Two-Stage SART Process: A Feasible Alternative for Gold Cyanidation Plants with High Zinc and Copper Contents
Abstract
:1. Introduction
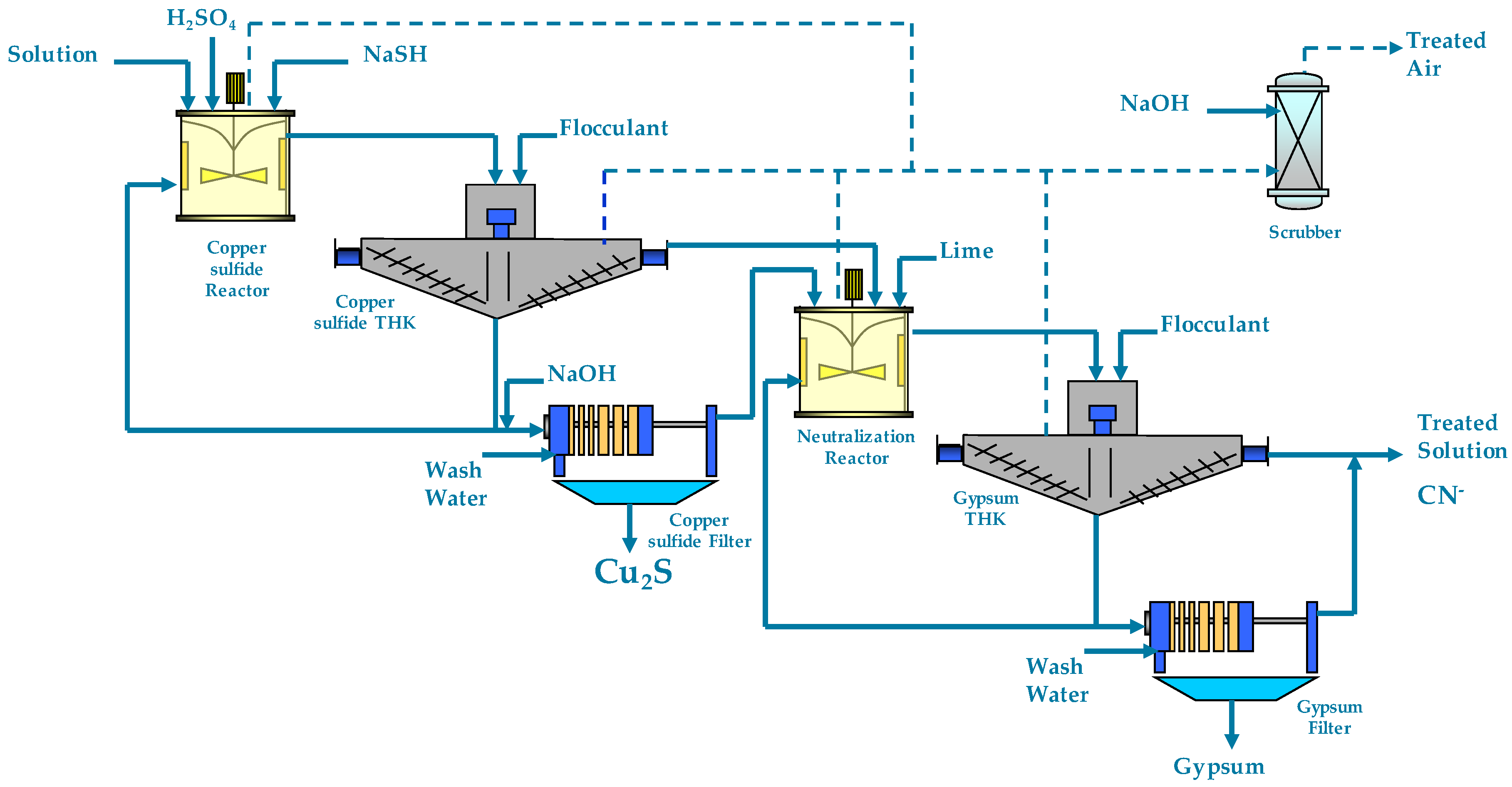
1.1. SART Process Description
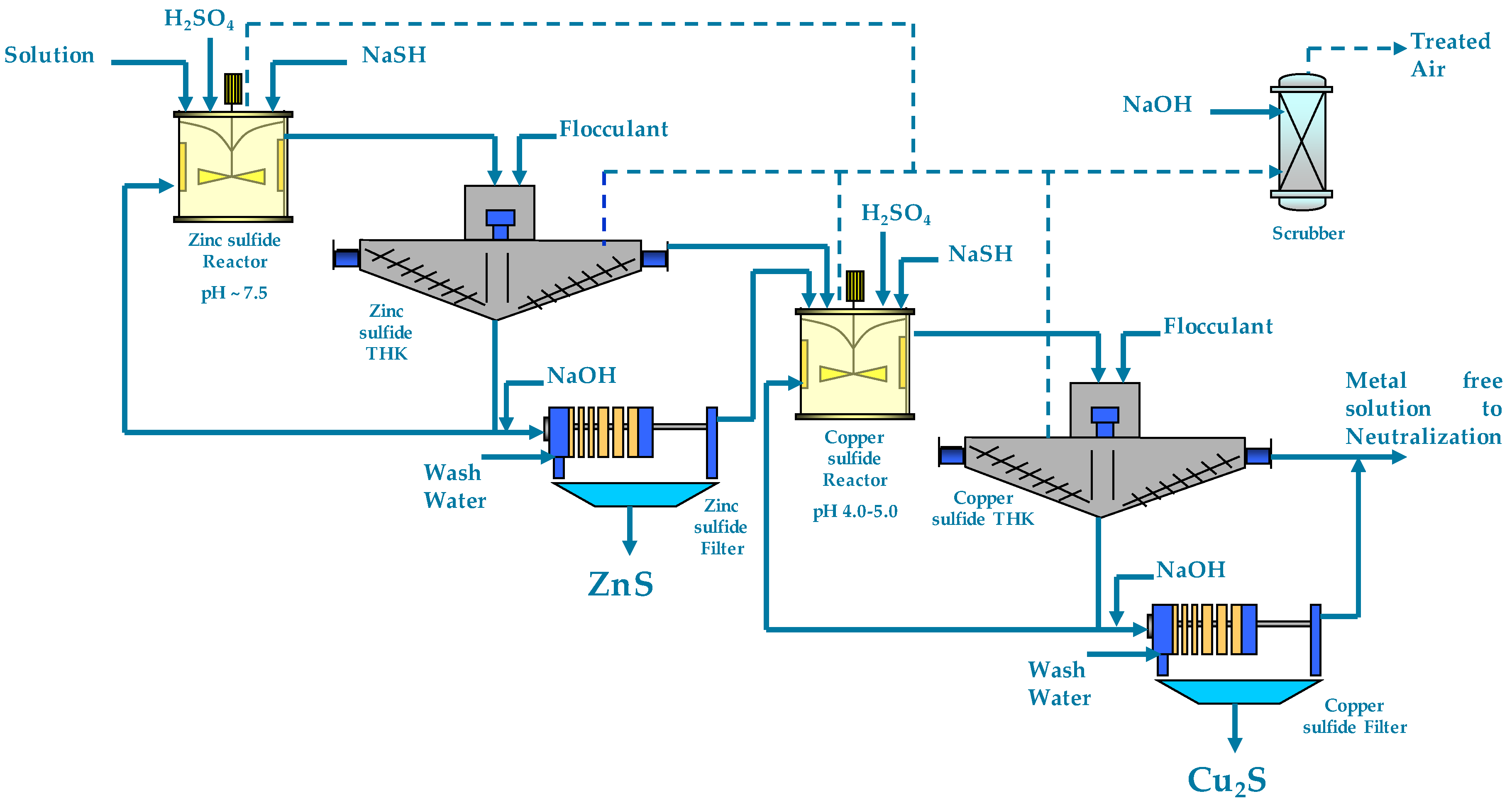
1.2. Two-Stage SART Process
2. Methods
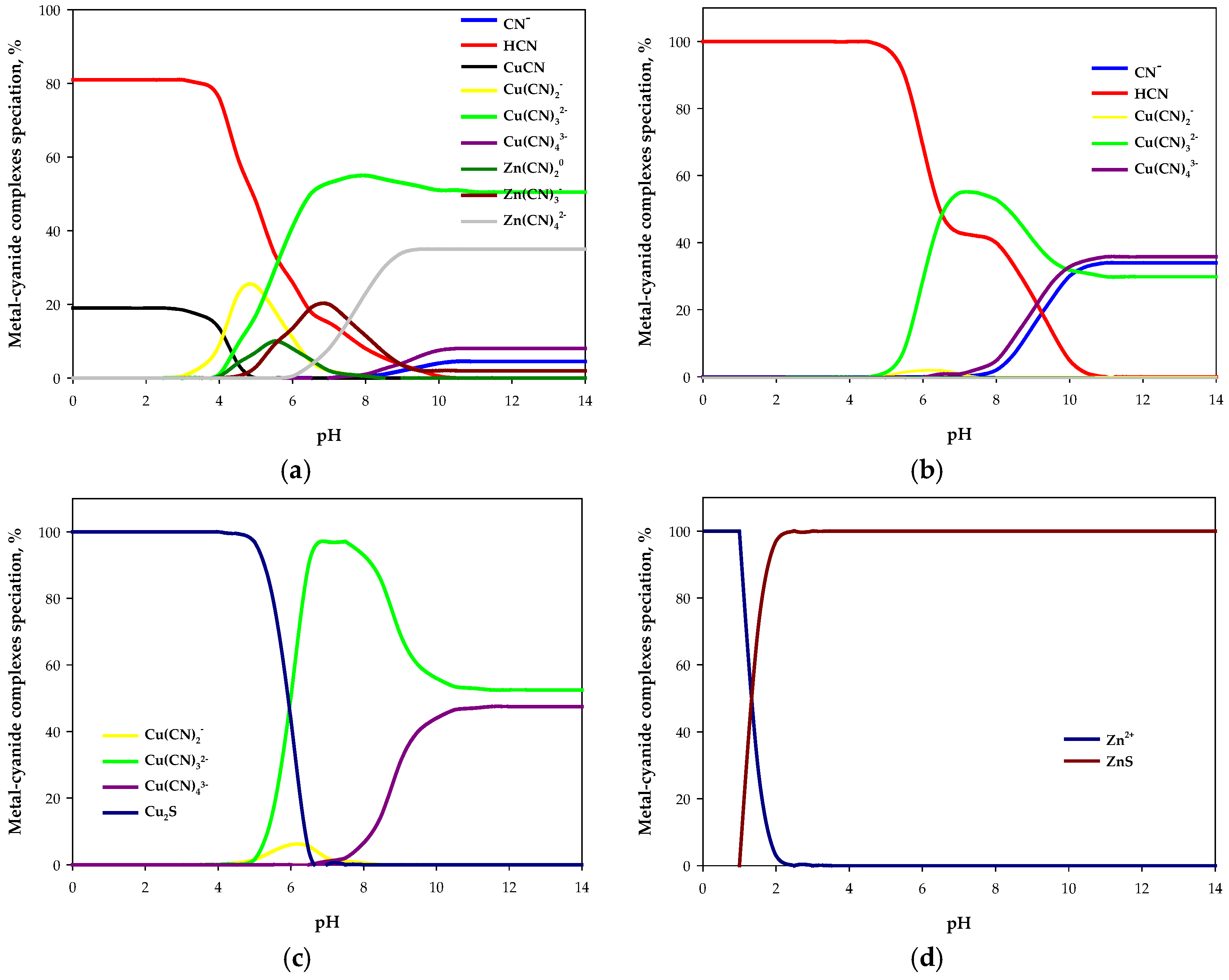
2.1. Estimation of Operational Parameters Using a Thermodynamic Model
- The model did not include other metals—such as iron, mercury, silver, or nickel—because these elements are typically present in low contents of cyanide solutions (<10 mg/L).
- The main reactions involved in sulfidization stage were Table 1.
2.2. Economic Comparison between Conventional and Two-Stage SART Process Options
3. Results and Discussion
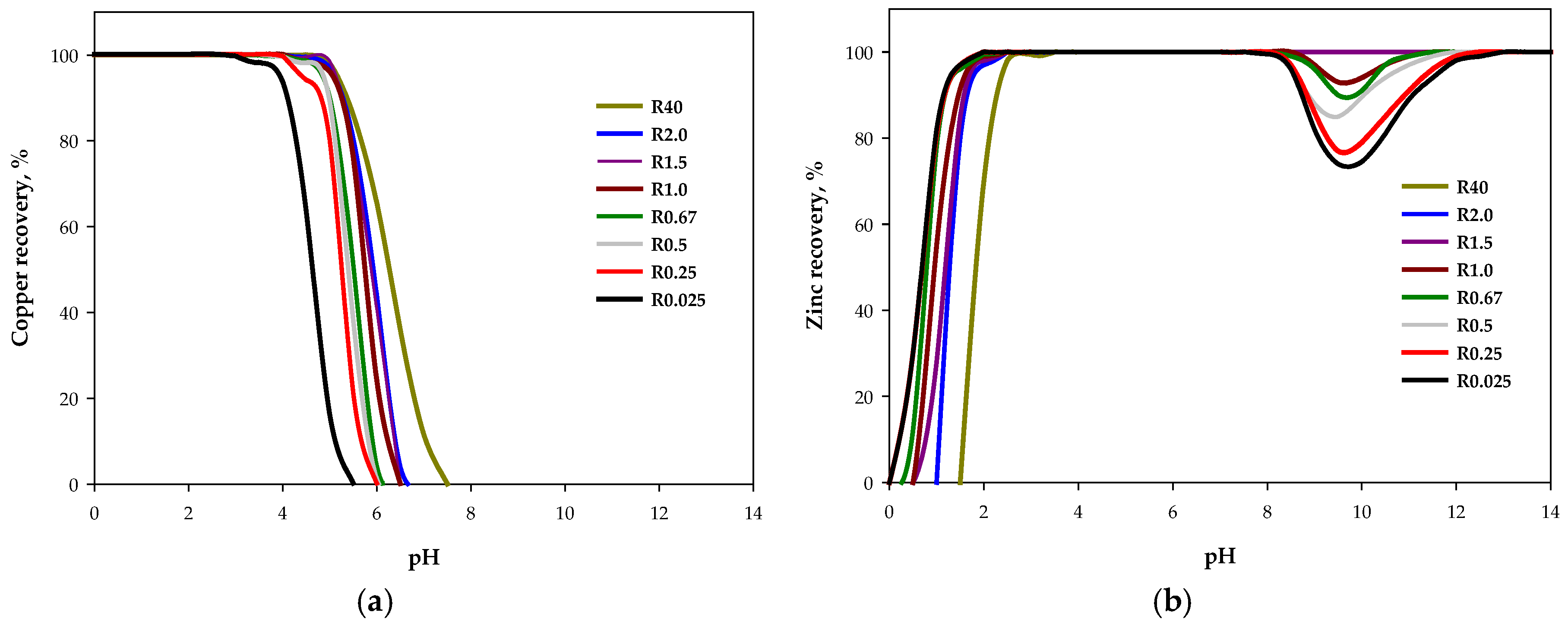
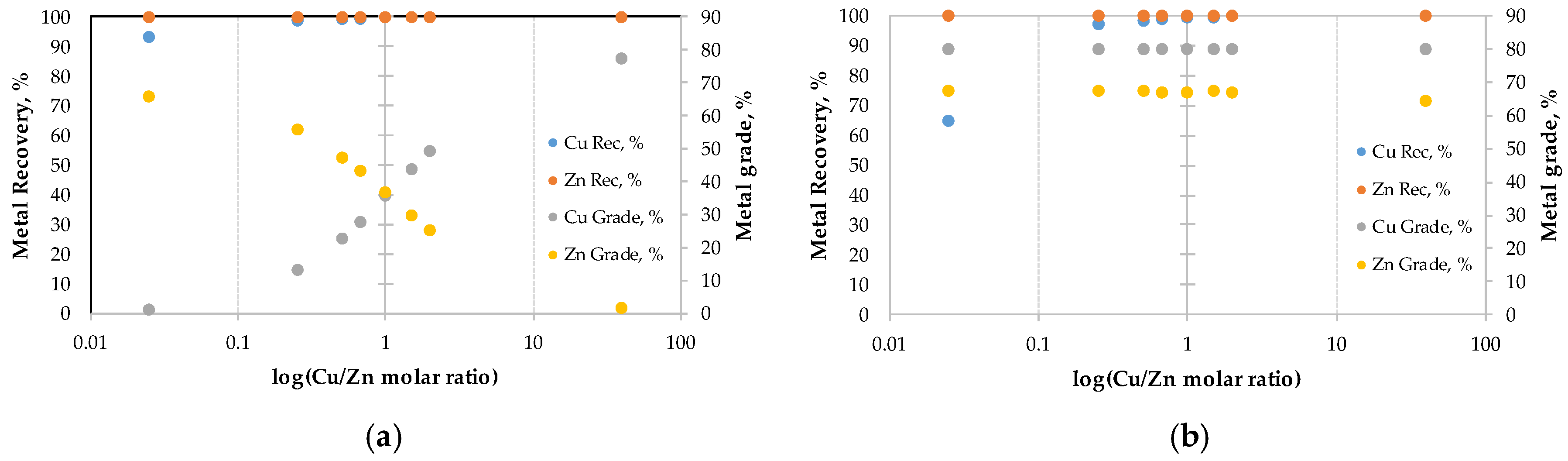
3.1. Operational Conditions and Metal Recoveries
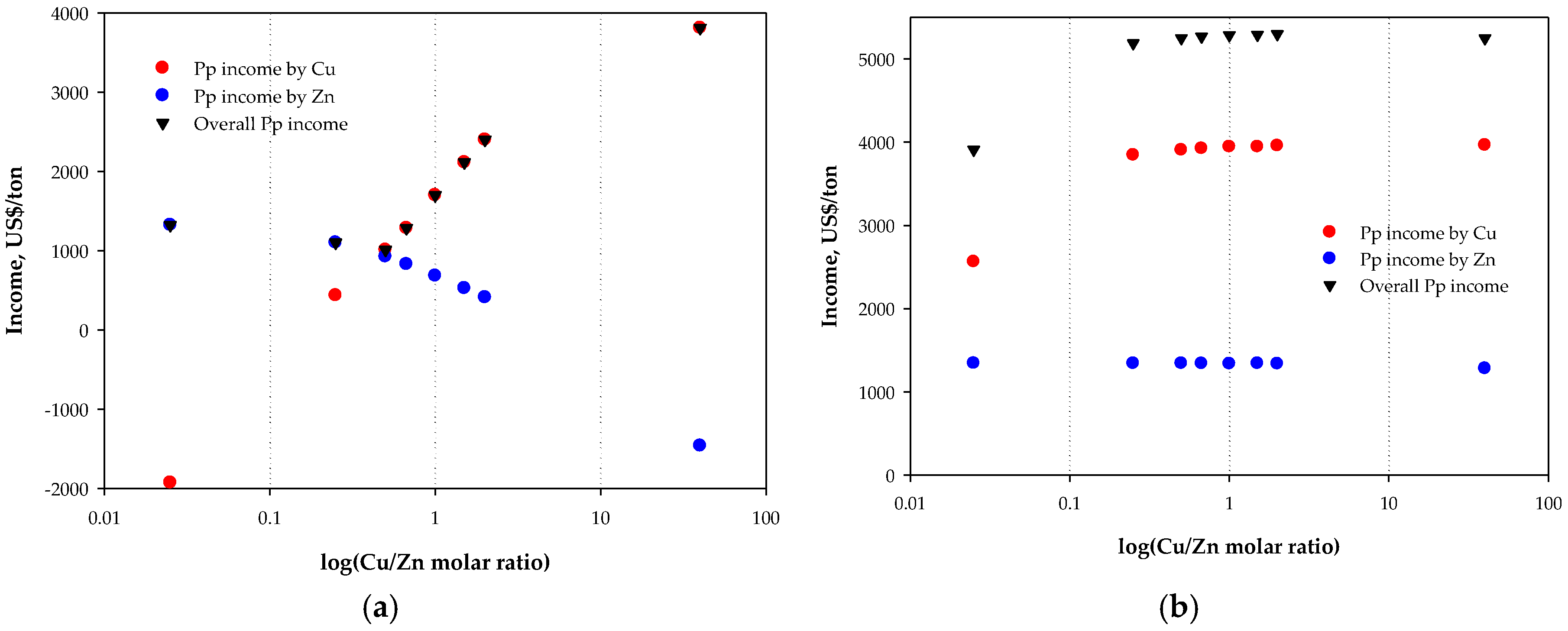
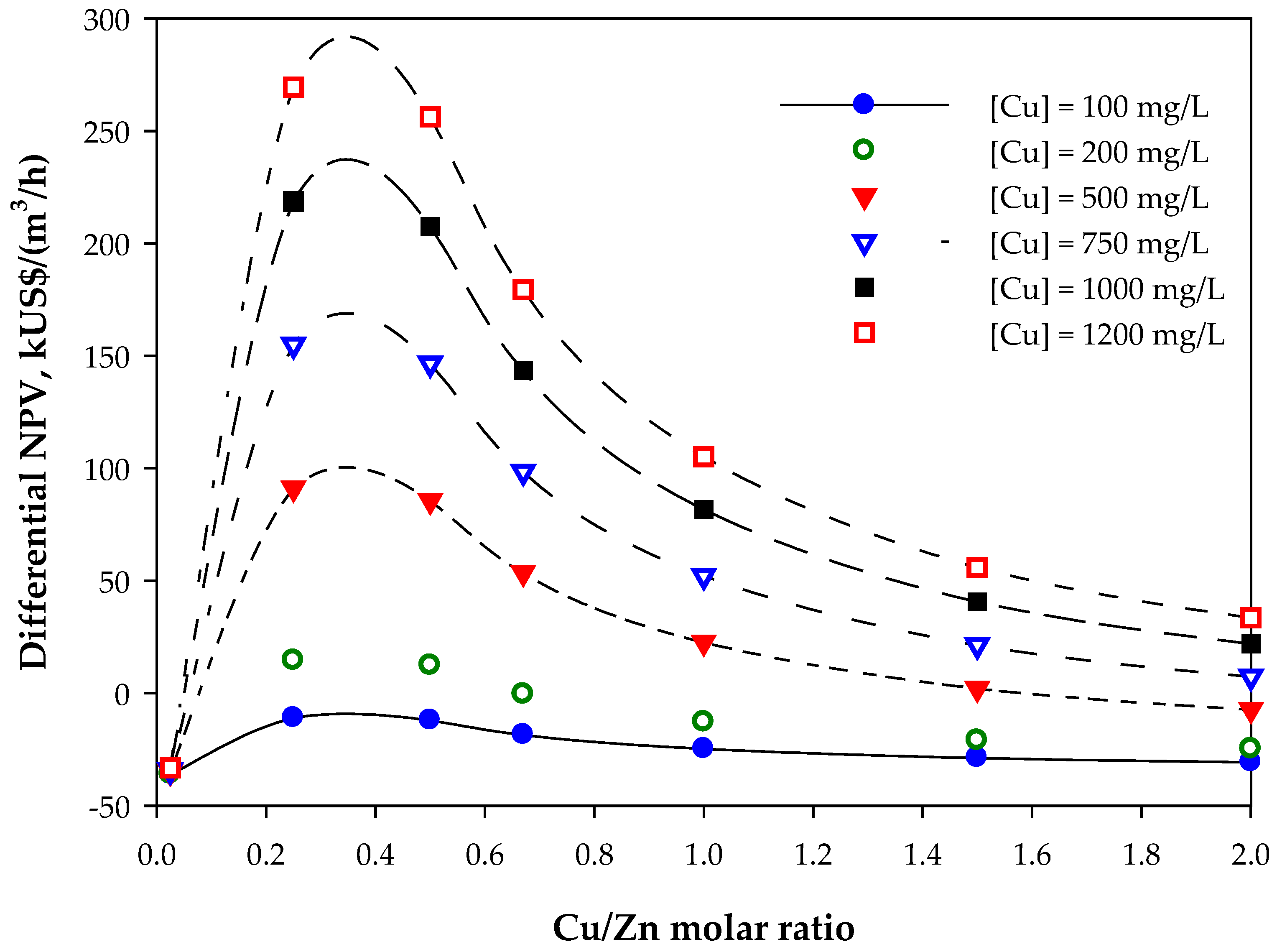
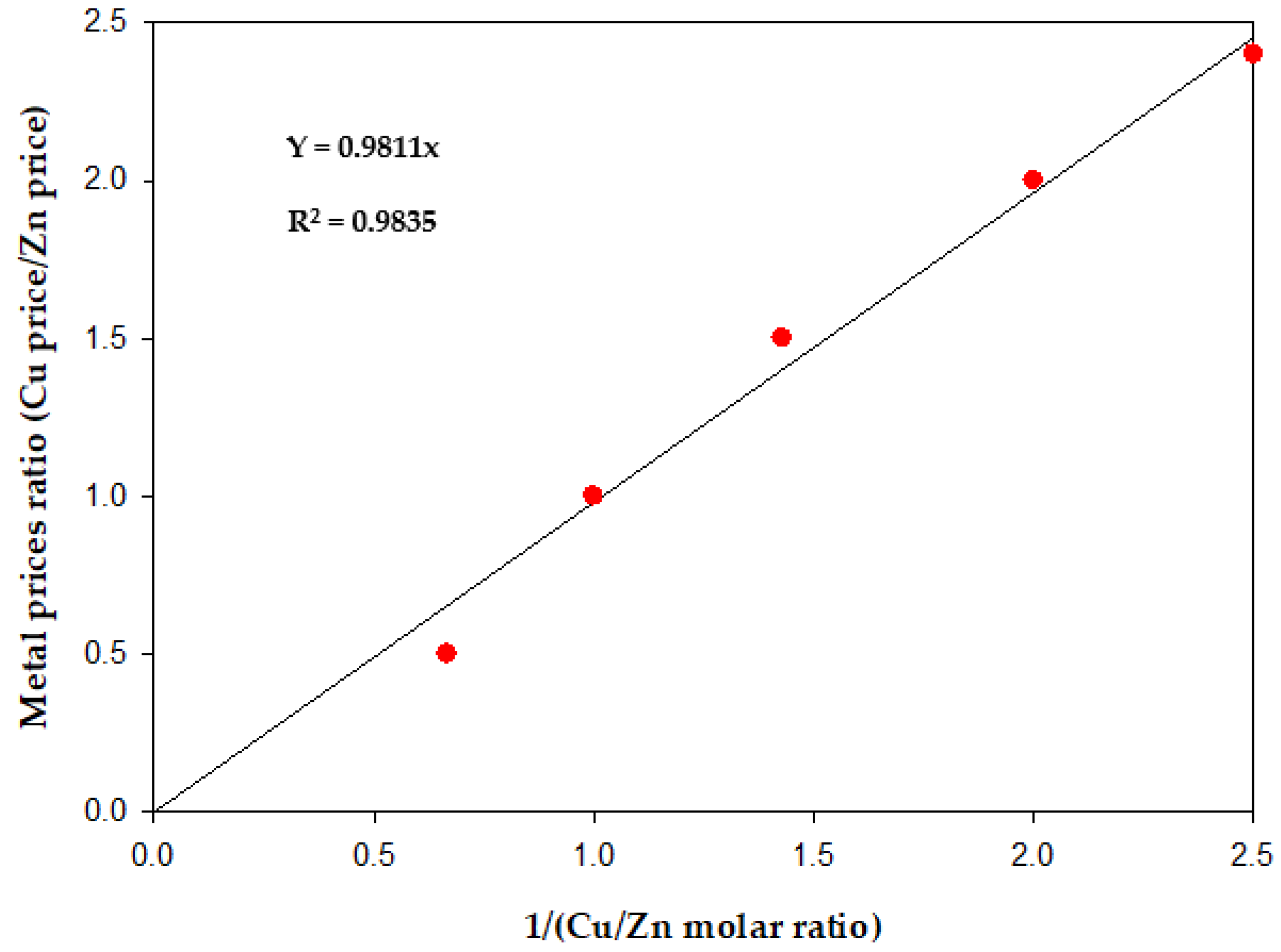
3.2. Economic Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Estay, H. Designing the SART process—A review. Hydrometallurgy 2018, 176, 147–165. [Google Scholar] [CrossRef]
- MacPhail, P.K.; Fleming, C.A.; Sarbutt, K. Cyanide Recovery by the SART Process for the Lobo-Marte Project, Chile. In Proceedings of the Randol Gold and Silver Forum ‘98, Denver, CO, USA, 26–29 April 1998. [Google Scholar]
- Barter, J.; Lane, G.; Mitchell, D.; Kelson, R.; Dunne, R.; Trang, C.; Dreisinger, D. Cyanide management by SART. In Cyanide: Social, Industrial and Economic Aspects; Young, C.A., Twidwell, L.G., Anderson, C.G., Eds.; TMS (The Minerals, Metals & Materials Society): Pittsburgh, PA, USA, 2001; pp. 549–562. [Google Scholar]
- Simons, A.; Breuer, P. The impact of residence time on copper recovery in Telfer Gold Mine’s cyanide recycling process. In Proceedings of the World Gold 2013, Brisbane, Australasian Institute of Mining and Metallurgy, Melbourne, QLD, Australia, 26–29 September 2013; pp. 189–196. [Google Scholar]
- Lawrence, R.; Lopez, O. Improving the economics of gold–copper ore projects using SART technology. In Proceedings of the World Gold 2011, CIMM, Calgary, AB, Canada, 2–5 October 2011. [Google Scholar]
- Vargas, L.; Guzman, G.; Roux, E.; Sevilla, L. Yanacocha Gold Mill and SART/AVR process 2013–2017. In Proceedings of the 1st Arcadis Gold Work, Santiago, Chile, 20 March 2013. [Google Scholar]
- Hedjazi, F.; Monhemius, A.J. Copper–gold ore processing with ion exchange and SART technology. Miner. Eng. 2014, 64, 120–125. [Google Scholar] [CrossRef]
- Kratochvil, D.; Chan, N.; Hall, A. Integrating SART into metallurgical flowsheets for cyanide recovery. In Proceedings of the ALTA 2013 Gold Conference, Perth, Australia, 25 May–1 June 2013. [Google Scholar]
- Ford, K.; Fleming, C.; Henderson, R. Application of the SART process to heap leaching of gold–copper ores at Maricunga, Chile. In Proceedings of the 40th Annual Meeting of the Canadian Mineral Processors, Ottawa, ON, Canada, 23–24 January 2008. [Google Scholar]
- Botz, M.; Kaczmarek, A.; Orser, S. Managing copper in leach solution at the copler gold mine: Laboratory testing and process design. Miner. Met. Process. 2011, 28, 133–138. [Google Scholar]
- Simons, A. A Fundamental Study of Copper and Cyanide Recovery from Gold Tailing by Sulfidisation. Ph.D. Thesis, Curtin University, Perth, WA, Australia, 2015. [Google Scholar]
- Walton, R. Zinc cementation. In Gold Ore Process, 2nd ed.; Adams, M.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 553–560. [Google Scholar]
- Botz, M.M.; Scola, J.C.; Fueyo, R.; de Moura, W. Cyanide recovery practice at cerro vanguardia. In Proceedings of the SME Annual Meeting, Denver, CO, USA, 26–28 February 2004. [Google Scholar]
- Ford, K.J.; Peacey, J.G.; Sevilla, L.; Villalobos, E. Processing of refractory sulphides at mantos de Oro, Chile. In Proceedings of the 41st Annual Meeting of the Canadian Mineral Processors, Ottawa, ON, Canada, 20–22 February 2009. [Google Scholar]
- Marsden, J.O.; House, C.I. The Chemistry of Gold Extraction, 2nd ed.; Society for Mining, Metallurgy, and Exploration, Inc. (SME): Littleton, CO, USA, 2006. [Google Scholar]
- Littlejohn, P.; Kratochvil, D.; Hall, A. Sulfidisation-acidification-recycling-thickening (SART) for complex gold ores. In Proceedings of the World Gold 2013, Brisbane, Australasian Institute of Mining and Metallurgy, Melbourne, QLD, Australia, 26–29 September 2013; pp. 149–155. [Google Scholar]
- Lopez, O.; Petrovic, N.; Krashenina, S. Copper and cyanide recovery from barren leach solution at the gold processing plant. In Proceedings of the XXVII International Mineral Processing Congress. (IMPC 2014), Santiago, Chile, 20–24 October 2014. [Google Scholar]
- Baker, B.; Rodriguez, F.; Littlejohn, P. SART implementation at gold mines in Latin America. In Proceedings of the World Gold 2017, Vancouver, BC, Canada, 27–30 August 2017. [Google Scholar]
- Fleming, C.A.; Trang, C.V. Review of options for cyanide recovery at gold and silver mines. In Proceedings of the Randol Gold and Silver Forum ‘98, Denver, CO, USA, 26–29 April 1998; pp. 313–318. [Google Scholar]
- Estay, H. Estimating the hydrogen cyanide generation: Thermodynamics and mass transfer. In Cyanide: Occurrence, Characteristics and Applications, 1st ed.; Soto-Blanco, B., Ed.; Nova Publishers: New York, NY, USA, 2013; pp. 47–86. [Google Scholar]
- Lu, J.; Dreisinger, D.; Cooper, W. Thermodynamics of the aqueous copper-cyanide system. Hydrometallurgy 2002, 66, 23–36. [Google Scholar] [CrossRef]
- Izzat, R.; Dee Johnston, H.; Watt, G.; Christensen, J. Thermodynamics of metalcyanide coordination. VI. Copper(I)-and silver(I)-cyanide systems. Inorg. Chem. 1967, 6, 132–135. [Google Scholar] [CrossRef]
- Izzatt, R.M.; Christensen, J.; Hansen, J.; Watt, G. Thermodynamics of metal cyanide coordination. IV. Values for ths Zinc(II)-Cyanide System at 25°. Inorg. Chem. 1965, 2, 718–721. [Google Scholar] [CrossRef]
- Izzatt, R.M.; Christensen, J.; Pack, R.; Bench, R. Thermodynamics of metal-cyanide coordination. I. Values as a funtion of temperature for hydrocyanic acid dissociation in aqueous solution. Inorg. Chem. 1961, 1, 828–831. [Google Scholar] [CrossRef]
- Banfalvi, G. Removal of insoluble heavy metal sulfides from water. Chemosphere 2006, 63, 1231–1234. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fang, Z.; Muhammed, M. On the solution chemistry of cyanidation of gold and silver bearing sulphide ores. A critical evaluation of thermodynamic calculations. Hydrometallurgy 1997, 46, 251–269. [Google Scholar] [CrossRef]
- Puigdomenech, I. Hydra-Medusa Software (Hydrochemical Equilibrium Constant, Database and Make Equilibrium Diagrams Using Sophisticated Algorithms), version 1; Inorganic Chemistry, Royal Institute of Technology: Stockholm, Sweden, 2004. [Google Scholar]
- Botz, M.; Guzman, G.; Sevilla, L. Campaign testing the yanacocha sart plant with high-copper feed solution. In Proceedings of the Society for Mining, Metallurgy, and Exploration, Inc. (SME), Denver, CO, USA, 15–18 February 2015. [Google Scholar]
| Reaction | logKi | Reference |
|---|---|---|
| 20 | [21] | |
| 3.94 | [22] | |
| 5.3 | [22] | |
| 1.5 | [22] | |
| 11.07 | [23] | |
| 4.98 | [23] | |
| 3.57 | [23] | |
| 9.21 | [24] | |
| 47.3 | [25] | |
| 23.08 | [25] | |
| 13.9 | [26] | |
| 7.02 | [26] |
| Parameter | Value | Unit |
|---|---|---|
| Copper price | 3.0 | US$/lb |
| Zinc price | 1.25 | US$/lb |
| Precipitate discount rate | 25 | % |
| Transport cost | 30 | US$/dry ton |
| Parameter | Value | Unit |
|---|---|---|
| Capital cost of additional equipment | 36.0 | kUS$/(m3/h) |
| Additional energy consumption | 0.047 | kWh/m3 |
| Energy price | 150 | US$/MWh |
| Additional flocculant consumption | 2.0 | g/m3 |
| Flocculant price | 3000 | US$/ton |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estay, H.; Gim-Krumm, M.; Quilaqueo, M. Two-Stage SART Process: A Feasible Alternative for Gold Cyanidation Plants with High Zinc and Copper Contents. Minerals 2018, 8, 392. https://doi.org/10.3390/min8090392
Estay H, Gim-Krumm M, Quilaqueo M. Two-Stage SART Process: A Feasible Alternative for Gold Cyanidation Plants with High Zinc and Copper Contents. Minerals. 2018; 8(9):392. https://doi.org/10.3390/min8090392
Chicago/Turabian StyleEstay, Humberto, Minghai Gim-Krumm, and Michelle Quilaqueo. 2018. "Two-Stage SART Process: A Feasible Alternative for Gold Cyanidation Plants with High Zinc and Copper Contents" Minerals 8, no. 9: 392. https://doi.org/10.3390/min8090392
APA StyleEstay, H., Gim-Krumm, M., & Quilaqueo, M. (2018). Two-Stage SART Process: A Feasible Alternative for Gold Cyanidation Plants with High Zinc and Copper Contents. Minerals, 8(9), 392. https://doi.org/10.3390/min8090392





