Comparative Studies of Quaternary Ammonium Salts on the Aggregation and Dispersion Behavior of Kaolinite and Quartz
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Sedimentation Tests
2.3. Adsorbed Amount Measurements
2.4. Zeta Potential Measurements
2.5. SEM Measurements
3. Results and Discussion
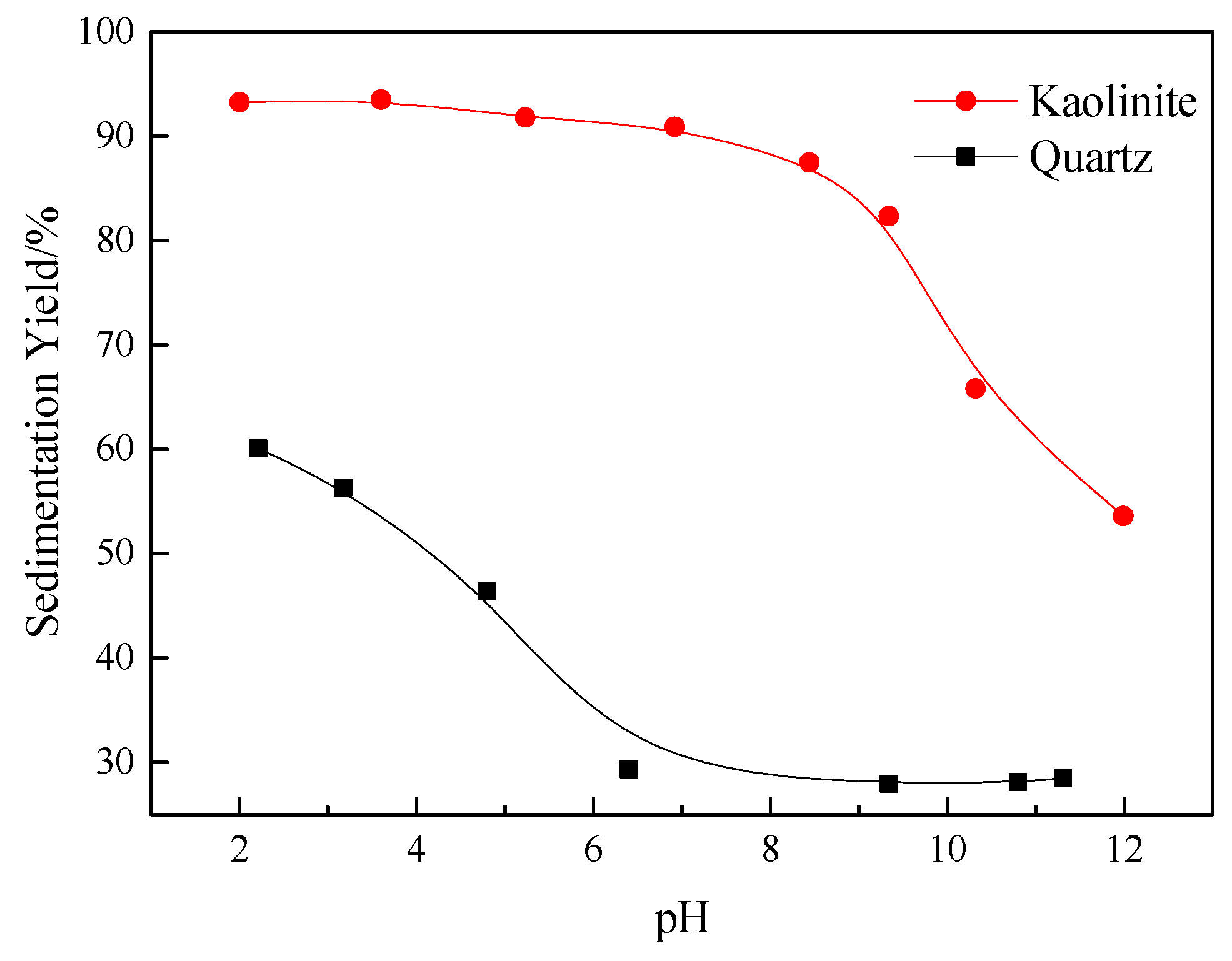
3.1. The Effect of Quaternary Ammonium Salts on Sedimentation Behavior of Kaolinite and Quartz
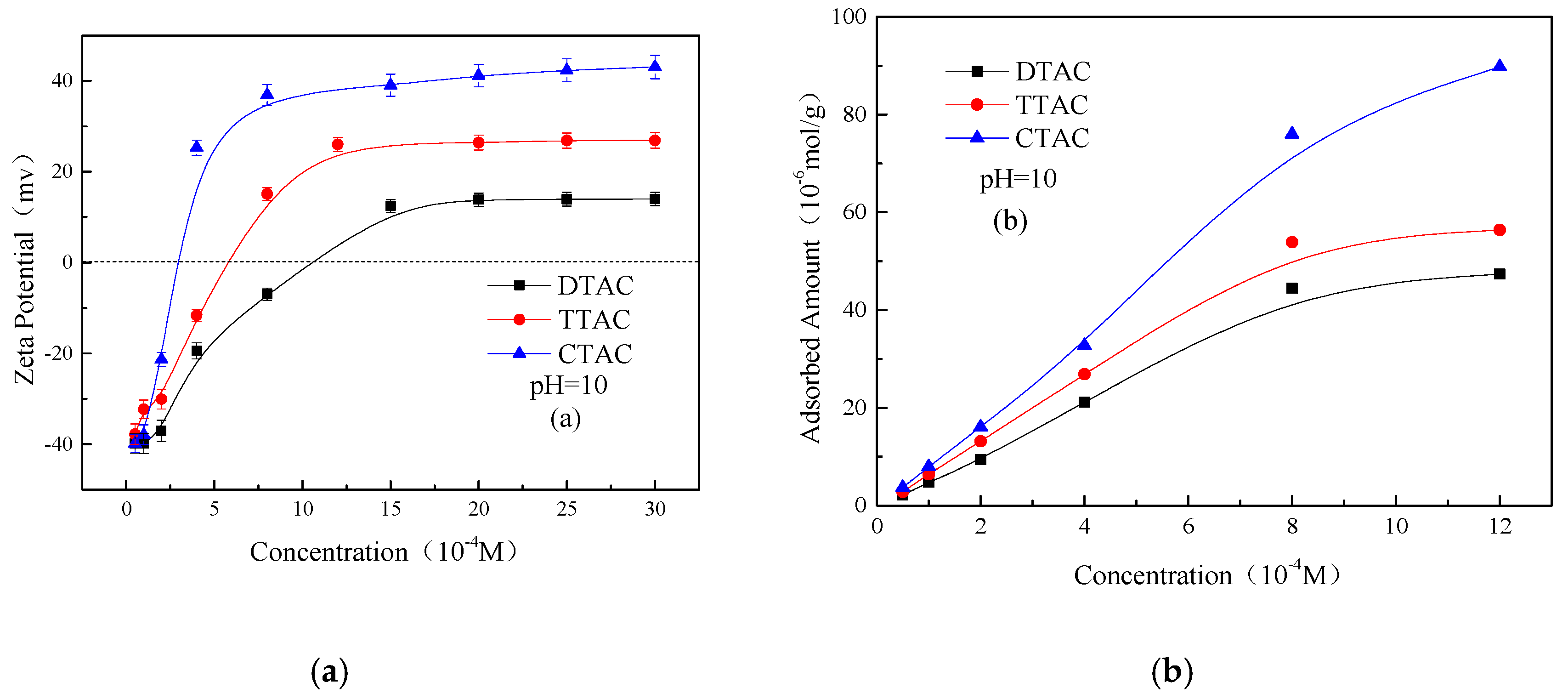
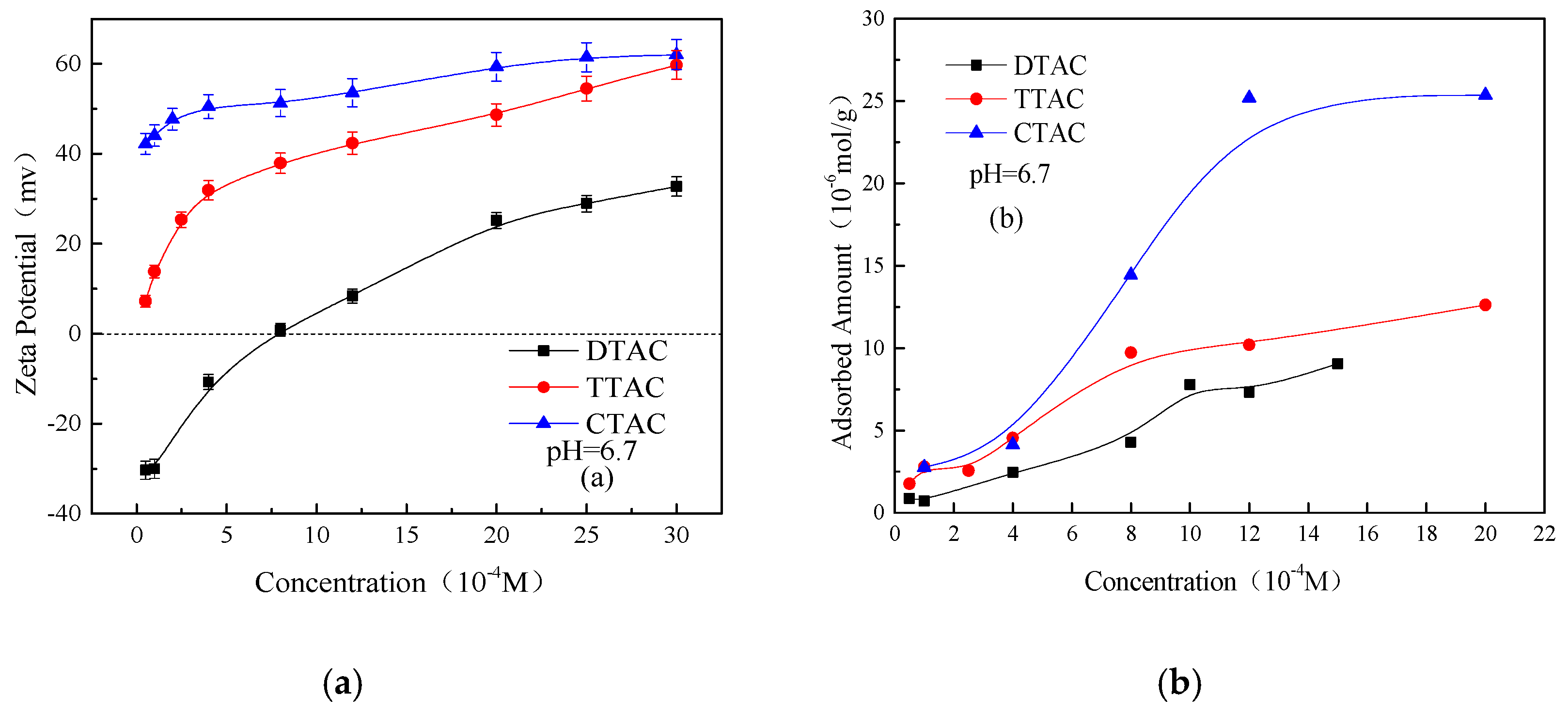
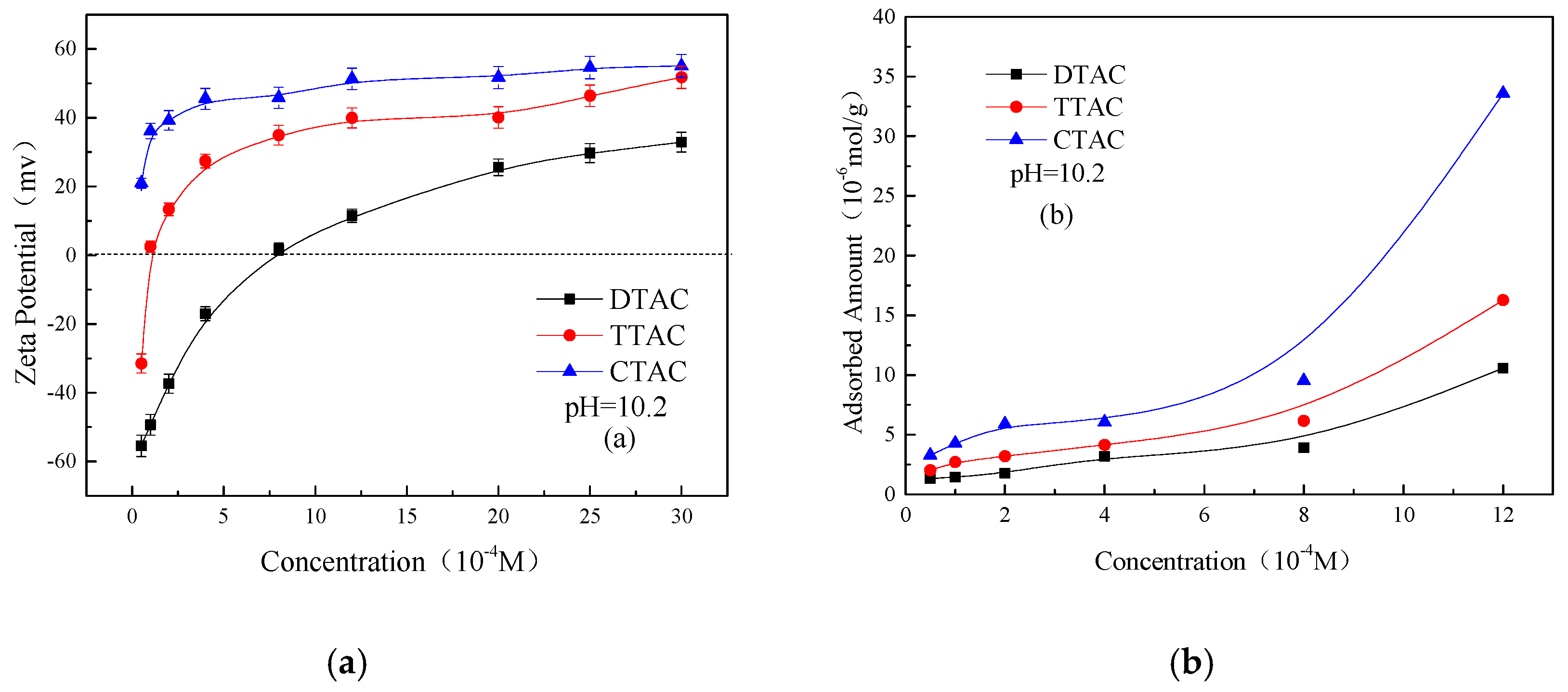
3.2. Adsorption of Quaternary Ammonium Salts on Kaolinite and Quartz Surfaces
3.3. SEM Image Analysis
4. Conclusions
- (1)
- In the absence of reagents, the kaolinite particles showed an improved aggregation behavior in neutral and acid conditions, at which the sedimentation yield of kaolinite was over 90%. Under the same experimental conditions, the quartz particles exhibited a comparatively lower aggregation behavior, and their sedimentation yield was less than 60%.
- (2)
- Except for CTAC, the additions of DTAC and TTAC increased the aggregation of the kaolinite and quartz particles in neutral and alkaline conditions. The aggregation yields of both minerals were increased significantly with the increasing the concentration of DTAC and TTAC. However, under the same conditions, the increasing concentration of CTAC resulted in an increase in the dispersion of kaolinite and quartz particles.
- (3)
- In neutral and alkaline conditions, increasing the concentration of the quaternary ammonium salts increased the Zeta potentials as well as the adsorbed amounts of all three salts on the two minerals. Moreover, the effect of CTAC on the adsorption amounts and Zeta potentials of two minerals was much greater than those of DTAC and TTAC. The greater affinity of CTAC towards the minerals was mainly due to its longer carbon chain compared to that of DTAC and TTAC; the longer the carbon chain, the greater the effect on the Zeta potential and adsorbed amount on the kaolinite and quartz surface.
- (4)
- The aggregation of kaolinite and quartz particles in the presence of quaternary ammonium salts mainly occurred either from basal to basal, or basal to edge planes in acidic conditions, whereas that in alkaline and neutral conditions mainly occurred between the basal planes.
Author Contributions
Funding
Conflicts of Interest
References
- Conceição, S.; Santos, N.; Velho, J.; Ferreira, J.; Santos, N.; Ferreira, J. Properties of paper coated with kaolin: The influence of the rheological modifier. Appl. Clay Sci. 2005, 30, 165–173. [Google Scholar] [CrossRef]
- Murray, H.H.; Kogel, J.E. Engineered clay products for the paper industry. Appl. Clay Sci. 2005, 29, 199–206. [Google Scholar] [CrossRef]
- Nithitanakul, M. Polymer layer silicate nanocomposites: Linear PEO and highly brancheddendrimer for organic wastewater treatment. Rev. Adv. Mater. 2003, 5, 187–192. [Google Scholar]
- Vahidhabanu, S.; Karuppasamy, D.; Adeogun, A.I.; Babu, B.R. Impregnation of zinc oxide modified clay over alginate beads: A novel material for the effective removal of congo red from wastewater. RSC Adv. 2017, 7, 5669–5678. [Google Scholar] [CrossRef]
- Gu, S.; Kang, X.; Wang, L.; Lichtfouse, E.; Wang, C. Clay mineral adsorbents for heavy metal removal from wastewater: A review. Environ. Chem. Lett. 2018, 17, 629–654. [Google Scholar] [CrossRef]
- Chang, P.-H.; Li, Z.; Jean, J.-S.; Jiang, W.-T.; Wang, C.-J.; Lin, K.-H. Adsorption of tetracycline on 2:1 layered non-swelling clay mineral illite. Appl. Clay Sci. 2012, 67–68, 158–163. [Google Scholar] [CrossRef]
- Galindo, A.L.; Viseras, C.; Cerezo, P. Compositional, technical and safety specifications of clays to be used as pharmaceutical and cosmetic products. Appl. Clay Sci. 2007, 36, 51–63. [Google Scholar]
- Götze, J. Chemistry, textures and physical properties of quartz—Geological interpretation and technical application. Miner. Mag. 2009, 73, 645–671. [Google Scholar] [CrossRef]
- Liu, P.; Niu, L.; Tao, X.; Li, X.; Zhang, Z.; Yu, L. Preparation of superhydrophobic-oleophilic quartz sand filter and its application in oil-water separation. Appl. Surf. Sci. 2018, 447, 656–663. [Google Scholar] [CrossRef]
- Oats, W.J.; Ozdemir, O.; Nguyen, A.V. Effect of mechanical and chemical clay removals by hydrocyclone and dispersants on coal flotation. Miner. Eng. 2010, 23, 413–419. [Google Scholar] [CrossRef]
- Ma, X.; Bruckard, W.; Holmes, R. Effect of collector, pH and ionic strength on the cationic flotation of kaolinite. Int. J. Miner. Process. 2009, 93, 54–58. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, X.; Xu, Z. Role of crystal structure in flotation separation of diaspore from kaolinite, pyrophyllite and illite. Miner. Eng. 2003, 16, 219–227. [Google Scholar] [CrossRef]
- Hu, Y.; Wei, S.; Hao, J.; Miller, J.; Fa, K. The anomalous behavior of kaolinite flotation with dodecyl amine collector as explained from crystal structure considerations. Int. J. Miner. Process. 2005, 76, 163–172. [Google Scholar] [CrossRef]
- Liu, C.; Hu, Y.; Feng, A.; Guo, Z.; Cao, X. The behavior of N,N-dipropyl dodecyl amine as a collector in the flotation of kaolinite and diaspore. Miner. Eng. 2011, 24, 737–740. [Google Scholar] [CrossRef]
- Zhang, N.; Nguyen, A.V.; Zhou, C. A review of the surface features and properties, surfactant adsorption and floatability of four key minerals of diasporic bauxite resources. Adv. Colloid Interface Sci. 2018, 254, 56–75. [Google Scholar] [CrossRef] [PubMed]
- Chorom, M.; Rengasamy, P. Dispersion and zeta potential of pure clays as related to net particle charge under varying pH, electrolyte concentration and cation type. Eur. J. Soil Sci. 1995, 46, 657–665. [Google Scholar] [CrossRef]
- Gupta, V.; Hampton, M.A.; Stokes, J.R.; Nguyen, A.V.; Miller, J.D. Particle interactions in kaolinite suspensions and corresponding aggregate structures. J. Colloid Interface Sci. 2011, 359, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Zbik, M. SEM evidence of structural re-arrangement from gelling to aggregation in Birdwood kaolinite. Colloids Surfaces A Physicochem. Eng. Asp. 2006, 287, 191–196. [Google Scholar] [CrossRef]
- Rao, F.; Ramirez-Acosta, F.J.; Sánchez-Leija, R.J.; Song, S.; Lopez-Valdivieso, A. Stability of kaolinite dispersions in the presence of sodium and aluminum ions. Appl. Clay Sci. 2011, 51, 38–42. [Google Scholar] [CrossRef]
- Lagaly, G.; Ziesmer, S. Colloid chemistry of clay minerals: The coagulation of montmorillonite dispersions. Adv. Colloid Interface Sci. 2003, 100–102, 105–128. [Google Scholar] [CrossRef]
- Yang, F.; Sun, W.; Hu, Y.; Long, S. Cationic flotation of scheelite from calcite using quaternary ammonium salts as collector: Adsorption behavior and mechanism. Miner. Eng. 2015, 81, 18–28. [Google Scholar] [CrossRef]
- Jiang, H.; Liu, G.; Hu, Y.; Xu, L.; Yu, Y.; Xie, Z.; Chen, H. Flotation and adsorption of quaternary ammonium salts collectors on kaolinite of different particle size. Int. J. Min. Sci. Technol. 2013, 23, 249–253. [Google Scholar] [CrossRef]
- Kewu, W. The crystal structure and surface property of kaolinite. Non Met. Mines 1992, 01, 48–53. [Google Scholar]
- Chen, Z.-Y.; Li, W.X.; Chen, X.Q.; Chen, X.Q.; Ma, J.W. Overview of the alumina silica crystal structure in bauxite. Light Met. 2008, 12, 6–9. [Google Scholar]
- Goumans, T.P.; Wander, A.; Brown, W.A.; Catlow, C.R.A. Structure and stability of the (001) alpha-quartz surface. Phys. Chem. Chem. Phys. 2007, 9, 2146–2152. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-P.; Hu, Y.H.; Huang, H.J.; Deng, M.J. Aggregation behavior of ultrafine kaolinite in water. J. China Univ. Min. Technol. 2007, 04, 514–517. [Google Scholar]
- Atkin, R.; Craig, V.; Wanless, E.; Biggs, S.; Craig, V.; Wanless, E. Mechanism of cationic surfactant adsorption at the solid–aqueous interface. Adv. Colloid Interface Sci. 2003, 103, 219–304. [Google Scholar] [CrossRef]
- Tian, J.; Xu, L.; Deng, W.; Jiang, H.; Gao, Z.; Hu, Y. Adsorption mechanism of new mixed anionic/cationic collectors in a spodumene-feldspar flotation system. Chem. Eng. Sci. 2017, 164, 99–107. [Google Scholar] [CrossRef]
- Fan, A.; Somasundaran, P.; Turro, N.J. Adsorption of Alkyltrimethylammonium Bromides on Negatively Charged Alumina. Langmuir 1997, 13, 506–510. [Google Scholar] [CrossRef]
- Zbik, M.; Horn, R.G. Hydrophobic attraction may contribute to aqueous flocculation of clays. Colloids Surfaces A Physicochem. Eng. Asp. 2003, 222, 323–328. [Google Scholar] [CrossRef]
- Zbik, M.; Smart, R. Dispersion of kaolinite and talc in aqueous solution: Nano-morphology and nano-bubble entrapment. Miner. Eng. 2002, 15, 277–286. [Google Scholar] [CrossRef]
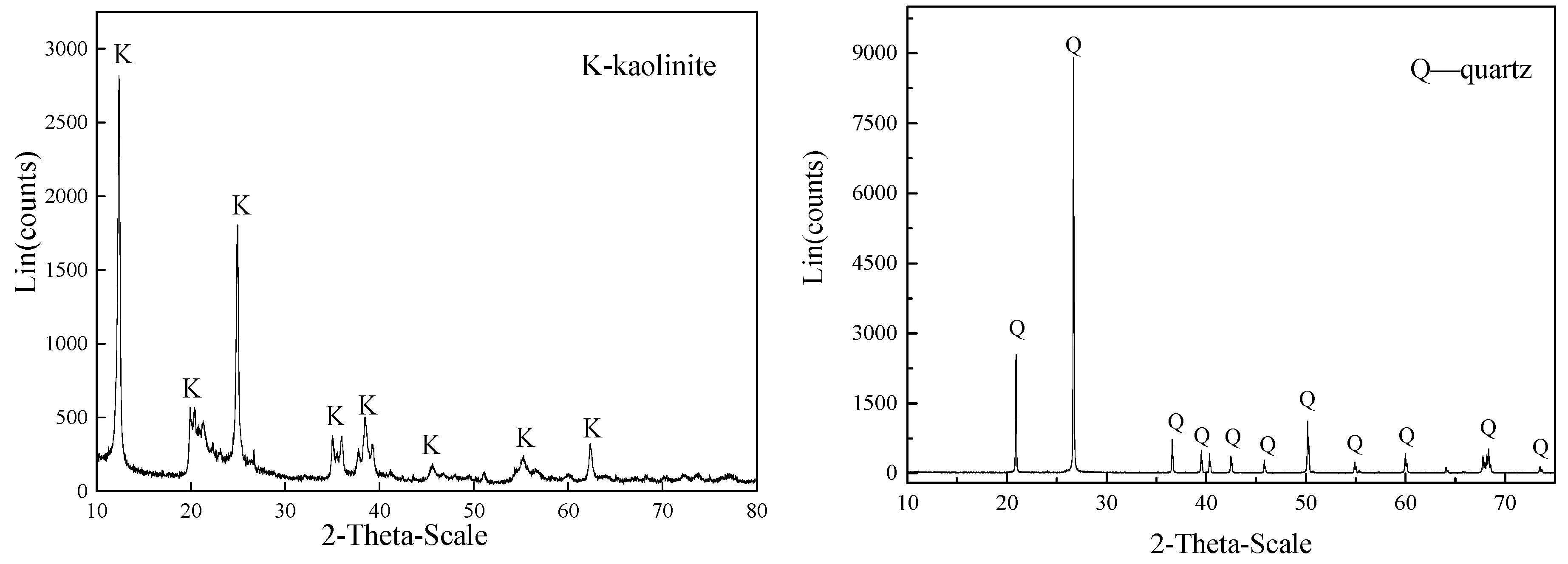








| Components | Al2O3 | SiO2 | Fe2O3 | TiO2 | CaO | MgO | K2O | Na2O | LOI a |
|---|---|---|---|---|---|---|---|---|---|
| Kaolinite | 39.2 | 43.67 | 0.32 | 1.98 | 0.01 | 0.068 | 0.094 | 0.028 | 13.98 |
| Quartz | 0.77 | 94.95 | 0.01 | 0.01 | 0.04 | 0 | 0.01 | 0 | 3.47 |
| Quaternary Ammonium Salts | Abbreviation | Molecular Formula | Weight Molecular | Carbon Chain Length |
|---|---|---|---|---|
| Dodecyltrimethylammonium chloride | DTAC | C12H25(CH3)3NCl | 263.90 | 12 |
| Tetradecyltrimethylammonium chloride | TTAC | C14H29(CH3)3NCl | 291.94 | 14 |
| Hexadecyltrimethylammonium chloride | CTAC | C16H33(CH3)3NCl | 320.00 | 16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, H.; Xiang, G.; Khoso, S.A.; Xie, J.; Huang, K.; Xu, L. Comparative Studies of Quaternary Ammonium Salts on the Aggregation and Dispersion Behavior of Kaolinite and Quartz. Minerals 2019, 9, 473. https://doi.org/10.3390/min9080473
Jiang H, Xiang G, Khoso SA, Xie J, Huang K, Xu L. Comparative Studies of Quaternary Ammonium Salts on the Aggregation and Dispersion Behavior of Kaolinite and Quartz. Minerals. 2019; 9(8):473. https://doi.org/10.3390/min9080473
Chicago/Turabian StyleJiang, Hao, Guoyuan Xiang, Sultan Ahmed Khoso, Jiahui Xie, Kai Huang, and Longhua Xu. 2019. "Comparative Studies of Quaternary Ammonium Salts on the Aggregation and Dispersion Behavior of Kaolinite and Quartz" Minerals 9, no. 8: 473. https://doi.org/10.3390/min9080473
APA StyleJiang, H., Xiang, G., Khoso, S. A., Xie, J., Huang, K., & Xu, L. (2019). Comparative Studies of Quaternary Ammonium Salts on the Aggregation and Dispersion Behavior of Kaolinite and Quartz. Minerals, 9(8), 473. https://doi.org/10.3390/min9080473






