Tsikourasite, Mo3Ni2P1+x (x < 0.25), a New Phosphide from the Chromitite of the Othrys Ophiolite, Greece
Abstract
1. Introduction
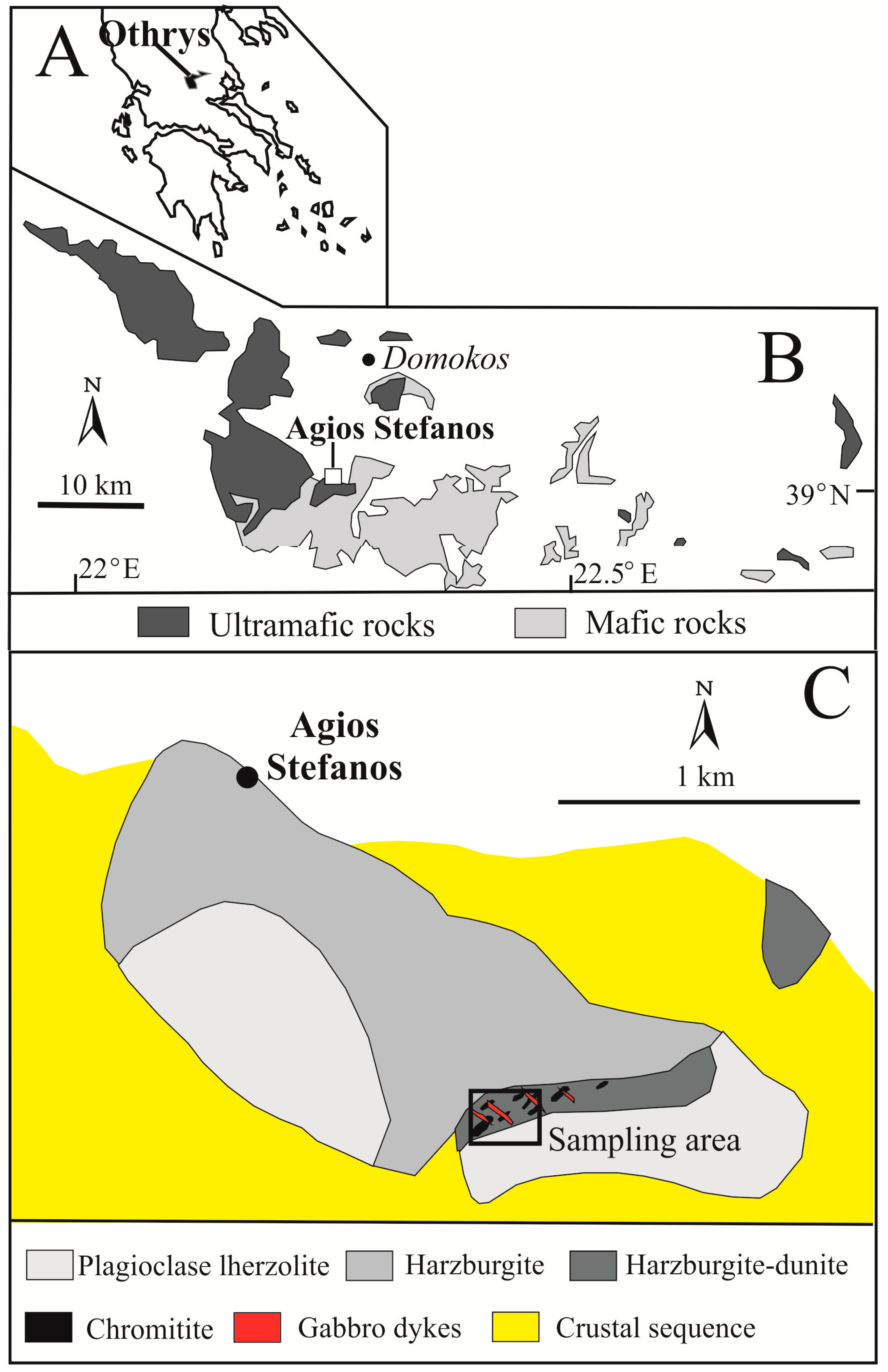
2. Geological Background and Occurrence of Tsikourasite
3. Analytical Methods
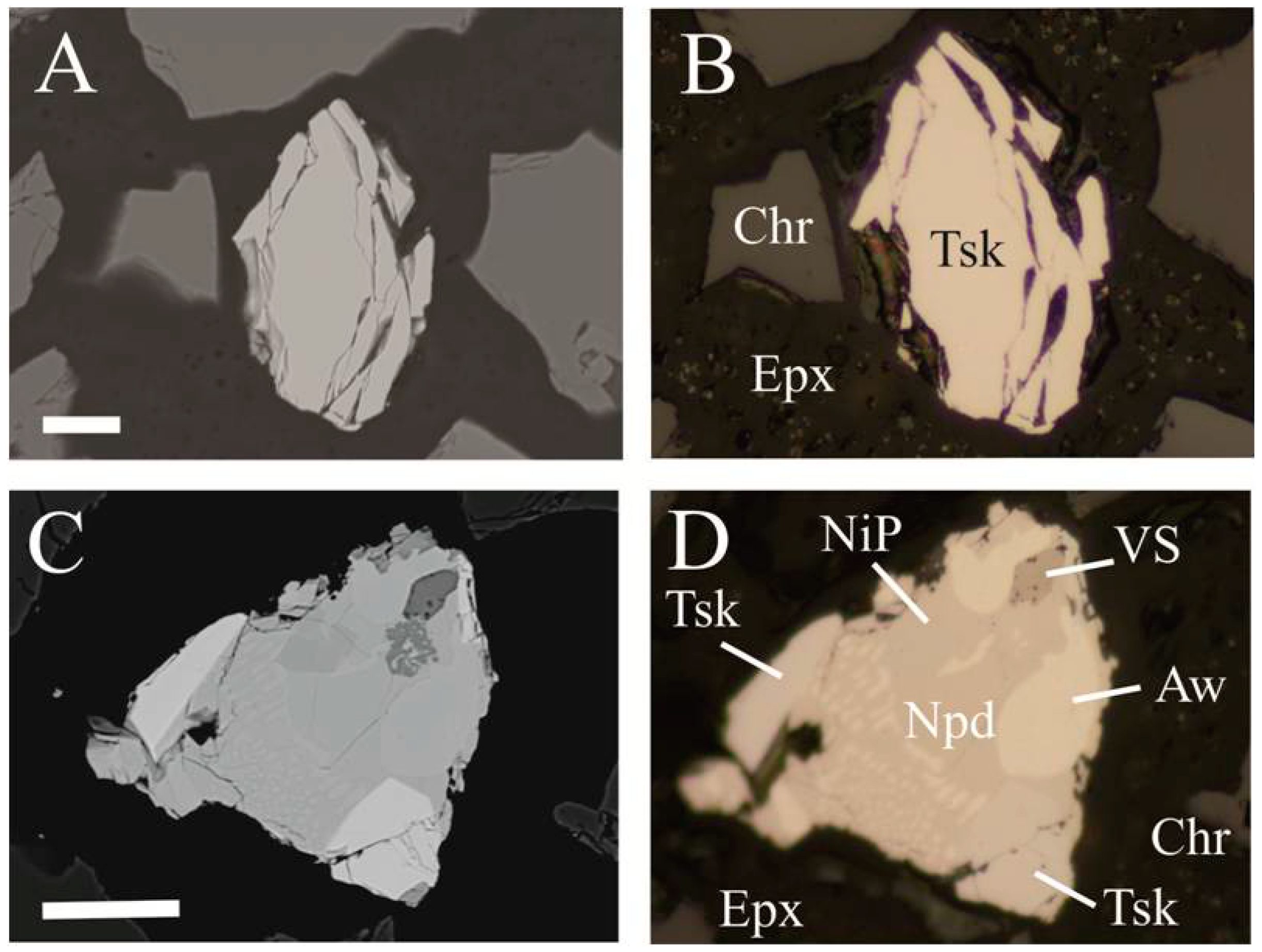
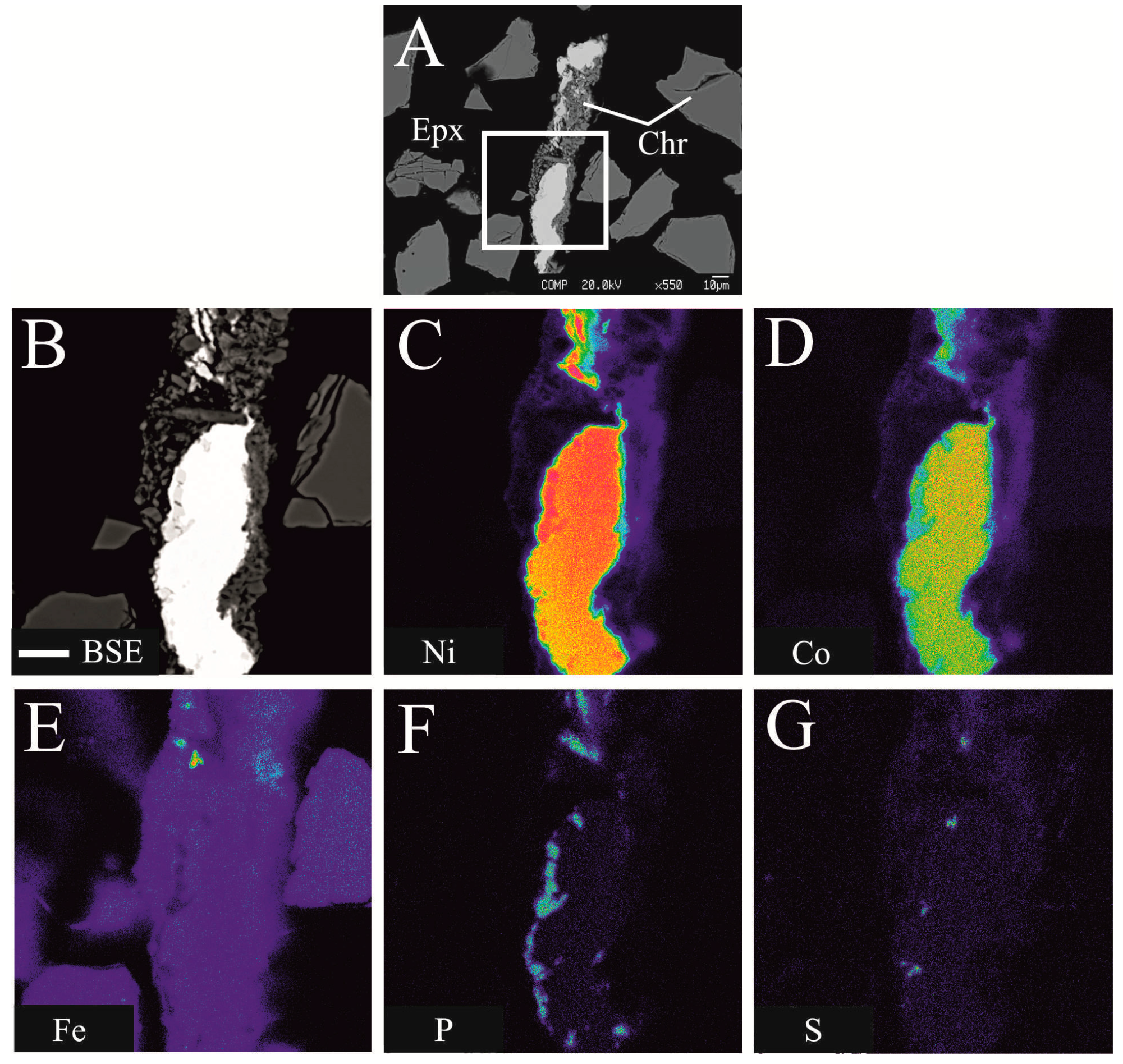
4. Physical and Optical Properties
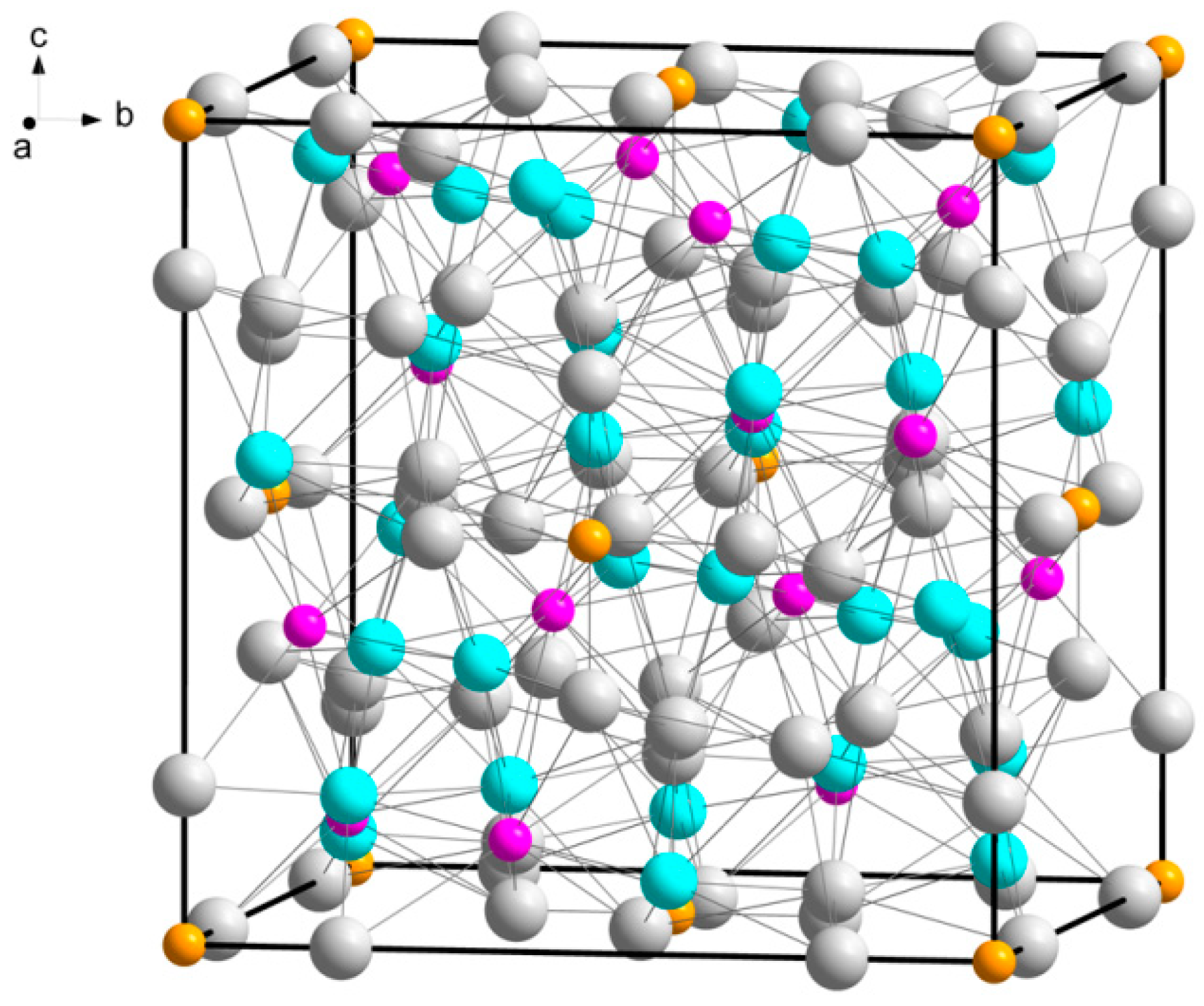
5. Chemical Composition and X-ray Crystallography
6. Relation to Other Species
7. Discussion
8. Genetic Models for the Precipitation of Tsikourasite
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Britvin, S.N.; Rudashevskii, N.S.; Krivovichev, S.V.; Burns, P.C.; Polekhovsky, Y.S. Allabogdanite, (Fe,Ni)2P, a new mineral from the Onello meteorite: The occurrence and crystal structure. Am. Mineral. 2002, 87, 1245–1249. [Google Scholar] [CrossRef]
- Zolensky, M.; Gounelle, M.; Mikouchi, T.; Ohsumi, K.; Le, L.; Hagiya, K.; Tachikawa, O. Andreyivanovite: A second new phosphide from the Kaidun meteorite. Am. Mineral. 2008, 93, 1295–1299. [Google Scholar] [CrossRef][Green Version]
- Buseck, P.R. Phosphide from meteorites: Barringerite, a new iron-nickel mineral. Science 1969, 165, 169–171. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.V.; Zolensky, M.E.; Saito, A.; Ohsumi, K.; Yang, S.V.; Kononkova, N.N.; Mikouchi, T. Florenskyite, FeTiP, a new phosphide from the Kaidun meteorite. Am. Mineral. 2000, 85, 1082–1086. [Google Scholar] [CrossRef]
- Britvin, S.N.; Murashko, M.N.; Vapnik, Y.; Polekhovsky, Y.S.; Krivovichev, S.V. Negevite, IMA 2013-104, Halamishite, IMA 2013-105, Transjordanite, IMA 2013-106, Zuktamrurite, IMA 2013-107. CNMNC Newsletter No. 19, February 2014. Mineral. Mag. 2014, 78, 165–170. [Google Scholar]
- Pratesi, G.; Bindi, L.; Moggi-Cecchi, V. Icosahedral coordination of phosphorus in the crystal structure of melliniite, a new phosphide mineral from the Northwest Africa 1054 acapulcoite. Am. Mineral. 2006, 91, 451–454. [Google Scholar] [CrossRef]
- Ma, C.; Beckett, J.R.; Rossman, G.R. Monipite, MoNiP, a new phosphide mineral in a Ca-Al-rich inclusion from the Allende meteorite. Am. Mineral. 2014, 99, 198–205. [Google Scholar] [CrossRef]
- Britvin, S.N.; Vapnik, Y.; Polekhovsky, Y.S.; Krivovichev, S.V. Murashkoite, IMA 2012-071. CNMNC Newsletter No. 15, February 2013. Mineral. Mag. 2013, 77, 1–12. [Google Scholar]
- Britvin, S.N.; Vapnik, Y.; Polekhovsky, Y.S.; Krivovichev, S.V.; Krzhizhanovskaya, M.G.; Gorelova, L.A.; Vereshchagin, O.S.; Shilovskikh, V.V.; Zaitsev, A.N. Murashkoite, FeP, a new terrestrial phosphide from pyrometamorphic rocks of the Hatrurim Formation, South Levant. Mineral. Petrol. 2019, 113, 237–248. [Google Scholar] [CrossRef]
- Britvin, S.N.; Kolomensky, V.D.; Boldyreva, M.M.; Bogdanova, A.N.; Krester, Y.L.; Boldyreva, O.N.; Rudashevsky, N.S. Nickelphosphide (Ni,Fe)3P-The nickel analogue of schreibersite. Zap. Vseross. Mineral. Obsh. 1999, 128, 64–72. [Google Scholar]
- Britvin, S.N.; Murashko, M.N.; Vereshchagin, O.S.; Vapnik, Y.; Shilovskikh, V.V.; Vlasenko, N.S. Polekhovskyite, IMA 2018-147. CNMNC Newsletter No. 48, April 2019. Mineral. Mag. 2019, 83. in press. [Google Scholar]
- Skala, R.; Cisarova, I. Crystal structure of meteoritic schreibersites: Determination of absolute structure. Phys. Chem. Miner. 2005, 31, 721–732. [Google Scholar] [CrossRef]
- Britvin, S.N.; Murashko, M.N.; Vapnik, Y.; Polekhovsky, Y.S.; Krivovichev, S.V.; Vereshchagin, O.S.; Vlasenko, N.S.; Shilovskikh, V.V.; Zaitsev, A.N. Zuktamrurite, FeP2, a new mineral, the phosphide analogue of löllingite, FeAs2. Phys. Chem. Mineral. 2019, 46, 361–369. [Google Scholar] [CrossRef]
- Zaccarini, F.; Pushkarev, E.; Garuti, G.; Kazakov, I. Platinum-Group Minerals and other accessory phases in chromite deposits of the Alapaevsk ophiolite, Central Urals, Russia. Minerals 2016, 6, 108. [Google Scholar] [CrossRef]
- Sideridis, A.; Zaccarini, F.; Grammatikopoulos, T.; Tsitsanis, P.; Tsikouras, B.; Pushkarev, E.; Garuti, G.; Hatzipanagiotou, K. First occurrences of Ni-phosphides in chromitites from the ophiolite complexes of Alapaevsk, Russia and GerakiniOrmylia, Greece. Ofioliti 2018, 43, 75–84. [Google Scholar]
- Ifandi, E.; Zaccarini, F.; Tsikouras, B.; Grammatikopoulos, T.; Garuti, G.; Karipi, S.; Hatzipanagiotou, K. First occurrences of Ni-V-Co phosphides in chromitite of Agios Stefanos mine, Othrys ophiolite, Greece. Ofioliti 2018, 43, 131–145. [Google Scholar]
- Zaccarini, F.; Ifandi, E.; Tsikouras, B.; Grammatikopoulos, T.; Garuti, G.; Mauro, D.; Bindi, L.; Stanley, C. Occurrences of of new phosphides and sulfide of Ni, Co, V, and Mo from chromitite of the Othrys ophiolite complex (Central Greece). Per. Ital. Mineral. 2019, in press. [Google Scholar]
- Oryshchyn, S.V.; Le Sénéchal, C.; Députier, S.; Bauer, J.; Guerin, R.; Akselrud, L.G. New Ternary Phases in the Mo–Ni–P System: Synthesis and Crystal Structures. J. Solid State Chem. 2001, 160, 156–166. [Google Scholar] [CrossRef]
- Rassios, A.; Smith, A.G. Constraints on the formation and emplacement age of western Greek ophiolites (Vourinos, Pindos, and Othris) inferred from deformation structures in peridotites. In Ophiolites and Oceanic Crust: New Insights from Field Studies and the Ocean Drilling Program; Dilek, Y., Moores, E., Elthon, D., Nicolas, A., Eds.; Geological Society of America: Boulder, CO, USA, 2000; pp. 473–484. [Google Scholar]
- Economou, M.; Dimou, E.; Economou, G.; Migiros, G.; Vacondios, I.; Grivas, E.; Rassios, A.; Dabitzias, S. Chromite deposits of Greece. In Chromites, UNESCO’s IGCP197 Project Metallogeny of Ophiolites; Petrascheck, W., Karamata, S., Kravchenko, G.G., Johan, Z., Economou, M., Engin, T., Eds.; Theophrastus Publications S.A.: Athens, Greece, 1986; pp. 129–159. [Google Scholar]
- Garuti, G.; Zaccarini, F.; Economou-Eliopoulos, M. Paragenesis and composition of laurite from chromitites of Othrys (Greece): Implications for Os-Ru fractionation in ophiolite upper mantle of the Balkan Peninsula. Mineral. Deposita 1999, 34, 312–319. [Google Scholar] [CrossRef]
- Tsikouras, B.; Ifandi, E.; Karipi, S.; Grammatikopoulos, T.A.; Hatzipanagiotou, K. Investigation of Platinum-Group Minerals (PGM) from Othrys chromitites (Greece) using superpanning concentrates. Minerals 2016, 6, 94. [Google Scholar] [CrossRef]
- Bortolotti, V.; Chiari, M.; Marcucci, M.; Photiades, A.; Principi, G.; Saccani, E. New geochemical and age data on the ophiolites from the Othrys area (Greece): Implication for the Triassic evolution of the Vardar ocean. Ofioliti 2008, 33, 135–151. [Google Scholar]
- Oxford Diffraction Ltd. CrysAlis RED; Oxford Diffraction Ltd.: Abingdon, UK, 2006. [Google Scholar]
- Oxford Diffraction Ltd. ABSPACK. In CrysAlis RED; Oxford Diffraction Ltd.: Abingdon, UK, 2006. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.J.C. International Tables for Crystallography: Mathematical, Physical, and Chemical Tables; International Union of Crystallography: Chester, UK, 1992; Volume 3. [Google Scholar]
- Mueller, M.H.; Knott, H.W. The crystal structures of Ti2Cu, Ti2Ni, Ti4Ni2O, and Ti4Cu2O. Trans. Met. Soc. AIME 1963, 227, 674–678. [Google Scholar]
- Smith, D.G.W.; Nickel, E.H. A system for codification for unnamed minerals: Report of the Subcommittee for Unnamed Minerals of the IMA Commission on New Minerals, Nomenclature and Classification. Can. Mineral. 2007, 45, 983–1055. [Google Scholar] [CrossRef]
- Guérin, R.; Sergent, M. Nouveaux phosphures ternaires: NiMoP2, NiWP2, CoMoP2 et CoWP2. J. Solid State Chem. 1976, 18, 317–323. [Google Scholar] [CrossRef]
- Rudashevsky, N.S.; Garuti, G.; Andersen, J.C.Ø.; Kretser, Y.L.; Rudashevsky, V.N.; Zaccarini, F. Separation of accessory minerals from rocks and ores by hydroseparation (HS) technology: Method and application to CHR-2 chromitite, Niquelândia intrusion, Brazil. Trans. Inst. Mining Metall. 2002, 111, B87–B94. [Google Scholar] [CrossRef]
- Kapsiotis, A.; Grammatikopoulos, T.A.; Tsikouras, B.; Hatzipanagiotou, K. Platinum-group mineral characterization in concentrates from high-grade PGE Al-rich chromitites of Korydallos area in the Pindos ophiolite complex (NW Greece). Resource Geol. 2010, 60, 178–191. [Google Scholar] [CrossRef]
- Grammatikopoulos, T.A.; Kapsiotis, A.; Tsikouras, B.; Hatzipanagiotou, K.; Zaccarini, F.; Garuti, G. Spinel composition, PGE geochemistry and mineralogy of the chromitites from the Vourinos ophiolite complex, northwestern Greece. Can. Mineral. 2011, 49, 1571–1598. [Google Scholar] [CrossRef]
- Malvoisin, B.; Chopin, C.; Brunet, F.; Matthieu, E.; Galvez, M.E. Low-temperature Wollastonite formed by carbonate reduction: A marker of serpentinite redox conditions. J. Petrol. 2012, 53, 159–176. [Google Scholar] [CrossRef]
- Etiope, G.; Ifandi, E.; Nazzari, M.; Procesi, M.; Tsikouras, B.; Ventura, G.; Steele, A.; Tardini, R.; Szatmari, P. Widespread abiotic methane in chromitites. Sci. Rep. 2018, 8, 8728. [Google Scholar] [CrossRef]
- Xiong, Q.; Griffin, W.L.; Huang, J.X.; Gain, S.E.M.; Toledo, V.; Pearson, N.J.; O’Reilly, S.Y. Super-reduced mineral assemblages in “ophiolitic” chromitites and peridotites: The view from Mount Carmel. Eur. J. Mineral. 2017, 29, 557–570. [Google Scholar] [CrossRef]
- Pasek, M.A.; Hammeijer, J.P.; Buick, R.; Gull, M.; Atlas, Z. Evidence for reactive reduced phosphorus species in the early Archean ocean. Proc. Natural Acad. Sci. U.S.A. 2013, 110, 100089–100094. [Google Scholar] [CrossRef] [PubMed]
- Ballhaus, C.; Wirth, R.; Fonseca, R.O.C.; Blanchard, H.; Pröll, W.; Bragagni, A.; Nagel, T.; Schreiber, A.; Dittrich, S.; Thome, V.; et al. Ultra-high pressure and ultra-reduced minerals in ophiolites may form by lightning strikes. Geochem. Perspec. Lett. 2017, 5, 42–46. [Google Scholar] [CrossRef]

| Sample | P | S | V | Fe | Co | Ni | Mo | Total |
|---|---|---|---|---|---|---|---|---|
| ot2gr17an4 | 7.59 | 0.64 | 14.19 | 1.14 | 7.47 | 23.78 | 43.56 | 98.37 |
| ot2gr17an3 | 7.96 | 0.67 | 14.18 | 1.18 | 7.72 | 23.77 | 44.04 | 99.52 |
| ot2gr17an5 | 8.03 | 0.65 | 14.16 | 1.18 | 7.53 | 24.16 | 44.16 | 99.87 |
| ot2gr17an2 | 8.05 | 0.71 | 13.98 | 1.19 | 7.56 | 23.86 | 44.39 | 99.74 |
| ot2gr17an1 | 8.20 | 0.66 | 14.13 | 1.20 | 7.67 | 23.91 | 44.65 | 100.42 |
| average | 7.97 | 0.67 | 14.13 | 1.18 | 7.59 | 23.90 | 44.16 | 99.60 |
| λ (nm) | R | λ (nm) | R |
|---|---|---|---|
| 400 | 54.6 | 560 | 57.0 |
| 420 | 54.9 | 589 | 57.5 |
| 440 | 55.2 | 580 | 57.3 |
| 460 | 55.5 | 600 | 57.6 |
| 470 | 55.7 | 620 | 58.0 |
| 480 | 55.8 | 640 | 58.3 |
| 500 | 56.1 | 650 | 58.5 |
| 520 | 56.4 | 660 | 58.6 |
| 546 | 56.8 | 680 | 58.9 |
| 540 | 56.7 | 700 | 59.2 |
| Atom | Wyckoff | x/a | y/b | z/c | Uiso |
|---|---|---|---|---|---|
| Mo1 | 24f | 0.1941(2) | 0 | 0 | 0.0162(8) |
| Mo2 | 24g | 0.4437(2) | ¼ | ¼ | 0.0169(8) |
| Ni1 | 16e | 0.8319(2) | 0.8319(2) | 0.8319(2) | 0.0189(7) |
| Ni2 | 16e | 0.4186(2) | 0.4186(2) | 0.4186(2) | 0.0151(7) |
| P1 | 16e | 0.6231(9) | 0.6231(9) | 0.6231(9) | 0.0288(6) |
| P2 | 4a | 0 | 0 | 0 | 0.056(17) |
| Mo1–P2 | 2.100(2) | Ni2–P1 (×3) | 2.303(11) |
|---|---|---|---|
| Mo1–Ni1 (×2) | 2.588(2) | Ni2–Ni2 (×3) | 2.492(5) |
| Mo1–Ni2 (×2) | 2.730(2) | Ni2–Mo2 (×3) | 2.594(2) |
| Mo1–P1 (×2) | 2.732(2) | Ni2–Mo1 (×3) | 2.730(2) |
| Mo1–Mo2 (×4) | 2.8384(2) | P1–Ni2 (×3) | 2.303(11) |
| Mo1–Mo1 | 2.970(3) | P1–Ni1 (×3) | 2.362(11) |
| Mo2–Ni2 (×2) | 2.594(2) | P1–Mo1 (×3) | 2.732(2) |
| Mo2–Ni1 (×2) | 2.733(2) | P1–Mo2 (×3) | 2.746(3) |
| Mo2–P1 (×2) | 2.746(3) | P2–Mo1 (×6) | 2.100(2) |
| Mo2–Mo1 (×4) | 2.8384(2) | ||
| Mo2–Mo2 (×2) | 2.964(3) | ||
| Ni1–P1 (×3) | 2.362(11) | ||
| Ni1–Ni1 (×3) | 2.507(5) | ||
| Ni1–Mo1 (×3) | 2.588(2) | ||
| Ni1–Mo2 (×3) | 2.733(2) |
| h | k | l | dcalc | Icalc |
|---|---|---|---|---|
| 2 | 2 | 2 | 3.1239 | 5 |
| 4 | 0 | 0 | 2.7054 | 13 |
| 3 | 3 | 1 | 2.4826 | 12 |
| 4 | 2 | 2 | 2.2089 | 42 |
| 5 | 1 | 1 | 2.0826 | 65 |
| 3 | 3 | 3 | 2.0826 | 35 |
| 4 | 4 | 0 | 1.9130 | 21 |
| 4 | 4 | 2 | 1.8036 | 8 |
| 5 | 5 | 1 | 1.5153 | 5 |
| 7 | 3 | 1 | 1.4088 | 5 |
| 7 | 3 | 3 | 1.3221 | 6 |
| 6 | 6 | 0 | 1.2753 | 14 |
| 8 | 2 | 2 | 1.2753 | 17 |
| 5 | 5 | 5 | 1.2496 | 7 |
| 9 | 3 | 3 | 1.0876 | 8 |
| 8 | 8 | 4 | 0.9018 | 5 |
| 11 | 5 | 1 | 0.8925 | 8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaccarini, F.; Bindi, L.; Ifandi, E.; Grammatikopoulos, T.; Stanley, C.; Garuti, G.; Mauro, D. Tsikourasite, Mo3Ni2P1+x (x < 0.25), a New Phosphide from the Chromitite of the Othrys Ophiolite, Greece. Minerals 2019, 9, 248. https://doi.org/10.3390/min9040248
Zaccarini F, Bindi L, Ifandi E, Grammatikopoulos T, Stanley C, Garuti G, Mauro D. Tsikourasite, Mo3Ni2P1+x (x < 0.25), a New Phosphide from the Chromitite of the Othrys Ophiolite, Greece. Minerals. 2019; 9(4):248. https://doi.org/10.3390/min9040248
Chicago/Turabian StyleZaccarini, Federica, Luca Bindi, Elena Ifandi, Tassos Grammatikopoulos, Chris Stanley, Giorgio Garuti, and Daniela Mauro. 2019. "Tsikourasite, Mo3Ni2P1+x (x < 0.25), a New Phosphide from the Chromitite of the Othrys Ophiolite, Greece" Minerals 9, no. 4: 248. https://doi.org/10.3390/min9040248
APA StyleZaccarini, F., Bindi, L., Ifandi, E., Grammatikopoulos, T., Stanley, C., Garuti, G., & Mauro, D. (2019). Tsikourasite, Mo3Ni2P1+x (x < 0.25), a New Phosphide from the Chromitite of the Othrys Ophiolite, Greece. Minerals, 9(4), 248. https://doi.org/10.3390/min9040248








