Evaluation of Microwave Intensified Vanadium Bearing-High Carbonaceous Shale Acid Extraction Process
Abstract
:1. Introduction
2. Materials and Methods
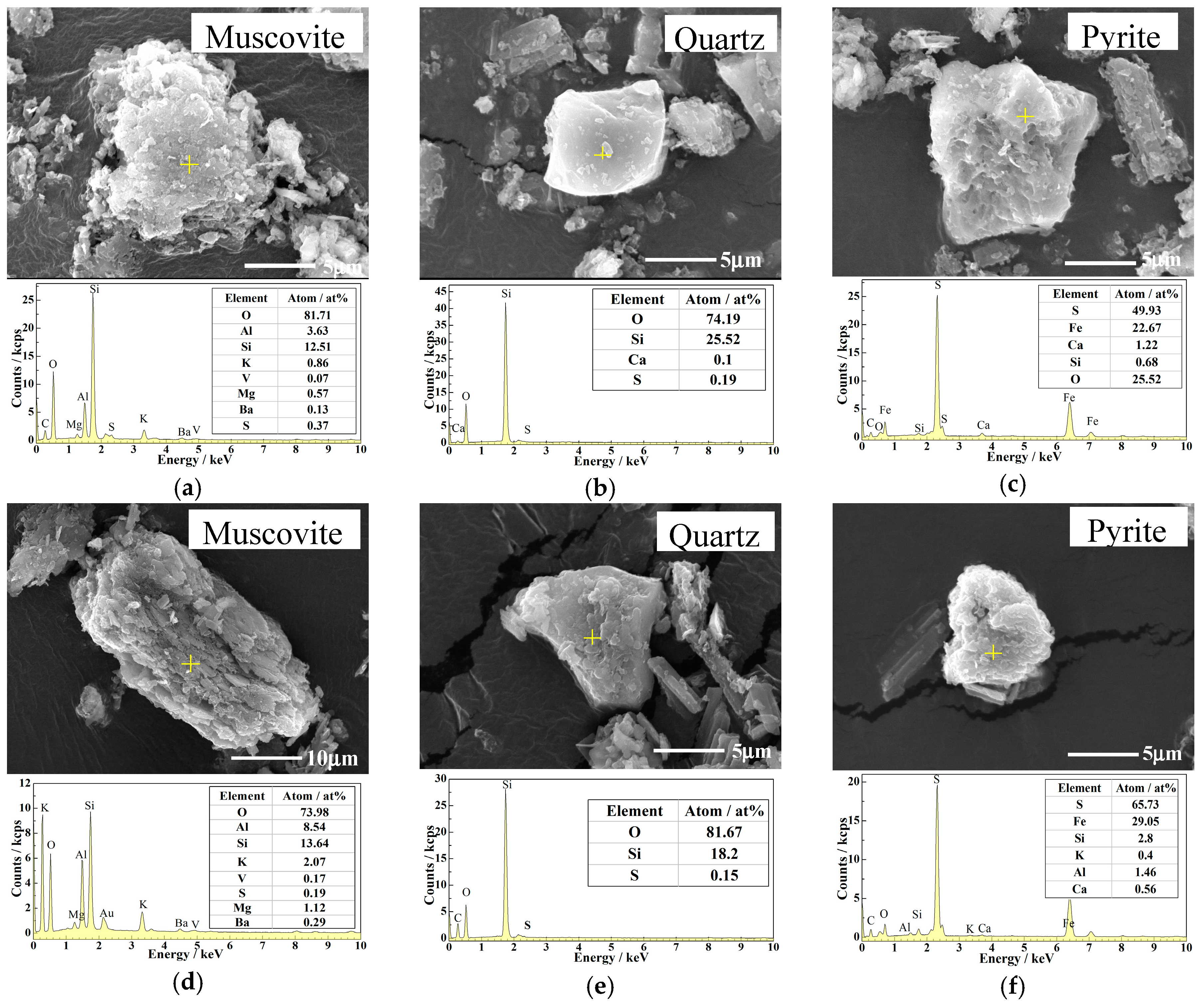
2.1. Materials
2.2. Equipment and Experimental
2.2.1. Leaching Procedure with Microwave Heating (MH)
2.2.2. Leaching Procedure with Conventional Heating (CH)
2.3. Experimental Design
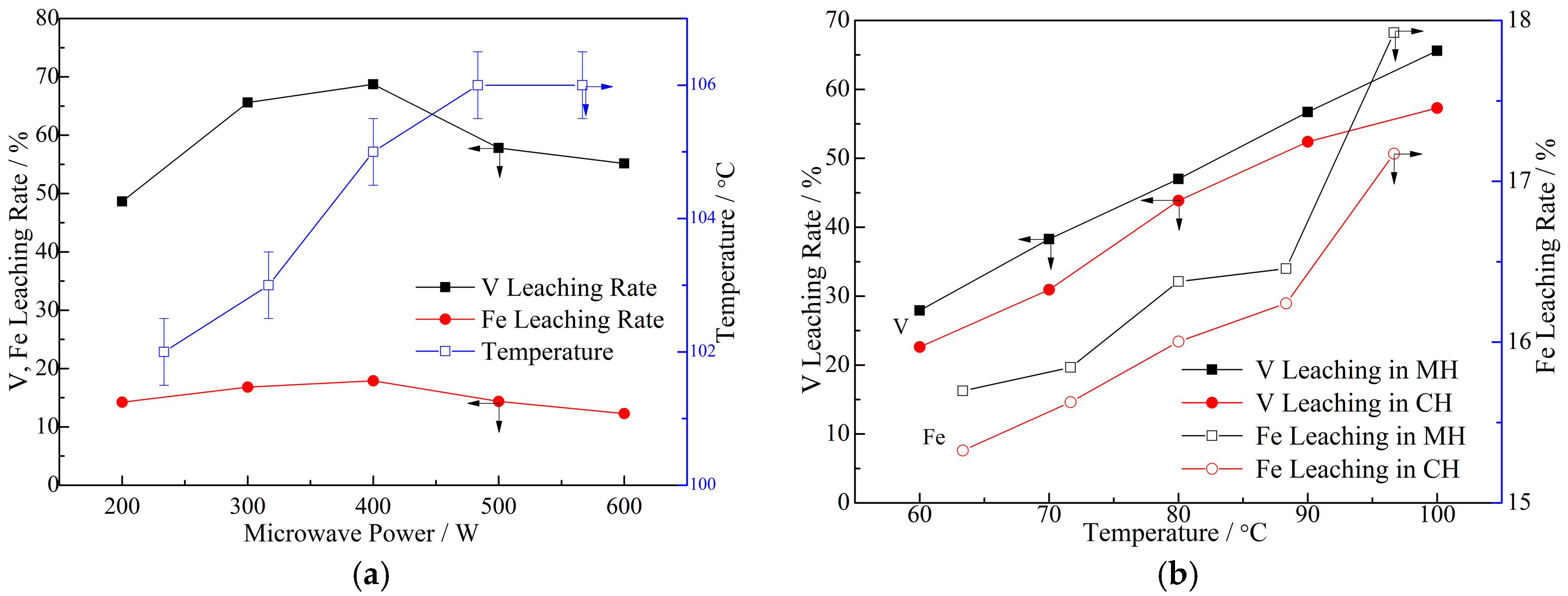
3. Results and Discussion
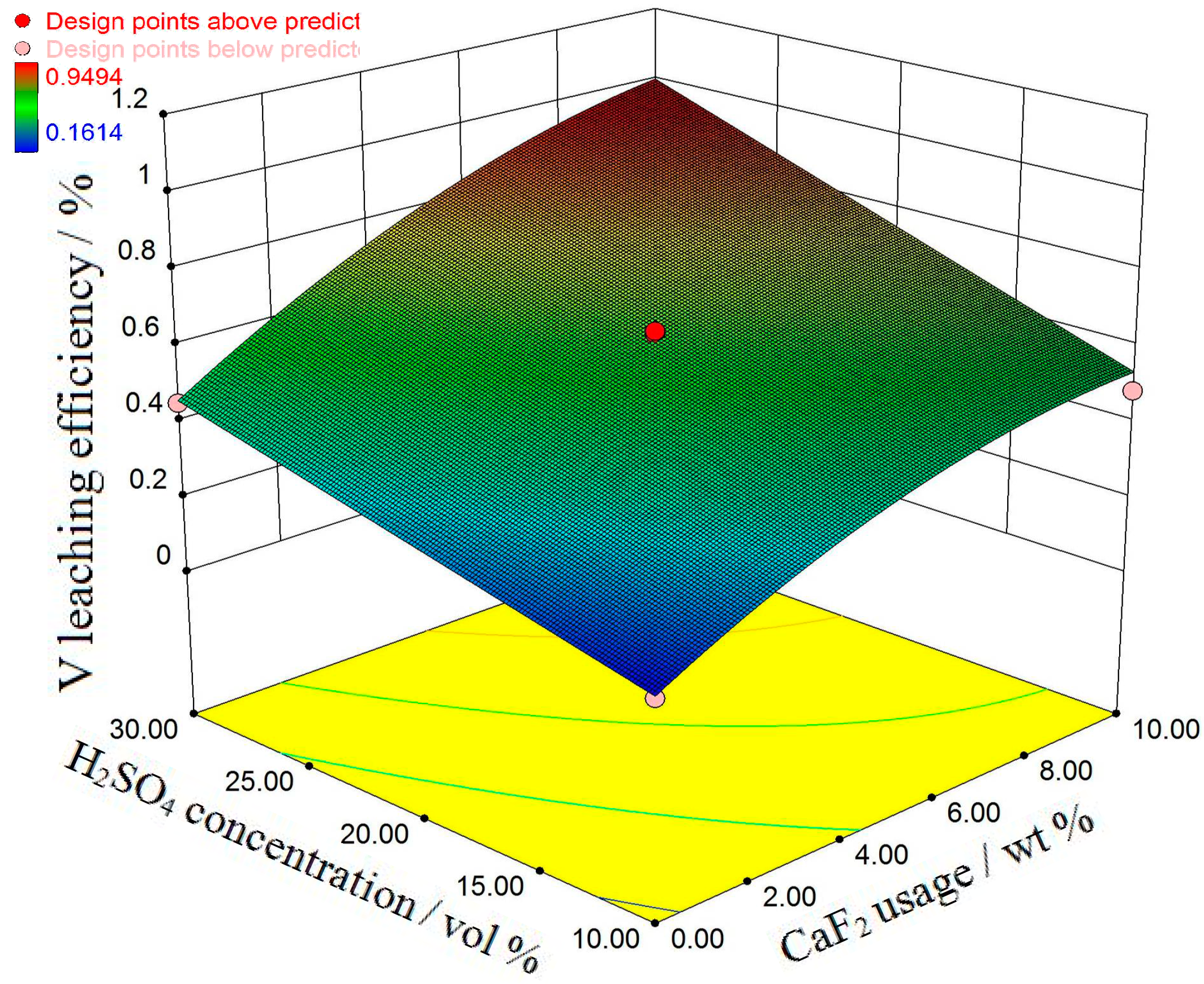
3.1. Response Analysis and Interpretation
3.2. Process Optimization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, Y.M.; Bao, S.X.; Liu, T.; Chen, T.J.; Huang, J. The technology of extracting vanadium from stone coal in China: History, current status and future prospects. Hydrometallurgy 2011, 109, 116–124. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, Y.M.; Bao, S.X. Vanadium recovery from stone coal through roasting and flotation. Trans. Nonferrous Met. Soc. China 2017, 27, 197–203. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Y.; Bao, S. Preparation of high purity V2O5 from a typical low-grade refractory stone coal using a pyro-hydrometallurgical process. Minerals 2016, 6, 69. [Google Scholar] [CrossRef]
- Hu, P.; Zhang, Y.; Liu, T.; Huang, J.; Yuan, Y.; Yang, Y. Separation and recovery of iron impurity from a vanadium-bearing stone coal via an oxalic acid leaching-reduction precipitation process. Sep. Purif. Technol. 2017, 180, 99–106. [Google Scholar] [CrossRef]
- Xiang, Z.; Wu, X. Ultrasonic-microwave assisted extraction of total flavonoids from scutellaria baicalensis using response surface methodology. Pharm. Chem. J. 2017, 51, 318–323. [Google Scholar] [CrossRef]
- Sadeghi, S.M.; Vanpeteghem, G.; Neto, I.F.F.; Soares, H.M.V.M. Selective leaching of Zn from spent alkaline batteries using environmentally friendly approaches. Waste Manage. 2017, 60, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Al-Harahsheh, M.; Kingman, S.W. Microwave-assisted leaching—a review. Hydrometallurgy 2004, 73, 189–203. [Google Scholar] [CrossRef]
- Tong, Z.; Ma, G.; Zhang, X.; Cai, Y. Microwave-supported leaching of electric arc furnace (EAF) slag by ammonium salts. Minerals 2017, 7, 119. [Google Scholar] [CrossRef]
- Shanshan, C.; Jianfeng, H.; Zhanwei, X.; Jiayin, L.; Haibo, O.; Liyun, C.; Lei, Z. Influence of microwave heating on the structure and electrochemical property of NH4V3O8 cathode material for lithium ion batteries. Ceram. Int. 2015, 41, 6747–6752. [Google Scholar] [CrossRef]
- Drunka, R.; Grabis, J.; Krumina, A. Microwave assisted synthesis, modification with platinum and photocatalytical properties of TiO2 nanofibers. Mater. Sci. 2016, 22, 138–141. [Google Scholar] [CrossRef]
- Zadaviciute, S.; Baltakys, K.; Bankauskaite, A. The effect of microwave and hydrothermal treatments on the properties of hydrotalcite. J. Therm. Anal. Calorim. 2017, 127, 189–196. [Google Scholar] [CrossRef]
- Keisandokht, S.; Haddad, N.; Gariepy, Y.; Orsat, V. Screening the microwave-assisted extraction of hydrocolloids from Ocimum basilicum L. seeds as a novel extraction technique compared with conventional heating-stirring extraction. Food Hydrocoll. 2018, 74, 11–22. [Google Scholar] [CrossRef]
- Falciglia, P.P.; De Guidi, G.; Catalfo, A.; Finocchiaro, G.; Farina, M.; Liali, M.; Lorenzano, G.; Valastro, G.; Vagliasindi, F.G.A. Glycerol-enhanced microwave heating for ultra-rapid effective remediation of marine sediments highly contaminated with hydrocarbons. Sep. Purify. Technol. 2017, 189, 11–19. [Google Scholar] [CrossRef]
- Li, X.; Wang, B.; Wu, S.; Kong, X.; Fang, Y.; Liu, J. Optimizing the conditions for the microwave-assisted pyrolysis of cotton stalk for bio-oil production using response surface methodology. Waste Biomass Valorization 2017, 8, 1361–1369. [Google Scholar] [CrossRef]
- Wen, T.; Zhao, Y.; Xiao, Q.; Ma, Q.; Kang, S.; Li, H.; Song, S. Effect of microwave-assisted heating on chalcopyrite leaching of kinetics, interface temperature and surface energy. Results Phys. 2017, 7, 2594–2600. [Google Scholar] [CrossRef]
- Hua, Y.; Cai, C.; Cui, Y. Microwave-enhanced roasting of copper sulfide concentrate in the presence of CaCO3. Sep. Purify. Technol. 2006, 50, 22–29. [Google Scholar] [CrossRef]
- Li, H.; Cui, J.; Liu, J.; Li, X.; Gao, X. Mechanism of the effects of microwave irradiation on the relative volatility of binary mixtures. AIChE J. 2017, 63, 1328–1337. [Google Scholar] [CrossRef]
- Wang, J.-P.; Zhang, Y.-M.; Huang, J.; Liu, T. Synergistic effect of microwave irradiation and CaF2 on vanadium leaching. Int. J. Miner. Metall. Mater. 2017, 24, 156–163. [Google Scholar] [CrossRef]
- Shariaty, P.; Lashaki, M.J.; Hashisho, Z.; Sawada, J.; Kuznicki, S.; Hutcheon, R. Effect of ETS-10 ion exchange on its dielectric properties and adsorption/microwave regeneration. Sep. Purify. Technol. 2017, 179, 420–427. [Google Scholar] [CrossRef]
- Lee, C.S.; Binner, E.; Winkworth-Smith, C.; John, R.; Gomes, R.; Robinson, J. Enhancing natural product extraction and mass transfer using selective microwave heating. Chem. Eng. Sci. 2016, 149, 97–103. [Google Scholar] [CrossRef]
- Li, L.; Guo, Z.; Han, W.; Wang, Q. The effect of microwave on the primary nucleation of CaSO4 from aqueous solutions. Powder Technol. 2017, 317, 189–196. [Google Scholar] [CrossRef]
- Janney, M.A.; Kimrey, H.D.; Allen, W.R.; Kiggans, J.O. Enhanced diffusion in sapphire during microwave heating. J. Mater. Sci. 1997, 32, 1347–1355. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, Y.; Huang, J.; Liu, T.; Cai, Z.; Xue, N. Selective leaching of vanadium from roasted stone coal by dilute sulfuric acid dephosphorization-two-stage pressure acid leaching. Minerals 2016, 6, 75. [Google Scholar] [CrossRef]
- Al-harahsheh, M.; Kingman, S.; Al-Makhadmah, L.; Hamilton, I.E. Microwave treatment of electric arc furnace dust with PVC: Dielectric characterization and pyrolysis-leaching. J. Hazard. Mater. 2014, 274, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Bayca, S.U. Microwave radiation leaching of colemanite in sulfuric acid solutions. Sep. Purify. Technol. 2013, 105, 24–32. [Google Scholar] [CrossRef]
- Bohlouli, A.; Afshar, M.R.; Aboutalebi, M.R.; Seyedein, S.H. Optimization of tungsten leaching from low manganese wolframite concentrate using Response Surface Methodology (RSM). Int. J. Refract. Met. Hard Mater. 2016, 61, 107–114. [Google Scholar] [CrossRef]
- Mpinga, C.N.; Eksteen, J.; Aldrich, C.; Dyer, L. Identification of the significant factors determining extractability of Ni and Cu after sulfation roasting of a PGM-bearing chromitite ore. Miner. Eng. 2017, 110, 153–165. [Google Scholar] [CrossRef]
- Ferdowsi, A.; Yoozbashizadeh, H. Process optimization and kinetics for leaching of cerium, lanthanum and neodymium elements from iron ore waste’s apatite by nitric acid. Trans. Nonferrous Met. Soc. China 2017, 27, 420–428. [Google Scholar] [CrossRef]
- Terry, B. Specific chemical rate constants for the acid dissolution of oxides and silicates. Hydrometallurgy 1983, 11, 315–344. [Google Scholar] [CrossRef]
- Larsen, E.; Kleiv, R.A. Flotation of quartz from quartz-feldspar mixtures by the HF method. Miner. Eng. 2016, 98, 49–51. [Google Scholar] [CrossRef]
- Baghurst, D.R.; Mingos, D.M.P. Superheating effects associated with microwave dielectric heating. J. Chem. Soc. Chem. Commun. 1992, 9, 674–677. [Google Scholar] [CrossRef]
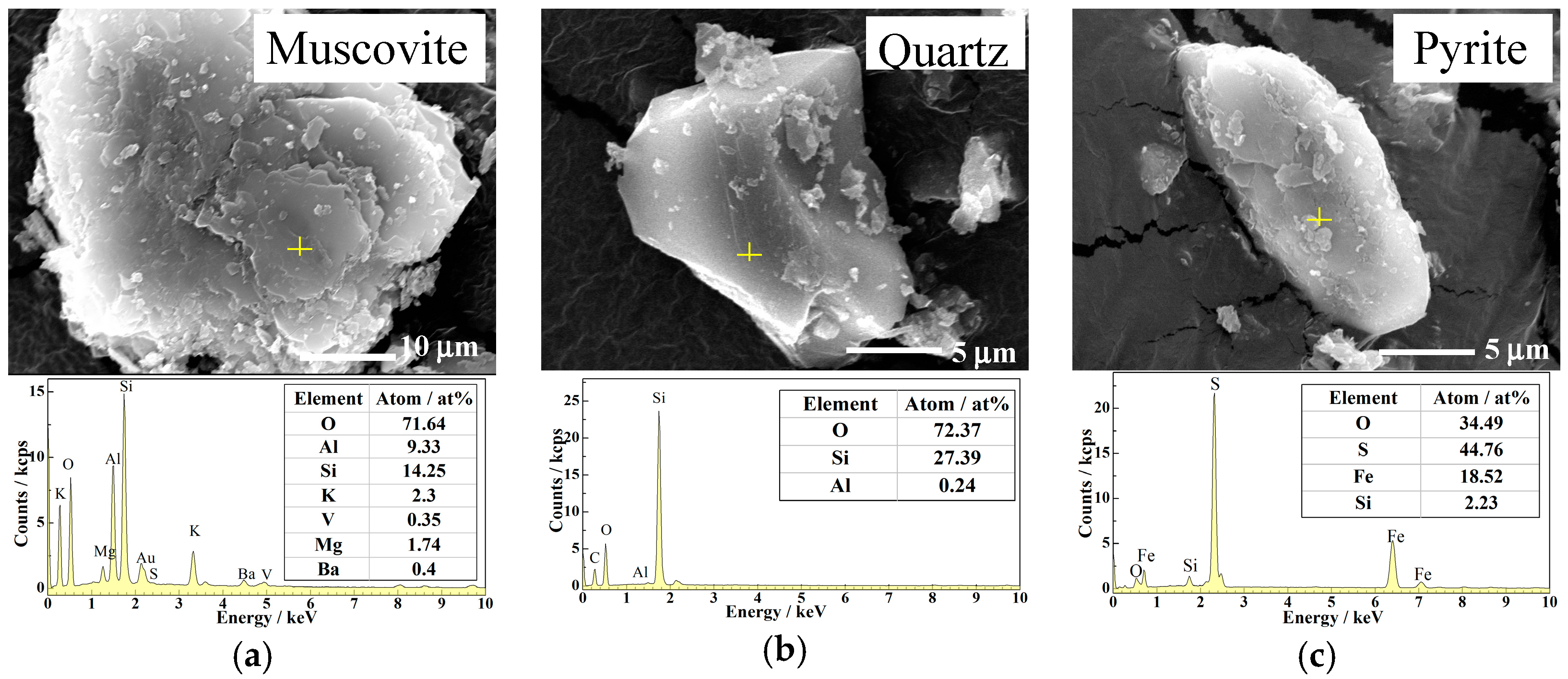
| V | Si | Al | Fe | Ca | Mg | K | Na | C | S |
|---|---|---|---|---|---|---|---|---|---|
| 0.40 | 25.20 | 7.07 | 3.01 | 3.71 | 1.45 | 1.50 | 0.22 | 10.77 | 1.22 |
| Mineral | ε′ | ε″ | Contents (wt %) |
|---|---|---|---|
| Muscovite | 3.096 | 0.589 | 15 |
| Quartz | 3.644 | 0.643 | 37 |
| Pyrite | 15.645 | 3.765 | 7 |
| Carbon | 25.584 | 8.162 | 11 |
| Feldspar | 3.232 | 0.607 | 10 |
| Calcite | 3.954 | 0.733 | 11 |
| Factors | Symbol | Range and Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| CaF2 dosage (wt %) | X1 | 0.0 | 5.0 | 10.0 |
| H2SO4 concentration (vol %) | X2 | 10.0 | 20.0 | 30.0 |
| Leaching time (min) | X3 | 60.0 | 150.0 | 240.0 |
| Power (W) | X4 | 150 | 350 | 550 |
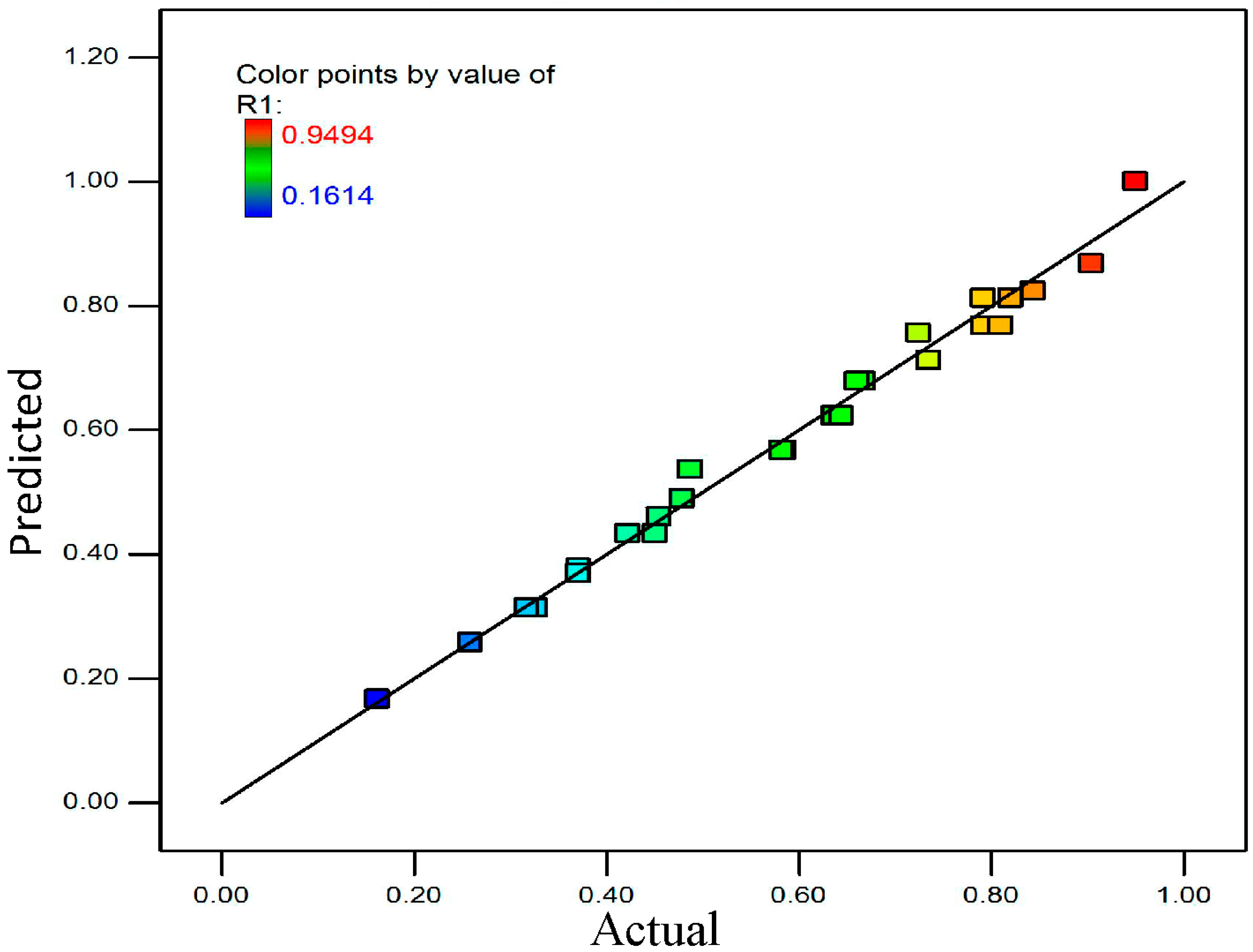
| Run | CaF2/wt % | H2SO4/vol % | Time/min | Power/W | Y1 (MH) | Y2 (CH) |
|---|---|---|---|---|---|---|
| 1 | 5.0 | 10.0 | 60.0 | 350 | 0.3703 | 0.3445 |
| 2 | 0.0 | 20.0 | 240.0 | 350 | 0.3701 | 0.2489 |
| 3 | 5.0 | 20.0 | 240.0 | 150 | 0.6663 | - |
| 4 | 5.0 | 10.0 | 150.0 | 550 | 0.4216 | - |
| 5 | 0.0 | 20.0 | 150.0 | 550 | 0.3165 | - |
| 6 | 10.0 | 20.0 | 150.0 | 150 | 0.7915 | - |
| 7 | 5.0 | 10.0 | 150.0 | 150 | 0.4500 | - |
| 8 | 10.0 | 30.0 | 150.0 | 350 | 0.9494 | 0.9282 |
| 9 | 5.0 | 20.0 | 60.0 | 550 | 0.5813 | - |
| 10 | 5.0 | 30.0 | 60.0 | 350 | 0.7238 | 0.6842 |
| 11 | 5.0 | 10.0 | 240.0 | 350 | 0.4779 | 0.4211 |
| 12 | 5.0 | 20.0 | 240.0 | 550 | 0.6601 | - |
| 13 | 5.0 | 20.0 | 150.0 | 350 | 0.6362 | 0.6235 |
| 14 | 0.0 | 10.0 | 150.0 | 350 | 0.1614 | 0.1303 |
| 15 | 5.0 | 30.0 | 240.0 | 350 | 0.9036 | 0.8785 |
| 16 | 10.0 | 20.0 | 150.0 | 550 | 0.8091 | - |
| 17 | 10.0 | 20.0 | 60.0 | 350 | 0.7345 | 0.5638 |
| 18 | 0.0 | 30.0 | 150.0 | 350 | 0.4539 | 0.3219 |
| 19 | 10.0 | 20.0 | 240.0 | 350 | 0.8433 | 0.8456 |
| 20 | 10.0 | 10.0 | 150.0 | 350 | 0.4864 | 0.4246 |
| 21 | 5.0 | 30.0 | 150.0 | 550 | 0.7910 | - |
| 22 | 5.0 | 20.0 | 60.0 | 150 | 0.5843 | - |
| 23 | 0.0 | 20.0 | 150.0 | 150 | 0.3258 | - |
| 24 | 5.0 | 30.0 | 150.0 | 150 | 0.8197 | - |
| 25 | 0.0 | 20.0 | 60.0 | 350 | 0.2577 | 0.1881 |
| 26 | 5.0 | 20.0 | 150.0 | 350 | 0.6439 | 0.614 |
| Source | F-Value | p-Value, Prob > F | ||
|---|---|---|---|---|
| Y1 | Y2 | Y1 | Y2 | |
| Model | 112.57 Significant | 147.21 Significant | <0.0001 | 0.0001 |
| X1 | 854.27 | 666.92 | <0.0001 | <0.0001 |
| X2 | 593.08 | 423.36 | <0.0001 | <0.0001 |
| X3 | 51.40 | 71.55 | <0.0001 | 0.0011 |
| X4 | 0.38 | - | 0.5478 | - |
| X1X2 | 10.00 | 37.01 | 0.0090 | 0.0037 |
| X1X3 | 0.0044 | 18.57 | 0.9480 | 0.0126 |
| X1X4 | 0.25 | - | 0.6277 | - |
| X2X3 | 1.79 | 5.27 | 0.2075 | 0.0834 |
| X2X4 | 0.000039 | - | 0.9951 | - |
| X3X4 | 0.00372 | - | 0.9524 | - |
| X12 | 47.37 | 100.90 | < 0.0001 | 0.0006 |
| X22 | 3.79 | 2.69 | 0.0774 | 0.1763 |
| X32 | 0.13 | 0.84 | 0.7280 | 0.4105 |
| X42 | 0.00527 | - | 0.9434 | - |
| Lack of Fit | 26.86 | 19.09 | 0.1492 Not significant | 0.1663 Not significant |
| Number | CaF2 Usage/wt % | H2SO4/vol % | Time/min | Y1 | Y2 |
|---|---|---|---|---|---|
| 1 | 10.00 | 15.56 | 60.0 | 0.6081 | 0.4545 |
| 2 | 9.98 | 15.23 | 60.0 | 0.6001 | 0.4468 |
| 3 | 10.00 | 14.58 | 60.0 | 0.5841 | 0.4291 |
| 4 | 10.00 | 15.81 | 62.1 | 0.6149 | 0.4643 |
| 5 | 0.12 | 30.00 | 240.0 | 0.5432 | 0.3773 |
| 6 | 0.04 | 30.00 | 240.0 | 0.5361 | 0.3671 |
| 7 | 0.24 | 30.00 | 240.0 | 0.5541 | 0.3930 |
| 8 | 0.41 | 30.00 | 240.0 | 0.5694 | 0.4150 |
| 9 | 0.16 | 30.00 | 237.9 | 0.5448 | 0.3824 |
| 10 | 0.57 | 30.00 | 240.0 | 0.5834 | 0.4351 |
| 11 | 0.21 | 29.13 | 240.0 | 0.5422 | 0.3819 |
| 12 | 9.71 | 15.12 | 60.0 | 0.5965 | 0.4512 |
| 13 | 0.44 | 28.67 | 240.0 | 0.5562 | 0.4053 |
| 14 | 0.15 | 30.00 | 216.9 | 0.5209 | 0.3746 |
| 15 | 10.00 | 11.38 | 147.3 | 0.5381 | 0.4606 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Zhang, Y.; Liu, T.; Huang, J. Evaluation of Microwave Intensified Vanadium Bearing-High Carbonaceous Shale Acid Extraction Process. Minerals 2018, 8, 113. https://doi.org/10.3390/min8030113
Wang J, Zhang Y, Liu T, Huang J. Evaluation of Microwave Intensified Vanadium Bearing-High Carbonaceous Shale Acid Extraction Process. Minerals. 2018; 8(3):113. https://doi.org/10.3390/min8030113
Chicago/Turabian StyleWang, Jingpeng, Yimin Zhang, Tao Liu, and Jing Huang. 2018. "Evaluation of Microwave Intensified Vanadium Bearing-High Carbonaceous Shale Acid Extraction Process" Minerals 8, no. 3: 113. https://doi.org/10.3390/min8030113
APA StyleWang, J., Zhang, Y., Liu, T., & Huang, J. (2018). Evaluation of Microwave Intensified Vanadium Bearing-High Carbonaceous Shale Acid Extraction Process. Minerals, 8(3), 113. https://doi.org/10.3390/min8030113




