Comparative Analysis of Attachment to Chalcopyrite of Three Mesophilic Iron and/or Sulfur-Oxidizing Acidophiles
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Immobilization of Cells on AFM Cantilevers
2.3. Preparation of Chalcopyrite Coupons and Powder
2.4. Force Measurement and Bacterial Topography Analysis by AFM
2.5. Attachment of Planktonic Cells to Chalcopyrite Powder
2.6. Bioleaching of Chalcopyrite Powder
2.7. Visualization of Attachment to Chalcopyrite Coupons by Combined AFM and EFM
3. Results and Discussion
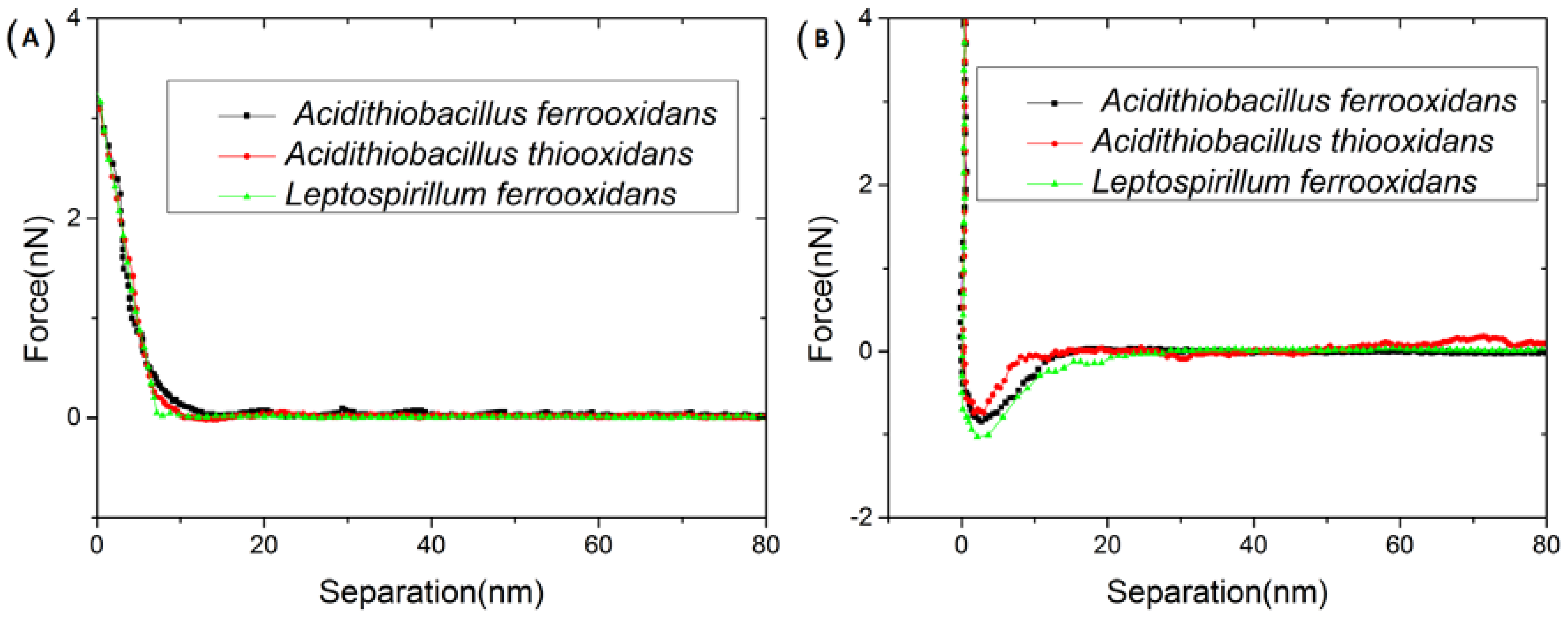
3.1. Interactions between Immobilized Cells and Surface of a Chalcopyrite Coupon
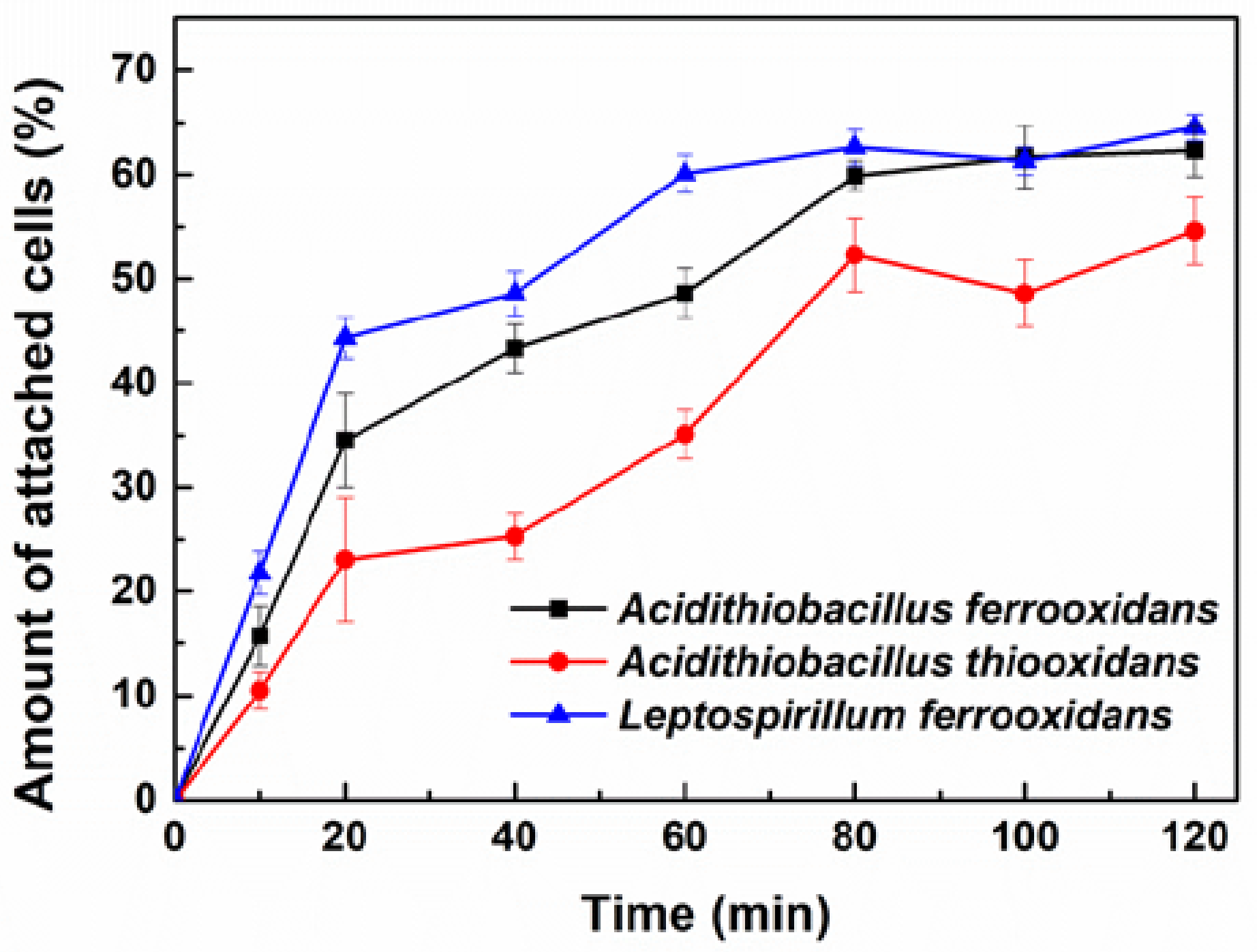
3.2. Attachment of Planktonic Cells to Chalcopyrite Powder
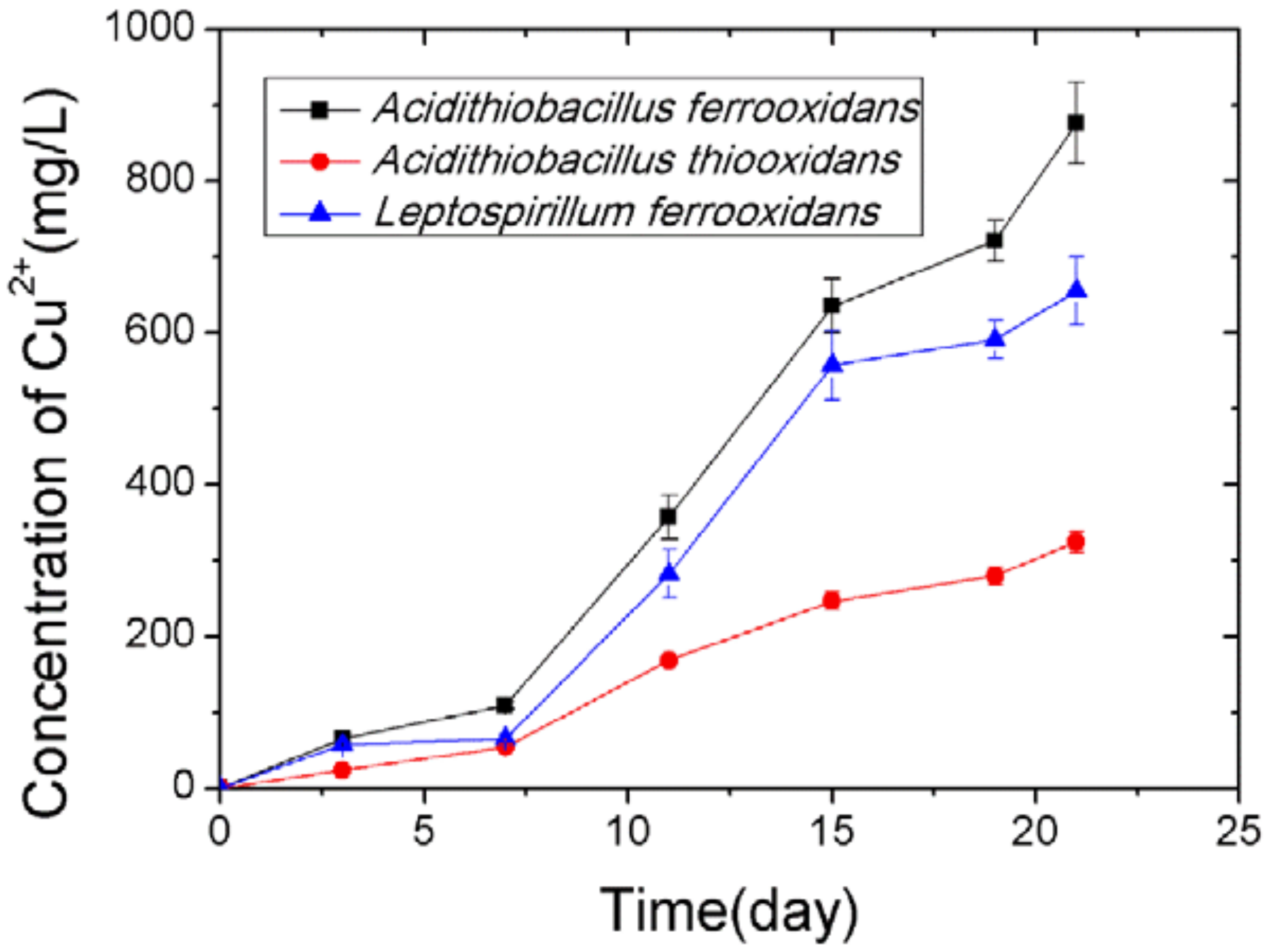
3.3. Bioleaching of Chalcopyrite Powder
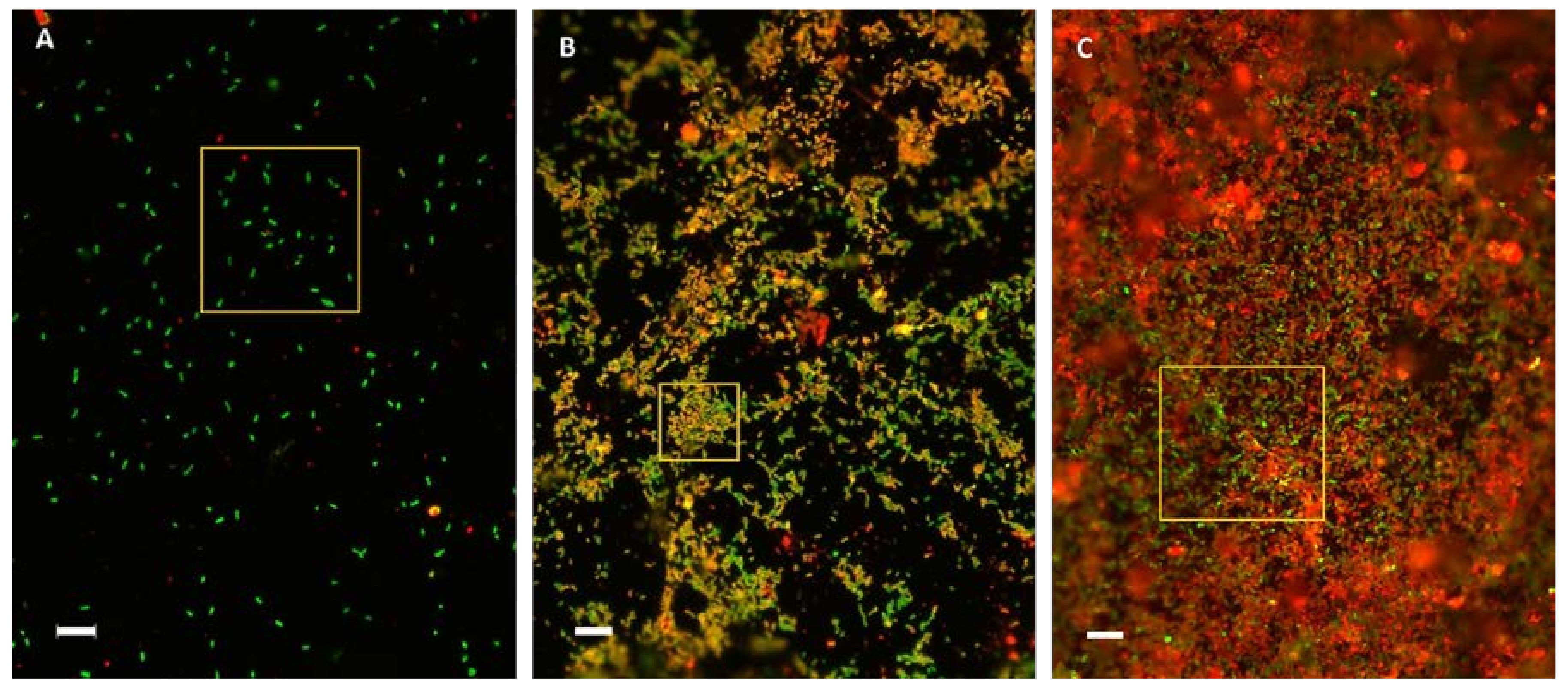
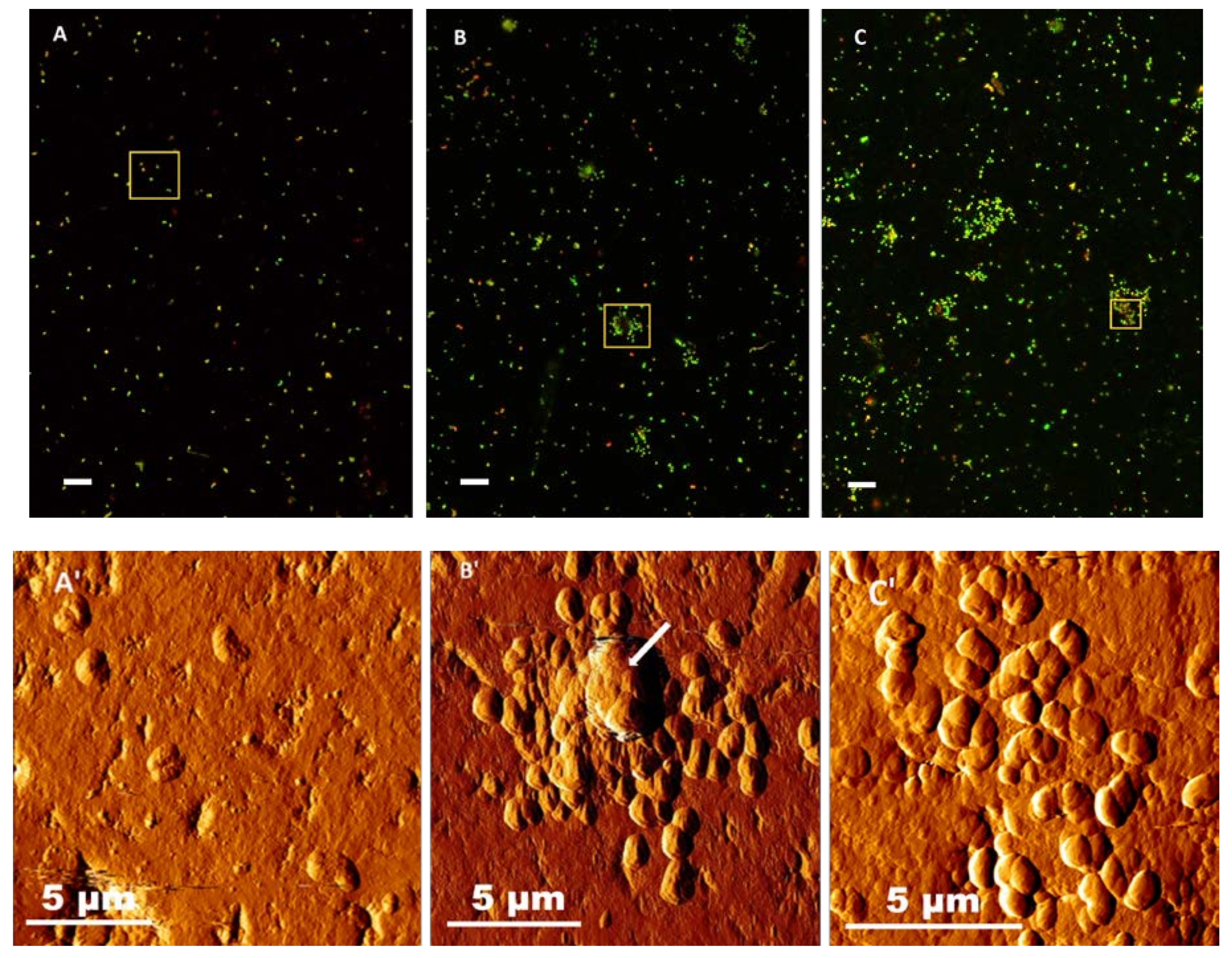
3.4. Visualization of Bacterial Biofilm Development on Chalcopyrite Coupons
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dreisinger, D. Copper leaching from primary sulfides: Options for biological and chemical extraction of copper. Hydrometallurgy 2006, 83, 10–20. [Google Scholar] [CrossRef]
- Hackl, R.; Dreisinger, D.; Peters, E.; King, J. Passivation of chalcopyrite during oxidative leaching in sulfate media. Hydrometallurgy 1995, 39, 25–48. [Google Scholar] [CrossRef]
- Pradhan, N.; Nathsarma, K.C.; Rao, K.S.; Sukla, L.B.; Mishra, B.K. Heap bioleaching of chalcopyrite: A review. Miner. Eng. 2008, 21, 355–365. [Google Scholar] [CrossRef]
- Ding, J.N.; Gao, J.; Wu, X.L.; Zhang, C.G.; Wang, D.Z.; Qiu, G.Z. Jarosite-type precipitates mediated by YN22, Sulfobacillus thermosulfidooxidans, and their influences on strain. Trans. Nonferrous Met. Soc. China 2007, 17, 1038–1044. [Google Scholar] [CrossRef]
- Li, H.J.; Yang, H.Y.; Chen, G.B. Catalytic performance of biological method seeds on jarosite process. Trans. Nonferrous Met. Soc. China 2016, 26, 557–564. [Google Scholar] [CrossRef]
- Vera, M.; Schippers, A.; Sand, W. Progress in bioleaching: fundamentals and mechanisms of bacterial metal sulfide oxidation-part A. Appl. Microbiol. Biotechnol. 2013, 97, 7529–7541. [Google Scholar] [CrossRef] [PubMed]
- Gan, M.; Jie, S.; Li, M.; Zhu, J.; Liu, X. Bioleaching of multiple metals from contaminated sediment by moderate thermophiles. Mar. Pollut. Bull. 2015, 97, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Gan, M.; Zhou, S.; Li, M.; Zhu, J.; Liu, X.; Chai, L. Bioleaching of multiple heavy metals from contaminated sediment by mesophile consortium. Environ. Sci. Pollut. Res. 2015, 22, 5807–5816. [Google Scholar] [CrossRef] [PubMed]
- Rohwerder, T.; Gehrke, T.; Kinzler, K.; Sand, W. Bioleaching review part a. Appl. Microbiol. Biotechnol. 2003, 63, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Harneit, K.; Göksel, A.; Kock, D.; Klock, J.H.; Gehrke, T.; Sand, W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans. Hydrometallurgy 2006, 83, 245–254. [Google Scholar] [CrossRef]
- Zhang, R.; Bellenberg, S.; Neu, T.R.; Sand, W.; Vera, M. The biofilm lifestyle of acidophilic metal/sulfur-oxidizing microorganisms. In Biotechnology of Extremophiles: Advances and Challenges; Rampelotto, H.P., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 177–213. [Google Scholar]
- Sand, W.; Gehrke, T.; Jozsa, P.G.; Schippers, A. (bio) chemistry of bacterial leaching—Direct vs. Indirect bioleaching. Hydrometallurgy 2001, 59, 159–175. [Google Scholar] [CrossRef]
- Wang, Y.; Zeng, W.; Qiu, G.; Chen, X.; Zhou, H. A moderately thermophilic mixed microbial culture for bioleaching of chalcopyrite concentrate at high pulp density. Appl. Environ. Microbiol. 2014, 80, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Yang, C.; Lai, S.; Wang, J.; Liu, K.; Zhang, B. Bioleaching of chalcopyrite by moderately thermophilic microorganisms. Bioresour. Technol. 2013, 129, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Liu, X.; Zeng, J.; Yin, C.; Gao, J.; Liu, J.; Qiu, G. Mechanism of enhanced bioleaching efficiency of Acidithiobacillus ferrooxidans after adaptation with chalcopyrite. Hydrometallurgy 2008, 92, 95–101. [Google Scholar]
- Stoodley, P.; Sauer, K.; Davies, D.; Costerton, J.W. Biofilms as complex differentiated communities. Annu. Rev. Microbiol. 2002, 56, 187–209. [Google Scholar] [CrossRef] [PubMed]
- Busscher, H.J.; Norde, W.; van der Mei, H.C. Specific molecular recognition and nonspecific contributions to bacterial interaction forces. Appl. Environ. Microbiol. 2008, 74, 2559–2564. [Google Scholar] [CrossRef] [PubMed]
- Farahat, M.; Hirajima, T.; Sasaki, K. Adhesion of Ferroplasma acidiphilum onto pyrite calculated from the extended DLVO theory using the van Oss–Good–Chaudhury approach. J. Colloid Interface Sci. 2010, 349, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Hanumantha Rao, K. Adhesion of Paenibacillus polymyxa on chalcopyrite and pyrite: Surface thermodynamics and extended DLVO theory. Colloids Surf. B Biointerfaces 2003, 29, 21–38. [Google Scholar] [CrossRef]
- Farahat, M.; Hirajima, T.; Sasaki, K.; Doi, K. Adhesion of Escherichia coli onto quartz, hematite and corundum: Extended DLVO theory and flotation behavior. Colloids Surf. B Biointerfaces 2009, 74, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Dufrêne, Y.F. Understanding forces in biofilms. Nanomedicine 2015, 10, 1219–1221. [Google Scholar] [CrossRef] [PubMed]
- Sheng, X.; Ting, Y.P.; Pehkonen, S.O. Force measurements of bacterial adhesion on metals using a cell probe atomic force microscope. J. Colloid Interface Sci. 2007, 310, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Boonaert, C.J.; Dufrêne, Y.F.; Derclaye, S.R.; Rouxhet, P.G. Adhesion of Lactococcus lactis to model substrata: Direct study of the interface. Colloids Surf. B Biointerfaces 2001, 22, 171–182. [Google Scholar] [CrossRef]
- Ong, Y.L.; Razatos, A.; Georgiou, G.; Sharma, M.M. Adhesion forces between E. coli bacteria and biomaterial surfaces. Langmuir 1999, 15, 2719–2725. [Google Scholar] [CrossRef]
- Diao, M.; Nguyen, T.A.; Taran, E.; Mahler, S.; Nguyen, A.V. Effect of energy source, salt concentration and loading force on colloidal interactions between Acidithiobacillus ferrooxidans cells and mineral surfaces. Colloids Surf. B Biointerfaces 2015, 132, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Diao, M.; Taran, E.; Mahler, S.; Nguyen, T.A.; Nguyen, A.V. Quantifying adhesion of acidophilic bioleaching bacteria to silica and pyrite by atomic force microscopy with a bacterial probe. Colloids Surf. B Biointerfaces 2014, 115, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Li, Q.; Jiao, W.; Jiang, H.; Sand, W.; Xia, J.; Liu, X.; Qin, W.; Qiu, G.; Hu, Y. Adhesion forces between cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans or Leptospirillum ferrooxidans and chalcopyrite. Colloids Surf. B Biointerfaces 2012, 94, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Noël, N.; Florian, B.; Sand, W. AFM & EFM study on attachment of acidophilic leaching organisms. Hydrometallurgy 2010, 104, 370–375. [Google Scholar]
- Lara, R.; Valdez-Pérez, D.; Rodríguez, A.; Navarro-Contreras, H.; Cruz, R.; García-Meza, J. Interfacial insights of pyrite colonized by Acidithiobacillus thiooxidans cells under acidic conditions. Hydrometallurgy 2010, 103, 35–44. [Google Scholar] [CrossRef]
- Silverman, M.P.; Lundgren, D.G. Studies on the chemoautotrophic iron bacterium Ferrobacillus Ferrooxidans: I. An improved medium and a harvesting procedure for securing high cell yields. J. Bacteriol. 1959, 77, 642–647. [Google Scholar] [PubMed]
- Postgate, J. Media for sulphur bacteria. Lab. Pract. 1966, 15, 1239–1244. [Google Scholar] [PubMed]
- Razatos, A.; Ong, Y.L.; Sharma, M.M.; Georgiou, G. Molecular determinants of bacterial adhesion monitored by atomic force microscopy. Proc. Natl Acad. Sci. USA 1998, 95, 11059–11064. [Google Scholar] [CrossRef] [PubMed]
- Weder, G.; Blondiaux, N.; Giazzon, M.; Matthey, N.; Klein, M.; Pugin, R.; Heinzelmann, H.; Liley, M. Use of force spectroscopy to investigate the adhesion of living adherent cells. Langmuir 2010, 26, 8180–8186. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, Q.; Zhou, S.; Li, Q.; Gan, M.; Jiang, H.; Qin, W.; Liu, X.; Hu, Y.; Qiu, G. Insights into the relation between adhesion force and chalcopyrite-bioleaching by Acidithiobacillus ferrooxidans. Colloids Surf. B: Biointerfaces 2015, 126, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.L.; Ou, Y.; Tan, J.X.; Wu, F.D.; Sun, J.; Miao, L.; Zhong, D.L. Effect of EPS on adhesion of Acidithiobacillus ferrooxidans on chalcopyrite and pyrite mineral surfaces. Trans. Nonferrous Met. Soc. China 2011, 21, 407–412. [Google Scholar] [CrossRef]
- Chandraprabha, M.N.; Somasundaran, P.; Natarajan, K.A. Modeling and analysis of nanoscale interaction forces between Acidithiobacillus ferrooxidans and AFM tip. Colloids Surf. B: Biointerfaces 2010, 75, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Meyer, E.E.; Rosenberg, K.J.; Israelachvili, J. Recent progress in understanding hydrophobic interactions. Proc. Natl Acad. Sci. 2006, 103, 15739–15746. [Google Scholar] [CrossRef] [PubMed]
- Hammer, M.U.; Anderson, T.H.; Chaimovich, A.; Shell, M.S.; Israelachvili, J. The search for the hydrophobic force law. Faraday Discuss. 2010, 146, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sand, W.; Zhang, R. Enhancement of biofilm formation on pyrite by Sulfobacillus thermosulfidooxidans. Minerals 2016, 6, 71. [Google Scholar] [CrossRef]
- Zhang, R.; Bellenberg, S.; Castro, L.; Neu, T.R.; Sand, W.; Vera, M. Colonization and biofilm formation of the extremely acidophilic archaeon Ferroplasma acidiphilum. Hydrometallurgy 2014, 150, 245–252. [Google Scholar] [CrossRef]
- Gehrke, T.; Telegdi, J.; Thierry, D.; Sand, W. Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching. Appl. Environ. Microbiol. 1998, 64, 2743–2747. [Google Scholar] [PubMed]
- Flemming, H.C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Strathmann, M.; Wingender, J.; Flemming, H.C. Application of fluorescently labelled lectins for the visualization and biochemical characterization of polysaccharides in biofilms of Pseudomonas aeruginosa. J. Microbiol. Methods 2002, 50, 237–248. [Google Scholar] [CrossRef]
- Zhang, R.; Neu, T.; Bellenberg, S.; Kuhlicke, U.; Sand, W.; Vera, M. Use of lectins to in situ visualize glycoconjugates of extracellular polymeric substances in acidophilic archaeal biofilms. Microb. Biotechnol. 2015, 8, 448–461. [Google Scholar] [CrossRef] [PubMed]
- Omoike, A.; Chorover, J.; Kwon, K.D.; Kubicki, J.D. Adhesion of bacterial exopolymers to α-feooh: Inner-sphere complexation of phosphodiester groups. Langmuir 2004, 20, 11108–11114. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sand, W. Mechanical and chemical studies on eps from Sulfobacillus thermosulfidooxidans: From planktonic to biofilm cells. Colloids Surf. B: Biointerfaces 2017, 153, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Becker, T.; Sand, W. Quantification of cell-substratum interactions by atomic force microscopy. Colloids Surf. B Biointerfaces 2017, 159, 639–643. [Google Scholar] [CrossRef] [PubMed]





© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Q.; Yang, B.; Zhu, J.; Jiang, H.; Li, J.; Zhang, R.; Sand, W. Comparative Analysis of Attachment to Chalcopyrite of Three Mesophilic Iron and/or Sulfur-Oxidizing Acidophiles. Minerals 2018, 8, 406. https://doi.org/10.3390/min8090406
Li Q, Yang B, Zhu J, Jiang H, Li J, Zhang R, Sand W. Comparative Analysis of Attachment to Chalcopyrite of Three Mesophilic Iron and/or Sulfur-Oxidizing Acidophiles. Minerals. 2018; 8(9):406. https://doi.org/10.3390/min8090406
Chicago/Turabian StyleLi, Qian, Baojun Yang, Jianyu Zhu, Hao Jiang, Jiaokun Li, Ruiyong Zhang, and Wolfgang Sand. 2018. "Comparative Analysis of Attachment to Chalcopyrite of Three Mesophilic Iron and/or Sulfur-Oxidizing Acidophiles" Minerals 8, no. 9: 406. https://doi.org/10.3390/min8090406
APA StyleLi, Q., Yang, B., Zhu, J., Jiang, H., Li, J., Zhang, R., & Sand, W. (2018). Comparative Analysis of Attachment to Chalcopyrite of Three Mesophilic Iron and/or Sulfur-Oxidizing Acidophiles. Minerals, 8(9), 406. https://doi.org/10.3390/min8090406




