Selective Mineralization and Recovery of Au(III) from Multi-Ionic Aqueous Systems by Bacillus licheniformis FZUL-63
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms and Growth Conditions
2.2. Metal Ion Solution and Analysis
2.3. Au(III) Uptake and Mineralization Experiments
2.4. X-ray Photoelectron Spectroscopy (XPS) Analysis
2.5. Selective Mineralization Experiments
2.6. Recovery of Au(III) Formaqua Regia-Based Metal Wastewater
3. Results and Discussion
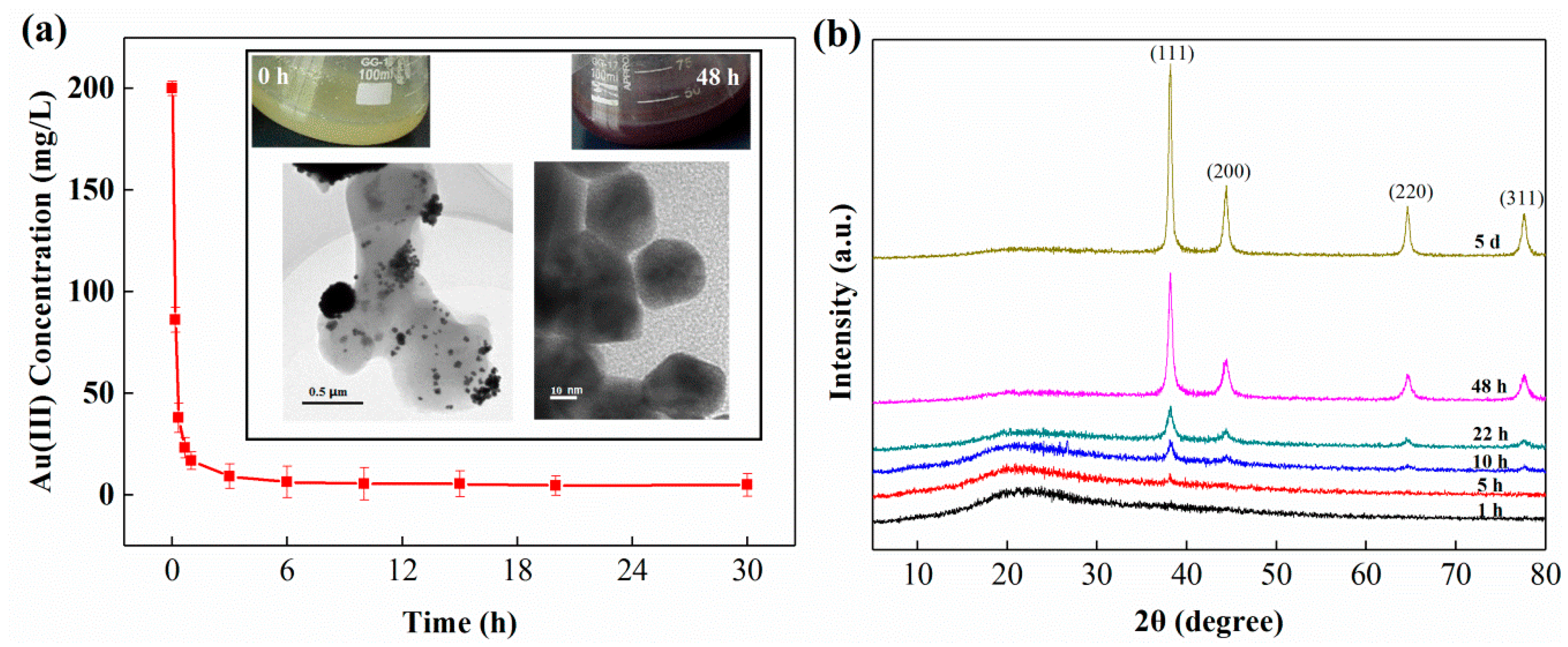
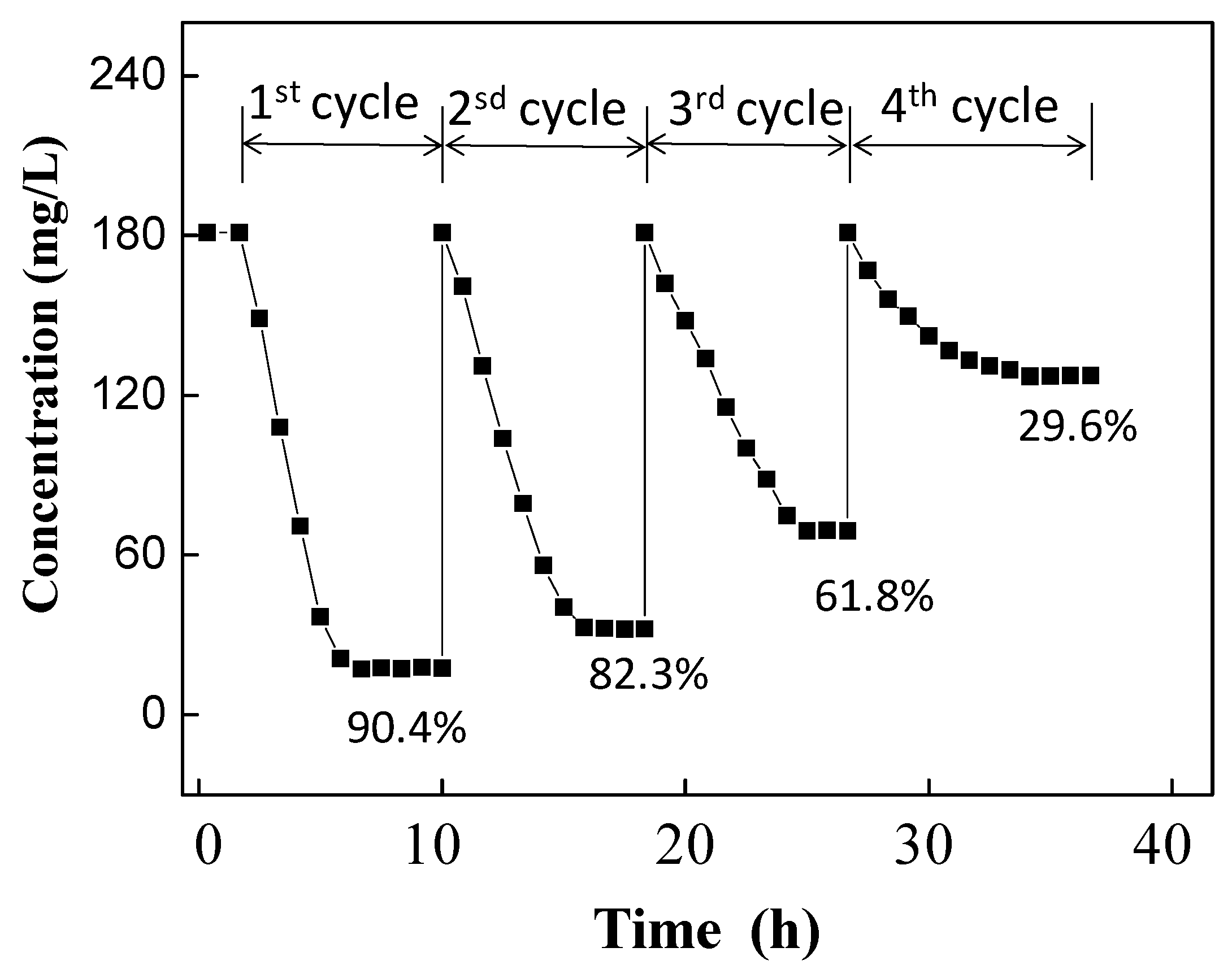
3.1. Au(III) Uptake and Mineralization
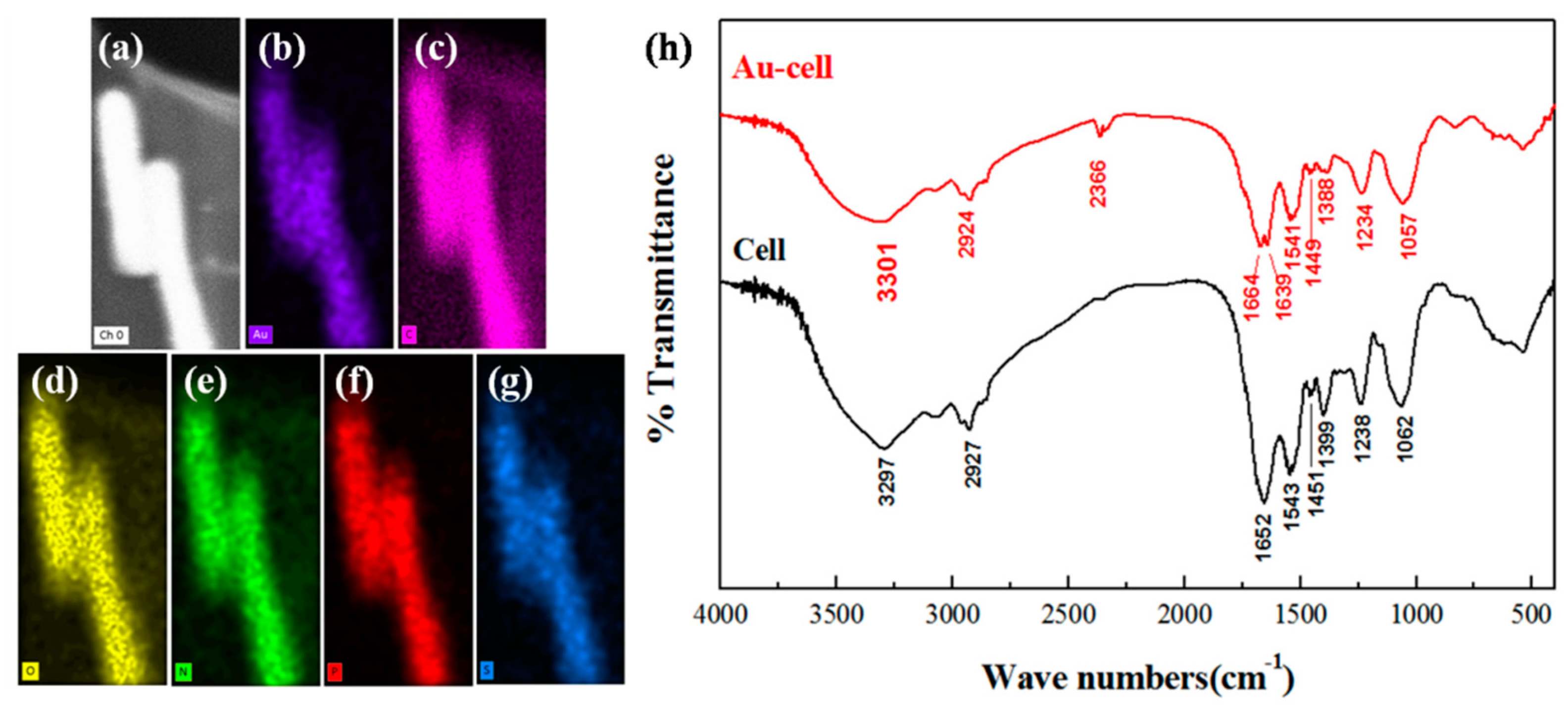
3.2. The Chemical Groups Involved in Gold Binding
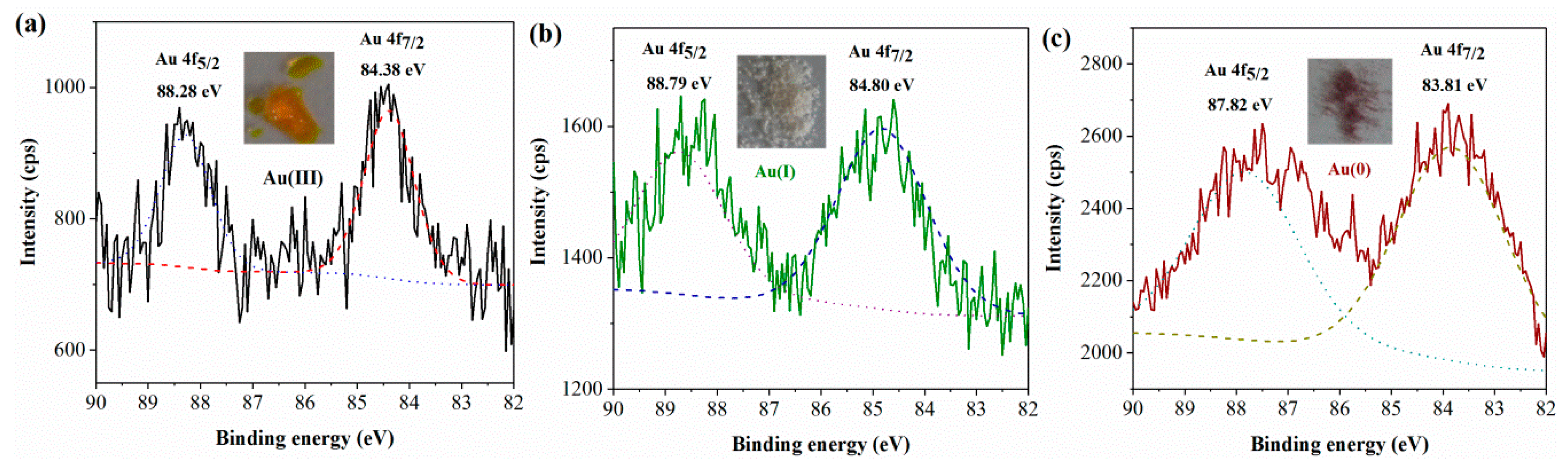
3.3. Changes in the Chemical Valence State of Gold
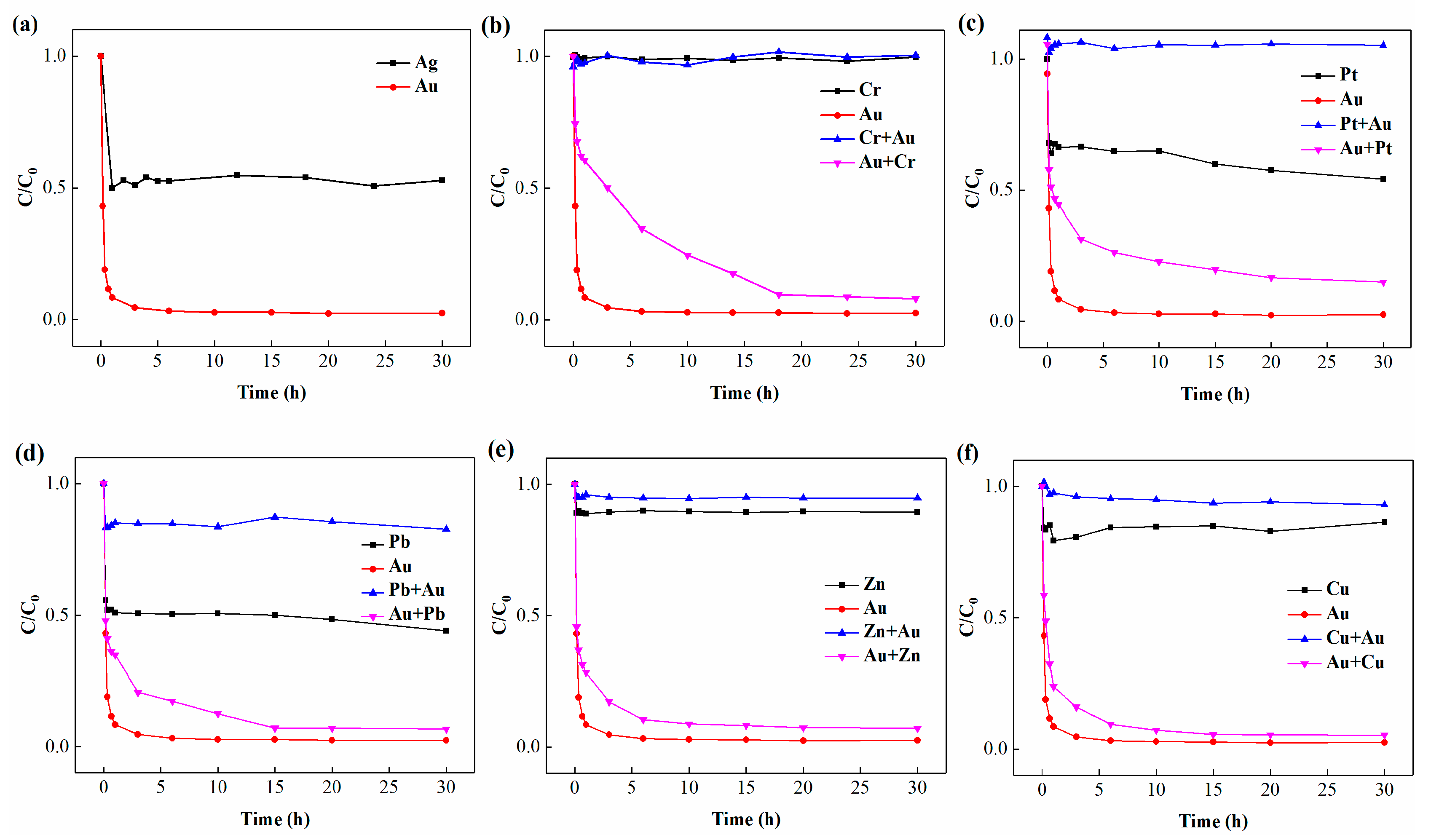
3.4. Coexisting Ions Affect the Mineralization and Recovery of Gold
3.5. Recovery of Au(III) Form Metal Wastewater
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yin, P.; Xu, M.Y.; Qu, R.J.; Chen, H.; Liu, X.G.; Zhang, J.; Xu, Q. Uptake of gold (III) from waste gold solution onto biomass-based adsorbents organophosphonic acid functionalized spent buckwheat hulls. Bioresour. Technol. 2013, 128, 36–43. [Google Scholar] [CrossRef]
- Fan, R.Y.; Xie, F.; Guan, X.L.; Zhang, Q.L.; Luo, Z.R. Selective adsorption and recovery of Au(III) from three kinds of acidic systems by persimmon residual based bio-sorbent: A method for gold recycling from e-wastes. Bioresour. Technol. 2014, 163, 167–171. [Google Scholar] [CrossRef]
- Kwak, I.S.; Yun, Y.S. Recovery of zero-valent gold from cyanide solution by a combined method of biosorption and incineration. Bioresour. Technol. 2010, 101, 8587–8592. [Google Scholar] [CrossRef]
- Hyder, M.; Ochiai, B. Selective recovery of Au(III), Pd(II), and Ag(I) from printed circuit boards using cellulose filter paper grafted with polymer chains bearing thiocarbamate moieties. Microsyst. Technol. 2018, 24, 683–690. [Google Scholar] [CrossRef]
- Choudhary, B.C.; Paul, D.; Borse, A.U.; Garole, D.J. Surface functionalized biomass for adsorption and recovery of gold from electronic scrap and refinery wastewater. Sep. Purif. Technol. 2018, 195, 260–270. [Google Scholar] [CrossRef]
- Van Eygen, E.; De Meester, S.; Tran, H.P.; Dewulf, J. Resource savings by urban mining: The case of desktop and laptop computers in Belgium. Resour. Conserv. Recy. 2016, 107, 53–64. [Google Scholar] [CrossRef]
- Syed, S. Recovery of gold from secondary sources—A review. Hydrometallurgy 2012, 115, 30–51. [Google Scholar] [CrossRef]
- Sheel, A.; Pant, D. Recovery of gold from electronic waste using chemical assisted microbial biosorption (hybrid) technique. Bioresour. Technol. 2018, 247, 1189–1192. [Google Scholar] [CrossRef]
- Alzate, A.; Lopez, M.E.; Serna, C. Recovery of gold from waste electrical and electronic equipment (WEEE) using ammonium persulfate. Waste Manag. 2016, 57, 113–120. [Google Scholar] [CrossRef]
- Natarajan, G.; Ting, Y.P. Gold biorecovery from e-waste: An improved strategy through spent medium leaching with pH modification. Chemosphere 2015, 136, 232–238. [Google Scholar] [CrossRef]
- Yap, C.Y.; Mohamed, N. An electrogenerative process for the recovery of gold from cyanide solutions. Chemosphere 2007, 67, 1502–1510. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, Y.; Helleur, R. Selective adsorption of Ag+ by ion-imprinted O-carboxymethyl chitosan beads grafted with thiourea-glutaraldehyde. Chem. Eng. J. 2015, 264, 56–65. [Google Scholar] [CrossRef]
- Maruyama, T.; Matsushita, H.; Shimada, Y.; Kamata, I.; Hanaki, M.; Sonokawa, S.; Kamiya, N.; Goto, M. Proteins and protein-rich biomass as environmentally friendly adsorbents selective for precious metal ions. Environ. Sci. Technol. 2007, 41, 1359–1364. [Google Scholar] [CrossRef]
- Devi, P.S.; Banerjee, S.; Chowdhury, S.R.; Kumar, G.S. Eggshell membrane: A natural biotemplate to synthesize fluorescent gold nanoparticles. Rsc. Adv. 2012, 2, 11578–11585. [Google Scholar] [CrossRef]
- Ju, X.H.; Igarashi, K.; Miyashita, S.; Mitsuhashi, H.; Inagaki, K.; Fujii, S.I.; Sawada, H.; Kuwabara, T.; Minoda, A. Effective and selective recovery of gold and palladium ions from metal wastewater using a sulfothermophilic red alga, Galdieria sulphuraria. Bioresour. Technol. 2016, 211, 759–764. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Mudunuru, B.M.; Hackl, R. The role of microorganisms in gold processing and recovery—A review. Hydrometallurgy 2014, 142, 70–83. [Google Scholar] [CrossRef]
- Das, N. Recovery of precious metals through biosorption—A review. Hydrometallurgy 2010, 103, 180–189. [Google Scholar] [CrossRef]
- Zhang, T.; Tu, Z.H.; Lu, G.N.; Duan, X.C.; Yi, X.Y.; Guo, C.L.; Dang, Z. Removal of heavy metals from acid mine drainage using chicken eggshells in column mode. J. Environ. Manag. 2017, 188, 1–8. [Google Scholar] [CrossRef]
- Malhotra, A.; Dolma, K.; Kaur, N.; Rathore, Y.S.; Ashish; Mayilraj, S.; Choudhury, A.R. Biosynthesis of gold and silver nanoparticles using a novel marine strain of Stenotrophomonas. Bioresour. Technol. 2013, 142, 727–731. [Google Scholar] [CrossRef]
- Johnston, C.W.; Wyatt, M.A.; Li, X.; Ibrahim, A.; Shuster, J.; Southam, G.; Magarvey, N.A. Gold biomineralization by a metallophore from a gold-associated microbe. Nat. Chem. Biol. 2013, 9, 241–243. [Google Scholar] [CrossRef]
- Nita, R.; Trammell, S.A.; Ellis, G.A.; Moore, M.H.; Soto, C.M.; Leary, D.H.; Fontana, J.; Talebzadeh, S.F.; Knight, D.A. Kinetic analysis of the hydrolysis of methyl parathion using citrate-stabilized 10 nm gold nanoparticles. Chemosphere 2016, 144, 1916–1919. [Google Scholar] [CrossRef]
- Uebe, R.; Schuler, D. Magnetosome biogenesis in magnetotactic bacteria. Nat. Rev. Microbiol. 2016, 14, 621–637. [Google Scholar] [CrossRef]
- Amor, M.; Busigny, V.; Louvat, P.; Gelabert, A.; Cartigny, P.; Durand-Dubief, M.; Ona-Nguema, G.; Alphandery, E.; Chebbi, I.; Guyot, F. Mass-dependent and -independent signature of Fe isotopes in magnetotactic bacteria. Science 2016, 352, 705–708. [Google Scholar] [CrossRef]
- Lu, A.H.; Li, Y.; Jin, S. Interactions between semiconducting minerals and bacteria under light. Elements 2012, 8, 125–130. [Google Scholar] [CrossRef]
- Pavlova, L.M.; Radomskaya, V.I. Biomineralization of precious metals. Biog. Abiogenic Interact. Nat. Anthropog. Syst. 2016, 15–27. [Google Scholar]
- Narayanan, K.B.; Park, H.H.; Han, S.S. Synthesis and characterization of biomatrixed-gold nanoparticles by the mushroom Flammulina velutipes and its heterogeneous catalytic potential. Chemosphere 2015, 141, 169–175. [Google Scholar] [CrossRef]
- Reith, F.; Etschmann, B.; Grosse, C.; Moors, H.; Benotmane, M.A.; Monsieurs, P.; Grass, G.; Doonan, C.; Vogt, S.; Lai, B.; et al. Mechanisms of gold biomineralization in the bacterium Cupriavidus metallidurans. Natl. Acad. Sci. USA 2009, 106, 17757–17762. [Google Scholar] [CrossRef]
- Das, S.K.; Liang, J.; Schmidt, M.; Laffir, F.; Marsili, E. Biomineralization mechanism of gold by zygomycete fungi rhizopous oryzae. Acs. Nano. 2012, 6, 6165–6173. [Google Scholar] [CrossRef]
- Lengke, M.F.; Fleet, M.E.; Southam, G. Morphology of gold nanoparticles synthesized by filamentous cyanobacteria from gold(I)-thiosulfate and gold(III)-chloride complexes. Langmuir 2006, 22, 2780–2787. [Google Scholar] [CrossRef]
- Shedbalkar, U.; Singh, R.; Wadhwani, S.; Gaidhani, S.; Chopade, B.A. Microbial synthesis of gold nanoparticles: Current status and future prospects. Adv. Colloid. Interfac. 2014, 209, 40–48. [Google Scholar] [CrossRef]
- Kalishwaralal, K.; Deepak, V.; Pandian, S.R.K.; Gurunathan, S. Biological synthesis of gold nanocubes from Bacillus licheniformis. Bioresour. Technol. 2009, 100, 5356–5358. [Google Scholar] [CrossRef]
- Lin, W.T.; Huang, Z.; Li, X.Z.; Liu, M.H.; Cheng, Y.J. Bio-remediation of acephate-Pb(II) compound contaminants by Bacillus subtilis FZUL-33. J. Environ. Sci. China 2016, 45, 94–99. [Google Scholar] [CrossRef]
- Cheng, Y.J.; Zhang, L.; Bian, X.J. Adsorption and mineralization of REE—Lanthanum onto bacterial cell surface. Environ. Sci. Pollut. Res. 2017, 25, 22334–22339. [Google Scholar] [CrossRef]
- Cheng, Y.J.; Xu, X.Y.; Yan, S.G.; Pan, X.H.; Chen, Z.; Lin, Z. Hydrothermal growth of large-size UO2 nanoparticles mediated by biomass and environmental implications. Rsc. Adv. 2014, 4, 62476–62482. [Google Scholar] [CrossRef]
- Singh, R.; Dong, H.L.; Liu, D.; Zhao, L.D.; Marts, A.R.; Farquhar, E.; Tierney, D.L.; Almquist, C.B.; Briggs, B.R. Reduction of hexavalent chromium by the thermophilic methanogen Methanothermobacter thermautotrophicus. Geochim. Cosmochim. Acta 2015, 148, 442–456. [Google Scholar] [CrossRef]
- Bao, X.Q.; Qin, Z.; Zhou, T.S.; Deng, J.J. In-situ generation of gold nanoparticles on MnO2 nanosheets for the enhanced oxidative degradation of basic dye (Methylene Blue). J. Environ. Sci.China 2018, 65, 236–245. [Google Scholar] [CrossRef]
- Miah, A.; Malakar, B.; Saikia, P. Gold over ceria-titania mixed oxides: Solar light induced catalytic activity for nitrophenol reduction. Catal. Lett. 2016, 146, 291–303. [Google Scholar] [CrossRef]
- Kumar, S.A.; Peter, Y.A.; Nadeau, J.L. Facile biosynthesis, separation and conjugation of gold nanoparticles to doxorubicin. Nanotechnology 2008, 19, 495101. [Google Scholar] [CrossRef]
- Choudhary, S.; Sar, P. Uranium biomineralization by a metal resistant Pseudomonas aeruginosa strain isolated from contaminated mine waste. J. Hazard Mater. 2011, 186, 336–343. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Papini, M.P.; Toro, L.; Trifoni, M.; Veglio, F. Biosorption of metal ions on Arthrobacter sp.: Biomass characterization and biosorption modeling. Environ. Sci. Technol. 2000, 34, 2773–2778. [Google Scholar] [CrossRef]
- Jiang, W.; Saxena, A.; Song, B.; Ward, B.B.; Beveridge, T.J.; Myneni, S.C.B. Elucidation of functional groups on gram-positive and gram-negative bacterial surfaces using infrared spectroscopy. Langmuir 2004, 20, 11433–11442. [Google Scholar] [CrossRef]
| Metal Concentration (mg/L) | Au3+ | Cu2+ | Zn2+ | Pb2+ | Pt4+ | Cr6+ | Fe3+ | Ni2+ | Ag+ |
|---|---|---|---|---|---|---|---|---|---|
| Without cell | 181.2 ± 3.2 | 348.5 ± 3.7 | 120.0 ± 2.3 | 39.2 ± 2.6 | 16.1 ± 1.1 | 27.3 ± 2.4 | 85.4 ± 3.5 | 48.7 ± 2.9 | ND |
| With cell | 17.4 ± 1.2 | 330.7 ± 2.5 | 115.2 ± 2.2 | 34.3 ± 1.7 | 15.9 ± 1.3 | 26.1 ± 1.9 | 80.6 ± 3.1 | 47.3 ± 2.7 | ND |
| Removal efficiency (%) | 90.4% | 5.1% | 4.0% | 12.5% | 1.3% | 4.4% | 5.6% | 2.9% | -- |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Y.; Ke, Z.; Bian, X.; Zhang, J.; Huang, Z.; Lv, Y.; Liu, M. Selective Mineralization and Recovery of Au(III) from Multi-Ionic Aqueous Systems by Bacillus licheniformis FZUL-63. Minerals 2019, 9, 392. https://doi.org/10.3390/min9070392
Cheng Y, Ke Z, Bian X, Zhang J, Huang Z, Lv Y, Liu M. Selective Mineralization and Recovery of Au(III) from Multi-Ionic Aqueous Systems by Bacillus licheniformis FZUL-63. Minerals. 2019; 9(7):392. https://doi.org/10.3390/min9070392
Chicago/Turabian StyleCheng, Yangjian, Zhibin Ke, Xiaojing Bian, Jianhua Zhang, Zhen Huang, Yuancai Lv, and Minghua Liu. 2019. "Selective Mineralization and Recovery of Au(III) from Multi-Ionic Aqueous Systems by Bacillus licheniformis FZUL-63" Minerals 9, no. 7: 392. https://doi.org/10.3390/min9070392
APA StyleCheng, Y., Ke, Z., Bian, X., Zhang, J., Huang, Z., Lv, Y., & Liu, M. (2019). Selective Mineralization and Recovery of Au(III) from Multi-Ionic Aqueous Systems by Bacillus licheniformis FZUL-63. Minerals, 9(7), 392. https://doi.org/10.3390/min9070392






