Formation Conditions and 40Ar/39Ar Age of the Gem-Bearing Boqueirão Granitic Pegmatite, Parelhas, Rio Grande do Norte, Brazil
Abstract
:1. Introduction
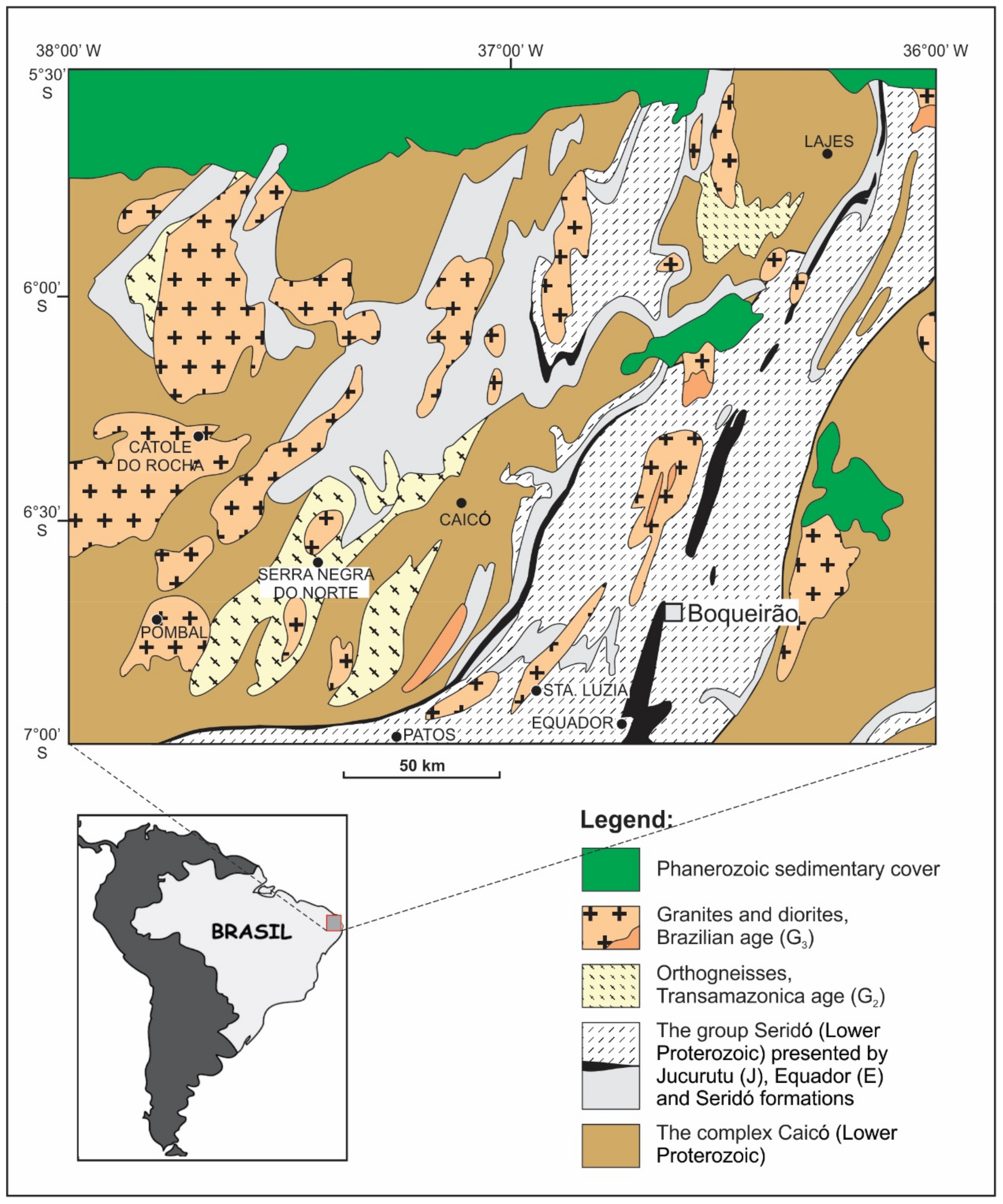
2. Geological Setting
3. Materials and Methods
4. Results
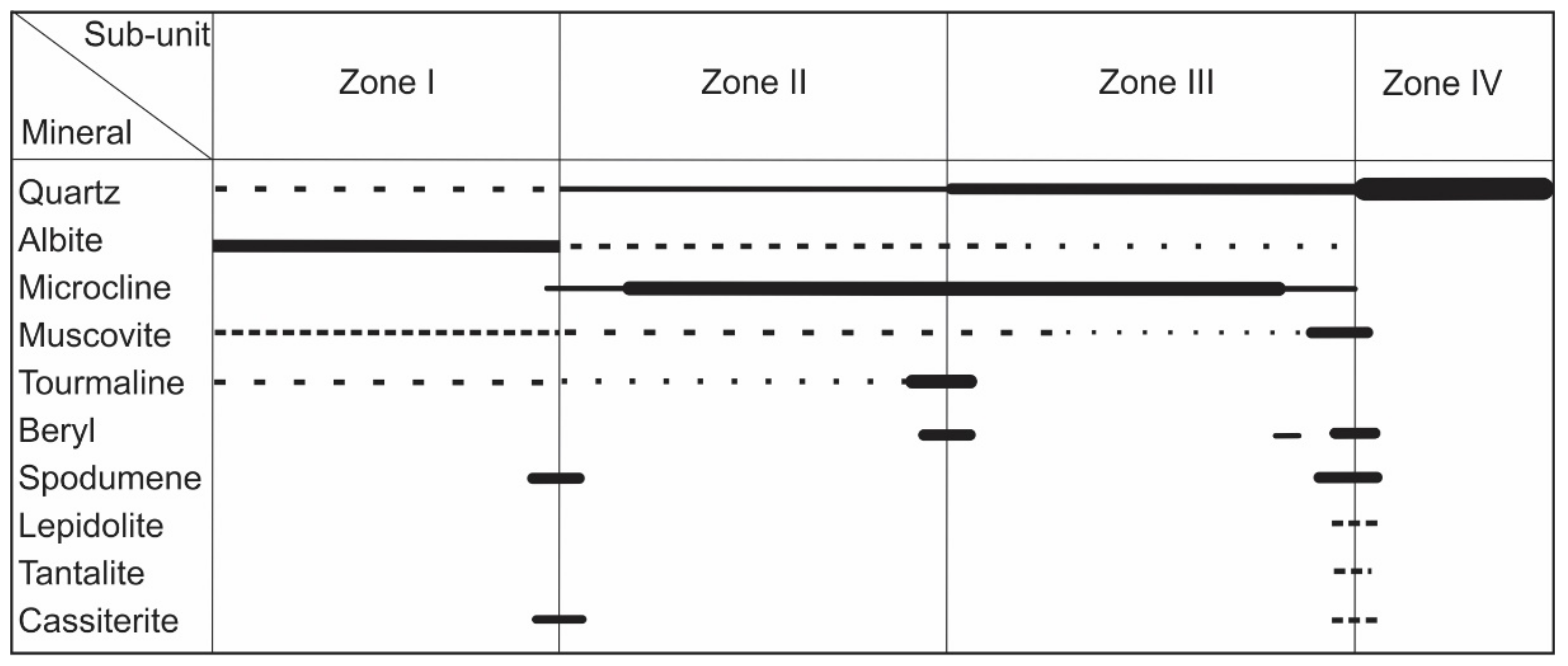
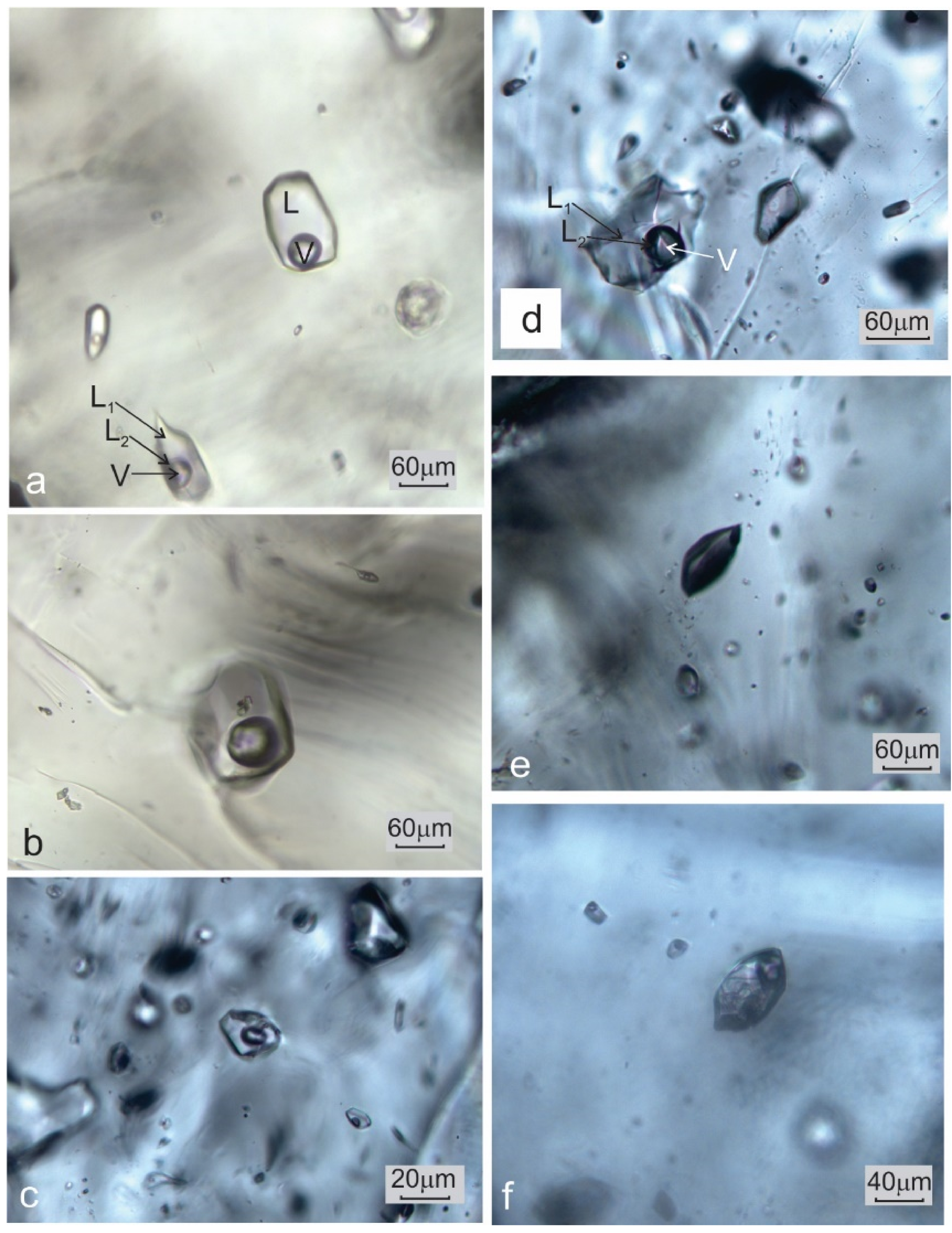
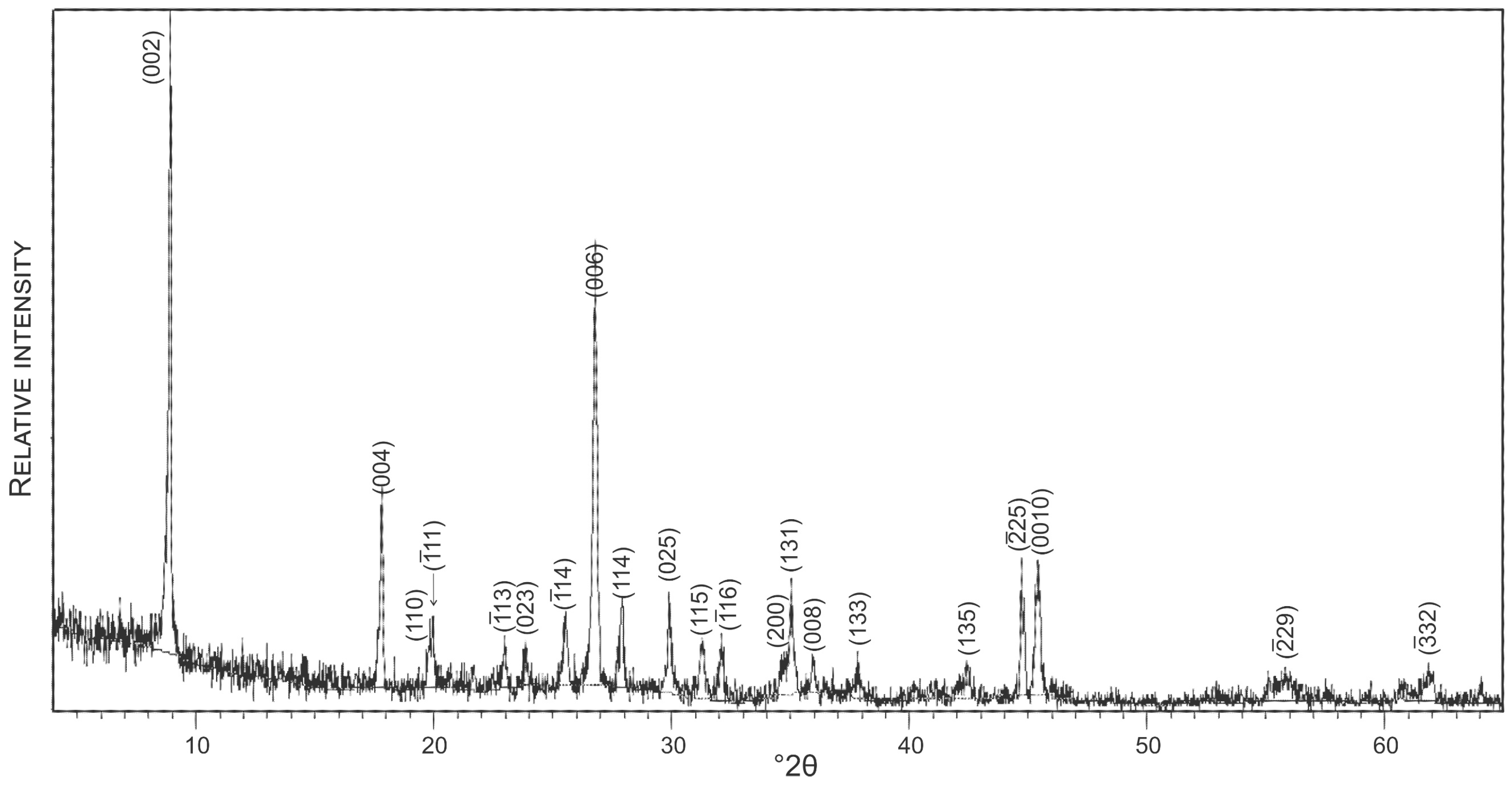
4.1. Petrography of Fluid Inclusions
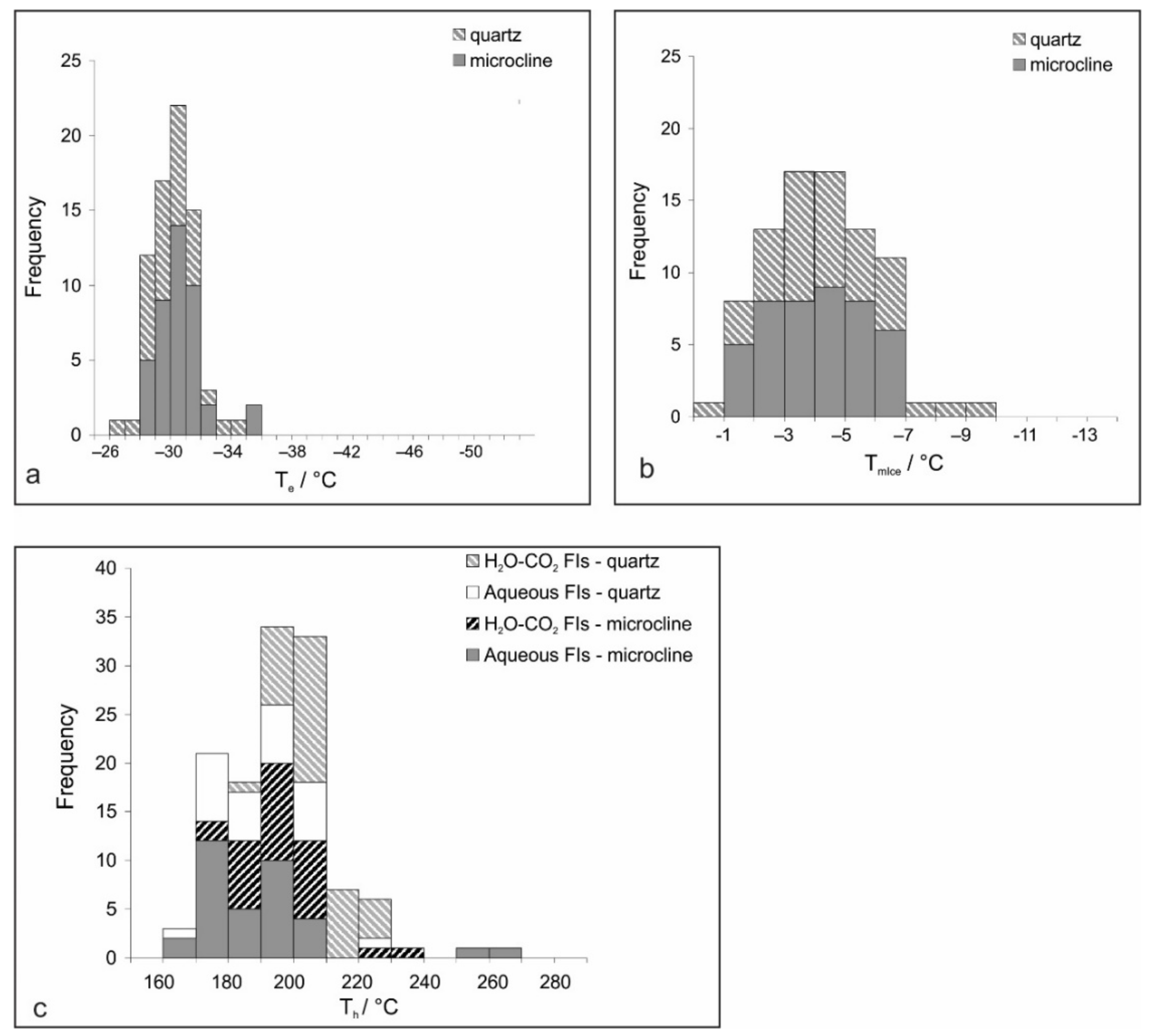
4.2. Microthermometry of Fluid Inclusions
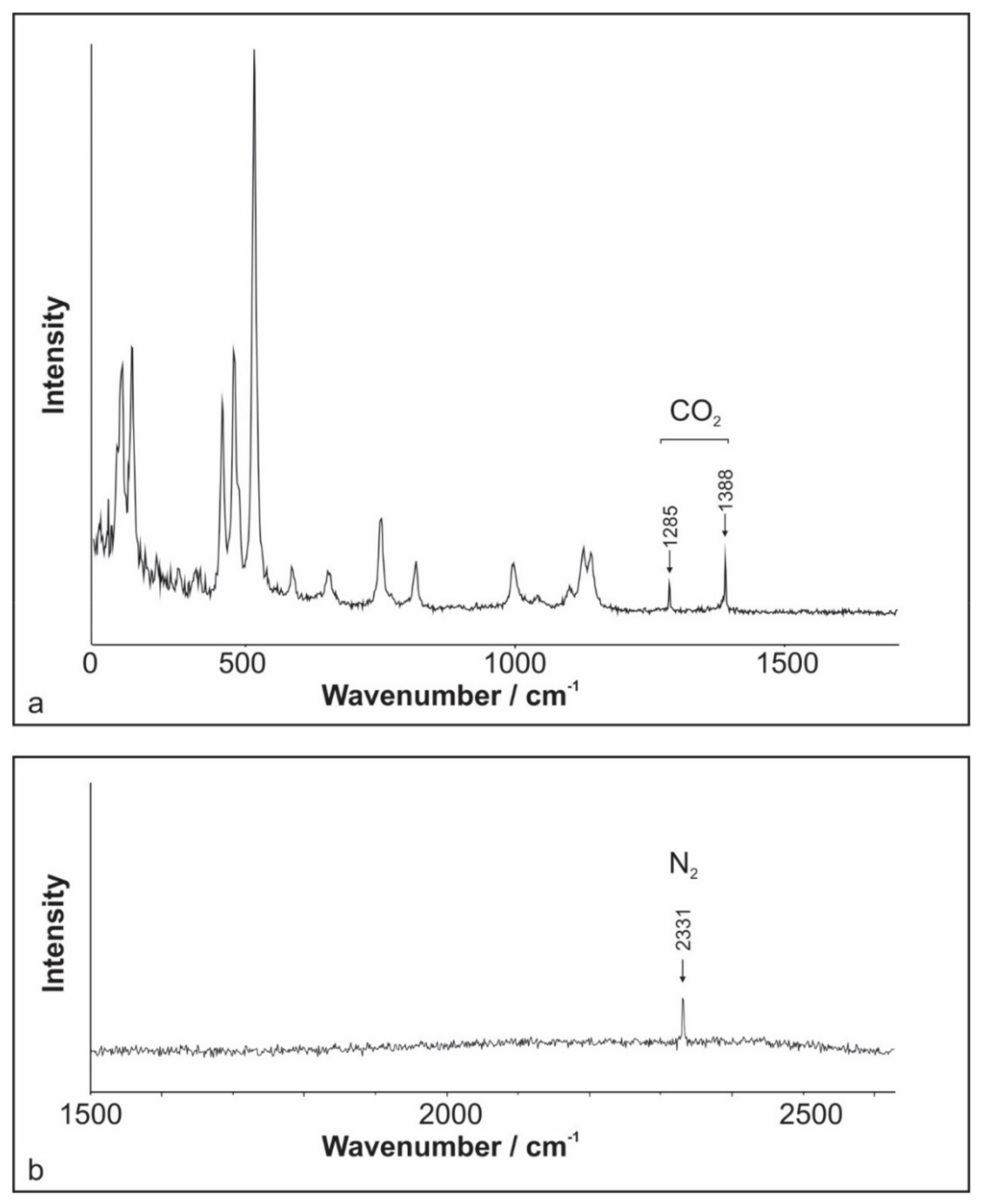
4.3. Raman Spectroscopy of Fluid Inclusions
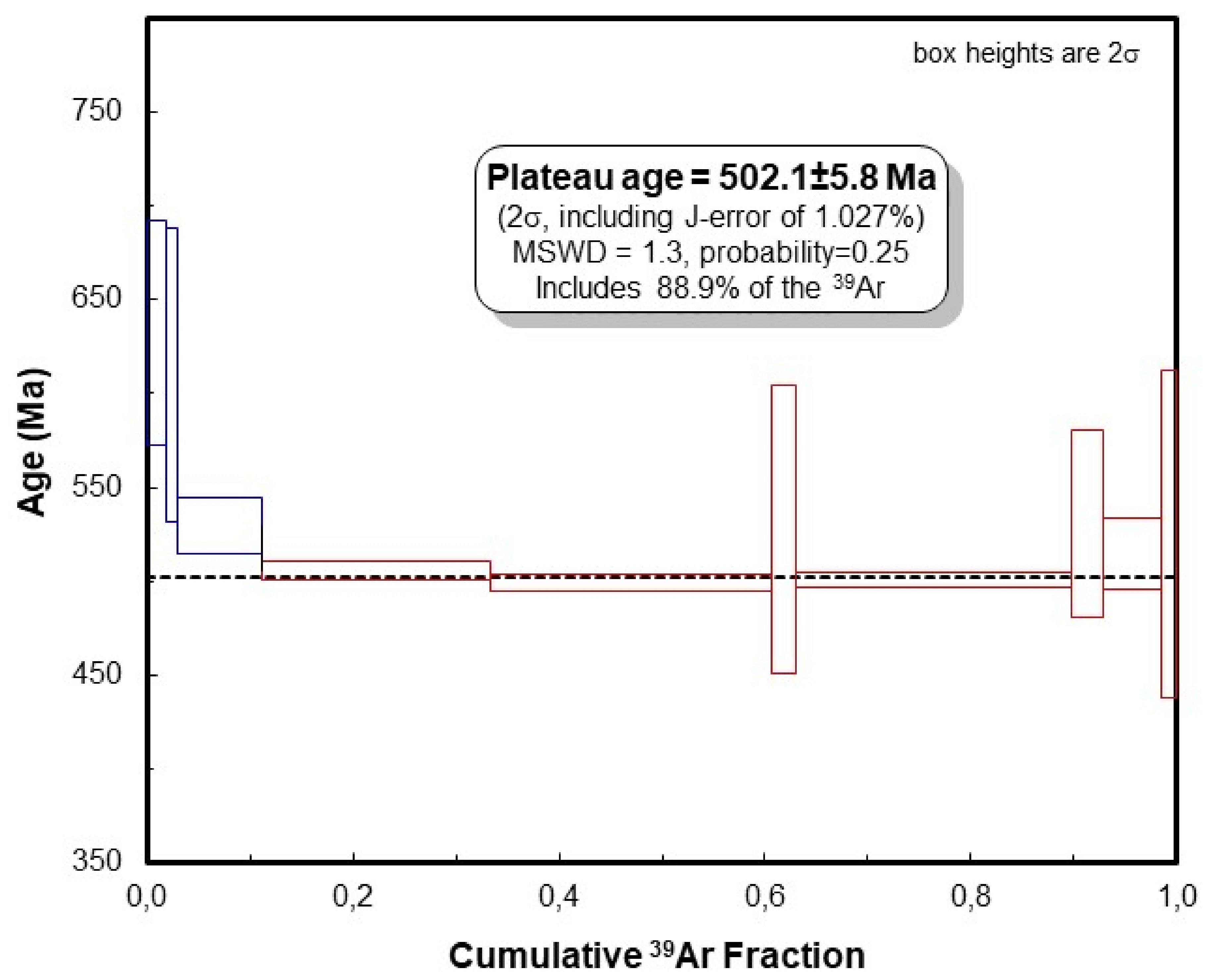
4.4. The 40Ar/39Ar Age
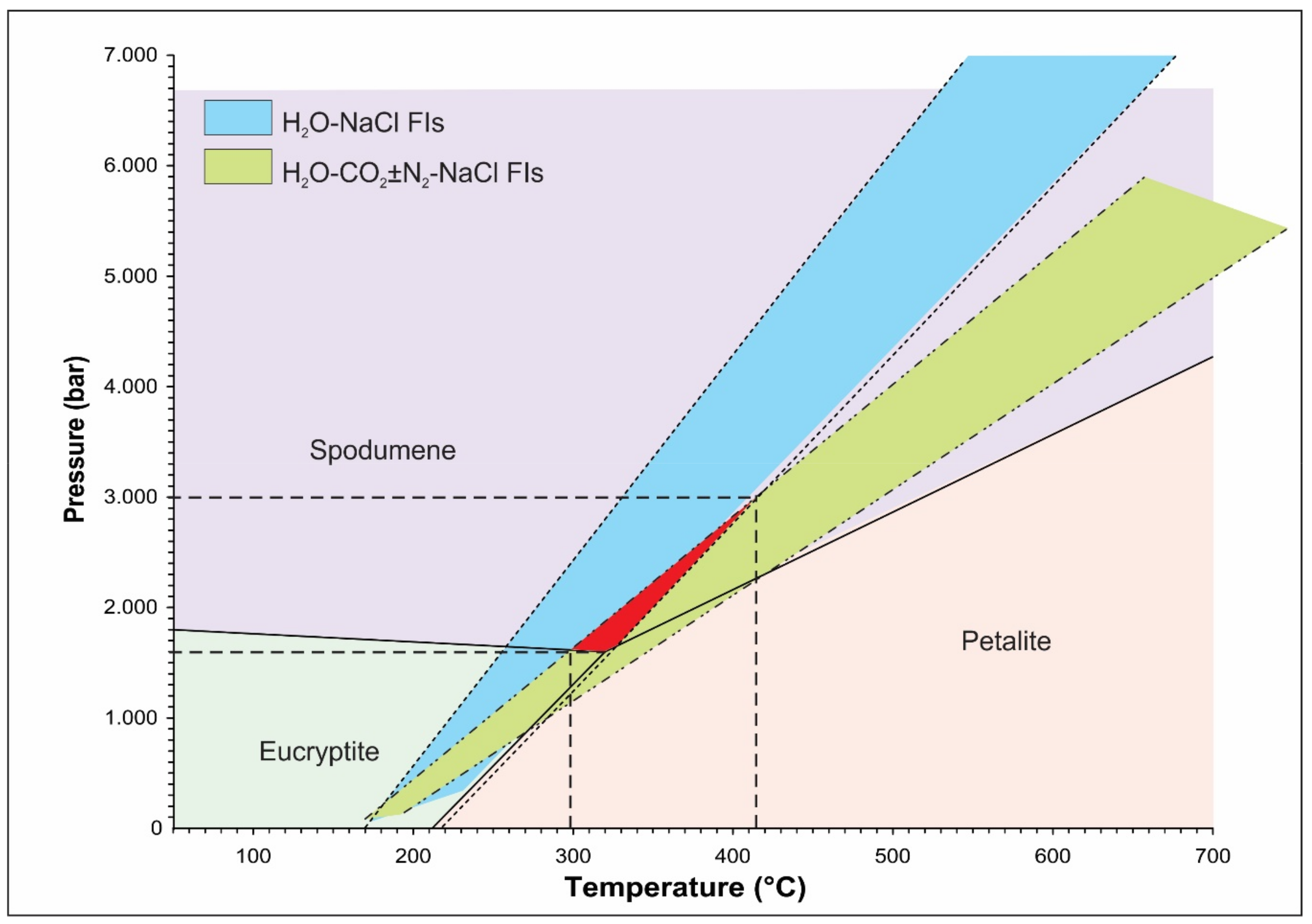
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- London, D. Ore-forming processes within granitic pegmatites. Ore Geol. Rev. 2018, 101, 349–383. [Google Scholar] [CrossRef]
- Pough, M.W. Simpsonite and the Northern Brazilian pegmatite region. Bull. Geol. Soc. Am. 1945, 56, 505–514. [Google Scholar] [CrossRef]
- Beurlen, H. The mineral resources of the Borborema Province in Northeastern Brazil and its sedimentary cover: A review. J. Am. Earth Sci. 1995, 8, 365–376. [Google Scholar] [CrossRef]
- Magyar, M.J. Tantalum. In Mineral Commodity Summaries 2007; U.S. Geological Survey: Reston, VA, USA, 2007; pp. 164–165. [Google Scholar]
- Beurlen, H.; Barreto, S.; Martin, R.; Melgarejo, J.; Da Silva, M.R.R.; Souza Neto, J.A. The Borborema pegmatite province. NE Brazil revisited. Estud. Geológicos 2009, 19, 62–66. [Google Scholar]
- Beurlen, H.; Da Silva, M.R.R.; Thomas, R.; Soares, D.R.; Olivier, P. Nb–Ta–(Ti–Sn) oxide mineral chemistry as tracer of rare-element granitic pegmatite fractionation in the Borborema Province, Northeastern Brazil. Miner. Depos. 2008, 43, 207–228. [Google Scholar] [CrossRef]
- Černy, P. Rare element granitic pegmatites. Part 1: Anatomy and internal evolution of pegmatite deposits. Geosci. Can. 1991, 18, 49–67. [Google Scholar]
- Černy, P.; Ercit, T.S. The Classification of Granitic Pegmatites Revisited. Can. Miner. 2005, 43, 2005–2026. [Google Scholar] [CrossRef]
- Soares, D.R.; Beurlen, H.; Barreto, S.D.B.; Da Silva, M.R.R.; Ferreira, A.C.M. Compositional variation of tourmaline-group minerals in the borborema pegmatite province, northeastern brazil. Can. Miner. 2008, 46, 1097–1116. [Google Scholar] [CrossRef]
- Beurlen, H.; da Silva, M.R.; de Castro, C. Fluid inclusion microthermometry in Be–Ta–(Li–Sn)-bearing pegmatites from the Borborema Province, northeast Brazil. Chem. Geol. 2001, 173, 107–123. [Google Scholar] [CrossRef]
- Thomas, R.; Davidson, P.; Beurlen, H. Tantalite-(Mn) from the Borborema Pegmatite Province. northeastern Brazil: Conditions of formation and melt- and fluid-inclusion constraints on experimental studies. Miner. Depos. 2011, 46, 749–759. [Google Scholar] [CrossRef]
- Strmić Palinkaš, S.; Wegner, R.; Čobić, A.; Palinkaš, L.A.; Barreto, S.D.B.; Váczi, T.; Bermanec, V. The role of magmatic and hydrothermal processes in the evolution of Be-bearing pegmatites: Evidence from beryl and its breakdown products. Am. Mineral. 2014, 99, 424–432. [Google Scholar] [CrossRef]
- Scorza, E.P. Província Pegmatítica da Borborema. DNPM. Div Geol Min Boletim. Rio de Janeiro 1944, 112, 57. [Google Scholar]
- Lima, E.S. Metamorphism and Tectonic Evolution in the Seridó Region. Northeastern Brazil. Ph.D. Thesis, Universty of California Los Angeles, Los Angeles, CA, USA, 1986; p. 215. [Google Scholar]
- Jardim de Sá, E.F.; Legrand, J.M.; McReath, I. Estratigrafia de rochas granitóides na Região do Seridó (RN-PB) com base em critérios estruturais. Rev. Bras. Geoc. 1981, 11, 50–57. [Google Scholar] [CrossRef]
- Araújo, M.N.C.; Silva, F.C.A.; Jardim, E.F. Pegmatite emplacement in the Seridó Belt. Northeastern Brazil: Late stage tectonics of the Brasiliano Orogen. Gondwana Res. 2001, 4, 75–85. [Google Scholar]
- DA Silva, M.R.R.; Höll, R.; Beurlen, H. Borborema Pegmatitic Province: Geological and geochemical characteristics. J. S. Am. Earth Sci. 1995, 8, 355–364. [Google Scholar] [CrossRef]
- Johnston, J.R. Beryl–tantalite pegmatites of Northeastern Brazil. Geol. Soc. Am. Bull. 1945, 56, 1015–1070. [Google Scholar] [CrossRef]
- Baumgartner, R.; Romer, R.L.; Möritz, R.; Sallet, R.; Chiaradia, M. Columbite-tantalite-bearing granitic pegmatites from the serido belt, northeastern brazil: Genetic constraints from U-Pb dating and Pb isotopes. Can. Miner. 2006, 44, 69–86. [Google Scholar] [CrossRef]
- Goldstein, R.H. Fluid inclusions in sedimentary and diagenetic systems. Lithos 2001, 55, 159–193. [Google Scholar] [CrossRef]
- Goldstein, R.H.; Reynolds, T.J. Systematics of fluid inclusions in diagenetic minerals. Soc. Sediment. Geol. Short Course 1994, 31, 199. [Google Scholar]
- Bodnar, R.J. Introduction to fluid inclusions. In Short Course, Fluid Inclusions: Analysis and Interpretation; Mineralogical Association of Canada: Ottawa, ON, Canada, 2003; Volume 32, pp. 1–8. [Google Scholar]
- Bakker, R.J.; Package, F. Computer programs for analysis of fluid inclusion data and for modelling bulk fluid properties. Chem. Geol. 2003, 194, 3–23. [Google Scholar] [CrossRef]
- Naden, J. Calcic Brine: A Microsoft Excel 5.0 Add-in for Calculating Salinities from Microthermometric Data in the System NaCl−CaCl2−H2O; Brown, P.E., Hagemann, S.G., Eds.; PACROFI VI: Madison, WI, USA, 1996; pp. 97–98. [Google Scholar]
- Collins, P.L. Gas hydrates in CO2-bearing fluid inclusions and the use of freezing data for estimation of salinity. Econ. Geol. 1979, 74, 1435–1444. [Google Scholar] [CrossRef]
- Wijbrans, J.R.; Pringle, M.S.; Koopers, A.A.; Schveers, R. Argon geochronology of small samples using the Vulkaan argon laserprobe. Proc. K. Ned. Akad. Van Wet. 1995, 98, 185–218. [Google Scholar]
- Mcdougall, I.; Harrison, M.T. Geochronology and Thermochronology by the 40Ar/39Ar Method, 2nd ed.; Oxford University Press: Oxford, UK, 1999; p. 26. [Google Scholar]
- Ludwig, K.R. Users Manual for ISOPLOT/Ex 3.75c: A Geochronological Toolkit for Microsoft Excel; Special Publication; Berkeley Geochronology Center: Berkeley, CA, USA, 2012. [Google Scholar]
- Steiger, R.H.; Jäger, E. Subcommission on geochronology: Convention on the use of decay constants in geo- and cosmochronology. Earth Planet. Sci. Lett. 1977, 36, 359–362. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Frantz, J.D. Determination of the homogenization temperatures and densities of supercritical fluids in the system NaCl-KCl-CaCl2-H2O using synthetic fluid inclusions. Chem. Geol. 1987, 64, 335–350. [Google Scholar] [CrossRef]
- Burrus, R.C. Hydrocarbon fluid inclusions in studies of sedimentary diagenesis. In Fluid Inclusions: Applications to Petrology; Short Course Notes; Mineralogical Association of Canada: Toronto, ON, Canada, 1981; Volume 6, pp. 138–156. [Google Scholar]
- Bowers, T.S.; Helgeson, H.C. Fortran programs for generating fluid inclusion isochores and fugacity coefficients for the system H2O-CO2-NaCl at high pressures and temperatures. Comput. Geosci. 1985, 11, 203–213. [Google Scholar] [CrossRef]
- Bakker, R.J. Adaptation of the Bowers and Helgeson (1983) equation of state to the H2O–CO2–CH4–N2–NaCl system. Chem. Geol. 1999, 154, 225–236. [Google Scholar] [CrossRef]
- Schrötter, H.W.; Klöckner, H.W. Raman scattering cross-sections in gases and liquids. In Raman Spectroscopy of Gases and Liquids; Weber, A., Ed.; Springer: Berlin, Germany, 1979; pp. 123–166. [Google Scholar]
- Wopenka, B.; Pasteris, J.D. Limitation to quantitative analysis of fluid inclusions in geological samples by laser Raman microprobe spectroscopy. Appl. Spectrosc. 1986, 40, 144–151. [Google Scholar] [CrossRef]
- Wopenka, B.; Pasteris, J.D. Raman intensities and detection limits of geochemically relevant gas mixtures for a laser Raman microprobe. Anal. Chem. 1987, 59, 2165–2170. [Google Scholar] [CrossRef]
- Dubessy, J.; Poty, B.; Ramboz, C. Advances in C-O-H-N-S fluid geochemistry based on micro-Raman spectroscopic analysis of fluid inclusions. Eur. J. Mineral. 1989, 1, 517–534. [Google Scholar] [CrossRef]
- Burke, E.A. Raman microspectrometry of fluid inclusions. Lithos 2001, 55, 139–158. [Google Scholar] [CrossRef]
- Harrison, T.M.; Célérier, J.; Aikman, A.B.; Hermann, J.; Heizler, M.T. Diffusion of 40Ar in muscovite. Geochim. Cosmochim. Acta 2009, 73, 1039–1051. [Google Scholar] [CrossRef]
- Roedder, E. Fluid inclusions. Rev. Mineral. 1984, 12, 644. [Google Scholar]
- Thomas, A.; Spooner, E. The volatile geochemistry of magmatic H2O-CO2 fluid inclusions from the Tanco zoned granitic pegmatite, southeastern Manitoba, Canada. Geochim. Cosmochim. Acta 1992, 56, 49–65. [Google Scholar] [CrossRef]
- London, D. Geochemical features of peraluminous granites. pegmatites. and rhyolites as sources of lithophile metal deposits. Magmas, Fluids and Ore Deposits. In Magmas, Fluids, and Ore Deposits; Short Course Handbook; Mineralogical Association of Canada: Nepean, ON, Canada, 1995; Volume 23, pp. 175–202. [Google Scholar]
- Webster, J.D.; Thomas, R.; Rhede, D.; Förster, H.-J.; Seltmann, R. Melt inclusions in quartz from an evolved peraluminous pegmatite: Geochemical evidence for strong tin enrichment in fluorine-rich and phosphorus-rich residual liquids. Geochim. Cosmochim. Acta 1997, 61, 2589–2604. [Google Scholar] [CrossRef]
- Thomas, R.; Webster, J.D.; Heinrich, W. Melt inclusions in pegmatite quartz: Complete miscibility between silicate melts and hydrous fluids at low pressure. Contrib. Mineral. Petrol. 2000, 139, 394–401. [Google Scholar] [CrossRef]
- Sirbescu, M.-L.C.; Nabelek, P.I. Crystallization conditions and evolution of magmatic fluids in the Harney Peak Granite and associated pegmatites, Black Hills, South Dakota—Evidence from fluid inclusions. Geochim. Cosmochim. Acta 2003, 67, 2443–2465. [Google Scholar] [CrossRef]
- Jahns, R.H.; Burnham, C.W. Experimental studies of pegmatite genesis: I. A model for the derivation and crystallization of granitic pegmatites. Econ. Geol. 1969, 64, 843–864. [Google Scholar] [CrossRef]
- Veksler, I.V. Liquid immiscibility and its role at the magmatic–hydrothermal transition: A summary of experimental studies. Chem. Geol. 2004, 210, 7–31. [Google Scholar] [CrossRef]
- London, D. Magmatic-hydrothermal transition in Tanco Pegmatite. Am. Mineral. 1986, 71, 76–395. [Google Scholar]
- Veksler, I.V.; Thomas, R.; Schmidt, C. Experimental evidence of three coexisting immiscible fluids in synthetic granitic pegmatite. Am. Mineral. 2002, 87, 775–779. [Google Scholar] [CrossRef]
- Sirbescu, M.-L.C.; Nabelek, P.I. Crustal melts below 400 °C. Geology 2003, 31, 685–688. [Google Scholar] [CrossRef]
- Kaeter, D.; Barros, R.; Menuge, J.F.; Chew, D.M. The magmatic–hydrothermal transition in rare-element pegmatites from southeast Ireland: LA-ICP-MS chemical mapping of muscovite and columbite–tantalite. Geochim. Cosmochim. Acta 2018, 240, 98–130. [Google Scholar] [CrossRef]
- Webster, J.D. The exsolution of magmatic hydrosaline chloride liquids. Chem. Geol. 2004, 210, 33–48. [Google Scholar] [CrossRef]
- Zajacz, Z.; Halter, W.E.; Pettke, T.; Guillong, M. Determination of fluid/melt partition coefficients by LA-ICPMS analysis of co-existing fluid and silicate melt inclusions: Controls on element partitioning. Geochim. Cosmochim. Acta 2008, 72, 2169–2197. [Google Scholar] [CrossRef]
- Nabelek, P.I.; Whittington, A.G.; Sirbescu, M.L.C. The role of H2O in rapid emplacement and crystallization of granite pegmatites: Resolving the paradox of large crystals in highly undercooled melts. Contrib. Mineral. Petrol. 2010, 160, 313–325. [Google Scholar] [CrossRef]
- London, D.; Burt, D.M. Lithium aluminosilicate occurrences in pegmatites and the lithium aluminosilicate phase diagram. Am. Mineral. 1982, 67, 483–493. [Google Scholar]
- London, D. Experimental phase equilibria in the system LiAlSiO4-SiO2-H2O: A petrogenetic grid for lithium-rich pegmatites. Am. Mineral. 1984, 69, 995–1004. [Google Scholar]
- Potter, R.W., II. Pressure corrections for fluid-inclusion homogenization temperatures based on the volumetric properties of the system NaCl-H2O. J. Res. U. S. Geol. Surv. 1977, 5, 603–607. [Google Scholar]
- Corsini, M.; De Figueiredo, L.L.; Caby, R.; Féraud, G.; Ruffet, G.; Vauchez, A. Thermal history of the Pan-African/Brasiliano Borborema Province of northeast Brazil deduced from 40Ar/39Ar analysis. Tectonophysics 1998, 285, 103–117. [Google Scholar] [CrossRef]

| Sample | FI | Microthermometry (°C) | Raman (mol %) | Bulk Composition (mol %) | Density (g∙cm−3) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Tmice | Tmclath | ThCO2 | Mode | CO2 | N2 | H2O | CO2 | N2 | |||
| Microcline | |||||||||||
| m-1 | 1 | −2.5 | 8.1 | 26.2 | V | 93.2 | 6.8 | 85.4 | 11.6 | 0.7 | 0.9366 |
| m-1 | 2 | −3.1 | 8.6 | 28.3 | V | 96.5 | 3.5 | 84.4 | 12.6 | 0.2 | 0.9576 |
| m-1 | 3 | −4.3 | 7.8 | 27.5 | L | 98.0 | 2.0 | 83.4 | 12.6 | 0.1 | 0.9691 |
| m-1 | 4 | −2.0 | 8.9 | 27.9 | V | 94.7 | 5.3 | 86.7 | 10.9 | 0.6 | 0.9253 |
| m-2 | 1 | −2.7 | 8.7 | 30.0 | L | 99.5 | 0.5 | 84.3 | 13.2 | <0.05 | 0.9671 |
| m-2 | 2 | −3.3 | 9.2 | 28.0 | L | 97.8 | 2.2 | 84.2 | 12.7 | 0.2 | 0.9608 |
| m-3 | 1 | −4.0 | 8.1 | 28.6 | V | 92.1 | 7.9 | 83.6 | 12.1 | 0.6 | 0.9621 |
| m-3 | 2 | −2.9 | 7.6 | 29.1 | V | 99.9 | 0.1 | 84.1 | 13.2 | <0.01 | 0.9694 |
| m-3 | 3 | −3.1 | 8.8 | 28.8 | V | 93.3 | 6.7 | 85.3 | 11.5 | 0.4 | 0.9359 |
| Quartz | |||||||||||
| q-1 | 1 | −4.0 | 8.2 | 20.1 | V | 93.4 | 6.6 | 83.7 | 12.0 | 0.8 | 0.9673 |
| q-1 | 2 | −3.1 | 8.9 | 18.5 | V | 99.7 | 0.3 | 82.8 | 14.4 | <0.05 | 0.9950 |
| q-1 | 3 | −2.9 | 8.4 | 17.9 | V | 92.6 | 7.4 | 83.5 | 13.0 | 0.9 | 0.9749 |
| J-Value: | 0.01850 | +/− | 0.00019 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Step | 36Ar Meas. | ±σ36 | 37Ar | ±σ37 | 38Ar | ±σ38 | 39Ar | ±σ39 | 40Ar | ±σ40 | 40Ar*/39ArK | ±σ | %40Ar* | %39Ar | Age (Ma) | ±(Ma) |
| Decay Corr. | Meas. | Decay Corr. | Meas. | 1-Sigma Abs. | ||||||||||||
| 1 | 62.9 | 9.3 | 56 | 25 | 120 | 7.9 | 7820 | 66.0 | 1.96 × 105 | 215 | 22.69 | 0.40 | 90.5 | 2.0 | 632.4 | 10.9 |
| 2 | 24.7 | 7.2 | 55 | 19 | 120 | 13 | 4070 | 26.2 | 9.59 × 104 | 183 | 21.74 | 0.54 | 92.4 | 1.1 | 610.0 | 13.9 |
| 3 | 56.1 | 12 | 52 | 29 | 360 | 14 | 33,100 | 68.6 | 6.27 × 105 | 253 | 18.44 | 0.11 | 97.3 | 8.6 | 529.6 | 5.5 |
| 4 | 17.7 | 8.7 | 46 | 22 | 940 | 15 | 90,500 | 119 | 1.59 × 106 | 916 | 17.49 | 0.04 | 99.6 | 23.5 | 505.7 | 4.6 |
| 5 | 99.8 | 1.3 | 37 | 25 | 1100 | 13 | 111,000 | 22.0 | 1.95 × 106 | 1340 | 17.24 | 0.04 | 98.4 | 28.9 | 499.5 | 4.6 |
| 6 | 27.8 | 18 | 63 | 32 | 130 | 11 | 9500 | 68.3 | 1.83 × 105 | 274 | 18.36 | 0.58 | 95.5 | 2.5 | 527.5 | 15.3 |
| 7 | 17.2 | 7.0 | 10 | 25 | 1100 | 18 | 108,000 | 142 | 1.88 × 106 | 1630 | 17.28 | 0.03 | 99.7 | 28.2 | 500.5 | 4.6 |
| 8 | 37.5 | 15 | 67 | 27 | 170 | 11 | 12,300 | 53.1 | 2.39 × 105 | 289 | 18.48 | 0.38 | 95.3 | 3.2 | 530.5 | 10.6 |
| 9 | 28.8 | 11 | 120 | 32 | 260 | 14 | 23,300 | 54.9 | 4.24 × 105 | 338 | 17.85 | 0.14 | 98.0 | 6.1 | 514.7 | 5.9 |
| 10 | 51.1 | 13 | 120 | 39 | 120 | 13 | 6060 | 43.0 | 1.26 × 105 | 187 | 18.25 | 0.67 | 88.0 | 1.6 | 524.7 | 17.3 |
| Total | - | - | - | - | - | - | - | - | - | - | - | - | - | 100.0 | 506.3 | 4.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strmić Palinkaš, S.; Palinkaš, L.; Neubauer, F.; Scholz, R.; Borojević Šoštarić, S.; Bermanec, V. Formation Conditions and 40Ar/39Ar Age of the Gem-Bearing Boqueirão Granitic Pegmatite, Parelhas, Rio Grande do Norte, Brazil. Minerals 2019, 9, 233. https://doi.org/10.3390/min9040233
Strmić Palinkaš S, Palinkaš L, Neubauer F, Scholz R, Borojević Šoštarić S, Bermanec V. Formation Conditions and 40Ar/39Ar Age of the Gem-Bearing Boqueirão Granitic Pegmatite, Parelhas, Rio Grande do Norte, Brazil. Minerals. 2019; 9(4):233. https://doi.org/10.3390/min9040233
Chicago/Turabian StyleStrmić Palinkaš, Sabina, Ladislav Palinkaš, Franz Neubauer, Ricardo Scholz, Sibila Borojević Šoštarić, and Vladimir Bermanec. 2019. "Formation Conditions and 40Ar/39Ar Age of the Gem-Bearing Boqueirão Granitic Pegmatite, Parelhas, Rio Grande do Norte, Brazil" Minerals 9, no. 4: 233. https://doi.org/10.3390/min9040233
APA StyleStrmić Palinkaš, S., Palinkaš, L., Neubauer, F., Scholz, R., Borojević Šoštarić, S., & Bermanec, V. (2019). Formation Conditions and 40Ar/39Ar Age of the Gem-Bearing Boqueirão Granitic Pegmatite, Parelhas, Rio Grande do Norte, Brazil. Minerals, 9(4), 233. https://doi.org/10.3390/min9040233





