Iron-Rich Nanoparticles in Natural Aquatic Environments
Abstract
1. Introduction
2. Methods for Evaluating Fe Nanomineralogy
2.1. Review of Current Methods of Studying Fe Nanominerals
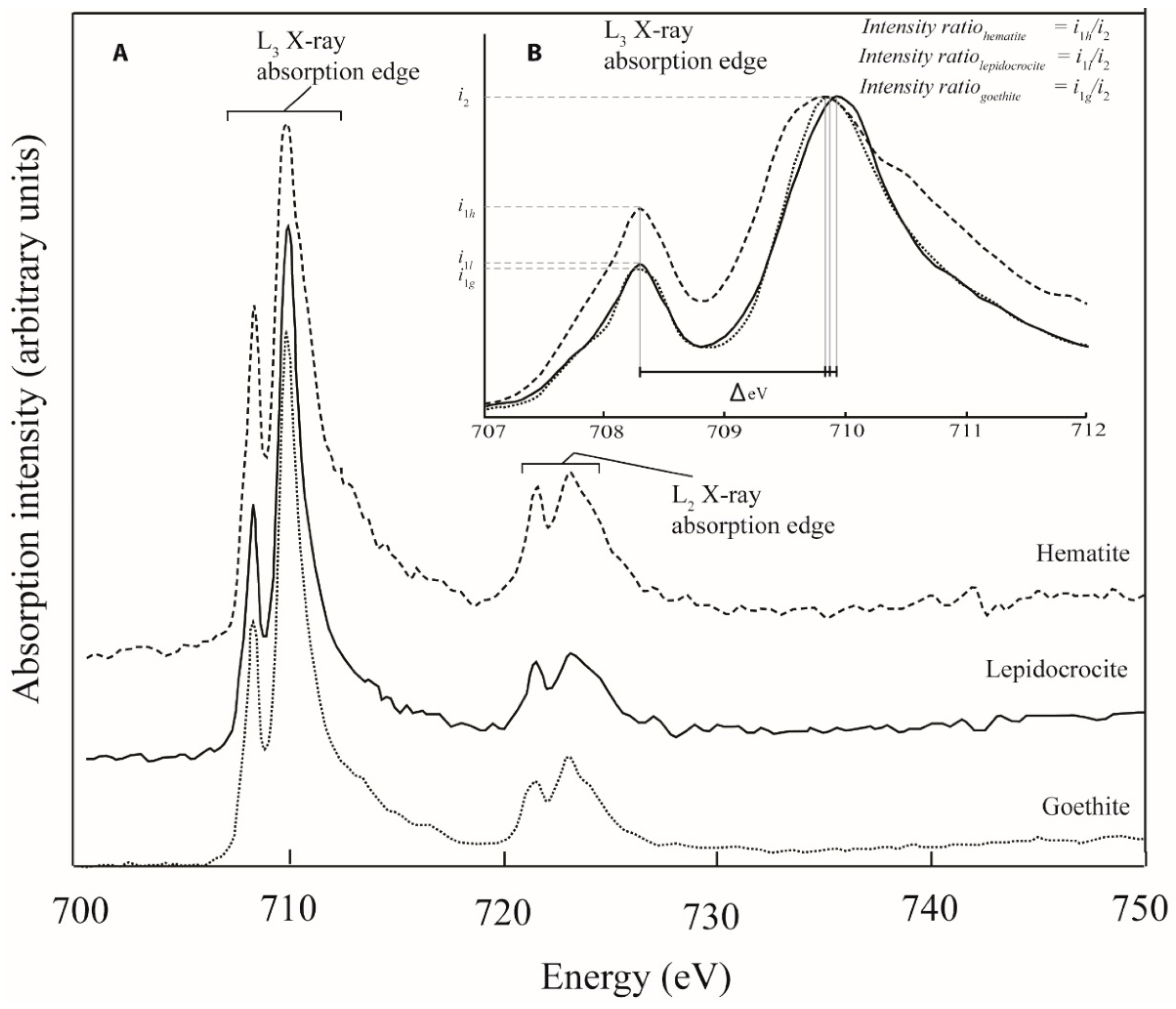
2.2. Fe L-Edge X-ray Absorption Spectroscopy as a Powerful Tool for Investigating Fe Nanomineralogy in Natural Systems
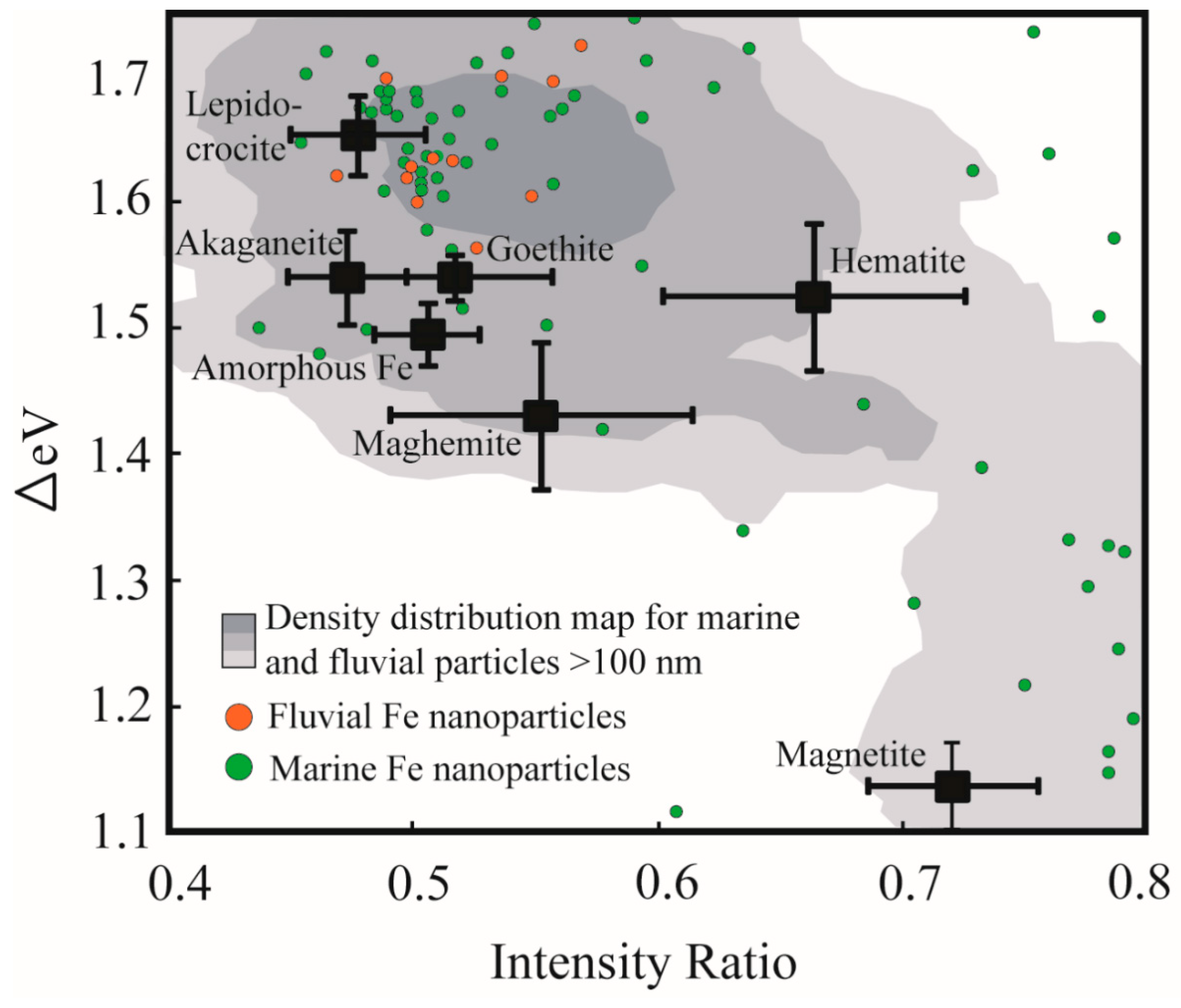
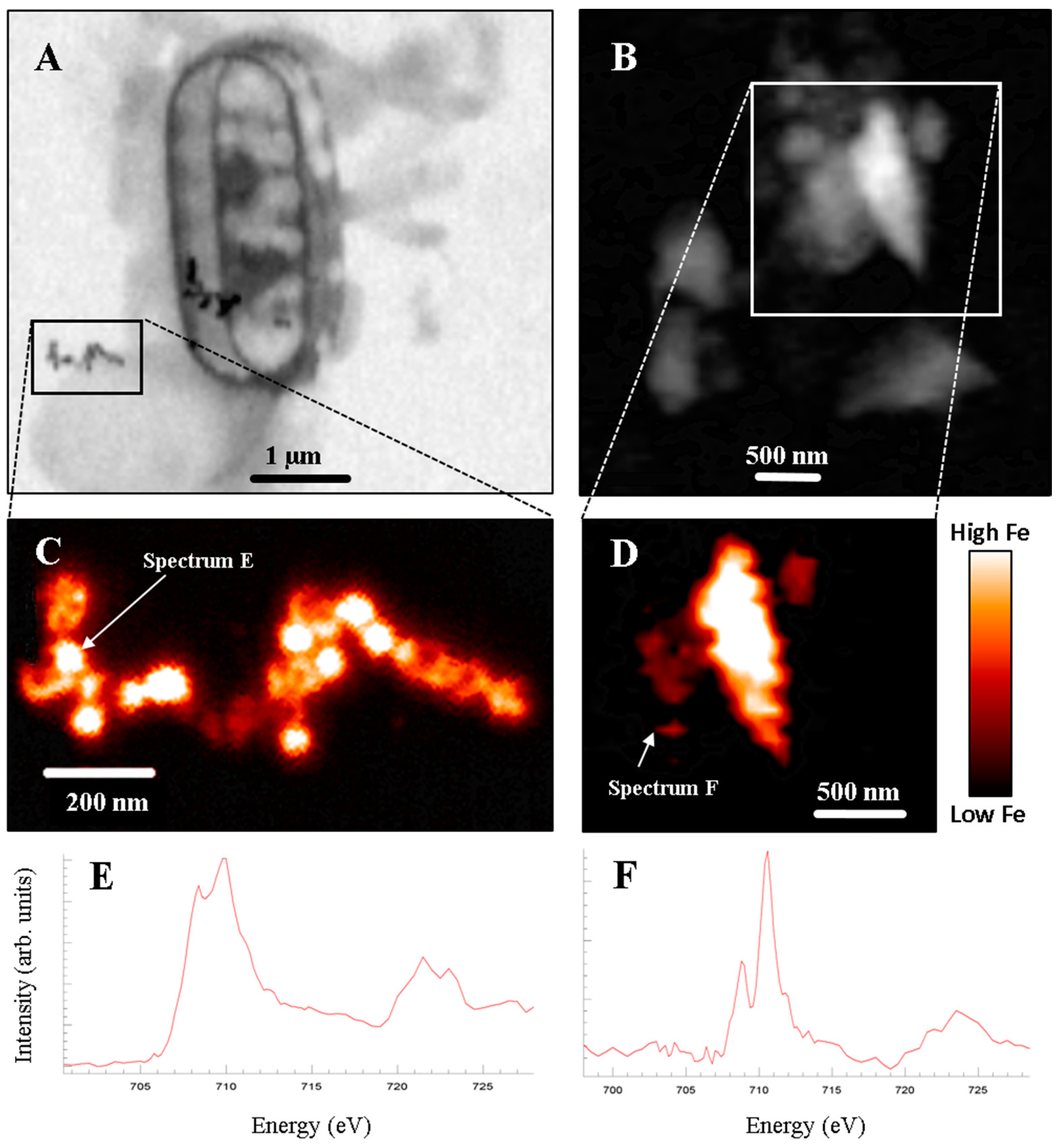
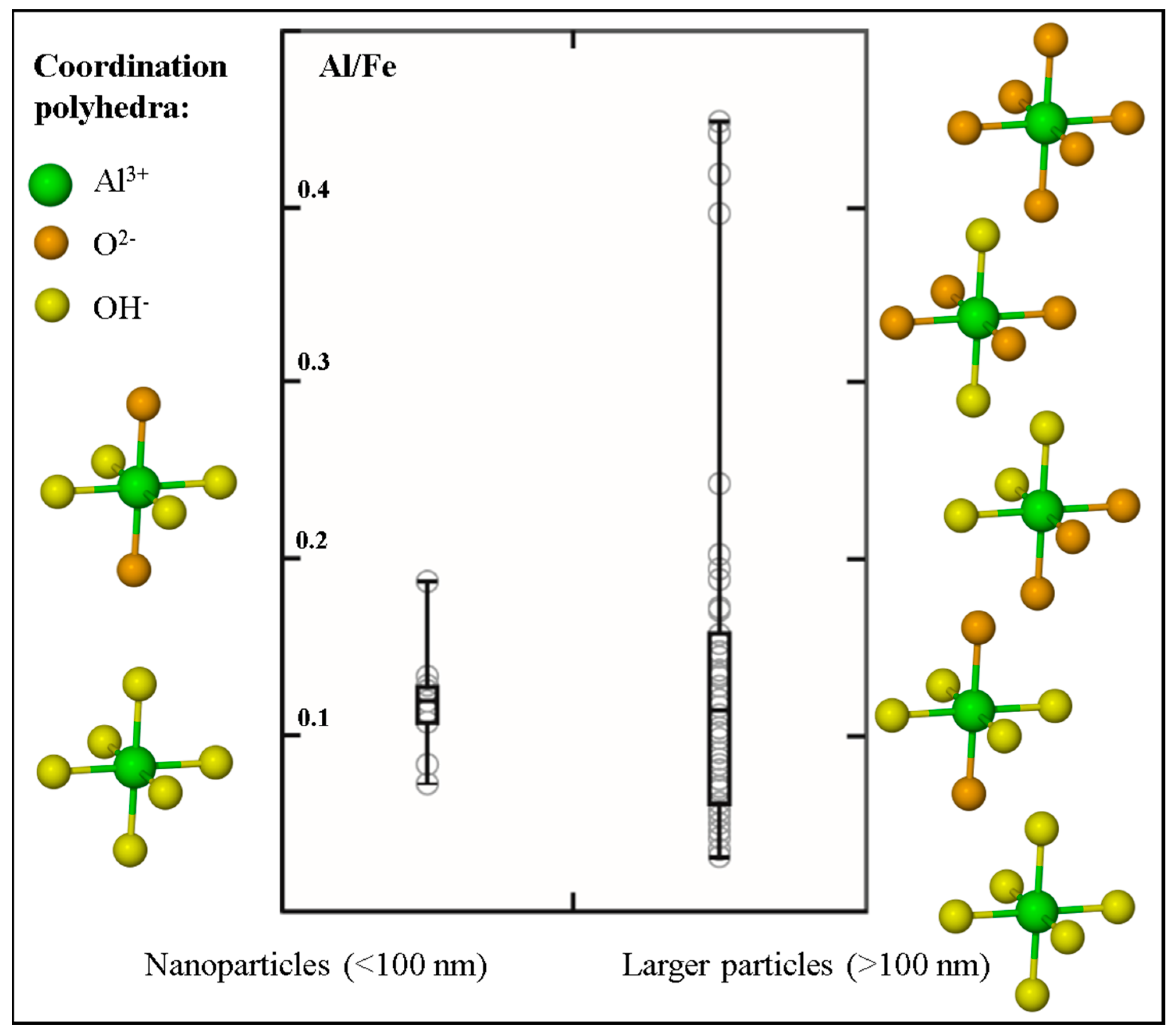
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lead, J.R.; Wilkinson, K.J. Aquatic colloids and nanoparticles: Current knowledge and future trends. Environ. Chem. 2006, 3, 159–171. [Google Scholar] [CrossRef]
- Hochella, M.F.J.; Mogk, D.W.; Ranville, J.; Allen, I.C.; Luther, G.W.; Marr, L.C.; Mcgrail, B.P.; Murayama, M.; Qafoku, N.P.; Rosso, K.M.; et al. Natural, incidental, and engineered nanomaterials and their impacts on the Earth system. Science 2019, 363, eaau8299. [Google Scholar] [CrossRef]
- Nowack, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Environ. Monit. 2011, 13, 1145–1155. [Google Scholar] [CrossRef]
- Hassellöv, M.; von der Kammer, F. Iron oxides as geochemical nanovectors for metal transport in soil-river systems. Elements 2008, 4, 401–406. [Google Scholar] [CrossRef]
- Hochella, M.F.; Moore, J.N.; Putnis, C.V.; Putnis, A.; Kasama, T.; Eberl, D.D. Direct observation of heavy metal-mineral association from the Clark Fork River Superfund Complex: Implications for metal transport and bioavailability. Geochim. Cosmochim. Acta 2005, 69, 1651–1663. [Google Scholar] [CrossRef]
- Yan, W.L.; Lien, H.-L.; Koel, B.E.; Zhang, W.-X. Iron nanoparticles for environmental clean-up: Recent developments and future outlook. Environ. Sci. Process. Impacts 2013, 15, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Tagliabue, A.; Bowie, A.R.; Boyd, P.W.; Buck, K.N.; Johnson, K.S.; Saito, M.A. The integral role of iron in ocean biogeochemistry. Nature 2017, 543, 51. [Google Scholar] [CrossRef]
- Boyd, P.W.; Ellwood, M.J. The biogeochemical cycle of iron in the ocean. Nat. Geosci. 2010, 3, 675–682. [Google Scholar] [CrossRef]
- Li, L.; Fan, M.; Brown, R.C.; Van Leeuwen, J.H.; Wang, W.; Song, Y.; Zhang, P. Synthesis, Properties, and Environmental Applications of Nanoscale Iron-Based Materials: A Review Applications of Nanoscale Iron-Based Materials: A Review. Crit. Rev. Environ. Sci. Technol. 2006, 36, 405–431. [Google Scholar] [CrossRef]
- Raychoudhury, T.; Scheytt, T. Potential of Zerovalent iron nanoparticles for remediation of environmental organic contaminants in water: A review. Water Sci. Technol. 2013, 68, 1425–1439. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Filip, J.; Zboril, R.; Varma, R.S. Natural inorganic nanoparticles—Formation, fate, and toxicity in the environment. Chem. Soc. Rev. 2015, 44, 8410–8423. [Google Scholar] [CrossRef]
- Neubauer, E.; Schenkeveld, W.D.; Plathe, K.L.; Rentenberger, C.; Von Der Kammer, F.; Kraemer, S.M.; Hofmann, T. The influence of pH on iron speciation in podzol extracts: Iron complexes with natural organic matter, and iron mineral nanoparticles. Sci. Total Environ. 2013, 461, 108–116. [Google Scholar] [CrossRef]
- Carbone, C.; Di Benedetto, F.; Marescotti, P.; Sangregorio, C. Natural Fe-oxide and -oxyhydroxide nanoparticles: An EPR and SQUID investigation. Mineral. Petrol. 2005, 85, 19–32. [Google Scholar] [CrossRef]
- Von der Heyden, B.P.; Roychoudhury, A.N.; Tyliszczak, T.; Myneni, S.C.B. Investigating nanoscale mineral compositions: Iron L3-edge spectroscopic evaluation of iron oxide and oxy-hydroxide coordination. Am. Mineral. 2017, 102, 674–685. [Google Scholar] [CrossRef]
- Tagliabue, A.; Aumont, O.; DeAth, R.; Dunne, J.P.; Dutkiewicz, S.; Galbraith, E.; Misumi, K.; Moore, J.K.; Ridgwell, A.; Sherman, E.; et al. How well do global ocean biogeochemistry models simulate dissolved iron distributions? Global Biogeochem. Cycles 2016, 32, 149–174. [Google Scholar] [CrossRef]
- Upadhyay, S.; Parekh, K.; Pandey, B. Influence of crystallite size on the magnetic properties of Fe3O4 nanoparticles. J. Alloys Compd. 2016, 678, 478–485. [Google Scholar] [CrossRef]
- Marcus, M.A.; Manceau, A.; Kersten, M. Mn, Fe, Zn and As speciation in a fast-growing ferromanganese marine nodule. Geochim. Cosmochim. Acta 2004, 68, 3125–3136. [Google Scholar] [CrossRef]
- Toner, B.M.; Santelli, C.M.; Marcus, M.A.; Wirth, R.; Chan, C.S.; McCollom, T.; Bach, W.; Edwards, K.J. Biogenic iron oxyhydroxide formation at mid-ocean ridge hydrothermal vents: Juan de Fuca Ridge. Geochim. Cosmochim. Acta 2009, 73, 388–403. [Google Scholar] [CrossRef]
- Křížek, M.; Pechoušek, J.; Tuček, J.; Šafářová, K.; Medřík, I.; Machala, L. Iron oxide nanoparticle powders with high surface area. AIP Conf. Proc. 2012, 1489, 88–94. [Google Scholar]
- Kenouche, S.; Larionova, J.; Bezzi, N.; Guari, Y.; Bertin, N.; Zanca, M.; Lartigue, L.; Cieslak, M.; Godin, C.; Morrot, G.; et al. NMR investigation of functionalized magnetic nanoparticles Fe3O4 as T1–T2 contrast agents. Powder Technol. 2014, 255, 60–65. [Google Scholar] [CrossRef]
- Sabale, S.; Jadhav, V.; Khot, V.; Zhu, X.; Xin, M.; Chen, H. Superparamagnetic MFe2O4 (M= Ni, Co, Zn, Mn) nanoparticles: Synthesis, characterization, induction heating and cell viability studies for cancer hyperthermia applications. J. Mater. Sci. Mater. Med. 2015, 26, 127. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.; Flores, D.; Ward, C.R.; Silva, L.F. Identification of nanominerals and nanoparticles in burning coal waste piles from Portugal. Sci. Total Environ. 2010, 408, 6032–6041. [Google Scholar] [CrossRef] [PubMed]
- Pyrgiotakis, G.; Blattmann, C.O.; Pratsinis, S.; Demokritou, P. Nanoparticle–nanoparticle interactions in biological media by atomic force microscopy. Langmuir 2013, 29, 11385–11395. [Google Scholar] [CrossRef] [PubMed]
- Raj, X.J.; Nishimura, T. Investigation of the Surface Potential on Iron Nanoparticles During the Corrosion by Atomic Force Microscopy (AFM) and Kelvin Probe Force Microscopy (KFM). Int. J. Electrochem. Sci. 2014, 9, 2090–2100. [Google Scholar]
- Poulton, S.W.; Raiswell, R. Chemical and physical characteristics of iron oxides in riverine and glacial meltwater sediments. Chem. Geol. 2005, 218, 203–221. [Google Scholar] [CrossRef]
- Allard, T.; Menguy, N.; Salomon, J.; Calligaro, T.; Weber, T.; Calas, G.; Benedetti, M.F. Revealing forms of iron in river-borne material from major tropical rivers of the Amazon Basin (Brazil). Geochim. Cosmochim. Acta 2004, 68, 3079–3094. [Google Scholar] [CrossRef]
- Allard, T.; Weber, T.; Bellot, C.; Damblans, C.; Bardy, M.; Bueno, G.; Nascimento, N.R.; Fritsch, E.; Benedetti, M.F. Tracing source and evolution of suspended particles in the Rio Negro Basin (Brazil) using chemical species of iron. Chem. Geol. 2011, 280, 79–88. [Google Scholar] [CrossRef]
- Liu, T.; Guo, L.; Tao, Y.; Hu, T.D.; Xie, Y.N.; Zhang, J. Bondlength alternation of nanoparticles Fe2O3 coated with organic surfactants probed by EXAFS. Nanostruct. Mater. 1999, 11, 1329–1334. [Google Scholar] [CrossRef]
- Sundman, A.; Karlsson, T.; Laudon, H.; Persson, P. XAS study of iron speciation in soils and waters from a boreal catchment. Chem. Geol. 2014, 364, 93–102. [Google Scholar] [CrossRef]
- Sun, Y.P.; Li, X.Q.; Cao, J.; Zhang, W.X.; Wang, H.P. Characterization of zero-valent iron nanoparticles. Adv. Colloid Interface Sci. 2006, 120, 47–56. [Google Scholar] [CrossRef]
- Hirst, C.; Andersson, P.S.; Shaw, S.; Burke, I.T.; Kutscher, L.; Murphy, M.J.; Maximov, T.; Pokrovsky, O.S.; Mörth, C.M.; Porcelli, D. Characterisation of Fe-bearing particles and colloids in the Lena River basin, NE Russia. Geochemica Cosmochim. Acta 2017, 213, 553–573. [Google Scholar] [CrossRef]
- von der Heyden, B.P.; Roychoudhury, A.N.; Mtshali, T.N.; Tyliszczak, T.; Myneni, S.C.B. Chemically and geographically distinct solid-phase iron pools in the Southern Ocean. Science 2012, 338, 1199–1201. [Google Scholar] [CrossRef]
- Von Der Heyden, B.P.; Frith, M.G.; Bernasek, S.; Tylizszak, T.; Roychoudhury, A.N.; Myneni, S.C.B. Geochemistry of Al and Fe in freshwater and coastal water colloids from the west coast of Southern Africa. Geochim. Cosmochim. Acta 2018, 241, 56–68. [Google Scholar] [CrossRef]
- Sheng, G.; Yang, P.; Tang, Y.; Hu, Q.; Li, H.; Ren, X.; Hu, B.; Wang, X.; Huang, Y. New insights into the primary roles of diatomite in the enhanced sequestration of UO22+ by zerovalent iron nanoparticles: An advanced approach utilizing XPS and EXAFS. Appl. Catal. B Environ. 2016, 193, 189–197. [Google Scholar] [CrossRef]
- Mourdikoudis, S.; Pallares, R.M.; Thanh, N.T. Characterization techniques for nanoparticles: Comparison and complementarity upon studying nanoparticle properties. Nanoscale 2018, 10, 12871–12934. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, M.; Srivastav, A.; Gandhi, S.; Rao, S.; Roychoudhury, A.; Kumar, A.; Singhal, R.K.; Kumar, S.; Singh, S.D. Monitoring of engineered nanoparticles in soil-plant system: A review. Environ. Nanotechnol. Monit. Manag. 2019, 11, 100218. [Google Scholar] [CrossRef]
- De Groot, F.M.F.; Glatzel, P.; Bergmann, U.; Van Aken, P.A.; Barrea, R.A.; Klemme, S.; Haevecker, M.; Knop-Gericke, A.; Heijboer, W.M.; Weckhuysen, B.M. 1s2p Resonant Inelastic X-ray Scattering of Iron Oxides. J. Phys. Chem. B 2005, 109, 20751–20762. [Google Scholar] [CrossRef] [PubMed]
- Bluhm, H.; Andersson, K.; Araki, T.; Benzerara, K.; Brown, G.E.; Dynes, J.J.; Ghosal, S.; Gilles, M.K.; Hansen, H.C.; Hemminger, J.C.; et al. Soft X-ray microscopy and spectroscopy at the molecular environmental science beamline at the Advanced Light Source. J. Electron. Spectros. Relat. Phenom. 2006, 150, 86–104. [Google Scholar] [CrossRef]
- Von Der Heyden, B.P.; Hauser, E.J.; Mishra, B.; Martinez, G.A.; Bowie, A.R.; Tyliszczak, T.; Mtshali, T.N.; Roychoudhury, A.N.; Myneni, S.C.B. Ubiquitous Presence of Fe(II) in Aquatic Colloids and Its Association with Organic Carbon. Environ. Sci. Technol. Lett. 2014, 1, 387–392. [Google Scholar] [CrossRef]
- Cressey, G.; Henderson, C.M.B.; van der Laan, G. Use of L-edge X-ray absorption spectroscopy to characterize multiple valence states of 3d transition metals; a new probe for mineralogical and geochemical research. Phys. Chem. Miner. 1993, 20, 111–119. [Google Scholar] [CrossRef]
- van Aken, P.A.; Liebscher, B. Quantification of ferrous/ferric ratios in minerals: New evaluation schemes of Fe L23 electron energy-loss near-edge spectra. Phys. Chem. Miner. 2002, 28, 188–200. [Google Scholar] [CrossRef]
- Calvert, C.C.; Brown, A.; Brydson, R. Determination of the local chemistry of iron in inorganic and organic materials. J. Electron. Spectros. Relat. Phenom. 2005, 143, 173–187. [Google Scholar] [CrossRef]
- Peak, D.; Regier, T. Direct observation of tetrahedrally coordinated Fe(III) in ferrihydrite. Environ. Sci. Technol. 2012, 46, 3163–3168. [Google Scholar] [CrossRef] [PubMed]
- Miot, J.; Benzerara, K.; Morin, G.; Kappler, A.; Bernard, S.; Obst, M.; Férard, C.; Skouri-Panet, F.; Guigner, J.M.; Posth, N.; et al. Iron biomineralization by anaerobic neutrophilic iron-oxidizing bacteria. Geochim. Cosmochim. Acta 2009, 73, 696–711. [Google Scholar] [CrossRef]
- Chan, C.S.; De Stasio, G.; Welch, S.A.; Girasole, M.; Frazer, B.H.; Nesterova, M.V.; Fakra, S.; Banfield, J.F. Microbial polysaccharides template assembly of nanocrystal fibers. Science 2004, 303, 1656–1658. [Google Scholar] [CrossRef]
- Chen, C.; Sparks, D.L. Multi-elemental scanning transmission X-ray microscopy-near edge X-ray absorption fine structure spectroscopy assessment of organo-mineral associations in soils from reduced environments. Environ. Chem. 2015, 12, 64–73. [Google Scholar] [CrossRef]
- Toner, B.M.; Fakra, S.C.; Manganini, S.J.; Santelli, C.M.; Marcus, M.A.; Moffett, J.W.; Rouxel, O.; German, C.R.; Edwards, K.J. Preservation of iron(II) by carbon-rich matrices in a hydrothermal plume. Nat. Geosci. 2009, 2, 197–201. [Google Scholar] [CrossRef]
- Todd, E.C.; Sherman, D.M.; Purton, J.A. Surface oxidation of chalcopyrite (CuFeS2) under ambient atmospheric and aqueous (pH 2-10) conditions: Cu, Fe L- and O K-edge X-ray spectroscopy. Geochim. Cosmochim. Acta 2003, 67, 2137–2146. [Google Scholar] [CrossRef]
- Hawkings, J.R.; Benning, L.G.; Raiswell, R.; Kaulich, B.; Araki, T.; Abyaneh, M.; Stockdale, A.; Koch-müller, M.; Wadham, J.L.; Tranter, M. Biolabile ferrous iron bearing nanoparticles in glacial sediments. Earth Planet. Sci. Lett. 2018, 493, 92–101. [Google Scholar] [CrossRef]
- Schwertmann, U.; Carlson, L. Aluminum Influence on Iron Oxides: XVII. Unit-Cell Parameters and Aluminum Substitution of Natural Goethites. Soil Sci. Soc. Am. J. 1994, 58, 256–261. [Google Scholar] [CrossRef]
- Myneni, S.; Hay, M.; Mishra, B. Applications of scanning transmission X-ray microscopy in studying clays and their chemical interactions. In Special Volume, Advanced Applications of Synchrotron Radiation in Clay Science; Clay Minerals Society: Chantilly, VA, USA, 2013; pp. 231–258. [Google Scholar]
- Baalousha, M.; Manciulea, A.; Cumberland, S.; Kendall, K.; Lead, J.R. Aggregation and surface properties of iron oxide nanoparticles: Influence of pH and natural organic matter. Environ. Toxicol. Chem. 2008, 27, 1875–1882. [Google Scholar] [CrossRef] [PubMed]
- Wigginton, N.S.; Haus, K.L.; Hochella, M.F. Aquatic environmental nanoparticles. J. Environ. Monit. 2007, 9, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Potter, H.A.B.; Yong, R.N. Influence of iron/aluminium ratio on the retention of lead and copper by amorphous iron–aluminium oxides. Appl. Clay Sci. 1999, 14, 1–26. [Google Scholar] [CrossRef]
- Jentzsch, T.L.; Penn, R.L. Effects of aluminum doping on the reactivity of ferrihydrite nanoparticles. J. Phys. Chem. B 2006, 110, 11746–11750. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2003; ISBN 3527302743. [Google Scholar]
- Lefevre, C.T.; Bazylinski, D.A. Ecology, diversity, and evolution of magnetotactic bacteria. Microbiol. Mol. Biol. Rev. 2013, 77, 497–526. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Gao, J.; Ai, H.; Chen, X. Applications and Potential Toxicity of Magnetic Iron Oxide Nanoparticles. Small 2013, 9, 1533–1545. [Google Scholar] [CrossRef]
- Phogat, N.; Khan, S.A.; Shankar, S.; Ansary, A.A.; Uddin, I. Fate of inorganic nanoparticles in agriculture. Adv. Mater. Lett. 2016, 7, 3–12. [Google Scholar] [CrossRef]
- Keller, A.A.; Lazareva, A. Predicted Releases of Engineered Nanomaterials: From Global to Regional to Local. Environ. Sci. Technol. Lett. 2013, 1, 65–70. [Google Scholar] [CrossRef]
- Liu, A.; Liu, J.; Han, J.; Zhang, W. Evolution of nanoscale zero-valent iron (nZVI) in water: Microscopic and spectroscopic evidence on the formation of nano- and micro-structured iron oxides. J. Hazard. Mater. 2017, 322, 129–135. [Google Scholar] [CrossRef] [PubMed]
| Technique | Acronym | Signal Analyzed | Property/Information Retrieved | Relevant References |
|---|---|---|---|---|
| Micro-focused X-ray diffraction | µXRD | Diffraction pattern of X-rays | Crystal structure, Grain size, Lattice parameters | [17,18,19] |
| Brunauer-Emmett-Teller Surface area analysis | BET | Gas sorption | Surface area, Pore analysis | [20] |
| Nuclear Magnetic Resonance Spectroscopy | NMR | Electromagnetic signal | Purity, Molecular structure | [21] |
| Fourier Transform Infrared Spectroscopy | FTIR | Absorpiton of electromagnetic radiations | Surface composition, Ligand binding | [22] |
| Scanning Electron Microscopy - Energy dispersive spectrometry | SEM-EDS | Elemental X-ray flourescence | Size, Shape, Composition | [23] |
| Atomic Force Microscopy | AFM | Force spectroscopy, imaging | Atomic structure, Crystal latice defects, Youngs modulus, Surface potential | [24,25] |
| Transmission Electron Microscopy | TEM | Electron beam flourescence | Size, Shape, Composition, Aggregation, Size distribution, Bonding environment | [26,27,28] |
| 3-D Tomography | µCT | X-ray Irradiation | Size, Shape, Aggregation, Porosity | - |
| Extended X-ray Absorption Fine Structure | EXAFS | X-ray absorption spectrum | X-ray absorption coefficent, Chemical state of species | [29,30] |
| X-ray Absorption Near Edge Structure | XANES | X-ray absorption spectrum, excitation of an inner shell electron | Vacant orbitals, Oxidation state, Chemical bonding environment | [31,32] |
| Scanning Transmission X-ray Microscopy | STXM | Elemental X-ray absroption and spectrum | Elemental concentration and speciation map, valency, coordination number | [33,34] |
| X-ray Photoelectron Spectroscopy | XPS | Kinetic energy of ejected photoelectron | Elemental concentration and speciation on particle surfaces | [31,35] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
von der Heyden, B.; Roychoudhury, A.; Myneni, S. Iron-Rich Nanoparticles in Natural Aquatic Environments. Minerals 2019, 9, 287. https://doi.org/10.3390/min9050287
von der Heyden B, Roychoudhury A, Myneni S. Iron-Rich Nanoparticles in Natural Aquatic Environments. Minerals. 2019; 9(5):287. https://doi.org/10.3390/min9050287
Chicago/Turabian Stylevon der Heyden, Bjorn, Alakendra Roychoudhury, and Satish Myneni. 2019. "Iron-Rich Nanoparticles in Natural Aquatic Environments" Minerals 9, no. 5: 287. https://doi.org/10.3390/min9050287
APA Stylevon der Heyden, B., Roychoudhury, A., & Myneni, S. (2019). Iron-Rich Nanoparticles in Natural Aquatic Environments. Minerals, 9(5), 287. https://doi.org/10.3390/min9050287






