G.; Oakley, F.; Hussain, Z.; Yeung, I.; Bryan-Lluka, L.J.; Ramm, G.A.; Mann, D.A. A Role for Serotonin (5-HT) in Hepatic Stellate Cell Function and Liver Fibrosis. Am. J. Pathol. 2006, 169, 861–876. [Google Scholar] [CrossRef]
- Kim, D.C.; Jun, D.W.; Kwon, Y.I.; Lee, K.N.; Lee, H.L.; Lee, O.Y.; Yoon, B.C.; Choi, H.S.; Kim, E.K. 5-HT 2A receptor antagonists inhibit hepatic stellate cell activation and facilitate apoptosis. Liver Int. 2013, 33, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimkhani, M.R.; Oakley, F.; Murphy, L.B.; Mann, J.; Moles, A.; Perugorria, M.J.; Ellis, E.; Lakey, A.F.; Burt, A.D.; Douglass, A.; et al. Stimulating healthy tissue regeneration by targeting the 5-HT2B receptor in chronic liver disease. Nat. Med. 2011, 17, 1668–1673. [Google Scholar] [CrossRef]
- Cataldo, L.R.; Mizgier, M.L.; Bravo Sagua, R.; Jaña, F.; Cárdenas, C.; Llanos, P.; Busso, D.; Olmos, P.; Galgani, J.E.; Santos, J.L.; et al. Prolonged Activation of the Htr2b Serotonin Receptor Impairs Glucose Stimulated Insulin Secretion and Mitochondrial Function in MIN6 Cells. PLoS ONE 2017, 12, e0170213. [Google Scholar] [CrossRef]
- Besseiche, A.; Riveline, J.-P.; Gautier, J.-F.; Bréant, B.; Blondeau, B. Metabolic roles of PGC-1α and its implications for type 2 diabetes. Diabetes Metab. 2015, 41, 347–357. [Google Scholar] [CrossRef]
- Nocito, A.; Dahm, F.; Jochum, W.; Jang, J.H.; Georgiev, P.; Bader, M.; Renner, E.L.; Clavien, P. Serotonin Mediates Oxidative Stress and Mitochondrial Toxicity in a Murine Model of Nonalcoholic Steatohepatitis. Gastroenterology 2007, 133, 608–618. [Google Scholar] [CrossRef]
- Ritze, Y.; Hengelhaupt, C.; Bárdos, G.; Ernst, B.; Thurnheer, M.; D’Haese, J.G.; Bischoff, S.C.; Schultes, B. Altered intestinal neuroendocrine gene expression in humans with obesity: Neuroendocrine Genes Are Altered in Obesity. Obesity 2015, 23, 2278–2285. [Google Scholar] [CrossRef]
- Ritze, Y.; Bárdos, G.; Hubert, A.; Böhle, M.; Bischoff, S.C. Effect of tryptophan supplementation on diet-induced non-alcoholic fatty liver disease in mice. Br. J. Nutr. 2014, 112, 1–7. [Google Scholar] [CrossRef] [PubMed]
| Variables | NW (n = 26) Mean ± SD | MO (n = 58) Mean ± SD | NL (n = 22) Mean ± SD | SS (n = 21) Mean ± SD | NASH (n = 15) Mean ± SD |
| Age (years) | 49.91 ± 9.47 | 45.08 ± 10.85 | 42.73 ± 10.01 | 45.45 ± 12.42 | 48.00 ± 9.52 |
| WC (cm2) | NA | 125.10 ± 11.71 | 124.08 ± 6.69 | 130.07 ± 12.39 | 120.42 ± 13.66 |
| Cholesterol (mg/dl) | 180.74 ± 14.27 | 170.41 ± 35.59 | 168.20 ± 36.25 | 171.56 ± 31.60 | 171.30 ± 8.62 |
| LDL–C (mg/dL) | 95.60 ± 28.18 | 136.34 ± 74.74 | 106.40 ± 36.21 | 99.78 ± 25.00 | 95.88 ± 30.27 |
| ALP (U/L) | 54.78 ± 4.90 * | 67.84 ± 14.22 | 63.54 ± 11.54 § | 75.72 ± 13.76 # | 61.23 ± 12.76 |
| Med (25th–75th) | Med (25th–75th) | Med (25th–75th) | Med (25th–75th) | Med (25th–75th) | |
| Weight (kg) | 58.00 | 116.40 | 117.50 | 116.80 | 113.50 |
| (52.75–60.38) * | (108.00–130.10) | (110.25–132.00) | (110.00–130.20) | (104.00–124.00) | |
| BMI (kg/m2) | 21.63 | 43.90 | 43.30 | 44.59 | 44.95 |
| (20.07–24.04) * | (40.89–46.85) | (40.89–47.53) | (40.84–46.81) | (41.14–48.83) | |
| Glucose (mg/dL) | 80.00 | 92.00 | 85.50 | 102.00 | 99.00 |
| (73.00–85.00) * | (80.50–107.00) | (76.75–93.00) § | (83.00–153.00) | (83.00–106.00) | |
| Insulin (mUI/L) | 5.66 | 9.42 | 9.43 | 9.80 | 6.57 |
| (4.94–7.89) * | (5.54–15.80) | (4.04–17.63) | (6.94–15.75) | (5.09–17.48) | |
| HOMA2–IR | 0.72 | 1.19 | 1.23 | 1.32 | 0.86 |
| (0.62–1.01) * | (0.71–2.27) | (0.46–2.27) | (0.93–2.28) | (0.61–2.42) | |
| HbA1c (%) | 5.40 | 5.60 | 5.40 | 5.60 | 5.60 |
| (5.20–5.70) | (5.30–5.98) | (5.30–5.70) | (5.30–6.10) | (5.20–6.33) | |
| HDL–C (mg/dL) | 68.00 | 38.50 | 39.00 | 45.00 | 38.00 |
| (59.00–81.00) * | (33.00–47.00) | (32.00–46.00) | (33.75–47.50) | (33.50–48.50) | |
| TG (mg/dL) | 62.00 | 123.00 | 105.00 | 128.00 | 128.00 |
| (49.00–71.00) * | (86.00–166.00) | (86.00–141.00) | (82.25–186.25) | (83.75–187.50) | |
| AST (U/L) | 17.00 | 23.00 | 20.00 | 27.00 | 30.00 |
| (14.00–22.00) * | (17.00–37.00) | (16.00–31.50) | (20.00–43.00) | (17.00–44.00) | |
| ALT (U/L) | 16.00 | 28.50 | 21.00 | 32.50 | 33.00 |
| (12.00–20.00) * | (18.00–35.00) | (15.50–30.00) § | (24.25–35.75) | (16.00–30.50) | |
| GGT (U/L) | 13.00 | 22.00 | 21.00 | 22.00 | 26.00 |
| (11.00–17.00) * | (16.00–31.00) | (16.00–29.00) | (16.25–31.75) | (12.50–30.50) | |
| SBP (mmHg) | 120.00 | 118.00 | 122.50 | 121.00 | 115.00 |
| (109.00–122.00) | (107.00–133.00) | (99.25–132.75) | (110.00–140.00) | (101.50–125.25) | |
| DBP (mmHg) | 65.00 | 66.00 | 70.50 | 62.00 | 66.00 |
| (65.00–66.00) | (59.00–75.50) | (56.75–79.50) | (59.00–77.00) | (59.00–71.50) | |
| RBC (1012/L) | 4.22 (4.09–4.45) | 4.26 (3.94–4.45) | 4.19 (3.89–4.43) | 4.20 (3.91–4.41) | 4.24 (3.93–4.49) |
| WBC (109/L) | 6.00 (5.24–7.00) * | 7.55 (6.17–9.12) | 6.13 (5.55–9.03) | 7.57 (6.38–9.37) | 7.17 (5.90–9.07) |
| PLT (109/L) | 269.00 | 255.00 | 228.00 | 256.00 | 247.50 |
| (230.00–326.00) | (218.00–310.00) | (188.00–285.00) | (234.00–318.00) | (200.50–304.25) | |
| HGB (g/dL) | 13.00 | 12.10 | 12.10 | 12.05 | 11.95 |
| (12.00–13.40) * | (11.00–13.00) | (11.90–12.60) | (11.30–12.77) | (10.72–13.17) | |
| HCT (%) | 37.80 | 36.00 | 35.60 | 35.90 | 35.85 |
| (36.00–39.00) * | (32.90–38.50) | (34.20–38.00) | (33.42–37.92) | (32.42–38.90) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binetti, J.; Bertran, L.; Riesco, D.; Aguilar, C.; Martínez, S.; Sabench, F.; Porras, J.A.; Camaron, J.; Castillo, D.D.; Richart, C.; et al. Deregulated Serotonin Pathway in Women with Morbid Obesity and NAFLD. Life 2020, 10, 245. https://doi.org/10.3390/life10100245
Binetti J, Bertran L, Riesco D, Aguilar C, Martínez S, Sabench F, Porras JA, Camaron J, Castillo DD, Richart C, et al. Deregulated Serotonin Pathway in Women with Morbid Obesity and NAFLD. Life. 2020; 10(10):245. https://doi.org/10.3390/life10100245
Chicago/Turabian StyleBinetti, Jessica, Laia Bertran, David Riesco, Carmen Aguilar, Salomé Martínez, Fàtima Sabench, Jose Antonio Porras, Javier Camaron, Daniel Del Castillo, Cristóbal Richart, and et al. 2020. "Deregulated Serotonin Pathway in Women with Morbid Obesity and NAFLD" Life 10, no. 10: 245. https://doi.org/10.3390/life10100245
APA StyleBinetti, J., Bertran, L., Riesco, D., Aguilar, C., Martínez, S., Sabench, F., Porras, J. A., Camaron, J., Castillo, D. D., Richart, C., & Auguet, T. (2020). Deregulated Serotonin Pathway in Women with Morbid Obesity and NAFLD. Life, 10(10), 245. https://doi.org/10.3390/life10100245






