Transformation of Vanadium-Bearing Titanomagnetite Concentrate in Additive-Free Roasting and Alkaline-Pressure Leaching for Extracting Vanadium (V)
Abstract
:1. Introduction
2. Thermodynamic Criteria for Roasting
ΔG°r = −10.987 kJ/mol at 1100 K
ΔG°r = −13.204 kJ/mol at 1100 K
3. Materials and Methods
3.1. Raw Materials
3.2. Experimental
4. Results and Discussion
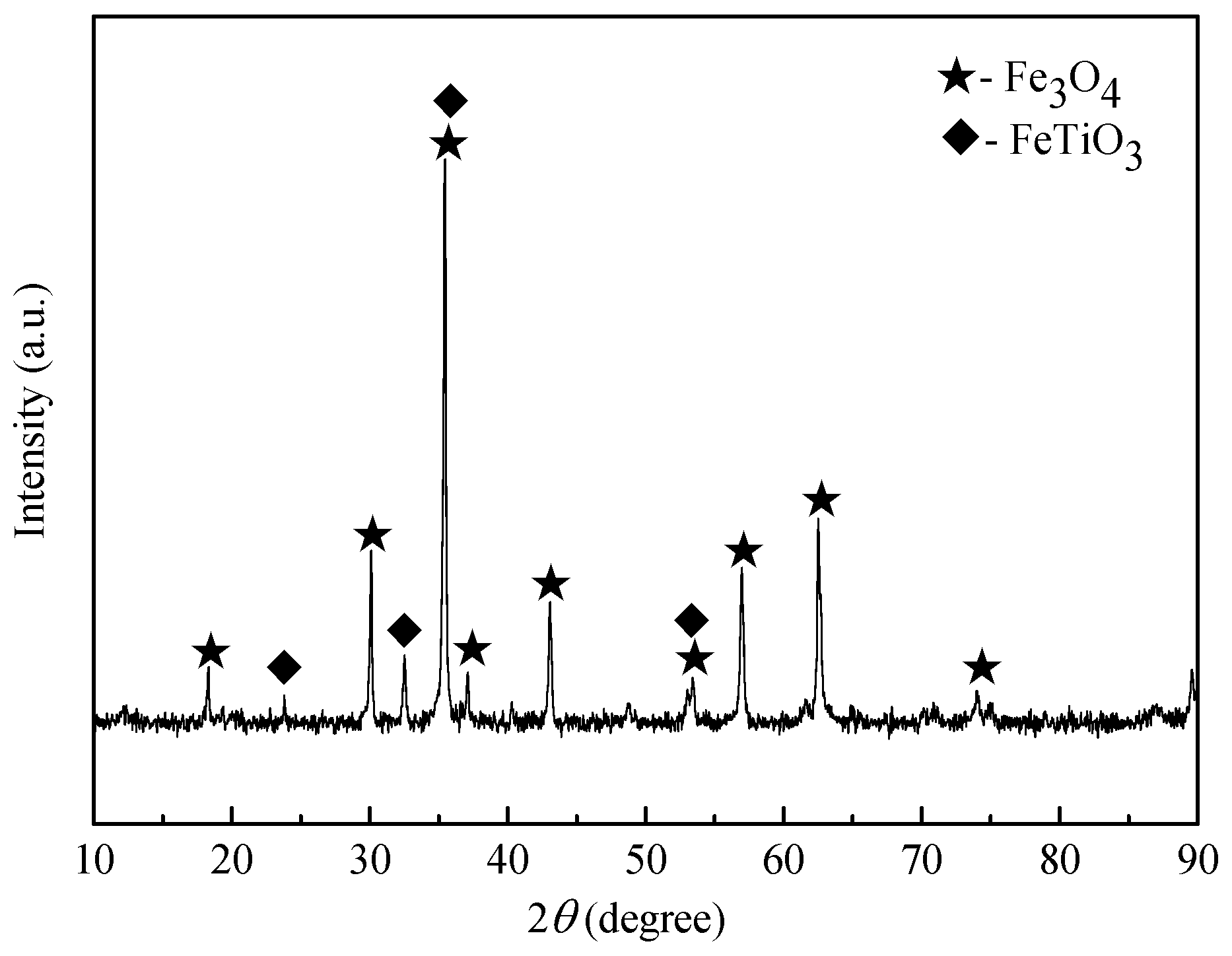
4.1. Mineral Analysis
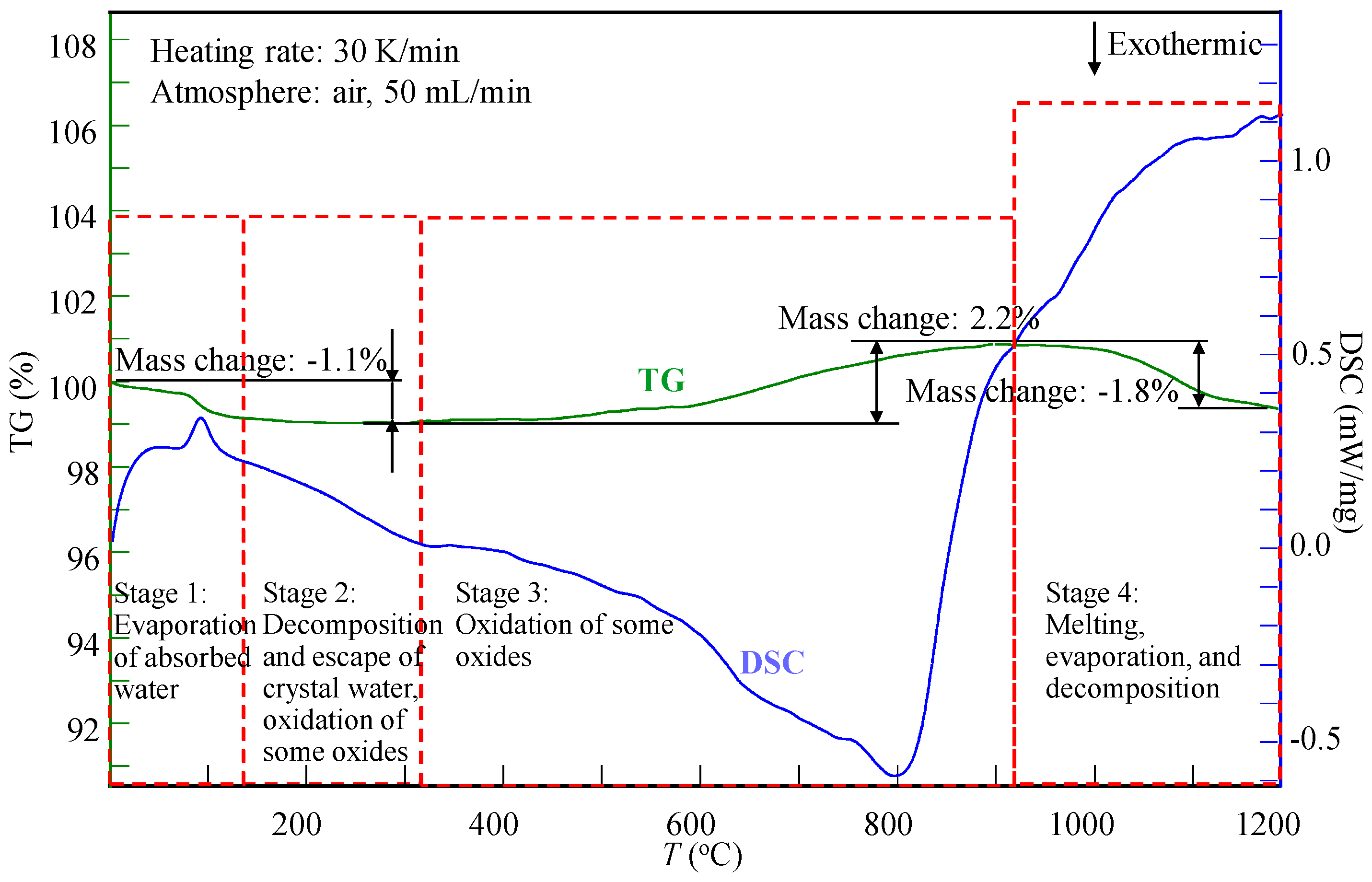
4.2. TG-DSC Analysis
TDec. = 460 °C, TBoi. = 550 °C;
ΔG°r = −74.028 kJ/mol, ΔG°r = −75.890 kJ/mol at 1100 K (Extrapolated data);
ΔH°r = 149.026 kJ/mol, ΔH°r = 166.00 kJ/mol at 1100 K (Extrapolated data)
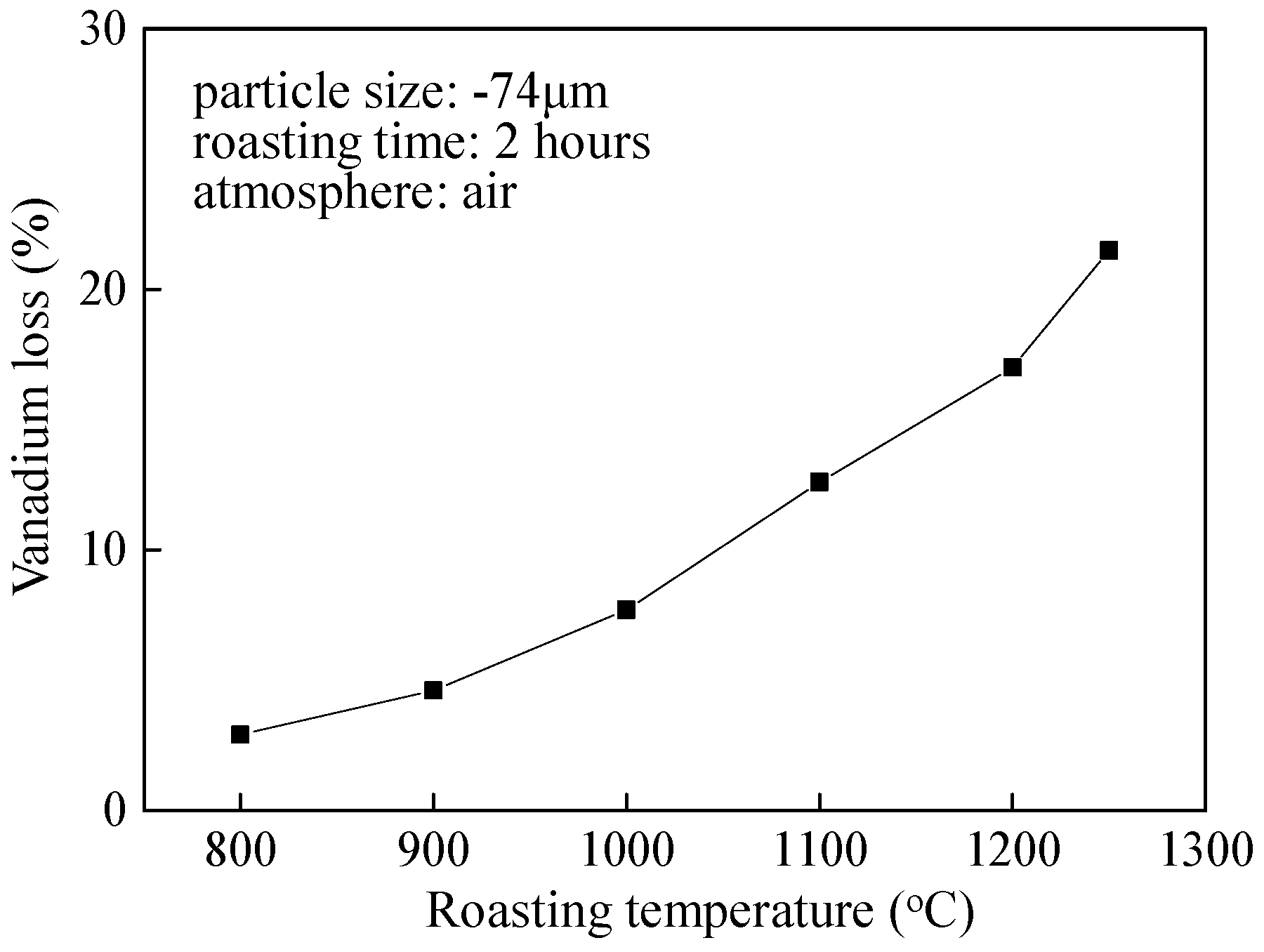
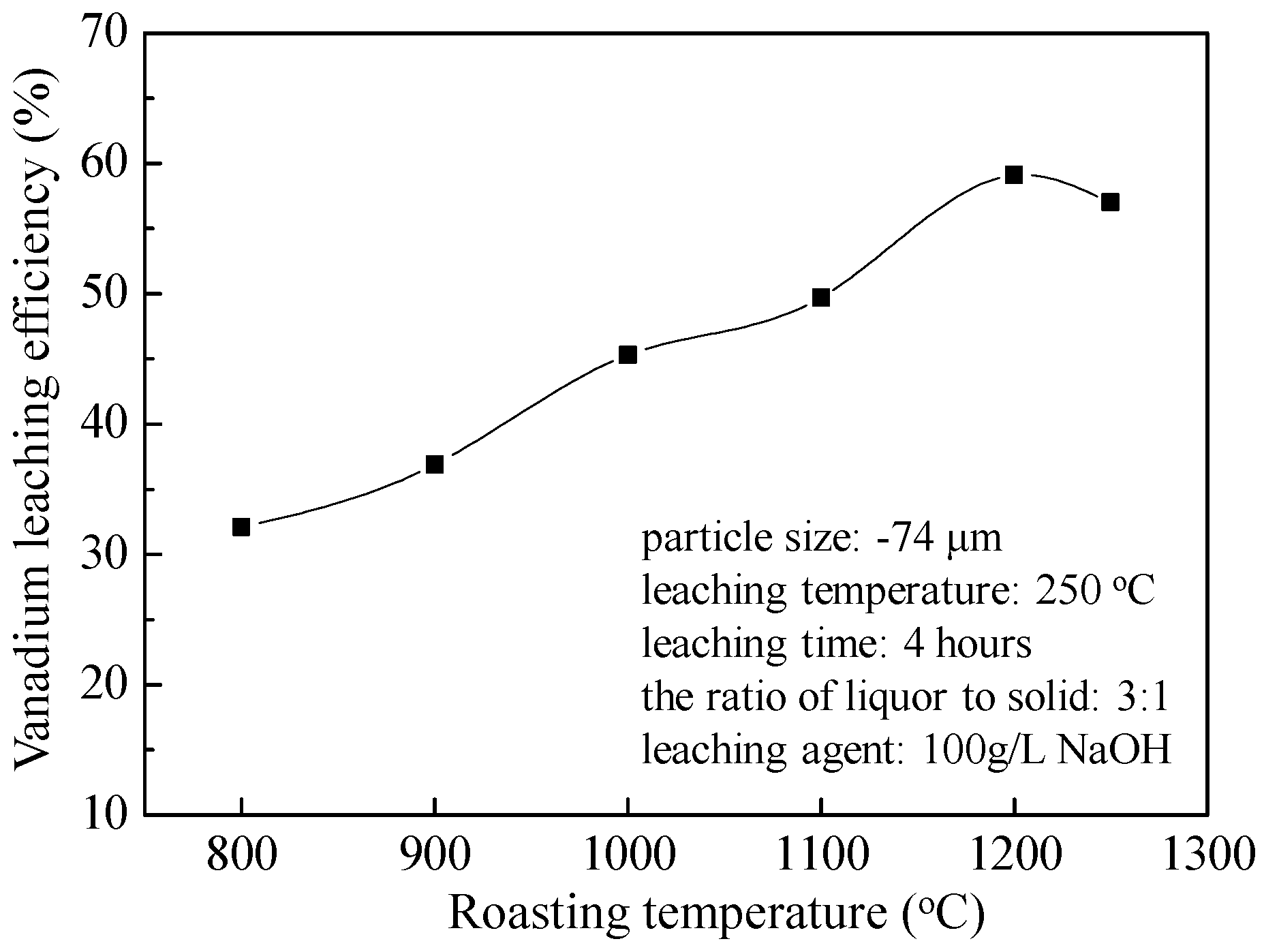
4.3. Vanadium Loss and Vanadium Leaching Efficiency
4.4. Transformation Reactions Analyses in Roasting
ΔG°r = −137.521 kJ/mol O2 at 1300 K
ΔG°r = −156.521 kJ/mol O2 at 1300 K
ΔG°r = −108.968 kJ/mol O2 at 1300 K
ΔG°r = −178.176 kJ/mol O2 at 900 K
ΔG°r = −215.877 kJ/mol O2 at 900 K, ΔG°r = −184.979 kJ/mol O2 at 1100 K
ΔG°r = −128.231 kJ/mol O2 at 1300 K
ΔG°r = −6.221 kJ/mol at 900 K, ΔG°r = −1.044 kJ/mol at 1100 K
5. Conclusions
- Thermodynamic analyses show that oxidation of V2O3 would happen in the roasting experiment over the range of 327–1327 °C and vanadium-iron spinel phase (FeV2O4) can be oxidized more easily than magnetite (Fe3O4) when the temperature is higher than 861 °C.
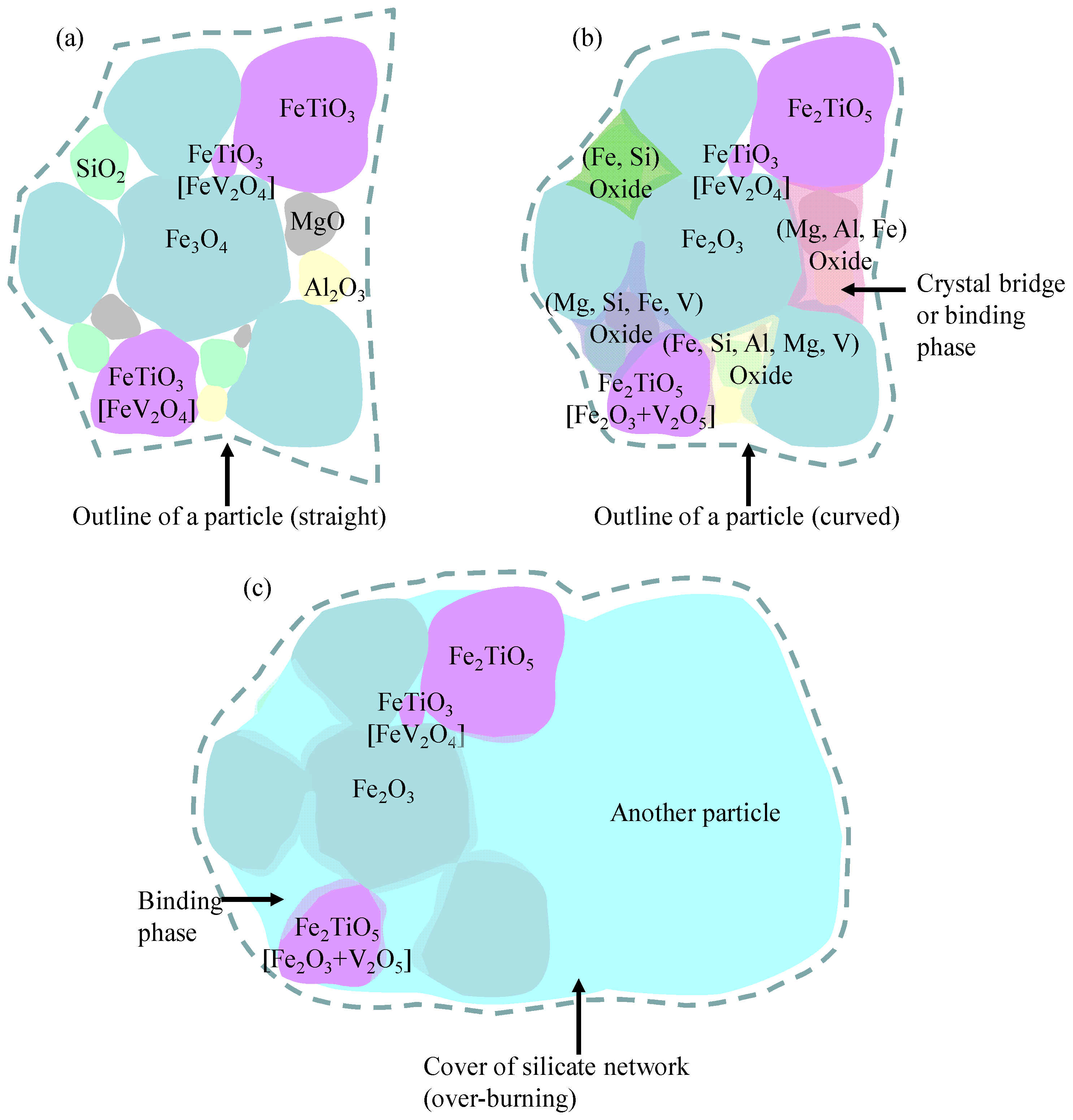
- The compounds (V2O5, Fe2Al4Si5O18, and Fe2SiO4) with low melting temperature were generated at low temperature and melted at elevated temperature; liquids were generated due to some chemical reactions or phase transformation reaction (Fe2V2O4(s) → Fe2O3(s) + liquid) happened at elevated temperature; main phases of Fe2O3 and Fe2TiO5 are connected and sintered with the binding phases of the compounds with low melting temperature or the mixtures with low liquidus temperature.
- Higher roasting temperature is beneficial for improving the vanadium leaching efficiency. However, over-burning causes some vanadium oxide wrapped by silicate network and reductions in the vanadium leaching efficiency from 59.1% at Troa. = 1200 °C to 57% at Troa. = 1250 °C (250 °C of leaching temperature with high pressure of 1.3 MPa, 4 h of leaching time, 3:1 of the ratio of liquor to solid, and 100 g/L NaOH as leaching agent).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hilliard, H. Minerals Yearbook; United States Department of the Interior, Bureau of Mines: Washington, DC, USA, 1993; pp. 1259–1278.
- Hilliard, H. The Materials Flow of Vanadium in the United States; Information Circular 9409; United States Department of the Interior, Bureau of Mines: Washington, DC, USA, 1994; pp. 1–20.
- Liao, S.; Bai, T. Vanadium Metallurgy in Foreign; Metallurgical Industry Press: Beijing, China, 1985; pp. 161–222. [Google Scholar]
- Moskalyk, R.R.; Alfantazi, A.M. Processing of vanadium: A review. Miner. Eng. 2003, 16, 793–805. [Google Scholar] [CrossRef]
- Du, H. Principle of Blast Furnace Smelting Vanadium-Bearing Titanomagnetite; Science Press: Beijing, China, 1996; pp. 16–20. [Google Scholar]
- Hukkanen, E.; Walden, H. The production of vanadium and steel from titanomagnetites. Int. J. Miner. Process. 1985, 15, 89–102. [Google Scholar] [CrossRef]
- Deng, J.; Xue, X.; Liu, G. Current situation and development of comprehensive utilization of vanadium-bearing titanomagnetite at Pangang. J. Mater. Metall. 2007, 6, 83–86. [Google Scholar]
- Zheng, H.; Sun, Y.; Lu, J.; Dong, J.; Zhang, W.; Sheng, F. Vanadium extraction from vanadium-bearing titanomagnetite by selective chlorination using chloride wastes (FeClX). J. Cent. South Univ. 2017, 24, 311–317. [Google Scholar] [CrossRef]
- Zwolak, I. Vanadium carcinogenic, immunotoxic and neurotoxic effects: A review of in vitro studies. Toxicol. Mech. Methods 2014, 24, 1–12. [Google Scholar]
- Boulassel, B.; Sadeg, N.; Roussel, O.; Perrin, M.; Belhadj-Tahar, H. Fatal poisoning by vanadium. Forensic Sci. Int. 2011, 206, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xia, Q.; Wang, Y.; Pan, P.; Luo, D. The progress in production technology of vanadium pentoxide. J. Panzhihua Sci. Technol. 1996, 2, 11–13. [Google Scholar]
- Zhang, Y.; Bao, S.; Liu, T.; Chen, T.; Huang, J. The technology of extracting vanadium from stone coal in China: History, current status and future prospects. Hydrometallurgy 2011, 109, 116–124. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, L.; Qi, T.; Chen, D.; Zhao, H.; Liu, Y. A novel method to extract iron, titanium, vanadium, and chromium from high-chromium vanadium-bearing titanomagnetite concentrates. Hydrometallurgy 2014, 149, 106–109. [Google Scholar] [CrossRef]
- Li, M.; Du, H.; Zhang, S.; Wang, S.; Zhang, Y.; Liu, B.; Dreisinger, D.B.; Zhang, Z. Extraction of vanadium from vanadium slag via non-salt roasting and ammonium oxalate leaching. JOM 2017, 69, 1970–1975. [Google Scholar] [CrossRef]
- Cai, Z.; Zhang, Y.; Liu, T.; Huang, J. Vanadium extraction from refractory stone coal using novel composite additive. JOM 2015, 67, 2629–2634. [Google Scholar] [CrossRef]
- Li, R.; Liu, T.; Zhang, Y.; Huang, J.; Xu, C. Efficient extraction of vanadium from vanadium-titanium magnetite concentrate by potassium salt roasting additives. Minerals 2018, 8, 25. [Google Scholar] [CrossRef]
- Li, X.; Xie, B.; Wang, G.; Li, X. Oxidation process of low-grade vanadium slag in presence of Na2CO3. Trans. Nonferrous Met. Soc. China 2011, 21, 1860–1867. [Google Scholar] [CrossRef]
- Takahashi, H.; Fujiwara, W.; Sun, H.; Yoshida, T.; Matsushima, Y. Extraction of vanadium from ammonia slag under near-atmospheric conditions. Metals 2018, 8, 414. [Google Scholar] [CrossRef]
- Zhu, X.; Li, W.; Guan, X. Vanadium extraction from titano-magnetite by hydrofluoric acid. Int. J. Miner. Process. 2016, 157, 55–59. [Google Scholar] [CrossRef]
- Xiang, J.; Huang, Q.; Lv, X.; Bai, C. Extraction of vanadium from converter slag by two-step sulfuric acid leaching process. J. Clean. Prod. 2018, 170, 1089–1101. [Google Scholar] [CrossRef]
- Chen, D.; Zhao, H.; Hu, G.; Qi, T.; Yu, H.; Zhang, G.; Wang, L.; Wang, W. An extraction process to recover vanadium from low-grade vanadium-bearing titanomagnetite. J. Hazard. Mater. 2015, 294, 35–40. [Google Scholar] [CrossRef]
- Ji, Y.; Shen, S.; Liu, J.; Xue, Y. Cleaner and effective process for extracting vanadium from vanadium slag by using an innovative three-phase roasting reaction. J. Clean. Prod. 2017, 149, 1068–1078. [Google Scholar] [CrossRef]
- Li, H.; Fang, H.; Wang, K.; Zhou, W.; Yang, Z.; Yan, X.; Ge, W.; Li, Q.; Xie, B. Asynchronous extraction of vanadium and chromium from vanadium slag by stepwise sodium roasting-water leaching. Hydrometallurgy 2015, 156, 124–135. [Google Scholar] [CrossRef]
- Li, H.; Wang, K.; Hua, W.; Yang, Z.; Zhou, W.; Xie, B. Selective leaching of vanadium in calcification-roasted vanadium slag by ammonium carbonate. Hydrometallurgy 2016, 160, 18–25. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, T.; David, D.; Lv, G.; Zhang, G.; Zhang, W.; Liu, Y. Extraction of vanadium from direct acid leach solution of converter vanadium slag. Can. Metall. Q. 2017, 56, 281–293. [Google Scholar] [CrossRef]
- Luo, X.; Dong, H.; Zhang, S.; Liu, Y. Study on the sodium oxidation properties of low-iron vanadium-titanium magnetite with high vanadium and titanium. Energy Sources 2018, 40, 1998–2008. [Google Scholar] [CrossRef]
- Zhang, Y.; Yi, L.; Wang, L.; Chen, D.; Wang, W.; Liu, Y.; Zhao, H.; Qi, T. A novel process for the recovery of iron, titanium, and vanadium from vanadium-bearing titanomagnetite: Sodium modification-direct reduction coupled process. Int. J. Miner. Metall. Mater. 2017, 24, 504–511. [Google Scholar] [CrossRef]
- Bauer, G.; Volker, G.; Hans, H.; Andreas, O.; Oskar, R.; Heinz, R.; Siegfried, S. Vanadium and vanadium compounds. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2005; Volume A27, pp. 367–386. [Google Scholar]
- Jena, B.C.; Dresler, W.; Reilly, I.G. Extraction of titanium, vanadium and iron from titanomagnetite deposits at pipestone lake, Manitoba, Canada. Miner. Eng. 1995, 8, 159–168. [Google Scholar] [CrossRef]
- Chen, J. Handbook of Hydrometallurgy; Metallurgical Industry Press: Beijing, China, 2005; pp. 935–966. [Google Scholar]
- Barin, I. Thermochemical Data of Pure Substances, 3rd ed.; Wiley-VCH GmbH: Weinheim, Germany, 1995; pp. 700, 702, 703, 713, 714, 716, 1047, 1505, 1692, 1778, 1780. [Google Scholar]
- ACerS-NIST Phase Equilibria Diagrams (PC Database Ver. 4.0, NIST Standard Reference Database 31); Figures No. 96-1, 696-A; American Ceramic Society: Westerville, OH, USA; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2014.
- Chen, H. Study on extraction of V2O5 from vanadium-bearing slag by lime roasting. Iron Steel Vanadium Titan. 1992, 6, 1–9. [Google Scholar]
- Wang, X. Ferrous Metallurgy; Metallurgical Industry Press: Beijing, China, 2014; Volume 1, p. 45. [Google Scholar]
- Huang, X. Principle of Iron and Steel Metallurgy; Metallurgical Industry Press: Beijing, China, 2017; p. 633. [Google Scholar]





| Composition | Fe2O3 | TiO2 | Al2O3 | SiO2 | MgO | CaO | V2O5 | MnO | Sum |
|---|---|---|---|---|---|---|---|---|---|
| Content (%) | 72.55 | 12.68 | 4.66 | 3.82 | 3.26 | 0.96 | 0.75 | 0.64 | 99.51 |
| Troa. (°C) | m1 (g) | α (%) | m2 (g) | β (%) | η (%) |
|---|---|---|---|---|---|
| 800 | 75.14 | 0.414 | 75.89 | 0.398 | 2.9 |
| 900 | 75.36 | 0.414 | 76.88 | 0.387 | 4.6 |
| 1000 | 75.10 | 0.414 | 75.89 | 0.378 | 7.7 |
| 1100 | 75.21 | 0.414 | 74.18 | 0.367 | 12.6 |
| 1200 | 75.16 | 0.414 | 73.16 | 0.353 | 17.0 |
| 1250 | 75.25 | 0.414 | 72.17 | 0.339 | 21.5 |
| Troa. (°C) | m3 (g) | ψ (%) | m4 (g) | φ (%) | γ (%) |
|---|---|---|---|---|---|
| 800 | 150 | 0.395 | 147.803 | 0.272 | 32.1 |
| 900 | 150 | 0.387 | 147.207 | 0.249 | 36.9 |
| 1000 | 150 | 0.375 | 147.113 | 0.209 | 45.3 |
| 1100 | 150 | 0.361 | 146.562 | 0.186 | 49.7 |
| 1200 | 150 | 0.355 | 146.121 | 0.149 | 59.1 |
| 1250 | 150 | 0.337 | 146.891 | 0.148 | 57.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, H.; Zhang, W.; Guo, Y.; Gao, Q.; Shen, F. Transformation of Vanadium-Bearing Titanomagnetite Concentrate in Additive-Free Roasting and Alkaline-Pressure Leaching for Extracting Vanadium (V). Minerals 2019, 9, 197. https://doi.org/10.3390/min9030197
Zheng H, Zhang W, Guo Y, Gao Q, Shen F. Transformation of Vanadium-Bearing Titanomagnetite Concentrate in Additive-Free Roasting and Alkaline-Pressure Leaching for Extracting Vanadium (V). Minerals. 2019; 9(3):197. https://doi.org/10.3390/min9030197
Chicago/Turabian StyleZheng, Haiyan, Weiling Zhang, Yongchun Guo, Qiangjian Gao, and Fengman Shen. 2019. "Transformation of Vanadium-Bearing Titanomagnetite Concentrate in Additive-Free Roasting and Alkaline-Pressure Leaching for Extracting Vanadium (V)" Minerals 9, no. 3: 197. https://doi.org/10.3390/min9030197
APA StyleZheng, H., Zhang, W., Guo, Y., Gao, Q., & Shen, F. (2019). Transformation of Vanadium-Bearing Titanomagnetite Concentrate in Additive-Free Roasting and Alkaline-Pressure Leaching for Extracting Vanadium (V). Minerals, 9(3), 197. https://doi.org/10.3390/min9030197





