Emerald Deposits: A Review and Enhanced Classification
Abstract
:1. Introduction
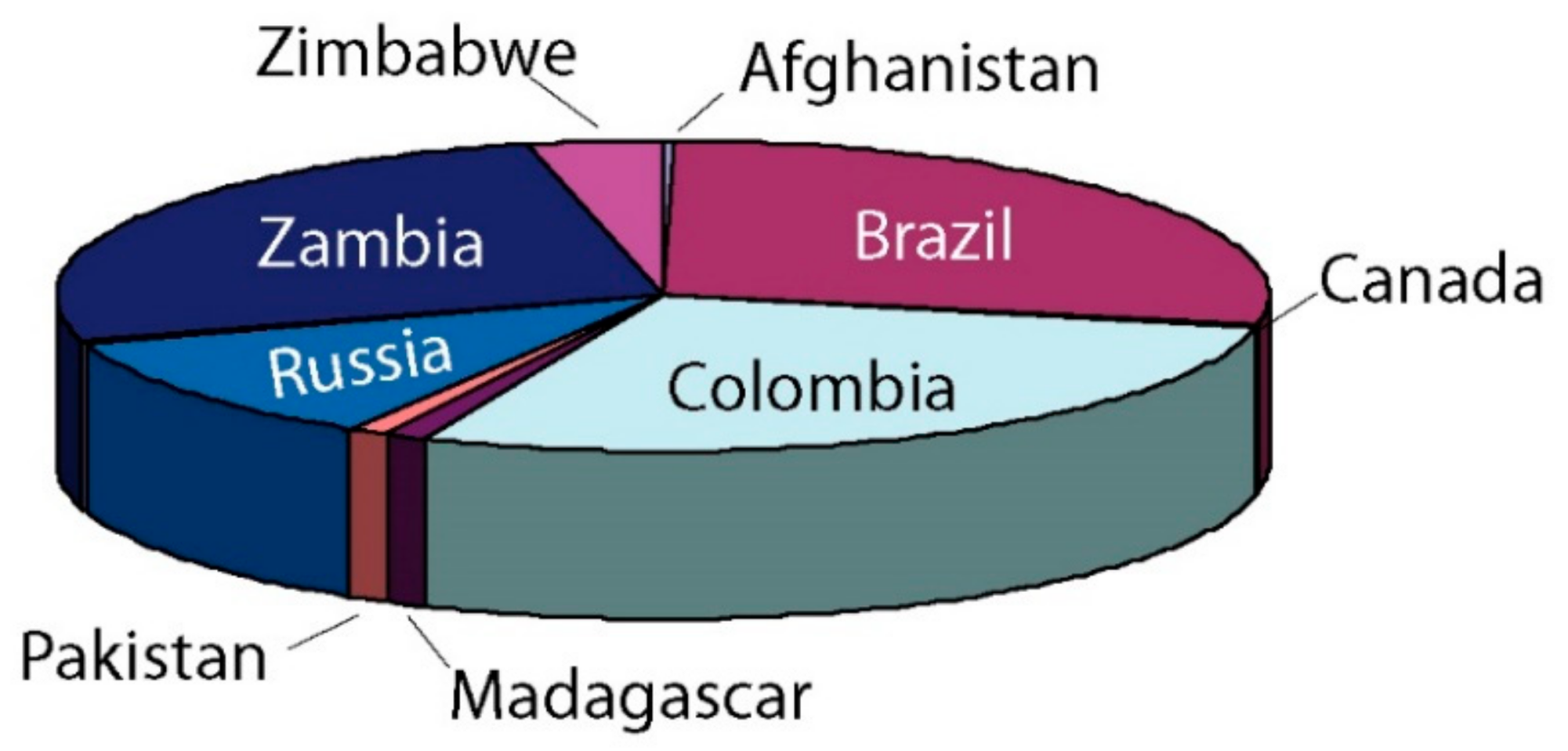
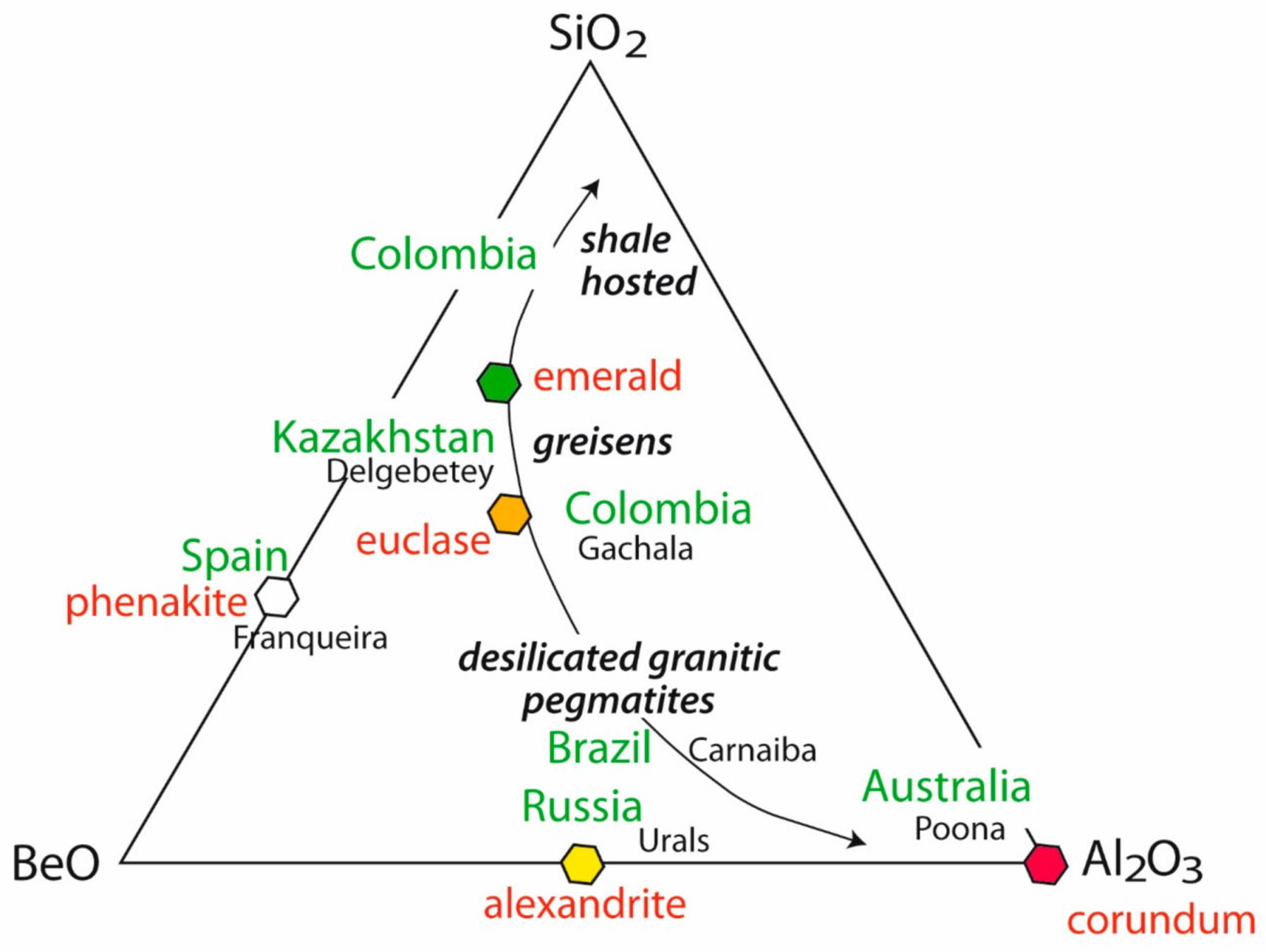
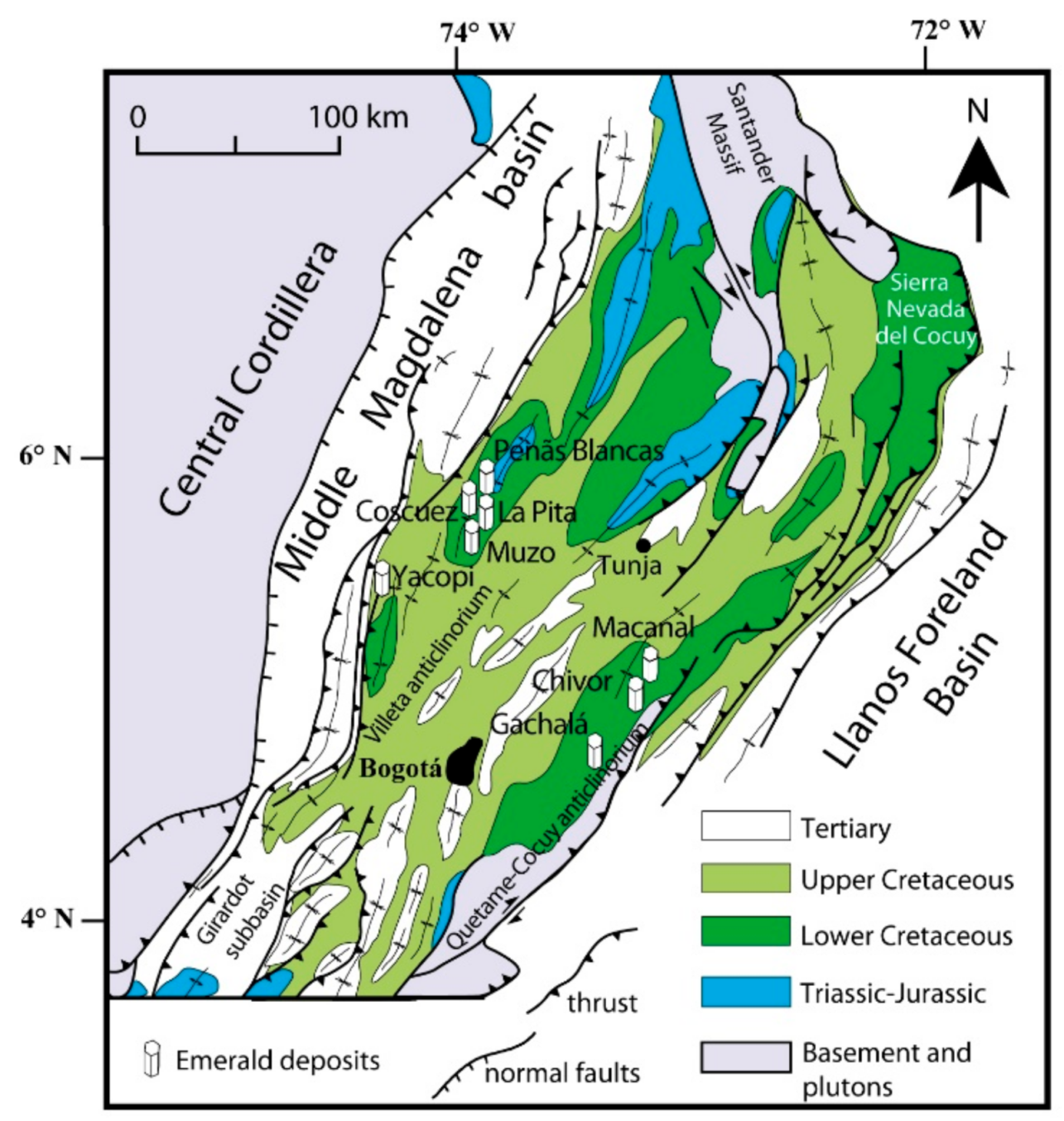
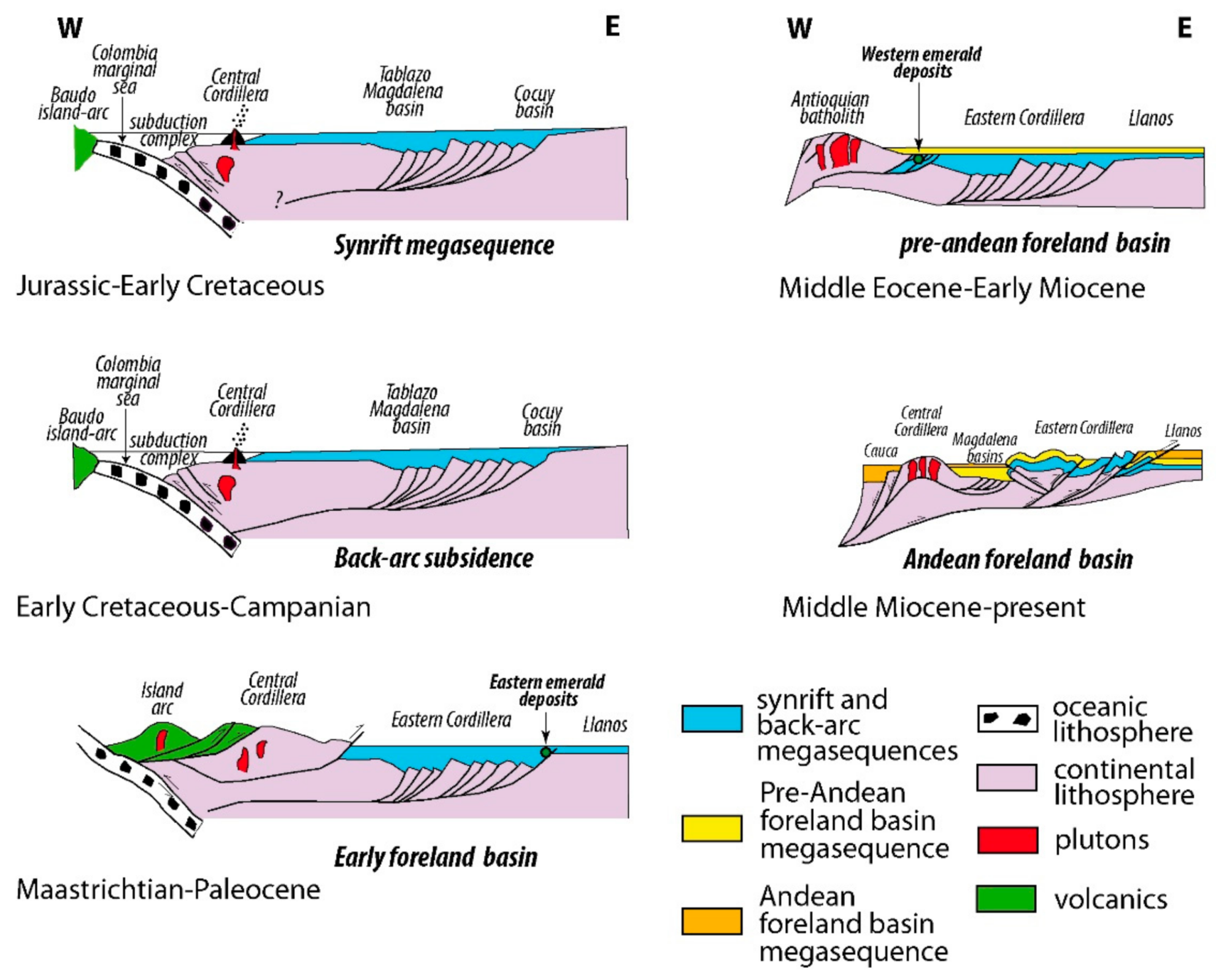
2. Worldwide Emerald Deposits
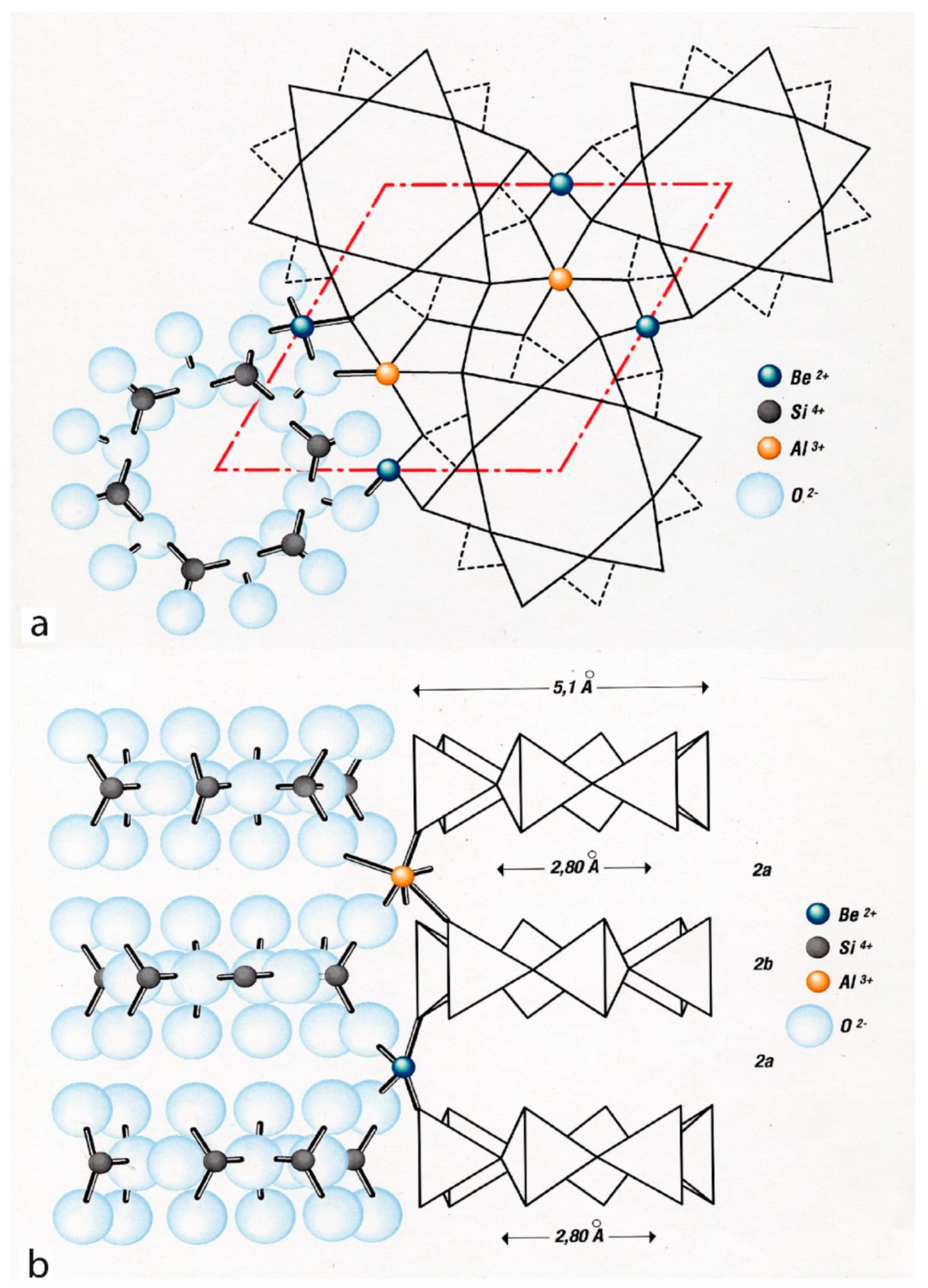
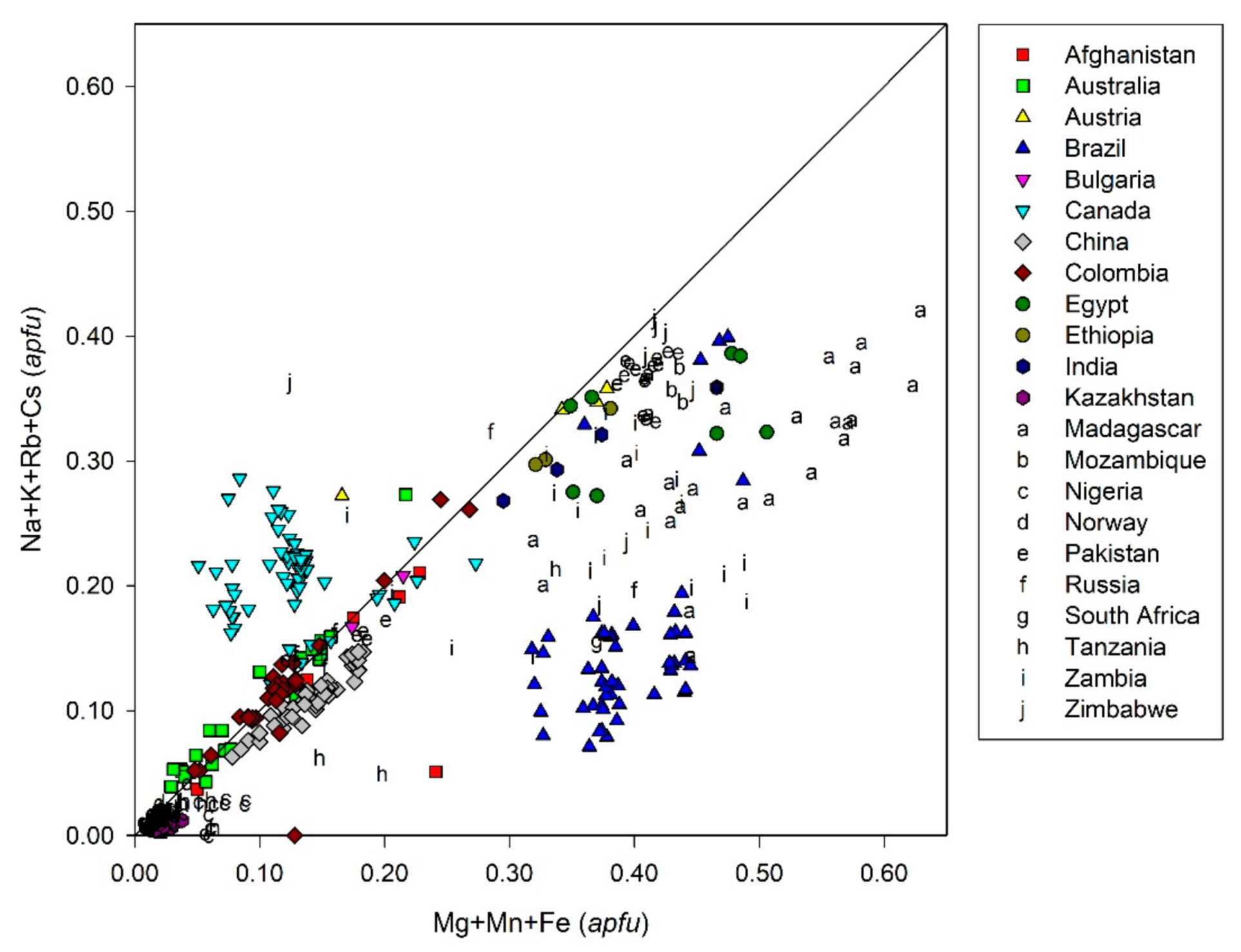
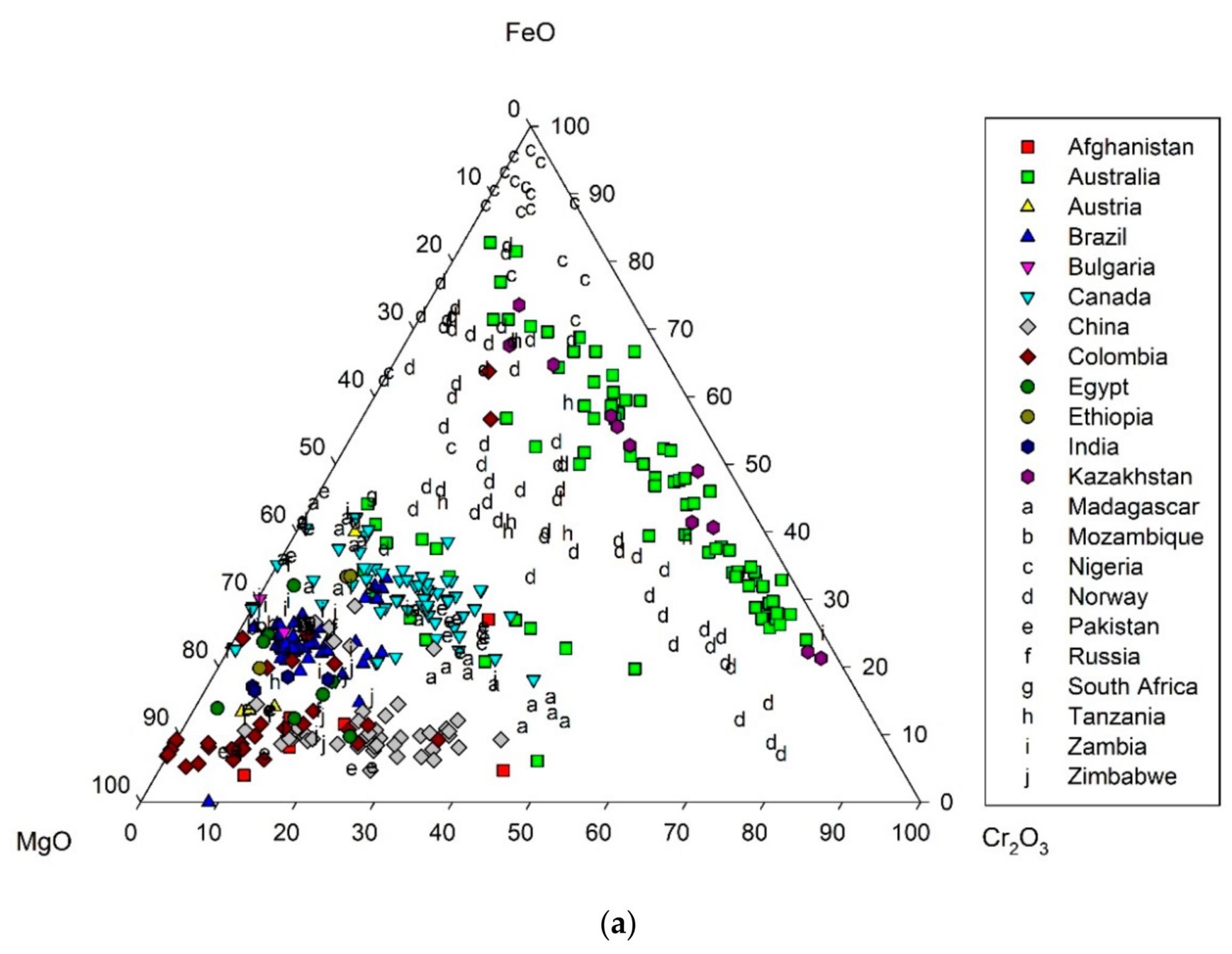
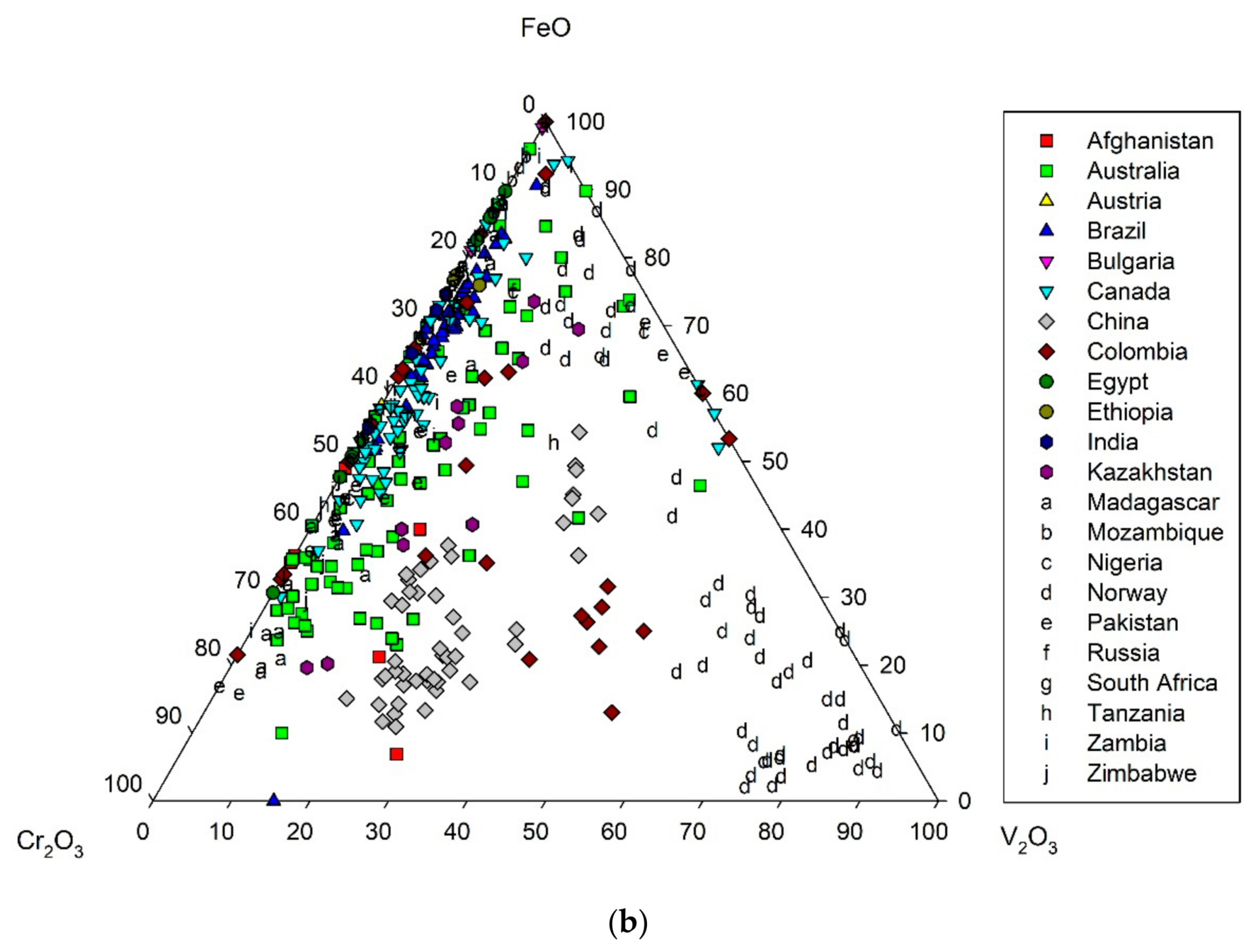
3. Crystal Chemistry of Emerald
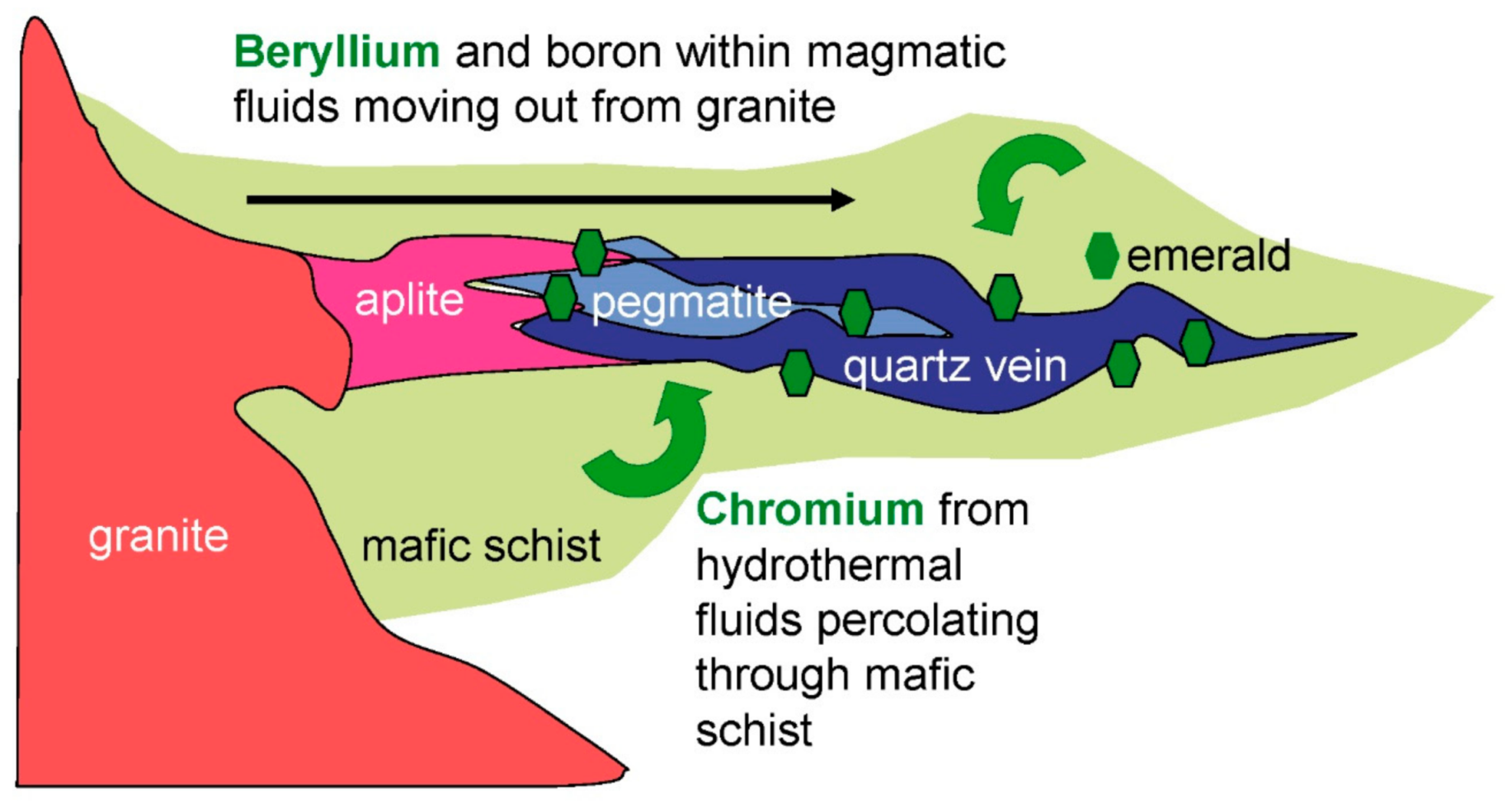
4. Sources of Be, Cr, and V: The Formation of Emerald
5. Classification of Emerald Deposits
5.1. Genetic Classifications
5.2. A Revised Classification for Emerald Deposits
- (Type I)
- Tectonic magmatic-related with sub-types hosted in:
- (IA)
- Mafic-ultramafic rocks (Brazil, Zambia, Russia, and others);
- (IB)
- Sedimentary rocks (China, Canada, Norway, Kazakhstan Australia);
- (IC)
- Granitic rocks (Nigeria).
- (Type II)
- Tectonic metamorphic-related with sub-types hosted in:
- (IIA)
- M-UMR (Brazil, Austria);
- (IIB)
- Sedimentary rocks-black shale (Colombia, Canada, USA);
- (IIC)
- Metamorphic rocks (China, Afghanistan, USA);
- (IID)
- Metamorphosed type I deposits or hidden-granitic intrusion-related (Austria, Egypt, Australia, Pakistan), and some unclassified deposits.
6. Different Types of Emerald Deposits
6.1. Tectonic Magmatic-Related (Type I)
6.1.1. Sub-Type IA: Tectonic Magmatic-Related Emerald Deposits Hosted in M-UMR (Brazil, Zambia, Russia, and Others; See Table 2 and Figure 2)
- Zone 6:
- Coarse-grained F-phlogopite + apatite + emerald + quartz;
- Zone 5:
- Fine-grained F-phlogopite + apatite + emerald;
- Zone 4:
- F-phlogopite + spinel (chromite + magnetite);
- Zone 3:
- F-phlogopite + spinel + amphibole (actinolite + tremolite);
- Zone 2:
- F-phlogopite + spinel + ampbibole + talc; and
- Zone 1:
- spinel + amphibole (or dolomite) + talc + serpentine + chlorite.
- At the Tsa da Gliza deposit in Canada, Be-bearing magmatic fluids from the neighboring granite reacted with the Cr- and V-bearing M-UMR [46]. Emeralds formed in aplite, pegmatite, and quartz veins surrounded by biotite schists. There is a continuum between the crystallization of granitic rocks, fluid rock-interaction, and emerald formation (Figure 18).
- In the Kafubu area in Zambia (Figure 1c), emerald is found predominantly in metamorphosed M-UMR with phlogopite schist or in quartz-tourmaline veins adjacent to pegmatites.
- At the Sandawana deposit in Zimbabwe, pegmatites intruded the M-UMR, but they are folded and fluid circulation in shear zones formed phlogopite schist. The fluid-rock interaction is coeval with the regional deformation [52]. Such phenomena are also found in the Carnaíba deposits in Brazil, where the dissolution of quartz from pegmatite is common [76].
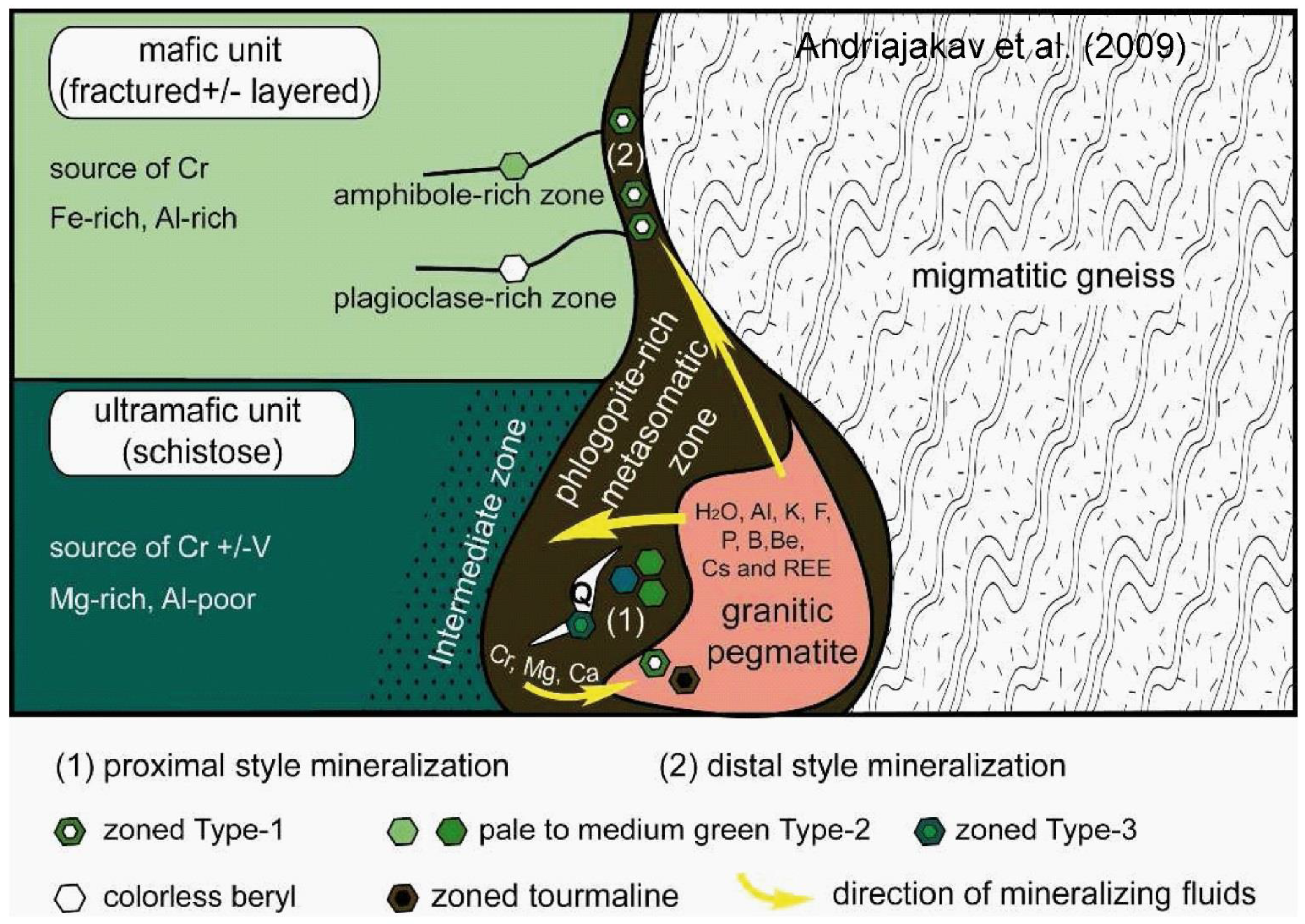
- At the Ianapera deposit in Madagascar, two coeval emerald deposits coexist (Figure 19; [11]): (1) A proximal one, formed at the contact between pegmatites and UMR; and (2) a distal one, hosted in biotite schist in fractures developed in mafic rocks with widespread fluid circulation affecting all the geological formations. Similarly, the Trecho Novo and Trecho Velho deposits at Carnaíba, Brazil [78] are also distal in nature.
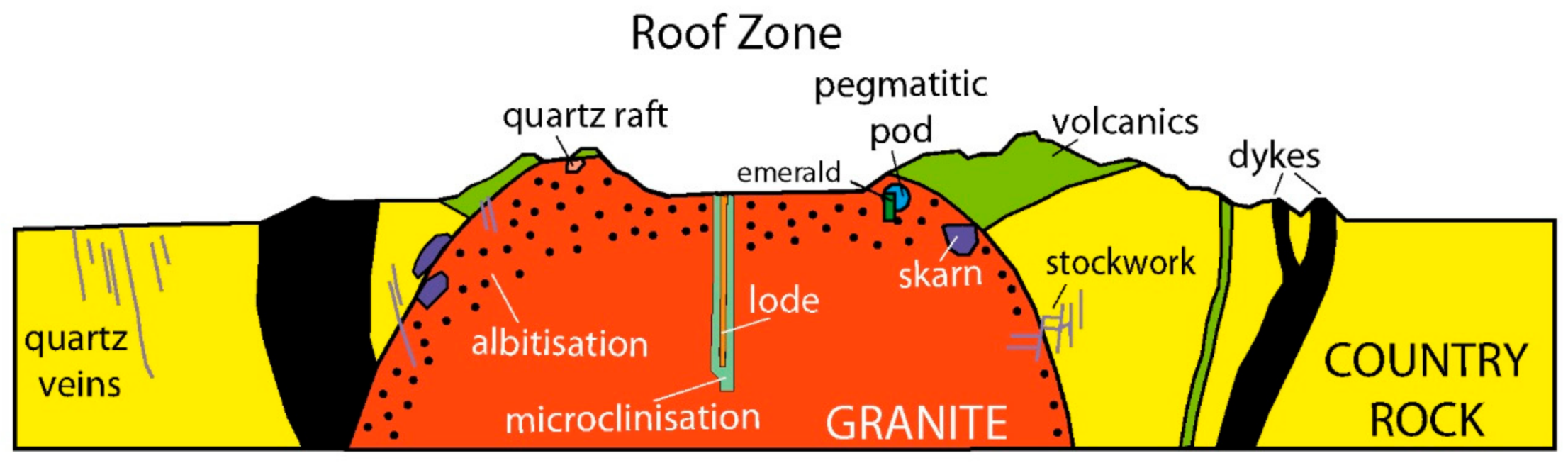
6.1.2. Sub-Type IB: Tectonic Magmatic-Related Emerald Deposits Hosted in (Meta)-Sedimentary Rocks (China, Canada, Kazakhstan, Norway, Australia; See Table 2)
- At Dyakou (China), the Lower Cretaceous porphyritic granite intruded biotite granofels, quartzite, gneiss, and plagioclase amphibolite of Lower and Upper Neoproterozoic formations. The intrusion formed skarns and dyke swarms of quartz veins, which are crosscut by pegmatites [85]. Emerald in quartz veins is less abundant than in the pegmatites, but of higher quality (Figure 1b). Some pegmatites show a local zoning with an outer zone enriched in K-feldspar and an inner zone of emerald and quartz.
- At the Lened V-rich emerald occurrence (Canada), Be and other incompatible elements (i.e., W, Sn, Li, B, and F) in the emerald, vein minerals, and surrounding skarn were derived during the terminal stages of crystallization of the proximal Lened pluton [21,47]. Decarbonation during pyroxene-garnet skarn formation in the host carbonate rocks probably caused local overpressuring and fracturing that allowed ingress of magma-derived fluids and formation of quartz-calcite-beryl-scheelite-tourmaline-pyrite veins. The vein fluid was largely igneous in origin, but the dominant emerald chromophore V was mobilized by metasomatism of V-rich sedimentary rocks (avg. 2000 ppm V) that underlie the emerald occurrence [21].
- At Delbegetey (Kazakhstan), the emerald mineralization is confined to the granite that hornfelsed carboniferous sandstones. Emerald is found in muscovite greisen formed in the wall-rocks of muscovite-tourmaline-fluorite-bearing quartz veins [51].
- At Eidswoll (Norway), the emerald Byrud Gård mine is related to Permo-Triassic alkaline intrusions. The V-bearing emerald occurs in Middle Triassic pegmatite veins that intruded Cambrian Alum shales and quartz syenite sills [20]. Vanadium and Cr were probably leached from the alum shales by the mineralizing fluids [13].
- At Emmaville-Torrington (Australia), emerald is located in pegmatite, aplite, and quartz veins associated with the Mole granite. The granite intrudes a Permian metasedimentary sequence consisting of meta-siltstones, slates, and quartzites [19]. The emerald-bearing pegmatite veins contain quartz, topaz, K-feldspar, and mica. Emerald is embedded in cavities and surrounded by dickite in the quartz-topaz veins. In the quartz lodes, emerald is associated with Sn-W-F minerals. At the Heffernan’s Wolfram mine, emerald occurs with wolframite in vugs in the pegmatites [86].
6.1.3. Sub-Type IC: Tectonic Magmatic-Related Emerald Deposits Hosted in Peralkaline Granites (Nigeria)
6.2. Tectonic Metamorphic-Related (Type II)
6.2.1. Sub-Type IIA: Tectonic Metamorphic-Related Emerald Deposits Hosted in M-UMR (Brazil, Austria, Pakistan; See Table 1)
- 1.
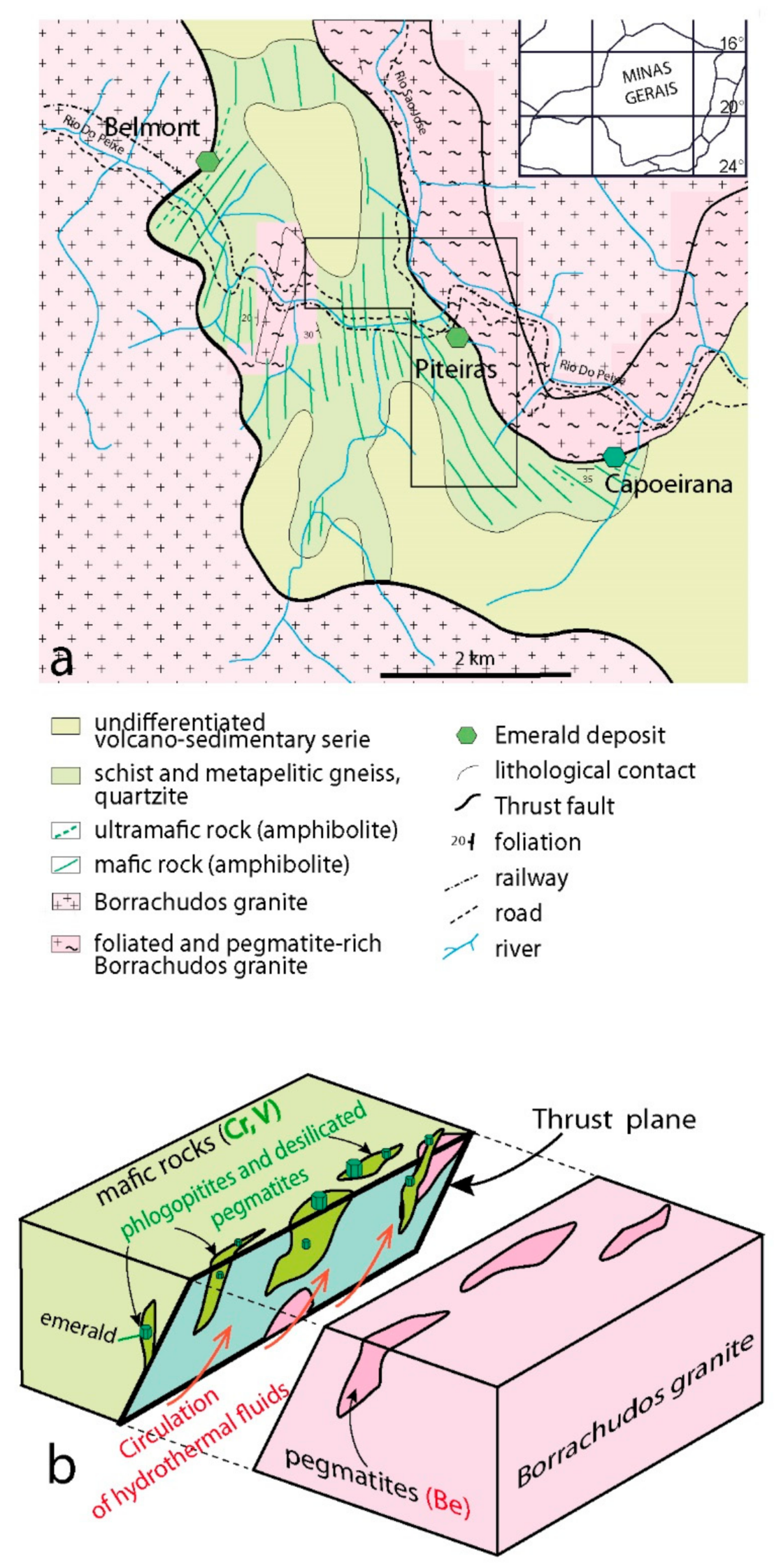
- The Santa Terezinha de Goiás deposit, located in central Goiás, produced 155 tons of emerald between 1981, date of the discovery, and 1988 [88]. The emerald grade was between 50 and 800 g/t. The infiltration of hydrothermal fluids is controlled by tectonic structures, such as the thrust and shear zones (Figure 21a). Pegmatite veins are absent and the mineralization is stratiform (Figure 21b). Emerald is disseminated within phlogopitites and phlogopitized carbonate-talc schists of the metavolcanic sedimentary sequence of Santa Terezinha [88,89,90]. Talc-schists provide the main sites for thrusting and the formation of sheath folds [91]. Emerald-rich zones are commonly found in the cores of sheath folds and along the foliation (Figure 21c). Two types of ore can be distinguished [88]: (i) A carbonate-rich ore composed of dolomite, talc, phlogopite, quartz, chlorite, tremolite, spinel, pyrite, and emerald; (ii) a phlogopite-rich ore composed of phlogopite, quartz, carbonates, chlorite, talc, pyrite, and emerald.The distal São José two-micas granite, located 5 km from the emerald deposit, is a syntectonic foliated granite, which underwent a polyphase ductile deformation coeval to that observed in the emerald deposit [92]. C and S structures in the granite indicate shear deformation along a typical frontal thrust ramp and the granite overthrusted the Santa Terezinha sequence where the emerald deposit is located. D’el-Rey Silva and Barros Neto [92] suggested that the granite most probably was the source of Be for the formation of emerald in the Santa Terezinha de Goiás deposit.
- 2.
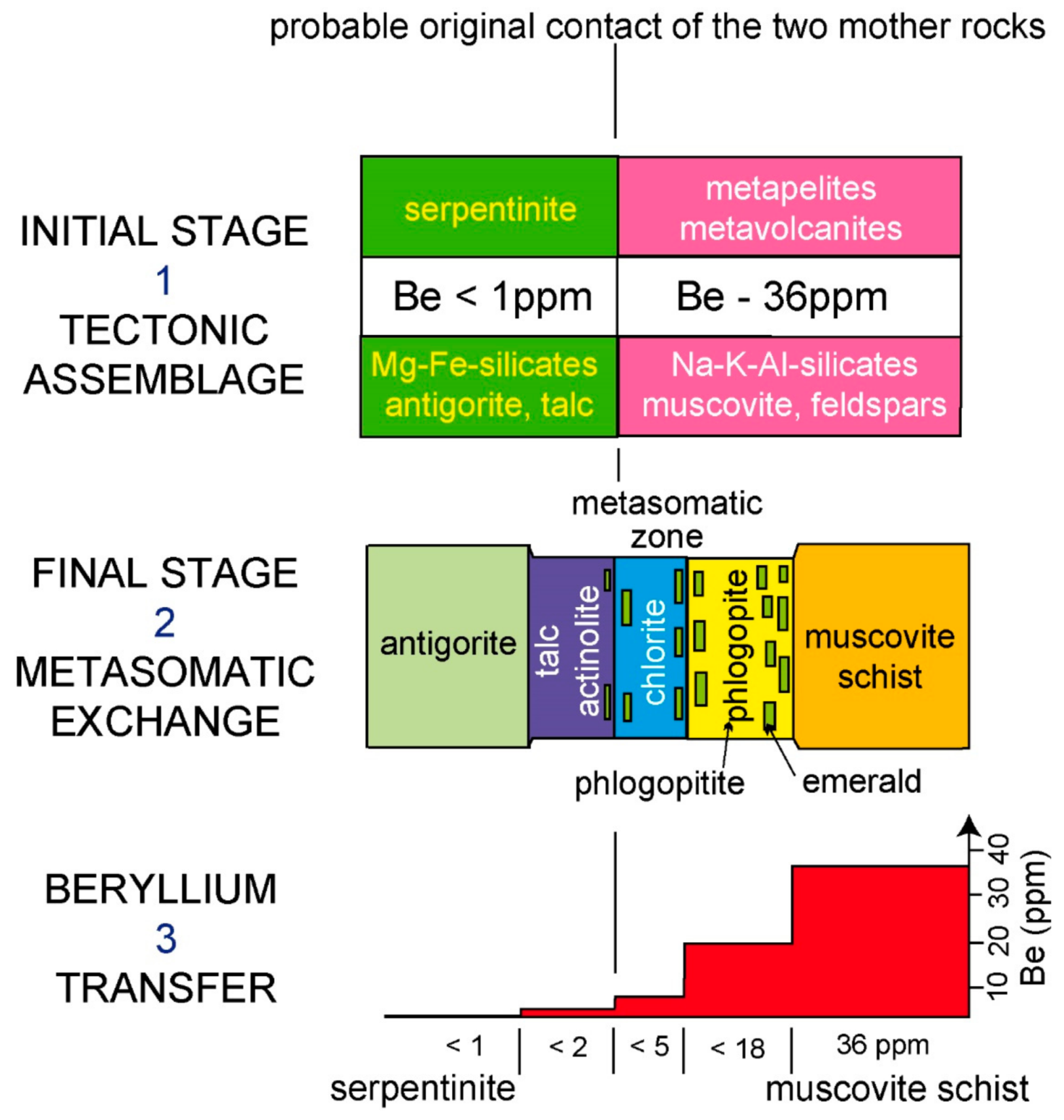
- The Habachtal deposit in the Austrian Alps has been studied in detail [77,93,94,95,96,97]. This alpine deposit is located in a contact zone, which overthrusts the volcano-sedimentary series of Habachtal (Habach Formation) on the ortho-augengneisses (central gneisses). The Paleozoic Habach formation is composed of a series of amphibolites, acid metavolcanics tranformed in muscovite schists, and black pelites with interlayered serpentinites and talc series. Two metamorphic events, one occurring before the Alpine event (P < 3 kb and T < 450 °C) and one occuring during the Alpine event (4.5 < P < 6 kb and 500 < T < 550 °C) were superimposed. The mineralized “blackwall zone”, the equivalent of a phlogopitite, is a tectonic or shear zone 100 m wide, formed from UMR (serpentinites) pinched between orthogneisses and amphibolites. Emerald is disseminated in the “blackwall zone” phlogopitites, talc-actinolite, and chlorite schists. The metasomatic process involves fluid percolation that extracted Be from the muscovite schists (average Be content = 36 ppm) and Cr from the serpentinites (Cr content = 304 ppm) to facilitate the crystallization of emerald (Figure 22). Fluid inclusions trapped by emerald belong to the H2O-CO2-NaCl system [96] with two generations of fluid inclusions: An early generation (XCO2 < 4 vol.%) and a late one (XCO2 up to 11 vol.%). Emerald-metasomatic fluids were related to hydration phenomena due to the alpine metamorphism [96].
- 3.
- The Swat-Mingora–Gujar Kili–Barang deposits are controlled by the Main Mantle Thrust [86,98,99,100]. The suture zone that marks the collision of the Indo-Pakistan plate with the Kohistan arc sequence is composed of a number of fault bounded rock melanges (blueschist, greenschist, and ophiolitic melanges).
6.2.2. Sub-Type IIB: Tectonic Metamorphic-Related Emerald Deposits Hosted in Sedimentary Rocks: Black Shales (Colombia, Canada, USA; Table 2)
- An emerald occurrence was described near Mountain River in the northern Canadian Cordillera [117]. These emerald veins are hosted within siliciclastic strata in the hanging wall of the Shale Lake thrust fault. The emerald formed as a result of inorganic thermal-chemical sulphate reduction via the circulation of deep-seated hydrothermal carbonic brines through basinal siliciclastic, carbonate, and evaporitic rocks (Figure 31). The deep-seated H2O-NaCl-CO2-N2 brines, with a salinity up to 24 wt.% equivalent NaCl, were driven along deep basement structures and reactivated normal faults related to tectonic activity associated with the development of a back-arc basin during the Late Devonian to Middle Mississippian (385–329 Ma). The Mountain River emerald occurrence thus represents a similar and small-scale variation of the Colombian-type emerald deposit model [117].
- Three emeralds were reported in the Uinta Mountains in Utah, USA [118,119]. The discovery was realized in the Neoproterozoic Red Pine shale, which is overlain by Paleozoic carbonate rocks. Based on the study of fibrous calcite hosted by the Mississippian carbonate units, subjacent to the hypothetic emerald-bearing shale, [120] proposed an amagmatic process for the formation of emeralds. The authors combined their chemical and isotope data on calcite, limestone, and the Red Pine shale using a model of formation similar to the Colombian type and involving thermal reduction of sulphates between 100 and 300 °C. Emeralds were not described [120] and one can question their existence in the Uinta Mountains. No one has described these emeralds since the discovery of [118].
6.2.3. Sub-Type IIC: Tectonic Metamorphic-Related Emerald Deposits Hosted in Metamorphic Rocks Other than M-UMR and Black Shales (Afghanistan, China, USA; Table 2)
- The Panjsher emerald deposits in Afghanistan (Figure 1a) are located in the Herat-Panjsher suture zone along the Panjsher Valley. The suture zone, which marks the collision of the Indo-Pakistan plate with the Kohistan arc sequence, contains a number of faults, such as the Herat-Panjsher strike-slip fault, which was mainly active during the Oligocene-Miocene [121]. The emerald deposits lie southeast of the Herat-Panjsher Fault in the Khendj, Saifitchir, and Dest-e-rewat Valleys. The deposits are hosted in the Proterozoic metamorphic basement formed by migmatite, gneiss, schist, marble, and amphibolite. The basement is overlain to the northwest by a Paleozoic metasedimentary sequence crosscut by Triassic granodiorite [86]. During the Oligocene, the Proterozoic rocks of the Panjsher valley were affected by the intrusion of granitoids [86] 20 km north and south of the emerald mining district [122].The emerald deposits are hosted by metamorphic schists that have been affected by intense fracturing, fluid circulation, and hydrothermal alteration, resulting in intense albitization and muscovite-tourmaline replacements [122]. Emerald is found in vugs and quartz veins associated with muscovite, tourmaline, albite, pyrite, rutile, dolomite, and Cl-apatite [122]. Ar-Ar dating on a muscovite from the emerald-bearing quartz veins at the Khendj mine gave an Oligocene age of 23 ± 1 Ma [123]. At the moment, the sources of Cr and Be remain unclear.
- The Davdar emerald deposit is located in the western part of Xinjiang Province, China. The deposit is formed by emerald-bearing quartz-carbonate veins associated with a major northwest-southeast trending fault zone [18]. The deposit is hosted by lower Permian meta-sedimentary rocks, including sandstone, dolomitic limestone, siltstone, and shale, which have been metamorphosed at upper greenschist conditions [124] to produce metasedimentary host rocks, which include quartzite, marble, schist, and phyllite, prior to the emplacement of the emerald-bearing veins. Basaltic dykes of an unknown age, which are up to 10 m wide and crop out along strike lengths of up to 200 m, are the only intrusive igneous rocks known in the area. The dykes are emplaced along the northwest-southeast fault zone; no visible contacts are exposed between the dykes and the emerald-bearing veins.The emerald-bearing veins, which are up to about 20 cm wide, contain epidote, K-feldspar, tourmaline group minerals, carbonates, and iron oxides. Quartz and emerald crystals up to a few centimeters long are found in the veins. Alteration haloes up to a few cm wide occur around the veins. In the sandstone and dolomitic limestone, the alteration halo is barely visible, but it is conspicuous as a bleached white halo in the phyllite. The alteration halo is generally enriched in fine-grained silica with variable amounts of quartz, biotite, muscovite, feldspar, carbonate, and tourmaline. It is representative of a retrograde metamorphic assemblage typical of greenschist facies minerals (epidote, plagioclase, potassic feldspar, quartz, biotite, and chlorite). Emerald typically occurs in the quartz veins and not in the host rocks or the alteration haloes.
- Hiddenite emerald was discovered in North Carolina northeast of the community of Hiddenite in 1875. Since then, a number of notable samples have been discovered, primarily from the Rist and North American Gem mines [125]. Over 3500 carats of emerald were extracted from the latter in the 1980s, including the 858 ct (uncut) “Empress Caroline” crystal [126].At the Rist property the emeralds occur in quartz veins and open cavities (50% of the veins) that occupy NE-trending sub-vertical fractures in folded metamorphic rocks [126,127]. The hiddenite area is underlain by Precambrian migmatitic schists, gneisses, and interlayered calc-silicate rocks, metamorphosed in the upper amphibolite facies. The area is locally intruded by the Rocky face leucogranite.The quartz veins range in size from 2 to 100 cm wide, 30 cm to 7 m long, and 10 cm to 5 m high. Most of the veins are not interconnected and represent tensional gash fractures that sharply crosscut the prominent metamorphic fabric of the host rocks, suggesting that they formed during late or post metamorphic brittle-ductile deformation [126].Wise and Anderson [127] identified four cavity assemblages: (1) An emerald-bearing assemblage composed of albite, beryl, calcite, dolomite, siderite, muscovite, cryptocrystalline quartz, rutile, and sulfides with clays; (2) a Cr-spodumene-bearing assemblage, which includes calcite, muscovite, and quartz. The green Cr-bearing spodumene, locally referred to as “hiddenite”, occurs in only minor amounts; (3) a calcite assemblage dominated by calcite and quartz; and (4) an amethyst assemblage characterized by amethystine quartz, calcite, muscovite, and chabazite. Emerald and spodumene rarely occur together in the same vein or cavity. Within the emerald-bearing cavities, beryl crystals up to 20 cm in length are closely associated with dolomite, muscovite, and quartz. The crystals are typically color-zoned with a pale green to colorless core and an emerald-green rim.Speer [126] described the veins and reported that the emeralds occur as free-standing crystals attached to cavity walls and as individual collapsed fragments. The collapsed crystals that have fallen from walls of the cavities show cementation phenomena, while the attached crystals exhibit dissolution, re-growth, and over-growth. Bleached wall-rock alteration halos up to 9 cm wide and rich in silica and chlorite are commonly peripheral to veins and crystal cavities.Wise and Anderson [127] pointed out that the emerald and Cr-spodumene mineralization in quartz veins and cavities is similar to what is seen in alpine-type fissures. In the absence of a pegmatitic or granitic body, the source of Be and Li remains in question; the source of Cr and V is also uncertain, given that M-UMR are unknown in the area. Speer [126] specified that the veins originated as hydrothermal filling of tensional sites during the waning ductile/brittle stages of metamorphism. Apparently, the geological setting and genesis of the Hiddenite emerald occurrences are unique.
6.2.4. Sub-Type IID: Tectonic Metamorphosed or Remobilized Type IA Deposits, Tectonic Hidden Granitic Intrusion-Related Emerald Deposits, and Some Unclassified Deposits (Egypt, Australia, perhaps also Brazil, Austria, Pakistan, Zambia; Table 2)
- Those for which the genesis is considered to be the consequence of regional metamorphism but with multi-stage emerald formation (Eastern desert of Egypt).
- The sub-type IIA (Santa Terezinha de Goiás, Habachtal, and Swat Valley) where metamorphic-metasomatic deep crustal fluids circulated along faults or shear zones and interacted with M-UMR with apparently no magmatic intrusion.
- The mineralization stages for the Poona deposit where emerald and ruby are associated.
7. Fluid Inclusions in Emerald
8. Discussion of the Model of Formation of the Emerald Deposits Associated with M-UMR
8.1. Sub-Type IA Emerald
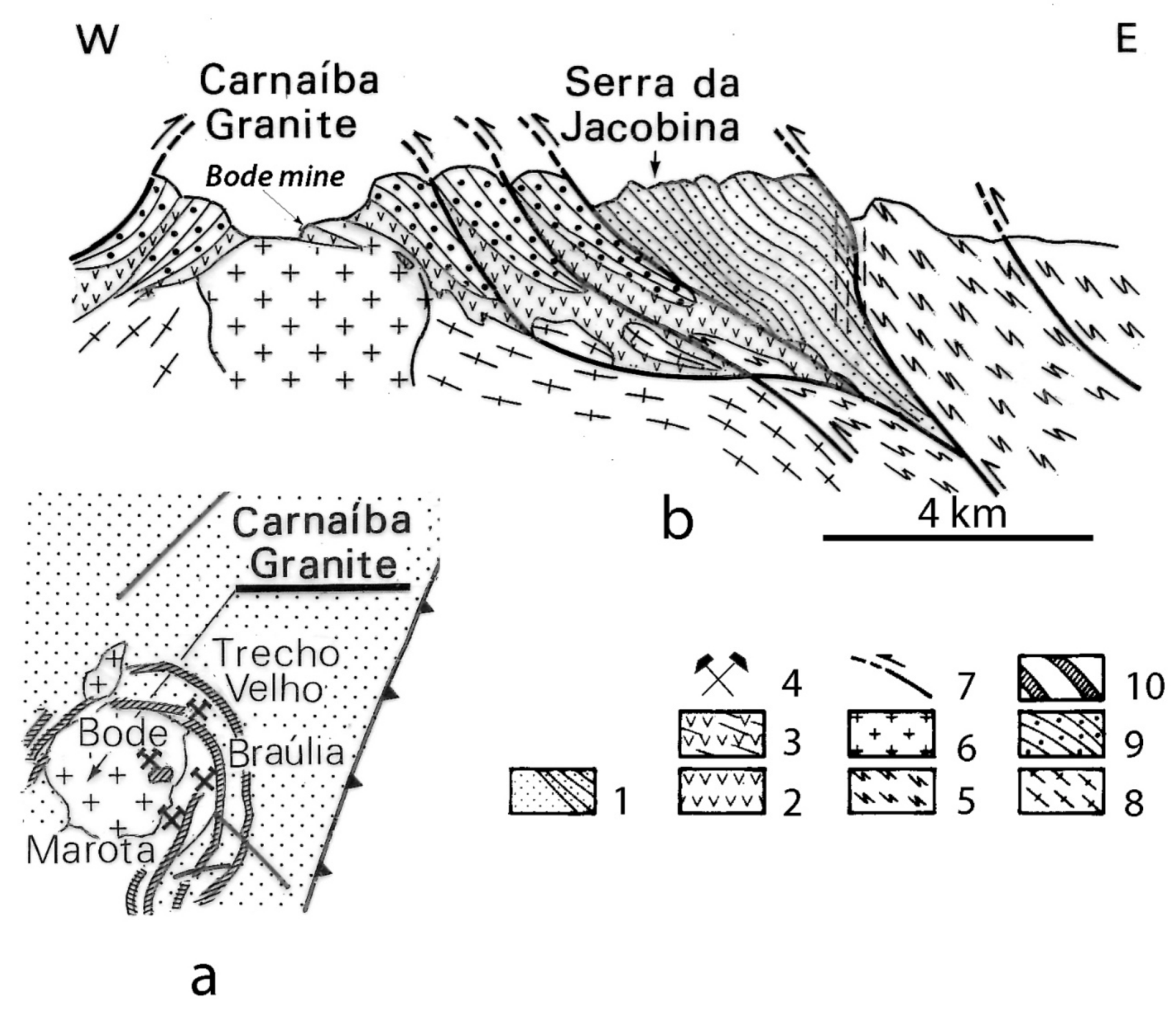
- The granite emplacement is related to a tectonic event, but the pegmatites are not deformed and metamorphosed. They are clearly intruding the M-UMR of the volcano-sedimentary series, and sometimes roof pendants on the granite as observed in Bode mine at Carnaíba (Figure 36) or at Franqueira;
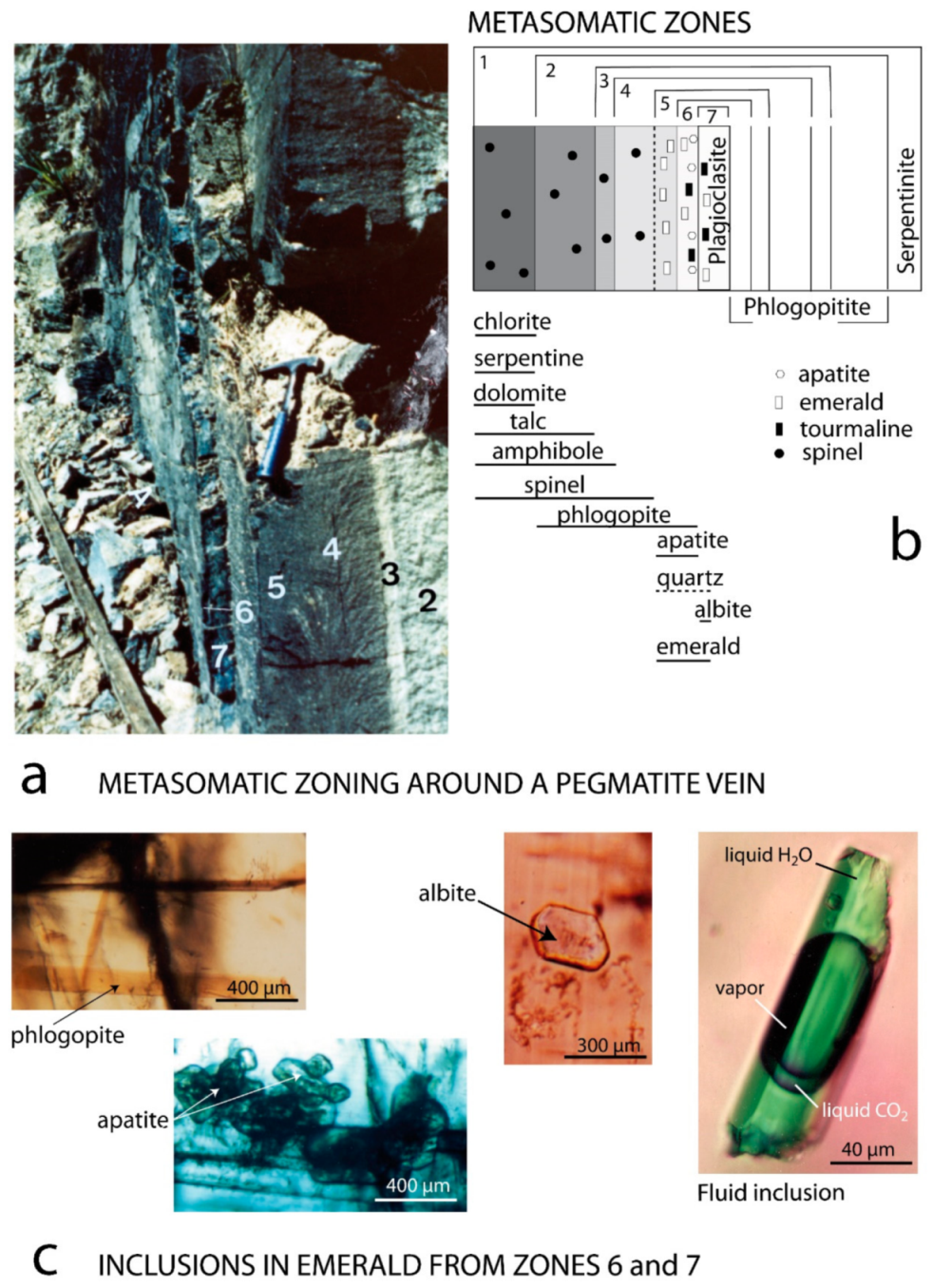
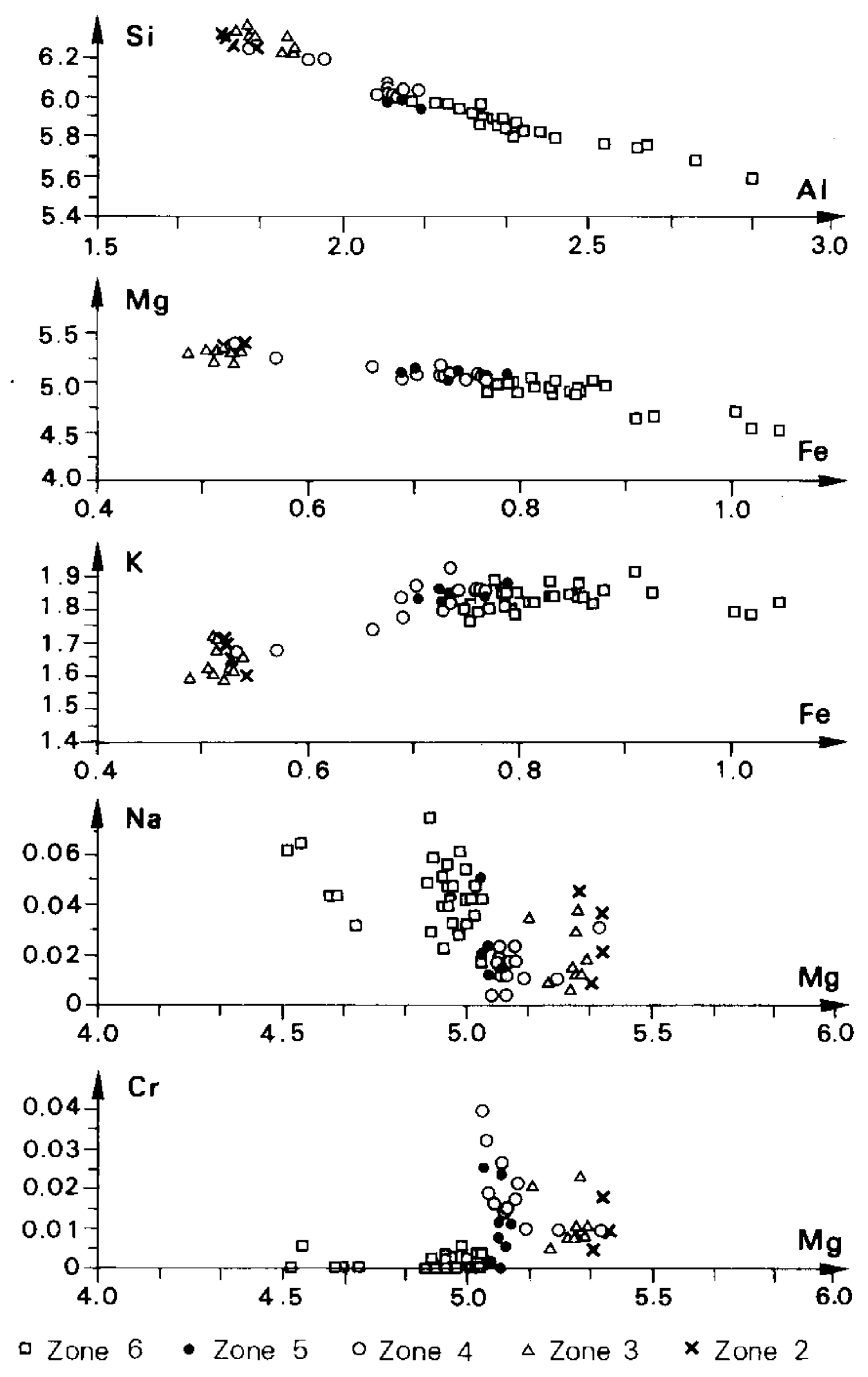
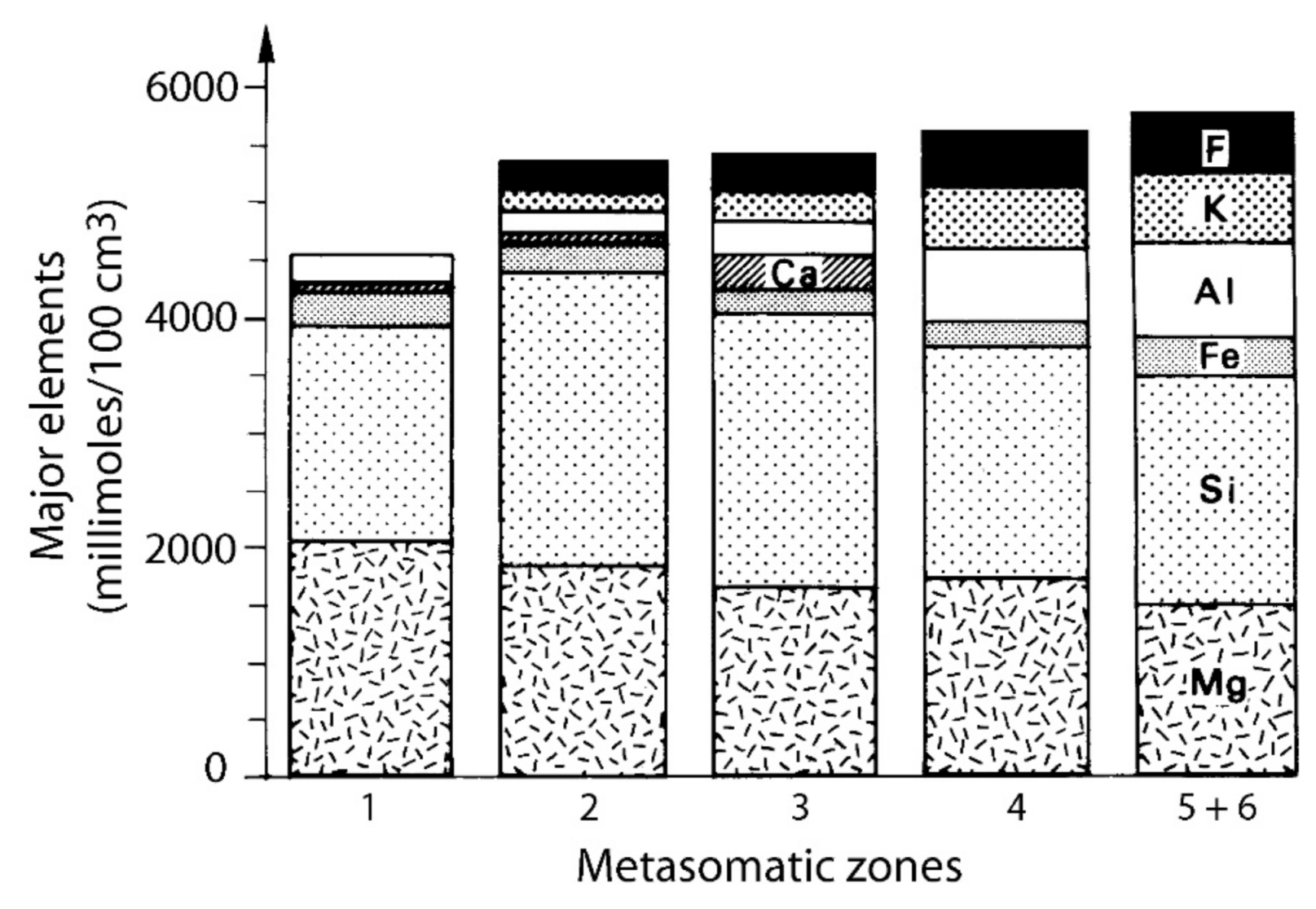
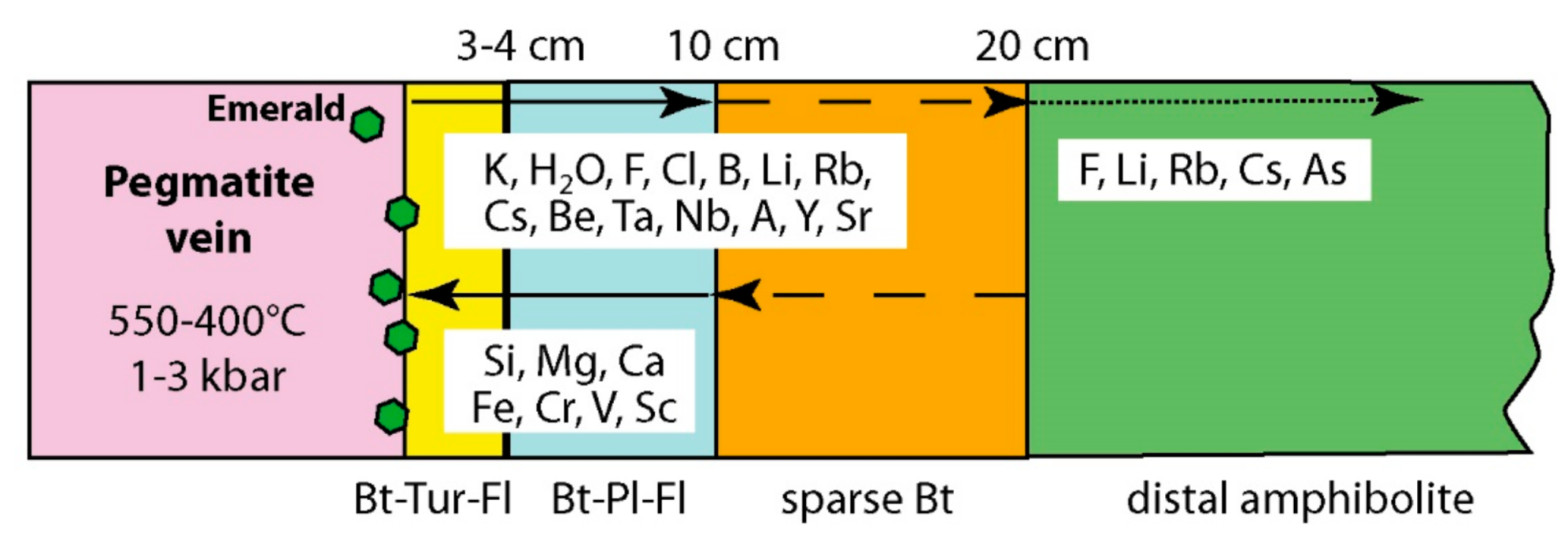
- At Carnaíba, the granite and pegmatite intrusions and fluid circulation are coeval [67], and the pegmatite is transformed into plagioclasite with disseminations of phlogopite (endo-plogopitite), and the M-UMR into phlogopitite (exo-phlogopitites). These exo-phlogopitites display clear zonation with a very sharp metasomatic front, i.e., metasomatic columns formed by infiltrational processes [173]. The metasomatic fronts where an additional phase appears in the mineral association correspond to the change of one of the determinant components from mobile to inert: Ca is displaced from the serpentinites to the center of the column for the formation of actinolite-tremolite, but also apatite at the border of the endo-phlogopitite. The occurrence of beryl is restricted to the most aluminous parts of the metasomatic zonation (plagioclasite, endo-phlogopitite, and exo-phlogopitites proximal to the plagioclasite). The inner part of the phlogopitite zonation plays the role of a filter for the Be-bearing fluids and constitutes a very efficient “metasomatic trap” where the mobile behavior of Cr favored the formation of emerald.At Khaltaro in Pakistan, non-deformed pegmatites and quartz veins crosscut amphibolites, which were metasomatized on 20 cm-wide selvages that are symmetrically zoned around the veins [83], as found at Carnaíba (see Figure 14). Mass-balance calculations on the metasomatic column (Figure 37) have shown that (a) in the inner and intermediate metasomatic zones, K, F, H2O, B, Li, Rb, Cs, Be, Ta, Nb, As, Y, and Sr are gained and Si, Mg, Ca, Fe, Cr, V, and Sc are lost; and (b) in the outer zone, F, Li, Rb, Cs, and As are gained. The oxygen isotope composition of the hydrothermal minerals indicated the circulation of a single fluid of magmatic origin.At Kafubu, the regional metamorphic event pre-dates the emerald formation. The F-B-Li-rich phlogopitites are located at the contact between tourmaline veins and pegmatites with Mg-metabasites. The pegmatites of the Lithium-Cesium-Tantalum family are linked to hidden fertile B-F-Nb-Ta-Li-Cs-rich granite.
- These deposits sometimes exhibit multi-stage Be-mineralization, as observed at the Carnaíba deposit: A second minor stage of metasomatism affected in some areas the emerald-bearing phlogopitites [15,90]. This stage is related to the intrusion of dyke swarms of quartz-muscovite veinlets with greisenisation of the granites, chloritisation, and muscovitisation of the phlogopitites, general silicification, and muscovitisation of the plagioclasites. This stage involves yellowish to whitish beryl, sometimes with molybdenite, scheelite, and schorlite.
- These emerald mineralizations are interpreted to be due to the efficiency of the metasomatic trap rather than significant pre-enrichment in Be (5 to 11 ppm of Be in the Carnaíba granite). The occurrence of strong chemical gradients in the zone of preferential circulation of the solutions constitutes highly favorable conditions for the beryl crystallization.
8.2. Multi-Stage Formation and Ages of Formation and Remobilization of Type IA Deposits
- The Precambrian deposits located in the volcano-sedimentary series or greenstone belts are often folded and sheared, but metasomatic processes during emerald formation are generally coeval with the deformation, as in the deposits of Piteiras, Fazenda Bonfim, and Socotó (Brazil); Sumbawanga and Manyara (Tanzania); Kafubu (Zambia); and recently in the Gubaranda area from Eastern India [174].
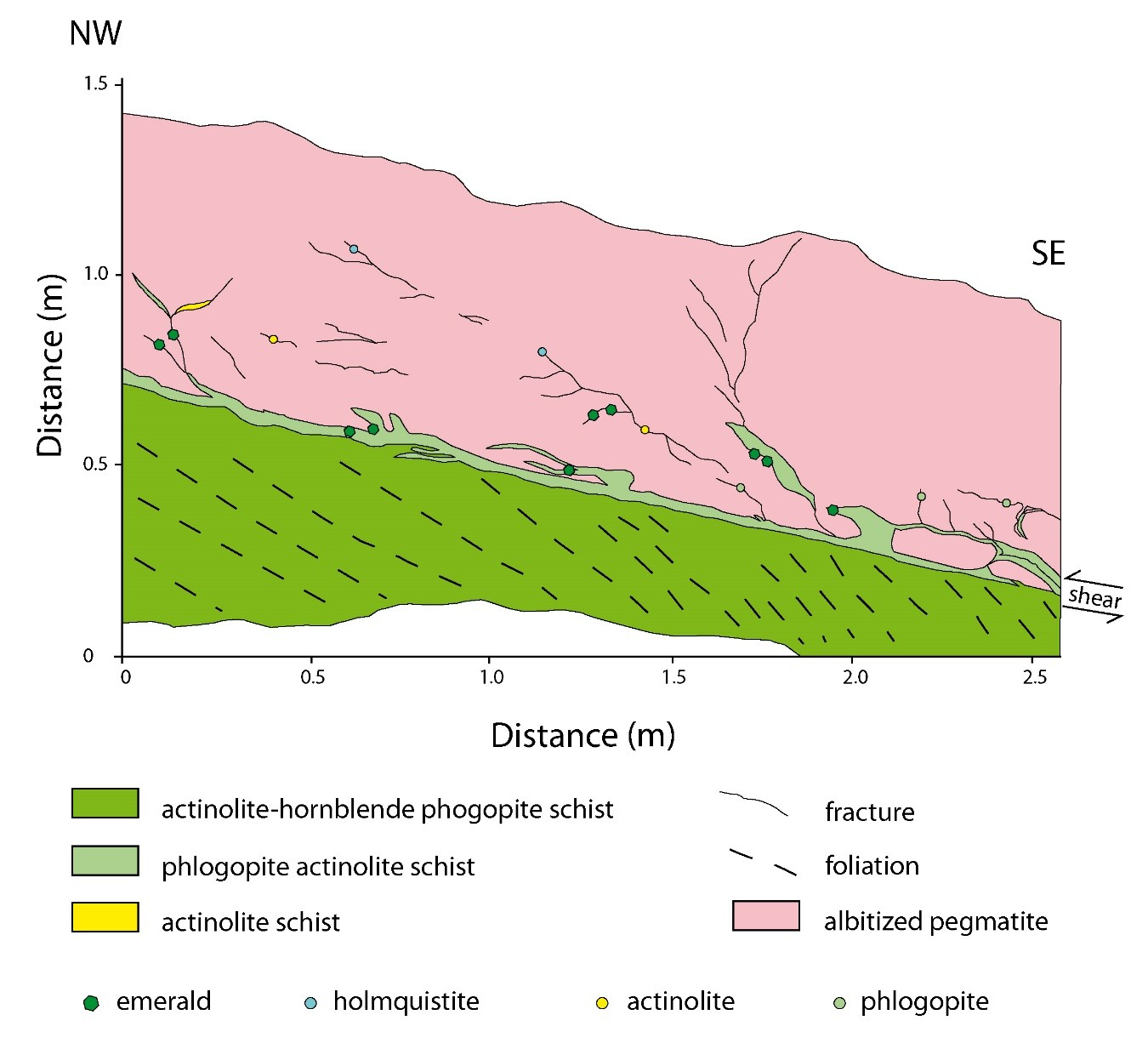
- The deposit at Sandawana in Zimbabwe [52,175] presents multi-stage formation; [169] advanced that the classical model of sub-Type IA cannot be applied. The Cs-Nb-Ta-bearing pegmatite veins that intruded UMR suffered the classical desilication with the formation of plagioclasite. During folding, shearing, and regional metamorphism, after the albitisation of the pegmatites, a reactive F-P-Be-Li-rich fluid of pegmatitic origin circulated in a shear zone, affecting the albitites and reacting with the UMR, to form emerald-bearing phlogopitites (Figure 38). Two generations of emerald are found: (i) Fine-grained crystals at the contact between albitite and phlogopitite and (ii) euhedral gem crystals formed later in phlogopitites either away from the albitite or in low-pressure zones next to the albitites. In that case, the albitites acted as incompetent levels, folded and sheared, and forming traps for euhedral emerald.The syntectonic pegmatites yielded an age of 2640 ± 40 Ma by U/Pb dating on monazite and an age of 2600 ± 100 Ma by the Pb-Pb method on microlite [176]. The 40Ar/39Ar dating on phlogopite and actinolite of the phlogopitites yielded a very disturbed age spectra and variable total gas ages between 2225 and 2447 Ma, with relative plateau ages of 1903 Ma for the phlogopite and of 1936 Ma for the amphibole [52]. Two ages were proposed for the formation of emerald: (i) An Archean age at 2640 Ma, which is the age of the intrusion of the pegmatites, or (ii) a Proterozoic age at around 2000 Ma, which corresponds to a major tectono-metamorphic episode that affected the Limpopo belt formed around the Zimbabwe craton. Zwaan [52] opted for the first hypothesis, considering that the deformation at circa 2000 Ma modified the isotopic argon clock of mica and amphibole, but these integrated Ar-ages between 2200 and 2500 Ma correlate with the Archean thermal event.The complexity of dating rocks that suffered deformational events and remobilization of material illustrates the complexity of classifying ore deposits. The Sandawana deposit belongs to sub-type IA and its genetic link with a magmatic source is obvious in terms of chemical elements, but it could be re-classified as sub-type IID if the age of the emerald is considered to be younger than 2400 Ma.
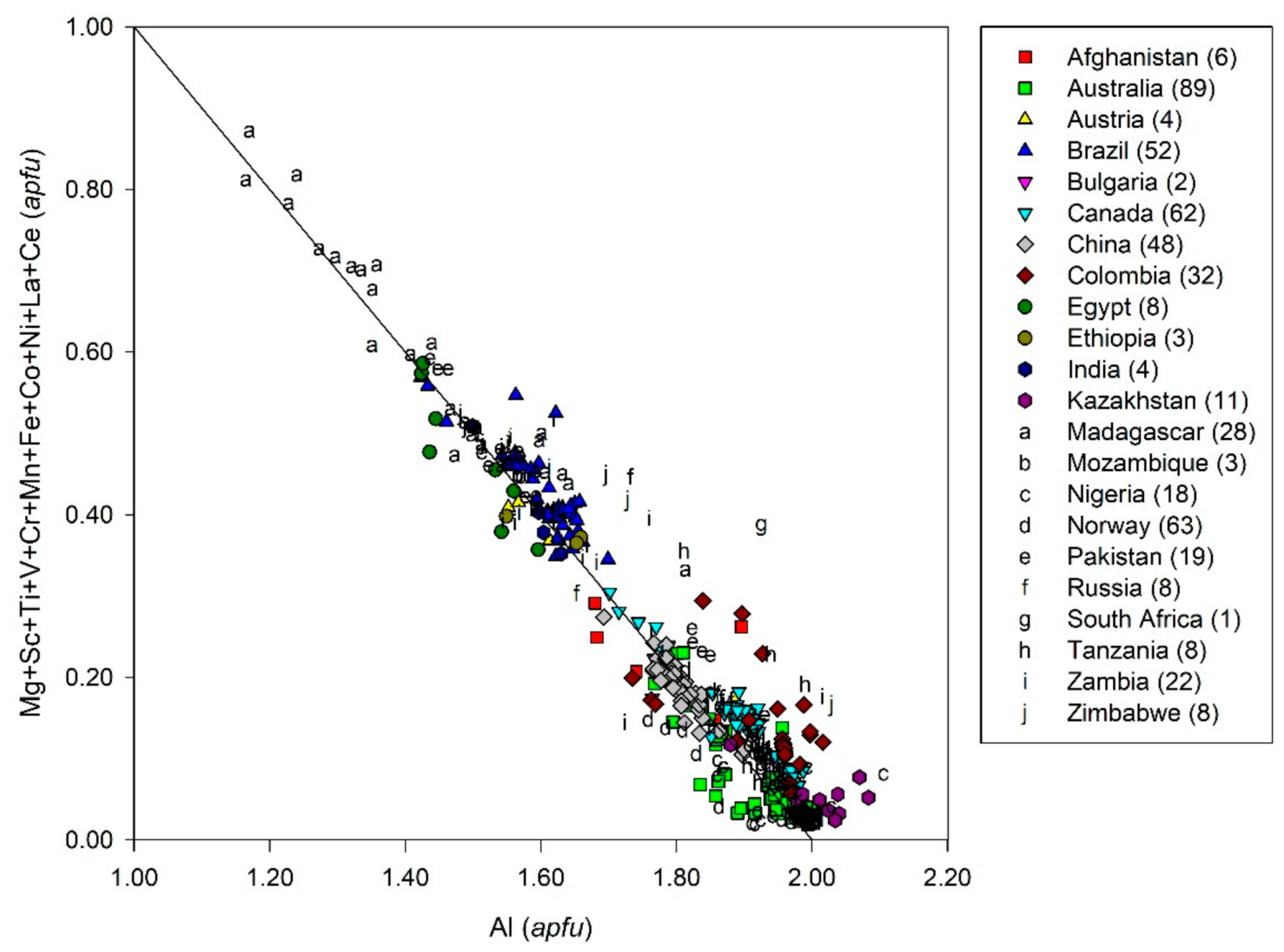
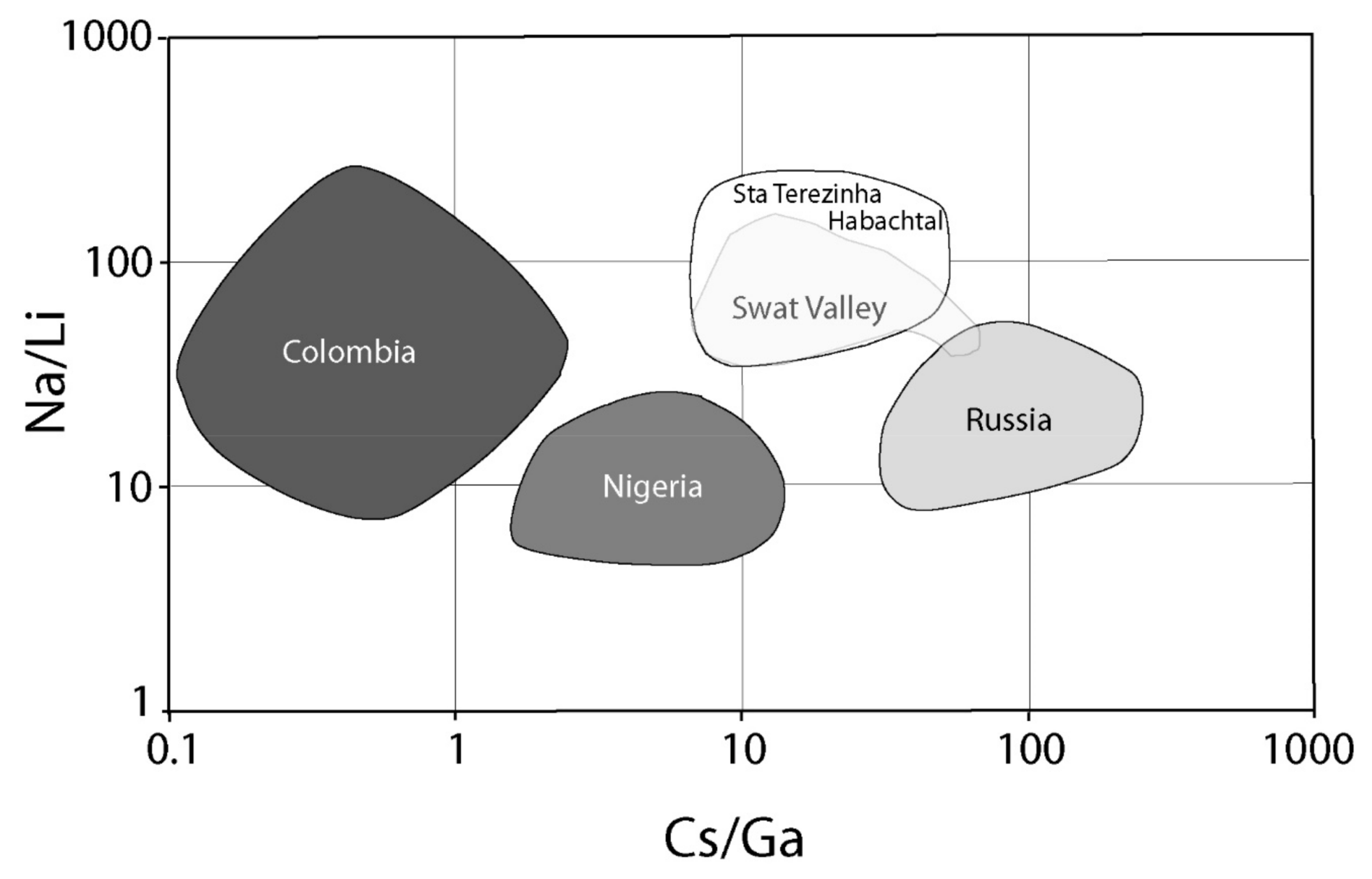
- The possible genesis of sub-type IIA deposits, such as those of Swat Valley, Santa Terezinha de Goiás, and Habachtal [168], was discussed previously. The presence of meta-pegmatites can be suspected based on either chemical data or O-H isotope composition of emerald and associated minerals, but has not been found up to now due to the tectonic regime (thrust and shear zone) and the level of observation. These deposits are classified as sub-type IIC based on the geological environment and are considered to be metamorphic with probable mixing of magmatic fluids (high Cs content for the Santa Terezinha emeralds). This hypothesis is strengthened by the Na/Li vs. Cs/Ga chemical diagram presented by Schwarz [65]. Figure 39 shows that these emerald deposits are grouped in one chemical field very different from those of Colombia, Russia, and Nigeria. They are characterized by a high Cs/Ga ratio, indicating appreciable to high amounts of Cs (magmatic source), and a high Na/Li ratio. The high Na content of this emerald is correlated with a high mean H2O content in the channels, as, determined for Santa Terezinha de Goiás (2.9 wt.%, n = 5), Habachtal (3.1 wt.%, n = 3), and Swat Valley (3.4 wt.%, n = 1) [15,16]. This is not just a coincidence, but is probably a genetic proxy Be-Cs source for emerald in these three deposits, i.e., magmatic sources with huge fluid circulation and metasomatism in a metamorphic environment.The Habachtal deposit is a complex deposit in terms of the genetic model and the previous discussion about the possible remobilization of Be-W enriched Hercynian pegmatites (possible sub-type IID deposit) by the regional metamorphism [168] opens the debate on the age of the formation of this deposit. The genesis of the emerald is metasomatic, but bound to the regional metamorphism of alpine age [77]. The K-Ar age obtained on phlogopite from the phlogopitites is 22 Ma, while the tracks of fission on apatite yielded ages of 9 Ma. The K-Ar age on muscovite from the muscovite schist is 27 Ma. The Rb/Sr dating realized on the zones of growth of garnets from the Schieferhülle formation, situated structurally above the Habach formation, indicated ages of crystallization between 62.0 and 30.2 Ma [177]. This age around 30 Ma is in agreement with the dates found for the end of the growth of garnet in the central Alps [178]. So, the best estimation established for the growth of the Habachtal emeralds would be situated around 30 Ma [179].
- The deposits of the Eastern desert in Egpyt and Poona in Australia are good examples of sub-type IID, where sub-type IA deposits were remobilized by regional metamorphism with deformation and remobilization of older rocks, following the genetic model proposed by Grundmann and Morteani [77].
9. Exploration Now and in the Future
10. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Groat, L.; Giuliani, G.; Marshall, D.; Turner, D. Emerald. In Geology of Gem Deposits; Raeside, E.R., Ed.; Mineralogical Association of Canada, Short Course Series 44: Tucson, AZ, USA, 2014; pp. 135–174. [Google Scholar]
- In the News. Gemfields’ October emerald auction: US$21.5 million in sales. Incolor 2018, 37, 22. [Google Scholar]
- Schmidt, S. Une des plus grandes émeraudes au monde a été découverte dans une mine en Zambie. Trust My Sicence, Nature. 2018. Available online: https://trustmyscience.com (accessed on 11 February 2019).
- Ibbetson, R. Miner finds giant 5,655-carat emeraldin Zambia worth up to £ 2m. Daily Mail Online, 30 October 2018. [Google Scholar]
- Yager, Y.G.; Menzie, W.D.; Olson, D.W. Weight of Production of Emeralds, Rubies, Sapphires, and Tanzanite from 1995 through 2005; Open File Report 2008–1013; USGS Reston: Reston, VA, USA, 2008; 9p.
- Wood, D.L.; Nassau, K. Characterization of beryl and emerald by visible and infrared absorption spectroscopy. Am. Mineral. 1968, 53, 777–800. [Google Scholar]
- Damon, P.E.; Kulp, J.L. Excess helium and argon in beryl and other minerals. Am. Mineral. 1958, 43, 433–459. [Google Scholar]
- Giuliani, G.; Marty, B.; Banks, D. Noble gases in fluid inclusions from emeralds: Implications for the origins of fluids and constraints on fluid-rock interactions. In Proceedings of the 18th Biennial Meeting of European Current Research on Fluid Inclusions, Siena, Italy, 4–5 July 2005. CD-ROM. [Google Scholar]
- Charoy, B. Cristallochimie du béryl: L’état des connaissances. In L’Émeraude; Giard, D., Giuliani, G., Cheilletz, A., Fritch, E., Gonthier, E., Eds.; AFG-CNRS-ORSTOM Edition: Paris, France, 1998; pp. 47–54. [Google Scholar]
- Schwarz, D.; Schmetzer, K. The definition of emerald: The green variety of beryl colored by chromium and/or vanadium. In Emeralds of the World, (2002) ExtraLapis English No. 2: The Legendary Green Beryl; Lapis International, LLC: East Hampton, CT, USA, 2002; pp. 74–78. [Google Scholar]
- Andrianjakavah, P.R.; Salvi, S.; Béziat, D.; Rakotondrazafy, A.F.M.; Giuliani, G. Proximal and distal styles of pegmatite-related metasomatic emerald mineralization at Ianapera, southern Madagascar. Miner. Deposita 2009, 44, 817–835. [Google Scholar] [CrossRef]
- Zwaan, J.C.; Jacob, D.E.; Häger, T.; Calvacanti Neto, M.T.O.; Kanis, J. Emeralds from the Fazenda Bonfim region, Rio Grande do Norte, Brazil. Gems Gemol. 2012, 48, 2–17. [Google Scholar] [CrossRef]
- Rondeau, B.; Fritsch, E.; Peucat, J.J.; Nordru, F.S.; Groat, L.A. Characterization of emeralds from a historical deposit: Byrud (Eidsvoll), Norway. Gems Gemol. 2008, 44, 108–122. [Google Scholar] [CrossRef]
- Artioli, G.; Rinaldi, R.; Stahl, K.; Zanazzi, P.F. Structure refinements of beryl by single-crystal neutron and X-ray diffraction. Am. Mineral. 1993, 78, 762–768. [Google Scholar]
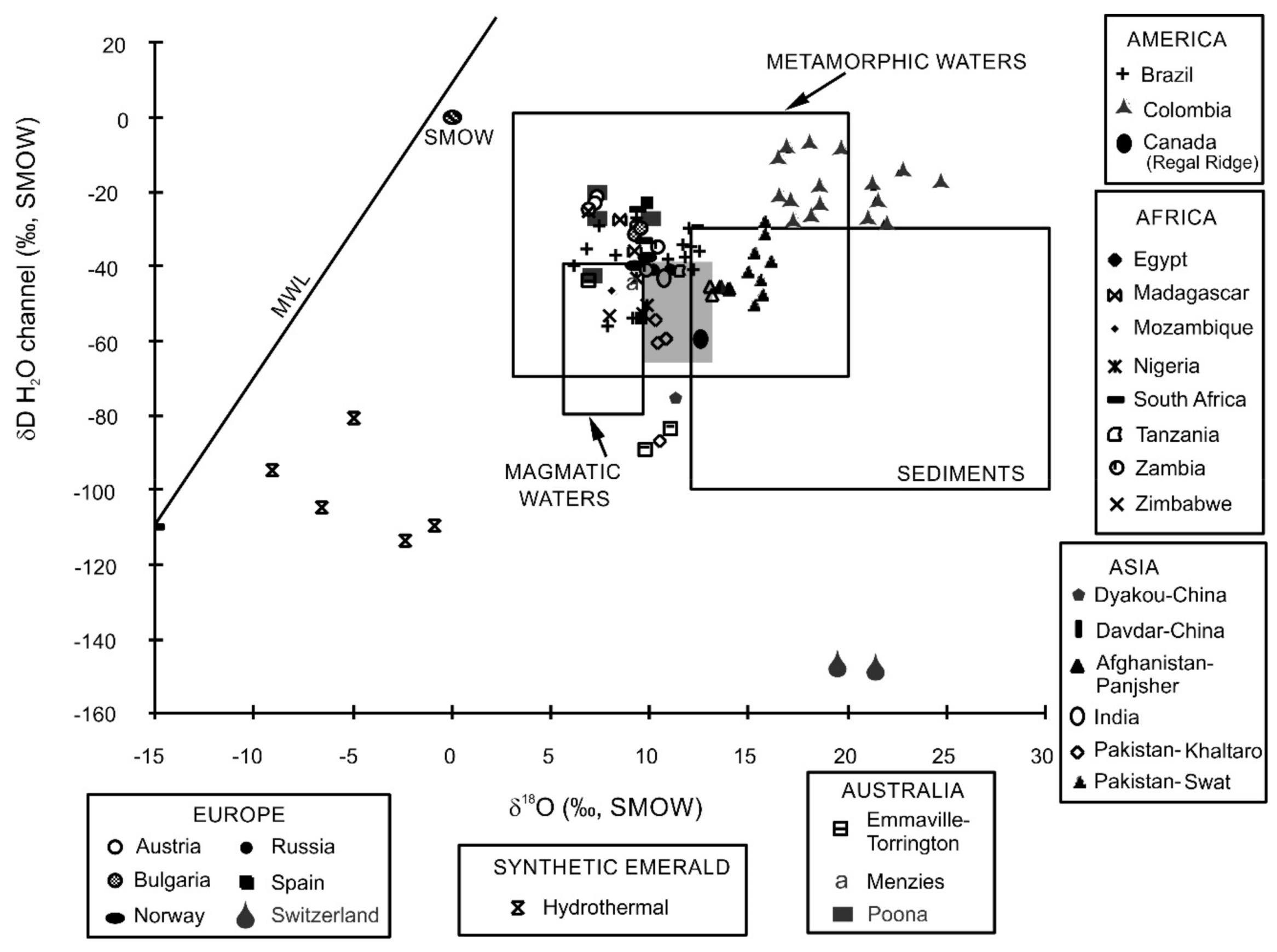
- Giuliani, G.; France-Lanord, C.; Zimmermann, J.-L.; Cheilletz, A.; Arboleda, C.; Charoy, B.; Coget, P.; Fontan, F.; Giard, D. Composition of fluids, δD of channel H2O and δ18O of lattice oxygen in beryls: Genetic implications for Brazilian, Colombian and Afghanistani emerald deposits. Int. Geol. Rev. 1997, 39, 400–424. [Google Scholar] [CrossRef]
- Marshall, D.; Downes, P.J.; Ellis, S.; Greene, R.; Loughrey, L.; Jones, P. Pressure–temperature-fluid constraints for the Poona emerald deposits, Western Australia: Fluid inclusion and stable isotope studies. Minerals 2016, 6, 130. [Google Scholar] [CrossRef]
- Brand, A.A.; Groat, L.A.; Linnen, R.L.; Garland, M.I.; Breaks, F.W.; Giuliani, G. Emerald mineralization associated with the Mavis Lake Pegmatite Group, near Dryden, Ontario. Can. Mineral. 2009, 47, 315–336. [Google Scholar] [CrossRef]
- Marshall, D.; Pardieu, V.; Loughrey, L.; Jones, P.; Xue, G. Conditions for emerald formation at Davdar, China: Fluid inclusion, trace element and stable isotope studies. Mineral. Mag. 2012, 76, 213–226. [Google Scholar] [CrossRef]
- Loughrey, L.; Marshall, D.; Jones, P.; Millsteed, P.; Main, A. Pressure-temperature-fluid constraints for the Emmaville-Torrington emerald deposit, New South Wales, Australia: Fluid inclusion and stable isotope studies. Cent. Eur. J. Geosci. 2012, 4, 287–299. [Google Scholar] [CrossRef]
- Loughrey, L.; Marshall, D.; Ihlen, P.; Jones, P. Boiling as a mechanism for colour zonations observed at the Byrud emerald deposit, Eidsvoll, Norway: Fluid inclusion, stable isotope and Ar-Ar studies. Geofluids 2013, 13, 542–558. [Google Scholar] [CrossRef]
- Lake, D.J.; Groat, L.; Falck, H.; Cempirek, J.; Kontak, D.; Marshall, D.; Giuliani, G.; Fayek, M. Genesis of emerald-bearing quartz veins associated with the Lened W-skarn mineralization, northwest territories, Canada. Can. Mineral. 2017, 55, 561–593. [Google Scholar] [CrossRef]
- Santiago, J.S.; Souza, V.S.; Filgueiras de, B.C.; Jiménez, F.A.C. Emerald from the Fazenda Bonfim deposit, northeastern Brazil: Chemical, fluid inclusions and oxygen isotope data. Braz. J. Geol. 2018, 1–16. [Google Scholar] [CrossRef]
- Aurisicchio, C.; Conte, A.M.; Medeghini, L.; Ottolini, L.; De Vito, C. Major and trace element geochemistry of emerald from several deposits: Implications for genetic models and classification schemes. Ore Geol. Rev. 2018, 94, 351–366. [Google Scholar] [CrossRef]
- Kovaloff, P. Geologist’s report on Somerset emeralds. S. Afr. Mining Eng. J. 1928, 39, 101–103. [Google Scholar]
- Zambonini, F.; Caglioto, V. Ricerche chimiche sulla rosterite di San Piero in Campo (Isola d’Elba) e sui berilli in generale. Gazz. Chim. Ital. 1928, 58, 131–152. [Google Scholar]
- Leitmeier, H. Das Smaragdvorkommen in Habachtal in Salzburg und seine Mineralien. Tscher. Miner. Petrog. 1937, 49, 245–368. [Google Scholar] [CrossRef]
- Otero Muñoz, G.; Barriga Villalba, A.M. Esmeraldas de Colombia; Banco de la Republica: Bogotá, Colombia, 1948. [Google Scholar]
- Simpson, E.S. Minerals of Western Australia; Government Printer: Perth, Australia, 1948; Volume 1, pp. 195–207.
- Gübelin, E.J. Emeralds from Sandawana. J. Gemmol. 1958, 6, 340–354. [Google Scholar] [CrossRef]
- Vlasov, K.A.; Kutakova, E.I. Izumrudnye Kopi; Moscow Akademiya Nauk SSGR: Moscow, Russia, 1960; 252p. [Google Scholar]
- Martin, H.J. Some observations on southern Rhodesian emeralds and chrysoberyls. Chamb. Mines J. 1962, 4, 34–38. [Google Scholar]
- Petrusenko, S.; Arnaudov, V.; Kostov, I. Emerald pegmatite from the Urdini Lakes, Rila Mountains. Annuaire de l’Université de Sofia Faculté de Géologie et Géographie 1966, 59, 247–268. [Google Scholar]
- Beus, A.A.; Mineev, D.A. Some Geological and Geochemical Features of the Muzo-Coscuez Emerald Zone, Cordillera Oriental, Colombia; Empresa Colombiana de Minas, Biblioteca: Bogotá, Colombia, 1972; 55p. [Google Scholar]
- Hickman, A.C.J. The Miku emerald deposit. Geol. Surv. Zambia Econ. Rep. 1972, 27, 35. [Google Scholar]
- Garstone, J.D. The geological setting and origin of emeralds from Menzies, Western Australia. J. R. Soc. West. Aust. 1981, 64, 53–64. [Google Scholar]
- Hanni, H.A.; Klein, H.H. Ein Smaragdvorkommen in Madagaskar. Zeitschrift der Deutschen Gemmologischen Gesellschaft 1982, 21, 71–77. [Google Scholar]
- Graziani, G.; Gübelin, E.; Lucchesi, S. The genesis of an emerald from the Kitwe District, Zambia. Neues Jb. Miner. Monat. 1983, 175–186. [Google Scholar]
- Kozlowski, A.; Metz, P.; Jaramillo, H.A.E. Emeralds from Sodomondoco, Colombia: Chemical composition, fluid inclusion and origin. Neues Jb. Miner. Abh. 1988, 59, 23–49. [Google Scholar]
- Hammarstrom, J.M. Mineral chemistry of emeralds and some minerals from Pakistan and Afghanistan: An electron microprobe study. In Emeralds of Pakistan; Kazmi, A.H., Snee, L.W., Eds.; Van Nostrand Reinhold: New York, NY, USA, 1989; pp. 125–150. [Google Scholar]
- Ottaway<sc>, T.L. The Geochemistry of the Muzo Emerald Deposit, Colombia. M.Sc. Thesis, University of Toronto, Toronto, ON, Canada, 1991. [Google Scholar]
- Schwarz, D. Australian emeralds. Austral. Gemmol. 1991, 17, 488–497. [Google Scholar]
- Schwarz, D.; Kanis, J.; Kinnaird, J. Emerald and green beryl from central Nigeria. J. Gemmol. 1996, 25, 117–141. [Google Scholar] [CrossRef]
- Abdalla, H.M.; Mohamed, F.H. Mineralogical and geochemical investigations of beryl mineralizations, Pan-African belt of Egypt: Genetic and exploration aspects. J. Afr. Earth Sci. 1999, 28, 581–598. [Google Scholar] [CrossRef]
- Gavrilenko, E.V.; Pérez, B.C. Characterisation of emeralds from the Delbegetey deposit, Kazakhstan. In Mineral Deposits: Processes to Processing; Stanley, C.J., Rankin, A.H., Bodnar, R.J., Naden, J., Yardley, B.W.D., Criddle, A.J., Hagni, R.D., Gize, A.P., Pasava, J.., Eds.; Balkema: Rotterdam, The Netherlands, 1999; pp. 1097–1100. [Google Scholar]
- Alexandrov, P.; Giuliani, G.; Zimmerman, J.L. Mineralogy, age and fluid geochemistry of the Rila Emerald deposits, Bulgaria. Econ. Geol. 2001, 96, 1469–1476. [Google Scholar] [CrossRef]
- Groat, L.A.; Marshall, D.D.; Giuliani, G.; Murphy, D.C.; Piercey, S.J.; Jambor, J.L.; Mortensen, J.K.; Ercit, T.S.; Gault, R.A.; Mattey, D.P.; et al. Mineralogical and geochemical study of the Regal Ridge showing emeralds, southeastern Yukon. Can. Mineral. 2002, 40, 1313–1338. [Google Scholar] [CrossRef]
- Marshall, D.D.; Groat, L.A.; Falck, H.; Douglas, H.L.; Giuliani, G. Fluid inclusions from the Lened emerald occurrence; Northwest Territories, Canada: Implications for Northern Cordilleran Emeralds. Can. Mineral. 2004, 42, 1523–1539. [Google Scholar] [CrossRef]
- Vapnik, Y.; Sabot, B.; Moroz, I. Fluid inclusions in Ianapera emerald, Southern Madagascar. Int. Geol. Rev. 2005, 47, 647–662. [Google Scholar] [CrossRef]
- Vapnik, Y.; Moroz, I.; Eliezri, I. Formation of emeralds at pegmatite-ultramafic contacts based on fluid inclusions in Kianjavato emerald, Mananjary deposits, Madagascar. Mineral. Mag. 2006, 70, 141–158. [Google Scholar] [CrossRef]
- Zwaan, J.C.; Seifert, A.V.; Vrána, S.; Laurs, B.M.; Anckar, B.; Simons, W.B.S.; Falster, A.U.; Lustenhouwer, W.J.; Muhlmeister, S.; Koivula, J.K.; et al. Emeralds from the Kafubu area, Zambia. Gems Gemol. 2005, 41, 116–148. [Google Scholar] [CrossRef]
- Gavrilenko, E.V.; Calvo Pérez, B.; Castroviejo Bolibar, R.; Garcia Del Amo, D. Emeralds from the Delbegetey deposit (Kazakhstan): Mineralogical characteristics and fluid-inclusion study. Mineral. Mag. 2006, 70, 159–173. [Google Scholar] [CrossRef]
- Zwaan, J.C. Gemmology, geology and origin of the Sandawana emerald deposits, Zimbabwe. Scr. Geol. 2006, 131, 211. [Google Scholar]
- Barton, M.D.; Young, S. Nonpegmatitic deposits of beryllium: Mineralogy, geology, phase equilibria and origin. In Beryllium: Mineralogy, Petrology, and Geochemistry; Grew, E.S., Ed.; Mineralogical Society of America: Washington, DC, USA, 2002; Volume 50, pp. 591–691. [Google Scholar]
- Giuliani, G.; Bourlès, D.; Massot, J.; Siame, L.L. Colombian emerald reserves inferred from leached beryllium of their host black shale. Explor. Min. Geol. 1999, 8, 109–116. [Google Scholar]
- Giuliani, G.; Branquet, Y.; Fallick, A.E.; Groat, L.; Marshall, D. Emerald deposits around the world, their similarities and differences. InColor 2015, special issue, 56–69. [Google Scholar]
- Sabot, B. Classification des gisements d’émeraude: Apports des études pétrographiques, minéralogiques et géochimiques. Thèse de Doctorat, Université de Nancy, Nancy, France, 2002. [Google Scholar]
- Dereppe, J.M.; Moreaux, C.; Chauvaux, B.; Schwarz, D. Classification of emeralds by artificial neural networks. J. Gemmol. 2000, 27, 93–105. [Google Scholar] [CrossRef]
- Schwarz, D.; Giuliani, G. Emerald deposits—A review. Austral. Gemmol. 2001, 21, 17–23. [Google Scholar]
- Schwarz, D.; Giuliani, G.; Grundmann, G.; Glas, M. Die Entstehung der Smaragde, ein vieldisskutiertes Thema. In Smaragd, der Kostbarste Beryll, der Teuerste Edelstein; Schwarz, D., Hochlitner, R., Eds.; ExtraLapis: Charleston, SC, USA, 2001; pp. 68–73. [Google Scholar]
- Schwarz, D.; Giuliani, G.; Grundmann, G.; Glas, M. The origin of emerald—A controversial topic. ExtraLapis Engl. 2002, 2, 18–21. [Google Scholar]
- Schwarz, D.; Klemm, L. Chemical signature of emerald. Int. Geol. Congr. Abstr. 2012, 34, 2812. [Google Scholar]
- Giuliani, G.; Groat, L.; Ohnenstetter, D.; Fallick, A.E.; Feneyrol, J. The geology of gems and their geographic origin. In Geology of Gem Deposits; Raeside, E.R., Ed.; Mineralogical Association of Canada, Short Course Series 44: Tucson, AZ, USA, 2014; pp. 113–134. [Google Scholar]
- Saeseasaw, S.; Pardieu, V.; Sangsawong, S. Three-phase inclusions in emerald and their impact on origin determination. Gems Gemol. 2014, 50, 114–132. [Google Scholar]
- Ochoa, C.J.C.; Daza, M.J.H.; Fortaleche, D.; Jiménez, J.F. Progress on the study of parameters related to the origin of Colombian emeralds. InColor 2015, special issue, 88–97. [Google Scholar]
- Schwarz, D. The geographic origin determination of emeralds. InColor 2015, special issue, 98–105. [Google Scholar]
- Hainschwang, T.; Notari, F. Standards and protocols for emerald analysis in gem testing laboratories. InColor 2015, special issue, 106–114. [Google Scholar]
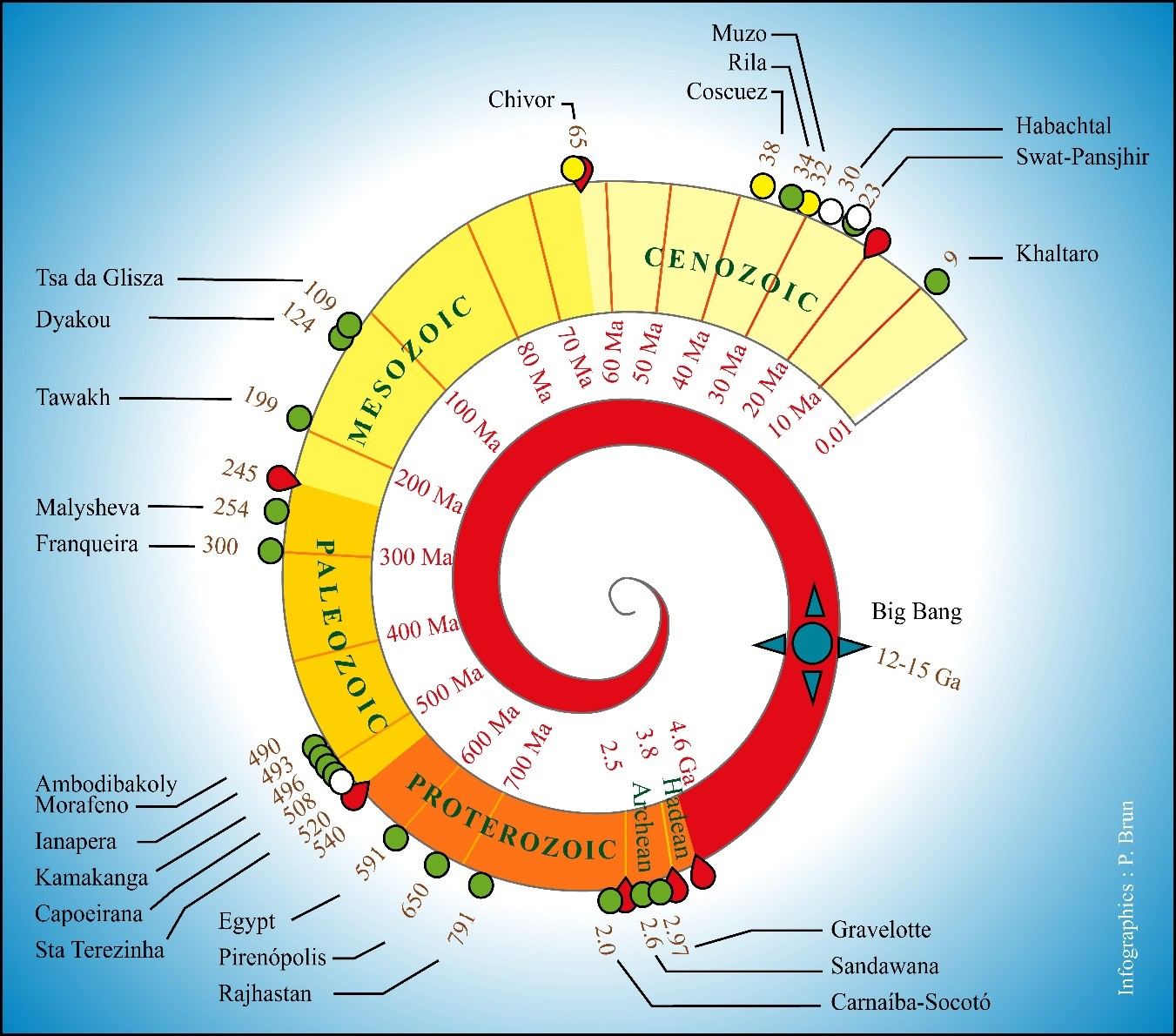
- Giuliani, G. La spirale du temps de l’émeraude. Règne Minéral 2011, 98, 31–44. [Google Scholar]
- Kinnaird, J.A. Hydrothermal alteration and mineralization of the alkaline anorogenic ring complexes of Nigeria. J. Afr. Earth Sci. 1985, 3, 229–251. [Google Scholar] [CrossRef]
- Larsen, B.T.; Olaussen, S.; Sundwoll, B.; Heeremans, M. The Permo-Carboniferous Oslo Rift through six stages and 65 million years. Episodes 2008, 31, 52–58. [Google Scholar]
- Seifert, A.V.; Žaček, V.; Vrána, S.; Pecina, V.; Zachariáš, J.; Zwaan, J.C. Emerald mineralization in the Kafubu area, Zambia. Bull. Geosci. 2004, 79, 1–40. [Google Scholar]
- Bastos, F.M. Emeralds from Itabira, Minas Gerais, Brazil. Lapid. J. 1981, 35, 1842–1848. [Google Scholar]
- Hänni, H.A.; Schwarz, D.; Fischer, M. The emeralds of the Belmont mine, Minas Gerais, Brazil. J. Gemmol. 1987, 20, 446–456. [Google Scholar] [CrossRef]
- de Souza, J.L.; Mendes, J.C.; da Silveira Bello, R.M.; Svisero, D.P.; Valarelli, J.V. Petrographic and microthermometrical studies of emeralds in the “Garimpo” of Capoeirana, Nova Era, Minas Gerais State, Brazil. Miner. Deposita 1992, 27, 161–168. [Google Scholar] [CrossRef]
- Rondeau, B.; Notari, F.; Giuliani, G.; Michelou, J.-C.; Martins, S.; Fritsch, E.; Respinger, A. La mine de Piteiras, Minas Gerais, nouvelle source d’émeraude de belle qualité au Brésil. Rev. Gemmol. AFG 2003, 148, 9–25. [Google Scholar]
- Walton, L. Exploration Criteria for Colored Gemstone Deposits in Yukon; Open file 2004-10; Geological Survey of Canada: Ottawa, ON, Canada, 2004; 184p.
- Giuliani, G.; Cheilletz, A.; Zimmermann, J.-L.; Ribeiro-Althoff, A.M.; France-Lanord, C.; Féraud, G. Les gisements d’émeraude du Brésil: Genèse et typologie. Chron. Rech. Min. BRGM 1997, 526, 17–60. [Google Scholar]
- Grundmann, G.; Morteani, G. Emerald mineralization during regional metamorphism: The Habachtal (Austria) and Leydsdorp (Transvaal, South Africa) deposits. Econ. Geol. 1989, 84, 1835–1849. [Google Scholar] [CrossRef]
- Rudowski, L.; Giuliani, G.; Sabaté, P. Les phlogopitites à émeraude au voisinage des granites de Campo Formoso et Carnaíba (Bahia, Brésil): Un exemple de minéralisation protérozoïque à Be, Wo et W dans les ultrabasiques métasomatisées. C. R. Acad. Sci. 1987, 304, 1129–1134. [Google Scholar]
- Rudowski, L. Pétrologie et géochimie des granites transamazoniens de Campo Formoso et Carnaíba (Bahia, Brésil) et des phlogopitites à émeraude associées. Thèse de Doctorat, Université Paris VI, Paris, France, 1989. [Google Scholar]
- Muñoz, J.L. F-OH and Cl-OH exchange in micas with applications to hydrothermal ore deposits. In Micas; Bailey, S.W., Ed.; Mineralogical Society of America: Washington, DC, USA, 1984; Volume 13, pp. 469–493. [Google Scholar]
- Guy, B. Contribution à l’étude des skarns de Costabonne (Pyrénées orientales, France) et à la théorie de la zonation métasomatique. Ph.D. Thesis, Université de Paris VI, Paris, France, 1979. [Google Scholar]
- Korzhinskii, D.S. The theory of systems with perfectly mobile components and processes of mineral formation. Am. J. Sci. 1965, 263, 193–205. [Google Scholar] [CrossRef]
- Laurs, B.M.; Dilles, J.H.; Snee, L.W. Emerald mineralization and metasomatism of amphibolite, Khaltaro granitic pegmatite hydrothermal vein system, Haramosh mountains, northern Pakistan. Can. Mineral. 1996, 34, 1253–1286. [Google Scholar]
- Grundmann, G.; Morteani, G. Ein neues Vorkommen von Smaragd, Alexandrit, Rubin und Saphir in einem Topas-führenden Phlogopit-felds von Poona, Cue District, West Australien. Zeitschrift der Deutschen Gemmologischen Gesellschaft 1995, 44, 11–31. [Google Scholar]
- Xue, G.; Marshall, D.; Zhang, S.; Ullrich, T.D.; Bishop, T.; Groat, L.A.; Thorkelson, D.J.; Giuliani, G.; Fallick, A.E. Conditions for early Cretaceous emerald formation at Dyakou, China: Fluid inclusion, Ar-Ar, and stable isotope studies. Econ. Geol. 2010, 105, 339–349. [Google Scholar] [CrossRef]
- Kazmi, A.H.; Snee, L.W. Geology of the world emerald deposits: A brief review. In Emeralds of Pakistan: Geology, Gemology and Genesis; Kazmi, A.H., Snee, L.W., Eds.; Van Nostrand Reinhold Company: New York, NY, USA, 1989; pp. 165–228. [Google Scholar]
- Martins, S. Brazilian emeralds. In Proceedings of the Oral Communication, II World Emerald Symposium, Bogotá, Colombia, 12–14 October 2018. [Google Scholar]
- Biondi, J.C. Depósitos de esmeralda de Santa Terezinha (GO). Rev. Bras. Geociências 1990, 20, 7–24. [Google Scholar] [CrossRef]
- Gusmão Costa, S.A. de Correlação da seqüência encaixante das esmeraldas de Santa Terezinha de Goiás com os terrenos do tipo greenstone belt de Crixás e tipologia dos depósitos. In Proceedings of the Boletim de Resumos Espandidos XXXVIII Congresso Brasileiro de Geologia Goiânia, Goiás, Brazil, 1986; Volume 2, pp. 597–614. [Google Scholar]
- Giuliani, G.; D’El-Rey Silva, L.J.; Couto, P.A. Origin of emerald deposits of Brazil. Miner. Deposita 1990, 25, 57–64. [Google Scholar] [CrossRef]
- D’el-Rey Silva, L.J.H.; Giuliani, G. Controle estrutural da jazida de esmeraldas de Santa Terezinha de Goiás: Implicações na gênese, na têctonica regional e no planajamento de lavra. In Proceedings of the Boletim de Resumos Espandidos XXXV Congresso Brasileiro de Geologia, Belém, PA, Brazil; 1988; Volume 1, pp. 413–427. [Google Scholar]
- D’el-Rey Silva, L.J.H.; de Barros Neto, L.S. The Santa Terezinha-Campos Verdes emerald district, central Brazil: Structural and Sm-Nd data to constrain the tectonic evolution of the Neoproterozoic Brasília belt. J. S. Am. Earth Sci. 2002, 15, 693–708. [Google Scholar] [CrossRef]
- Morteani, G.; Grundmann, G. The emerald porphyroblasts in the penninic rocks of the central Tauern Window. Neues Jb. Miner. Monat. 1977, 11, 509–516. [Google Scholar]
- Grundmann, G. Polymetamorphose und Abschätzung der Bildungsb edin-gungen der smaragd-fürhrenden gesteinsserien der leckbachscharte, Habachtal, Österreich. Fortschr. Mineral. 1980, 58, 39–41. [Google Scholar]
- Grundmann, G.; Morteani, G. Die geologie des smaragdvorkommens im Habachtal (Land Salzburg, Österreich). Archiv. Lagerstättenforsch. Geol. Bundensanst 1982, 2, 71–107. [Google Scholar]
- Nwe, Y.Y.; Grundmann, G. Evolution of metamorphic fluids in shear zones: The record from the emeralds of Habachtal, Tauern window, Austria. Lithos 1990, 25, 281–304. [Google Scholar] [CrossRef]
- Trumbull, R.B.; Krienitz, M.-S.; Grundmann, G.; Wiedenbeck, M. Tourmaline geochemistry and δ11B variations as a guide to fluid-rock interaction in the Habachtal emerald deposit, Tauern window, Austria. Contrib. Mineral. Petr. 2009, 157, 411–427. [Google Scholar] [CrossRef]
- Bowersox, G.; Snee, L.W.; Foord, E.E.; Seal, R.R., II. Emeralds of the Panjsher Valley, Afghanistan. Gems Gemol. 1991, 27, 26–39. [Google Scholar] [CrossRef]
- Hussain, S.S.; Chaudhry, M.N.; Dawood, H. Emerald mineralization of Barang, Bajaur Agency, Pakistan. J. Gemmol. 1993, 23, 402–408. [Google Scholar] [CrossRef]
- Dilles, J.H.; Snee, L.W.; Laurs, B.M. Geology, Ar-Ar age and stable isotopes geochemistry of suture-related emerald mineralization, Swat, Pakistan, Himalayas. In Proceedings of the Geological Society of America, Annual Meeting, Seattle, WA, USA, 1994; Abstracts. Volume 26, p. A-311. [Google Scholar]
- Arif, M.; Fallick, A.E.; Moon, C.J. The genesis of emeralds and their host rocks from Swat, northwestern Pakistan: A stable-isotope investigation. Miner. Deposita 1996, 31, 255–268. [Google Scholar] [CrossRef]
- Arif, M.; Henry, D.J.; Moon, C.J. Host rock characteristics and source of chromium and beryllium for emerald mineralization in the ophiolitic rocks of the Indus suture zone in Swat, NW Pakistan. Ore Geol. Rev. 2011, 39, 1–20. [Google Scholar] [CrossRef]
- Cheilletz, A.; Féraud, G.; Giuliani, G.; Rodriguez, C.T. Time-pressure-temperature formation of Colombian emerald: An 40Ar/39Ar laser-probe and fluid inclusion-microthermometry contribution. Econ. Geol. 1994, 89, 361–380. [Google Scholar] [CrossRef]
- Ottaway, T.L.; Wicks, F.J.; Bryndzia, L.T.; Kyser, T.K.; Spooner, E.T.C. Formation of the Muzo hydrothermal emerald deposit in Colombia. Nature 1994, 369, 552–554. [Google Scholar] [CrossRef]
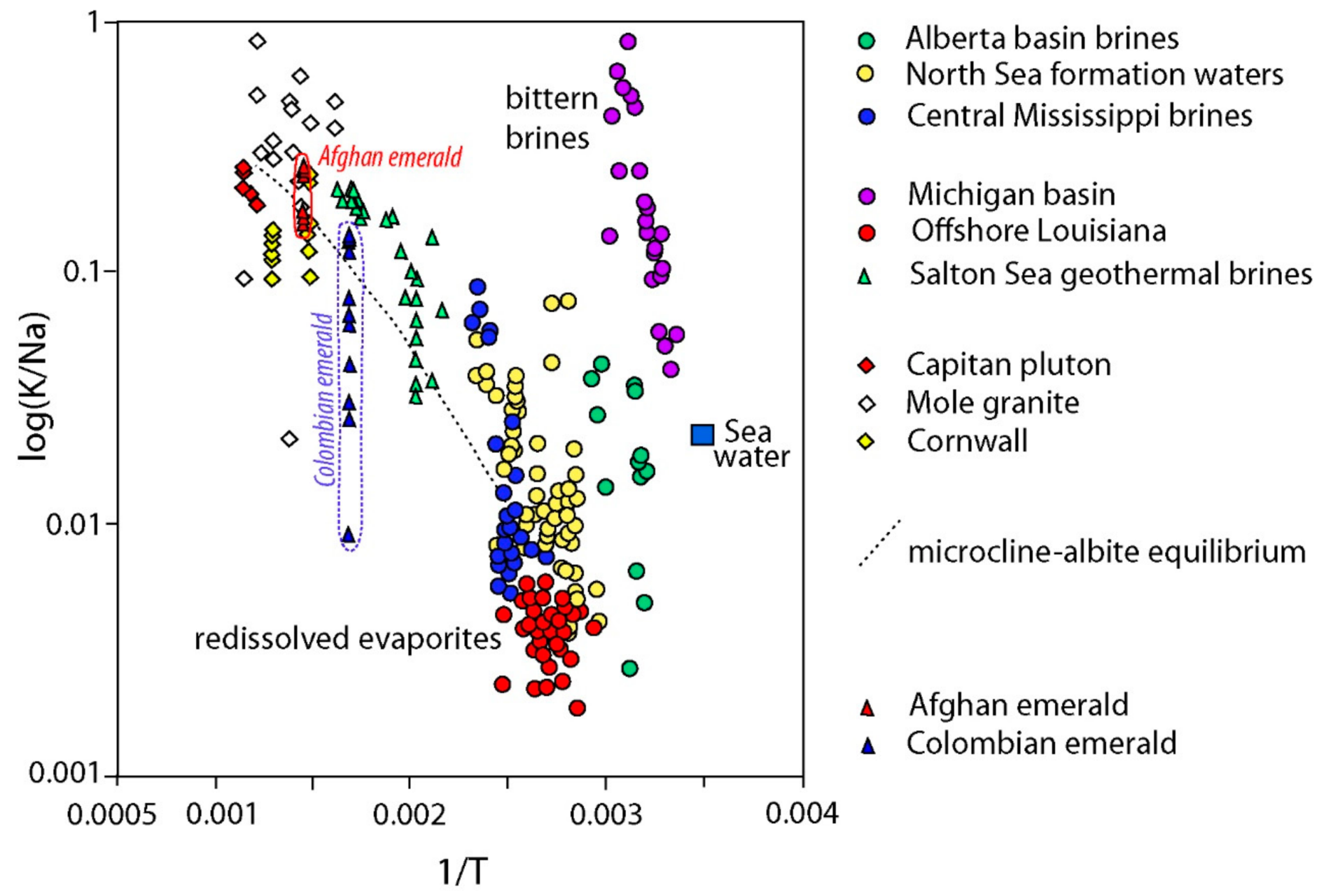
- Banks, D.; Giuliani, G.; Yardley, B.W.D.; Cheilletz, A. Emerald mineralisation in Colombia: Fluid chemistry and the role of brine mixing. Miner. Deposita 2000, 35, 699–713. [Google Scholar] [CrossRef]
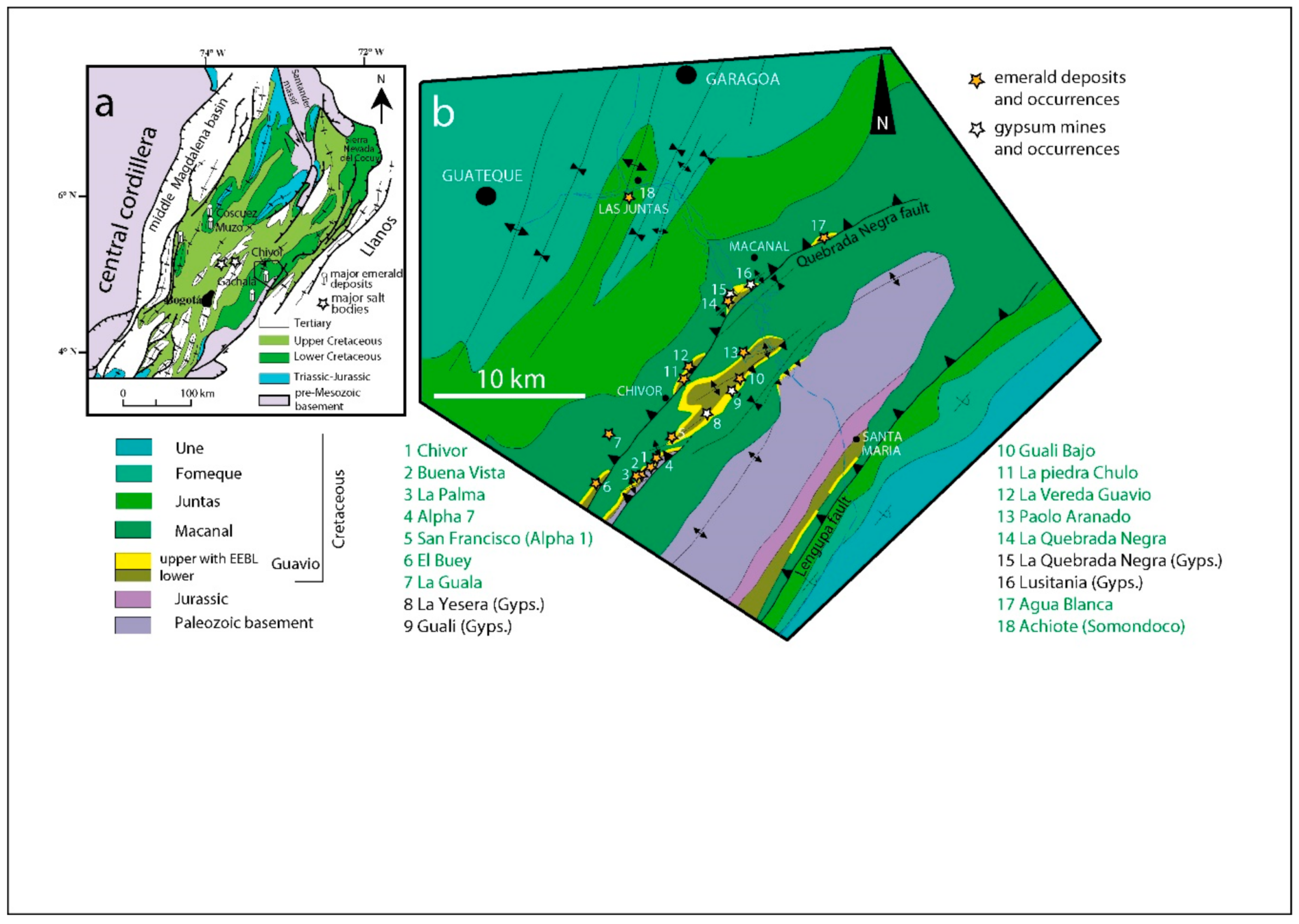
- Cheilletz, A.; Giuliani, G.; Branquet, Y.; Laumonier, B.; Sanchez, A.J.M.; Féraud, G.; Arhan, T. Datation K-Ar et 40Ar/39Ar à 65 ± 3 Ma des gisements d’émeraude du district de Chivor-Macanal: Argument en faveur d’une déformation précoce dans la Cordillère Orientale de Colombie. C. R. Acad. Sci. 1997, 324, 369–377. [Google Scholar]
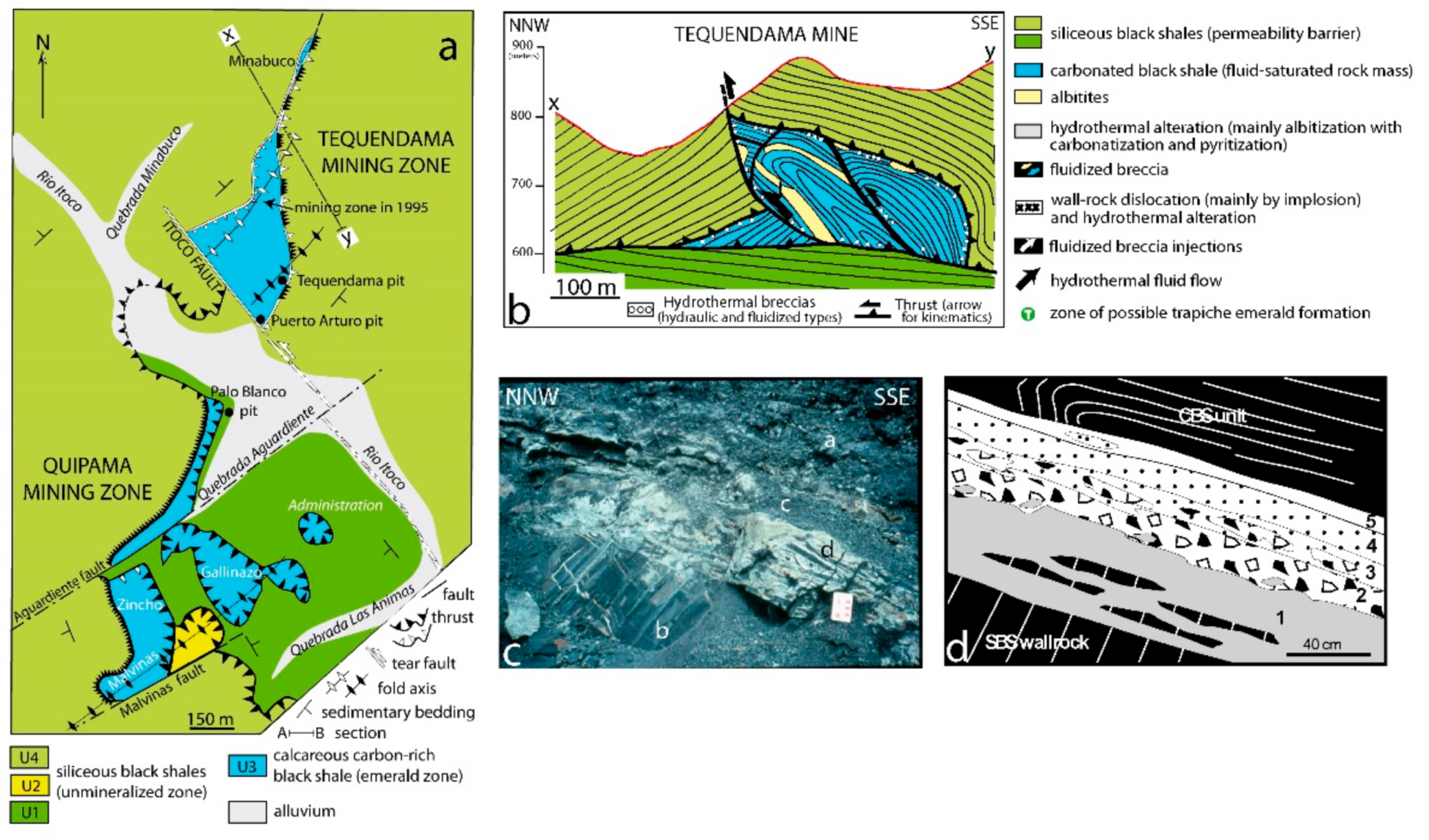
- Branquet, Y.; Cheilletz, A.; Giuliani, G.; Laumonier, B. Fluidized hydrothermal breccia in dilatant faults during thrusting: The Colombian emerald deposits case. In Fractures, Fluid Flow and Mineralization; McCaffrey, K.J.W., Lonergan, L., Wilkinson, J.J., Eds.; Geological Society London: London, UK, 1999; Volume 155, pp. 183–195. [Google Scholar]
- Branquet, Y.; Giuliani, G.; Cheilletz, A.; Laumonier, B. Colombian emeralds and evaporites: Tectono-stratigraphic significance of a regional emerald-bearing evaporitic breccia level. In Proceedings of the 13th SGA Biennal Meeting, Nancy, France, 24–27 August 2015; Volume 4, pp. 1291–1294. [Google Scholar]
- Cooper, M.A.; Addison, F.T.; Alvarez, R.; Coral, M.; Graham, R.; Hayward, A.B.; Howe, S.; Martinez, J.; Naar, J.; Pena, R.; et al. Basin development and tectonic history of the Llanos basin, Eastern Cordillera, and Middle Magdalena valley, Colombia. AAPG Bull. 1995, 79, 1421–1443. [Google Scholar]
- Branquet, Y.; Cheilletz, A.; Cobbold, P.R.; Baby, P.; Laumonier, B.; Giuliani, G. Andean deformation and rift inversion, eastern edge of Cordillera Oriental (Guateque, Medina area), Colombia. J. S. Am. Earth Sci. 2002, 15, 391–407. [Google Scholar] [CrossRef]
- Bayona, G.; Cortès, M.; Jaramillo, C.; Ojeda, G.; Aristizabal, J.J.; Harker, A.R. An integrated analysis of an orogen-sedimentary basin pair: Latest Cretaceous (Cenozoic evolution of the linked Eastern Cordillera orogen and the Llanos foreland basin of Colombia. Geol. Soc. Am. Bull. 2008, 120, 1171–1197. [Google Scholar] [CrossRef]
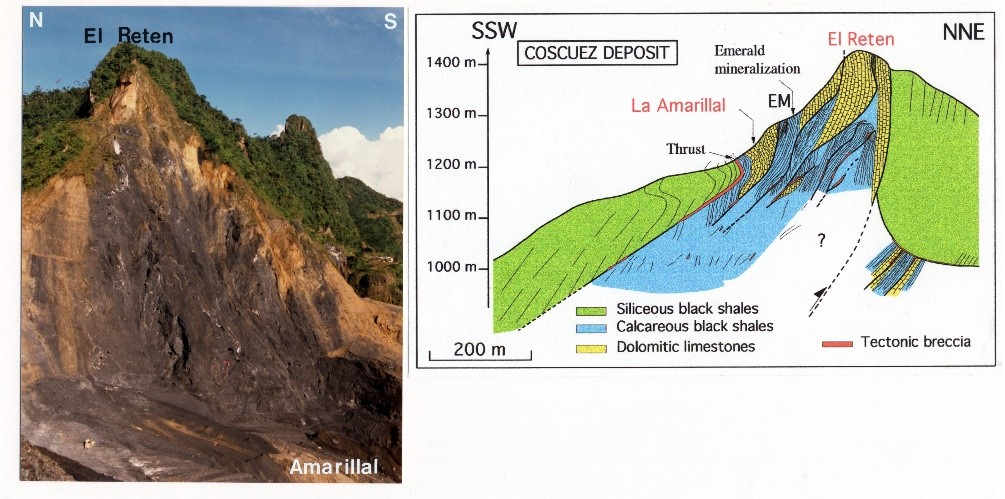
- Laumonier, B.; Branquet, Y.; Lopès, B.; Cheilletz, A.; Giuliani, G.; Rueda, F. Mise en évidence d’une tectonique compressive Eocène-Oligocène dans l’Ouest de la Cordillère orientale de Colombie, d’après la structure en duplex des gisements d’émeraude de Muzo et de Coscuez. C. R. Acad. Sci. 1996, 323, 705–712. [Google Scholar]
- Branquet, Y.; Laumonier, B.; Cheilletz, A.; Giuliani, G. Emeralds in the Eastern Cordillera of Colombia: Two tectonic settings for one mineralization. Geology 1999, 27, 597–600. [Google Scholar] [CrossRef]
- Pignatelli, I.; Giuliani, G.; Ohnenstetter, D.; Agrosì, G.; Mathieu, S.; Morlot, C.; Branquet, Y. Colombian trapiche emeralds: Recent advances in understanding their formation. Gems Gemol. 2015, 51, 222–259. [Google Scholar] [CrossRef]
- Giuliani, G.; France-Lanord, C.; Cheilletz, A.; Coget, P.; Branquet, Y.; Laumonier, B. Sulfate reduction by organic matter in Colombian emerald deposits: Chemical and stable isotope (C, O, H) evidence. Econ. Geol. 2000, 95, 1129–1153. [Google Scholar] [CrossRef]
- Bosshart, G. Emeralds from Colombia. J. Gemmol. 1991, 22, 409–425. [Google Scholar] [CrossRef]
- Hewton, M.L.; Marshall, D.D.; Ootes, L.; Loughrey, L.E.; Creaser, R.A. Colombian-style emerald mineralization in the northern Canadian Cordillera: Integration into a regional Paleozoic fluid flow regime. Can. J. Earth Sci. 2013, 50, 857–871. [Google Scholar] [CrossRef]
- Keith, J.D.; Thompson, T.J.; Ivers, S. The Uinta emerald and the emerald-bearing potential of the Red Pine shale, Uinta Mountains, Utah. Geol. Soc. Am. Abstr. Program 1996, 28, 85. [Google Scholar]
- Keith, J.D.; Nelson, S.T.; Thompson, T.J.; Dorais, M.J.; Olcott, J.; Duerichen, E.; Constenius, K.N. The genesis of fibrous calcite and shale-hosted emerald in a nonmagmatic hydrothermal system, Uinta mountains, Utah. Geol. Soc. Am. Abstr. Program 2002, 34, 55. [Google Scholar]
- Nelson, S.T.; Keith, J.D.; Constenius, K.N.; Duerichen, E.; Tingey, D.G. Genesis of fibrous calcite and emerald by amagmatic processes in the southwestern Uinta Mountains, Utah. Rocky Mt. Geol. 2008, 43, 1–21. [Google Scholar] [CrossRef]
- Tapponnier, P.; Mattauer, M.; Proust, F.; Cassaigneau, C. Mesozoic ophiolites, sutures, and large-scale tectonic movements in Afghanistan. Earth Planet. Sci. Lett. 1981, 52, 335–371. [Google Scholar] [CrossRef]
- Sabot, B.; Cheilletz, A.; de Donato, P.; Banks, D.; Levresse, G.; Barrès, O. Afghan emeralds face Colombian cousins. Chron. R. Min. BRGM 2000, 541, 111–114. [Google Scholar]
- Sabot, B.; Cheilletz, A.; de Donato, P.; Banks, D.; Levresse, G.; Barrès, O. The Panjsher-Afghanistan emerald deposit: New field and geochemical evidence for Colombian style mineralisation. In Proceedings of the European Union Geoscience XI, Strasbourg, France, 8–13 April 2001; Section OS 06. p. 548. [Google Scholar]
- An, Y. The geological characteristics of the emerald deposit in Tashen Kuergan, Xinjiang. Xinjiang Non-Ferrous Met. 2009, 29, 9–10. [Google Scholar]
- Wise, M.A. New finds in North Carolina. ExtraLapis Engl. 2002, 2, 64–65. [Google Scholar]
- Speer, W.E. Hiddenite district, Alexander Co, North Carolina. In Fieldtrip Guidebook. In Proceedings of the Southeastern Section 57th Annual Meeting; Charlotte, NC, USA, 10–11 April 2008, The Geological Society of America: Boulder, CO, USA, 2008; 28p. [Google Scholar]
- Wise, M.A.; Anderson, A.J. The emerald- and spodumene-bearing quartz veins of the Rist emerald mine, Hiddenite, North Carolina. Can. Mineral. 2006, 44, 1529–1541. [Google Scholar] [CrossRef]
- Grundmann, G.; Morteani, G. “Smaragdminen der Cleopatra”: Zabara, Sikait und Umm Kabo in Ägypten. Lapis 1993, 7–8, 27–39. [Google Scholar]
- Grundmann, G.; Morteani, G. Emerald formation during regional metamorphism: The Zabara, Sikeit and Umm Kabo deposits (Eastern Desert, Egypt). In Geoscientific Research in Northeast Africa; Schandelmeier, T., Ed.; A.A. Balkema: Rotterdam, The Netherlands, 1993; pp. 495–498. [Google Scholar]
- Grundmann, G.; Morteani, G. Multi-stage emerald formation during Pan-African regional metamorphism: The Zabara, Sikait, Umm Kabo deposits, South Eastern desert of Egypt. J. Afr. Earth Sci. 2008, 50, 168–187. [Google Scholar] [CrossRef]
- Surour, A.A.; Takla, M.A.; Omar, S.A. EPR spectra and age determination of beryl from the Eastern Desert of Egypt. Ann. Geol. Surv. Egypt 2002, 25, 389–400. [Google Scholar]
- Giuliani, G.; France-Lanord, C.; Coget, P.; Schwarz, D.; Cheilletz, A.; Branquet, Y.; Giard, D.; Pavel, A.; Martin-Izard, A.; Piat, D.H. Oxygen isotope systematics of emerald—Relevance for its origin and geological significance. Miner. Deposita 1998, 33, 513–519. [Google Scholar] [CrossRef]
- Duarte, L.D.C.; Pulz, G.M.; D’el-Rey Silva, L.J.H.; Ronchi, L.H.; de Brun, T.M.M.; Juchem, P.L. Microtermometria das inclusões fluidas na esmeralda do distrito mineiro de Campos Verdes, Goiás. In Caracterização e Modelamento de depósitos Minerais; Ronchi, L.H., Althoff, F.J., Eds.; Eitoras Unisinos: São Leopoldo, RS, Brazil, 2003; pp. 267–292. [Google Scholar]
- Olivera, J.A.P.; Ali, S.H. Gemstone mining as a develoment cluster: A study of Brazil’s emerald mines. Resour. Policy 2011, 36, 132–141. [Google Scholar] [CrossRef]
- Salvi, S.; Giuliani, G.; Andrianjakavah, P.R.; Moine, B.; Beziat, D.; Fallick, A.E. Fluid inclusion and stable isotope constraints on the formation of the Ianapera emerald deposit, Southern Madagascar. Can. Mineral. 2017, 55, 619–650. [Google Scholar] [CrossRef]
- de Wit, M.J.; Hart, R.A.; Hart, R.J. The Jamestown ophiolite complex, Barberton mountain belt: A section through 3.5 Ga oceanic crust. J. Afr. Earth Sci. 1987, 6, 681–730. [Google Scholar] [CrossRef]
- Biondi, J.C.; Poidevin, J.L. Idade da mineralização e da sequência Santa Terezinha (Goiás, Brasil). In Proceedings of the Boletim de Resumos Espandidos XXXVIII Congresso Brasileiro de Geologia, Camboriú, Brasil, 23–28 October 1994; pp. 302–304. [Google Scholar]
- Ribeiro-Althoff, A.M.; Cheilletz, A.; Giuliani, G.; Féraud, G.; Barbosa Camacho, G.; Zimmermann, J.L. Evidences of two periods (2 Ga and 650-500 Ma) of emerald formation in Brazil by K-Ar and 40Ar/39Ar dating. Int. Geol. Rev. 1997, 39, 924–937. [Google Scholar] [CrossRef]
- Calligaro, T.; Dran, J.-C.; Poirot, J.-P.; Querré, G.; Salomon, J.; Zwaan, J.C. PIXE/PIGE characterization of emeralds using an external micro-beam. Nucl. Instrum. Meth. B 2000, 161–163, 769–774. [Google Scholar] [CrossRef]
- Höll, R.; Maucher, A.; Westenberger, H. Synsedimentary diagenetic ore fabrics in the strata- and time-bound scheelite deposits of Kleinarltal and Felbertal in the Eastern Alps. Miner. Deposita 1972, 7, 217–226. [Google Scholar] [CrossRef]
- Raith, J.G.; Montanuiversität Leoben, Austria. Personal communication, 2010.
- Kozlik, M.; Raith, J.G.; Gerdes, A. U-Pb, Lu-Hf and trace element characteristics of zircon from the Felbertal scheelite deposit (Austria): New constraints on timing and source of W mineralization. Chem. Geol. 2016, 421, 112–126. [Google Scholar] [CrossRef]
- Maluski, H.; Matte, P. Ages of alpine tectonometamorphic events in the northwestern Himalaya (northern Pakistan) by 39Ar/40Ar method. Tectonophysics 1984, 3, 1–18. [Google Scholar]
- Grundmann, G.; Morteani, G. Alexandrite, emerald, sapphire, ruby and topaz in a biotite-phlogopite fels from the Poona, Cue district, Western Australia. Austral. Gemmol. 1998, 20, 159–167. [Google Scholar]
- Vapnik, Y.; Moroz, I. Fluid inclusions in Panjshir emerald (Afghanistan). In Proceedings of the 16th European Current Research on Fluid Inclusions, Faculdade de Ciências do Porto, Porto, Portugal, 17–20 July 2001; Noronha, F., Doria, A., Guedes, A., Eds.; Memoria n° 7: New York, NY, USA; pp. 451–454. [Google Scholar]
- Lapointe, M.; Anderson, A.J.; Wise, M. Fluid inclusion constraints on the formation of emerald-bearing quartz veins at the Rist tract, Heddenite, North Carolina. Atl. Geol. Abstr. 2004, 4, 146. [Google Scholar]
- Diamond, L.W. Introduction to gas-bearing aqueous fluid inclusions. In Fluid Inclusions: Analysis and Interpretation; Samson, I., Anderson, A., Marshall, D., Eds.; Mineralalogical Association of Canada: Quebec City, QC, Canada, 2003; Volume 32, pp. 101–158. [Google Scholar]
- Bodnar, R.J. Introduction to aqueous-electrolyte fluid inclusions. In Fluid Inclusions: Analysis and Interpretation; Samson, I., Anderson, A., Marshall, D., Eds.; Mineralogical Association of Canada: Quebec City, QC, Canada, 2003; Volume 32, pp. 81–100. [Google Scholar]
- Audétat, A.; Gunter, D.; Heinrich, C. Formation of a magmatic-hydrothermal ore deposit: Insights with LA-ICP-MS analysis of fluid inclusions. Science 2012, 279, 2091–2094. [Google Scholar]
- Diamond, L.W.; Marshall, D.; Jackman, J.; Skippen, G.B. Elemental analysis of individual fluid inclusions in minerals by Secondary Ion Mass Spectrometry (SIMS): Application to cation ratios of fluid inclusions in an Archaean mesothermal gold-quartz vein. Geochim. Cosmochim. Acta 1990, 54, 545–552. [Google Scholar] [CrossRef]
- Rosasco, G.; Roedder, E.; Simmons, J. Laser-excited Raman spectroscopy for non-destructive analysis of individual phases in fluid inclusions in minerals. Science 1975, 190, 557–560. [Google Scholar] [CrossRef]
- Delhaye, M.; Dhamelincourt, P. Raman microprobe and microscope with Laser excitation. J. Raman Spectrosc. 1975, 3, 33–43. [Google Scholar] [CrossRef]
- Dhamelincourt, P.; Schubnel, H. La microsonde moléculaire à laser et son application à la minéralogie et la gemmologie. Rev. Gemmol. AFG 1977, 52, 11–14. [Google Scholar]
- Burruss, R.C. Raman spectroscopy of fluid inclusions. In Fluid Inclusions: Analysis and Interpretation; Samson, I., Anderson, A., Marshall, D., Eds.; Mineralogical Association of Canada: Quebec City, QC, Canada, 2003; Volume 32, pp. 279–290. [Google Scholar]
- Burke, E.A.J. Raman microspectrometry of fluid inclusions. Lithos 2001, 55, 139–158. [Google Scholar] [CrossRef]
- Taylor, R.; Fallick, A.; Breaks, F. Volatile evolution in Archean rare-element granitic pegmatites: Evidence from the hydrogen-isotopic composition of channel H2O in beryl. Can. Mineral. 1992, 30, 877–893. [Google Scholar]
- Marshall, D.; Meisser, N.; Ellis, S.; Jones, P.; Bussy, F.; Mumenthaler, T. Formational conditions for the Binntal emerald occurrence, Valais, Switzerland: Fluid inclusion, chemical composition and stable isotope studies. Can. Mineral. 2017, 55, 725–741. [Google Scholar] [CrossRef]
- Groat, L.A.; Giuliani, G.; Marshall, D.D.; Turner, D. Emerald deposits and occurrences: A review. Ore Geol. Rev. 2008, 34, 87–112. [Google Scholar] [CrossRef]
- Sheppard, S.M.F. Characterization and isotopic variations in natural waters. In Stable Isotopes in High Temperature Geological Processes; Valley, J.W., Taylor, H.P., O’Neil, J.R., Eds.; Mineralogical Association of America: Chantilly, VA, USA, 1986; Volume 16, pp. 165–183. [Google Scholar]
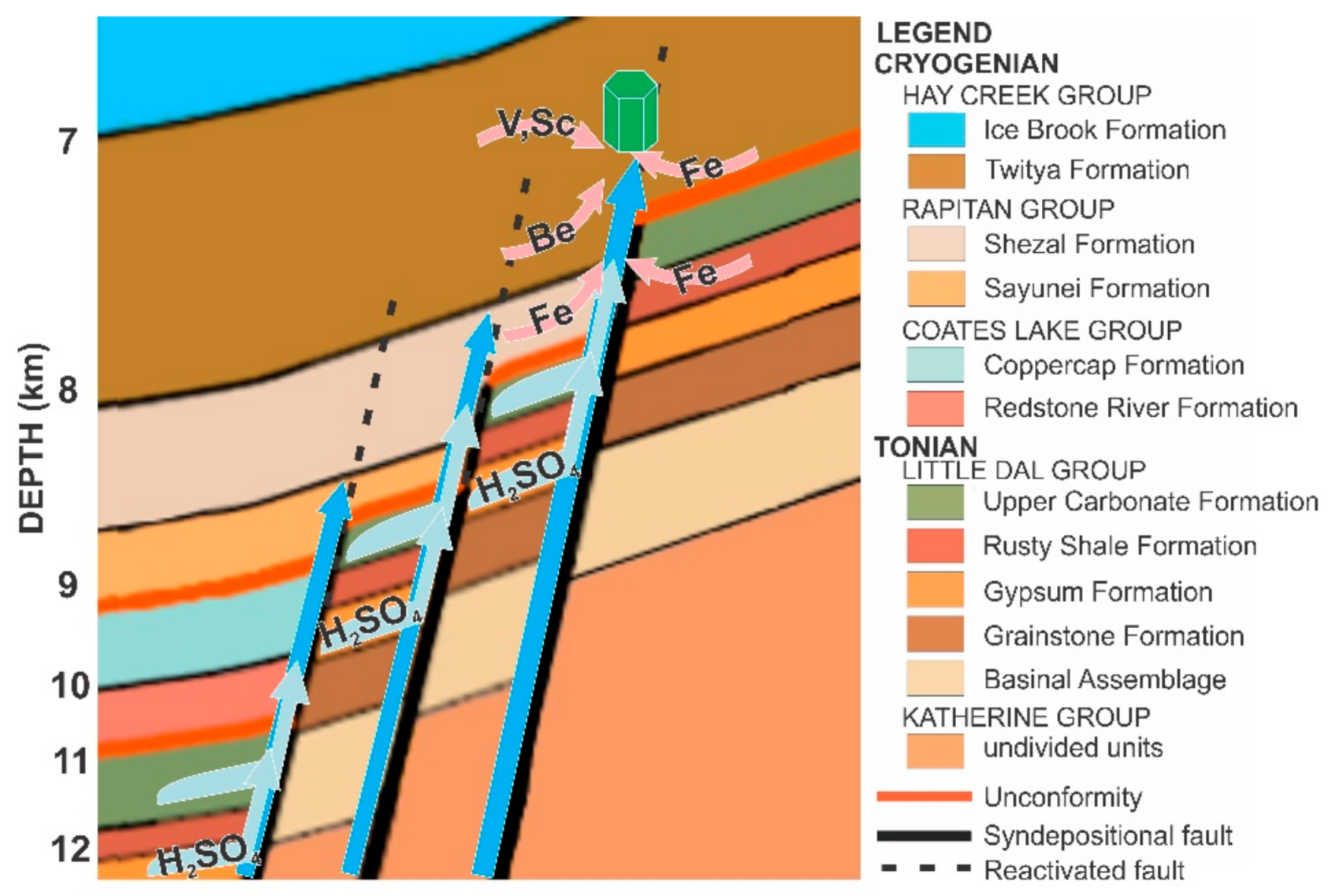
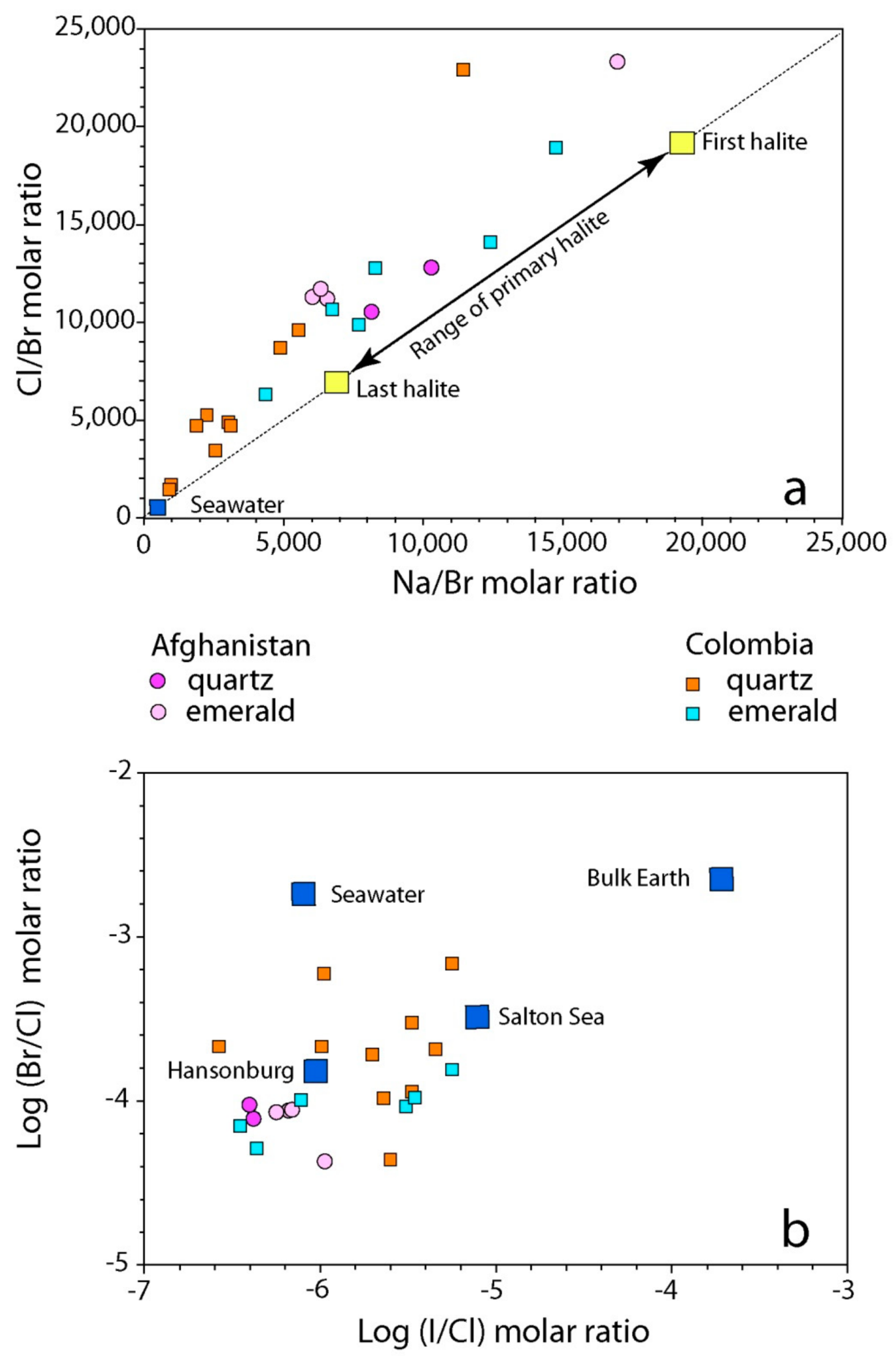
- Giuliani, G.; Dubessy, J.; Ohnenstetter, D.; Banks, D.; Branquet, Y.; Feneyrol, J.; Fallick, A.E.; Martelat, E. The role of evaporites in the formation of gems during metamorphism of carbonate platforms: A review. Miner. Deposita 2017, 53, 1–20. [Google Scholar] [CrossRef]
- Yardley, B.W.D.; Bodnar, R.J. Fluids in the Continental Crust. Geochem. Perspect. 2014, 3, 1–127. [Google Scholar] [CrossRef]
- Yardley, B.W.D.; Cleverley, J.S. The Role of Metamorphic Fluids in the Formation of Ore Deposits; Geological Society: London, UK, 2013; pp. 1–393. [Google Scholar]
- Williams, A.E.; McKibben, M.A. A brine interface in the Salton Sea Geothermal System, California: Fluid geochemical and isotopic characteristics. Geochim. Cosmochim. Acta 1989, 53, 1905–1920. [Google Scholar] [CrossRef]
- Bohlke, J.K.; Irwin, J.J. Laser microprobe analyses of Cl, Br, I, and K in fluid inclusions: Implications for sources of salinity in some ancient hydrothermal fluids. Geochim. Cosmochim. Acta 1992, 56, 203–225. [Google Scholar] [CrossRef]
- Poujol, M.; Robb, L.J.; Respaut, J.P. Origin of gold and emerald mineralization in the Murchinson greenstone belt, Kaapval craton, South Africa. Mineral. Mag. 1998, 62A, 1206–1207. [Google Scholar] [CrossRef]
- Beus, A.A. Geochemistry of Beryllium and Genetic Types of Beryllium Deposits; W.H. Freeman: San Francisco, CA, USA; London, UK, 1966; 401p. [Google Scholar]
- Smirnov, V.L. Deposits of beryllium. In Ore deposits of the U.S.S.R.; Pitman Publications: London, UK, 1977; Volume 3, pp. 320–371. [Google Scholar]
- Franz, G.; Gilg, H.A.; Grundmann, G.; Morteani, G. Metasomatism at a granitic pegmatite-dunite contact in Galicia: The Franqueira occurrence of chrysoberyl (alexandrite), emerald, and phenakite: Discussion. Can. Mineral. 1996, 34, 1329–1331. [Google Scholar]
- Zwaan, J.C.; Touret, J. Emeralds in greenstone belts: The case of Sandawana, Zimbabwe. Münchener Geologische Hefte 2000, 28, 245–258. [Google Scholar]
- Franz, G.; Morteani, G. Be-minerals: Synthesis, stability, and occurrence in metamorphic rocks. In Beryllium: Mineralogy, Petrology, and Geochemistry; Grew, E.S., Ed.; Mineralogical Society of America: Washington, DC, USA, 2002; Volume 50, pp. 551–589. [Google Scholar]
- Martin-Izard, A.; Paniagua, A.; Moreiras, D. Metasomatism at a granitic pegmatite-dunite contact in Galicia: The Franqueira occurrence of chrysoberyl (alexandrite), emerald and phenakite. Can. Mineral. 1995, 33, 775–792. [Google Scholar]
- Martin-Izard, A.; Paniagua, A.; Moreiras, D.; Acevedo, R.D.; Marcos-Pascual, C. Metasomatism at a granitic pegmatite-dunite contact in Galicia: The Franqueira occurrence of chrysoberyl (alexandrite), emerald, and phenakite: Reply. Can. Mineral. 1996, 34, 1332–1336. [Google Scholar]
- Korzhinskii, D.S. Theory of Metasomatic Zoning; Clarendon Press: Oxford, UK, 1970. [Google Scholar]
- Sahu, S.S.; Singh, S.; Stapathy, J.S. Lithological and structural controls on the genesis of emerald occurrences and their exploration implications in and around Gubarabanda area, Singhbhum crustal province Eastern India. J. Geol. Soc. India 2018, 92, 291–297. [Google Scholar] [CrossRef]
- Zwaan, J.C.; Kanis, J.; Petsch, J. Update on emeralds from the Sandawana mines, Zimbabwe. Gems Gemol. 1997, 33, 80–101. [Google Scholar] [CrossRef]
- Holmes, A. The oldest dated minerals of the Rhodesian shield. Nature 1954, 173, 612–614. [Google Scholar] [CrossRef]
- Christensen, J.N.; Selverstone, J.; Rosenfeld, J.L.; DePaolo, D.J. Correlation by Rb/Sr geochronology of garnet growth histories from different structural levels within the Tauern Window, Eastern Alps. Contrib. Mineral. Petr. 1994, 118, 1–12. [Google Scholar] [CrossRef]
- Vance, D.; O’nions, R.K. Isotopic chronometry of zone garnets growth kinetics and metamorphic histories. Earth Planet. Sci. Lett. 1990, 114, 113–129. [Google Scholar] [CrossRef]
- Gilg, A.; Technische Universität München, Germany. Personal comunication, 2009.
- Lake, D.J. Are there Colombian-type emeralds in Canada’s Northern Cordillera? Insights from regional silt geochemistry, and the genesis of emerald at Lened, NWT. M.Sc. Thesis, University of British Columbia, Vancouver, BC, Canada, 2017. [Google Scholar]
- Escobar, R. Geology and geochemical expression of the Gachalá emerald district, Colombia. Geol. Soc. Am. Abstr. Program 1978, 10, 397. [Google Scholar]
- Beus, A.A. Sodium—A geochemical indicator of emerald mineralization in the Cordillera Oriental, Colombia. J. Geochem. Explor. 1979, 11, 195–208. [Google Scholar] [CrossRef]
- Ringsrud, R. The Coscuez mine: A major source of Colombian emeralds. Gems Gemol. 1986, 22, 67–79. [Google Scholar] [CrossRef]
- Cheilletz, A.; Giuliani, G. The genesis of Colombian emeralds: A restatement. Miner. Deposita 1996, 31, 359–364. [Google Scholar] [CrossRef]
- Rohtert, W.R.; Independant geologist. Personal comunication, 2017.
- Gutiérrez, L.H.O. Evaluación magnetométrica, radiométrica y geoeléctrica de depósitos esmeraldíferos. Geofísica Colombiana 2003, 7, 13–18. [Google Scholar]
- Arif, M.; Henry, D.J.; Moon, C.J. Cr-bearing tourmaline associated with emerald deposits from Swat, NW Pakistan: Genesis and its exploration significance. Am. Mineral. 2010, 95, 799–809. [Google Scholar] [CrossRef]
- Wise, M.A. Crystal Morphology of quartz, calcite, pyrite and rutile; Potential tool for emerald exploration in the Hiddenite area (North Carolina). Geol. Soc. Am. Abstr. Programs 2012, 44, 25. [Google Scholar]
- Murphy, D.C.; Lipovsky, P.S.; Stuart, A.; Fonseca, A.; Piercey, S.J.; Groat, L. What about those emeralds, eh? Geological setting of emeralds at Regal Ridge (SE Yukon) provides clues to their origin and to other places to explore. Geol. Assoc. Canada Mineral. Assoc. Canada Abstr. Program 2002, 28, 154. [Google Scholar]
- Lewis, L.L.; Hart, C.J.R.; Murphy, D.C. Roll out the beryl. In Yukon Geoscience Forum; Publications du Gouvernement du Canada: Yukon Territory, NT, Canada, 2003. [Google Scholar]









| Elementss | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 64.50 | 64.94 | 63.58 | 65.98 | 67.13 | 66.74 | 66.30 | 66.04 | 64.71 | 64.75 | 65.57 | 65.57 | 66.49 | 66.28 | 67.13 | 64.39 |
| TiO2 | 0.00 | 0.00 | n.d. | 0.01 | 0.00 | 0.00 | 0.01 | 0.01 | n.d. | n.d. | 0.04 | 0.01 | 0.00 | 0.06 | 0.01 | 0.01 |
| Al2O3 | 11.54 | 10.61 | 17.36 | 16.81 | 18.77 | 18.67 | 18.60 | 18.17 | 17.23 | 14.83 | 17.24 | 14.51 | 18.29 | 18.10 | 16.52 | 14.48 |
| Sc2O3 | n.d. | n.d. | n.d. | 0.06 | 0.01 | 0.00 | 0.05 | 0.01 | 0.03 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.02 |
| V2O3 | 0.15 | 0.10 | 0.02 | 0.17 | 0.05 | 0.01 | 0.10 | 0.01 | 0.23 | 0.03 | 0.07 | 0.00 | 0.17 | 0.05 | 0.00 | 0.03 |
| Cr2O3 | 1.83 | 3.39 | 0.25 | 0.35 | 0.20 | 0.26 | 0.02 | 0.05 | 0.01 | 0.57 | 0.07 | 0.09 | 0.05 | 0.24 | 0.37 | 0.17 |
| Fe2O3 | 1.34 | 1.27 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| La2O3 | 0.06 | 0.12 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Ce2O3 | 0.02 | 0.00 | n.d. | 0.02 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| BeO | 13.40 | 13.45 | 12.85 | 13.65 | 13.99 | 13.91 | 13.82 | 13.73 | 13.54 | 13.50 | 13.47 | 13.59 | 13.83 | 13.78 | 13.80 | 13.37 |
| MgO | 3.49 | 3.36 | 0.58 | 0.95 | 0.04 | 0.03 | 0.06 | 0.15 | 0.88 | 2.24 | 0.87 | 2.63 | 0.05 | 0.16 | 0.87 | 2.38 |
| CaO | 0.21 | 0.17 | n.d. | 0.00 | 0.00 | 0.00 | 0.01 | 0.01 | 0.02 | 0.02 | 0.00 | 0.06 | 0.00 | 0.00 | 0.00 | 0.04 |
| MnO | 0.10 | 0.00 | n.d. | 0.03 | 0.00 | 0.01 | 0.01 | 0.00 | n.d. | 0.01 | 0.00 | 0.01 | 0.00 | 0.00 | 0.01 | 0.01 |
| FeO | n.d. | n.d. | 0.37 | 0.17 | 0.21 | 0.19 | 0.12 | 0.15 | 0.26 | 0.65 | 0.33 | 0.96 | 0.71 | 0.40 | 0.14 | 0.63 |
| Li2O | n.d. | n.d. | 0.28 | 0.03 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.11 | 0.02 | 0.01 | 0.01 | 0.01 | n.d. |
| Na2O | 0.83 | 0.89 | 1.13 | 0.62 | 0.05 | 0.06 | 0.08 | 0.27 | 0.82 | 1.76 | 0.82 | 1.84 | 0.04 | 0.10 | 0.70 | 1.86 |
| K2O | 2.04 | 1.88 | 0.05 | 0.03 | 0.01 | 0.02 | 0.00 | 0.01 | 0.02 | 0.03 | 0.00 | 0.24 | 0.02 | 0.05 | 0.00 | 0.04 |
| Rb2O | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.00 | n.d. | 0.14 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Cs2O | 0.02 | 0.02 | 0.13 | n.d. | n.d. | n.d. | 0.01 | 0.11 | 0.10 | 0.03 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| H2O | 2.09 | 2.12 | 2.25 | 1.93 | 0.57 | 0.67 | 0.82 | 1.48 | 2.08 | 2.49 | 2.08 | 2.52 | 0.45 | 0.94 | 1.99 | 2.52 |
| Total | 101.62 | 102.32 | 98.85 | 100.81 | 101.04 | 100.57 | 100.01 | 100.20 | 99.94 | 101.05 | 100.67 | 102.05 | 100.11 | 100.17 | 101.55 | 99.96 |
| apfu | ||||||||||||||||
| Si4+ | 6.012 | 6.030 | 5.963 | 6.015 | 5.992 | 5.991 | 5.991 | 6.005 | 5.970 | 5.991 | 5.998 | 6.012 | 5.998 | 6.001 | 6.066 | 6.013 |
| Ti4+ | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | 0.003 | 0.001 | 0.000 | 0.004 | 0.001 | 0.001 |
| Al3+ | 1.268 | 1.161 | 1.919 | 1.806 | 1.975 | 1.975 | 1.981 | 1.947 | 1.873 | 1.617 | 1.858 | 1.568 | 1.945 | 1.931 | 1.759 | 1.594 |
| Sc3+ | 0.000 | 0.000 | 0.000 | 0.005 | 0.001 | 0.000 | 0.004 | 0.001 | 0.002 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.002 |
| V3+ | 0.011 | 0.007 | 0.002 | 0.012 | 0.004 | 0.001 | 0.007 | 0.001 | 0.017 | 0.002 | 0.005 | 0.000 | 0.012 | 0.004 | 0.000 | 0.002 |
| Cr3+ | 0.135 | 0.249 | 0.019 | 0.025 | 0.014 | 0.018 | 0.001 | 0.004 | 0.001 | 0.042 | 0.005 | 0.007 | 0.004 | 0.017 | 0.026 | 0.013 |
| Fe3+ | 0.094 | 0.089 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| La3+ | 0.002 | 0.004 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Ce3+ | 0.001 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Be2+ | 3.000 | 3.000 | 2.894 | 2.989 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 2.960 | 2.993 | 2.996 | 2.996 | 2.996 | 3.000 |
| Mg2+ | 0.485 | 0.465 | 0.081 | 0.129 | 0.005 | 0.004 | 0.008 | 0.020 | 0.121 | 0.309 | 0.119 | 0.359 | 0.007 | 0.022 | 0.117 | 0.331 |
| Ca2+ | 0.021 | 0.017 | 0.000 | 0.000 | 0.000 | 0.000 | 0.001 | 0.001 | 0.002 | 0.002 | 0.000 | 0.006 | 0.000 | 0.000 | 0.000 | 0.004 |
| Mn2+ | 0.008 | 0.000 | 0.000 | 0.002 | 0.000 | 0.001 | 0.001 | 0.000 | 0.000 | 0.001 | 0.000 | 0.001 | 0.000 | 0.000 | 0.001 | 0.001 |
| Fe2+ | 0.000 | 0.000 | 0.029 | 0.013 | 0.016 | 0.014 | 0.009 | 0.011 | 0.020 | 0.050 | 0.025 | 0.074 | 0.054 | 0.030 | 0.011 | 0.049 |
| Li+ | 0.000 | 0.000 | 0.106 | 0.011 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.040 | 0.007 | 0.004 | 0.004 | 0.004 | 0.000 |
| Na+ | 0.150 | 0.160 | 0.205 | 0.110 | 0.009 | 0.010 | 0.014 | 0.048 | 0.147 | 0.316 | 0.145 | 0.327 | 0.007 | 0.018 | 0.123 | 0.337 |
| K+ | 0.243 | 0.223 | 0.006 | 0.003 | 0.001 | 0.002 | 0.000 | 0.001 | 0.002 | 0.004 | 0.000 | 0.028 | 0.002 | 0.006 | 0.000 | 0.005 |
| Rb+ | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.008 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Cs+ | 0.001 | 0.001 | 0.005 | 0.000 | 0.000 | 0.000 | 0.000 | 0.004 | 0.004 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Type of Deposit | Tectonic-Metamorphic-Related | Tectonic-Magmatic-Related | ||||||
|---|---|---|---|---|---|---|---|---|
| Geological Environment | SEDIMENTARY | Metamorphic | Granitic | |||||
| Metamorphic Conditions | Anchizone to Greenschist facies | Greenschist to granulite facies | Greenschist to granulite facies | |||||
| Host-rocks | Sedimentary Rocks | Metamorphic rocks | Mafic-UltraMafic Rocks (M-UMR) | Sedimentary Rocks (SR) | Granitoids | |||
| Type | TYPE IIB | TYPE IIC | TYPE IIA | TYPE IID | TYPE IA | TYPE IB | TYPE IC | |
| carbonate platform sediments | Metamorphism of SR | Migmatites | Metamorphism of M-UMR | Metamorphosed Type IA, Mixed IA and IIA in M-UMR, and unknown | pegmatite- aplite- quartz- greisen veins, pods, metasomatites | |||
| Mineralization Style | veins and/or metasomatites | veins | veins | shear zone | shear zone, metasomatites, veins, boudins, fault | veins and/or metasomatites, skarns | pods | |
| Origin of the Fluid | Metasomatic-hydrothermal | Metasomatic-hydrothermal | Hydrothermal | Metamorphic-metasomatic | (Magmatic-Metasomatic) with a metamorphic remobilization | Metasomatic-hydrothermal | ||
| Deposits | Colombia (Eastern and western emerald zones) | China (Davdar) | USA (Heddenite) | Austria (Habachtal) | Austria (Habachtal) ?: Probably metamorphic remobilization of type IA deposit | Brazil (Carnaíba, Socotó, Itabira, Fazenda Bonfim, Pirenópolis, etc.) | Norway (Eidsvoll) | Nigeria (Kaduna) |
| Afghanistan (Panjsher) | ||||||||
| Canada (Mountain River) | Brazil (Itaberai, Santa Terezinha de Goiás) | China (Dyakou) | ||||||
| USA (Uinta (?): question about the presence of emerald) | Pakistan (Swat-Mingora–Gujar-Kili, Barang) | Brazil (Santa Terezinha de Goias) ?: Probably related to hidden granitic intrusive cut by thrust and emerald-bearing shear-zone | Canada (Tsa da Gliza, Taylor 2) | Canada (Lened) | ||||
| Bulgaria (Rila) | Australia (Emmavile, Torrington) | |||||||
| Urals (Malysheva, etc.) | ||||||||
| Pakistan (Swat-Mingora)? Probably related to undeformed hidden y intrusives | Pakistan (Khaltaro) | Kazakstan (Delgebetey) | ||||||
| Afghanistan (Tawak) | ||||||||
| India (Rajhastan, Gubaranda) | ||||||||
| Australia (Poona): Probably metamorphic remobilization of Tye IA deposit | South Africa (Gravelotte) | |||||||
| Zambia (Kafubu, etc.) | ||||||||
| Egypt (Djebel Sikait, Zabara, Umm Kabu): Probably metamorphic remobilization of Type IA deposit | Tanzania (Manyara, Sumbawanga) | |||||||
| Mozambique (Rio Maria, etc.) | ||||||||
| Zambia (Musakashi): Unknown genesis, vein style, fluid inclusion indicates affinities with Types IIB and IIC | Australia (Menzies, Wodgina, etc.) | |||||||
| Ethiopia (Kenticha) | ||||||||
| Madagascar (Ianapera, Mananjary) | ||||||||
| Zimbabwe (Sandawana, Masvingo, Filibusi) | ||||||||
| Somalia (Boorama) | ||||||||
| Ukraine (Wolodarsk) | ||||||||
| Type of Environment and Deposit | Tectonic Magmatic-Related | Tectonic Metamorphic-Related | ||
|---|---|---|---|---|
| Granitic Rocks in M-UM and SR | (Meta) Sedimentary Rocks | Metamorphic Rocks | ||
| (Type I) | (Type IIB) | (Types IIA- IIC-IID) | ||
| Temperature | 300-680°C | 300-330°C* | 350-400°C *1 | |
| 260-550°C *2 | ||||
| Pressure | 0.5 to 7 kbar | 3 to 4 kbar | 1.6 kbar *1D | |
| 4-4 to 5 kbar *2 | ||||
| Salinity | 2 to 45 wt.% eq. NaCl | 40 wt.% eq. NaCl | 30 to 33 wt.% eq. NaCl *1P | |
| 35 to 41 wt.% eq. NaCl *1D | ||||
| 2 to 38 wt.% eq.NaCl *2 | ||||
| Composition | H2O-NaCl-(±CO2)- (±N2)-(±CH4)- (K,Be,F,B,Li,P,Cs) | H2O-NaCl-(±CO2)- (±N2)-(±hydrocarbon liquid)- F15(K,Mg,Fe,Li,SO4,Pb,Zn) | H2O-NaCl-(±CO2) *1D | |
| H2O-NaCl-KCl-FeCl2-(±CO2) *1P | ||||
| H2O-NaCl-(±CO2) *2 | ||||
| CO2 or CO2-H2O-NaCl-(±N2 (±CH4) *2 | ||||
| or H2O-CH4-CO2-NaCl *2 | ||||
| Oxygen isotopes | 6.0 < δ18O < 15‰ | 16.2 < δ18O < 24.5‰ | Panjsher: | 13.25 < δ18O < 13.9‰ |
| Davdar: | 14.4 < δ18O < 15.8‰ | |||
| Sta Terezinha: | 12.0 < δ18O < 12.4‰ | |||
| Gravelotte: | 9.5 < δ18O < 9.7‰ | |||
| Gebel Sikait: | 9.8 < δ18O < 10.7‰ | |||
| Habachtal: | 6.5 < δ18O < 7.3‰ | |||
| Origin of the fluid | Metasomatic-Hydrothermal | Basinal brines that have dissolved evaporites | Metamorphic-Metasomatic | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giuliani, G.; Groat, L.A.; Marshall, D.; Fallick, A.E.; Branquet, Y. Emerald Deposits: A Review and Enhanced Classification. Minerals 2019, 9, 105. https://doi.org/10.3390/min9020105
Giuliani G, Groat LA, Marshall D, Fallick AE, Branquet Y. Emerald Deposits: A Review and Enhanced Classification. Minerals. 2019; 9(2):105. https://doi.org/10.3390/min9020105
Chicago/Turabian StyleGiuliani, Gaston, Lee A. Groat, Dan Marshall, Anthony E. Fallick, and Yannick Branquet. 2019. "Emerald Deposits: A Review and Enhanced Classification" Minerals 9, no. 2: 105. https://doi.org/10.3390/min9020105
APA StyleGiuliani, G., Groat, L. A., Marshall, D., Fallick, A. E., & Branquet, Y. (2019). Emerald Deposits: A Review and Enhanced Classification. Minerals, 9(2), 105. https://doi.org/10.3390/min9020105





