The Effect of Sodium Alginate on Chlorite and Serpentine in Chalcopyrite Flotation
Abstract
1. Introduction
2. Material and Methods
2.1. Pure Minerals and Reagents
2.2. Experiments
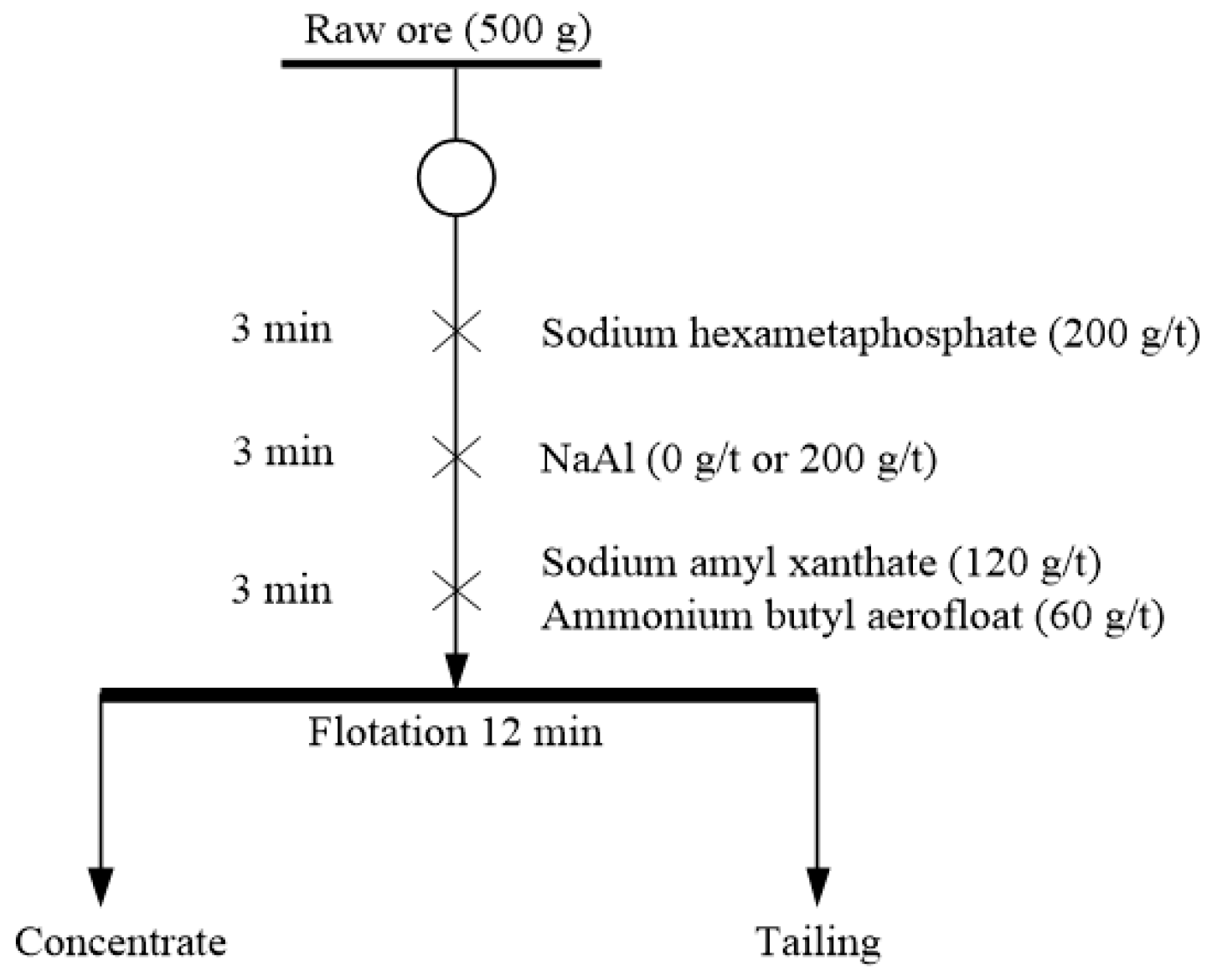
2.2.1. Micro-Flotation Experiments
2.2.2. Batch Flotation Tests
2.2.3. Adsorption Tests
2.2.4. Zeta Potential Measurements
2.2.5. FT-IR Studies
3. Results and Discussion
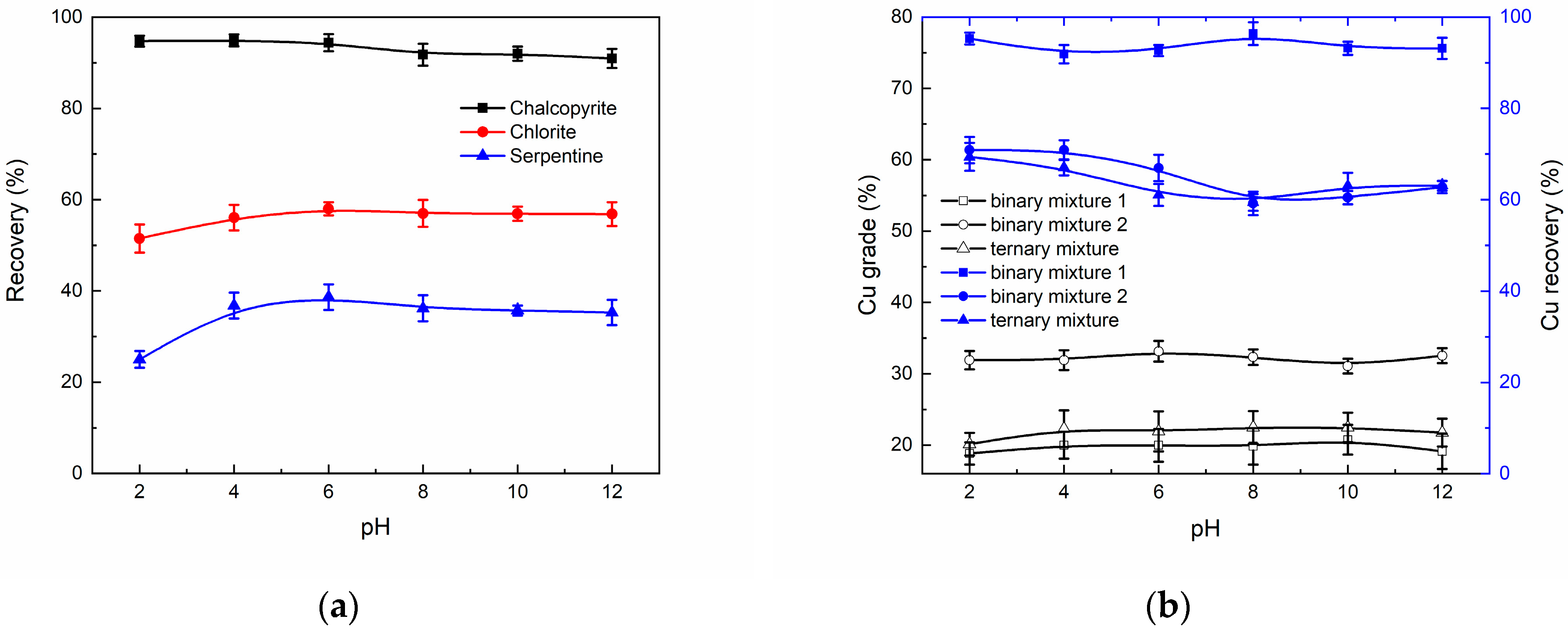
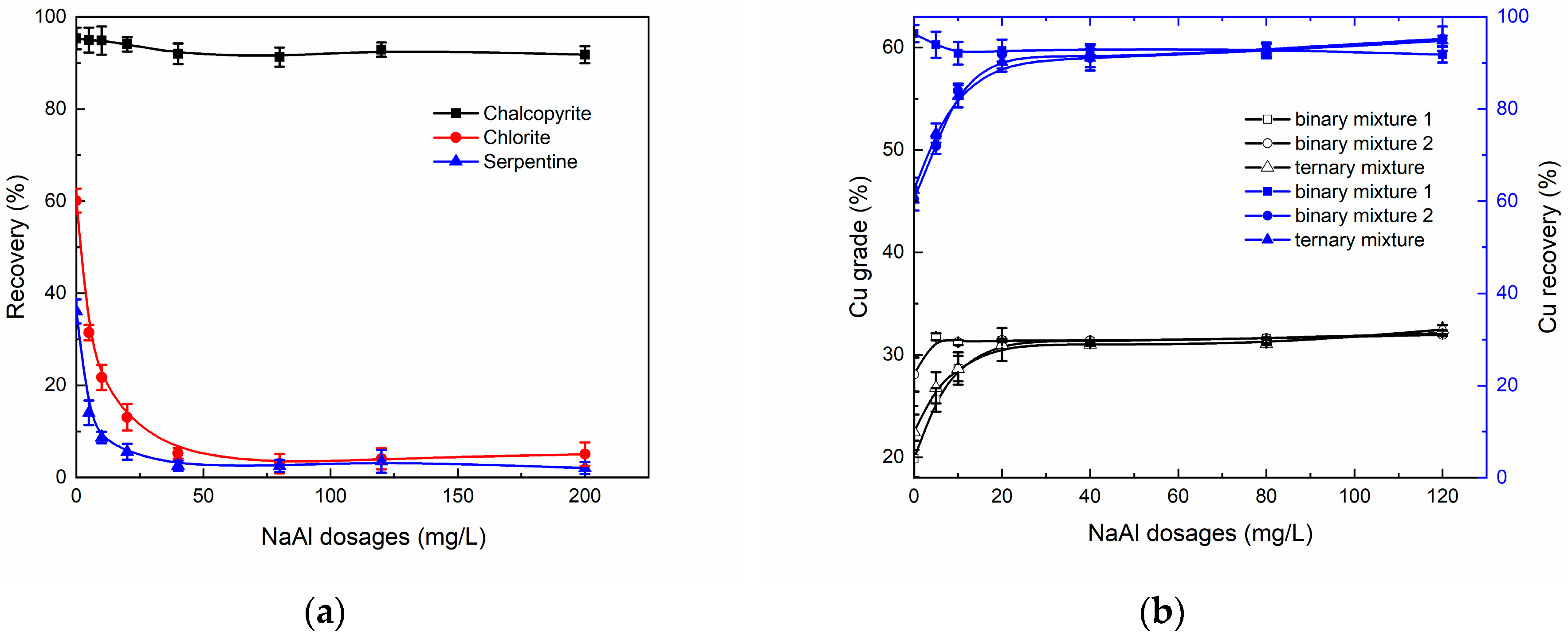
3.1. Micro-Flotation Experiments
3.2. Real Ore Flotation Experiments
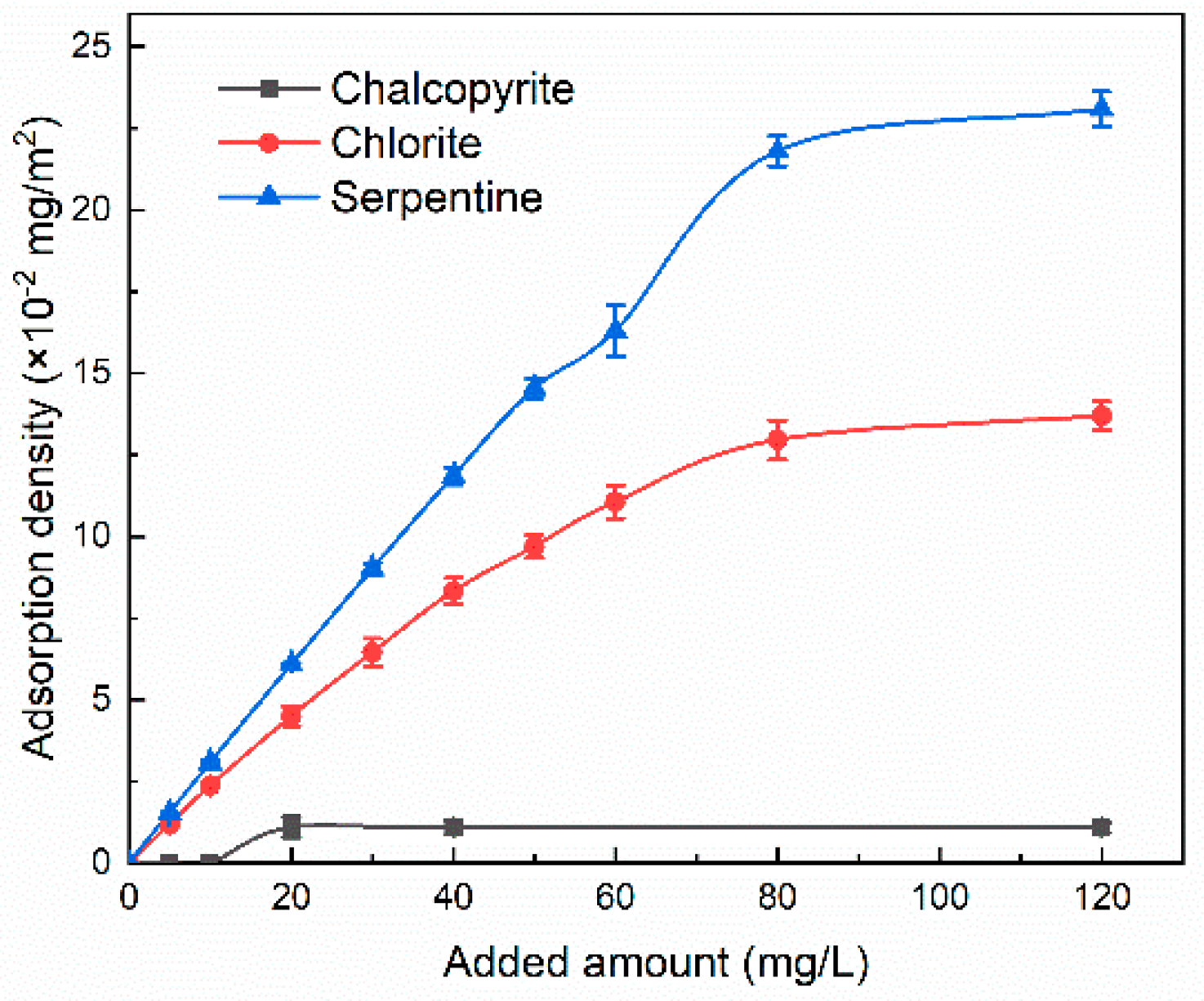
3.3. Adsorption Experiments
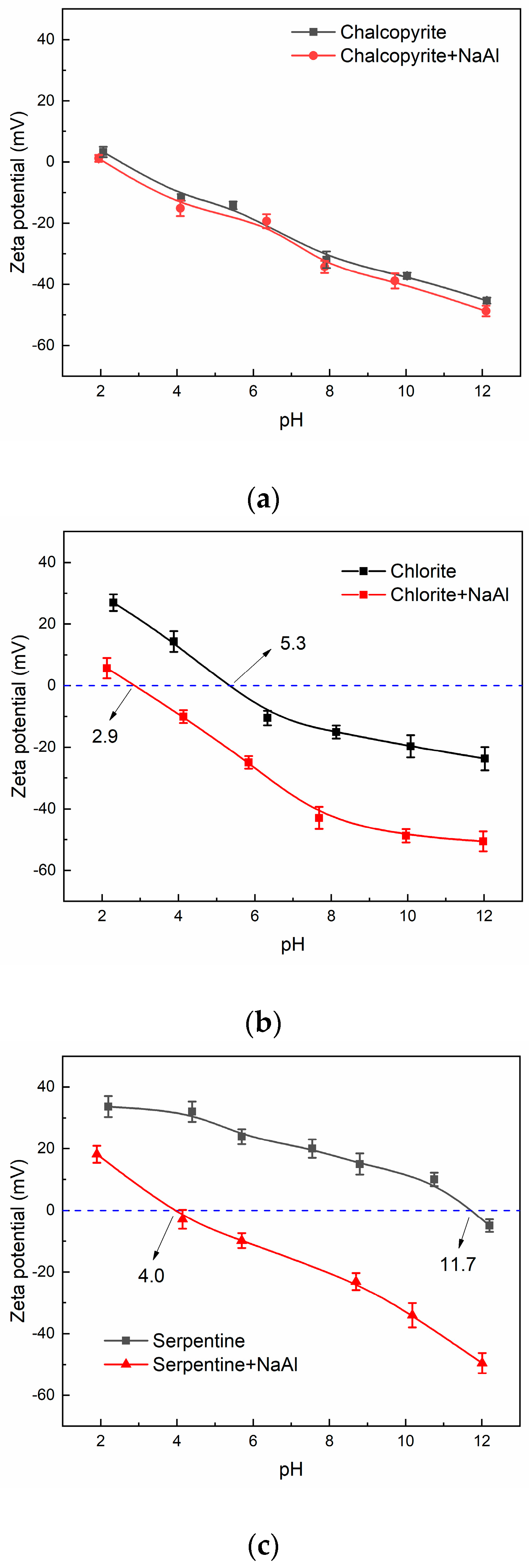
3.4. Zeta Potential Measurement Results
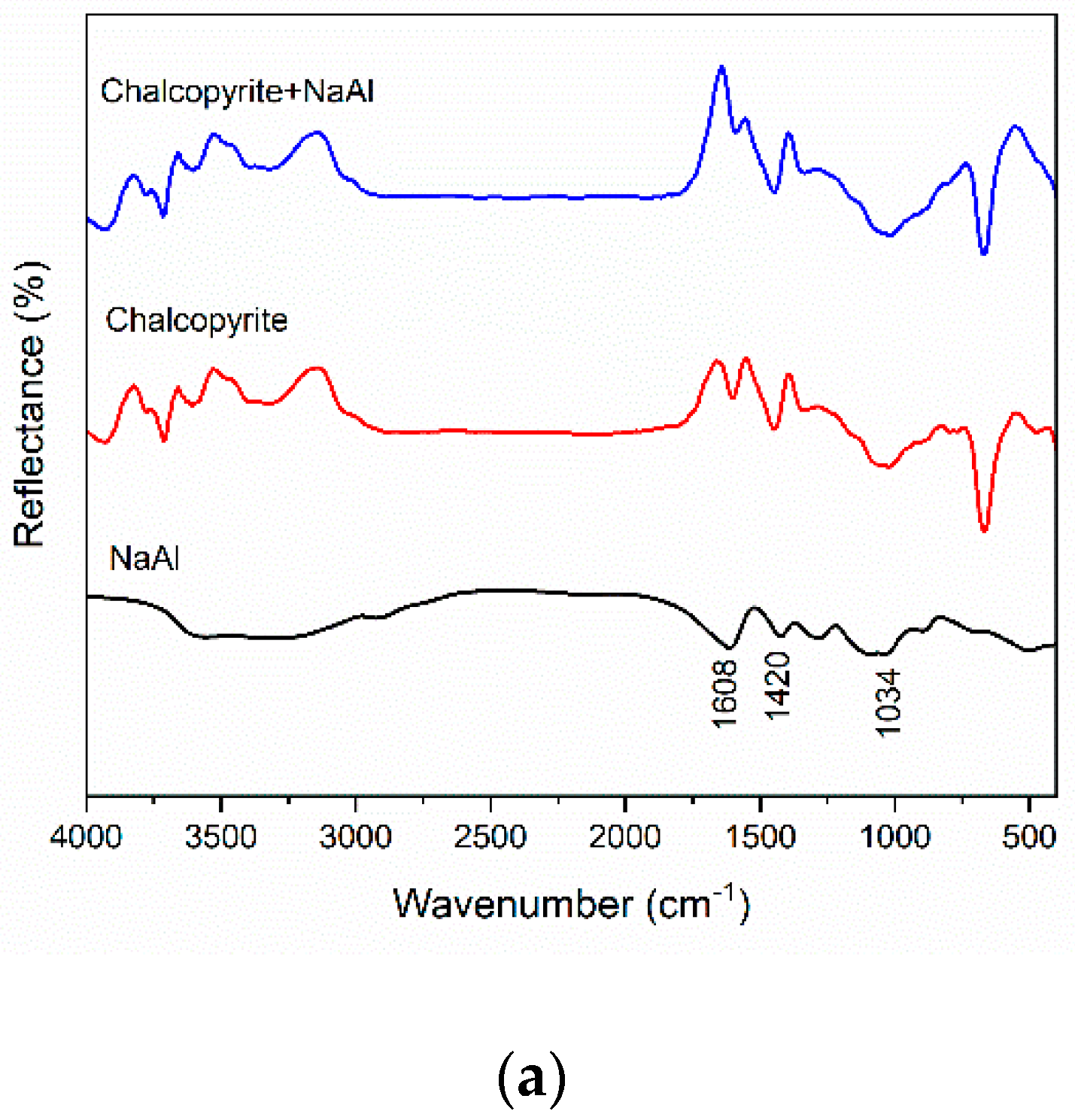
3.5. IR Spectroscopic Analysis
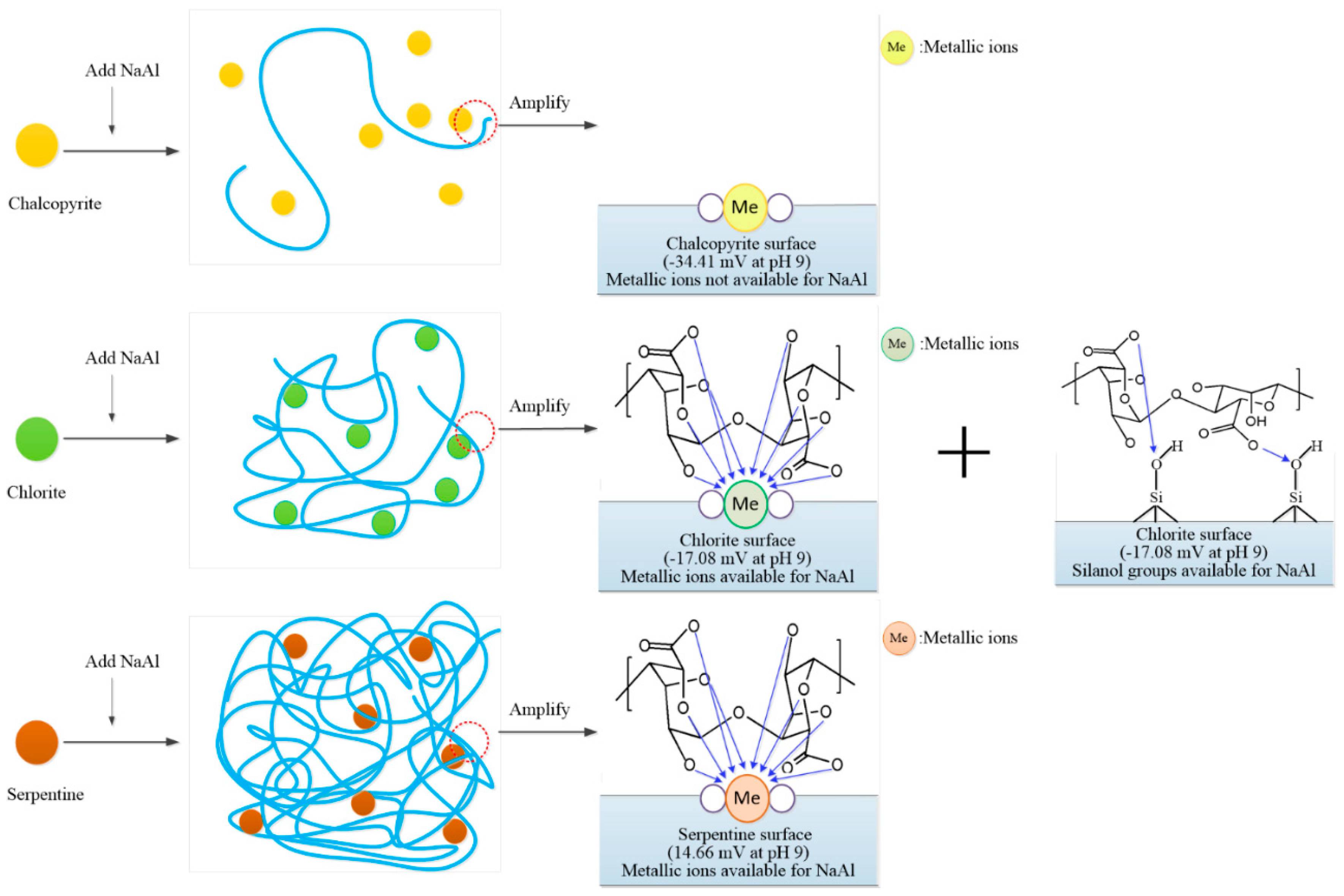
3.6. Depression Mechanism of NaAl
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Suárez, S.; Nieto, F.; Velasco, F.; Martín, F.J. Serpentine and chlorite as effective Ni-Cu sinks during weathering of the Aguablanca sulphide deposit (SW Spain). TEM evidence for metal-retention mechanisms in sheet silicates. Eur. J. Mineral. 2011, 23, 179–196. [Google Scholar] [CrossRef]
- Gur’yanov, V.A.; Prikhod’ko, V.S.; Perestoronin, A.N.; Petukhova, L.L.; Pototskii, Y.P.; Sobolev, L.P. New Type of Copper-Nickel Deposits in the Southeastern Aldan-Stanovoi Shield. Dokl. Earth Sci. 2009, 425, 363–366. [Google Scholar] [CrossRef]
- Hamer, M.; Graham, R.C.; Amrheim, C.; Bozhilov, K.N. Dissolution of ripidolite (Mg, Fe-chlorite) in organic and inorganic acid solutions. Soil Sci. Soc. Am. J. 2003, 67, 654–661. [Google Scholar] [CrossRef]
- Palandri, J.L.; Reed, M.H. Geochemical models of metasomatism in ultramafic systems: Serpentinization, rodingitization, and sea floor carbonate chimney precipitation 1. Geochim. Cosmochim. Acta 2004, 68, 1115–1133. [Google Scholar] [CrossRef]
- Austrheim, H.; Prestvik, T. Rodingitization and hydration of the oceanic lithosphere as developed in the Leka ophiolite, north–central Norway. Lithos 2008, 104, 177–198. [Google Scholar] [CrossRef]
- Zhang, W.; Zhu, G.C.; Wu, Y.Z. The enrichment mechanism of PGE and characteristic of MSZ in HW mining area in Great Dyke Zimbabwe. Adv. Mater. Res. 2013, 616, 90–95. [Google Scholar] [CrossRef]
- Parman, S.W.; Grove, T.L. Petrology and geochemistry of Barberton komatiites and basaltic komatiites: Evidence of Archean fore-arc magmatism. Dev. Precambrian Geol. 2004, 13, 539–565. [Google Scholar]
- Bobicki, E.R.; Liu, Q.; Xu, Z. Effect of microwave pre-treatment on ultramafic nickel ore slurry rheology. Miner. Eng. 2014, 61, 97–104. [Google Scholar] [CrossRef]
- Tan, H.; Skinner, W.; Addai-Mensah, J. Influence of fluorite on the isothermal leaching and rheological behaviours of chlorite mineral pulps at low pH. Int. J. Miner. Process. 2013, 123, 1–8. [Google Scholar] [CrossRef]
- Chen, X.; Peng, Y. Managing clay minerals in froth flotation—A critical review. Miner. Process. Extr. Metall. Rev. 2018, 39, 289–307. [Google Scholar] [CrossRef]
- Feng, B.; Lu, Y.; Feng, Q.; Zhang, M.; Gu, Y. Talc-serpentine interactions and implications for talc depression. Miner. Eng. 2012, 32, 68–73. [Google Scholar] [CrossRef]
- Jin, S.; Shi, Q.; Li, Q.; Ou, L.; Ouyang, K. Effect of calcium ionic concentrations on the adsorption of carboxymethyl cellulose onto talc surface: Flotation, adsorption and AFM imaging study. Powder Technol. 2018, 331, 155–161. [Google Scholar] [CrossRef]
- Silvester, E.J.; Bruckard, W.J.; Woodcock, J.T. Surface and chemical properties of chlorite in relation to its flotation and depression. Miner. Process. Extr. Metall. 2011, 120, 65–70. [Google Scholar] [CrossRef]
- Zheng, G.; Liu, L.; Liu, J.; Wang, Y.; Cao, Y. Study of chlorite flotation and its influencing factors. Procedia Earth Planet. Sci. 2009, 1, 830–837. [Google Scholar]
- Fornasiero, D.; Ralston, J. Cu(II) and Ni(II) activation in the flotation of quartz, lizardite and chlorite. Int. J. Miner. Process. 2005, 76, 75–81. [Google Scholar] [CrossRef]
- Alvarez-Silva, M.; Uribe-Salas, A.; Mirnezami, M.; Finch, J.A. The point of zero charge of phyllosilicate minerals using the Mular–Roberts titration technique. Miner. Eng. 2010, 23, 383–389. [Google Scholar] [CrossRef]
- Bo, F.; Lu, Y.; Feng, Q.; Peng, D.; Na, L.U.O. Mechanisms of surface charge development of serpentine mineral. Trans. Nonferrous Met. Soc. China 2013, 23, 1123–1128. [Google Scholar]
- Feng, B.; Feng, Q.; Lu, Y. The effect of lizardite surface characteristics on pyrite flotation. Appl. Surf. Sci. 2012, 259, 153–158. [Google Scholar] [CrossRef]
- Peng, Y.; Bradshaw, D. Mechanisms for the improved flotation of ultrafine pentlandite and its separation from lizardite in saline water. Miner. Eng. 2012, 36, 284–290. [Google Scholar] [CrossRef]
- Feng, B.; Feng, Q.; Lu, Y. A novel method to limit the detrimental effect of serpentine on the flotation of pentlandite. Int. J. Miner. Process. 2012, 114, 11–13. [Google Scholar] [CrossRef]
- Jin, S.; Shi, Q.; Feng, Q.; Zhang, G.; Chang, Z. The role of calcium and carbonate ions in the separation of pyrite and talc. Miner. Eng. 2018, 119, 205–211. [Google Scholar] [CrossRef]
- Feng, B.; Peng, J.; Guo, W.; Zhang, W.; Ai, G.; Wang, H. The effect of changes in pH on the depression of talc by chitosan and the associated mechanisms. Powder Technol. 2018, 325, 58–63. [Google Scholar] [CrossRef]
- Zhao, K.; Gu, G.; Wang, C.; Rao, X.; Wang, X.; Xiong, X. The effect of a new polysaccharide on the depression of talc and the flotation of a nickel–copper sulfide ore. Miner. Eng. 2015, 77, 99–106. [Google Scholar] [CrossRef]
- Liu, G.; Feng, Q.; Ou, L.; Lu, Y.; Zhang, G. Adsorption of polysaccharide onto talc. Miner. Eng. 2006, 19, 147–153. [Google Scholar] [CrossRef]
- Ma, X.; Pawlik, M. The effect of lignosulfonates on the floatability of talc. Int. J. Miner. Process. 2007, 83, 19–27. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Daemi, H.; Barikani, M. Synthesis and characterization of calcium alginate nanoparticles, sodium homopolymannuronate salt and its calcium nanoparticles. Sci. Iran. 2012, 19, 2023–2028. [Google Scholar] [CrossRef]
- Pawar, S.N.; Edgar, K.J. Alginate derivatization: A review of chemistry, properties and applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- Thakur, S.; Sharma, B.; Verma, A.; Chaudhary, J.; Tamulevicius, S.; Thakur, V.K. Recent progress in sodium alginate based sustainable hydrogels for environmental applications. J. Clean. Prod. 2018, 198, 143–159. [Google Scholar] [CrossRef]
- Gombotz, W.R.; Wee, S.F. Protein release from alginate matrices. Adv. Drug Deliv. Rev. 2012, 64, 194–205. [Google Scholar] [CrossRef]
- Topuz, F.; Henke, A.; Richtering, W.; Groll, J. Magnesium ions and alginate do form hydrogels: A rheological study. Soft Matter 2012, 8, 4877–4881. [Google Scholar] [CrossRef]
- Jana, S.; Gandhi, A.; Sheet, S.; Sen, K.K. Metal ion-induced alginate–locust bean gum IPN microspheres for sustained oral delivery of aceclofenac. Int. J. Biol. Macromol. 2015, 72, 47–53. [Google Scholar] [CrossRef]
- Tønnesen, H.H.; Karlsen, J. Alginate in drug delivery systems. Drug Dev. Ind. Pharm. 2002, 28, 621–630. [Google Scholar] [CrossRef]
- Pongjanyakul, T.; Priprem, A.; Puttipipatkhachorn, S. Investigation of novel alginate-magnesium aluminum silicate microcomposite films for modified-release tablets. J. Control. Release 2005, 107, 343–356. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Feng, Q.; Zhang, G.; Yang, Q.; Zhang, C. The effect of sodium alginate on the flotation separation of scheelite from calcite and fluorite. Miner. Eng. 2017, 113, 1–7. [Google Scholar] [CrossRef]
- Chen, Z.; Gu, G.; Li, S.; Wang, C.; Zhu, R. The Effect of Seaweed Glue in the Separation of Copper–Molybdenum Sulphide Ore by Flotation. Minerals 2018, 8, 41. [Google Scholar] [CrossRef]
- Wellham, E.J.; Elber, L.; Yan, D.S. The role of carboxy methyl cellulose in the flotation of a nickel sulphide transition ore. Miner. Eng. 1992, 5, 381–395. [Google Scholar] [CrossRef]
- Duarte, A.C.P.; Grano, S.R. Mechanism for the recovery of silicate gangue minerals in the flotation of ultrafine sphalerite. Miner. Eng. 2007, 20, 766–775. [Google Scholar] [CrossRef]
- Miller, J.D.; Li, J.; Davidtz, J.C.; Vos, F. A review of pyrrhotite flotation chemistry in the processing of PGM ores. Miner. Eng. 2005, 18, 855–865. [Google Scholar] [CrossRef]
- Zouboulis, A.I.; Kydros, K.A.; Matis, K.A. Adsorbing flotation of copper hydroxo precipitates by pyrite fines. Sep. Sci. Technol. 1992, 27, 2143–2155. [Google Scholar] [CrossRef]
- Bellamy, L.J. The infrared spectra of complex molecules. In The Infrared Spectra of Complex Molecules; Springer Science & Business Media: Berlin, Germany, 2013; pp. 183–202. ISBN 0412138506. [Google Scholar]
- Rath, R.K.; Subramanian, S.; Pradeep, T. Surface chemical studies on pyrite in the presence of polysaccharide-based flotation depressants. J. Colloid Interface Sci. 2000, 229, 82–91. [Google Scholar] [CrossRef]
- Puttipipatkhachorn, S.; Pongjanyakul, T.; Priprem, A. Molecular interaction in alginate beads reinforced with sodium starch glycolate or magnesium aluminum silicate, and their physical characteristics. Int. J. Pharm. 2005, 293, 51–62. [Google Scholar] [CrossRef]
- Sondi, I.; Milat, O.; Pravdić, V. Electrokinetic potentials of clay surfaces modified by polymers. J. Colloid Interface Sci. 1997, 189, 66–73. [Google Scholar] [CrossRef]
- Sondi, I.; Pravdić, V. Electrokinetics of natural and mechanically modified ripidolite and beidellite clays. J. Colloid Interface Sci. 1996, 181, 463–469. [Google Scholar] [CrossRef]
- Sondi, I.; Pravdic, V. The colloid and surface chemistry of clays in natural waters. Croat. Chem. Acta 1998, 71, 1061–1074. [Google Scholar]
- Zaikowski, A. Infrared spectra of the Orgueil (C-1) chondrite and serpentine minerals. Geochim. Cosmochim. Acta 1979, 43, 943–945. [Google Scholar] [CrossRef]
- Heller-Kallai, L.; Yariv, S.; Gross, S. Hydroxyl-stretching frequencies of serpentine minerals. Mineral. Mag. 1975, 40, 197–200. [Google Scholar] [CrossRef]
- Andersen, T.; Auk-Emblem, P.; Dornish, M. 3D Cell Culture in Alginate Hydrogels. Microarrays 2015, 4, 133–161. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.M.; Deng, J.S.; Xian, Y.J.; Dan, L. Theory analysis and vestigial information of surface relaxation of natural chalcopyrite mineral crystal. Trans. Nonferrous Met. Soc. China 2013, 23, 796–803. [Google Scholar] [CrossRef]
- Li, X.; Wang, L.; Lu, A. A discussion on activation mechanism of atom groups in serpentine. ACTA Petrol. Mineral. 2003, 22, 386–390. [Google Scholar]
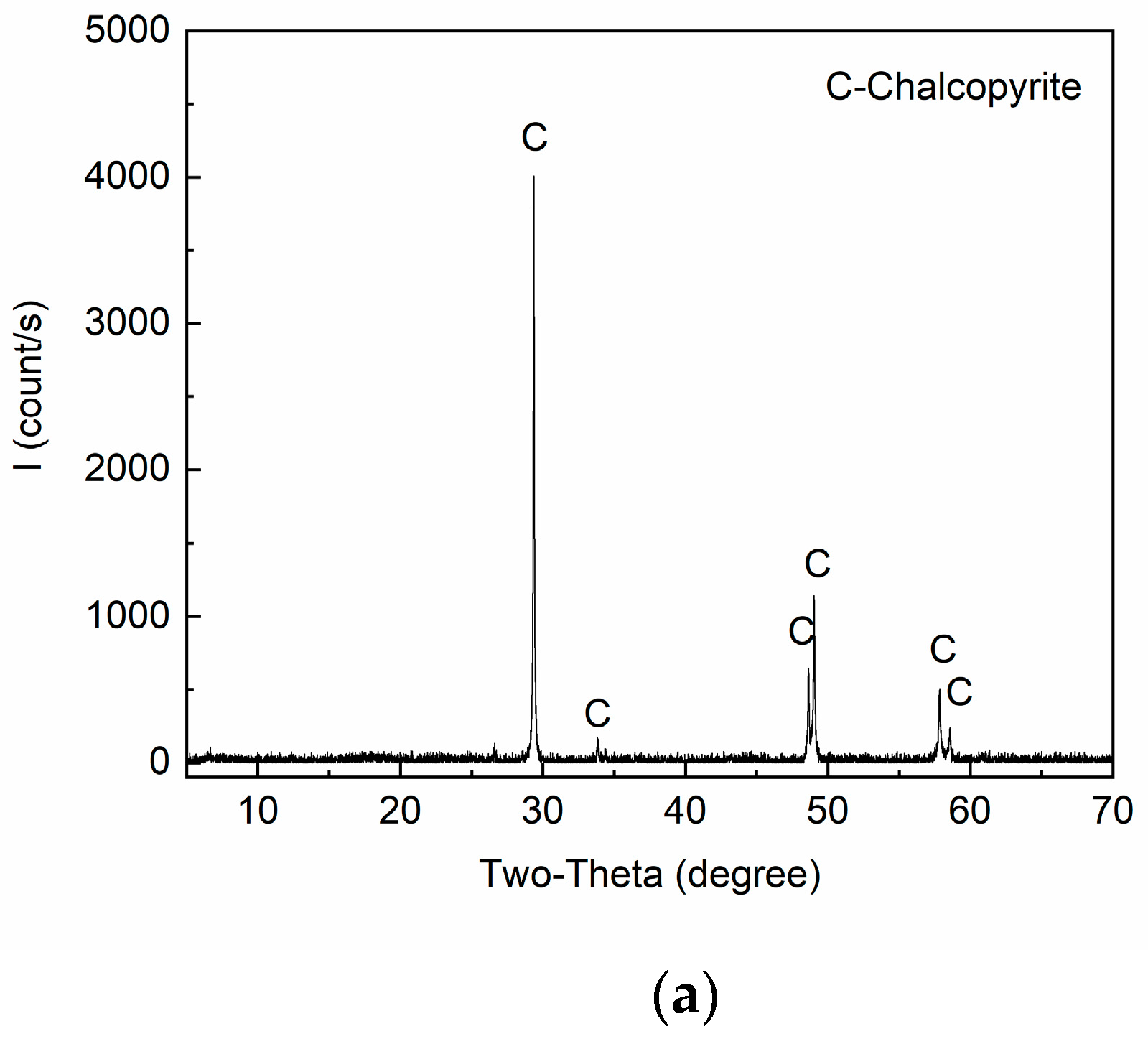
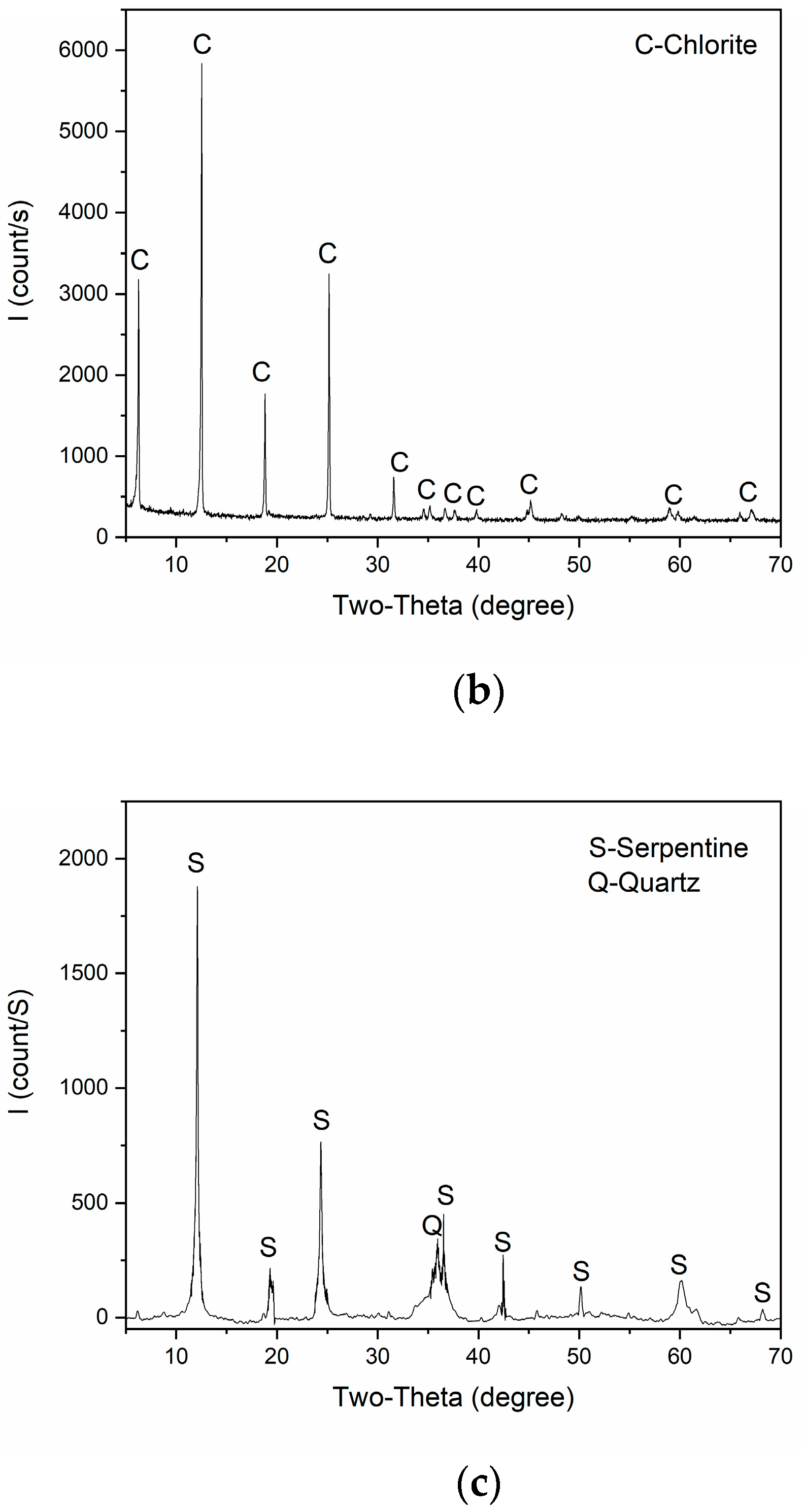

| Elements | Cu | Fe | S | MgO | SiO2 | Al2O3 | CaO | Others |
|---|---|---|---|---|---|---|---|---|
| Chalcopyrite | 33.88 | 29.45 | 33.86 | / | 2.81 | / | / | / |
| Chlorite | / | 31.69 | / | 15.41 | 41.10 | 11.32 | 0.28 | 0.20 |
| Serpentine | / | 1.08 | / | 38.92 | 46.13 | 1.16 | 0.61 | 12.10 |
| Conditions | Product | Yield (%) | Cu Grade (%) | Cu Recovery (%) |
|---|---|---|---|---|
| NaAl 0 g/t | Concentrate | 33.5 | 2.6 | 65.5 |
| Tailing | 66.5 | 0.7 | 34.5 | |
| Feed | 100.0 | 1.3 | 100.0 | |
| NaAl 200 g/t | Concentrate | 30.2 | 3.2 | 73.2 |
| Tailing | 69.8 | 0.5 | 26.8 | |
| Feed | 100.0 | 1.3 | 100.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, G.; Zhang, G.; Shi, Q.; Chen, W. The Effect of Sodium Alginate on Chlorite and Serpentine in Chalcopyrite Flotation. Minerals 2019, 9, 196. https://doi.org/10.3390/min9030196
Pan G, Zhang G, Shi Q, Chen W. The Effect of Sodium Alginate on Chlorite and Serpentine in Chalcopyrite Flotation. Minerals. 2019; 9(3):196. https://doi.org/10.3390/min9030196
Chicago/Turabian StylePan, Guangjiu, Guofan Zhang, Qing Shi, and Wei Chen. 2019. "The Effect of Sodium Alginate on Chlorite and Serpentine in Chalcopyrite Flotation" Minerals 9, no. 3: 196. https://doi.org/10.3390/min9030196
APA StylePan, G., Zhang, G., Shi, Q., & Chen, W. (2019). The Effect of Sodium Alginate on Chlorite and Serpentine in Chalcopyrite Flotation. Minerals, 9(3), 196. https://doi.org/10.3390/min9030196




