A Mosaic of Colors: Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Glassy Matrices
3.2. Colorless Tesserae
3.3. Turquoise, Blue and White Tesserae
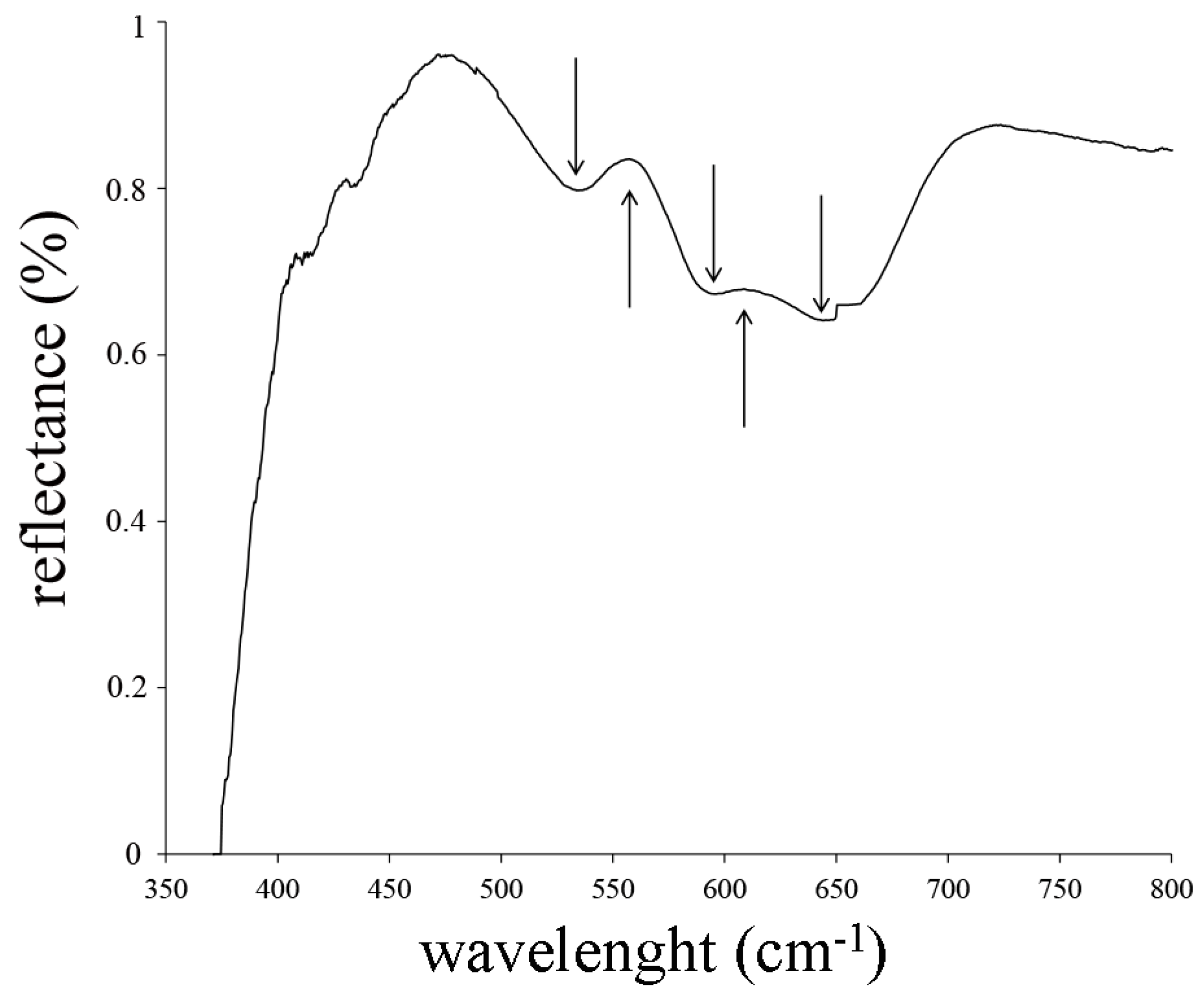
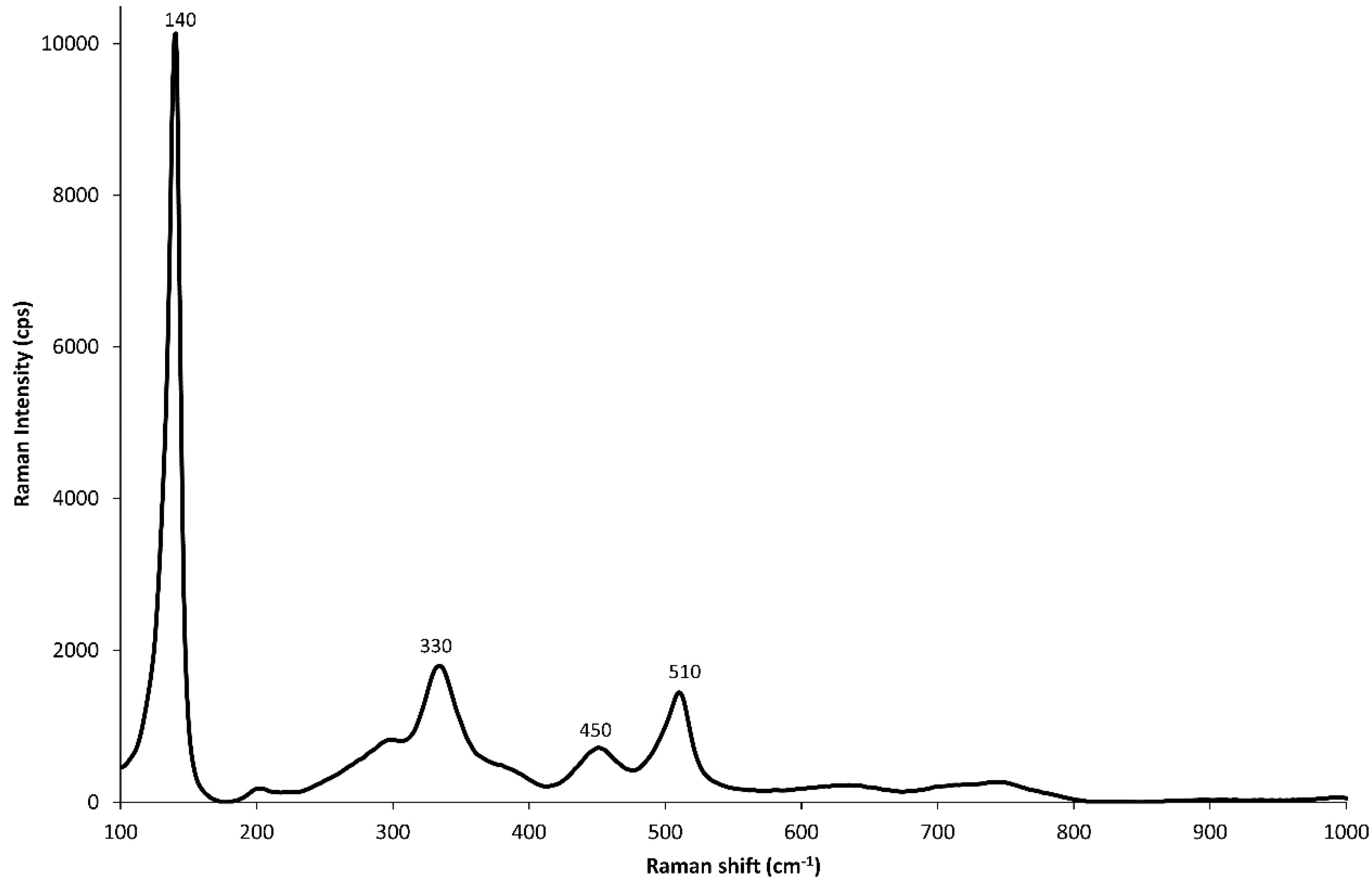
3.4. Yellow and Green Tesserae
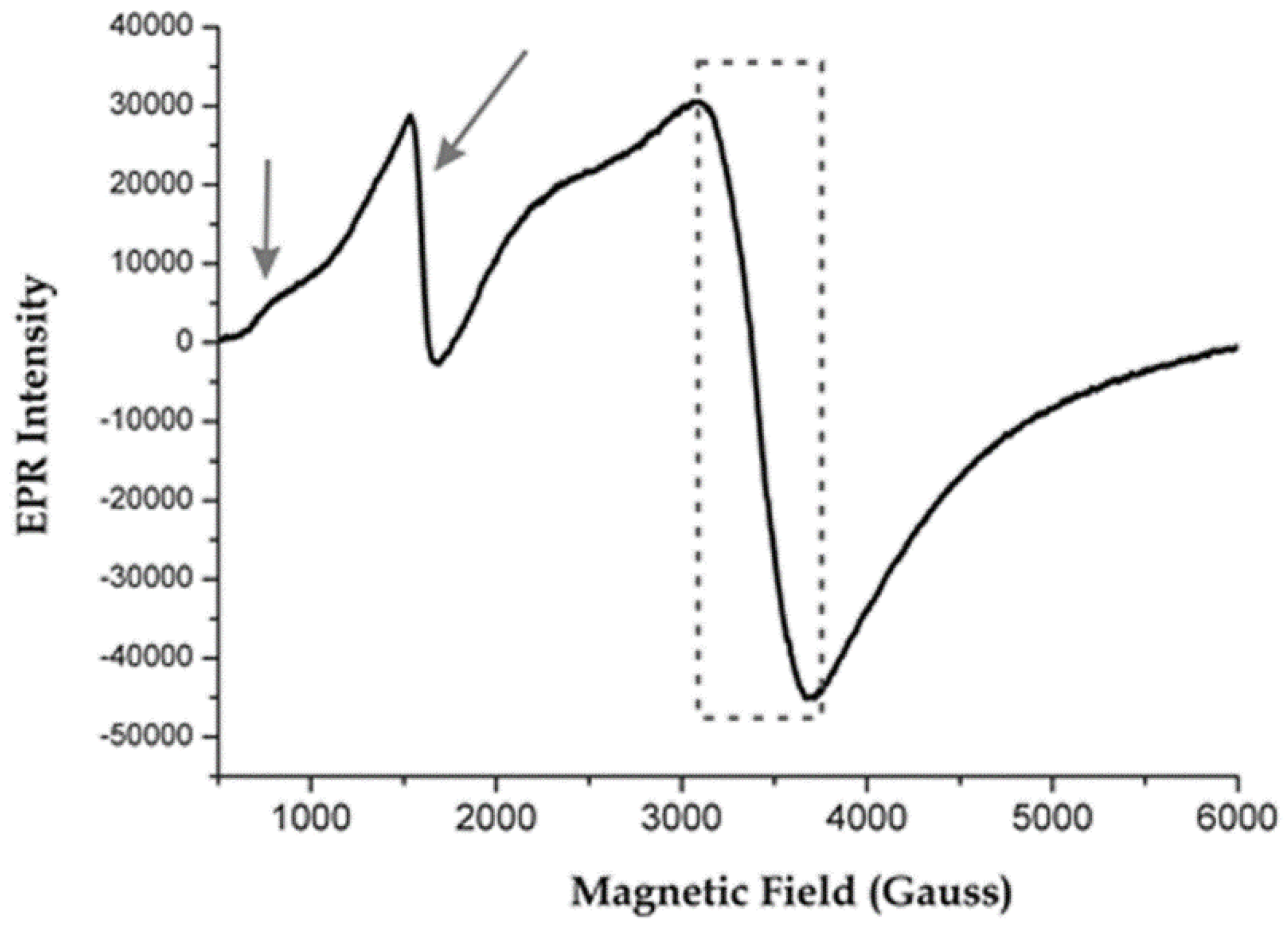
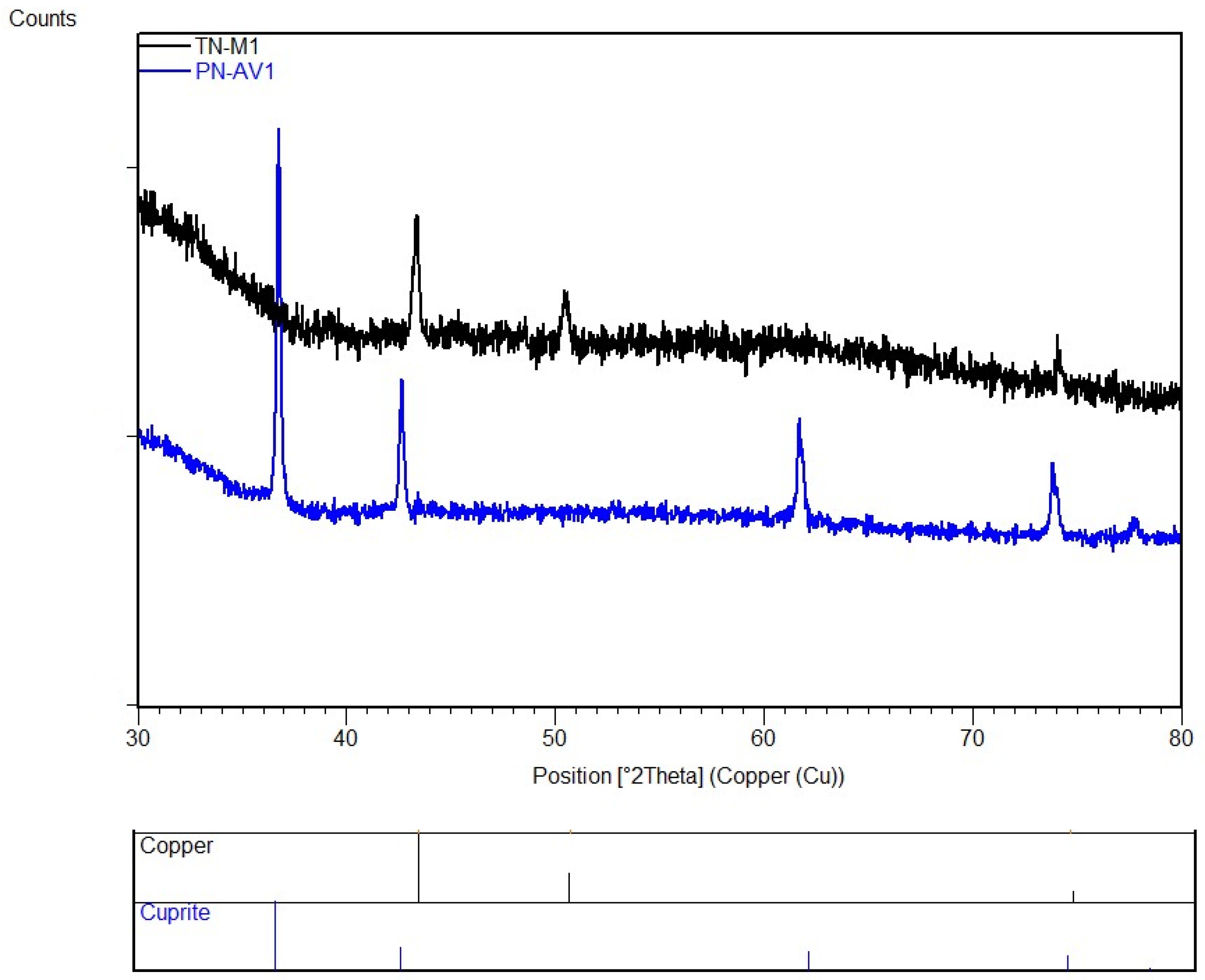
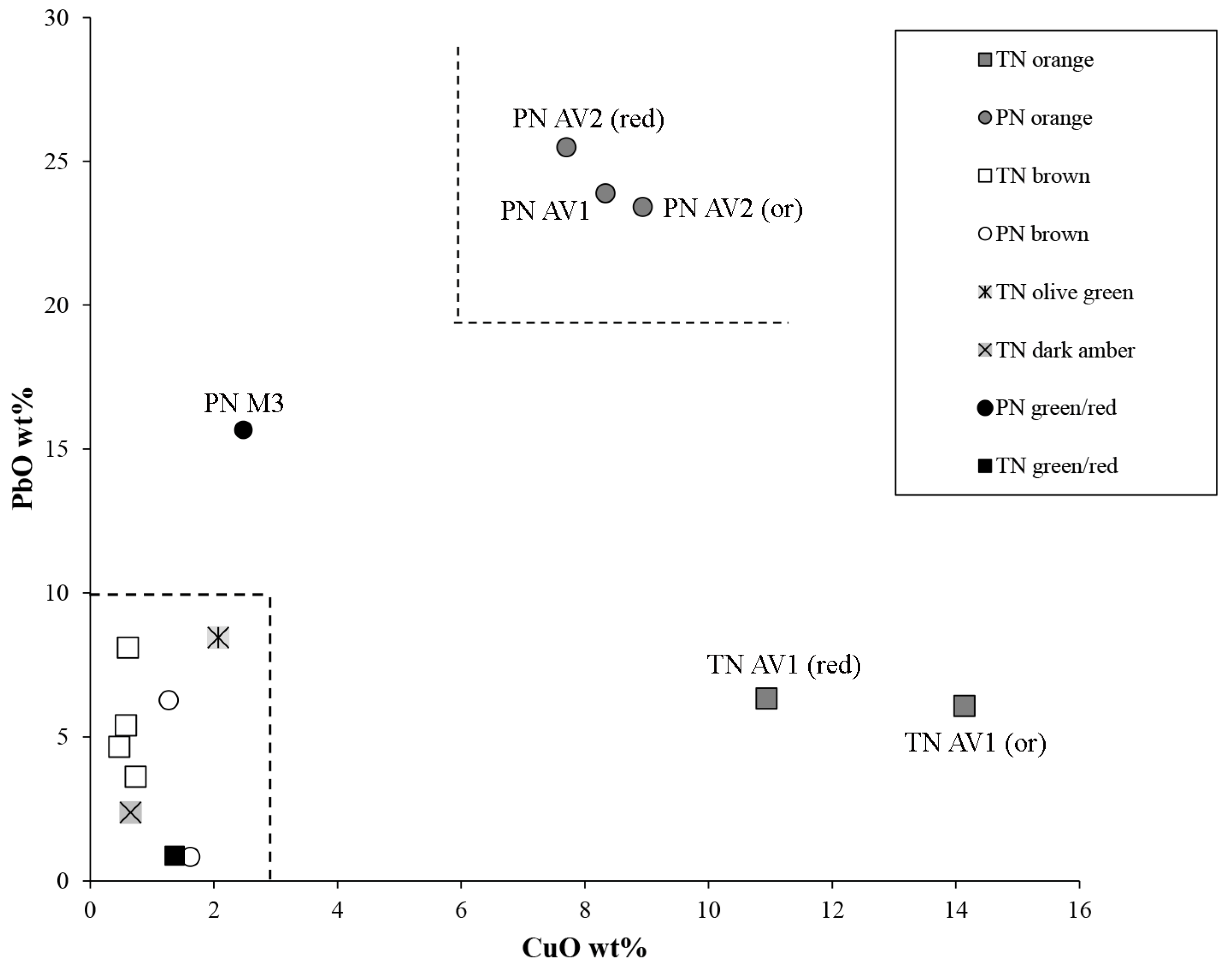
3.5. Red Tesserae
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Verità, M. Tecniche di fabbricazione dei materiali musivi vitrei. Indagini chimiche e mineralogiche. In Medieval Mosaics Light, Color, Materials; Borsook, E., Gioffredi Superbi, F., Pagliarulo, G., Eds.; Silvana Editoriale S.p.A.: Cinisello Balsamo, Italy, 2000; pp. 47–64. ISBN 88-8215-265-0. [Google Scholar]
- Galli, S.; Mastelloni, M.; Ponterio, R.; Sabatino, G.; Triscari, M. Raman and scanning electron microscopy and energy-dispersive X-ray techniques for the characterization of colouring and opaquening agents in Roman mosaic glass tesserae. J. Raman Spectrosc. 2004, 35, 622–627. [Google Scholar] [CrossRef]
- Ricciardi, P.; Colomban, P.; Tournié, A.; Macchiarola, M.; Ayed, N. A non-invasive study of Roman Age mosaic glass tesserae by means of Raman spectroscopy. J. Archaeol. Sci. 2009, 36, 2551–2559. [Google Scholar] [CrossRef]
- Neri, E.; Morvan, C.; Colomban, P.; Guerra, M.F.; Prigent, V. Late Roman and Byzantine mosaic opaque “glass-ceramics” tesserae (5th–9th Century). Ceram. Int. 2016, 42, 18859–18869. [Google Scholar] [CrossRef]
- Neri, E.; Jackson, M.; O’Hea, M.; Gregory, T.; Blet-Lemarquand, M.; Schibille, N. Analyses of glass tesserae from Kilise Tepe: New insights into an early Byzantine production technology. J. Archaeol. Sci. Rep. 2017, 11, 600–612. [Google Scholar] [CrossRef]
- Van der Werf, I.; Mangone, A.; Giannossa, L.C.; Traini, A.; Laviano, R.; Coralini, A.; Sabbatini, L. Archaeometric investigation of Roman tesserae from Herculaneum (Italy) by the combined use of complementary micro-destructive analytical techniques. J. Archaeol. Sci. 2009, 36, 2625–2634. [Google Scholar] [CrossRef]
- Silvestri, A.; Tonietto, S.; Molin, G. The palaeo-Christian glass mosaic of St. Prosdocimus (Padova, Italy): Archaeometric characterisation of “gold” tesserae. J. Archaeol. Sci. 2011, 38, 3402–3414. [Google Scholar] [CrossRef]
- Gliozzo, E.; Santagostino Barbone, A.; Turchiano, M.; Memmi, I.; Volpe, G. The coloured tesserae decorating the vaults of the Faragola balneum (Ascoli Satriano, Foggia, Southern Italy). Archaeometry 2012, 54, 311–331. [Google Scholar] [CrossRef]
- Silvestri, A.; Tonietto, S.; Molin, G.; Guerriero, P. The palaeo-Christian glass mosaic of St. Prosdocimus (Padova, Italy): Archaeometric characterisation of tesserae with antimony- or phosphorus-based opacifiers. J. Archaeol. Sci. 2012, 39, 2177–2190. [Google Scholar] [CrossRef]
- Di Bella, M.; Quartieri, S.; Sabatino, G.; Santalucia, F.; Triscari, M. The glass mosaics tesserae of “Villa del Casale” (Piazza Armerina, Italy): A multi-technique archaeometric study. Archaeol. Anthropol. Sci. 2013, 6, 345–362. [Google Scholar] [CrossRef]
- Silvestri, A.; Tonietto, S.; Molin, G.; Guerriero, P. The palaeo-Christian glass mosaic of St. Prosdocimus (Padova, Italy): Archaeometric characterisation of tesserae with copper- or tin-based opacifiers. J. Archaeol. Sci. 2014, 42, 51–67. [Google Scholar] [CrossRef]
- Paynter, S.; Kearns, T.; Cool, H.; Chenery, S. Roman coloured glass in the Western provinces: The glass cakes and tesserae from West Clacton in England. J. Archaeol. Sci. 2015, 62, 66–81. [Google Scholar] [CrossRef]
- Silvestri, A.; Tonietto, S.; Molin, G.; Guerriero, P. Multi-methodological study of palaeo-Christian glass mosaic tesserae of St. Maria Mater Domini (Vicenza, Italy). Eur. J. Mineral. 2015, 27, 225–245. [Google Scholar] [CrossRef]
- Shortland, A.J. The use and origin of antimonate colorants in early Egyptian glass. Archaeometry 2002, 44, 517–530. [Google Scholar] [CrossRef]
- Freestone, I.C.; Stapleton, C.P.; Rigby, V. The production of red glass and enamel in the Late Iron Age, Roman and Byzantine periods. In Through a Glass Brightly: Studies in Byzantine and Medieval Art and Archaeology; Presented to David Buckton; Entwistle, C., Buckton, D., Eds.; Oxbow Books: Oxford, UK, 2003; pp. 142–154. ISBN 978-1785702518. [Google Scholar]
- Lahlil, S.; Biron, I.; Galoisy, L.; Morin, G. Technological processes to produce antimonate opacified glass throughout history. In Proceedings of the Annales du 17e Congrés de l’Association Internationale pour l’Histoire du Verre, Antwerp, Belgium, 4–8 September 2006; Janssens, K., Degryse, P., Cosyns, P., Caen, J., Van’t dack, L., Eds.; University Press Antwerp: Antwerp, Belgium, 2009; pp. 571–578. [Google Scholar]
- Molina, G.; Odin, G.P.; Pradell, T.; Shortland, A.J.; Tite, M.S. Production technology and replication of lead antimonate yellow glass from New Kingdom Egypt and the Roman Empire. J. Archaeol. Sci. 2014, 41, 171–184. [Google Scholar] [CrossRef]
- Lahlil, S.; Biron, I.; Galoisy, L.; Morin, G. Rediscovering ancient glass technologies through the examination of opacifier crystals. Appl. Phys. A 2008, 92, 109–116. [Google Scholar] [CrossRef]
- Lahlil, S.; Biron, I.; Cotte, M.; Susini, J. New insight on the in situ crystallization of calcium antimonate opacified glass during the Roman period. Appl. Phys. A 2010, 100, 683–692. [Google Scholar] [CrossRef]
- Lahlil, S.; Cotte, M.; Biron, I.; Szlachetko, J.; Menguy, N.; Susini, J. Synthesizing lead antimonate in ancient and modern opaque glass. J. Anal. At. Spectrom. 2011, 26, 1040–1050. [Google Scholar] [CrossRef]
- Brill, R.H.; Cahill, N.D. A red opaque glass from Sardis and some thoughts on red opaques in general. J. Glass Stud. 1988, 30, 16–27. [Google Scholar]
- Barber, D.J.; Freestone, I.C.; Moulding, K.M. Ancient copper red glasses: Investigation and analysis by microbeam techniques. In From Mine to Microscope. Advances in the Study of Ancient Technology; Shortland, A.J., Freestone, I.C., Rehren, T., Eds.; Oxbow Books: Oxford, UK, 2009; pp. 115–127. ISBN 978-1-84217-259-9. [Google Scholar]
- Schibille, N.; McKenzie, J. Glass tesserae from Hagios Polyeuktos, Constantinopole: Their early Byzantine affiliations. In Neighbours and Successors of Rome. Tradition of Glass Production and Use in Europe and the Middle East in the Later 1st Millenium AD; Keller, D., Price, J., Jackson, C.M., Eds.; Oxbow Books: Oxford, UK, 2014; pp. 114–127. ISBN 978178297397. [Google Scholar]
- Verità, M. Analisi di tessere musive vitree del battistero della Basilica di San Marco in Venezia. In Proceedings of the Scienza e Tecnica del Restauro della Basilica di San Marco: Atti del Convegno Internazionale di Studi, Venezia, Italy, 16–19 May 1995; Vio, E., Lepschy, A., Eds.; Istituto Veneto di Scienze, Lettere ed Arti: Venezia, Italy, 1999; pp. 567–585. ISBN 8886166796. [Google Scholar]
- Tite, M.S.; Shortland, A.J. Production technology for copper-and cobalt-blue vitreous materials from the New Kingdom site of Amarna—A reappraisal. Archaeometry 2003, 45, 285–312. [Google Scholar] [CrossRef]
- Vichy, M.; Picon, M.; Thirion-Merle, V. Le manganèse comme impureté, décolorant ou colorant des verres au natron. Bull. l’AFAV 2002, 15–17. Available online: http://www.afaverre.fr/pdf/bull2002/07_Bull2002_M._Vichy%20et%20alii_p15-17.pdf (accessed on 15 June 2018).
- Barca, D.; Basso, E.; Bersani, D.; Galli, G.; Invernizzi, C.; La Russa, M.F.; Lottici, P.P.; Malagodi, M.; Ruffolo, S.A. Vitreous tesserae from the calidarium mosaics of the Villa dei Quintili, Rome. Chemical composition and production technology. Microchem. J. 2016, 124, 726–735. [Google Scholar] [CrossRef]
- Conte, A.; Salvadori, M.; Tirone, C. La Villa Romana di Torre di Pordenone. Tracce della Residenza di un Ricco Dominus Nella Cisalpina Orientale; Quasar: Roma, Italy, 1999; ISBN 8871401654. [Google Scholar]
- Guaitoli, M.T. Il progetto di Santa Maria Maggiore (Trento). Relazione preliminare: Dallo scavo alla diffusione dei dati. J. Fasti Online 2011, 238, 1–18. [Google Scholar]
- Maltoni, S.; Silvestri, A. Innovation and tradition in the fourth century mosaic of the Casa delle Bestie Ferite in Aquileia, Italy: Archaeometric characterisation of the glass tesserae. Archaeol. Anthropol. Sci. 2018, 10, 415–429. [Google Scholar] [CrossRef]
- Brill, R.H. Chemical Analyses of Early Glasses. Volume 2 Tables of Analyses; The Corning Museum of Glass: Corning, NY, USA, 1999; ISBN 0-872900-143-2. [Google Scholar]
- Savitzky, A.; Golay, M.J.E. Smoothing and differentiation of data by simplified least squares procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Lilyquist, C.; Brill, R.H. Studies in Early Egyptian Glass; Metropolitan Museum of Art: New York, NY, USA, 1993; ISBN 978-0300200195. [Google Scholar]
- Jackson, C.M.; Cottam, S. “A green thought in a green shade”; Compositional and typological observations concerning the production of emerald green glass vessels in the 1st century A.D. J. Archaeol. Sci. 2015, 61, 139–148. [Google Scholar] [CrossRef]
- Van Der Linden, V.; Cosyns, P.; Schalm, O.; Cagno, S.; Nys, K.; Janssens, K.; Nowak, A.; Wagner, B.; Bulska, E. Deeply Coloured and black glass in the northern provinces of the Roman empire: Differences and similarities in chemical composition before and after AD 150. Archaeometry 2009, 51, 822–844. [Google Scholar] [CrossRef]
- Maltoni, S.; Silvestri, A.; Molin, G. Opaque red glass tesserae from Roman and early-Byzantine sites of north-eastern Italy: New light on production technologies. In Proceedings of the Annales du 20e Congrès de l’Association Internationale pour l’Histoire du Verre, Fribourg, Switzerland, 7–11 September 2015; Wolf, S., de Pury-Gysel, A., Eds.; Verlag Marie Leidorf GmbH: Rahden, Germany, 2017; pp. 280–287. [Google Scholar]
- Jackson, C.M.; Paynter, S. A Great Big Melting Pot: Exploring Patterns of Glass Supply, Consumption and Recycling in Roman Coppergate, York. Archaeometry 2015, 58, 68–95. [Google Scholar] [CrossRef]
- Schibille, N.; Sterrett-Krause, A.; Freestone, I.C. Glass groups, glass supply and recycling in late Roman Carthage. Archaeol. Anthropol. Sci. 2017, 9, 1223–1241. [Google Scholar] [CrossRef]
- Silvestri, A.; Gallo, F.; Maltoni, S.; Degryse, P.; Ganio, M.; Longinelli, A.; Molin, G. Things that travelled—A review of the Roman glass from Northern Adriatic Italy. In Things That Travelled: Mediterranean Glass in the First Millennium CE; Daniela Rosenow, D., Phelps, M., Meek, A., Freestone, I.C., Eds.; UCL Press: London, UK, 2018; pp. 346–367. ISBN 978-1-78735-117-2. [Google Scholar]
- Silvestri, A. The coloured glass of Iulia Felix. J. Archaeol. Sci. 2008, 35, 1489–1501. [Google Scholar] [CrossRef]
- Silvestri, A.; Molin, G.; Salviulo, G. The colourless glass of Iulia Felix. J. Archaeol. Sci. 2008, 35, 331–341. [Google Scholar] [CrossRef]
- Gallo, F.; Silvestri, A.; Molin, G. Glass from the Archaeological Museum of Adria (North–East Italy): New insights into Early Roman production technologies. J. Archaeol. Sci. 2013, 40, 2589–2605. [Google Scholar] [CrossRef]
- Maltoni, S.; Silvestri, A.; Marcante, A.; Molin, G. The transition from Roman to Late Antique glass: New insights from the Domus of Tito Macro in Aquileia (Italy). J. Archaeol. Sci. 2016, 73, 1–16. [Google Scholar] [CrossRef]
- Neri, E.; Verità, M. Glass and metal analyses of gold leaf tesserae from 1st to 9th century mosaics. A contribution to technological and chronological knowledge. J. Archaeol. Sci. 2013, 40, 4596–4606. [Google Scholar] [CrossRef]
- Verità, M.; Zecchin, S. Scientific investigation of Byzantine glass tesserae from the mosaics on the south chapel of Torcello’s Basilica, Venice. In Proceedings of the Annales du 18e Congrès de l’Association International pour l’Histoire du Verre, Thessaloniki, Greece, 20–25 September 2009; Ignatiadou, D., Antonaras, A., Eds.; ZITI Publishing: Thessaloniki, Greece, 2012; pp. 315–320. [Google Scholar]
- Gedzevičiūtė, V.; Welter, N.; Schüssler, U.; Weiss, C. Chemical composition and colouring agents of Roman mosaic and millefiori glass, studied by electron microprobe analysis and Raman microspectroscopy. Archaeol. Anthropol. Sci. 2009, 1, 15–29. [Google Scholar] [CrossRef]
- Basso, E.; Invernizzi, C.; Malagodi, M.; La Russa, M.F.; Bersani, D.; Lottici, P.P. Characterization of colorants and opacifiers in roman glass mosaic tesserae through spectroscopic and spectrometric techniques. J. Raman Spectrosc. 2014, 45, 238–245. [Google Scholar] [CrossRef]
- Lahlil, S.; Biron, I.; Cotte, M.; Susini, J.; Menguy, N. Synthesis of calcium antimonate nano-crystals by the 18th dynasty Egyptian glassmakers. Appl. Phys. A 2010, 98, 1–8. [Google Scholar] [CrossRef]
- Mass, J.L.; Stone, R.E.; Wypyski, M.T. The mineralogical and metallurgical origins of Roman opaque colored glasses. In Prehistory and History of Glassmaking Technology; McCray, P., Kingery, D.W., Eds.; American Ceramic Society: Columbus, OH, USA, 1998; pp. 121–145. ISBN 9781574980417. [Google Scholar]
- Gratuze, B.; Soulier, I.; Barrandon, J.N.; Foy, D. De l’origine du cobalt dans les verres. Rev. d’Archéométrie 1992, 16, 97–108. [Google Scholar] [CrossRef]
- Henderson, J. The Science and Archaeology of Materials: An Investigation of Inorganic Materials; Routledge: London, UK, 2000; ISBN 0415199336. [Google Scholar]
- Bamford, C.R. Colour Generation and Control in Glass; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1977; Volume 3, ISBN 1520-6378. [Google Scholar]
- Cascales, C.; Alonso, J.A.; Rasines, I. NEW PYROCHLORES Pb2(MSb) O 6.5 (M equals Ti,Zr,Sn,Hf). J. Mater. Sci. Lett. 1986, 5, 675–677. [Google Scholar] [CrossRef]
- Rosi, F.; Manuali, V.; Miliani, C.; Brunetti, B.G.; Sgamellotti, A.; Grygar, T.; Hradil, D. Raman scattering features of lead pyroantimonate compounds. Part I: XRD and Raman characterization of Pb2Sb2O7 doped with tin and zinc. J. Raman Spectrosc. 2009, 40, 107–111. [Google Scholar] [CrossRef]
- Rosi, F.; Manuali, V.; Grygar, T.; Bezdicka, P.; Brunetti, B.G.; Sgamellotti, A.; Burgio, L.; Seccaroni, C.; Miliani, C. Raman scattering features of lead pyroantimonate compounds: Implication for the non-invasive identification of yellow pigments on ancient ceramics. Part II. In situ characterisation of Renaissance plates by portable micro-Raman and XRF studies. J. Raman Spectrosc. 2011, 42, 407–414. [Google Scholar] [CrossRef]
- Wypyski, M.T.; Becker, L. Glassmaking Technology at Antioch. In The Arts of Antioch: Art Historical and Scientific Approaches to Roman Mosaics and a Catalogue of the Worchester Art Museum Antioch Collection; Becker, L., Kondoleon, C., Eds.; Princeton University Press: Princeton, NJ, USA, 2005; pp. 115–175. ISBN 9780691122328. [Google Scholar]
- Verità, M.; Maggetti, M.; Saguì, L.; Santopadre, P. Colors of Roman glass: An investigation of the yellow sectilia in the Gorga collection. J. Glass Stud. 2013, 55, 39–52. [Google Scholar]
- Sandalinas, C.; Ruiz-Moreno, S. Lead-Tin-Antimony Yellow: Historical Manufacture, Molecular Characterization and Identification in Seventeenth-Century Italian Paintings. Stud. Conserv. 2004, 49, 41–52. [Google Scholar] [CrossRef]
- Hradil, D.; Grygar, T.; Hradilová, J.; Bezdicka, P.; Grunwaldová, V.; Fogaš, I.; Miliani, C. Microanalytical identification of Pb-Sb-Sn yellow pigment in historical European paintings and its differentiation from lead tin and Naples yellows. J. Cult. Herit. 2007, 8, 377–386. [Google Scholar] [CrossRef]
- Freestone, I.C. Chemical analysis of raw glass fragments. In Excavation at Carthage, The Circular Harbour, North Side. The Site and Finds Other than Pottery; British Academy Monographs in Archeology; Hurst, H.R., Ed.; Oxford University Press: Oxford, UK, 1994; Volume 2, p. 290. ISBN 0-19-727003-4 hbk. [Google Scholar]
- Paynter, S.; Kearns, T. West Clacton Reservoir, Great Bentley, Essex. Analysis of Glass Tesserae; Technology Report; English Heritage: Portsmouth, UK, 2011; Volume 44. [Google Scholar]
- Jackson, C.M.; Cottam, S.; Lazar, I. The green, green glass of Rome. In Proceedings of the Annales du 19e Congrès de l’Association International pour l’Histoire du Verre, Piran, Slovenia, 17–21 September 2012; Lazar, I., Ed.; AIHV: Thessaloniki, Greece, 2015; pp. 109–117. [Google Scholar]
- Schibille, N.; Degryse, P.; Corremans, M.; Specht, C.G. Chemical characterisation of glass mosaic tesserae from sixth-century Sagalassos (south–west Turkey): Chronology and production techniques. J. Archaeol. Sci. 2012, 39, 1480–1492. [Google Scholar] [CrossRef]
- Rehren, T.; Pusch, E.B. Late Bronze Age glass production at Qantir-Piramesses, Egypt. Science 2005, 308, 1756–1758. [Google Scholar] [CrossRef] [PubMed]
- Cable, M.; Smedley, J.W. The replication of an opaque red glass from Nimrud. In Early Vitreous Materials; British Museum Occasional Paper 56; Bimson, M., Freestone, I.C., Eds.; British Museum: London, UK, 1987; pp. 151–164. ISBN 978-0861590568. [Google Scholar]
- Ahmed, A.A.; Ashour, G.M. Effect of heat treatment on the crystallisation of cuprous oxide in glass. Glass Technol. 1981, 22, 24–33. [Google Scholar]
- Freestone, I.C. Composition and microstructure of early opaque red glass. In Early Vitreous Material; British Museum Occasional Paper 56; Bimson, M., Freestone, I.C., Eds.; British Museum: London, UK, 1987; pp. 173–191. ISBN 9780861590568. [Google Scholar]
- Stapleton, C.P.; Freestone, I.C.; Bowman, S.G.E. Composition and origin of Early Mediaeval opaque red enamel from Britain and Ireland. J. Archaeol. Sci. 1999, 26, 913–921. [Google Scholar] [CrossRef]

 ). Compositional ranges of literature groups (Sb-glass; Sb/Mn glass; Mn-glass) calculated from raw data published in Silvestri [40], Silvestri et al. [41], Gallo et al. [42] and Maltoni et al. [43].
). Compositional ranges of literature groups (Sb-glass; Sb/Mn glass; Mn-glass) calculated from raw data published in Silvestri [40], Silvestri et al. [41], Gallo et al. [42] and Maltoni et al. [43].
 ). Compositional ranges of literature groups (Sb-glass; Sb/Mn glass; Mn-glass) calculated from raw data published in Silvestri [40], Silvestri et al. [41], Gallo et al. [42] and Maltoni et al. [43].
). Compositional ranges of literature groups (Sb-glass; Sb/Mn glass; Mn-glass) calculated from raw data published in Silvestri [40], Silvestri et al. [41], Gallo et al. [42] and Maltoni et al. [43].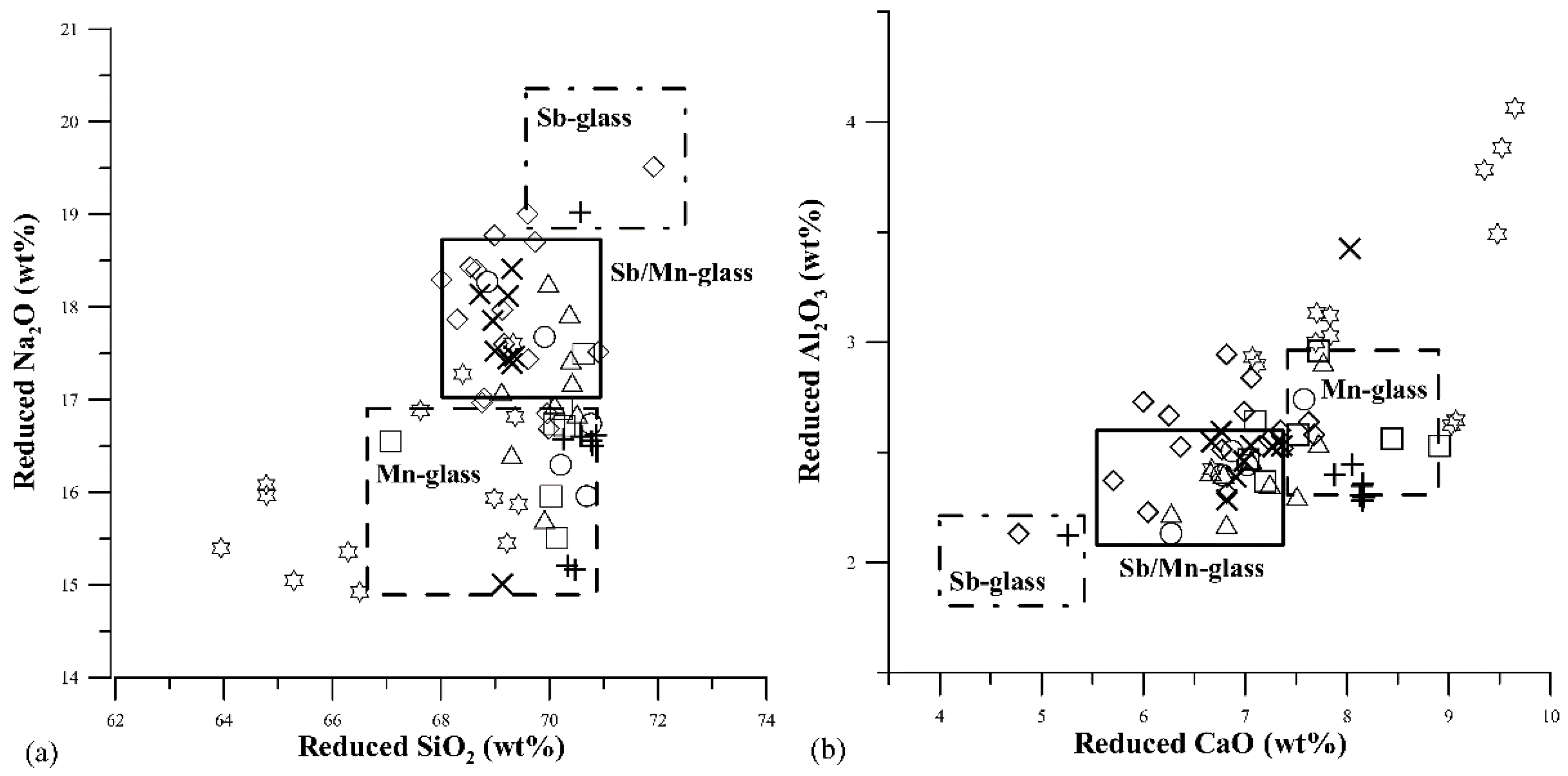


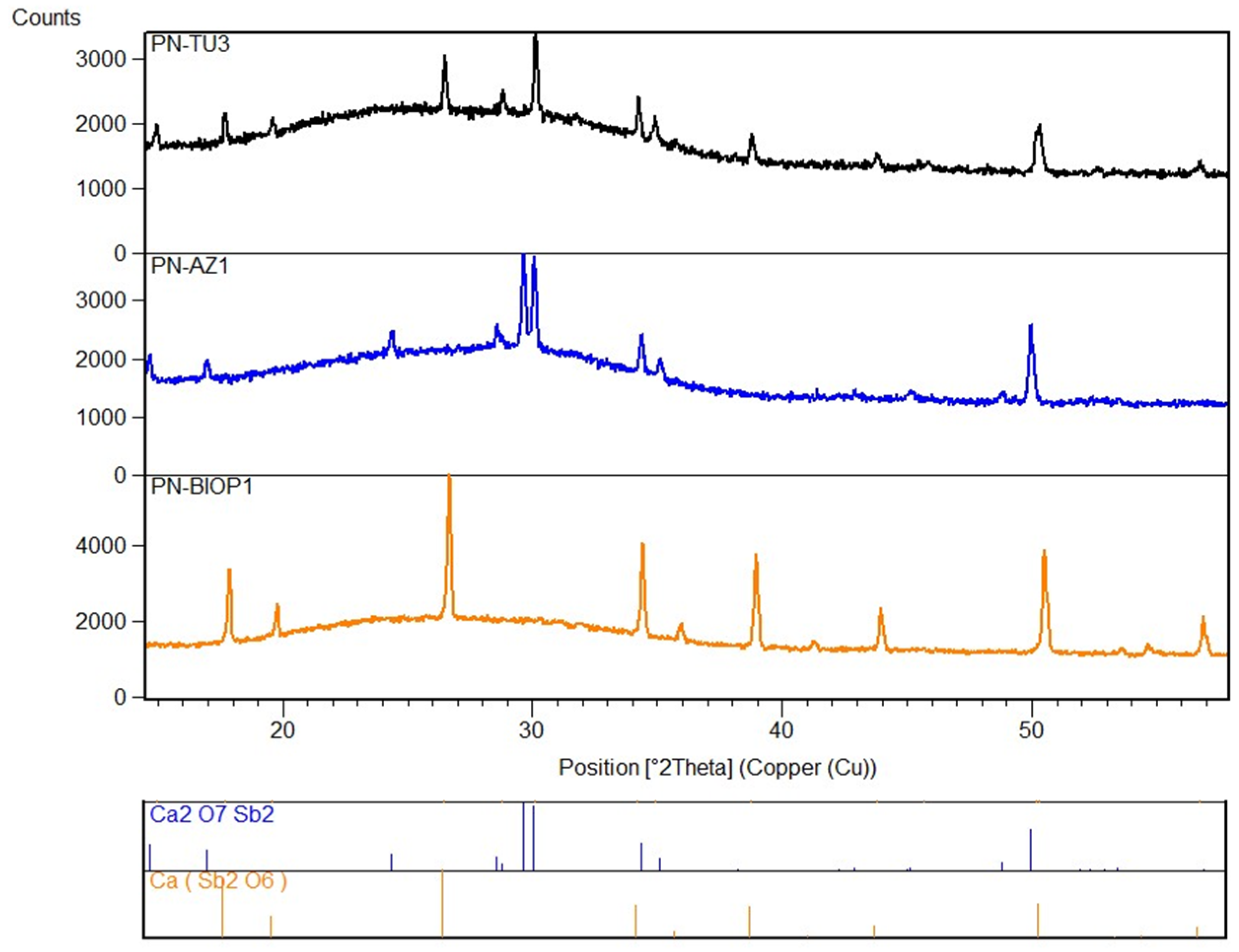
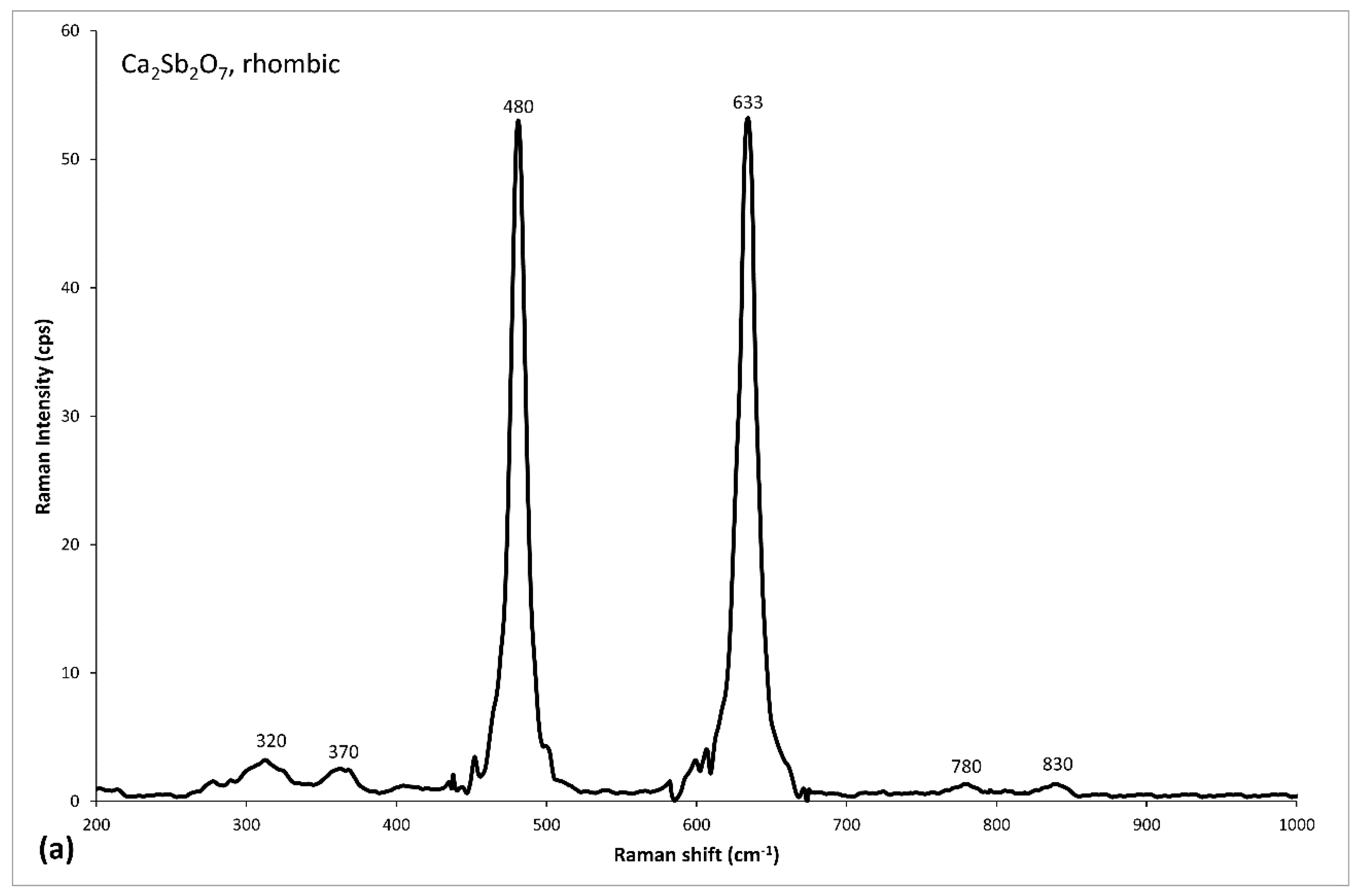
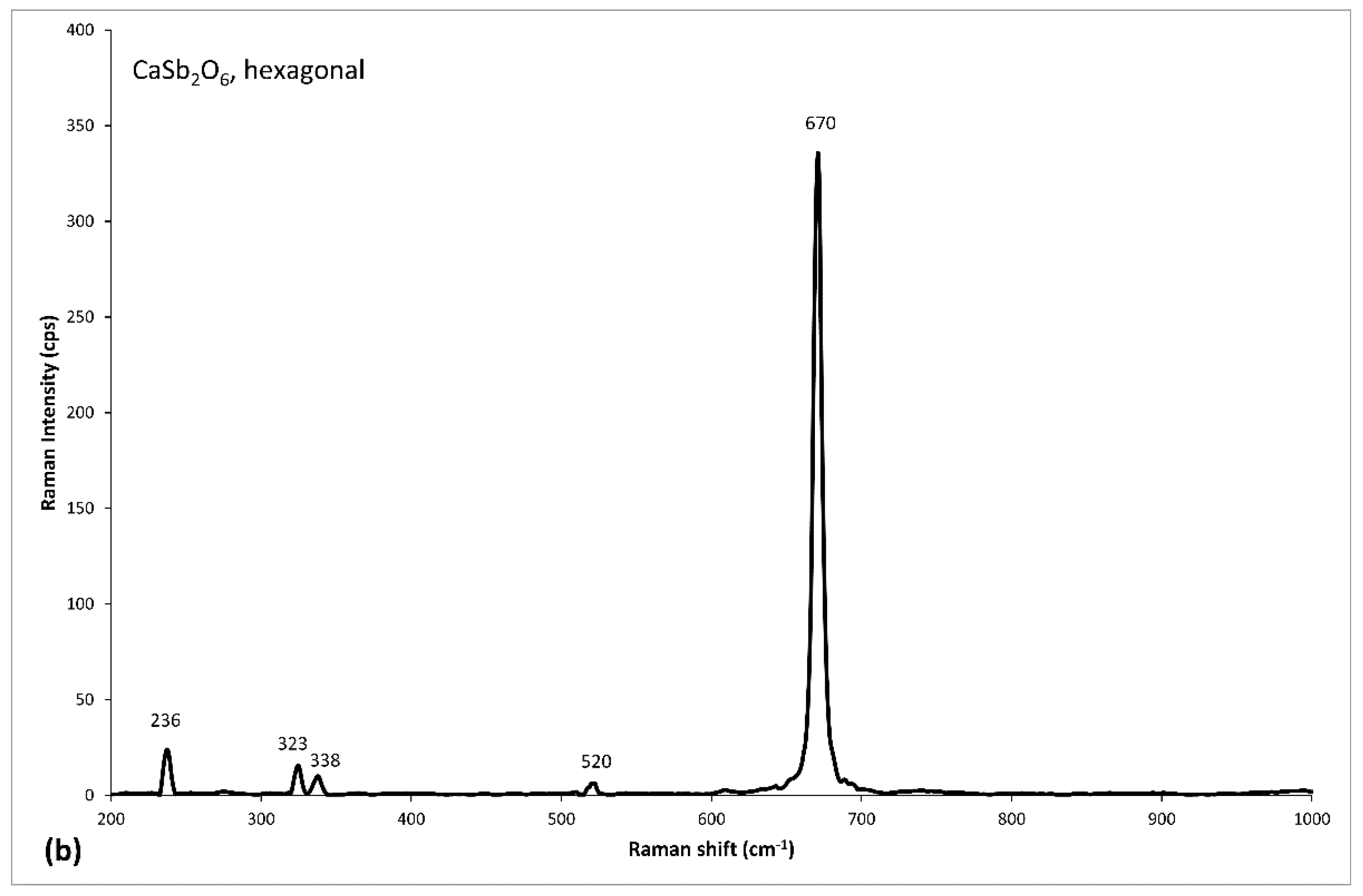





| Tessera | Color Macro-Group | Color | Colorimetric Coordinates | Diaphaneity | Flux | Glassy Matrix | Opacifier | Ionic Colorant/Decoloriser | ||
|---|---|---|---|---|---|---|---|---|---|---|
| L* | a* | b* | ||||||||
| PN AU1 | colorless | gold | n.m. | transparent | N | Mn-glass | Mn | |||
| PN AU2 | colorless | gold | n.m. | transparent | N | Mn-glass | Mn | |||
| TN AU1 | colorless | gold | n.m. | transparent | N | Mn-glass | ||||
| PN AG1 | colorless | colorless | n.m. | transparent | N | Mn-glass | Mn | |||
| TN TR INC 1 | colorless | colorless | n.m. | transparent | N | Sb-glass | Sb | |||
| PN TU1 | turquoise | turquoise | 53 | −21 | 1 | opaque | N | Sb/Mn glass | Ca-antimonate | Cu |
| PN TU2 | turquoise | turquoise | 53 | −20 | 11 | translucent with opaque bands | N | Sb/Mn glass | Ca-antimonate | Cu |
| PN TU3 | turquoise | turquoise | 51 | −13 | −30 | opaque | N | Sb-glass | Ca-antimonate | Cu |
| TN TU1 | turquoise | turquoise | 48 | −19 | −18 | opaque | N | Mn-glass | Ca-antimonate | Cu |
| TN TU2 | turquoise | turquoise | 52 | −16 | −11 | opaque | N | Sb/Mn glass | Ca-antimonate | Cu |
| TN TU3 | turquoise | turquoise | 60 | −24 | −8 | opaque | N | Sb/Mn glass | Ca-antimonate | Cu |
| PN BS1 | turquoise | dark blue | 25 | −1 | −17 | translucent | N | Sb/Mn glass | Cu | |
| TN BS1 | turquoise | dark blue | 27 | −1 | −21 | translucent | N | Sb/Mn glass | Cu | |
| PN CE1 | turquoise | pale blue | 69 | −18 | 5 | opaque | N | Mn-glass | Ca-antimonate | Cu |
| TN CE1 | turquoise | pale blue | 70 | −22 | 1 | opaque | N | Sb/Mn glass | Ca-antimonate | Cu |
| PN AQ1 | turquoise | aquamarine | 52 | −9 | −17 | semi-opaque | N | Sb/Mn glass | Quartz + bubbles + Ca-antimonate | Cu |
| TN AQ1 | turquoise | aquamarine | 49 | −5 | −18 | opaque | N | Mn-glass | Ca-antimonate | Cu |
| TN AQ2 | turquoise | aquamarine | 54 | −13 | −14 | opaque | N | Mn-glass | Ca-antimonate | Cu |
| PN VCH1 | turquoise | pale green | 65 | −13 | 0 | opaque | N | Mn-glass | Ca-antimonate | Cu |
| TN VCH1 | turquoise | pale green | 63 | −25 | 4 | opaque | N | Sb/Mn glass | Ca-antimonate | Cu |
| PN BO1 | blue | blue | 27 | 16 | −62 | opaque | N | Mn-glass | Ca-antimonate | Co |
| TN BO1 | blue | blue | 27 | 13 | −45 | opaque | N | Mn-glass | Ca-antimonate | Co |
| TN B1 | blue | blue | 38 | 21 | −71 | translucent | N | Mn-glass | Co | |
| PN AZ1 | blue | azure | 42 | 4 | −31 | opaque | N | Mn-glass | Ca-antimonate | Co |
| PN AZ2 | blue | azure | 48 | 0 | −26 | translucent with opaque bands | N | Mn-glass | Ca-antimonate | Co |
| TN AZ1 | blue | azure | 65 | −3 | −13 | opaque | N | Mn-glass | Ca-antimonate | Co |
| TN AZ2 | blue | azure | 53 | −1 | −42 | opaque | N | Mn-glass | Ca-antimonate | Co |
| PN BIOP1 | white | white | 83 | 2 | −6 | opaque | N | Mn-glass | Ca-antimonates | |
| TN BIOP1 | white | white | 87 | −1 | −2 | opaque | N | Mn-glass | Ca-antimonates | |
| PN GR1 | white | grey | 46 | 2 | −4 | opaque | N | Sb/Mn glass | Ca-antimonates | Co |
| TN GR1 | white | grey | 62 | 1 | −3 | opaque | N | Sb/Mn glass | Ca-antimonates | Co |
| TN GR2 | white | grey | 66 | −6 | −2 | opaque | N | Mn-glass | Ca-antimonates | Co |
| PN GSO1 | yellow | yellow | 75 | 1 | 63 | opaque | N | Sb/Mn glass | Pb-antimonate | |
| PN GSO2 | yellow | yellow | 71 | 7 | 60 | translucent with opaque bands | N | Mn-glass | Pb-antimonate | |
| TN GSO1 | yellow | yellow | 81 | −7 | 76 | opaque | N | Sb/Mn glass | Pb-antimonate | |
| TN GSO2 | yellow | yellow | 79 | −6 | 76 | opaque | N | Sb/Mn glass | Pb-antimonate | |
| PN VG1 | yellow | yellow green | 69 | −12 | 43 | opaque | N | Sb/Mn glass | Pb-antimonate | |
| TN VG1 | yellow | yellow green | 56 | −2 | 39 | semi-opaque | N | Sb/Mn glass | Pb-antimonate | |
| TN VG2 | yellow | yellow green | 65 | −11 | 38 | translucent | N | Mn-glass | ||
| PN NC1 | yellow | light amber | 65 | 2 | 31 | semi-opaque | N | Mn-glass | Pb-antimonate | |
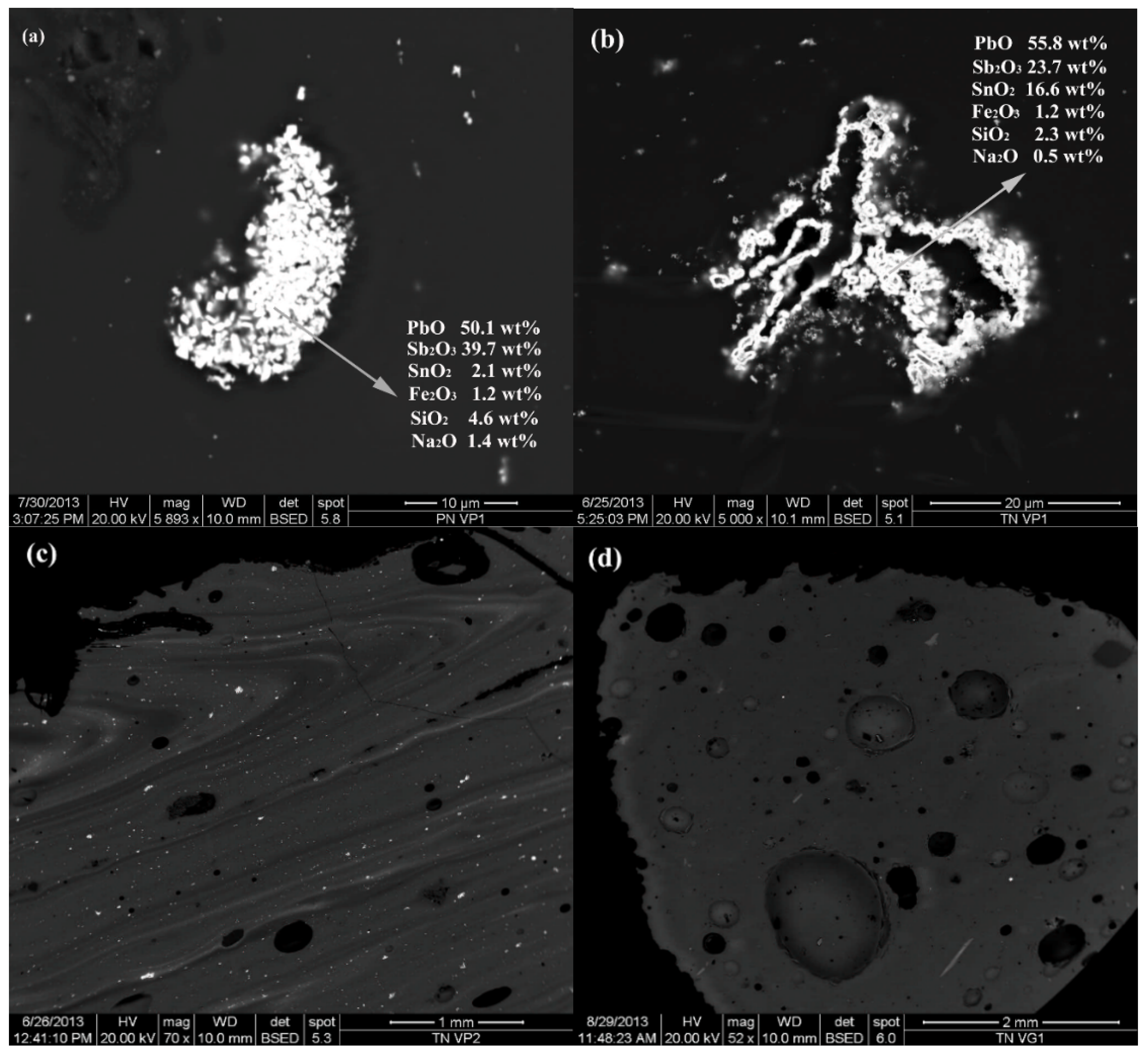
| PN VP1 | green | green | 54 | −28 | 19 | opaque | N | Sb/Mn glass | Pb-antimonate | Cu |
| TN VP1 | green | green | 74 | −27 | 29 | opaque | N | Sb/Mn glass | Pb-antimonate | Cu |
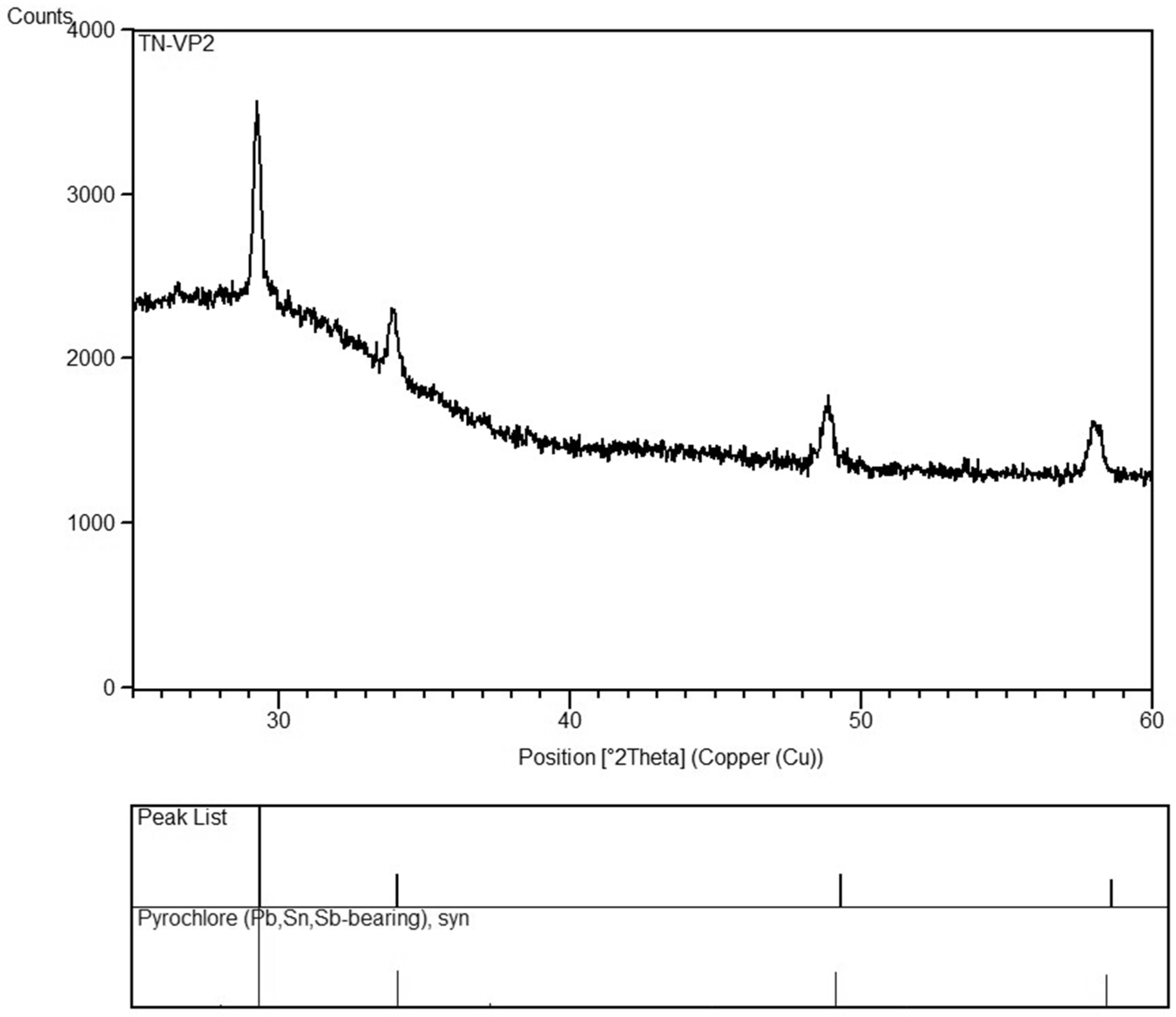
| TN VP2 | green | green | 66 | −21 | 41 | opaque | N | Sb/Mn glass | Pb-antimonate | Cu |
| PN VS1 | green | dark green | 41 | −29 | 1 | opaque | N | Sb/Mn glass | Pb-antimonate | Cu |
| TN VS1 | green | dark green | 49 | −22 | 5 | translucent | N | Sb/Mn glass | Cu | |
| TN VS2 | green | dark green | 50 | −27 | 8 | opaque | N | Sb/Mn glass | Pb-antimonate | Cu |
| PN VTR1 | green | dark green | 29 | −25 | 8 | translucent | N | Sb/Mn glass | Fe | |
| PN VTR2 | green | dark green | 30 | −25 | 4 | translucent | N | Sb/Mn glass | Fe | |
| TN VOL2 | green | olive green | 62 | −10 | 35 | translucent | N | n.a. | Cu | |
| PN AV1 | red | orange | 63 | 33 | 66 | opaque | N | n.a. | cuprite | |
| PN AV2 | red | orange/red | 58 | 34 | 59 | opaque | N | n.a. | cuprite | |
| TN AV1 | red | orange/red | 66 | 38 | 58 | opaque | N | n.a. | cuprite | |
| PN M1 | red | brown | 31 | 25 | 10 | opaque | N | n.a. | metallic copper | |
| PN M2 | red | brown | 29 | 35 | 6 | opaque | A? | n.a. | metallic copper | |
| TN M1 | red | brown | 22 | 26 | 21 | opaque | N | n.a. | metallic copper | |
| TN M2 | red | brown | 33 | 32 | 15 | opaque | N | n.a. | metallic copper | |
| TN M3 | red | brown | 39 | 41 | 29 | opaque | N | n.a. | metallic copper | |
| PN M3 | red | green | 72 | −12 | 41 | translucent with opaque bands | N | n.a. | metallic copper (red bands) | Cu (green glass) |
| red | 33 | 42 | 42 | n.a. | ||||||
| TN VOL1 | red | green | 61 | −5 | 43 | translucent with opaque bands | N | n.a. | metallic copper (red bands) | Cu (green glass) |
| red | 40 | 28 | 18 | n.a. | ||||||
| TN NS1 | red | dark amber | 53 | 7 | 20 | semi-opaque | N | Sb/Mn glass | metallic copper | |
| Sample | Color Macro-Group | Color | Diaphaneity | SiO2 | Na2O | CaO | Al2O3 | MnO | Fe2O3 | MgO | K2O | TiO2 | P2O5 | SO3 | Cl | CoO | CuO | ZnO | SnO2 | Sb2O3 | PbO | TOT |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PN AU1 | colorless | gold | transparent | 69.35 | 16.22 | 7.97 | 2.26 | 1.90 | 0.37 | 0.64 | 0.55 | 0.05 | 0.09 | 0.12 | 1.24 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.76 |
| 0.28 | 0.30 | 0.09 | 0.11 | 0.28 | 0.05 | 0.01 | 0.06 | 0.01 | 0.03 | 0.04 | 0.05 | |||||||||||
| PN AU1 (cart) | colorless | gold | transparent | 69.49 | 16.20 | 8.00 | 2.24 | 1.73 | 0.41 | 0.66 | 0.50 | 0.06 | 0.09 | 0.11 | 1.30 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.79 |
| 0.28 | 0.17 | 0.01 | 0.13 | 0.11 | 0.02 | 0.02 | 0.03 | 0.00 | 0.02 | 0.01 | 0.00 | |||||||||||
| PN AU2 | colorless | gold | transparent | 68.91 | 16.26 | 7.99 | 2.30 | 1.80 | 0.36 | 0.67 | 0.55 | 0.08 | 0.09 | 0.13 | 1.26 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.40 |
| 0.19 | 0.24 | 0.09 | 0.09 | 0.12 | 0.02 | 0.01 | 0.04 | 0.02 | 0.01 | 0.01 | 0.02 | |||||||||||
| PN AU2 (cart) | colorless | gold | transparent | 69.45 | 16.29 | 7.99 | 2.31 | 1.87 | 0.37 | 0.65 | 0.53 | 0.06 | 0.09 | 0.12 | 1.21 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.96 |
| 0.29 | 0.18 | 0.09 | 0.06 | 0.03 | 0.06 | 0.05 | 0.03 | 0.02 | 0.02 | 0.01 | 0.02 | |||||||||||
| TN AU1 | colorless | gold | transparent | 72.10 | 15.09 | 8.01 | 2.43 | 0.38 | 0.31 | 0.53 | 0.42 | 0.07 | 0.10 | 0.16 | 1.11 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.72 |
| 0.41 | 0.29 | 0.11 | 0.02 | 0.14 | 0.03 | 0.00 | 0.05 | 0.01 | 0.04 | 0.02 | 0.04 | |||||||||||
| TN AU1 (cart) | colorless | gold | transparent | 71.99 | 15.14 | 7.84 | 2.39 | 0.35 | 0.33 | 0.53 | 0.42 | 0.07 | 0.08 | 0.15 | 1.11 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.38 |
| 0.27 | 0.22 | 0.04 | 0.15 | 0.04 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.03 | |||||||||||
| PN AG1 | colorless | colorless | transparent | 69.26 | 16.29 | 8.01 | 2.25 | 1.77 | 0.38 | 0.65 | 0.54 | 0.06 | 0.09 | 0.13 | 1.29 | <0.03 | <0.03 | <0.04 | <0.04 | <0.04 | <0.08 | 100.72 |
| 0.38 | 0.16 | 0.08 | 0.09 | 0.07 | 0.02 | 0.02 | 0.05 | 0.01 | 0.02 | 0.03 | 0.04 | |||||||||||
| TN TR INC 1 | colorless | colorless | transparent | 68.04 | 18.87 | 7.20 | 2.11 | 0.01 | 0.56 | 0.68 | 0.57 | 0.09 | 0.03 | 0.31 | 1.36 | <0.03 | <0.03 | <0.04 | <0.04 | 0.66 | <0.08 | 100.50 |
| 0.37 | 0.16 | 0.08 | 0.13 | 0.01 | 0.04 | 0.02 | 0.07 | 0.01 | 0.03 | 0.02 | 0.03 | 0.04 | ||||||||||
| PN TU1 | turquoise | turquoise | opaque | 65.27 | 15.72 | 6.75 | 2.39 | 0.63 | 0.69 | 0.93 | 0.84 | 0.09 | 0.30 | 0.38 | 0.81 | <0.03 | 2.89 | <0.04 | 0.29 | 2.52 | 0.40 | 100.90 |
| 0.63 | 0.24 | 0.05 | 0.10 | 0.04 | 0.04 | 0.05 | 0.07 | 0.01 | 0.02 | 0.02 | 0.03 | 0.80 | 0.08 | 0.42 | 0.05 | |||||||
| PN TU2 (tr) | turquoise | turquoise | translucent | 62.86 | 17.83 | 5.54 | 2.52 | 0.22 | 0.88 | 0.87 | 0.69 | 0.13 | 0.31 | 0.37 | 1.15 | <0.03 | 4.49 | 0.11 | 0.14 | 1.45 | 1.21 | 100.77 |
| 1.87 | 0.27 | 0.16 | 0.26 | 0.05 | 0.15 | 0.08 | 0.09 | 0.02 | 0.04 | 0.04 | 0.05 | 0.55 | 0.05 | 0.10 | 0.37 | 0.56 | ||||||
| PN TU2 (op) | turquoise | turquoise | opaque | 64.86 | 17.71 | 5.32 | 2.21 | 0.14 | 0.74 | 0.67 | 0.56 | 0.11 | 0.11 | 0.41 | 1.10 | <0.03 | 3.08 | 0.06 | 0.09 | 2.56 | 0.89 | 100.63 |
| 1.35 | 0.35 | 0.05 | 0.02 | 0.05 | 0.01 | 0.01 | 0.05 | 0.03 | 0.02 | 0.04 | 0.06 | 0.64 | 0.01 | 0.05 | 0.65 | 0.05 | ||||||
| PN TU3 | turquoise | turquoise | opaque | 68.09 | 17.53 | 4.52 | 2.02 | <0.05 | 0.52 | 0.39 | 0.50 | 0.07 | 0.01 | 0.21 | 1.38 | <0.03 | 2.83 | 0.10 | 0.21 | 1.89 | 0.31 | 100.60 |
| 0.24 | 0.14 | 0.03 | 0.10 | 0.04 | 0.02 | 0.03 | 0.03 | 0.02 | 0.02 | 0.01 | 0.07 | 0.03 | 0.01 | 0.02 | 0.05 | |||||||
| TN TU1 | turquoise | turquoise | opaque | 65.13 | 16.11 | 7.22 | 2.50 | 0.73 | 0.67 | 0.70 | 0.72 | 0.09 | 0.20 | 0.39 | 0.89 | <0.03 | 2.32 | <0.04 | 0.22 | 1.64 | 0.40 | 99.92 |
| 0.45 | 0.21 | 0.10 | 0.10 | 0.03 | 0.02 | 0.03 | 0.07 | 0.01 | 0.02 | 0.02 | 0.02 | 0.05 | 0.01 | 0.28 | 0.04 | |||||||
| TN TU2 | turquoise | turquoise | opaque | 64.49 | 15.93 | 5.69 | 2.43 | 0.61 | 0.70 | 0.74 | 0.63 | 0.13 | 0.06 | 0.34 | 1.09 | <0.03 | 4.49 | <0.04 | 0.24 | 3.06 | 0.72 | 101.36 |
| 0.94 | 0.24 | 0.17 | 0.19 | 0.03 | 0.03 | 0.03 | 0.10 | 0.03 | 0.04 | 0.02 | 0.04 | 0.09 | 0.03 | 1.76 | 0.05 | |||||||
| TN TU3 | turquoise | turquoise | opaque | 64.95 | 16.03 | 7.25 | 2.44 | 0.74 | 0.66 | 0.71 | 0.70 | 0.09 | 0.18 | 0.39 | 0.89 | <0.03 | 2.34 | <0.04 | 0.21 | 1.78 | 0.41 | 99.78 |
| 0.58 | 0.28 | 0.08 | 0.15 | 0.05 | 0.04 | 0.04 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.06 | 0.03 | 0.50 | 0.05 | |||||||
| TN BS1 | turquoise | dark blue | translucent | 64.50 | 17.55 | 5.95 | 2.36 | 0.53 | 0.81 | 0.70 | 0.68 | 0.14 | 0.22 | 0.28 | 1.34 | <0.03 | 5.07 | <0.04 | 0.13 | 0.56 | 0.26 | 101.07 |
| 0.55 | 0.21 | 0.10 | 0.04 | 0.05 | 0.03 | 0.02 | 0.05 | 0.00 | 0.02 | 0.01 | 0.04 | 0.89 | 0.07 | 0.06 | 0.06 | |||||||
| PN BS1 | turquoise | dark blue | translucent | 66.89 | 19.32 | 5.85 | 2.16 | 0.57 | 0.63 | 0.65 | 0.61 | 0.11 | 0.11 | 0.31 | 1.39 | <0.03 | 1.81 | <0.04 | 0.14 | 0.62 | 0.09 | 101.28 |
| 0.59 | 0.33 | 0.18 | 0.10 | 0.05 | 0.05 | 0.02 | 0.04 | 0.01 | 0.02 | 0.05 | 0.06 | 0.66 | 0.03 | 0.02 | 0.04 | |||||||
| PN CE1 | turquoise | pale blue | opaque | 65.36 | 15.58 | 6.37 | 2.75 | 0.43 | 0.98 | 0.74 | 0.68 | 0.11 | 0.18 | 0.41 | 0.73 | <0.03 | 2.21 | <0.04 | 0.10 | 3.74 | 0.13 | 100.50 |
| 0.38 | 0.21 | 0.15 | 0.12 | 0.04 | 0.05 | 0.04 | 0.03 | 0.02 | 0.03 | 0.08 | 0.04 | 0.34 | 0.06 | 0.35 | 0.01 | |||||||
| TN CE1 | turquoise | pale blue | opaque | 63.88 | 16.76 | 6.62 | 2.66 | 0.23 | 0.98 | 0.79 | 0.67 | 0.11 | 0.18 | 0.47 | 0.87 | <0.03 | 2.70 | <0.04 | 0.14 | 3.02 | 0.09 | 100.17 |
| 0.40 | 0.35 | 0.11 | 0.11 | 0.03 | 0.06 | 0.02 | 0.04 | 0.01 | 0.02 | 0.02 | 0.03 | 0.04 | 0.04 | 0.21 | 0.05 | |||||||
| PN AQ1 | turquoise | aquamarine | semi-opaque | 66.04 | 17.70 | 6.51 | 2.42 | 0.40 | 0.74 | 0.74 | 0.68 | 0.12 | 0.17 | 0.29 | 1.29 | <0.03 | 2.30 | <0.04 | 0.11 | 0.79 | 0.20 | 100.50 |
| 0.62 | 0.23 | 0.08 | 0.11 | 0.03 | 0.06 | 0.02 | 0.05 | 0.03 | 0.02 | 0.03 | 0.03 | 0.05 | 0.03 | 0.12 | 0.07 | |||||||
| TN AQ1 | turquoise | aquamarine | opaque | 66.37 | 16.62 | 7.00 | 2.48 | 0.89 | 0.52 | 0.64 | 0.69 | 0.09 | 0.06 | 0.28 | 1.23 | <0.03 | 2.42 | <0.04 | 0.19 | 0.83 | 0.33 | 100.67 |
| 0.49 | 0.30 | 0.09 | 0.17 | 0.03 | 0.03 | 0.04 | 0.10 | 0.02 | 0.04 | 0.02 | 0.06 | 0.08 | 0.03 | 0.16 | 0.08 | |||||||
| TN AQ2 | turquoise | aquamarine | opaque | 66.28 | 16.87 | 7.06 | 2.41 | 0.85 | 0.58 | 0.69 | 0.68 | 0.10 | 0.17 | 0.30 | 1.07 | <0.03 | 2.17 | <0.04 | 0.16 | 0.69 | 0.28 | 100.36 |
| 0.71 | 0.16 | 0.16 | 0.09 | 0.05 | 0.06 | 0.03 | 0.05 | 0.02 | 0.02 | 0.01 | 0.05 | 0.26 | 0.05 | 0.13 | 0.07 | |||||||
| PN VCH1 | turquoise | pale green | opaque | 68.21 | 18.29 | 6.67 | 2.26 | 0.58 | 0.44 | 0.68 | 0.54 | 0.08 | 0.10 | 0.26 | 1.24 | <0.03 | 0.68 | <0.03 | <0.04 | 0.88 | <0.08 | 100.91 |
| 0.34 | 0.25 | 0.05 | 0.07 | 0.04 | 0.03 | 0.03 | 0.04 | 0.01 | 0.02 | 0.02 | 0.03 | 0.03 | 0.06 | |||||||||
| TN VCH1 | turquoise | pale green | opaque | 64.06 | 17.23 | 6.53 | 2.51 | 0.20 | 0.91 | 0.77 | 0.62 | 0.12 | 0.14 | 0.40 | 1.01 | <0.03 | 2.84 | <0.04 | 0.16 | 3.22 | 0.12 | 100.85 |
| 1.02 | 0.34 | 0.15 | 0.08 | 0.01 | 0.01 | 0.01 | 0.04 | 0.01 | 0.04 | 0.01 | 0.04 | 0.08 | 0.02 | 0.62 | 0.02 | |||||||
| PN BO1 | blue | blue | opaque | 66.11 | 15.06 | 6.70 | 2.49 | 0.63 | 1.12 | 0.80 | 0.74 | 0.09 | 0.24 | 0.42 | 0.68 | 0.17 | 0.26 | <0.04 | <0.04 | 4.40 | 0.11 | 100.03 |
| 0.35 | 0.40 | 0.06 | 0.15 | 0.04 | 0.08 | 0.02 | 0.03 | 0.02 | 0.02 | 0.02 | 0.01 | 0.03 | 0.02 | 0.06 | 0.03 | |||||||
| TN BO1 | blue | blue | opaque | 68.95 | 16.40 | 7.06 | 2.32 | 0.78 | 0.84 | 0.63 | 0.59 | 0.07 | 0.19 | 0.24 | 1.15 | 0.10 | 0.17 | <0.04 | <0.04 | 0.76 | <0.08 | 100.25 |
| 1.39 | 0.36 | 0.21 | 0.10 | 0.15 | 0.39 | 0.06 | 0.04 | 0.01 | 0.03 | 0.05 | 0.14 | 0.05 | 0.02 | 0.87 | ||||||||
| TN B1 | blue | blue | translucent | 69.42 | 18.18 | 6.61 | 1.93 | 1.54 | 0.48 | 0.53 | 0.43 | 0.05 | 0.17 | 0.17 | 1.63 | 0.06 | 0.06 | <0.04 | <0.04 | <0.04 | <0.08 | 101.24 |
| 0.35 | 0.17 | 0.04 | 0.08 | 0.08 | 0.06 | 0.03 | 0.03 | 0.01 | 0.02 | 0.04 | 0.05 | 0.01 | 0.02 | |||||||||
| PN AZ1 | blue | azure | opaque | 61.01 | 15.96 | 7.68 | 2.33 | 1.16 | 1.89 | 0.64 | 0.58 | 0.03 | 0.21 | 0.57 | 0.69 | 0.19 | 0.57 | <0.04 | <0.04 | 6.39 | <0.08 | 99.90 |
| 0.59 | 0.27 | 0.15 | 0.11 | 0.04 | 0.07 | 0.03 | 0.03 | 0.02 | 0.02 | 0.05 | 0.02 | 0.02 | 0.02 | 0.57 | ||||||||
| PN AZ2 | blue | azure | translucent with opaque bands | 68.47 | 15.13 | 8.69 | 2.47 | 0.87 | 0.68 | 0.63 | 0.53 | 0.06 | 0.22 | 0.32 | 0.95 | 0.03 | 0.04 | <0.04 | <0.04 | 1.37 | <0.08 | 100.48 |
| 0.22 | 0.23 | 0.15 | 0.12 | 0.15 | 0.08 | 0.04 | 0.04 | 0.02 | 0.03 | 0.05 | 0.04 | 0.01 | 0.02 | 0.30 | ||||||||
| TN AZ1 | blue | azure | opaque | 67.52 | 16.12 | 7.24 | 2.49 | 0.81 | 0.63 | 0.67 | 0.68 | 0.07 | 0.17 | 0.40 | 0.85 | <0.03 | 0.15 | <0.04 | <0.04 | 2.49 | 0.19 | 100.48 |
| 0.49 | 0.20 | 0.07 | 0.03 | 0.05 | 0.03 | 0.02 | 0.09 | 0.01 | 0.02 | 0.03 | 0.03 | 0.01 | 0.18 | 0.04 | ||||||||
| TN AZ2 | blue | azure | opaque | 67.47 | 16.24 | 6.76 | 2.37 | 0.57 | 0.82 | 0.69 | 0.60 | 0.10 | 0.19 | 0.42 | 0.94 | 0.03 | 0.12 | <0.04 | <0.04 | 2.88 | 0.29 | 100.50 |
| 0.34 | 0.28 | 0.10 | 0.08 | 0.02 | 0.04 | 0.03 | 0.03 | 0.02 | 0.02 | 0.10 | 0.06 | 0.02 | 0.13 | 0.07 | ||||||||
| PN BIOP1 | white | white | opaque | 67.82 | 14.89 | 5.85 | 1.99 | 0.66 | 0.39 | 0.46 | 0.54 | 0.04 | 0.18 | 0.36 | 0.63 | <0.03 | <0.3 | <0.04 | <0.04 | 4.67 | 1.29 | 99.78 |
| 0.54 | 0.17 | 0.05 | 0.09 | 0.03 | 0.02 | 0.02 | 0.09 | 0.01 | 0.02 | 0.03 | 0.02 | 0.18 | 0.13 | |||||||||
| TN BIOP1 | white | white | opaque | 67.39 | 15.64 | 7.28 | 2.63 | 0.80 | 0.64 | 0.66 | 0.74 | 0.08 | 0.08 | 0.39 | 0.83 | <0.03 | 0.07 | <0.04 | <0.04 | 2.88 | 0.23 | 100.32 |
| 1.11 | 0.45 | 0.11 | 0.10 | 0.03 | 0.08 | 0.03 | 0.07 | 0.02 | 0.03 | 0.18 | 0.15 | 0.04 | 1.24 | 0.18 | ||||||||
| PN GR1 | white | grey | opaque | 67.48 | 17.91 | 6.73 | 2.46 | 0.59 | 0.74 | 0.73 | 0.69 | 0.11 | 0.20 | 0.29 | 1.14 | <0.03 | 0.04 | <0.04 | <0.04 | 1.29 | <0.08 | 100.39 |
| 0.37 | 0.13 | 0.04 | 0.13 | 0.02 | 0.02 | 0.02 | 0.09 | 0.02 | 0.02 | 0.03 | 0.01 | 0.01 | 0.07 | |||||||||
| TN GR1 | white | grey | opaque | 67.80 | 17.14 | 6.57 | 2.32 | 0.31 | 0.70 | 0.76 | 0.71 | 0.09 | 0.06 | 0.34 | 0.95 | <0.03 | 0.03 | <0.04 | <0.04 | 2.60 | <0.08 | 100.38 |
| 0.35 | 0.17 | 0.08 | 0.08 | 0.04 | 0.04 | 0.05 | 0.05 | 0.02 | 0.02 | 0.02 | 0.03 | 0.01 | 0.28 | |||||||||
| TN GR2 | white | grey | opaque | 69.12 | 16.35 | 6.86 | 2.39 | 0.55 | 0.69 | 0.73 | 0.58 | 0.10 | 0.11 | 0.25 | 1.05 | <0.03 | 0.06 | <0.04 | <0.04 | 1.49 | 0.19 | 100.50 |
| 0.14 | 0.31 | 0.06 | 0.11 | 0.03 | 0.05 | 0.03 | 0.07 | 0.02 | 0.03 | 0.02 | 0.01 | 0.02 | 0.11 | 0.02 | ||||||||
| PN GSO1 | yellow | yellow | opaque | 66.22 | 17.28 | 4.99 | 2.10 | 0.67 | 0.88 | 0.66 | 0.62 | 0.14 | 0.08 | 0.28 | 0.99 | <0.03 | <0.03 | <0.03 | <0.04 | 0.85 | 3.76 | 99.51 |
| 0.34 | 0.22 | 0.07 | 0.13 | 0.03 | 0.09 | 0.03 | 0.05 | 0.02 | 0.03 | 0.02 | 0.04 | 0.04 | 0.48 | |||||||||
| PN GSO2 (tr) | yellow | yellow | translucent | 65.67 | 15.88 | 7.04 | 2.16 | 0.54 | 0.58 | 0.48 | 0.54 | 0.06 | 0.11 | 0.22 | 1.00 | <0.03 | <0.03 | <0.04 | 0.10 | 0.52 | 5.11 | 99.98 |
| 0.55 | 0.29 | 0.18 | 0.08 | 0.07 | 0.19 | 0.02 | 0.04 | 0.02 | 0.01 | 0.02 | 0.06 | 0.06 | 0.30 | 0.14 | ||||||||
| PN GSO2 (op) | yellow | yellow | opaque | 61.23 | 13.76 | 6.76 | 2.23 | 0.44 | 0.95 | 0.49 | 0.49 | 0.07 | 0.06 | 0.17 | 0.90 | <0.03 | <0.03 | <0.04 | 0.09 | 0.25 | 11.52 | 99.39 |
| 2.97 | 0.53 | 0.17 | 0.07 | 0.06 | 0.82 | 0.03 | 0.11 | 0.01 | 0.02 | 0.05 | 0.10 | 0.02 | 0.32 | 1.37 | ||||||||
| TN GSO1 | yellow | yellow | opaque | 68.88 | 17.55 | 6.67 | 2.13 | 1.09 | 0.26 | 0.57 | 0.53 | 0.08 | 0.04 | 0.29 | 1.20 | <0.03 | <0.03 | <0.04 | <0.04 | 0.45 | 0.51 | 100.26 |
| 0.44 | 0.28 | 0.10 | 0.11 | 0.08 | 0.22 | 0.02 | 0.04 | 0.02 | 0.03 | 0.03 | 0.04 | 0.04 | 0.10 | |||||||||
| TN GSO2 | yellow | yellow | opaque | 66.86 | 16.56 | 6.45 | 2.28 | 0.78 | 0.45 | 0.53 | 0.57 | 0.11 | 0.05 | 0.26 | 1.13 | <0.03 | <0.03 | <0.04 | 0.09 | 0.49 | 3.63 | 100.23 |
| 0.46 | 0.25 | 0.08 | 0.07 | 0.05 | 0.17 | 0.02 | 0.04 | 0.10 | 0.04 | 0.02 | 0.03 | 0.05 | 0.15 | 0.21 | ||||||||
| PN VG1 | yellow | yellow-green | opaque | 67.12 | 16.04 | 6.34 | 2.30 | 0.72 | 0.93 | 0.76 | 0.78 | 0.13 | 0.21 | 0.19 | 0.95 | <0.03 | 0.04 | <0.04 | 0.11 | 0.34 | 3.59 | 100.54 |
| 0.37 | 0.16 | 0.11 | 0.11 | 0.03 | 0.06 | 0.03 | 0.05 | 0.01 | 0.02 | 0.01 | 0.03 | 0.02 | 0.08 | 0.16 | 0.38 | |||||||
| TN VG1 | yellow | yellow-green | semi-opaque | 66.25 | 16.38 | 6.94 | 2.26 | 0.43 | 1.36 | 0.84 | 0.72 | 0.09 | 0.15 | 0.29 | 1.22 | <0.03 | 0.59 | <0.04 | 0.09 | 0.64 | 2.27 | 100.52 |
| 0.16 | 0.19 | 0.10 | 0.10 | 0.05 | 0.03 | 0.03 | 0.06 | 0.01 | 0.02 | 0.03 | 0.02 | 0.04 | 0.02 | 0.06 | 0.10 | |||||||
| TN VG2 | yellow | yellow- green | translucent | 67.10 | 15.89 | 7.52 | 2.82 | 0.97 | 1.59 | 0.77 | 0.86 | 0.10 | 0.12 | 0.28 | 1.05 | <0.03 | 0.89 | <0.04 | 0.04 | 0.44 | 0.50 | 100.94 |
| 0.29 | 0.29 | 0.13 | 0.15 | 0.04 | 0.08 | 0.03 | 0.04 | 0.01 | 0.03 | 0.02 | 0.04 | 0.09 | 0.02 | 0.04 | 0.07 | |||||||
| PN NC1 | yellow | light amber | semi-opaque | 68.67 | 16.76 | 6.88 | 2.41 | 0.35 | 0.51 | 0.59 | 0.56 | 0.09 | 0.09 | 0.19 | 1.21 | <0.03 | <0.03 | <0.04 | <0.04 | 0.26 | 1.82 | 100.38 |
| 0.41 | 0.20 | 0.14 | 0.09 | 0.03 | 0.05 | 0.02 | 0.05 | 0.02 | 0.03 | 0.04 | 0.05 | 0.03 | 0.26 | |||||||||
| PN VP1 | green | green | opaque | 66.11 | 17.30 | 6.46 | 2.48 | 0.50 | 0.60 | 0.67 | 0.69 | 0.11 | 0.11 | 0.24 | 1.21 | <0.03 | 1.06 | <0.04 | 0.06 | 0.38 | 2.50 | 100.48 |
| 0.54 | 0.26 | 0.07 | 0.09 | 0.02 | 0.03 | 0.03 | 0.06 | 0.02 | 0.02 | 0.03 | 0.04 | 0.04 | 0.03 | 0.06 | 0.41 | |||||||
| TN VP1 | green | green | opaque | 64.83 | 16.47 | 6.63 | 2.38 | 0.62 | 0.74 | 0.67 | 0.65 | 0.10 | 0.12 | 0.23 | 1.18 | <0.03 | 1.13 | <0.04 | 0.13 | 0.56 | 3.58 | 100.00 |
| 0.46 | 0.22 | 0.10 | 0.07 | 0.04 | 0.05 | 0.02 | 0.04 | 0.01 | 0.01 | 0.03 | 0.06 | 0.05 | 0.03 | 0.04 | 0.20 | |||||||
| TN VP2 | green | green | opaque | 64.91 | 16.35 | 6.54 | 2.30 | 0.61 | 0.69 | 0.62 | 0.65 | 0.11 | 0.12 | 0.24 | 1.18 | <0.03 | 0.97 | <0.4 | 0.16 | 0.66 | 3.99 | 100.09 |
| 0.64 | 0.26 | 0.06 | 0.13 | 0.06 | 0.04 | 0.02 | 0.06 | 0.02 | 0.02 | 0.03 | 0.05 | 0.08 | 0.10 | 0.19 | 0.49 | |||||||
| PN VS1 | green | dark green | opaque | 66.04 | 17.54 | 6.36 | 2.43 | 0.44 | 0.64 | 0.72 | 0.73 | 0.12 | 0.13 | 0.24 | 1.23 | <0.03 | 2.52 | <0.04 | 0.10 | 0.41 | 1.26 | 100.92 |
| 0.24 | 0.30 | 0.06 | 0.09 | 0.03 | 0.04 | 0.02 | 0.05 | 0.01 | 0.02 | 0.04 | 0.04 | 0.13 | 0.03 | 0.05 | 0.20 | |||||||
| TN VS1 | green | dark green | translucent | 66.32 | 17.51 | 7.10 | 2.44 | 0.34 | 0.98 | 0.71 | 0.71 | 0.11 | 0.14 | 0.28 | 1.24 | <0.03 | 1.84 | <0.04 | 0.10 | 0.72 | 0.49 | 101.04 |
| 0.53 | 0.34 | 0.03 | 0.15 | 0.03 | 0.03 | 0.01 | 0.05 | 0.01 | 0.03 | 0.01 | 0.03 | 0.07 | 0.01 | 0.02 | 0.06 | |||||||
| TN VS2 | green | dark green | opaque | 66.42 | 16.67 | 6.97 | 2.42 | 0.76 | 0.76 | 0.66 | 0.62 | 0.10 | 0.13 | 0.23 | 1.21 | <0.03 | 1.70 | <0.04 | 0.11 | 0.50 | 1.07 | 100.35 |
| 0.22 | 0.29 | 0.17 | 0.08 | 0.05 | 0.03 | 0.03 | 0.06 | 0.02 | 0.03 | 0.02 | 0.04 | 0.10 | 0.03 | 0.03 | 0.13 | |||||||
| PN VTR1 | green | dark green | translucent | 62.59 | 16.20 | 6.19 | 2.07 | 0.77 | 1.92 | 0.54 | 0.55 | 0.07 | 0.10 | 0.20 | 1.13 | <0.03 | <0.03 | <0.04 | 0.13 | 0.38 | 7.25 | 100.09 |
| 1.13 | 0.27 | 0.26 | 0.18 | 0.11 | 0.39 | 0.03 | 0.05 | 0.02 | 0.02 | 0.02 | 0.03 | 0.05 | 0.08 | 1.25 | ||||||||
| PN VTR2 | green | dark green | translucent | 63.50 | 16.00 | 6.34 | 2.19 | 0.92 | 2.79 | 0.57 | 0.56 | 0.09 | 0.09 | 0.20 | 1.13 | <0.03 | <0.03 | <0.04 | 0.11 | 0.31 | 5.37 | 100.16 |
| 1.05 | 0.28 | 0.06 | 0.08 | 0.04 | 0.76 | 0.02 | 0.06 | 0.02 | 0.02 | 0.02 | 0.04 | 0.02 | 0.05 | 0.14 | ||||||||
| TN VOL2 | green | olive green | translucent | 59.35 | 12.89 | 6.89 | 2.94 | 1.28 | 2.46 | 0.69 | 0.83 | 0.11 | 0.12 | 0.31 | 0.80 | <0.03 | 2.07 | <0.04 | 0.40 | 0.77 | 8.45 | 100.34 |
| 0.47 | 0.37 | 0.07 | 0.14 | 0.13 | 0.10 | 0.04 | 0.07 | 0.02 | 0.03 | 0.03 | 0.04 | 0.07 | 0.03 | 0.05 | 0.21 | |||||||
| PN AV1 | red | orange | opaque | 41.67 | 10.03 | 6.29 | 2.65 | 0.26 | 1.99 | 1.35 | 0.85 | 0.24 | 0.43 | 0.33 | 0.74 | <0.03 | 8.34 | <0.04 | 1.36 | 0.56 | 23.88 | 100.98 |
| 0.37 | 0.25 | 0.06 | 0.18 | 0.02 | 0.08 | 0.03 | 0.05 | 0.01 | 0.05 | 0.03 | 0.03 | 1.28 | 0.06 | 0.04 | 0.18 | |||||||
| PN AV2 (or) | red | orange | opaque | 43.25 | 9.71 | 6.19 | 2.52 | 0.28 | 1.20 | 0.71 | 0.64 | 0.14 | 0.16 | 0.38 | 0.56 | <0.03 | 8.94 | 0.15 | 1.00 | 1.40 | 23.41 | 100.67 |
| 0.31 | 0.21 | 0.07 | 0.10 | 0.04 | 0.03 | 0.03 | 0.06 | 0.02 | 0.03 | 0.01 | 0.02 | 0.19 | 0.02 | 0.02 | 0.04 | 0.22 | ||||||
| PN AV2 (red) | red | red | opaque | 42.57 | 9.86 | 6.00 | 2.43 | 0.27 | 1.16 | 0.69 | 0.64 | 0.12 | 0.14 | 0.36 | 0.56 | <0.03 | 7.71 | 0.13 | 0.97 | 1.38 | 25.48 | 100.49 |
| 0.87 | 0.26 | 0.25 | 0.12 | 0.03 | 0.07 | 0.05 | 0.03 | 0.04 | 0.03 | 0.06 | 0.03 | 1.15 | 0.01 | 0.02 | 0.09 | 0.93 | ||||||
| TN AV1 (or) | red | orange | opaque | 50.62 | 11.67 | 7.03 | 2.05 | 0.27 | 1.28 | 1.92 | 1.47 | 0.18 | 0.65 | 0.54 | 0.80 | <0.03 | 14.14 | 0.07 | 1.01 | 0.96 | 6.07 | 100.74 |
| 0.73 | 0.32 | 0.06 | 0.09 | 0.02 | 0.05 | 0.04 | 0.10 | 0.02 | 0.03 | 0.04 | 0.01 | 0.48 | 0.03 | 0.03 | 0.03 | 0.20 | ||||||
| TN AV1 (red) | red | red | opaque | 52.13 | 12.94 | 7.26 | 2.11 | 0.28 | 1.36 | 1.94 | 1.45 | 0.17 | 0.62 | 0.49 | 0.85 | <0.03 | 10.94 | 0.06 | 1.01 | 0.91 | 6.33 | 100.86 |
| 1.02 | 0.41 | 0.08 | 0.11 | 0.03 | 0.06 | 0.05 | 0.09 | 0.02 | 0.05 | 0.05 | 0.03 | 0.67 | 0.03 | 0.09 | 0.09 | 0.22 | ||||||
| PN M1 | red | brown | opaque | 61.57 | 15.37 | 7.13 | 2.84 | 0.44 | 1.24 | 0.82 | 0.77 | 0.15 | 0.15 | 0.11 | 0.88 | <0.03 | 1.27 | 0.19 | 0.34 | 0.40 | 6.27 | 99.95 |
| 0.79 | 0.26 | 0.10 | 0.10 | 0.03 | 0.08 | 0.03 | 0.04 | 0.02 | 0.04 | 0.03 | 0.10 | 0.26 | 0.05 | 0.06 | 0.02 | 0.57 | ||||||
| PN M2 | red | brown | opaque | 61.59 | 15.83 | 7.85 | 2.06 | 0.65 | 1.61 | 3.05 | 2.14 | 0.19 | 1.21 | 0.52 | 1.20 | <0.03 | 1.62 | 0.04 | 0.19 | 0.85 | 0.83 | 101.44 |
| 0.63 | 0.30 | 0.12 | 0.08 | 0.05 | 0.06 | 0.10 | 0.08 | 0.02 | 0.08 | 1.07 | 0.05 | 1.47 | 0.02 | 0.09 | 0.18 | 0.09 | ||||||
| TN M1 | red | brown | opaque | 62.84 | 14.51 | 7.01 | 2.73 | 0.84 | 4.10 | 0.81 | 0.85 | 0.10 | 0.18 | 0.28 | 0.98 | <0.03 | 0.74 | <0.04 | 0.24 | 0.64 | 3.61 | 100.47 |
| 0.54 | 0.44 | 0.26 | 0.21 | 0.06 | 0.13 | 0.02 | 0.06 | 0.02 | 0.03 | 0.03 | 0.02 | 0.13 | 0.04 | 0.02 | 0.25 | |||||||
| TN M2 (dark) | red | brown | opaque | 62.82 | 14.36 | 6.97 | 2.84 | 0.86 | 3.89 | 0.81 | 0.80 | 0.11 | 0.16 | 0.27 | 0.94 | <0.03 | 0.47 | <0.04 | 0.21 | 0.75 | 4.65 | 100.91 |
| 0.79 | 0.42 | 0.17 | 0.12 | 0.04 | 0.52 | 0.03 | 0.03 | 0.03 | 0.02 | 0.04 | 0.07 | 0.29 | 0.07 | 0.14 | 1.09 | |||||||
| TN M2 (pale) | red | brown | opaque | 62.76 | 14.01 | 7.10 | 2.75 | 0.87 | 2.71 | 0.81 | 0.87 | 0.10 | 0.15 | 0.29 | 0.93 | <0.03 | 0.58 | <0.04 | 0.17 | 0.85 | 5.40 | 100.35 |
| 1.25 | 0.39 | 0.19 | 0.15 | 0.05 | 0.42 | 0.02 | 0.08 | 0.02 | 0.03 | 0.03 | 0.03 | 0.10 | 0.04 | 0.16 | 0.97 | |||||||
| TN M3 | red | brown | opaque | 60.61 | 15.31 | 6.27 | 2.60 | 0.34 | 2.62 | 0.75 | 0.79 | 0.11 | 0.11 | 0.27 | 0.96 | <0.03 | 0.61 | <0.04 | 0.31 | 0.68 | 8.09 | 100.42 |
| 0.30 | 0.14 | 0.49 | 0.11 | 0.04 | 0.10 | 0.02 | 0.04 | 0.02 | 0.03 | 0.02 | 0.05 | 0.37 | 0.05 | 0.03 | 0.32 | |||||||
| PN M3 | red | green/red | translucent with opaque bands | 51.78 | 12.76 | 7.58 | 2.79 | 0.34 | 1.42 | 0.92 | 0.78 | 0.16 | 0.16 | 0.18 | 0.98 | <0.03 | 2.48 | 0.06 | 0.39 | 0.78 | 15.66 | 99.20 |
| 0.30 | 0.34 | 0.26 | 0.13 | 0.02 | 0.06 | 0.02 | 0.09 | 0.03 | 0.04 | 0.03 | 0.15 | 0.19 | 0.02 | 0.03 | 0.04 | 0.50 | ||||||
| TN VOL 1 | red | green/red | translucent with opaque bands | 66.44 | 16.11 | 6.81 | 2.78 | 0.58 | 1.91 | 0.90 | 0.81 | 0.16 | 0.17 | 0.27 | 1.11 | <0.03 | 1.37 | <0.04 | 0.15 | 0.57 | 0.87 | 101.00 |
| 0.28 | 0.17 | 0.15 | 0.35 | 0.03 | 0.21 | 0.05 | 0.09 | 0.01 | 0.04 | 0.04 | 0.08 | 0.47 | 0.03 | 0.05 | 0.16 | |||||||
| TN NS1 | red | dark amber | semi-opaque | 66.04 | 16.77 | 6.35 | 2.31 | 0.64 | 1.59 | 0.73 | 0.74 | 0.10 | 0.10 | 0.28 | 1.08 | <0.03 | 0.66 | <0.04 | 0.06 | 0.66 | 2.38 | 100.50 |
| 0.48 | 0.23 | 0.25 | 0.09 | 0.05 | 0.03 | 0.02 | 0.06 | 0.02 | 0.04 | 0.03 | 0.04 | 0.03 | 0.01 | 0.03 | 0.10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maltoni, S.; Silvestri, A. A Mosaic of Colors: Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy. Minerals 2018, 8, 255. https://doi.org/10.3390/min8060255
Maltoni S, Silvestri A. A Mosaic of Colors: Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy. Minerals. 2018; 8(6):255. https://doi.org/10.3390/min8060255
Chicago/Turabian StyleMaltoni, Sarah, and Alberta Silvestri. 2018. "A Mosaic of Colors: Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy" Minerals 8, no. 6: 255. https://doi.org/10.3390/min8060255
APA StyleMaltoni, S., & Silvestri, A. (2018). A Mosaic of Colors: Investigating Production Technologies of Roman Glass Tesserae from Northeastern Italy. Minerals, 8(6), 255. https://doi.org/10.3390/min8060255





