Hydrotalcite and Hydrocalumite in Mortar Binders from the Medieval Castle of Portilla (Álava, North Spain): Accurate Mineralogical Control to Achieve More Reliable Chronological Ages
Abstract
1. Introduction
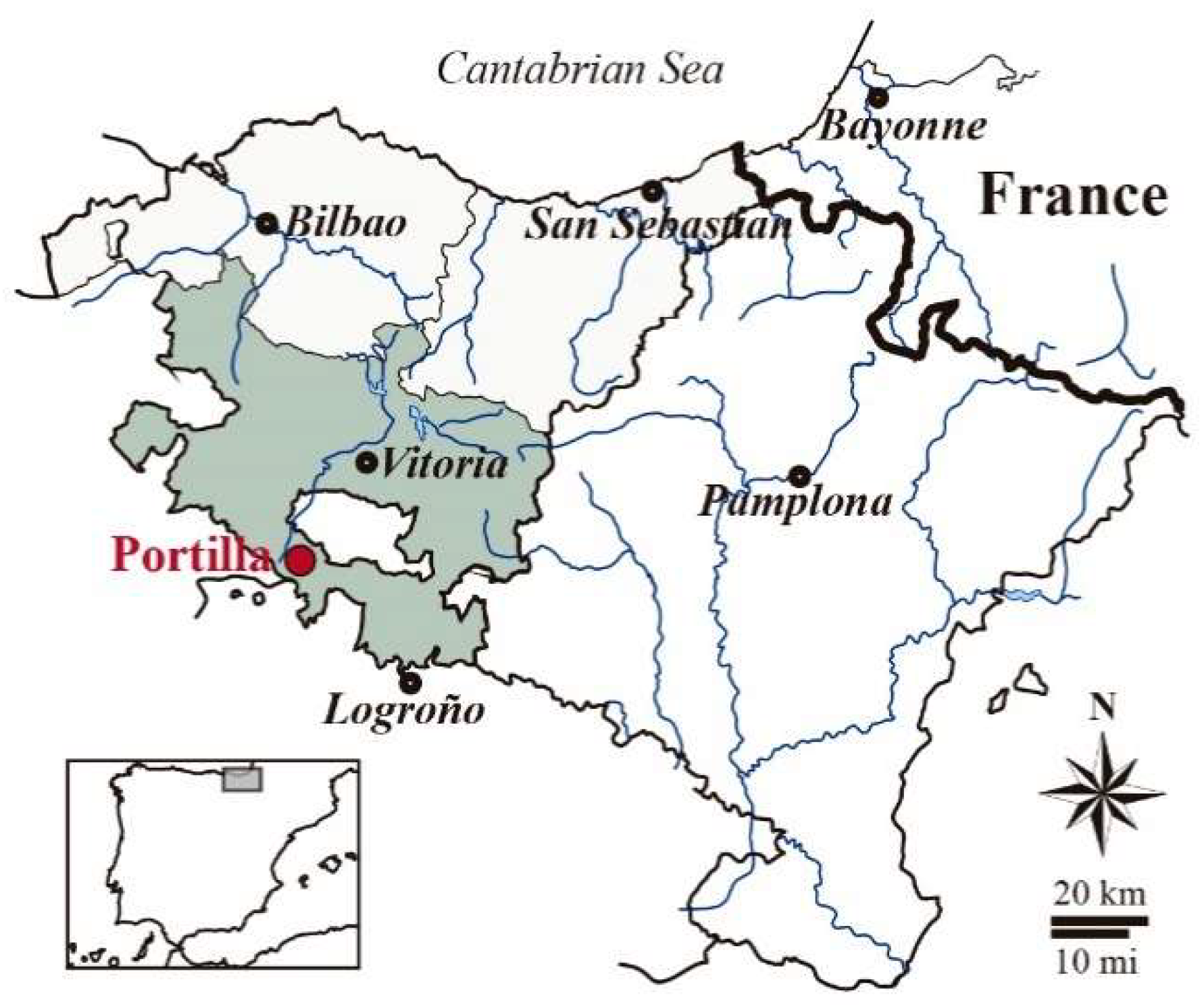
2. Archaeological Background
3. Materials and Methods
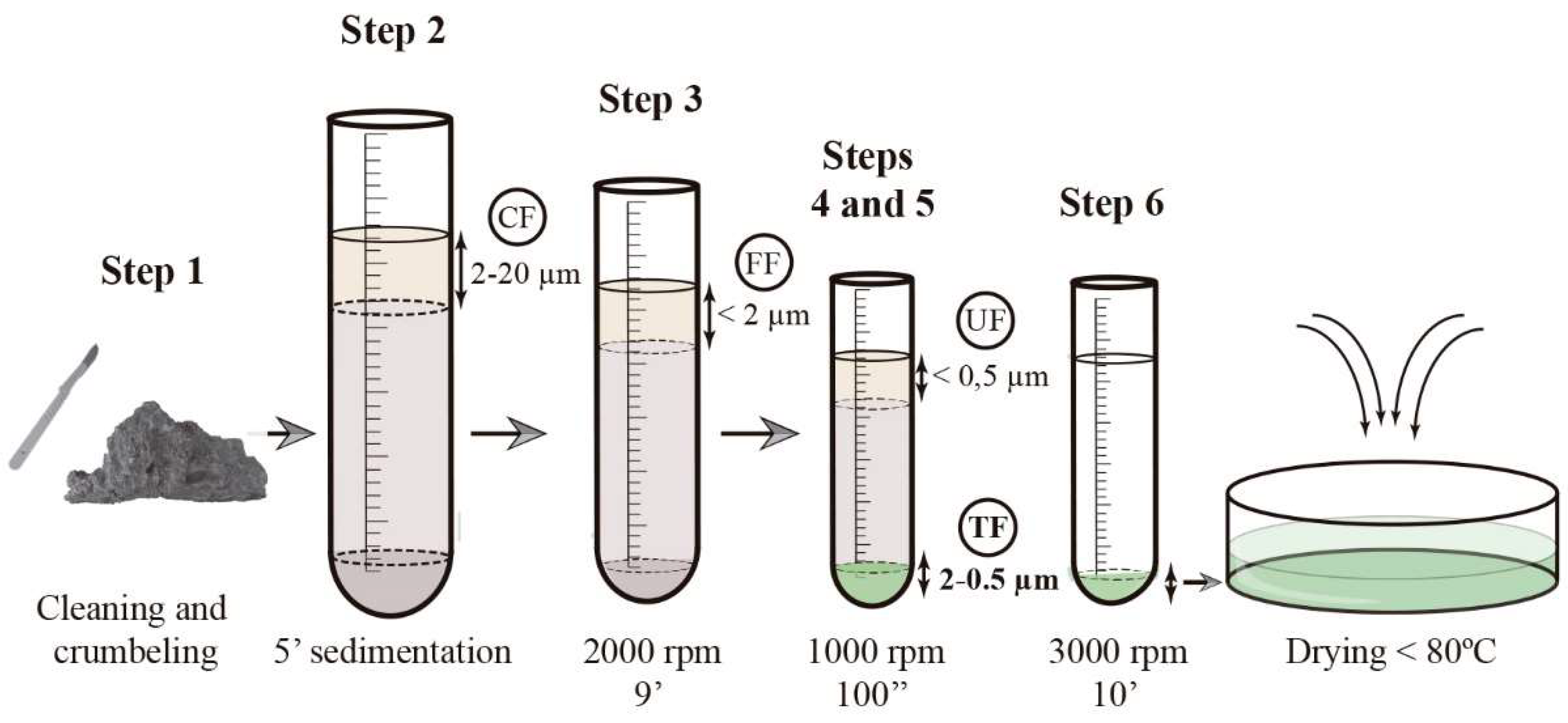
Extraction Procedure
- The few upper millimeters of mortar surface were removed using a scraper to eliminate the altered surfaces that may interfere in the analytical results. Then mortars were crumbled manually, which is described by Casadio et al. [65]. The fragments of charcoal produced during lime production were picked out and removed.
- The sample was placed in an ultrasonic bath with 150 mL of ultrapure water for 10 min. To promote further crumbling, the test tube was shaken vigorously and then left to rest for 5 min. Then, the topmost 50 mL of suspended volume corresponding to particle fraction size 2 μm to 20 μm was extracted. This fraction was referred to as coarse fraction (CF).
- Coarse fraction enriched in the binder was re-suspended in 65 mL of ultrapure water and placed in the ultrasonic bath to promote particle disaggregation. The suspension was centrifuged for 100 s at 1000 rpm. The uppermost 15 mL were collected corresponding to a grain size fraction of <2 μm and were referred to as a fine fraction (FF).
- From the fine fraction, the particles of grain size <0.5 µm were removed. To this end, the sample was re-suspended again in 65 mL of ultrapure water and placed in an ultrasonic bath. The suspension was centrifuged for 9 min at 2000 rpm and the uppermost 15 mL were collected and referred to as ultrafine fraction (UF).
- The suitable fraction enriched in neoformation calcite was obtained as a result of removing the ultrafine fraction (UF) from the fine fraction (FF). This fraction was named a target fraction (TF) corresponding to a grain size between 0.5 µm to 2 µm.
- The collected fractions were concentrated via centrifugation at 3000 rpm for 10 min using a Kubota 3000 centrifuge (Kubota Corp., Tokyo, Japan).
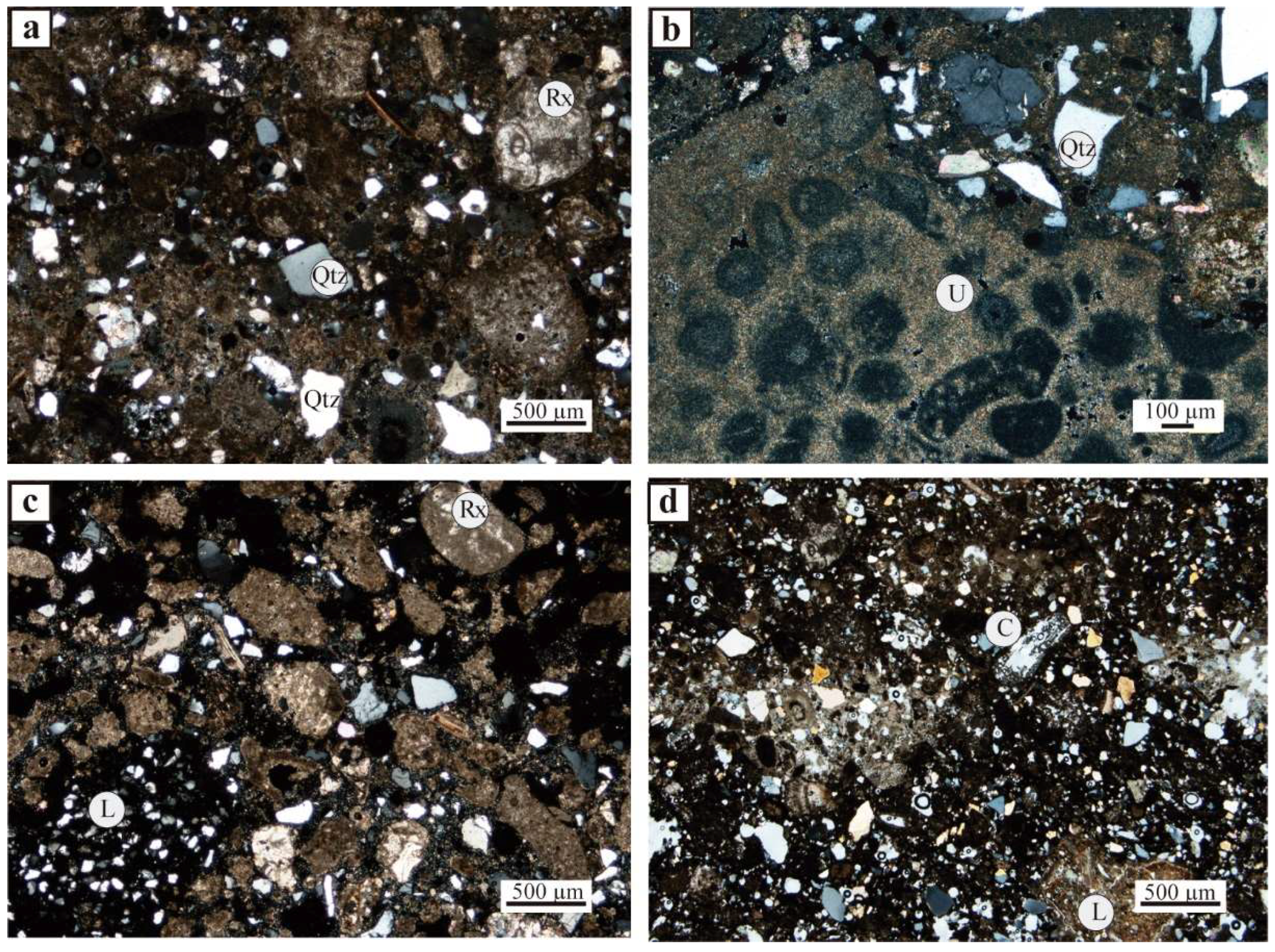
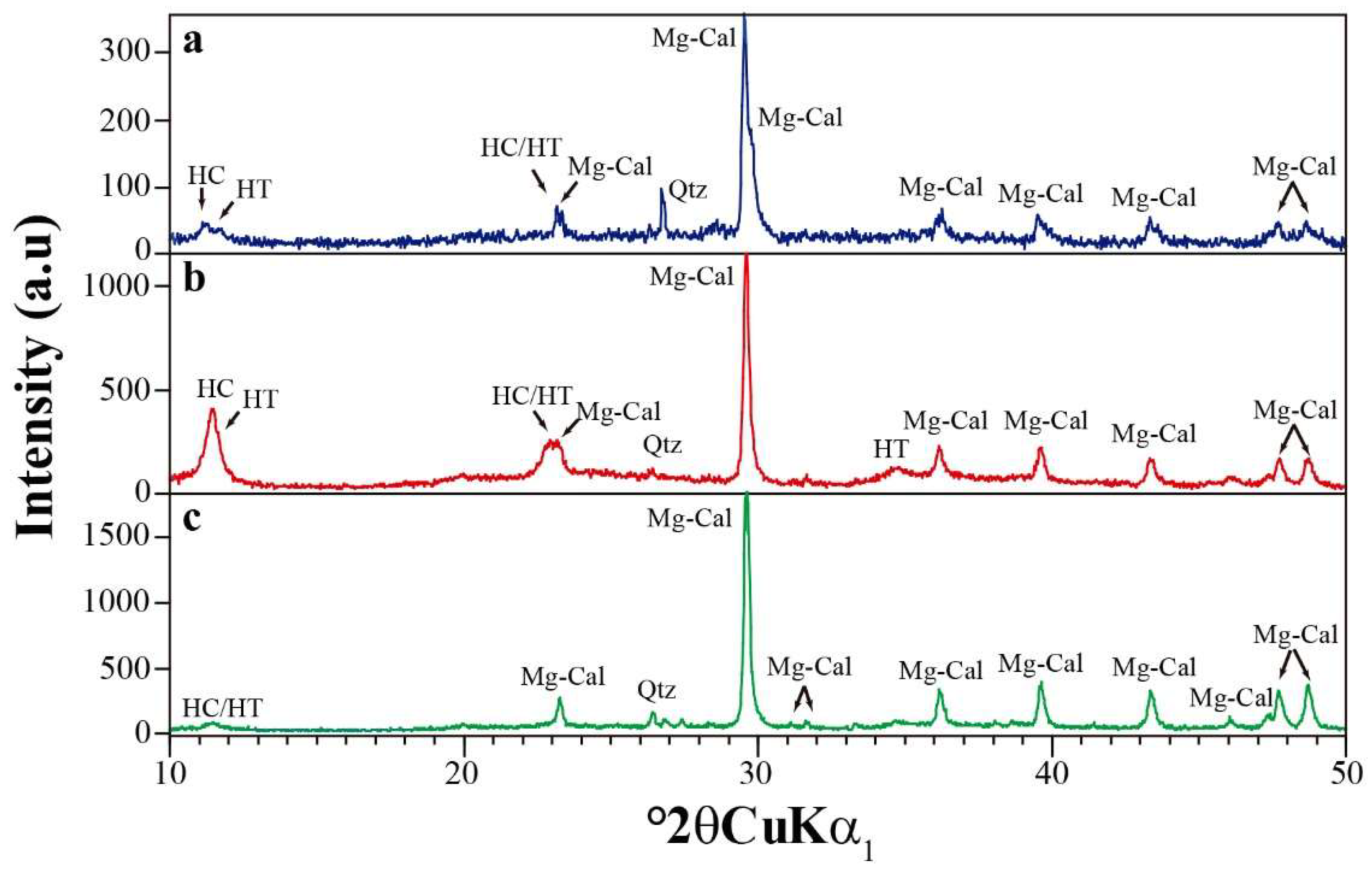
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schiffer, M. Radiocarbon Dating and the “Old Wood” Problem: The Case of the Hohokam Chronology; Elsevier: New York, NY, USA, 1986; Volume 13, pp. 13–30. [Google Scholar]
- Mc Fadgen, B.G. Dating New Zealand archaeology by radiocarbon. N. Z. J. Sci. 1982, 25, 379–392. [Google Scholar]
- Gavin, D.G. Estimation of inbuilt age in radiocarbon ages of soil charcoal for fire history studies. Radiocarbon 2001, 43, 27–44. [Google Scholar] [CrossRef]
- Bowman, S. Radiocarbon Dating; British Museum Press: London, UK, 1990. [Google Scholar]
- Taylor, R.; Bar-Yosef, O. Radiocarbon Dating: An Archaeological Perspective; Left Coast Press: Walnut Creek, CA, USA, 2014. [Google Scholar]
- Boaretto, E.; Poduska, K.M. Materials science challenges in radiocarbon dating: The case of archaeological plasters. J. Mater. 2013, 65, 481–488. [Google Scholar] [CrossRef]
- Kingery, D.W.; Pamela, B.V.; Martha, P. The Beginnings of Pyrotechnology, Part II: Production and Use of Lime and Gypsum Plaster in the Pre-Pottery Neolithic Near East; Taylor and Francis: New York, NY, USA, 1988; Volume 15, pp. 219–243. [Google Scholar]
- Labeyrie, J.; Delibrias, G. Dating of old mortar by Carbon-14 method. Nature 1964, 201, 742–743. [Google Scholar] [CrossRef]
- Stuiver, M.; Smith, C.S. Radiocarbon Dating of Ancient Mortar and Plaster, Proceedings of the 6th International 14C Conference, Montreal, Canada, 8–15 September 1965; Chatters, R.M., Olson, C.A., Eds.; Clearinghouse for Federal Scientific and Technical Information: Springfield, IL, USA; pp. 338–343.
- Pachiaudi, C.; Marechal, J.; Van Strydonck, M.; Dupas, M.; Dauchotdehon, M. Isotopic fractionation of carbon during CO2 absorption by mortar. Radiocarbon 1986, 28, 691–697. [Google Scholar] [CrossRef]
- Ambers, J. Stable carbon isotope ratios and their relevance to the determination of accurate radiocarbon dates for lime mortars. J. Archaeol. Sci. 1987, 14, 569–576. [Google Scholar] [CrossRef]
- Baxter, M.S.; Walton, A. Radiocarbon dating of mortars. Nature 1970, 225, 937–938. [Google Scholar] [CrossRef] [PubMed]
- Folk, R.L.; Valastro, S.J. Successful technique for dating of lime mortar by carbon-14. J. Field Archaeol. 1976, 3, 203–208. [Google Scholar] [CrossRef]
- Van Strydonck, M.; Dupas, M.; Dauchot-Dehon, M. Radiocarbon dating of old mortars, 14C and Archaeology, Proceedings; Mook, W.G., Waterbolk, H.T., Eds.; PACT: Phnom Penh, Cambodia, 1983; pp. 337–343. [Google Scholar]
- Van Strydonck, M.; Dupas, M.; Dauchotdehon, M.; Pachiaudi, C.; Marechal, J. The influence of contaminating (fossil) carbonate and the variations of delta-C-13 in mortar dating. Radiocarbon 1986, 28, 702–710. [Google Scholar] [CrossRef]
- Van Strydonck, M.J.Y.; Van der Borg, K.; De Jong, A.F.M.; Keppens, E. Radiocarbon dating of lime fractions and organic material from buildings. Radiocarbon 1992, 34, 873–879. [Google Scholar] [CrossRef]
- Lindroos, A.; Heinemeier, J.; Ringbom, Å.; Braskén, M.; Sveinbjörnsdóttir, Á. Mortar dating using AMS 14C and sequential dissolution: Examples from medieval, non-hydraulic lime mortars from the Åland Islands, SW Finland. Radiocarbon 2007, 49, 47–67. [Google Scholar] [CrossRef]
- Sonninen, E.; Jungner, H. An improvement in preparation of mortar for radiocarbon dating. Radiocarbon 2001, 43, 271–273. [Google Scholar] [CrossRef]
- Hale, J.; Heinemeier, J.; Lancaster, L.; Lindroos, A.; Ringbom, Å. Dating ancient mortar. Am. Sci. 2003, 91, 130–137. [Google Scholar] [CrossRef]
- Nawrocka, D.; Michniewicz, J.; Pawlyta, J.; Pazdur, A. Application of radiocarbon method for dating of lime mortars. Geochronometria 2005, 24, 109–115. [Google Scholar]
- Heinemeier, J.; Jungner, H.G.; Lindroos, A.; Ringbom, S.; von Konow, T.; Rud, N. AMS 14C dating of lime mortar. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 1997, 123, 487–495. [Google Scholar] [CrossRef]
- Tubbs, L.E.; Kinder, T.N. The use of AMS for the dating of lime mortars. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms. 1990, 52, 438. [Google Scholar] [CrossRef]
- Toffolo, M.; Maeir, A.M.; Chadwick, J.R.; Boaretto, E. Characterization of contexts for radiocarbon dating: Results from the early Iron Age at Tell es-Safi/Gath, Israel. Radiocarbon 2012, 54, 371–390. [Google Scholar] [CrossRef]
- Asscher, Y.; Lehmann, G.; Rosen, S.A.; Weiner, S.; Boaretto, E. Absolute dating of the late bronze to iron age transition and the appearance of philistine culture in qubur el-walaydah, southern Levant. Radiocarbon 2015, 57, 77–97. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Hansen, E.; Ginell, W.S. Calcium hydroxide crystal evolution upon aging of lime putty. J. Am. Ceram. Soc. 1998, 81, 3032–3034. [Google Scholar] [CrossRef]
- Boynton, R.S. Chemistry and Technology of Lime and Limestone; John Wiley & Sons, Inc.: New York, NY, USA, 1980. [Google Scholar]
- Zouridakis, N.M.; Saliege, J.F.; Person, A.; Filippakis, S.E. Radiocarbon dating of mortars from ancient greek palaces. Archaeometry 1987, 29, 60–68. [Google Scholar] [CrossRef]
- Ortega, L.A.; Zuluaga, M.C.; Alonso-Olazabal, A.; Murelaga, X.; Insausti, M.; Ibañez-Etxeberria, A. Historic lime-mortar 14C dating of Santa María la Real (Zarautz, northern Spain): Extraction of suitable grain size for reliable 14C dating. Radiocarbon 2012, 54, 23–36. [Google Scholar] [CrossRef]
- Marzaioli, F.; Lubritto, C.; Nonni, S.; Passariello, I.; Capano, M.; Terrasi, F. Mortar radiocarbon dating: Preliminary accuracy evaluation of a novel methodology. Anal. Chem. 2011, 83, 2038–2045. [Google Scholar] [CrossRef] [PubMed]
- Marzaioli, F.; Nonni, S.; Passariello, I.; Capano, M.; Ricci, P.; Lubritto, C.; De Cesare, N.; Eramo, G.; Quirós Castillo, J.A.; Terrasi, F. Accelerator mass spectrometry 14C dating of lime mortars: Methodological aspects and field study applications at CIRCE (Italy). Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 2013, 294, 246–251. [Google Scholar] [CrossRef]
- Folk, R.L.; Valastro, S. Successful Technique for Dating of Lime Mortar by Carbon-14. J. Field Archaeol. 1976, 3, 203–208. [Google Scholar] [CrossRef]
- Goslar, T.; Nawrocka, D.; Czernik, J. Foraminiferous limestone in C-14 dating of mortar. Radiocarbon 2009, 51, 987–993. [Google Scholar] [CrossRef]
- Heinemeier, J.; Ringbom, A.; Lindroos, A.; Sveinbjornsdottir, A.E. Successful AMS 14C dating of non-hydraulic lime mortars from the medieval churches of the Aland Islands, Finland. Radiocarbon 2010, 52, 171–204. [Google Scholar] [CrossRef]
- Al-Bashaireh, K. Plaster and mortar radiocarbon dating of Nabatean and Islamic structures, South Jordan. Archaeometry 2013, 55, 329–354. [Google Scholar] [CrossRef]
- Ringbom, A.; Lindroos, A.; Heinemeier, J.; Sonck-Koota, P. 19 years of mortar dating: Learning from experience. Radiocarbon 2014, 56, 619–635. [Google Scholar] [CrossRef]
- Nonni, S.; Marzaioli, F.; Secco, M.; Passariello, I.; Capano, M.; Lubritto, C.; Mignardi, S.; Tonghini, C.; Terrasi, F. 14C mortar dating: The case of the medieval shayzar citadel, Syria. Radiocarbon 2013, 55, 514–525. [Google Scholar] [CrossRef]
- Pesce, G.; Quarta, G.; Calcagnile, L.; D’Elia, M.; Cavaciocchi, P.; Lastrico, C.; Guastella, R. Radiocarbon dating of lumps from aerial lime mortars and plasters: Methodological issues and results from San Nicolò of Capodimonte church (Camogli, Genoa, Italy). Radiocarbon 2009, 51, 867–872. [Google Scholar] [CrossRef]
- Pesce, G.L.A.; Ball, R.J.; Quarta, G.; Calcagnile, L. Identification, extraction, and preparation of reliable lime samples for 14C dating of plasters and mortars with the “pure lime lumps” technique. Radiocarbon 2012, 54, 933–942. [Google Scholar] [CrossRef]
- Beruto, D.T.; Vecchiattini, R.; Giordani, M. Solid products and rate-limiting step in the thermal half decomposition of natural dolomite in a CO2 (g) atmosphere. Thermochim. Acta 2003, 405, 183–194. [Google Scholar] [CrossRef]
- Schork, J. Dolomitic Lime in the US. J. Archit. Conserv. 2012, 18, 7–25. [Google Scholar] [CrossRef]
- Kuzel, H.-J.; Baier, H. Hydration of calcium aluminate cements in the presence of calcium carbonate. Eur. J. Mineral. 1996, 129–142. [Google Scholar] [CrossRef]
- Evans, D.G.; Slade, R.C.T. Structural aspects of layered double hydroxides. In Layered Double Hydroxides; Duan, X., Evans, D.G., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 1–87. [Google Scholar]
- Forano, C.; Hibino, T.; Leroux, F.; Taviot-Guého, C. Chapter 13.1 layered double hydroxides. Dev. Clay Sci. 2006, 1, 1021–1095. [Google Scholar]
- Guo, Q.; Reardon, E.J. Calcined dolomite: Alternative to lime for minimizing undesirable element leachability from fly ash. Ind. Eng. Chem. Res. 2012, 51, 9106–9116. [Google Scholar] [CrossRef]
- Miyata, S.; Okada, A. Synthesis of hydrotalcite-like compounds and their physico-chemical properties—The systems Mg2+-Al3+-SO42−- and Mg2+-Al3+-CrO42−. Clays Clay Min. 1977, 25, 14–18. [Google Scholar] [CrossRef]
- Miyata, S. Physico-chemical properties of synthetic hydrotalcites in relation to composition. Clays Clay Min. 1980, 28, 50–56. [Google Scholar] [CrossRef]
- Miyata, S. Anion-exchange properties of hydrotalcite-like compounds. Clays Clay Min. 1983, 31, 305–311. [Google Scholar] [CrossRef]
- Reichle, W.T. Synthesis of anionic clay minerals (mixed metal hydroxides, hydrotalcite). Solid State Ionics 1986, 22, 135–141. [Google Scholar] [CrossRef]
- Cavani, F.; Trifirò, F.; Vaccari, A. Hydrotalcite type anionic clays, preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Mills, S.J.; Christy, A.G.; Génin, J.-M.R.; Kameda, T.; Colombo, F. Nomenclature of the hydrotalcite supergroup: Natural layered double hydroxides. Mineral. Mag. 2012, 76, 1289–1336. [Google Scholar] [CrossRef]
- Kutus, B.; Gacsi, A.; Pallagi, A.; Palinko, I.; Peintler, G.; Sipos, P. A comprehensive study on the dominant formation of the dissolved Ca(OH)2 (aq) in strongly alkaline solutions saturated by Ca(ii). RSC Adv. 2016, 6, 45231–45240. [Google Scholar] [CrossRef]
- Pallagi, A.; Tasi, A.; Gácsi, A.; Csáti, M.; Pálinkó, I.; Peintler, G.; Sipos, P. The solubility of Ca(OH)2 in extremely concentrated NaOH solutions at 25 °C. Cent. Eur. J. Chem. 2012, 10, 332–337. [Google Scholar] [CrossRef]
- Reardon, E.J.; Della Valle, S. Anion sequestering by the formation of anionic clays: Lime treatment of fly ash slurries. Environ. Sci. Technol. 1997, 31, 1218–1223. [Google Scholar] [CrossRef]
- Rosenberg, S.P.; Armstrong, L. Layered double hydroxides in the Bayer process: Past, present and future. In Essential Readings in Light Metals; Springer: Berlin, Germany, 2005; pp. 157–161. [Google Scholar]
- Sipos, P. The structure of Al (III) in strongly alkaline aluminate solutions—A review. J. Mol. Liq. 2009, 146, 1–14. [Google Scholar] [CrossRef]
- Gácsi, A.; Kutus, B.; Kónya, Z.; Kukovecz, Á.; Pálinkó, I.; Sipos, P. Estimation of the solubility product of hydrocalumite-hydroxide, a layered double hydroxide with the formula of [Ca2Al(OH)6]OH·nH2O. J. Phys. Chem. Solids 2016, 98, 167–173. [Google Scholar] [CrossRef]
- Artioli, G.; Secco, M.; Addis, A.; Bellotto, M. 5 Role of hydrotalcite-type layered double hydroxides in delayed pozzolanic reactions and their bearing on mortar dating. In Cementitious Materials: Composition, Properties, Application; Walter de Gruyter: Berlin, Germany, 2017. [Google Scholar]
- Siedel, H.; Michalski, S.; Ullrich, B. Characterisation of dolomitic lime mortars from the Benedictine monastery in Riesa, Saxony (Germany). In Historic Mortars; Springer: Dordrecht, The Netherlands, 2012; pp. 115–124. [Google Scholar]
- Brandon, C.J.; Hohlfelder, R.L.; Jackson, M.D.; Oleson, J.P. Building for Eternity: The History and Technology of Roman Concrete Engineering in the Sea; Oxbow Books: Oxford, UK; Philadelphia, PA, USA, 2014. [Google Scholar]
- Massazza, F. Pozzolana and pozzolanic cements. In Lea’s Chemistry of Cement and Concrete, 4th ed.; Hewlett, P., Ed.; Elsevier-Butterworth-Heinemann: Oxford, UK, 1998; pp. 471–635. [Google Scholar]
- Sagredo, I. Navarra: Castillos que Defendieron el Reino (Tomo III) La Navarra Occidental, la Frontera del Mar Álava, Bizkaia, el Duranguesado, Guipuzkoa; PAMIELA: Pamplona, Spain, 2007. [Google Scholar]
- Solaun, J.L.; Azcárate, A. El castillo de Portilla (Zambrana, Alava). Origen y significado de una fortaleza plenomedieval en territorio alavés (siglos XI-XII). Munibe Antropol.-Arkeol. 2016, 67, 167–183. [Google Scholar]
- Ramsey, C.B.; Scott, M.; van der Plicht, H. Calibration for archaeological and environmental terrestrial samples in the time range 26–50 ka cal BP. Radiocarbon 2013, 55, 2021–2027. [Google Scholar] [CrossRef]
- Reimer, P.J.; Bard, E.; Bayliss, A.; Beck, J.W.; Blackwell, P.G.; Ramsey, C.B.; Buck, C.E.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. IntCal13 and marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 2013, 55, 1869–1887. [Google Scholar] [CrossRef]
- Casadio, F.; Chiari, G.; Simon, S. Evaluation of binder/aggregate ratios in archaeological lime mortars with carbonate aggregate: A comparative assessment of chemical, mechanical and microscopic approaches. Archaeometry 2005, 47, 671–689. [Google Scholar] [CrossRef]
- Warkentin, B.P.; Maeda, T. Physical and mechanical characteristics of andisols. In Soils with Variable Charge; Theng, B.K.G., Ed.; New Zealand Society of Soil Science: Lower Hutt, New Zealand, 1980; pp. 281–302. [Google Scholar]
- Langmuir, D. Aqueous Environmental Geochemistry; Prentice Hall: London, UK, 1997. [Google Scholar]
- Grover, K.; Komarneni, S.; Katsuki, H. Synthetic hydrotalcite-type and hydrocalumite-type layered double hydroxides for arsenate uptake. Appl. Clay Sci. 2010, 48, 631–637. [Google Scholar] [CrossRef]
- Bakolas, A.; Biscontin, G.; Moropoulou, A.; Zendri, E. Characterization of structural byzantine mortars by thermogravimetric analysis. Thermochim. Acta 1998, 321, 151–160. [Google Scholar] [CrossRef]
- Paama, L.; Pitkänen, I.; Rönkkömäki, H.; Perämäki, P. Thermal and infrared spectroscopic characterization of historical mortars. Thermochim. Acta 1998, 320, 127–133. [Google Scholar] [CrossRef]
- Moropoulou, A.; Bakolas, A.; Anagnostopoulou, S. Composite materials in ancient structures. Cem. Concr. Compos. 2005, 27, 295–300. [Google Scholar] [CrossRef]
- Yang, W.; Kim, Y.; Liu, P.K.T.; Sahimi, M.; Tsotsis, T.T. A study by in situ techniques of the thermal evolution of the structure of a Mg-Al-CO3 layered double hydroxide. Chem. Eng. Sci. 2002, 57, 2945–2953. [Google Scholar] [CrossRef]
- León, M.; Díaz, E.; Bennici, S.; Vega, A.; Ordóñez, S.; Auroux, A. Adsorption of CO2 on hydrotalcite-derived mixed oxides: Sorption mechanisms and consequences for adsorption irreversibility. Ind. Eng. Chem. Res. 2010, 49, 3663–3671. [Google Scholar] [CrossRef]


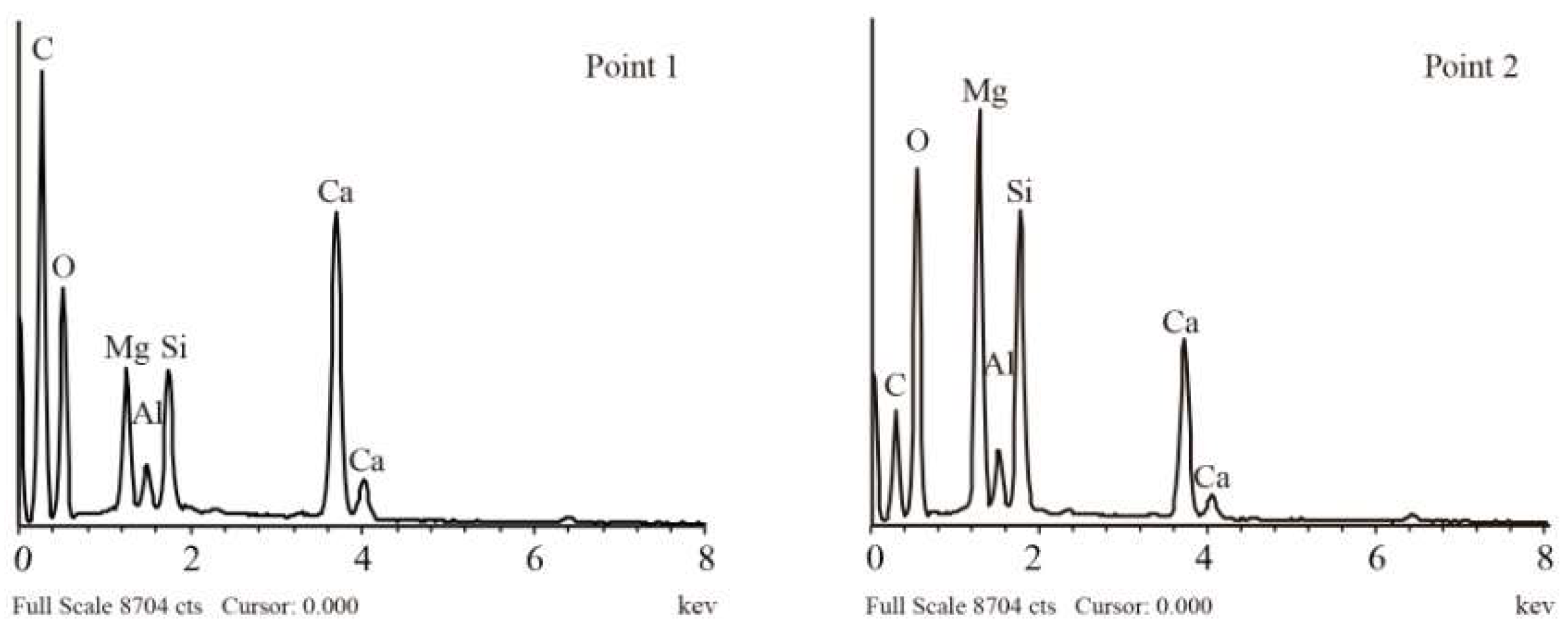
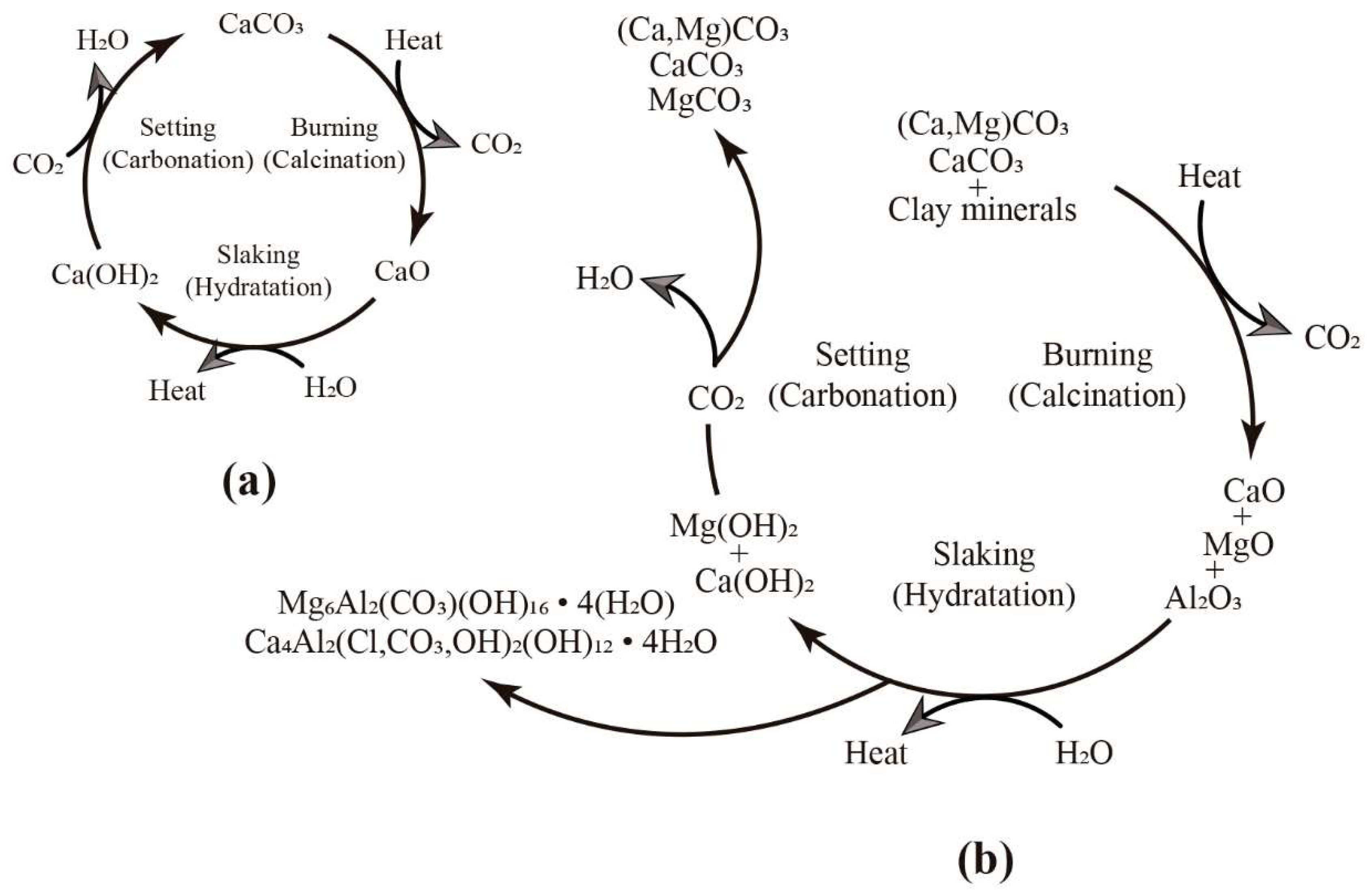


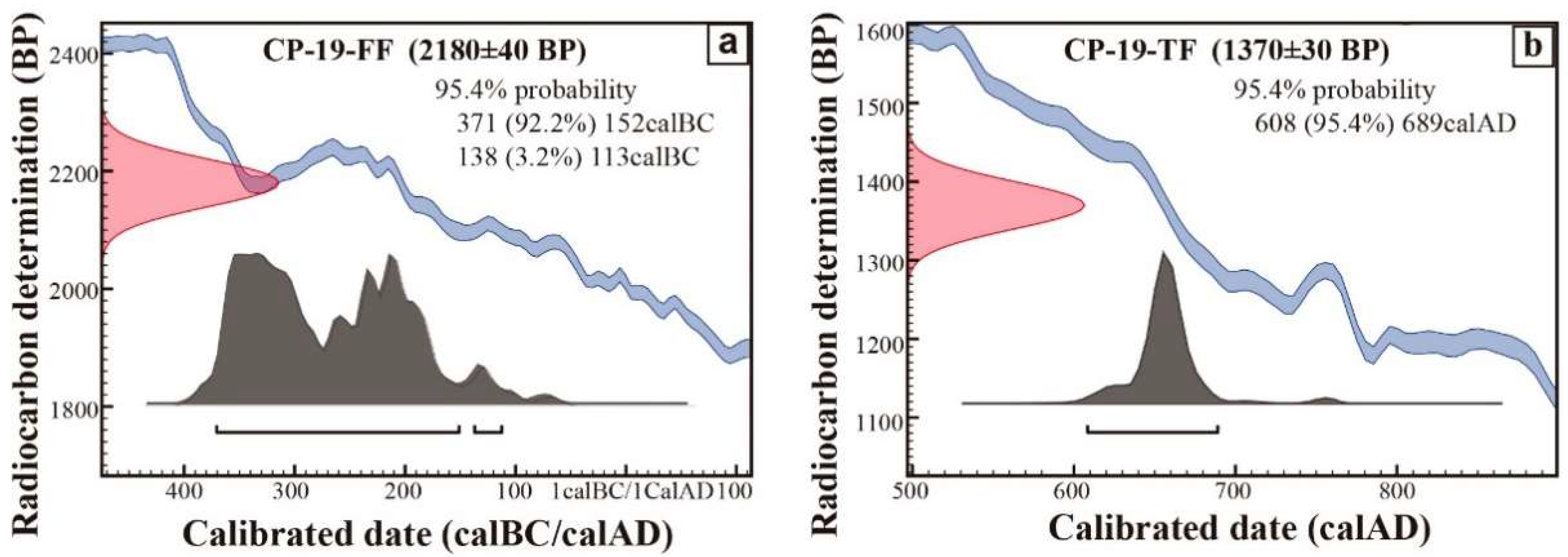
| Stratigraphic Unit | Archaeological Structure | Sample |
|---|---|---|
| SU-2 | West Town (A14) | CP-2 |
| SU-4 | Tower Keep (A12) | CP-4 |
| CP-13-6 | ||
| CP-13-8 | ||
| CP-13-10 | ||
| SU-19 | East Town (A11) | CP-19 |
| CP-13-3 |
| Sample | Target Fraction (%) | Ultrafine Fraction (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mg-Cal | HT | HC | Qtz | Total LDHs | Mg-Cal | HT | HC | Qtz | Total LDHs | |
| CP-2 | 88 | 3 | 6 | 3 | 9 | 64 | 12 | 24 | - | 36 |
| CP-4 | 84 | 8 | 8 | - | 16 | 58 | 16 | 26 | - | 42 |
| CP-13-6 | 62 | 10 | 28 | - | 38 | 38 | 28 | 34 | - | 62 |
| CP-13-8 | 79 | 9 | 10 | 2 | 19 | 48 | 28 | 24 | - | 52 |
| CP-13-10 | 85 | 4 | 10 | 1 | 14 | 55 | 22 | 23 | - | 45 |
| CP-19 | 89 | 5 | 5 | 1 | 10 | 55 | 24 | 20 | - | 44 |
| CP-13-3 | 68 | 13 | 18 | 1 | 31 | 61 | 11 | 28 | - | 39 |
| Sample | Pattern Type | H2O(Itp) | H2O(Itl) | OH− | CO32− |
|---|---|---|---|---|---|
| CP-2-TF | A | 1.46 | 1.35 | 10.40 | 23.47 |
| CP-4-TF | A | 2.86 | 1.82 | 12.8 | 16.28 |
| CP-13-6-TF | B | 2.30 | 2.78 | 19.64 | 18.46 |
| CP-13-8-TF | B | 1.89 | 2.28 | 15.41 | 23.04 |
| CP-13-10-TF | B | 1.89 | 2.12 | 17.01 | 21.83 |
| CP-19-TF | A | 2.54 | 1.41 | 10.31 | 23.29 |
| CP-13-3-TF | B | 2.77 | 2.45 | 13.56 | 18.06 |
| Lab Code | Sample | Binder Grain-Size | Conventional Age | δ13C | Calibrate Age (95.4%) |
|---|---|---|---|---|---|
| BETA375404 | CP-19-TF | 0.5–2 µm | 1370 ± 30 BP | −16.5 | Cal AD 640–675 |
| BETA343295 | CP-19-FF | <2 µm | 2180 ± 40 BP | −21.3 | Cal BC 380–160 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ponce-Antón, G.; Ortega, L.A.; Zuluaga, M.C.; Alonso-Olazabal, A.; Solaun, J.L. Hydrotalcite and Hydrocalumite in Mortar Binders from the Medieval Castle of Portilla (Álava, North Spain): Accurate Mineralogical Control to Achieve More Reliable Chronological Ages. Minerals 2018, 8, 326. https://doi.org/10.3390/min8080326
Ponce-Antón G, Ortega LA, Zuluaga MC, Alonso-Olazabal A, Solaun JL. Hydrotalcite and Hydrocalumite in Mortar Binders from the Medieval Castle of Portilla (Álava, North Spain): Accurate Mineralogical Control to Achieve More Reliable Chronological Ages. Minerals. 2018; 8(8):326. https://doi.org/10.3390/min8080326
Chicago/Turabian StylePonce-Antón, Graciela, Luis Angel Ortega, Maria Cruz Zuluaga, Ainhoa Alonso-Olazabal, and Jose Luis Solaun. 2018. "Hydrotalcite and Hydrocalumite in Mortar Binders from the Medieval Castle of Portilla (Álava, North Spain): Accurate Mineralogical Control to Achieve More Reliable Chronological Ages" Minerals 8, no. 8: 326. https://doi.org/10.3390/min8080326
APA StylePonce-Antón, G., Ortega, L. A., Zuluaga, M. C., Alonso-Olazabal, A., & Solaun, J. L. (2018). Hydrotalcite and Hydrocalumite in Mortar Binders from the Medieval Castle of Portilla (Álava, North Spain): Accurate Mineralogical Control to Achieve More Reliable Chronological Ages. Minerals, 8(8), 326. https://doi.org/10.3390/min8080326





