Colloidal and Thermal Behaviors of Some Venezuelan Kaolin Pastes for Therapeutic Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Paste Formulation
2.2. pH Measurement
2.3. Apparent Viscosity Measurement
2.4. Zeta Potential Measurement
2.5. Desorbed Cation Measurements
2.6. Thermal Measurements
3. Results and Discussion
3.1. Mineralogy
3.2. Rheology
3.3. pH
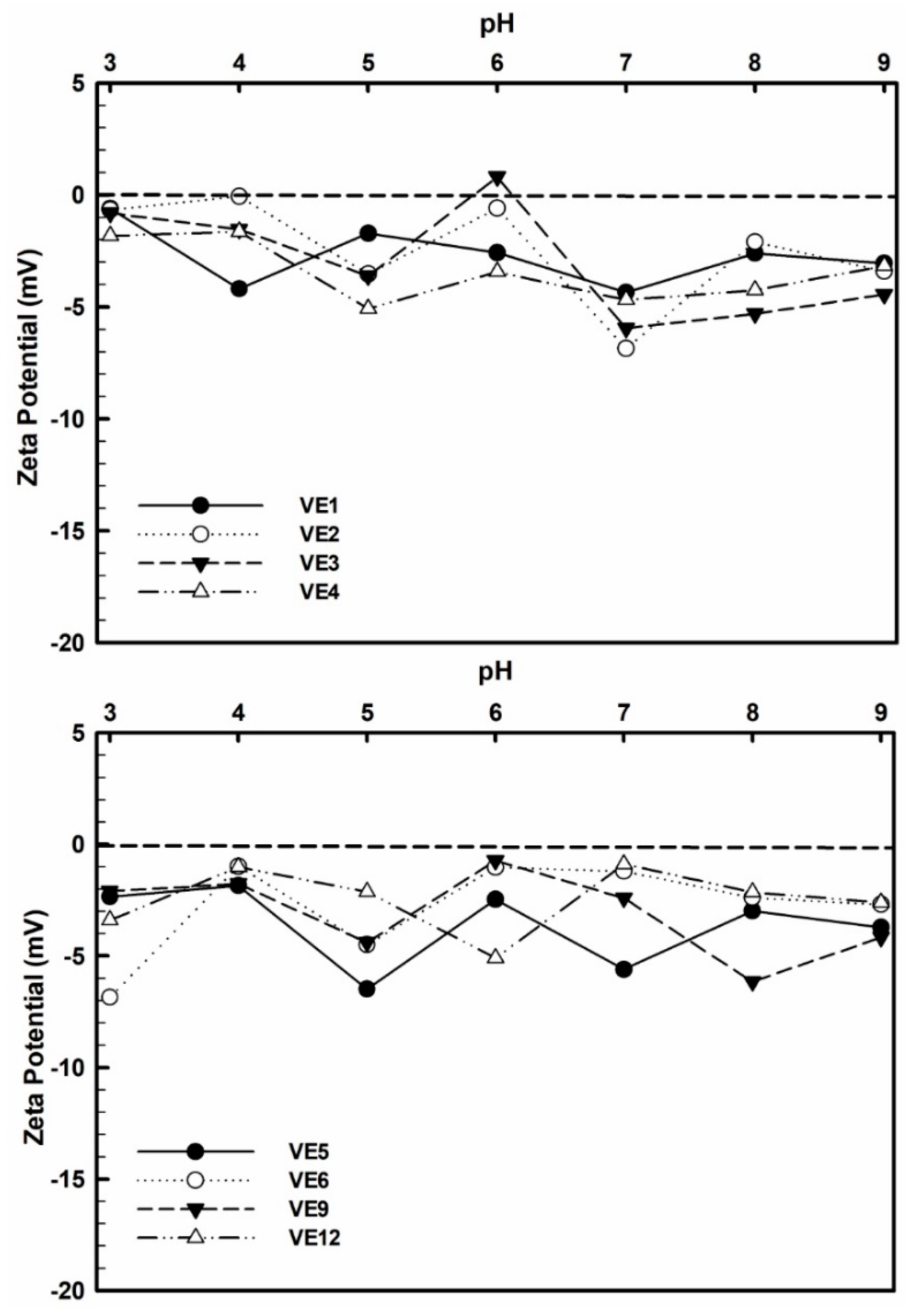
3.4. Zeta Potential
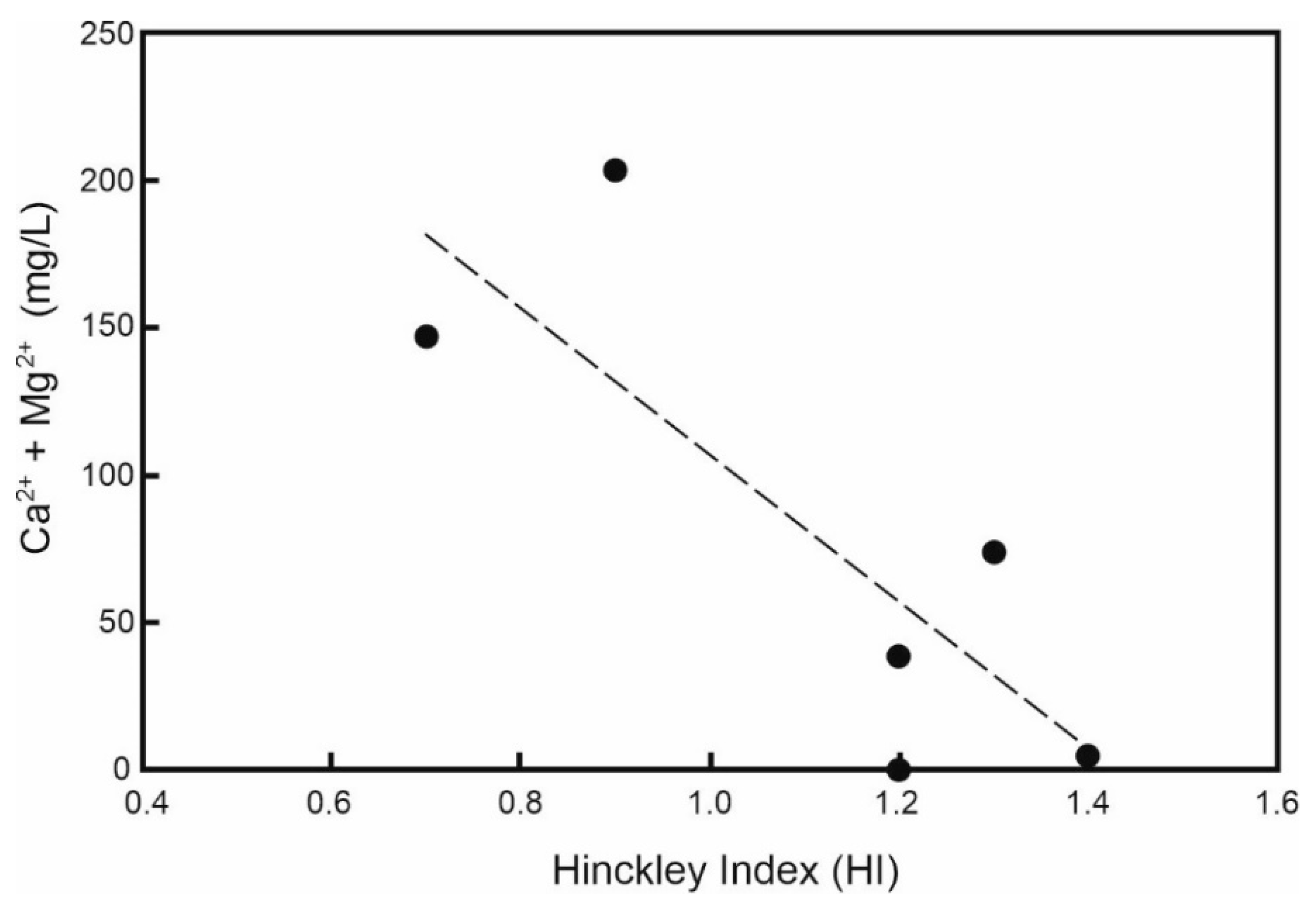
3.5. Desorbed Cations
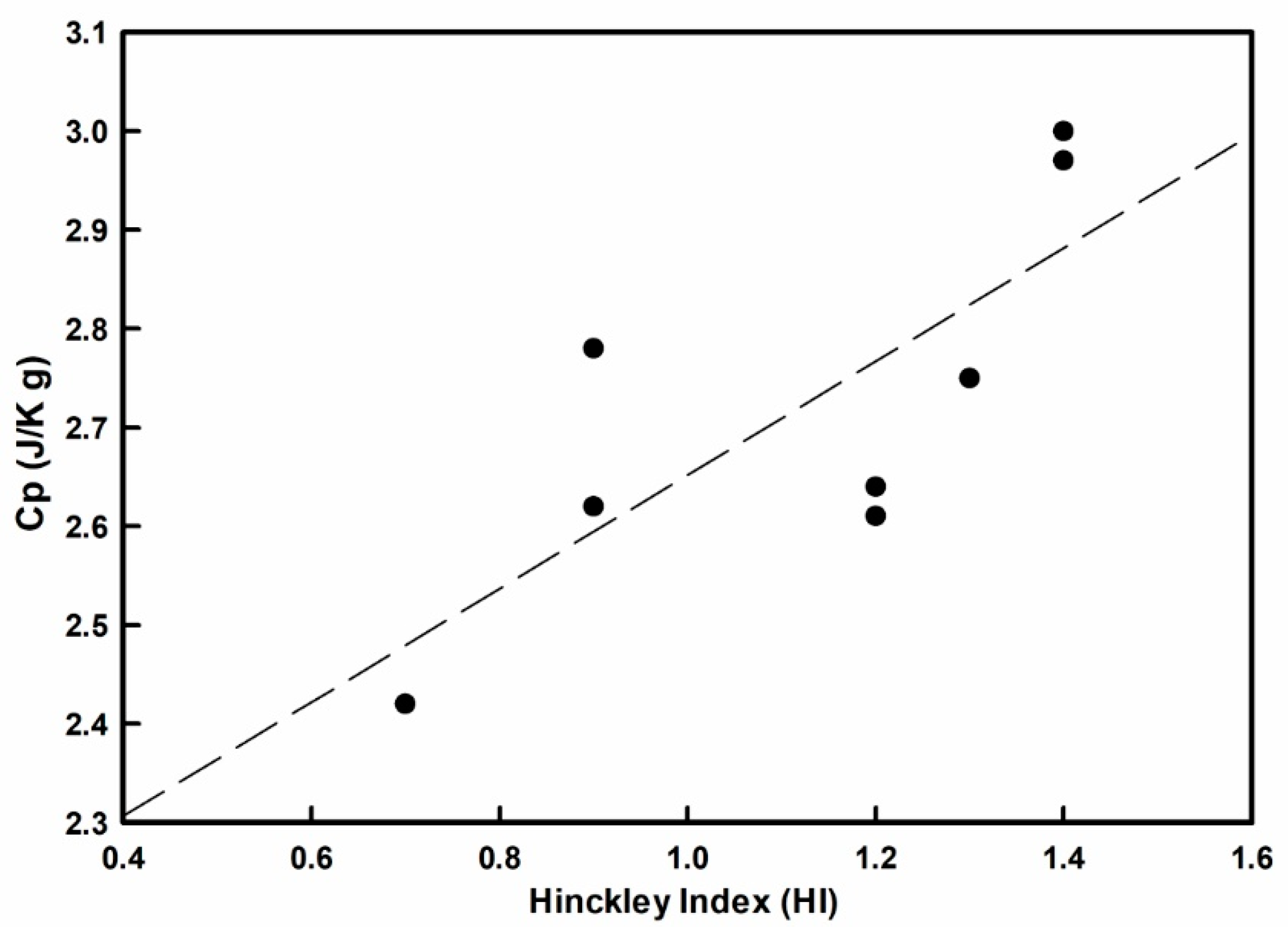
3.6. Thermal Characteristics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Summa, V.; Tateo, F. The use of pelitic raw materials in termal centres: Mineralogy, geochemistry, grain size and leaching tests. Examples from Lucania area (Southern Italy). Appl. Clay Sci. 1998, 12, 403–417. [Google Scholar] [CrossRef]
- Viseras, C.; López-Galindo, A. Pharmaceutical applications of some Spanish clays (sepiolite, palygorskite, bentonite): Some preformulation studies. Appl. Clay Sci. 1999, 14, 69–82. [Google Scholar] [CrossRef]
- Cara, S.; Carcangiu, G.; Padalino, G.; Palomba, M.; Tamanini, M. The bentonites in pelotherapy: Thermal properties of clay pastes from Sardinia (Italy). Appl. Clay Sci. 2000, 16, 125–132. [Google Scholar] [CrossRef]
- Carretero, M.I. Clay minerals and their beneficial effects upon human health: A review. Appl. Clay Sci. 2002, 21, 155–163. [Google Scholar] [CrossRef]
- Veniale, F.; Barberis, E.; Carcangiu, G.; Morandi, N.; Setti, M.; Tamanini, M.; Tessier, D. Formulation of muds for pelotherapy: Effects of “maturation” by different mineral waters. Appl. Clay Sci. 2004, 25, 135–148. [Google Scholar] [CrossRef]
- Carretero, M.I.; Gomes, C.S.F.; Tateo, F. Clays and human health. In Handbook of Clay Science; Bergaya, F., Theng, B.K.G., Lagaly, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 1, pp. 717–741. [Google Scholar]
- Carretero, I.; Pozo, M. Mineralogia Applicada: Salud y Medio Ambiente; Paraninfo: Madrid, Spain, 2007; 424p. [Google Scholar]
- Tateo, F.; Summa, V. Element mobility in clays for healing use. Appl. Clay Sci. 2007, 36, 64–76. [Google Scholar] [CrossRef]
- Veniale, F.; Bettero, A.; Jobstraibizer, P.; Setti, M. Thermal muds: Perspectives of innovations. Appl. Clay Sci. 2007, 36, 141–147. [Google Scholar] [CrossRef]
- Viseras, C.; Aguzzi, C.; Cerezo, P.; López-Galindo, A. Uses of clay minerals in semisolid health care and therapeutic products. Appl. Clay Sci. 2007, 36, 37–50. [Google Scholar] [CrossRef]
- Dolmaa, G.; Tserenpil, S.; Ugtakhbayar, O.; Shevchenko, S.G.; Kliba, L.V.; Voronkov, M.G. Characterization and organic compounds in peloids from Mongolia. Proc. Mong. Acad. Sci. 2009, 49, 3–21. [Google Scholar] [CrossRef]
- Carretero, M.I.; Pozo, M.; Martín-Rubí, J.A.; Pozo, E.; Maraver, F. Mobility of elements in interaction between artificial sweat and peloids used in Spanish spa. Appl. Clay Sci. 2010, 48, 506–515. [Google Scholar] [CrossRef]
- Karakaya, M.; Karakaya, N.; Sarioglan, S.; Koral, M. Some properties of thermal muds of some spas in Turkey. Appl. Clay Sci. 2010, 48, 531–537. [Google Scholar] [CrossRef]
- Matike, D.M.E.; Ekosse, G.I.E.; Ngole, V.M. Physico-chemical properties of clayey soils used traditionally for cosmetics in Eastern Cape, South Africa. Int. J. Phys. Sci. 2011, 6, 7557–7566. [Google Scholar]
- Sánchez-Espejo, R.; Cerezo, P.; Aguzzi, C.; López-Galindo, A.; Machado, J.; Viseras, C. Physicochemical and in vitro cation release relevance of therapeutic muds “maturation”. Appl. Clay Sci. 2015, 116, 1–7. [Google Scholar] [CrossRef]
- Lagaly, G. Principles of flow of kaolin and bentonite dispersions. Appl. Clay Sci. 1989, 4, 105–123. [Google Scholar] [CrossRef]
- Bundy, W.M.; Ishley, J.N. Kaolin in paper filling and coating. Appl. Clay Sci. 1991, 5, 397–420. [Google Scholar] [CrossRef]
- Murray, H.H.; Keller, W.D. Kaolins, kaolins, kaolins. In Kaolin Genesis and Utilization; Murray, H.H., Bundy, W., Harvey, C., Eds.; The Clay Minerals Society: Boulder, CO, USA, 1993; Spec. Pub.1; pp. 1–24. [Google Scholar]
- De Bernardi, M.; Pedrinazzi, G.M. Azioni biologiche dei fanghi termali. In Atti Convegno Argille Curative; Veniale, F., Ed.; Gruppo Italiano AIPEA: Pavia, Italy, 1996; pp. 17–24. [Google Scholar]
- Yuan, J.; Murray, H.H. The importance of crystal morphology on the viscosity of concentrated suspensions of kaolins. Appl. Clay Sci. 1997, 12, 209–219. [Google Scholar] [CrossRef]
- Minguzzi, N.; Morandi, N.; Tagnin, S.; Tateo, F. Le argille curative in uso negli stabilimenti termali Emiliano-Romagnoli: Verifica della composizione e delle proprieta. In Argille Per Fanghi Peloidi Termali E Per Trattamenti Dermatologici E Cosmetici; Veniale, F., Ed.; Symp. Gruppo Ital. AIPEA: Montecatini Terme, Italy, 1999; Volume XLII, pp. 287–298. [Google Scholar]
- Veniale, F. Argille Curative Per Fanghi Peloidi Termali E Per Trattamienti Dermatologici E Cosmetici; Symp. Gruppo Ital. AIPEA: Montecatini Terme, Italy, 1999; Volume XLII, pp. 261–335. [Google Scholar]
- Millman, N. Some factors that influence the viscosity of paper coating compositions. Tappi 1964, 47, 168A–175A. [Google Scholar]
- Ferrand, T.; Yvon, J. Thermal properties of clay pastes for pelotherapy. Appl. Clay Sci. 2000, 6, 751–760. [Google Scholar] [CrossRef]
- Legido, J.L.; Mourelle, M.L. Investigaciones En El Ámbito Iberoamericano Sobre Peloides Termales; Universidad de Vigo: Vigo, Spain, 2008; p. 308. [Google Scholar]
- Armijo, F.; Maraver, F.; Pozo, M.; Carretero, M.I.; Armijo, O.; Fernández-Torán, M.A.; Fernández-González, M.V.; Corvillo, I. Thermal behaviour of clays and clay-water mixtures for pelotherapy. Appl. Clay Sci. 2016, 126, 50–56. [Google Scholar] [CrossRef]
- Awad, M.E.; López-Galindo, A.; Setti, M.; El-Rahmany, M.M.; Iborra, C.V. Kaolinite in pharmaceutics and biomedicine. Int. J. Pharm. 2017, 533, 34–48. [Google Scholar] [CrossRef]
- Ramasamy, S.; Hussin, K.; Abdullah, M.M.A.; Ruzaidi, C.M.; Binhussain, M.; Sandu, A.V. Effect of solid/liquid ratio on mechanical properties of kaolin coated teak wood via geopolymer technology. App. Mech. Mater. 2015, 754, 708–713. [Google Scholar] [CrossRef]
- Awad, M.E.; López-Galindo, A.; Sánchez-Espejo, R.; El-Rahmany, M.M.; Iborra, C.V. Thermal properties of some Egyptian kaolin pastes for pelotherapeutic applications: Influence of particle geometry on thermal dosage release. Appl. Clay Sci. 2018, 160, 193–200. [Google Scholar] [CrossRef]
- Awad, M.E.; López-Galindo, A.; Sánchez-Espejo, R.; Sainz-Díaz, C.I.; El-Rahmany, M.M.; Viseras, C. Crystallite size as a function of kaolinite structural order-disorder and kaolin chemical variability: Sedimentological implication. Appl. Clay Sci. 2018, 162, 261–267. [Google Scholar] [CrossRef]
- Hernández, A.C.; Sánchez-Espejo, R.; Meléndez, W.; González, G.; López-Galindo, A.; Viseras, C. Characterization of Venezuelan kaolins as health care ingredients. Appl. Clay Sci. 2019, 175, 30–39. [Google Scholar] [CrossRef]
- Williams, D.J.A.; Williams, K.P. Electrophoresis and zeta potential of kaolinite. J. Colloid Interface Sci. 1977, 65, 79–87. [Google Scholar] [CrossRef]
- Schott, H. Reología. In Remington. Farmacia; Gennaro, A.R., Ed.; Editorial Médica Panamericana: Buenos Aires, Argentina, 1995; Volume I, pp. 426–455. [Google Scholar]
- Conceição, S.; Santos, N.F.; Velho, J.; Ferreira, J.M.F. Properties of paper coated with kaolin: The influence of the rheological modifier. Appl. Clay Sci. 2005, 30, 165–173. [Google Scholar] [CrossRef]
- De Azerêdo, A.F.; Azeredo, G.; Carneiro, A.M. Study of rheological parameters of lime-metakaolin paste made of kaolin wastes and lime paste. Key Eng. Mater. 2015, 668, 419–432. [Google Scholar] [CrossRef]
- Awad, M.E.; López-Galindo, A.; El Rahmany, M.M.; El-Desoky, H.M.; Viseras, C. Characterization of Egyptian kaolins for health-care uses. Appl. Clay Sci. 2017, 135, 176–189. [Google Scholar] [CrossRef]
- Qiu, X.; Liu, Y.; Alshameri, A.; Zhu, X.; Yan, C. Viscosity of kaolin slurries: Effects of dispersant and urea-intercalation. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2017, 32, 51–57. [Google Scholar] [CrossRef]
- Aguzzi, C.; Sánchez-Espejo, R.; Cerezo, P.; Machado, J.; Bonferoni, M.C.; Rossi, S.; Salcedo, I.; Viseras, C. Networking and rheology of concentrated clay suspensions “matured” in mineral medicinal water. Int. J. Pharm. 2013, 453, 473–479. [Google Scholar] [CrossRef]
- Modabberi, S.; Namayandeh, A.; López-Galindo, A.; Viseras, C.; Setti, M.; Ranjbaran, M. Characterization of Iranian bentonites to be used as pharmaceutical materials. Appl. Clay Sci. 2015, 116, 193–201. [Google Scholar] [CrossRef]
- Sánchez-Espejo, R.; Aguzzi, C.; Cerezo, P.; Salcedo, I.; López-Galindo, A.; Viseras, C. Folk pharmaceutical formulations in western Mediterranean: Identification and safety of clays used in pelotherapy. J. Ethnopharmacol. 2014, 155, 810–814. [Google Scholar] [CrossRef] [PubMed]
- Conde-Pumpido, R.; Ferrón, J.J.; Campillo, G. Influence of granulometric factors on the rheology of kaolins of Galicia, Spain. Appl. Clay Sci. 1988, 3, 177–185. [Google Scholar] [CrossRef]
- Wierenga, A.M.; Lenstra, T.A.J.; Philipse, A.P. Aqueous dispersions of colloidal gibbsite platelets: Synthesis, characterisation and intrinsic viscosity measurements. Colloids Surf. Physicochem. Eng. Asp. 1998, 134, 359–371. [Google Scholar] [CrossRef]
- Lambers, H.; Piessens, S.; Bloem, A.; Pronk, H.; Finkel, P. Natural skin surface pH is on average below 5, which is beneficial for its resident flora. Int. J. Cosm. Sci. 2006, 28, 359–370. [Google Scholar] [CrossRef]
- Fluhr, J.W.; Elias, P.M. Stratum corneum pH: Formation and function of the ‘acid mantle’. Exog. Dermatol. 2002, 1, 163–175. [Google Scholar] [CrossRef]
- Driscoll, C.T.; Postek, K.M. The chemistry of aluminum in surface waters. In The Environmental Chemistry of Aluminum, 2nd ed.; Sposito, G., Ed.; Lewis Publishers: New York, NY, USA, 1996; pp. 363–418. [Google Scholar]
- Krewski, D.; Yokel, R.A.; Nieboer, E.; Borchelt, D.; Cohen, J.; Harry, J.; Kacew, S.; Lindsay, J.; Mahfouz, A.M.; Rondeau, V. Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J. Toxicol. Environ. Health B 2007, 10, 1–269. [Google Scholar] [CrossRef]
- Becker, L.C.; Boyer, I.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; et al. Safety assessment of alumina and aluminum hydroxide as used in Cosmetics. Int. J. Toxicol. 2016, 35, 16S–33S. [Google Scholar] [CrossRef]
- Yukselen, Y.; Kaya, A. A study of factors affecting on the zeta potential of kaolinite and quartz powder. Environ. Earth Sci. 2011, 62, 697–705. [Google Scholar] [CrossRef]
- Qiu, X.; Lei, X.; Alshameri, A.; Wang, H.; Yan, C. Comparison of the physicochemical properties and mineralogy of Chinese (Beihai) and Brazilian kaolin. Ceram. Int. 2014, 40, 5397–5405. [Google Scholar] [CrossRef]
- Ndlovu, B.; Farrokhpay, S.; Forbes, E.; Bradshaw, D. Characterisation of kaolinite colloidal and flow behaviour via crystallinity measurements. Powder Technol. 2015, 269, 505–512. [Google Scholar] [CrossRef]
- Usuga-Manco, L.M.; López-Valdivieso, A.; Bustamante-Rúa, M.O. Hydrophobicity study of kaolinite from La Unión, Antioquia. TecnoLógicas 2015, 18, 71–81. [Google Scholar] [CrossRef]
- Au, P.I.; Leong, Y.K. Surface chemistry and rheology of slurries of kaolinite and montmorillonite from different sources. Kona Powder Part. J. 2016, 33, 17–32. [Google Scholar] [CrossRef]
- Adekola, F.; Fédoroff, M.; Geckeis, H.; Kupcik, T.; Lefèvre, G.; Lützenkirchen, J.; Plaschke, M.; Preocanin, T.; Rabung, T.; Schild, D. Characterization of acid–base properties of two gibbsite samples in the context of literature results. J. Coll. Interface Sci. 2011, 354, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Cunha, F.O.; Torem, M.L.; D’Abreu, J.C. On the fundamentals of kaolin rheology applied to the paper industry. Miner. Eng. 2006, 19, 1462–1464. [Google Scholar] [CrossRef]
- Du, J.; Morris, G.; Pushkarova, R.A.; Smart, R.S.C. Effect of surface structure of kaolinite on aggregation, settling rate, and bed density. Langmuir 2010, 26, 13227–13235. [Google Scholar] [CrossRef] [PubMed]
- Yukselen, Y.; Kaya, A. Zeta potential of kaolinite in the presence of alkali, alkaline earth and hydrolyzable metal ions. Waterairand Soil Pollut. 2003, 145, 155–168. [Google Scholar] [CrossRef]
- Bolland, M.D.A.; Posner, A.M.; Quirk, J.P. pH-independent and pH-dependent surface charges on kaolinite. Clays Clay Miner. 1980, 28, 412–418. [Google Scholar] [CrossRef]
- Atesok, G.; Somasundaran, P.; Morgan, L.J. Adsorption properties of Ca2+ on Na-kaolinite and its effect on flocculation using polyacrylamides. Colloids Surf. 1988, 32, 127–138. [Google Scholar] [CrossRef]
- Legido, J.L.; Medina, C.; Mourelle, M.L.; Carretero, M.I.; Pozo, M. Comparative study of the cooling rates of bentonite, sepiolite and common clays for their use in pelotherapy. Appl. Clay Sci. 2007, 36, 148–160. [Google Scholar] [CrossRef]
| Samples | Kaolinite (%) | HI | Associated Mineral Impurities | Fe2O3 (%) | Desorbed Cations (mg/L) | |
|---|---|---|---|---|---|---|
| Ca2+ | Mg2+ | |||||
| VE1 | 75 | 1.2 | Gibbsite (20%), Fe–Ti oxide minerals (4%), quartz (1%) | 1.79 | 29.8 | 8.59 |
| VE2 | 92 | 0.7 | Gibbsite (4%), Fe–Ti oxide minerals (3%), quartz (1%) | 0.70 | 70.57 | 75.08 |
| VE3 | 94 | 1.3 | Gibbsite (5%), anatase (1%) | 0.34 | 26.33 | 46.92 |
| VE4 | 99 | 1.2 | Gibbsite (1%). | 0.37 | 0.77 | - |
| VE5 | 88 | 1.4 | Mica (5%), quartz (6%), anatase (1%) | 0.37 | - | - |
| VE6 | 92 | 1.4 | Gibbsite (5%), Fe–Ti oxide minerals (2%), quartz (1%) | 2.06 | 4.61 | - |
| VE9 | 94 | 0.9 | Quartz (4%), Fe–Ti oxide minerals (2%) | 0.81 | - | - |
| VE12 | 73 | 0.9 | Gibbsite (13%), quartz (3%), zircon (2%), Fe–Ti oxide minerals (9%). | 7.76 | 155.80 | 47.08 |
| Samples | Flow Character | Viscosity (Pa·s) ± S.D. (n = 4) | Suspension pH ± S.D. (n = 3) | D50 (μm) |
|---|---|---|---|---|
| VE1 | Pseudoplastic | 1.82 ± 0.10 | 4.30 ± 0.01 | 1.05 |
| VE2 | Dilatant | 0.70 ± 0.05 | 4.50 ± 0.02 | 1.12 |
| VE3 | Dilatant | 1.13 ± 0.30 | 4.40 ± 0.07 | 1.24 |
| VE4 | Dilatant | 1.44 ± 0.20 | 4.52 ± 0.09 | 1.55 |
| VE5 | Dilatant | 0.32 ± 0.10 | 4.50 ± 0.04 | 2.29 |
| VE6 | Dilatant | 2.98 ± 0.20 | 4.70 ± 0.05 | 5.1 |
| VE9 | Pseudoplastic | 3.96 ± 0.20 | 5.90 ± 0.12 | 1.81 |
| VE12 | Dilatant | 0.03 ± 0.10 | 3.86 ± 0.89 | 1.83 |
| Sample | Cp (J/K·g) | t32 °C (min) | T20 min (°C) | |||
|---|---|---|---|---|---|---|
| Theor. | Exp. | Theor. | Exp. | Theor. | Exp. | |
| VE1 | 2.49 | 2.64 | 25.4 | 24.3 | 34.2 | 34.1 |
| VE2 | 2.55 | 2.42 | 24.1 | 23.6 | 33.7 | 33.7 |
| VE3 | 2.55 | 2.75 | 24.1 | 23.7 | 33.7 | 33.7 |
| VE4 | 2.56 | 2.61 | 23.5 | 23.0 | 33.4 | 33.4 |
| VE5 | 2.55 | 2.97 | 27.4 | 25.9 | 34.8 | 34.8 |
| VE6 | 2.54 | 3.00 | 26.5 | 26.1 | 34.7 | 34.7 |
| VE9 | 2.55 | 2.62 | 27.0 | 26.2 | 34.8 | 34.8 |
| VE12 | 2.47 | 2.78 | 27.0 | 26.1 | 35.0 | 35.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández, A.C.; Awad, M.E.; Meléndez, W.; González, G.; López-Galindo, A.; Sánchez-Espejo, R.; García-Villén, F.; Viseras, C. Colloidal and Thermal Behaviors of Some Venezuelan Kaolin Pastes for Therapeutic Applications. Minerals 2019, 9, 756. https://doi.org/10.3390/min9120756
Hernández AC, Awad ME, Meléndez W, González G, López-Galindo A, Sánchez-Espejo R, García-Villén F, Viseras C. Colloidal and Thermal Behaviors of Some Venezuelan Kaolin Pastes for Therapeutic Applications. Minerals. 2019; 9(12):756. https://doi.org/10.3390/min9120756
Chicago/Turabian StyleHernández, Ana C., Mahmoud E. Awad, Williams Meléndez, Gema González, Alberto López-Galindo, Rita Sánchez-Espejo, Fátima García-Villén, and César Viseras. 2019. "Colloidal and Thermal Behaviors of Some Venezuelan Kaolin Pastes for Therapeutic Applications" Minerals 9, no. 12: 756. https://doi.org/10.3390/min9120756
APA StyleHernández, A. C., Awad, M. E., Meléndez, W., González, G., López-Galindo, A., Sánchez-Espejo, R., García-Villén, F., & Viseras, C. (2019). Colloidal and Thermal Behaviors of Some Venezuelan Kaolin Pastes for Therapeutic Applications. Minerals, 9(12), 756. https://doi.org/10.3390/min9120756






