Biomineralization Forming Process and Bio-inspired Nanomaterials for Biomedical Application: A Review
Abstract
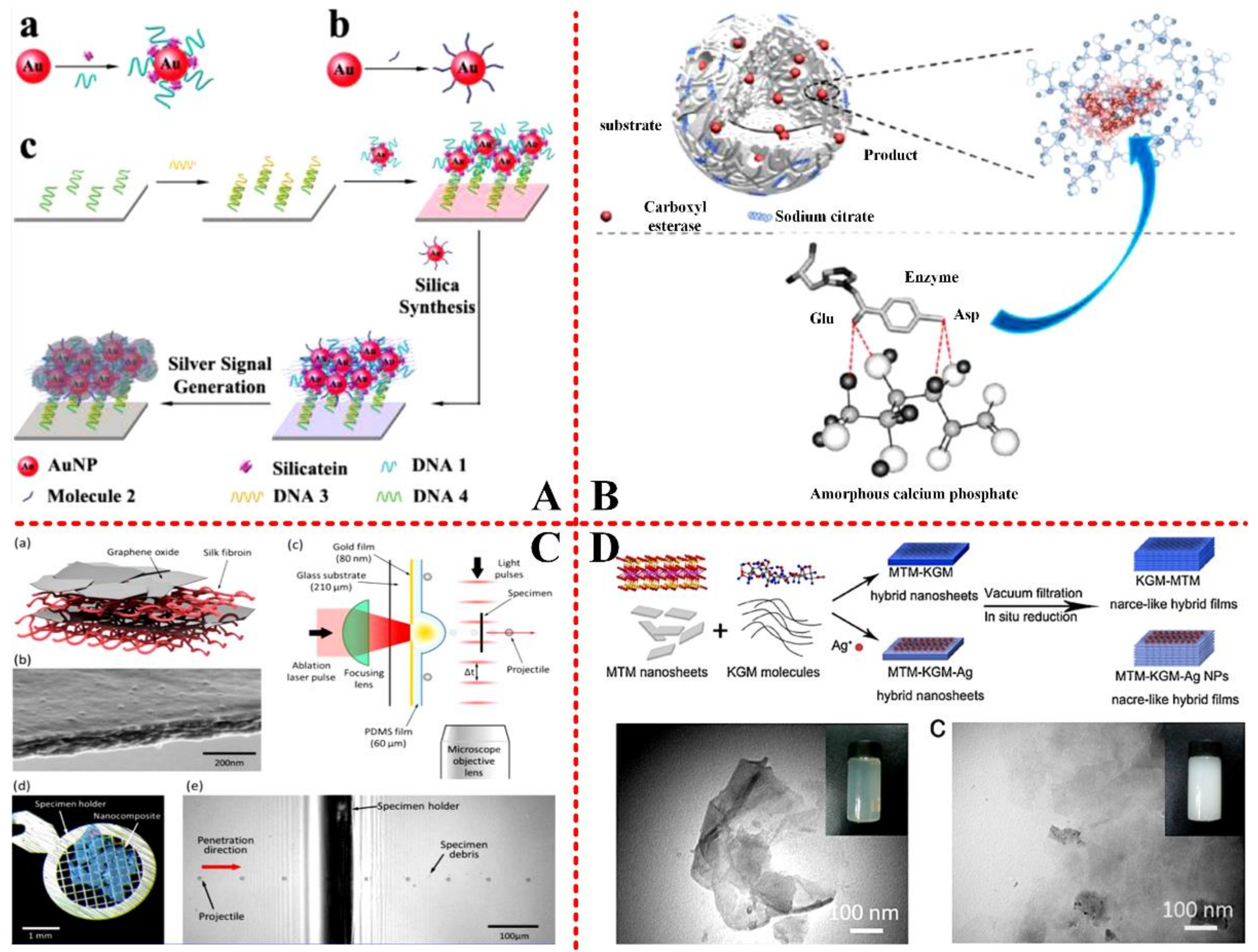
1. Introduction
2. Biomineralization Forming Process
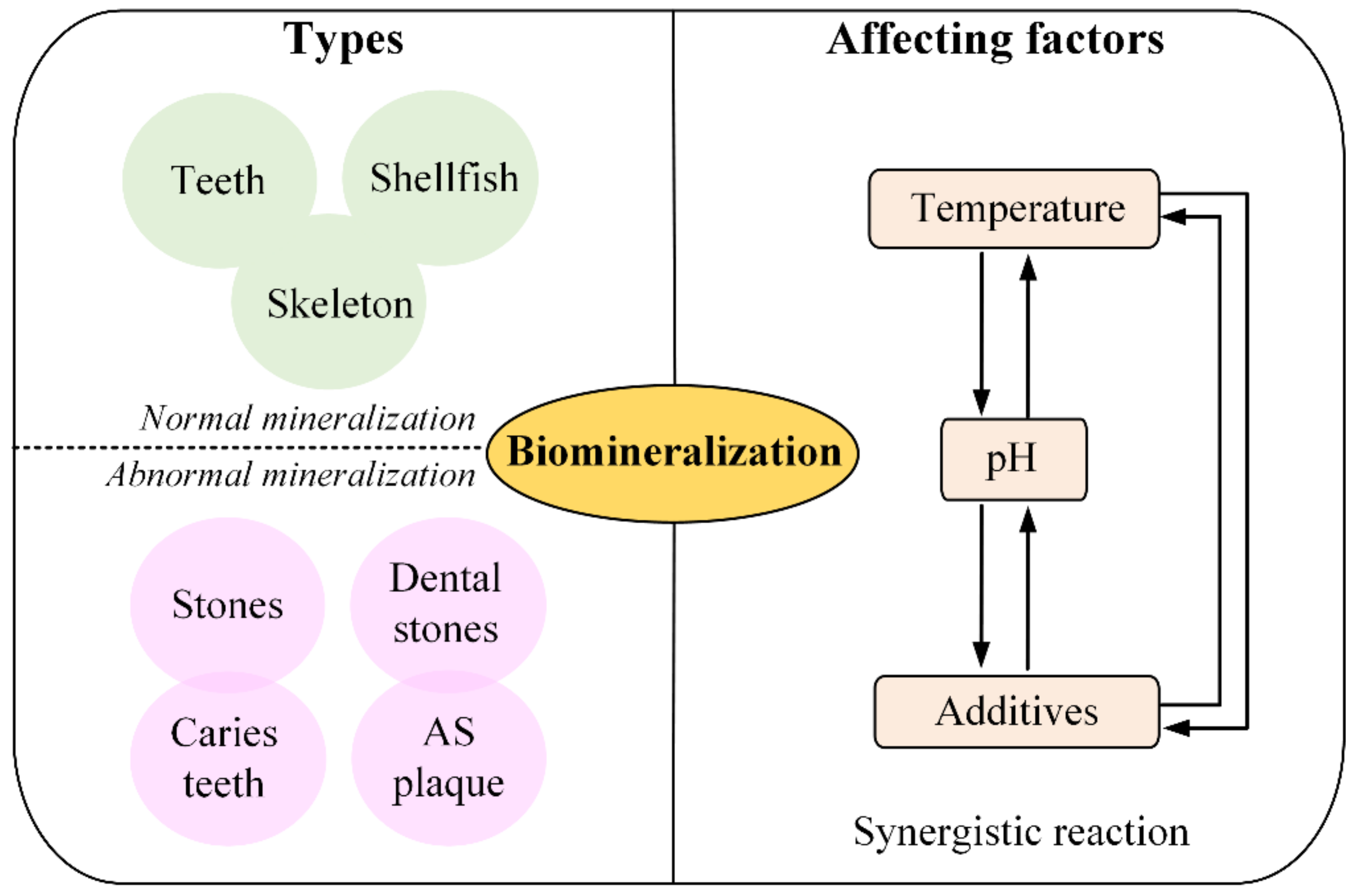
2.1. The Type of Mineralization
2.2. Factors Controlling Biomineralization
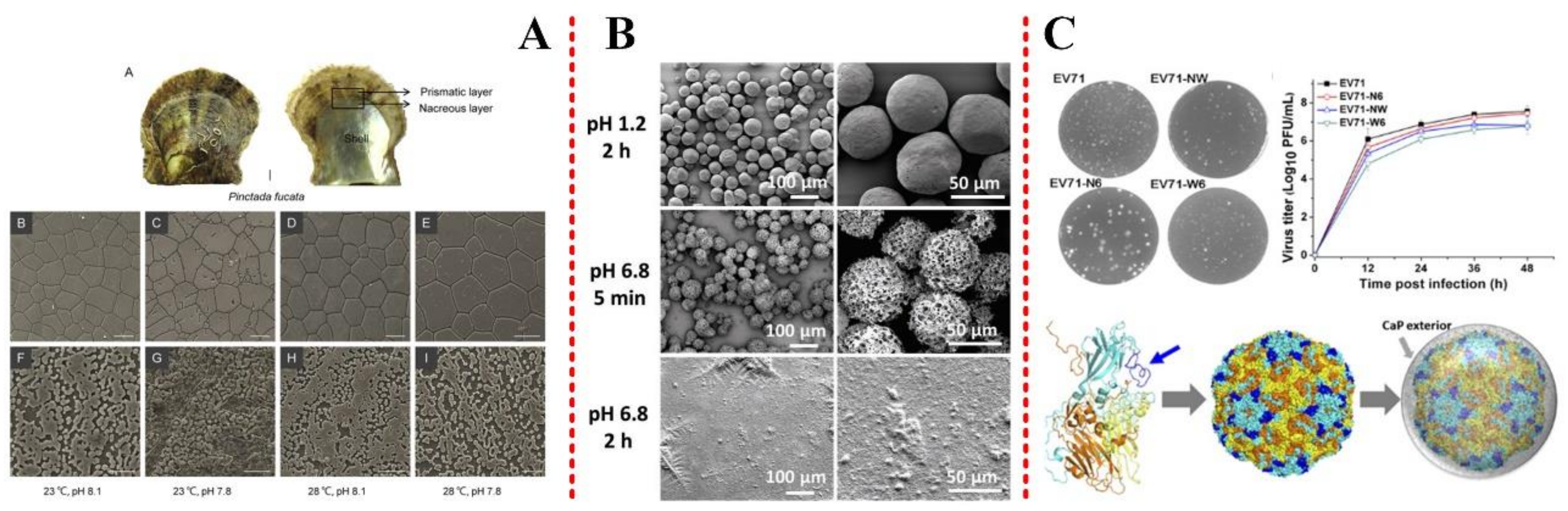
2.2.1. Temperature and pH
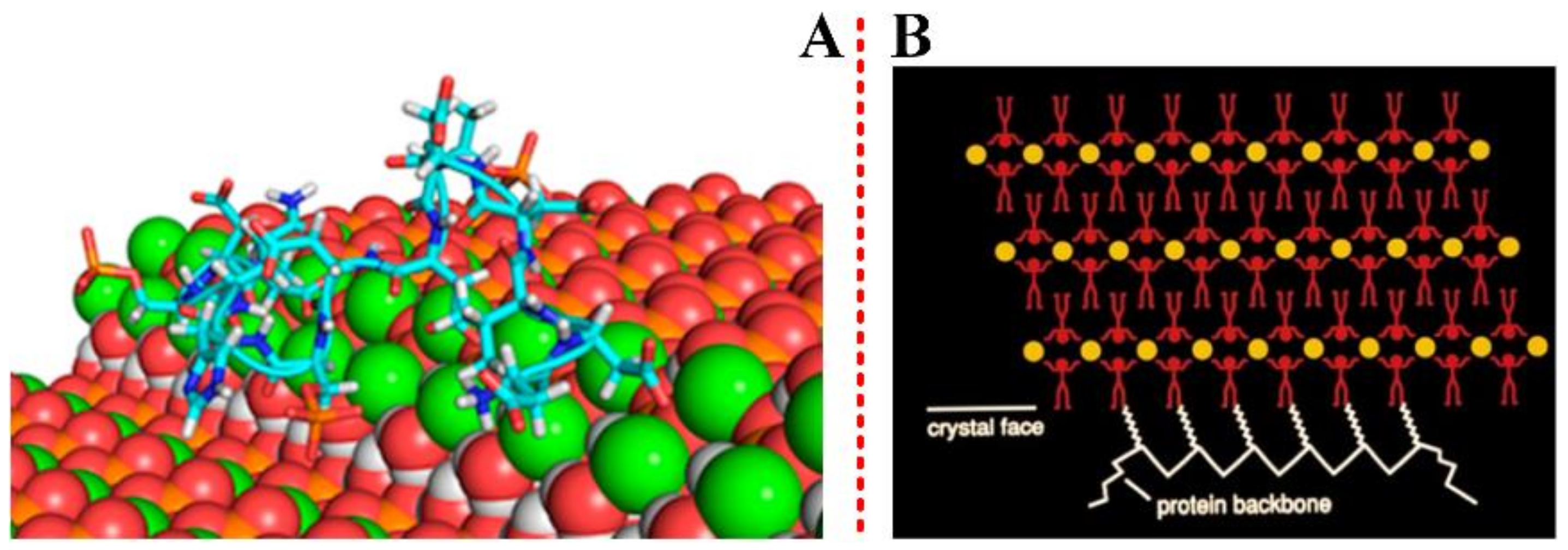
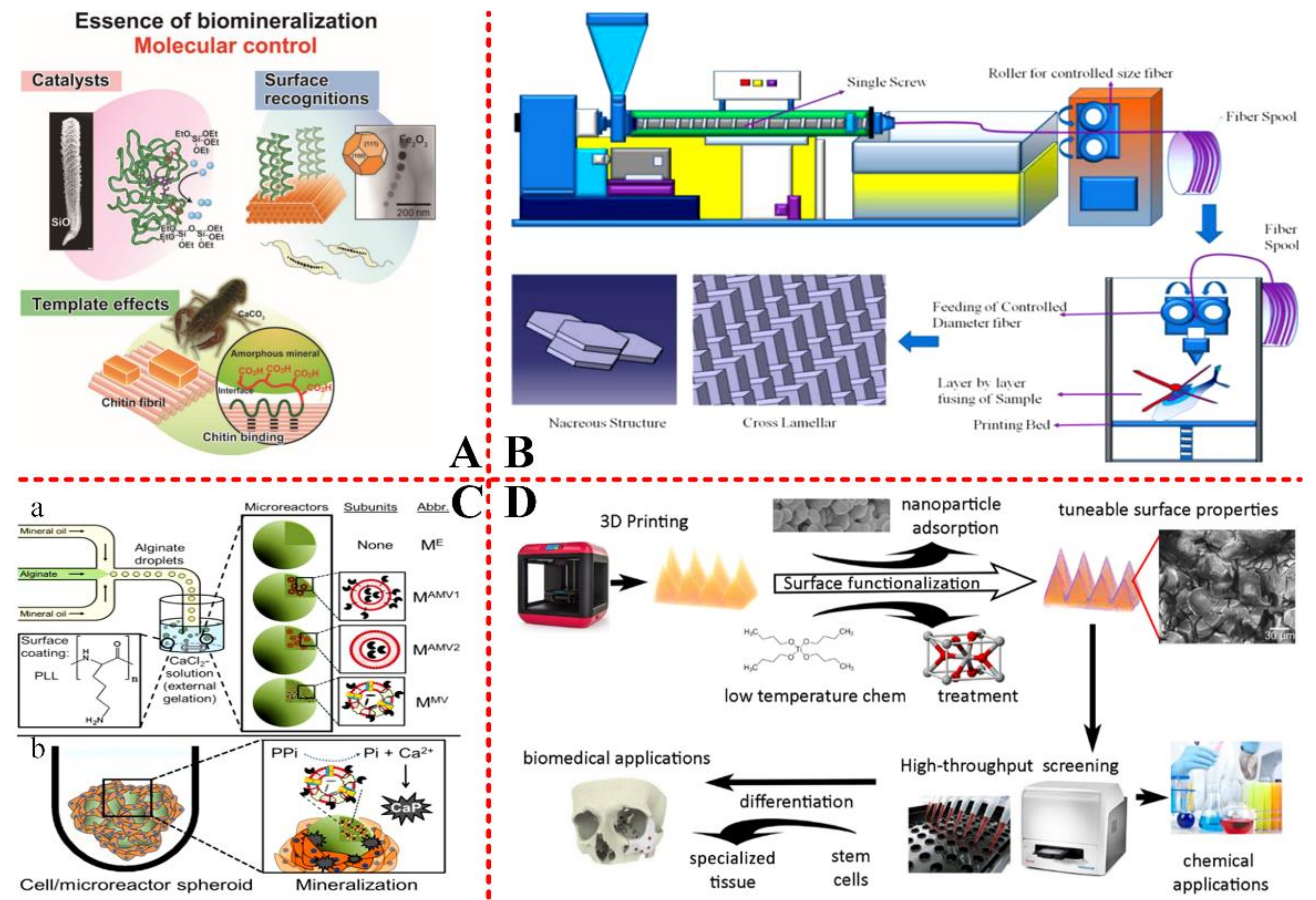
2.2.2. Guidance of Organic Matrix on Biominerals
2.2.3. Additives
2.3. Mechanism and Approach of Mineralization
2.4. Biomimetic Biomineralization
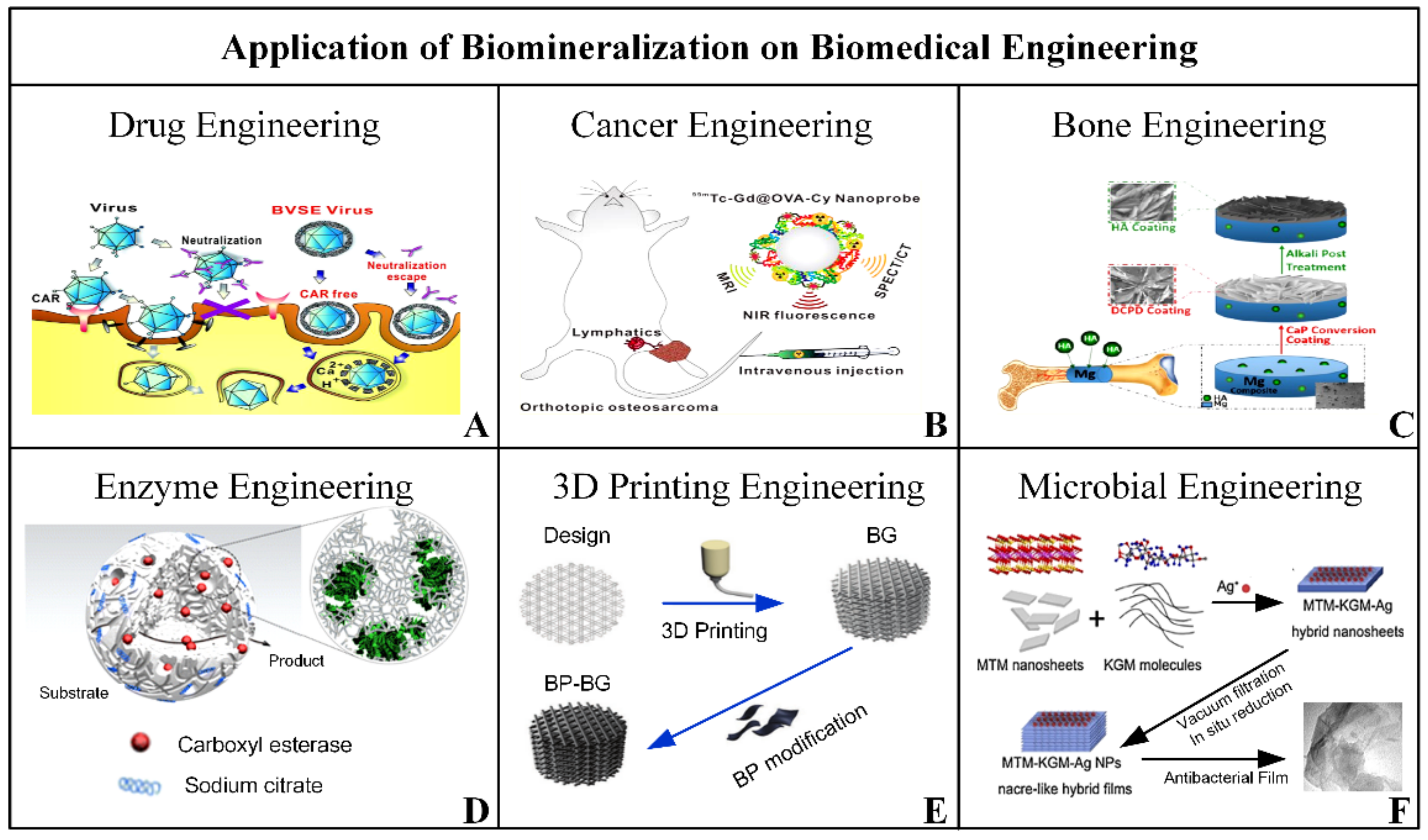
3. Application of Biomineralization in Biomedical Engineering
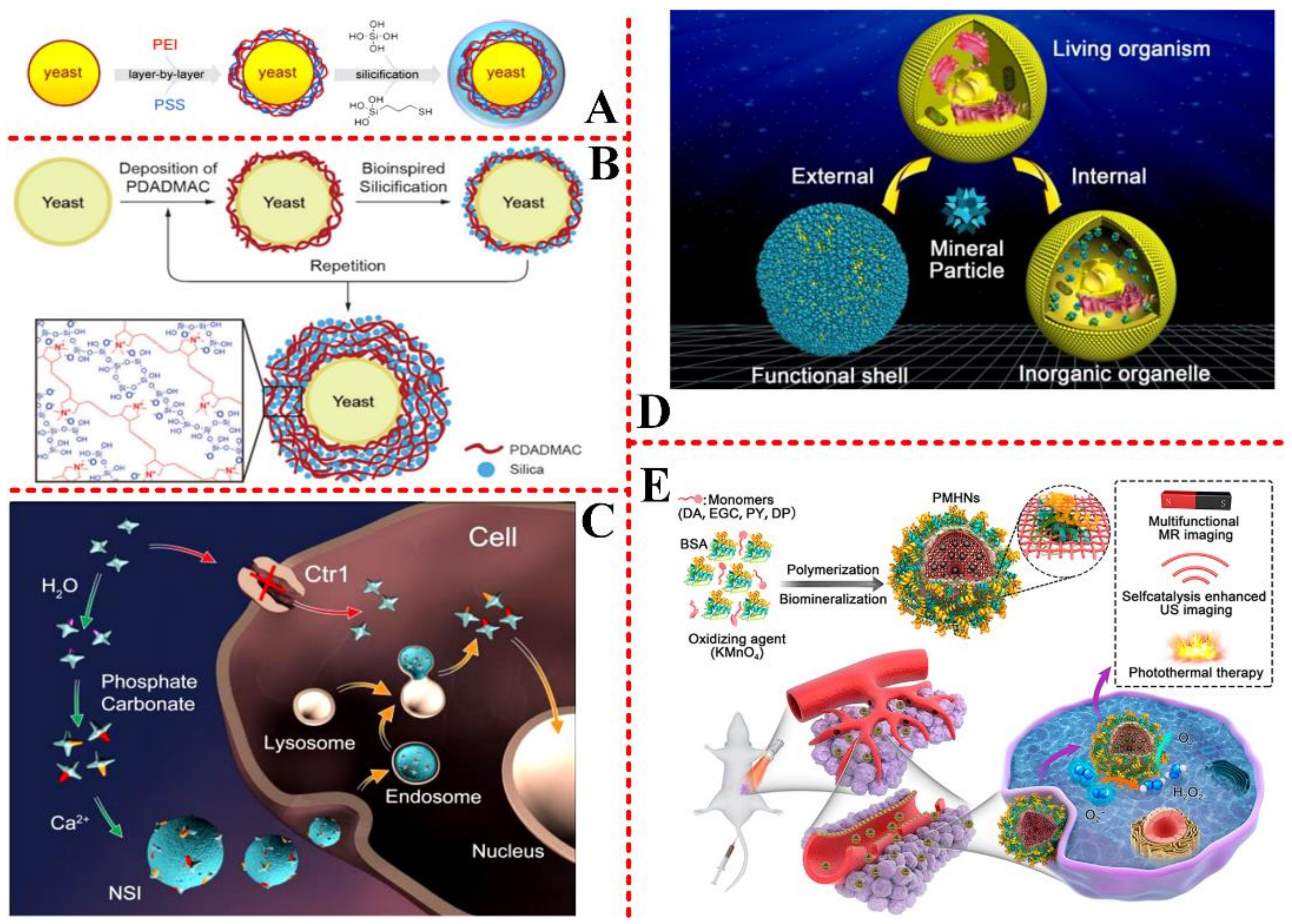
3.1. Drug and Cell-Therapy Engineering
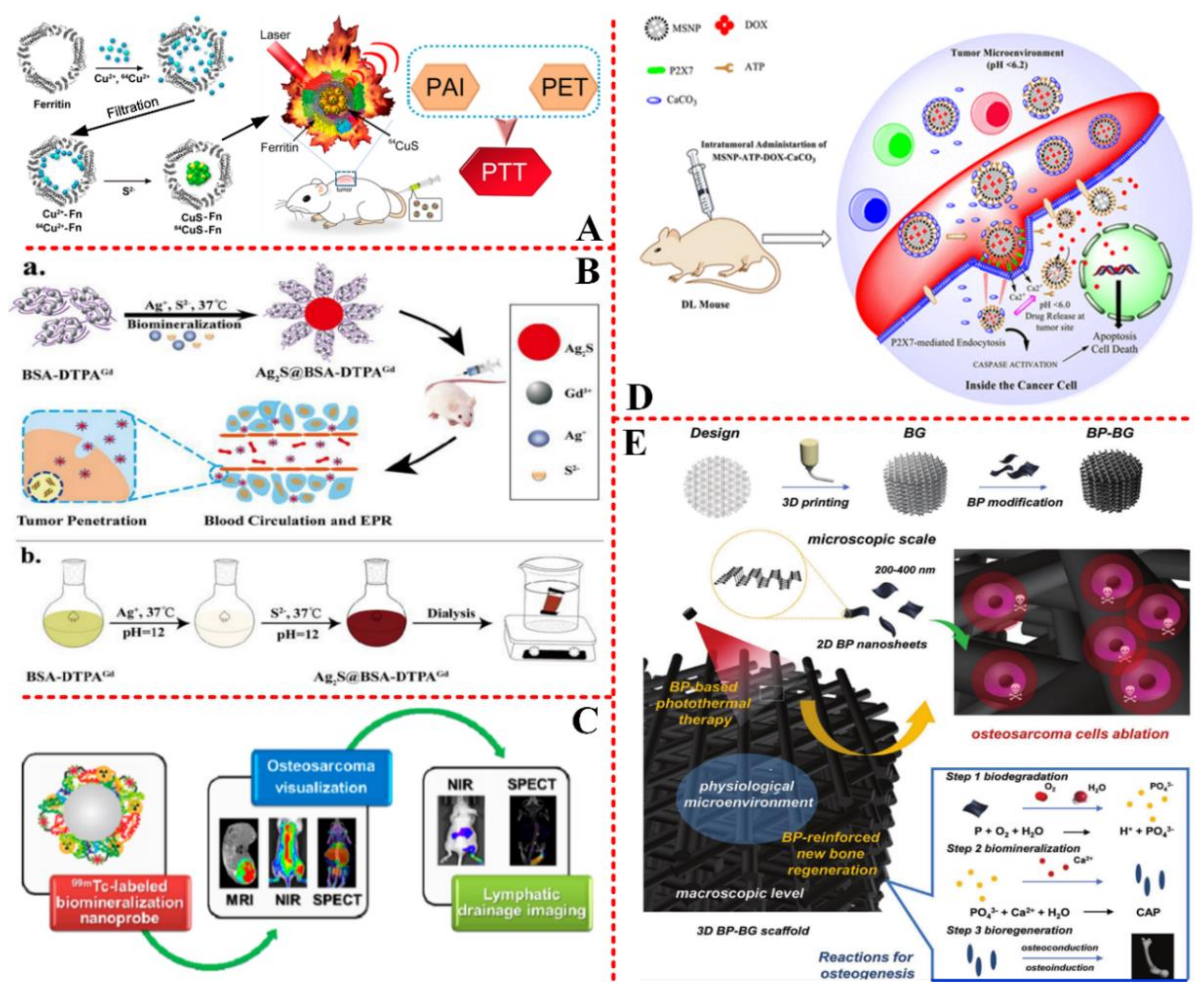
3.2. Cancer/Tumor Target Engineering
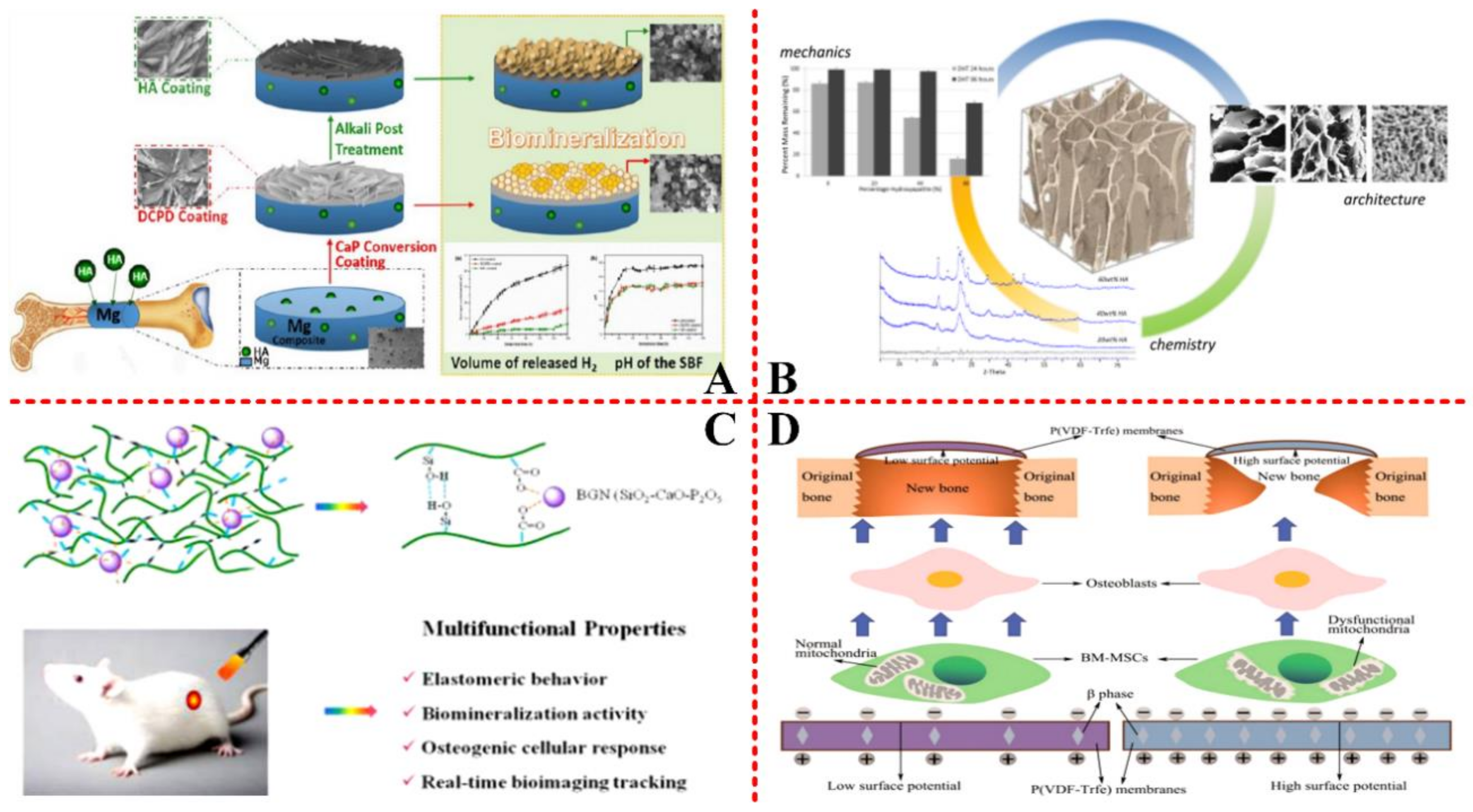
3.3. Bone Tissue Engineering
3.4. Bionic and 3D Printing Engineering
3.5. Other Biomedical Applications
4. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Estroff, L.A. Introduction: Biomineralization. Chem. Rev. 2008, 108, 4329–4331. [Google Scholar] [CrossRef] [PubMed]
- Gal, A.; Weiner, S.; Addadi, L. A perspective on underlying crystal growth mechanisms in biomineralization: Solution mediated growth versus nanosphere particle accretion. CrystEngComm 2015, 17, 2606–2615. [Google Scholar] [CrossRef]
- Addadi, L.; Steve, W. Biomineralization: Mineral formation by organisms. Phys. Scr. 2014, 89, 098003. [Google Scholar] [CrossRef]
- Arakaki, A.; Shimizu, K.; Oda, M.; Sakamoto, T.; Nishimura, T.; Kato, T. Biomineralization-inspired synthesis of functional organic/inorganic hybrid materials: Organic molecular control of self-organization of hybrids. Org. Biomol. Chem. 2015, 13, 974–989. [Google Scholar] [CrossRef] [PubMed]
- DeVol, R.T.; Sun, C.Y.; Marcus, M.A.; Coppersmith, S.N.; Myneni, S.C.; Gilbert, P.U. Nanoscale Transforming Mineral Phases in Fresh Nacre. J. Am. Chem. Soc. 2015, 137, 13325–13333. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Huerta, A.; Dauphin, Y.; Cusack, M. Biogenic calcite granules—Are brachiopods different? Micron 2013, 44, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, S.; Kajiyama, S.; Nishimura, T.; Kato, T. Formation of Helically Structured Chitin/CaCO3 Hybrids through an Approach Inspired by the Biomineralization Processes of Crustacean Cuticles. Small 2015, 11, 5127–5133. [Google Scholar] [CrossRef]
- Kokubo, T. Design of bioactive bone substitutes based on biomineralization process. Mater. Sci. Eng. C 2005, 25, 97–104. [Google Scholar] [CrossRef]
- Sone, E.D.; Weiner, S.; Addadi, L. Biomineralization of limpet teeth: A cryo-TEM study of the organic matrix and the onset of mineral deposition. J. Struct. Biol. 2007, 158, 428–444. [Google Scholar] [CrossRef]
- Märkel, K.; Röser, U.; Mackenstedt, U.; Klostermann, M. Ultrastructural investigation of matrix-mediated biomineralization in echinoids (Echinodermata, Echinoida). Zoomorphology 1986, 106, 232–243. [Google Scholar] [CrossRef]
- Cuif, J.P.; Dauphin, Y. The two-step mode of growth in the Scleractinian coral skeletons from the micrometre to the overall scale. J. Struct. Biol. 2005, 150, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Ingalls, A.E.; Lee, C.; Druffel, E.R. Preservation of organic matter in mound-forming coral skeletons. Geochim. Cosmochim. Acta 2003, 67, 2827–2841. [Google Scholar] [CrossRef]
- Kopp, C.; Meibom, A.; Beyssac, O.; Stolarski, J.; Djediat, S.; Szlachetko, J.; Domart-Coulon, I. Calcareous sponge biomineralization: Ultrastructural and compositional heterogeneity of spicules in Leuconia johnstoni. J. Struct. Biol. 2011, 173, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.G. Skeletal Biomineralization; Short Courses in Geology; American Geophysical Union: Washington, DC, USA, 1989; Volume 5. [Google Scholar]
- Hodge, A.J. Recent studies with the electron microscope on ordered aggregates of the tropocollagen macromolecule. In Aspects of Protein Structure; Ramachandran, G.N., Ed.; Academic Press: New York, NY, USA, 1963; pp. 289–300. [Google Scholar]
- Glimcher, M.J.; Hodge, A.J.; Schmitt, F.O. Macromolecular aggregation states in relation to mineralization: The collagen-hydroxyapatlte system as studied in vitro. PNAS 1957, 43, 860–867. [Google Scholar] [CrossRef]
- Xu, Z.; Yang, Y.; Zhao, W.; Wang, Z.; Landis, W.J.; Cui, Q.; Sahai, N. Molecular mechanisms for intrafibrillar collagen mineralization in skeletal tissues. Biomaterials 2015, 39, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Olszta, M.J.; Cheng, X.G.; Jee, S.S.; Kumar, R.; Kim, Y.Y.; Kaufman, M.J.; Douglas, E.P.; Gower, L.B. Bone structure and formation: A new perspective. Mater. Sci. Eng. R Rep. 2007, 58, 77–116. [Google Scholar] [CrossRef]
- Yu, J.G.; Lei, M.; Cheng, B.; Zhao, X.J. Effects of PAA additive and temperature on morphology of calcium carbonate particles. J. Solid State Chem. 2004, 177, 681–689. [Google Scholar] [CrossRef]
- Ma, Y.F.; Gao, Y.H.; Feng, Q.L. Effects of pH and temperature on CaCO3 crystallization in aqueous solution with water soluble matrix of pearls. J. Cryst. Growth 2010, 312, 3165–3170. [Google Scholar] [CrossRef]
- Chen, A.L.; Ma, P.Y.; Fu, Z.Y.; Wu, Y.; Kong, W. Crystallization and assembling behavior of calcium carbonate controlled by Ca-organic fibers. J. Cryst. Growth 2013, 377, 136–142. [Google Scholar] [CrossRef]
- Clark, M.S.; Thorne, M.A.; Amaral, A.; Vieira, F.; Batista, F.M.; Reis, J.; Power, D.M. Identification of molecular and physiological responses to chronic environmental challenge in an invasive species: The Pacific oyster, Crassostrea gigas. Ecol. Evol. 2013, 3, 3283–3297. [Google Scholar] [CrossRef] [PubMed]
- Matoo, O.B.; Ivanina, A.V.; Ullstad, C.; Beniash, E.; Sokolova, I.M. Interactive effects of elevated temperature and CO2 levels on metabolism and oxidative stress in two common marine bivalves (Crassostrea virginica and Mercenaria mercenaria). Comp. Biochem. Phys. A 2013, 164, 545–553. [Google Scholar] [CrossRef]
- Dickinson, G.H.; Ivanina, A.V.; Matoo, O.B.; Portner, H.O.; Lannig, G.; Bock, C.; Beniash, E.; Sokolova, I.M. Interactive effects of salinity and elevated CO2 levels on juvenile eastern oysters, Crassostrea virginica. J. Exp. Biol. 2012, 215, 29–43. [Google Scholar] [CrossRef]
- Zheng, X.Y.; Shen, Y.H.; Wang, X.Y.; Wang, T.S. Effect of pH on uranium(VI) biosorption and biomineralization by Saccharomyces cerevisiae. Chemosphere 2018, 203, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Huang, J.; Liu, C.; Liu, Y.; Zheng, G.; Xie, L.; Zhang, R. Interactive Effects of Seawater Acidification and Elevated Temperature on the Transcriptome and Biomineralization in the Pearl Oyster Pinctada fucata. Environ. Sci. Technol. 2016, 50, 1157–1165. [Google Scholar] [CrossRef]
- Mucci, A.; Morse, J.W. The incorporation of Mg2+ and Sr2+ into calcite overgrowths: Influences of growth rate and solution composition. Geochim. Cosmochim. Acta 1983, 47, 217–223. [Google Scholar] [CrossRef]
- Bertoni, S.; Liu, Z.; Correia, A.; Martins, J.P.; Rahikkala, A.; Fontana, F.; Kemell, M.; Liu, D.; Albertini, B.; Passerini, N.; et al. pH and reactive oxygen species-sequential responsive nano-in-micro composite for targeted therapy of inflammatory bowel disease. Adv. Funct. Mater. 2018, 28. [Google Scholar] [CrossRef]
- Wang, G.; Cao, R.Y.; Chen, R.; Mo, L.; Han, J.F.; Wang, X.; Xu, X.; Jiang, T.; Deng, Y.Q.; Lyu, K.; et al. Rational design of thermostable vaccines by engineered peptide-induced virus self-biomineralization under physiological conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 7619–7624. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, L.B.; De Schryver, A.M.; Hendriks, A.J.; Huijbregts, M.A. Calcifying species sensitivity distributions for ocean acidification. Environ. Sci. Technol. 2015, 49, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.; Hendriks, I.E.; Ramajo, L.; Singh, G.S.; Duarte, C.M.; Gattuso, J.P. Impacts of ocean acidification on marine organisms: Quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 2013, 19, 1884–1896. [Google Scholar] [CrossRef]
- Welladsen, H.M.; Southgate, P.C.; Heimann, K. The effects of exposure to near-future levels of ocean acidification on shell characteristics of Pinctada fucata (Bivalvia: Pteriidae). Molluscan Res. 2010, 30, 125–130. [Google Scholar] [CrossRef]
- Ding, H.; Pan, H.; Xu, X.; Tang, R. Toward a Detailed Understanding of Magnesium Ions on Hydroxyapatite Crystallization Inhibition. Cryst. Growth Des. 2014, 14, 763–769. [Google Scholar] [CrossRef]
- You, H.; Fang, J. Particle-mediated nucleation and growth of solution-synthesized metal nanocrystals: A new story beyond the LaMer curve. Nano Today 2016, 11, 145–167. [Google Scholar] [CrossRef]
- Gebauer, D. How Can Additives Control the Early Stages of Mineralisation? Minerals 2018, 8. [Google Scholar] [CrossRef]
- Yang, J.; Liu, Y.; Wen, T.; Wei, X.; Li, Z.; Cai, Y.; Su, Y.; Wang, D. Controlled Mineralization of Calcium Carbonate on the Surface of Nonpolar Organic Fibers. Cryst. Growth Des. 2012, 12, 29–32. [Google Scholar] [CrossRef]
- Wang, T.; Leng, B.X.; Che, R.C.; Shao, Z.Z. Biomimetic Synthesis of Multilayered Aragonite Aggregates Using Alginate as Crystal Growth Modifier. Langmuir 2010, 26, 13385–13392. [Google Scholar] [CrossRef] [PubMed]
- Pastero, L.; Costa, E.; Bruno, M.; Rubbo, M.; Sgualdino, G.; Aquilano, D. Morphology of calcite (CaCO3) crystals growing from aqueous solutions in the presence of Li+ ions. Surface behavior of the {0001} form. Cryst. Growth Des. 2004, 4, 485–490. [Google Scholar] [CrossRef]
- Liu, Y.; Tan, Y.; Ren, J.; Chen, H.; Li, H. Assessing the synergy effect of additive and matrix on single-crystal growth: Morphological revolution resulted from gel-mediated enhancement on CIT-calcite interaction. Chin. Chem. Lett. 2018, 29, 1296–1300. [Google Scholar] [CrossRef]
- Lei, M.; Li, P.G.; Sun, Z.B.; Tang, W.H. Effects oforganic additives on the morphology of calcium carbonate particles in the presence of CTAB. Mater. Lett. 2006, 60, 1261–1264. [Google Scholar] [CrossRef]
- Liu, X.; Zeng, S.; Dong, S.; Jin, C.; Li, J. A Novel Matrix Protein Hic31 from the Prismatic Layer of Hyriopsis Cumingii Displays a Collagen-Like Structure. PLoS ONE 2015, 10, e0135123. [Google Scholar] [CrossRef]
- Pacella, M.S.; Gray, J.J. A Benchmarking Study of Peptide–Biomineral Interactions. Cryst. Growth Des. 2018, 18, 607–616. [Google Scholar] [CrossRef]
- Addadi, L.; Weiner, S.; Geva, M. On how proteins interact with crystals and their effect on crystal formation. Clin. Res. Cardiol. 2001, 90, 92–98. [Google Scholar] [CrossRef]
- Aizenberg, J.; Muller, D.A.; Grazul, J.L.; Hamann, D.R. Direct Fabrication of Large Micropatterned Single Crystals. Science 2003, 299, 1205–1208. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Horiuchi, N.; Nakamura, M.; Nozaki, K.; Nagai, A.; Yamashita, K. Calcite Crystallization on Polarized Single Calcite Crystal Substrates in the Presence of Poly-Lysine. Cryst. Growth Des. 2018, 18, 872–878. [Google Scholar] [CrossRef]
- Shirazi, S.F.; Gharehkhani, S.; Mehrali, M.; Yarmand, H.; Metselaar, H.S.; Adib Kadri, N.; Osman, N.A. A review on powder-based additive manufacturing for tissue engineering: Selective laser sintering and inkjet 3D printing. Sci. Technol. Adv. Mater. 2015, 16, 033502. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, P.; Jiang, W.; Pan, H.; Xu, X.; Tang, R. Yeast Cells with an Artificial Mineral Shell: Protection and Modification of Living Cells by Biomimetic Mineralization. Angew. Chem. Int. Ed. Engl. 2008, 47, 3560–3564. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Li, M.; Wang, B.; Liu, M.F.; Shao, C.; Pan, H.; Lu, Y.; Xu, B.B.; Li, S.; Zhan, D.; et al. Total morphosynthesis of biomimetic prismatic-type CaCO3 thin films. Nat. Commun. 2017, 8, 1398. [Google Scholar] [CrossRef]
- Li, X.; Zhang, W.; Lu, J.; Huang, L.; Nan, D.; Webb, M.A.; Hillion, F.; Wang, L. Templated Biomineralization on Self-Assembled Protein Nanofibers Buried in Calcium Oxalate Raphides of Musa spp. Chem. Mater. 2014, 26, 3862–3869. [Google Scholar] [CrossRef]
- Lin, S.; Zhong, Y.; Zhao, X.; Sawada, T.; Li, X.; Lei, W.; Wang, M.; Serizawa, T.; Zhu, H. Synthetic Multifunctional Graphene Composites with Reshaping and Self-Healing Features via a Facile Biomineralization-Inspired Process. Adv. Mater. 2018, e1803004. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, Z.; Guo, L. Nacre-inspired composites with different macroscopic dimensions: Strategies for improved mechanical performance and applications. NPG Asia Mater. 2018, 10, 1–22. [Google Scholar] [CrossRef]
- Zhang, S.; Jiang, Z.; Qian, W.; Shi, J.; Wang, X.; Tang, L.; Zou, H.; Liu, H. Preparation of Ultrathin, Robust Nanohybrid Capsules through a “Beyond Biomineralization” Method. ACS Appl. Mater. Interfaces 2017, 9, 12841–12850. [Google Scholar] [CrossRef]
- Xu, Z.; Huang, J.; Zhang, C.; Daryadel, S.; Behroozfar, A.; McWilliams, B.; Boesl, B.; Agarwal, A.; Minary-Jolandan, M. Bioinspired Nacre-Like Ceramic with Nickel Inclusions Fabricated by Electroless Plating and Spark Plasma Sintering. Adv. Eng. Mater. 2018, 20, 1700782. [Google Scholar] [CrossRef]
- Yan, Y.; Shi, X.R.; Miao, M.; He, T.; Dong, Z.H.; Zhan, K.; Yang, J.H.; Zhao, B.; Xia, B.Y. Bio-inspired design of hierarchical FeP nanostructure arrays for the hydrogen evolution reaction. Nano Res. 2018, 11, 3537–3547. [Google Scholar] [CrossRef]
- Hou, Y.K.; Pan, G.L.; Sun, Y.Y.; Gao, X.P. Li-Rich Layered Oxide Microspheres Prepared by the Biomineralization as High-Rate and Cycling-Stable Cathode for Li-Ion Batteries. ACS Appl. Energy Mater. 2018. [Google Scholar] [CrossRef]
- Hou, Y.K.; Pan, G.L.; Sun, Y.Y.; Gao, X.P. LiMn0.8Fe0.2PO4/Carbon Nanospheres@Graphene Nanoribbons Prepared by the Biomineralization Process as the Cathode for Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2018, 10, 16500–16510. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhou, G.; Dai, H.; Yang, M.; Fu, Y.; Ying, Y.; Li, Y. Biomineralization-mimetic preparation of hybrid membranes with ultra-high loading of pristine metal–organic frameworks grown on silk nanofibers for hazard collection in water. Lab Chip 2018, 6, 3402–3413. [Google Scholar] [CrossRef]
- Xiong, W.; Yang, Z.; Zhai, H.; Wang, G.; Xu, X.; Ma, W.; Tang, R. Alleviation of high light-induced photoinhibition in cyanobacteria by artificially conferred biosilica shells. Chem. Commun. 2013, 49, 7525–7527. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ju, Y.; Zong, Y.; Qi, H.; Zhao, K. In Situ Real-Time Study on Dynamics of Microbially Induced Calcium Carbonate Precipitation at a Single-Cell Level. Environ. Sci. Technol. 2018, 52, 9266–9276. [Google Scholar] [CrossRef]
- Chen, W.; Wang, G.; Tang, R. Nanomodification of living organisms by biomimetic mineralization. Nano Res. 2014, 7, 1404–1428. [Google Scholar] [CrossRef]
- Wang, X.; Deng, Y.; Li, S.; Wang, G.; Qin, E.; Xu, X.; Tang, R.; Qin, C. Biomineralization-Based Virus Shell-Engineering: Towards Neutralization Escape and Tropism Expansion. Adv. Healthc. Mater. 2012, 1, 443–449. [Google Scholar] [CrossRef]
- Walsh, P.; Fee, K.; Clarke, S.; Julius, M.; Buchanan, F. Blueprints for the Next Generation of Bioinspired and Biomimetic Mineralised Composites for Bone Regeneration. Mar. Drugs 2018, 16. [Google Scholar] [CrossRef]
- Julius Rebek, J. Introduction to the Molecular Recognition and Self-Assembly Special Feature. PNAS 2009, 106, 10423–10424. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Ko, E.H.; Jung, Y.H.; Choi, I.S. Bioinspired functionalization of silica-encapsulated yeast cells. Angew. Chem. Int. Ed. Engl. 2011, 50, 6115–6118. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Hong, D.; Choi, J.Y.; Kim, J.Y.; Lee, S.H.; Kim, H.M.; Yang, S.H.; Choi, I.S. Layer-by-Layer-Based Silica Encapsulation of Individual Yeast with Thickness Control. ChemViews 2014. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, Y.; Han, J.; Xu, Q.; Ren, J.; Xu, J.; Wang, Y.; Chai, Z. Noninvasive Multimodal Imaging of Osteosarcoma and Lymph Nodes Using a 99mTc-Labeled Biomineralization Nanoprobe. Anal. Chem. 2018, 90, 4529–4534. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Li, D.; Su, Y.; Lu, C.; Niu, L.; Lian, J.; Li, G. Improvement of the Biodegradation Property and Biomineralization Ability of Magnesium–Hydroxyapatite Composites with Dicalcium Phosphate Dihydrate and Hydroxyapatite Coatings. ACS Biomater. Sci. Eng. 2016, 2, 818–828. [Google Scholar] [CrossRef]
- Hwang, E.T.; Tatavarty, R.; Chung, J.; Gu, M.B. New Functional Amorphous Calcium Phosphate Nanocomposites by Enzyme-Assisted Biomineralization. ACS Appl. Mater. Interfaces 2013, 5, 532–537. [Google Scholar] [CrossRef]
- Yang, B.; Yin, J.; Chen, Y.; Pan, S.; Yao, H.; Gao, Y.; Shi, J. 2D-Black-Phosphorus-Reinforced 3D-Printed Scaffolds: A Stepwise Countermeasure for Osteosarcoma. Adv. Mater. 2018, 30. [Google Scholar] [CrossRef]
- Zhu, W.; Li, J.; Lei, J.; Li, Y.; Chen, T.; Duan, T.; Yao, W.; Zhou, J.; Yu, Y.; Liu, Y. Silver nanoparticles incorporated konjac glucomannan-montmorillonite nacre-like composite films for antibacterial applications. Carbohydr. Polym. 2018, 197, 253–259. [Google Scholar] [CrossRef]
- Chen, W.; Xiao, Y.; Liu, X.; Chen, Y.; Zhang, J.; Xu, X.; Tang, R. Overcoming cisplatin resistance in chemotherapy by biomineralization. Chem. Commun. 2013, 49, 4932–4934. [Google Scholar] [CrossRef]
- Xiao, B.; Zhou, X.; Xu, H.; Wu, B.; Hu, D.; Hu, H.; Pu, K.; Zhou, Z.; Liu, X.; Tang, J.; et al. Integration of Polymerization and Biomineralization as a Strategy to Facilely Synthesize Nanotheranostic Agents. ACS Nano 2018. [Google Scholar] [CrossRef]
- Wang, Z.; Huang, P.; Jacobson, O.; Wang, Z.; Liu, Y.; Lin, L.; Lin, J.; Lu, N.; Zhang, H.; Tian, R.; et al. Biomineralization-Inspired Synthesis of Copper Sulfide-Ferritin Nanocages as Cancer Theranostics. ACS Nano 2016, 10, 3453–3460. [Google Scholar] [CrossRef]
- Zhang, J.; Hao, G.; Yao, C.; Yu, J.; Wang, J.; Yang, W.; Hu, C.; Zhang, B. Albumin-Mediated Biomineralization of Paramagnetic NIR Ag2S QDs for Tiny Tumor Bimodal Targeted Imaging in Vivo. ACS Appl. Mater. Interfaces 2016, 8, 16612–16621. [Google Scholar] [CrossRef]
- Srivastava, P.; Hira, S.K.; Srivastava, D.N.; Singh, V.K.; Gupta, U.; Singh, R.; Singh, R.A.; Manna, P.P. ATP-Decorated Mesoporous Silica for Biomineralization of Calcium Carbonate and P2 Purinergic Receptor-Mediated Antitumor Activity against Aggressive Lymphoma. ACS Appl. Mater. Interfaces 2018, 10, 6917–6929. [Google Scholar] [CrossRef]
- Jiang, J.X.; Riquelme, M.A.; Zhou, J.Z. ATP, a double-edged sword in cancer. Oncoscience 2015, 2, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Pawelec, K.M.; Kluijtmans, S.G. Biomineralization of Recombinant Peptide Scaffolds: Interplay among Chemistry, Architecture, and Mechanics. ACS Biomater. Sci. Eng. 2017, 3, 1100–1108. [Google Scholar] [CrossRef]
- Yi, L.; Wen, G.; Mi, N.; Yumeng, C.; Juan, X.; Bo, M. Biodegradable Multifunctional Bioactive Glass-Based Nanocomposite Elastomers with Controlled Biomineralization Activity, Real-Time Bioimaging Tracking, and Decreased Inflammatory Response. ACS Appl. Mater. Interfaces 2018, 10, 17722–17731. [Google Scholar] [CrossRef]
- Hess, R.; Jaeschke, A.; Neubert, H.; Hintze, V.; Moeller, S.; Schnabelrauch, M.; Wiesmann, H.P.; Hart, D.A.; Scharnweber, D. Synergistic effect of defined artificial extracellular matrices and pulsed electric fields on osteogenic differentiation of human MSCs. Biomaterials 2012, 33, 8975–8985. [Google Scholar] [CrossRef] [PubMed]
- Kuzyk, P.R.; Schemitsch, E.H. The science of electrical stimulation therapy for fracture healing. Indian J. Orthop. 2009, 43, 127–131. [Google Scholar]
- Zhang, C.; Liu, W.; Cao, C.; Zhang, F.; Tang, Q.; Ma, S.; Zhao, J.; Hu, L.; Shen, Y.; Chen, L. Modulating Surface Potential by Controlling the β Phase Content in Poly(vinylidene fluoridetrifluoroethylene) Membranes Enhances Bone Regeneration. Adv. Healthc. Mater. 2018, 7, 1701466. [Google Scholar] [CrossRef]
- Yadav, R.; Goud, R.; Dutta, A.; Wang, X.; Naebe, M.; Kandasubramanian, B. Biomimicking of Hierarchal Molluscan Shell Structure Via Layer by Layer 3D Printing. Ind. Eng. Chem. Res. 2018, 57, 10832–10840. [Google Scholar] [CrossRef]
- Yadav, R.; Naebe, M.; Wang, X.; Kandasubramanian, B. Review on 3D Prototyping of Damage Tolerant Interdigitating Brick Arrays of Nacre. Ind. Eng. Chem. Res. 2017, 56, 10516–10525. [Google Scholar] [CrossRef]
- Itel, F.; Skovhus Thomsen, J.; Städler, B. Matrix Vesicles-Containing Microreactors as Support for Bonelike Osteoblasts to Enhance Biomineralization. ACS Appl. Mater. Interfaces 2018, 10, 30180–30190. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Gao, G.; Jang, J.; Cho, D.W. 3D printed structures for delivery of biomolecules and cells: Tissue repair and regeneration. J. Mater. Chem. B 2016, 4, 7521–7539. [Google Scholar] [CrossRef]
- Jackson, R.J.; Patrick, P.S.; Page, K.; Powell, M.J.; Lythgoe, M.F.; Miodownik, M.A.; Parkin, I.P.; Carmalt, C.J.; Kalber, T.L.; Bear, J.C. Chemically Treated 3D Printed Polymer Scaffolds for Biomineral Formation. ACS Omega 2018, 3, 4342–4351. [Google Scholar] [CrossRef]
- Wang, S.; Ni, D.; Yue, H.; Luo, N.; Xi, X.; Wang, Y.; Shi, M.; Wei, W.; Ma, G. Exploration of Antigen Induced CaCO3 Nanoparticles for Therapeutic Vaccine. Small 2018, 14. [Google Scholar] [CrossRef]
- Zhou, X.; Xia, S.; Lu, Z.; Tian, Y.; Yan, Y.; Zhu, J. Biomineralization-Assisted Ultrasensitive Detection of DNA. J. Am. Chem. Soc. 2010, 132, 6932–6934. [Google Scholar] [CrossRef] [PubMed]
- Taton, T.A.; Mirkin, C.A.; Letsinger, R.L. Scanometric DNA array detection with nanoparticle probes. Science 2000, 289, 1757–1760. [Google Scholar] [CrossRef]
- Xie, W.; Tadepalli, S.; Park, S.H.; Kazemi-Moridani, A.; Jiang, Q.; Singamaneni, S.; Lee, J.H. Extreme Mechanical Behavior of Nacre-Mimetic Graphene-Oxide and Silk Nanocomposites. Nano Lett. 2018, 18, 987–993. [Google Scholar] [CrossRef] [PubMed]
- Fairbrother, L.; Etschmann, B.; Brugger, J.; Shapter, J.; Southam, G.; Reith, F. Biomineralization of gold in biofilms of Cupriavidus metallidurans. Environ. Sci. Technol. 2013, 47, 2628–2635. [Google Scholar] [CrossRef]
- Fleming, D.O.; Hunt, D.L. (Eds.) Biological Safety: Principles and Practice; Diagnostic Microbiology & Infectious Disease; ASM Press: Washington, DC, USA, 2001; Volume 39, pp. 203–203. [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Feng, Y.; Deveaux, J.G.; Masoud, M.A.; Chandra, F.S.; Chen, H.; Zhang, D.; Feng, L. Biomineralization Forming Process and Bio-inspired Nanomaterials for Biomedical Application: A Review. Minerals 2019, 9, 68. https://doi.org/10.3390/min9020068
Chen Y, Feng Y, Deveaux JG, Masoud MA, Chandra FS, Chen H, Zhang D, Feng L. Biomineralization Forming Process and Bio-inspired Nanomaterials for Biomedical Application: A Review. Minerals. 2019; 9(2):68. https://doi.org/10.3390/min9020068
Chicago/Turabian StyleChen, Yuanyuan, Yanmin Feng, John Gregory Deveaux, Mohamed Ahmed Masoud, Felix Sunata Chandra, Huawei Chen, Deyuan Zhang, and Lin Feng. 2019. "Biomineralization Forming Process and Bio-inspired Nanomaterials for Biomedical Application: A Review" Minerals 9, no. 2: 68. https://doi.org/10.3390/min9020068
APA StyleChen, Y., Feng, Y., Deveaux, J. G., Masoud, M. A., Chandra, F. S., Chen, H., Zhang, D., & Feng, L. (2019). Biomineralization Forming Process and Bio-inspired Nanomaterials for Biomedical Application: A Review. Minerals, 9(2), 68. https://doi.org/10.3390/min9020068





