Low-Carbon Binder for Cemented Paste Backfill: Flowability, Strength and Leaching Characteristics
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Result and Discussion
3.1. Mix Proportion for Binder
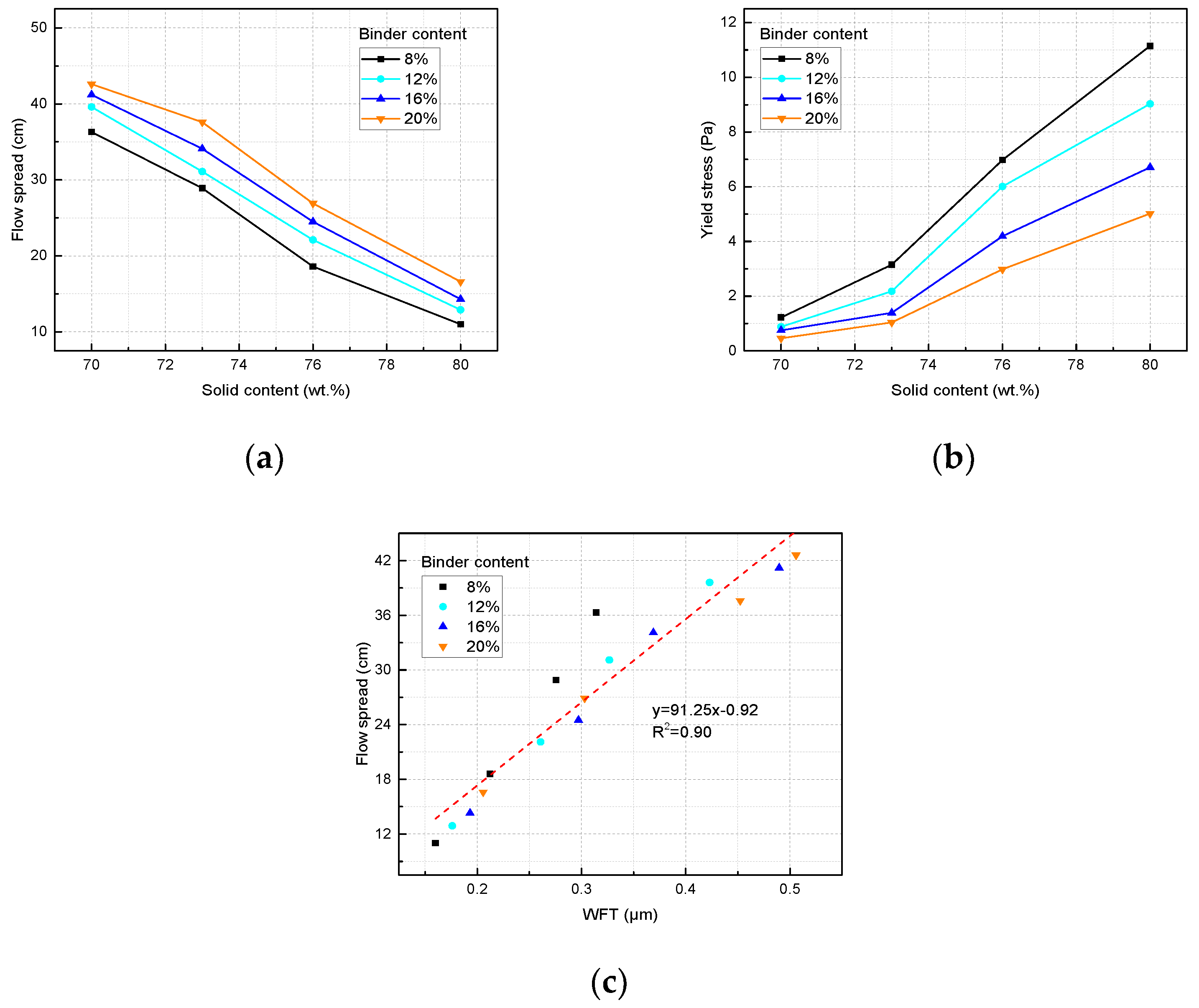
3.2. Flowability of the CPB Slurry
3.3. Strength Properties
3.3.1. Multivariate Analysis
3.3.2. Sensitivity Analysis
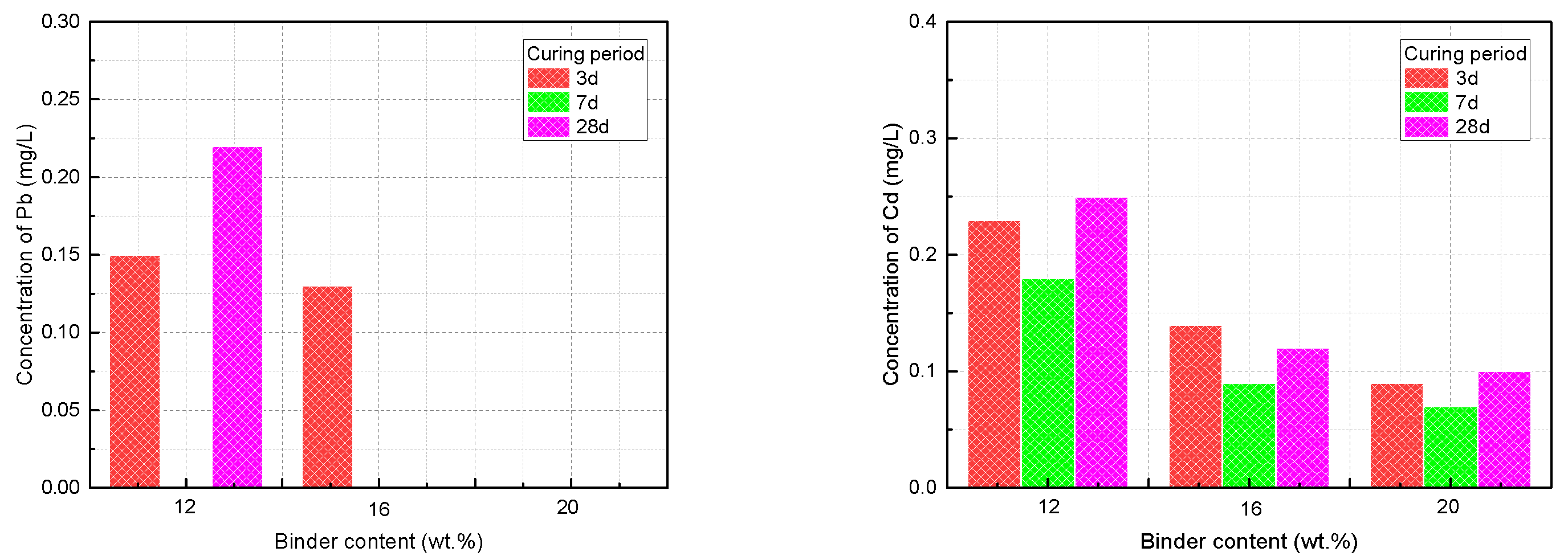
3.4. Leaching Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Luukkonen, T.; Abdollahnejad, Z.; Yliniemi, J.; Kinnunen, P.; Illikainen, M. One-part alkali-activated materials: A review. Cem. Concr. Res. 2018, 103, 21–34. [Google Scholar] [CrossRef]
- Zhu, W.; Teoh, P.J.; Liu, Y.; Chen, Z.; Yang, E.-H. Strategic utilization of municipal solid waste incineration bottom ash for the synthesis of lightweight aerated alkali-activated materials. J. Clean. Prod. 2019, 235, 603–612. [Google Scholar] [CrossRef]
- Zhu, W.; Chen, X.; Zhao, A.; Struble, L.J.; Yang, E.-H. Synthesis of high strength binders from alkali activation of glass materials from municipal solid waste incineration bottom ash. J. Clean. Prod. 2019, 212, 261–269. [Google Scholar] [CrossRef]
- Zhang, X.; Han, Y.; Sun, Y.; Li, Y. Innovative utilization of refractory iron ore via suspension magnetization roasting: A pilot-scale study. Powder Technol. 2019, 352, 16–24. [Google Scholar] [CrossRef]
- Jiang, H.; Qi, Z.; Yilmaz, E.; Han, J.; Qiu, J.; Dong, C. Effectiveness of alkali-activated slag as alternative binder on workability and early age compressive strength of cemented paste backfills. Constr. Build. Mater. 2019, 218, 689–700. [Google Scholar] [CrossRef]
- Ishwarya, G.; Singh, B.; Deshwal, S.; Bhattacharyya, S.K. Effect of sodium carbonate/sodium silicate activator on the rheology, geopolymerization and strength of fly ash/slag geopolymer pastes. Cem. Concr. Compos. 2019, 97, 226–238. [Google Scholar] [CrossRef]
- Cihangir, F.; Ercikdi, B.; Kesimal, A.; Turan, A.; Deveci, H. Utilisation of alkali-activated blast furnace slag in paste backfill of high-sulphide mill tailings: Effect of binder type and dosage. Miner. Eng. 2012, 30, 33–43. [Google Scholar] [CrossRef]
- Zhao, Y.; Soltani, A.; Taheri, A.; Karakus, M.; Deng, A. Application of Slag–Cement and Fly Ash for Strength Development in Cemented Paste Backfills. Minerals 2019, 9, 22. [Google Scholar] [CrossRef]
- GB 16889-2008. Domestic Waste Landfill Control Standards; Chinese Academy of Environmental Sciences: Beijing, China, 2008. [Google Scholar]
- ASTM D2166/D2166M-16. Standard Test Method for Unconfined Compressive Strength of Cohesive Soil; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- ASTM C1437-15. Standard Test Method for Flow of Hydraulic Cement Mortar; ASTM International: West Conshohocken, PA, USA, 2015. [Google Scholar]
- Kashani, A.; Provis, J.L.; Qiao, G.G.; van Deventer, J.S.J. The interrelationship between surface chemistry and rheology in alkali activated slag paste. Constr. Build. Mater. 2014, 65, 583–591. [Google Scholar] [CrossRef]
- Guo, B.; Liu, B.; Yang, J.; Zhang, S. The mechanisms of heavy metal immobilization by cementitious material treatments and thermal treatments: A review. J. Environ. Manag. 2017, 193, 410–422. [Google Scholar] [CrossRef]
- Tu, W.; Zhu, Y.; Fang, G.; Wang, X.; Zhang, M. Internal curing of alkali-activated fly ash-slag pastes using superabsorbent polymer. Cem. Concr. Res. 2019, 116, 179–190. [Google Scholar] [CrossRef]
- Thomas, J.J.; Allen, A.J.; Jennings, H.M. Density and water content of nanoscale solid C–S–H formed in alkali-activated slag (AAS) paste and implications for chemical shrinkage. Cem. Concr. Res. 2012, 42, 377–383. [Google Scholar] [CrossRef]
- Lee, N.K.; Jang, J.G.; Lee, H.K. Shrinkage characteristics of alkali-activated fly ash/slag paste and mortar at early ages. Cem. Concr. Compos. 2014, 53, 239–248. [Google Scholar] [CrossRef]
- Qiu, J.; Yang, L.; Sun, X.; Xing, J.; Li, S. Strength Characteristics and Failure Mechanism of Cemented Super-Fine Unclassified Tailings Backfill. Minerals 2017, 7, 58. [Google Scholar] [CrossRef]
- Yang, L.; Qiu, J.; Jiang, H.; Hu, S.; Li, H.; Li, S. Use of Cemented Super-Fine Unclassified Tailings Backfill for Control of Subsidence. Minerals 2017, 7, 216. [Google Scholar] [CrossRef]
- Nath, P.; Sarker, P.K. Use of OPC to improve setting and early strength properties of low calcium fly ash geopolymer concrete cured at room temperature. Cem. Concr. Compos. 2015, 55, 205–214. [Google Scholar] [CrossRef]
- Görhan, G.; Kürklü, G. The influence of the NaOH solution on the properties of the fly ash-based geopolymer mortar cured at different temperatures. Compos. Part B Eng. 2014, 58, 371–377. [Google Scholar] [CrossRef]
- Rattanasak, U.; Chindaprasirt, P. Influence of NaOH solution on the synthesis of fly ash geopolymer. Miner. Eng. 2009, 22, 1073–1078. [Google Scholar] [CrossRef]
- Phoo-ngernkham, T.; Maegawa, A.; Mishima, N.; Hatanaka, S.; Chindaprasirt, P. Effects of sodium hydroxide and sodium silicate solutions on compressive and shear bond strengths of FA–GBFS geopolymer. Constr. Build. Mater. 2015, 91, 1–8. [Google Scholar] [CrossRef]
- Puertas, F.; Fernández-Jiménez, A.; Blanco-Varela, M.T. Pore solution in alkali-activated slag cement pastes. Relation to the composition and structure of calcium silicate hydrate. Cem. Concr. Res. 2004, 34, 139–148. [Google Scholar] [CrossRef]
- Zuo, Y.; Nedeljković, M.; Ye, G. Pore solution composition of alkali-activated slag/fly ash pastes. Cem. Concr. Res. 2019, 115, 230–250. [Google Scholar] [CrossRef]
- Ahmari, S.; Zhang, L. Production of eco-friendly bricks from copper mine tailings through geopolymerization. Constr. Build. Mater. 2012, 29, 323–331. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, Y.; Jia, Y.; She, W.; Liu, G.; Wu, Z.; Sun, W. Influence of sodium hydroxide on the performance and hydration of lime-based low carbon cementitious materials. Constr. Build. Mater. 2019, 200, 604–615. [Google Scholar] [CrossRef]
- Mota, B.; Matschei, T.; Scrivener, K. Impact of NaOH and Na2SO4 on the kinetics and microstructural development of white cement hydration. Cem. Concr. Res. 2018, 108, 172–185. [Google Scholar] [CrossRef]
- Jeong, Y.; Oh, J.E.; Jun, Y.; Park, J.; Ha, J.-h.; Sohn, S.G. Influence of four additional activators on hydrated-lime [Ca(OH)2] activated ground granulated blast-furnace slag. Cem. Concr. Compos. 2016, 65, 1–10. [Google Scholar] [CrossRef]
- de la Grée, G.C.H.D.; Yu, Q.L.; Brouwers, H.J.H. Assessing the effect of CaSO4 content on the hydration kinetics, microstructure and mechanical properties of cements containing sugars. Constr. Build. Mater. 2017, 143, 48–60. [Google Scholar] [CrossRef]
- Tian, H.; Kong, X.; Su, T.; Wang, D. Comparative study of two PCE superplasticizers with varied charge density in Portland cement and sulfoaluminate cement systems. Cem. Concr. Res. 2019, 115, 43–58. [Google Scholar] [CrossRef]
- Park, H.; Jeong, Y.; Jun, Y.; Jeong, J.-H.; Oh, J.E. Strength enhancement and pore-size refinement in clinker-free CaO-activated GGBFS systems through substitution with gypsum. Cem. Concr. Compos. 2016, 68, 57–65. [Google Scholar] [CrossRef]
- Neto, A.A.M.; Cincotto, M.A.; Repette, W. Mechanical properties, drying and autogenous shrinkage of blast furnace slag activated with hydrated lime and gypsum. Cem. Concr. Compos. 2010, 32, 312–318. [Google Scholar] [CrossRef]
- Simon, D.; Grabinsky, M. Apparent yield stress measurement in cemented paste backfill. Int. J. Min. Reclam. Environ. 2013, 27, 231–256. [Google Scholar] [CrossRef]
- Steins, P.; Poulesquen, A.; Diat, O.; Frizon, F. Structural Evolution during geopolymerization from an early age to consolidated material. Langmuir 2012, 28, 8502–8510. [Google Scholar] [CrossRef] [PubMed]
- Yammine, J.; Chaouche, M.; Guerinet, M.; Moranville, M.; Roussel, N. From ordinary rhelogy concrete to self compacting concrete: A transition between frictional and hydrodynamic interactions. Cem. Concr. Res. 2008, 38, 890–896. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, A.; Tang, M.; Liu, X. The filling role of pozzolanic material. Cem. Concr. Res. 1996, 26, 943–947. [Google Scholar] [CrossRef]
- Fung, W.W.S.; Kwan, A.K.H. Role of water film thickness in rheology of CSF mortar. Cem. Concr. Compos. 2010, 32, 255–264. [Google Scholar] [CrossRef]
- Ji, Z.; Pei, Y. Bibliographic and visualized analysis of geopolymer research and its application in heavy metal immobilization: A review. J. Environ. Manag. 2019, 231, 256–267. [Google Scholar] [CrossRef]
- Xing, J.; Zhao, Y.; Qiu, J.; Sun, X. Microstructural and Mechanical Properties of Alkali Activated Materials from Two Types of Blast Furnace Slags. Materials (Basel) 2019, 12, 2089. [Google Scholar] [CrossRef]
- Qiu, J.; Zhao, Y.; Xing, J.; Sun, X. Fly Ash/Blast Furnace Slag-Based Geopolymer as a Potential Binder for Mine Backfilling: Effect of Binder Type and Activator Concentration. Adv. Mater. Sci. Eng. 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Li, Y.; Min, X.; Ke, Y.; Fei, J.; Liu, D.; Tang, C. Immobilization potential and immobilization mechanism of arsenic in cemented paste backfill. Miner. Eng. 2019, 138, 101–107. [Google Scholar] [CrossRef]
- Gineys, N.; Aouad, G.; Damidot, D. Managing trace elements in Portland cement—Part I: Interactions between cement paste and heavy metals added during mixing as soluble salts. Cem. Concr. Compos. 2010, 32, 563–570. [Google Scholar] [CrossRef]
- Qiu, J.; Zhao, Y.; Xing, J.; Sun, X. Fly ash-based geopolymer as a potential adsorbent for Cr(VI) removal. DWT 2017, 70, 201–209. [Google Scholar] [CrossRef]
- Rasaki, S.A.; Bingxue, Z.; Guarecuco, R.; Thomas, T.; Minghui, Y. Geopolymer for use in heavy metals adsorption, and advanced oxidative processes: A critical review. J. Clean. Prod. 2019, 213, 42–58. [Google Scholar] [CrossRef]
- Bumanis, G.; Novais, R.M.; Carvalheiras, J.; Bajare, D.; Labrincha, J.A. Metals removal from aqueous solutions by tailored porous waste-based granulated alkali-activated materials. Appl. Clay Sci. 2019, 179, 105147. [Google Scholar] [CrossRef]
- Shao, N.; Tang, S.; Liu, Z.; Li, L.; Yan, F.; Liu, F.; Li, S.; Zhang, Z. Hierarchically Structured Calcium Silicate Hydrate-Based Nanocomposites Derived from Steel Slag for Highly Efficient Heavy Metal Removal from Wastewater. ACS Sustain. Chem. Eng. 2018, 6, 14926–14935. [Google Scholar] [CrossRef]
- Kuwahara, Y.; Tamagawa, S.; Fujitani, T.; Yamashita, H. A novel conversion process for waste slag: synthesis of calcium silicate hydrate from blast furnace slag and its application as a versatile adsorbent for water purification. J. Mater. Chem. A 2013, 1, 7199. [Google Scholar] [CrossRef]
- Wu, J.; Zhu, Y.-J.; Chen, F. Ultrathin calcium silicate hydrate nanosheets with large specific surface areas: synthesis, crystallization, layered self-assembly and applications as excellent adsorbents for drug, protein, and metal ions. Small 2013, 9, 2911–2925. [Google Scholar] [CrossRef]
- Siyal, A.A.; Shamsuddin, M.R.; Rabat, N.E.; Zulfiqar, M.; Man, Z.; Low, A. Fly ash based geopolymer for the adsorption of anionic surfactant from aqueous solution. J. Clean. Prod. 2019, 229, 232–243. [Google Scholar] [CrossRef]
- Chen, X.; Guo, Y.; Ding, S.; Zhang, H.; Xia, F.; Wang, J.; Zhou, M. Utilization of red mud in geopolymer-based pervious concrete with function of adsorption of heavy metal ions. J. Clean. Prod. 2019, 207, 789–800. [Google Scholar] [CrossRef]
- Demir, F.; Derun, E.M. Modelling and optimization of gold mine tailings based geopolymer by using response surface method and its application in Pb2+ removal. J. Clean. Prod. 2019, 237, 117766. [Google Scholar] [CrossRef]
- Ferone, C.; Capasso, I.; Bonati, A.; Roviello, G.; Montagnaro, F.; Santoro, L.; Turco, R.; Coiifi, R. Sustainable management of water potabilization sludge by means of geopolymers production. J. Clean. Prod. 2019, 229, 1–9. [Google Scholar] [CrossRef]
- Novais, R.M.; Carvalheiras, J.; Tobaldi, D.M.; Seabra, M.P.; Pullar, R.C.; Labrincha, J.A. Synthesis of porous biomass fly ash-based geopolymer spheres for efficient removal of methylene blue from wastewaters. J. Clean. Prod. 2019, 207, 350–362. [Google Scholar] [CrossRef]
- Chrysochoou, M.; Dermatas, D. Evaluation of ettringite and hydrocalumite formation for heavy metal immobilization: literature review and experimental study. J. Hazard. Mater. 2006, 136, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Lasheras-Zubiate, M.; Navarro-Blasco, I.; Fernández, J.M.; Alvarez, J.I. Encapsulation, solid-phases identification and leaching of toxic metals in cement systems modified by natural biodegradable polymers. J. Hazard. Mater. 2012, 233, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Mollah, M.Y.A.; Kesmez, M.; Cocke, D.L. An X-ray diffraction (XRD) and Fourier transform infrared spectroscopic (FT-IR) investigation of the long-term effect on the solidification/stabilization (S/S) of arsenic(V) in Portland cement type-V. Sci. Total Environ. 2004, 325, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Macias, A.; Kindness, A.; Glasser, F.P. Impact of carbon dioxide on the immobilization potential of cemented wastes: Chromium. Cem. Concr. Res. 1997, 27, 215–225. [Google Scholar] [CrossRef]
- Baur, I.; Johnson, C.A. The solubility of selenate-AFt (3CaO·Al2O3·3CaSeO4·37.5H2O) and selenate-AFm (3CaO·Al2O3·CaSeO4·xH2O). Cem. Concr. Res. 2003, 33, 1741–1748. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Tyrer, M.; Hills, C.D.; Yang, X.M.; Carey, P. Immobilisation of heavy metal in cement-based solidification/stabilisation: A review. Waste Manag. 2009, 29, 390–403. [Google Scholar] [CrossRef]
- Zhang, M.; Reardon, E.J. Removal of B, Cr, Mo, and Se from wastewater by incorporation into hydrocalumite and ettringite. Environ. Sci. Technol. 2003, 37, 2947–2952. [Google Scholar] [CrossRef]
- Segni, R.; Vieille, L.; Leroux, F.; Taviot-Guého, C. Hydrocalumite-type materials: 1. Interest in hazardous waste immobilization. J. Phys. Chem. Solids 2006, 67, 1037–1042. [Google Scholar] [CrossRef]
- Van Jaarsveld, J.G.S.; Van Deventer, J.S.J.; Lorenzen, L. Factors affecting the immobilization of metals in geopolymerized flyash. Metall. Mater. Trans. B 1998, 29, 283–291. [Google Scholar] [CrossRef]
- Zhang, J.; Provis, J.L.; Feng, D.; van Deventer, J.S.J. Geopolymers for immobilization of Cr6+, Cd2+, and Pb2+. J. Hazard. Mater. 2008, 157, 587–598. [Google Scholar] [CrossRef]
- Palomo, A.; Palacios, M. Alkali-activated cementitious materials: Alternative matrices for the immobilisation of hazardous wastes: Part II. Stabilisation of chromium and lead. Cem. Concr. Res. 2003, 33, 289–295. [Google Scholar] [CrossRef]
- Perera, D.S.; Aly, Z.; Vance, E.R.; Mizumo, M. Immobilization of Pb in a Geopolymer Matrix. J. Am. Ceram. Soc. 2005, 88, 2586–2588. [Google Scholar] [CrossRef]
- Li, X.; Chen, Q.; Zhou, Y.; Tyrer, M.; Yu, Y. Stabilization of heavy metals in MSWI fly ash using silica fume. Waste Manag. 2014, 34, 2494–2504. [Google Scholar] [CrossRef]
- Ma, W.; Chen, D.; Pan, M.; Gu, T.; Zhong, L.; Chen, G.; Yan, B.; Cheng, Z. Performance of chemical chelating agent stabilization and cement solidification on heavy metals in MSWI fly ash: A comparative study. J. Environ. Manag. 2019, 247, 169–177. [Google Scholar] [CrossRef]
| Materials | CaO | SiO2 | Al2O3 | MgO | K2O | Fe2O3 | Na2O | BET Surface Area (m2/kg) |
|---|---|---|---|---|---|---|---|---|
| BFS | 48.83 | 35.46 | 8.99 | 3.07 | 0.87 | 0.63 | 0.51 | 567.87 |
| OPC | 57.21 | 21.65 | 5.84 | 4.79 | 0.45 | 1.77 | 0.86 | 586.22 |
| Gypsum | 38.85 | 0.29 | 0.01 | 2.32 | 0 | 0.06 | 0 | 521.09 |
| Heavy Metals | Leaching Concentration (mg/L) | GB 16889-2008 [9] (mg/L) |
|---|---|---|
| Cu | 0.55 ± 0.03 | 40.00 |
| Pb | 0.65 ± 0.06 | 0.25 |
| Zn | 2.68 ± 0.12 | 100.00 |
| Cd | 0.39 ± 0.04 | 0.15 |
| Factors | Level 1 | Level 2 | Level 3 |
|---|---|---|---|
| A: NaOH content (wt. %) | 3 | 4 | 5 |
| B: OPC dosage (wt. %) | 7 | 8 | 9 |
| C: gypsum content (wt. %) | 4 | 5 | 6 |
| No. T-j | A | B | C | Compressive Strength/MPa |
|---|---|---|---|---|
| T-1 | 1 (3%) | 1 (7%) | 1 (4%) | 29.55 |
| T-2 | 1 | 2 (8%) | 2 (5%) | 26.71 |
| T-3 | 1 | 3 (9%) | 3 (6%) | 22.00 |
| T-4 | 2 (4%) | 1 | 2 | 25.91 |
| T-5 | 2 | 2 | 3 | 22.66 |
| T-6 | 2 | 3 | 1 | 24.28 |
| T-7 | 3 (5%) | 1 | 3 | 26.75 |
| T-8 | 3 | 2 | 1 | 25.64 |
| T-9 | 3 | 3 | 2 | 21.68 |
| Kj1 | 78 | 82 | 79 | - |
| Kj2 | 72 | 75 | 74 | - |
| Kj3 | 74 | 67 | 71 | - |
| R | 1.8 | 4.8 | 2.7 | - |
| Optimal level | A1 | B1 | C1 | - |
| Order | B > C > A | - | ||
| A1B1C1 | - | |||
| Binder Dosage (wt. %) | Solid Content a (wt. %) | Flow Spread (cm) | Yield Stress (Pa) | WFT (μm) |
|---|---|---|---|---|
| 8 | 70 | 47.6 | 1.22 | 0.3311 |
| 73 | 38.6 | 3.15 | 0.2757 | |
| 76 | 26.9 | 6.98 | 0.2121 | |
| 80 | 16.6 | 11.15 | 0.1600 | |
| 12 | 70 | 39.6 | 0.82 | 0.4230 |
| 73 | 31.1 | 2.18 | 0.3268 | |
| 76 | 22.1 | 6.01 | 0.2609 | |
| 80 | 12.9 | 9.03 | 0.1762 | |
| 16 | 70 | 39.6 | 0.65 | 0.4897 |
| 73 | 31.1 | 1.39 | 0.3690 | |
| 76 | 22.1 | 4.19 | 0.2972 | |
| 80 | 12.9 | 6.71 | 0.1932 | |
| 20 | 70 | 42.6 | 0.56 | 0.5234 |
| 73 | 37.6 | 1.04 | 0.4523 | |
| 76 | 26.9 | 2.98 | 0.3031 | |
| 80 | 16.6 | 5.02 | 0.2057 |
| Binder Dosage (wt. %) | Solid Content (wt. %) | Compressive (MPa) | |||
|---|---|---|---|---|---|
| 3 Days | 7 Days | 14 Days | 28 Days | ||
| 20% | 70 | 0.69 | 1.12 | 2.26 | 2.72 |
| 73 | 0.94 | 1.55 | 2.52 | 2.85 | |
| 76 | 1.15 | 1.70 | 2.66 | 3.40 | |
| 80 | 1.68 | 2.64 | 3.77 | 4.87 | |
| 16% | 70 | 0.51 | 0.69 | 1.78 | 2.13 |
| 73 | 0.65 | 0.96 | 1.91 | 2.36 | |
| 76 | 0.89 | 1.48 | 2.52 | 3.03 | |
| 80 | 1.03 | 1.66 | 2.87 | 3.85 | |
| 12% | 70 | 0.64 | 0.85 | 0.92 | 1.15 |
| 73 | 0.84 | 0.91 | 1.08 | 1.26 | |
| 76 | 0.91 | 1.11 | 1.27 | 1.66 | |
| 80 | 0.96 | 1.23 | 1.76 | 2.20 | |
| 8% | 70 | 0.26 | 0.44 | 0.60 | 0.90 |
| 73 | 0.46 | 0.61 | 0.78 | 1.17 | |
| 76 | 0.59 | 0.93 | 0.98 | 1.26 | |
| 80 | 0.89 | 1.02 | 1.23 | 1.61 | |
| Curing Period (d) | Fitting Equation | Standard Errors | Correlation Coefficient (R2) |
|---|---|---|---|
| 3 | y1 = 27.5665 − 19.2973x12 + 50.6791x22 − 24.0004x1 − 75.1738x2 + 45.9925x1x2 | 0.1368 | 0.9016 |
| 7 | y2 = 60.3656 − 27.5251x12 + 105.9263x22 − 53.7166x1 − 160.9699x2 + 94.3906x1x2 | 0.1428 | 0.9578 |
| 14 | y3 = 100.3304 − 112.6050x12 + 192.4251x22 + 5.1676x1 − 282.8351x2 + 64.1722x1x2 | 0.1807 | 0.9614 |
| 28 | y4 = 151.7099 − 129.9277x12 + 280.8141 x22 − 7.3326x1 − 418.3366x2 + 119.5032x1x2 | 0.2525 | 0.9549 |
| Binder Dosage (Wt. %) | Solid Content (Wt. %) | 3 Days | 7 Days | 14 Days | 28 Days | ||||
|---|---|---|---|---|---|---|---|---|---|
| Predicted Value | Deviation | Predicted Value | Deviation | Predicted Value | Deviation | Predicted Value | Deviation | ||
| 20% | 70% | 0.620 | 0.069 | 0.960 | 0.160 | 2.118 | 0.141 | 2.432 | 0.287 |
| 73% | 0.884 | 0.056 | 1.483 | 0.067 | 2.369 | 0.150 | 2.826 | 0.024 | |
| 76% | 1.239 | 0.089 | 1.896 | 0.196 | 2.867 | 0.207 | 3.624 | 0.224 | |
| 80% | 1.854 | 0.174 | 2.813 | 0.173 | 4.103 | 0.333 | 5.408 | 0.538 | |
| 16% | 70% | 0.607 | 0.097 | 0.785 | 0.095 | 1.854 | 0.074 | 2.250 | 0.120 |
| 73% | 0.757 | 0.105 | 0.873 | 0.087 | 1.945 | 0.035 | 2.345 | 0.014 | |
| 76% | 0.996 | 0.106 | 1.450 | 0.029 | 2.382 | 0.137 | 2.945 | 0.085 | |
| 80% | 1.158 | 0.128 | 1.851 | 0.191 | 3.204 | 0.334 | 4.331 | 0.481 | |
| 12% | 70% | 0.613 | 0.027 | 0.705 | 0.144 | 1.135 | 0.215 | 1.383 | 0.233 |
| 73% | 0.792 | 0.048 | 0.874 | 0.036 | 1.146 | 0.066 | 1.427 | 0.167 | |
| 76% | 0.775 | 0.135 | 1.034 | 0.076 | 1.403 | 0.133 | 1.878 | 0.218 | |
| 80% | 1.109 | 0.149 | 1.477 | 0.247 | 2.118 | 0.358 | 2.765 | 0.565 | |
| 8% | 70% | 0.340 | 0.080 | 0.551 | 0.111 | 0.516 | 0.083 | 0.805 | 0.094 |
| 73% | 0.461 | 0.001 | 0.549 | 0.061 | 0.579 | 0.201 | 0.961 | 0.209 | |
| 76% | 0.609 | 0.019 | 0.738 | 0.191 | 0.788 | 0.191 | 1.022 | 0.238 | |
| 80% | 0.948 | 0.058 | 1.186 | 0.166 | 1.538 | 0.308 | 2.089 | 0.479 | |
| No. | ζ01(k) | ζ02(k) | ζ03(k) |
|---|---|---|---|
| 1 | 1.000 | 1.000 | 1.000 |
| 2 | 0.763 | 0.979 | 0.769 |
| 3 | 0.584 | 0.832 | 0.592 |
| 4 | 0.399 | 0.638 | 0.404 |
| 5 | 0.962 | 0.958 | 0.949 |
| 6 | 0.923 | 0.751 | 1.000 |
| 7 | 0.535 | 0.888 | 0.571 |
| 8 | 0.519 | 0.448 | 0.548 |
| 9 | 0.831 | 0.926 | 0.945 |
| 10 | 0.758 | 0.879 | 0.863 |
| 11 | 0.828 | 0.546 | 0.923 |
| 12 | 0.752 | 0.348 | 0.837 |
| 13 | 0.819 | 0.933 | 0.965 |
| 14 | 0.809 | 0.803 | 0.939 |
| 15 | 0.846 | 0.532 | 0.979 |
| 16 | 0.816 | 0.333 | 0.930 |
| Correlation degree | 0.759 | 0.737 | 0.826 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, J.; Zhao, Y.; Long, H.; Guo, Z.; Xing, J.; Sun, X. Low-Carbon Binder for Cemented Paste Backfill: Flowability, Strength and Leaching Characteristics. Minerals 2019, 9, 707. https://doi.org/10.3390/min9110707
Qiu J, Zhao Y, Long H, Guo Z, Xing J, Sun X. Low-Carbon Binder for Cemented Paste Backfill: Flowability, Strength and Leaching Characteristics. Minerals. 2019; 9(11):707. https://doi.org/10.3390/min9110707
Chicago/Turabian StyleQiu, Jingping, Yingliang Zhao, Hui Long, Zhenbang Guo, Jun Xing, and Xiaogang Sun. 2019. "Low-Carbon Binder for Cemented Paste Backfill: Flowability, Strength and Leaching Characteristics" Minerals 9, no. 11: 707. https://doi.org/10.3390/min9110707
APA StyleQiu, J., Zhao, Y., Long, H., Guo, Z., Xing, J., & Sun, X. (2019). Low-Carbon Binder for Cemented Paste Backfill: Flowability, Strength and Leaching Characteristics. Minerals, 9(11), 707. https://doi.org/10.3390/min9110707




